Abstract
Chlorphenesin functions as a biocide in cosmetics and is used at concentrations up to 0.32% in rinse-off products and up to 0.3% in leave-on products. The Cosmetic Ingredient Review Expert Panel (Panel) noted that chlorphenesin was well absorbed when applied to the skin of rats; however, any safety concern was minimized because available data demonstrated an absence of toxicity. The Panel concluded that chlorphenesin is safe in the present practices of use and concentration.
Keywords
Introduction
As stated in the International Cosmetic Ingredient Dictionary and Handbook, 1 chlorphenesin functions as a biocide (preservative) in cosmetic products. The Expert Panel (Panel) noted that chlorphenesin (CAS No. 104-29-0) may be confused with the muscle relaxant drug, chlorphenesin carbamate (CAS No. 886-74-8), which has also been known as chlorphenesin. Chlorphenesin carbamate is not a cosmetic ingredient and is not reviewed in this safety assessment.
Chemistry
Definition and Structure
As given in the International Cosmetic Ingredient Dictionary and Handbook, chlorphenesin (CAS No. 104-29-0) is a chlorophenol derivative defined as the organic compound that conforms to the formula shown 1 in Figure 1. Other names for this chemical include 3-(4-chlorophenoxy)-1,2-propanediol; 1,2-propanediol,3-(4-chlorophenoxy)-; and p-chlorophenyl glyceryl ether. 1
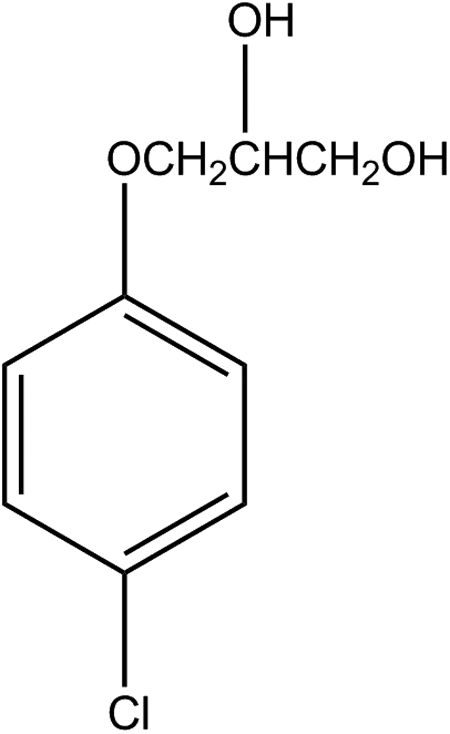
Chlorphenesin.
Chemical and Physical Properties
An ultraviolet (UV) spectrum of 0.01% aqueous chlorphenesin solution exhibited an absorption maximum at 279 nm. 2 Additional properties of chlorphenesin are given in Table 1.
Properties of Chlorphenesin.a

a Complies with British Pharmacopoeia specifications for chlorophenol as a component. 41
Methods of Production
Chlorphenesin is prepared by condensing equimolar amounts of p-chlorophenol and glycidol in the presence of a tertiary amine or a quaternary ammonium salt as a catalyst. 3
Use
Cosmetic
Chlorphenesin reportedly functions as a biocide in cosmetic products. 1 Reportedly, chlorphenesin (ELESTAB CPN; concentration of use = 0.10%-0.30%) has bactericidal activity against gram-positive and gram-negative bacteria, fungicidal activity against Aspergillus niger IMI 149007, and Penicillium pinophilum IMI 87160 (fungi), and is also active against Candida albicans NCPF 3179 and Saccharomyces cerevisiae NCPF 3275 (yeasts). 4
According to information supplied to the US Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP) in 2011, chlorphenesin is used in 1386 cosmetic products. 5 These data are summarized in Table 2. Results from a survey of ingredient use concentrations provided by the Personal Care Products Council (also included in Table 2) in 2011 indicate that chlorphenesin is used at similar concentrations in rinse-off and leave-on cosmetic products—up to 0.32% in rinse-off products and up to 0.3% in leave-on products. 6
Current Frequency and Concentration of Use According to Duration and Type of Exposure Provided in 2011and 2012.a,5, 6
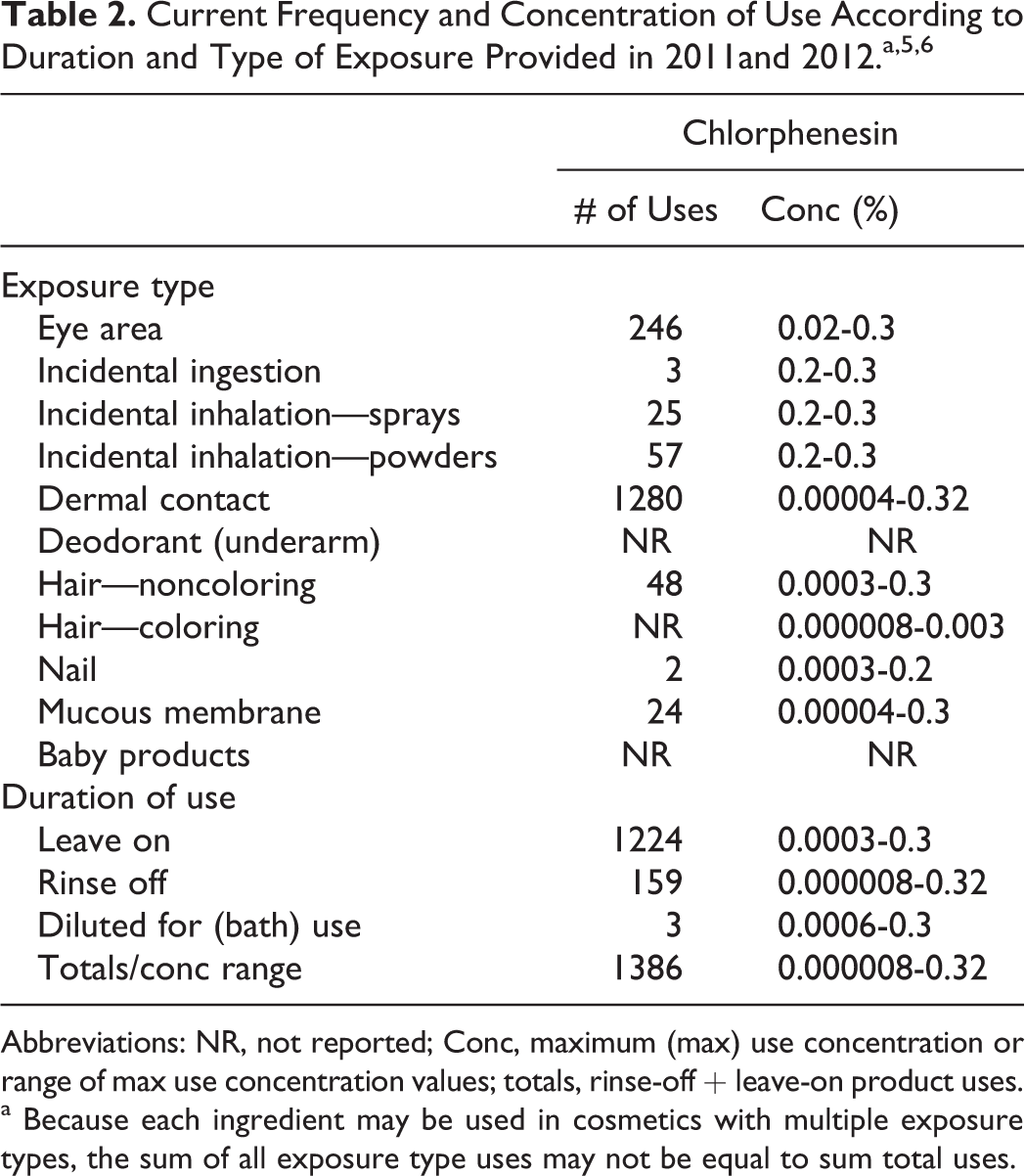
Abbreviations: NR, not reported; Conc, maximum (max) use concentration or range of max use concentration values; totals, rinse-off + leave-on product uses.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not be equal to sum total uses.
Cosmetic products containing chlorphenesin may be applied to the skin and hair or, incidentally, may come in contact with the eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
Chlorphenesin is used in hair, foot, and suntan sprays and could possibly be inhaled. In practice, 95% to 99% of the particles released from cosmetic sprays have aerodynamic equivalent diameters in the 10 to 110 µm range, with propellant sprays yielding a greater fraction of droplets/particles below this range when compared to pump sprays. 7,8 Therefore, most aerosols incidentally inhaled from these sprays are deposited in the nasopharyngeal region and are not respirable to any appreciable level. 9,10 Thus, toxicological concerns could arise from direct effects on nasopharyngeal tissues or from swallowing of the resulting minute amounts. Based on further toxicological assessments contained herein, such exposures would pose no identifiable risks.
According to the European Union Cosmetics Directive, chlorphenesin is listed among the preservatives that may be contained in cosmetic products marketed in the European Union (EU). The maximum authorized use concentration for this ingredient is 0.3%. 11
Noncosmetic
Chlorphenesin (0.10%) is one of the ingredients in an antimicrobial product identified as Miol cream. The reason for adding chlorphenesin as an ingredient was not stated. 12
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
The absorption, distribution, and metabolic fate of chlorphenesin was evaluated using male Sprague-Dawley rats and Beagle dogs. 13 In the first experiment (4 rats), a 16.7 mg oral dose of chlorphenesin-1,3- 14 C (in physiological saline) was administered via oral gavage, after which concentrations in the blood were determined. In a second experiment, chlorphenesin-1,3- 14 C (15.2 mg) was administered intraperitonelly (ip) to 1 rat and the distribution of administered radioactivity was determined. A third experiment was performed to isolate chlorphenesin metabolites from the urine. Nonradioactive chlorphenesin (500 mg/kg) was administered orally to 2 Beagle dogs and urine was collected for 24 hours. Urine from 2 Beagle dogs was also collected after the animals received 2 ip injections of nonradioactive chlorphenesin (250 mg/kg, 6 hours apart). In a fourth experiment to identify conjugated metabolites, 4 male rats were injected ip with chlorphenesin UL-ring- 14 C (30 mg) and urine was collected for 24 hours.
Following oral ingestion, chlorphenesin- 14 C was absorbed rapidly in the rat. Radioactivity reached a peak blood concentration in 30 minutes, and the half-life of serum radioactivity was approximately 140 minutes.
Results of the distribution experiment indicated that over half of the administered ip dose of chlorphenesin-1,3- 14 C in the single rat studied was excreted in the urine after 4 hours. The remainder was found primarily in the gastrointestinal tract and carcass. A small portion of the radioactivity was recovered as exhaled CO2.
The urinary end products (expressed as % of urinary radioactivity) identified after administration of the compound to rats and dogs were 3-p-chlorophenoxylactic acid (57.3% in dogs; 41.8% in rats), p-chlorophenoxyacetic acid (12% in dogs; 22.8% in rats), and unchanged chlorphenesin (30.4% in dogs; 35.5% in rats). Additional urinary end products identified as a conjugate of chlorophenol and a conjugate of chlorphenesin were observed after rats were injected ip with chlorphenesin UL-ring- 14 C. 13
Percutaneous Absorption
The percutaneous absorption of 14 C-chlorphenesin was evaluated using 16 male rats of the Sprague-Dawley CD strain (∼6 weeks old). 14 C-chlorphenesin (in 0.05% weight/weight cold cream; mean dose = 1.14 mg/kg [∼ 14 µCi]) was applied topically to shaved skin on the back (9 cm2). Application sites were occluded with aluminum foil until the animals were killed. 14 After test substance application, the animals were placed in individual metabolism cages for the collection of urine and feces. Pairs of animals were killed at various intervals, beginning at 1 hour and ending at 96 hours. The mean total recovery of radioactivity (application site, excreta, selected tissues, and residual carcass) was 92.35% dose + 3.11 (standard deviation) for the 0 to 96 hours time period. The proportion of administered 14 C-chlorphenesin dose that remained at the application site (in and on the skin) decreased from ∼89% at 1 hour to ∼43% at 96 hours. During the 0 to 96 hours time period, ∼48% (mean value) of the applied dose was excreted in the urine. Approximately 0.5% was excreted in the feces and ∼0.7% was recovered in cage washings. Thus, practically all of the absorbed dose was excreted in the urine over a period of 96 hours.
Not more than 1% of the applied dose was present in any tissues during the 1 to 96 hours time frame although up to 57% of the dose was absorbed. At 96 hours, ∼7% to 8% of the administered dose remained. Apparently, the radioactivity was absorbed biphasically, with initial and terminal half-lives for absorption ≈4 hours and 126 hours, respectively. The urinary excretion rate was proportional to plasma radioactivity concentrations during 0 to 96 hours, suggesting that the renal clearance of radioactivity was concentration independent. The terminal excretion half-life (∼22 hours) was considerably shorter than the terminal absorption half-life (∼126 hours). Thus, the excretion of radioactivity was absorption rate limited, causing plasma concentrations to remain quite low. 14
Toxicology
Acute Oral Toxicity
The acute oral toxicity of chlorphenesin (in 0.5% carboxymethylcellulose aqueous gel) was evaluated using 5 groups of 10 (5 males, 5 females/group; ∼6 weeks old) Sprague Dawley rats. 15 The 5 groups received single oral doses of 1200, 1620, 2187, 2952, and 3985 mg/kg, respectively. Dosing was followed by a 14-day observation period, after which all surviving animals were killed. The following signs were observed after test substance administration of each dose: dyspnea, decrease in spontaneous activity, hypotonia, piloerection, and loss of reflex. Necropsy findings for animals that died were mainly an intestinal meteroism and lung congestion. A mean lethal dose, 50% (LD50) of 3000 mg/kg (95% confidence interval = 2830-3180 mg/kg) was reported.
Repeated Dose Toxicity
A repeated dose oral toxicity study on chlorphenesin was performed using 4 groups of 16 rats of the Charles River Crl: CD(SD) BR strain (8 males, 8 females/group; 47 days old). 16 Chlorphenesin (suspension in 1% aqueous methylcellulose) was administered by gavage to 3 groups at doses of 10, 100, and 1000 mg/kg/d (dose volume = 10 mL/kg/day), respectively, for 28 consecutive days. Control rats were dosed similarly with 1% aqueous methylcellulose. Except for 1 animal killed during week 4, the animals were killed on day 29. Microscopic examination of the rat (high-dose male) killed during week 4 revealed renal tubular dilatation and necrosis of the papillary tip, both treatment related. No microscopic changes were observed in high-dose female rats or the remaining high-dose male rats. Clinical findings in the highest dose group included hunched posture, abnormal gait, pallor, lethargy, ptosis, a badly groomed appearance, noisy respiration, and piloerection. A badly groomed appearance was also observed in rats of the low-dose (according to the authors, not toxicologically significant) and intermediate-dose groups, and increased salivation was also observed in the intermediate-dose group. Compared to controls, a statistically significant reduction (P < 0.01) in body weight gain was noted for male and female rats of the highest dose group. The decreased body weight gain correlated with the decreased food intake. Significantly lower hemoglobin levels were reported for high-dose males and females and intermediate-dose males.
Statistically significant increases (P < 0.01) in glutamic pyruvic transaminase were reported for high-dose males and females. Alkaline phosphatase levels in high-dose males were slightly higher when compared to controls, but the difference was not statistically significant. Potassium and calcium ion concentrations were significantly lower (P < 0.05) in high-dose females. Immunoglobulin (Ig) G and IgM serum levels in high-dose females, when adjusted for predose levels, were significantly higher than control values at the end of dosing. The authors considered these changes as a reflection of hematological and biochemical changes due to treatment with chlorphenesin and not a specific effect on the immune system. Absolute spleen weights (high-dose males and females) and thymus weights (high-dose males) were significantly lower (P < 0.05 or P < 0.01) when compared to controls. At macroscopic examination, general brown staining of the fur was observed in all 5 high-dose female rats examined, compared to the absence of this finding in controls. The only microscopic finding (in kidney) is mentioned in the preceding paragraph. The reported changes in the high- and intermediate-dose groups were considered treatment related. A dose of 10 mg/kg/d was considered the no adverse effect level in this study. 16
Ocular Irritation
The ocular irritation potential of chlorphenesin (1% [weight/volume, w/v] in distilled water) was evaluated using 3 New Zealand albino rabbits (ages not stated). 17 The test substance (0.1 mL) was instilled into the right eye of each animal and the lids were held together for approximately 10 seconds. Untreated left eyes served as controls. The eyes were examined for ocular reactions at 1 hour and then at days 1, 2, 3, 4, and 7 postinstillation. Slight conjunctival irritation (enanthema, chemosis, and lacrimation) was reported for each rabbit and these reactions had cleared by 24 hours postinstillation. Chlorphenesin was classified as a weak ocular irritant (maximum ocular irritation index = 6 [at 1 hour postinstillation]).
Skin Irritation
Nonhuman
The skin irritation potential of chlorphenesin was evaluated using 6 male New Zealand albino rabbits (age not stated). 17 A 2.5 × 2.5 cm occlusive patch containing chlorphenesin (1% [w/v] in distilled water, 0.5 mL) was applied to the shaved flanks of each animal. The right flank was abraded and the left remained intact. Patches were secured with fastening tape and the trunk was wrapped with an elastic bandage secured with adhesive tape. At 24 hours, the patches were removed. Slight, reversible erythema was observed in 2 rabbits and there was no evidence of structural modification. Chlorphenesin was classified as a nonirritant (primary irritation index = 0.1).
Human
A study was performed to investigate the side effects of cosmetic preservatives by evaluating objective and subjective skin irritants. 18 In a 24-hour occlusive patch test involving 30 patients (20 females, 10 males; mean age = 33.7 years), 2% chlorphenesin (20 µL) was applied to filter paper discs on IQ test chambers and patches remained in contact with the forearm for 24 hours. Reactions were evaluated at 30 minutes and 1 day after patch removal. A mean irritation score of 0.17 ± 0.38 was reported. A cumulative skin irritation test was performed using 15 healthy patients (8 females, 7 males; mean age = 29.7 years). The formulations tested were an emulsion base with a preservative mixture consisting of 0.2% methylparaben, 0.1% propylparaben, and 0.25% chlorphenesin and an emulsion base containing 0.2% methylparaben, 0.1% propylparaben, 0.25% chlorphenesin, and 0.3% phenoxyethanol. Each formulation (20 µL) was applied according to the preceding method 3 times per week over a 21-day period. Each patient received 9 applications (same site) of the test substance. For type 1 formulations tested, the highest reported total cumulative irritation mean score was 0.40 ± 0.91. For type 2 formulations, a mean score of 0.87 ± 1.19 was the highest reported.
A facial sensory irritation test was performed using 16 healthy participants (6 females, 10 males; mean age = 28.3 years). A cotton swab soaked with 0.4% chlorphenesin (in 0.5% carbopol solution, 0.5 mL volume) was rubbed briskly and applied (under occlusion) to each side of the nasolabial fold and cheek. Any evidence of a stinging/burning reaction was recorded over a period of 9 minutes. Carbopol (0.5%) solution served as the vehicle control. The sensory irritation potential of 0.4% chlorphenesin (mean score = 0.54) was greater than the control (mean score = 0.22). Emulsion bases (with or without chlorphenesin in preservatives mixture) were tested according to the same procedure. Facial sensory irritation induced by the formula containing methylparaben, propylparaben, and chlorphenesin was greater when compared to the same formula without chlorphenesin. 18
Facial sensory irritation testing was initially proposed by Frosch and Kligman. 19 In a previous Cosmetic Ingredient Review (CIR) safety assessment of α-hydroxy acids (AHAs), 20 for example, it was concluded that the sensitivity of tissue around the area of the eye to sensory irritation was such that AHA-containing products intended for use near the eye be formulated in such a way as to reduce stinging and burning reactions. The AHAs were also used as dermal irritants.
The acute dose skin irritation potential of 0.3% chlorphenesin (in water) was evaluated using 25 patients (20 females, 5 males; 19 to 62 years old). 21 An occlusive patch containing the test substance (0.1 mL) was applied to the back of each patient for 48 hours. Reactions were scored 20 minutes after patch removal. Faint, minimal erythema was observed in 2 patients and erythema (score = 1) was observed in a third patient. Chlorphenesin was classified as having negligible dermal irritation potential.
Skin Irritation and Sensitization
Nonhuman
Prior to initiation of the sensitization study subsequently, a range-finding test was performed to determine the maximal nonirritant concentration of chlorphenesin. 22 The test involved 3 male albino Dunkin Hartley guinea pigs (ages not stated). A dorsal surface area of ∼60 cm2 was clipped free of hair, and, on both sides of the spinal column, 3 symmetrical intradermal injections (0.1 mL) of the following preparations were made: (1) 50% Freund Complete Adjuvant (FCA) in distilled water, (2) distilled water, and (3) a 50/50 mixture of 1 and 2. Sites were clipped free of hair 7 days later, and the following concentrations of chlorphenesin (0.5 mL volume) were applied under an occlusive patch for 24 hours: 0.1%, 0.25%, 0.5%, and 1.0% in distilled water. Irritation reactions were scored at 24 hours and 48 hours after patch removal. Irritation was not induced by any of the concentrations tested. Test concentrations of 0.5% and 1.0% were designated for use during the challenge phase of the sensitization study.
The skin sensitization potential of chlorphenesin was evaluated in a modified guinea pig maximization test using 30 female albino Dunkin-Hartley guinea pigs (ages not stated). Test and control groups consisted of 20 and 10 guinea pigs, respectively. Dorsal skin was clipped free of hair, and 3 symmetrical intradermal injections (0.1 mL) of 1% chlorphenesin (in distilled water), 1% chlorphenesin (in a mixture of FCA and distilled water), and a mixture of FCA and distilled water, respectively, were made on both sides of the spinal column (scapular level) during induction of test animals. During induction, control animals were injected with FCA/distilled water mixtures and distilled water. Induction injections were followed by a single 48-hour application of an occlusive patch (2 × 4 cm) moistened with 1% chlorphenesin in distilled water (0.5 mL, test animals) or distilled water (0.5 mL, controls). During the challenge phase, chlorphenesin (1% or 0.5% in distilled water, 0.5 mL) was applied, under occlusive patch (2 × 2 cm), to a new test site for 24 hours. Reactions were evaluated at 24 and 48 hours after patch removal. Chlorphenesin did not induce sensitization in guinea pigs at a concentration of 1%, followed by challenge with 0.5% or 1.0%. 22
Human
A human repeated insult patch test was used to evaluate the skin irritation and sensitization potential of a test material containing 5% to 9% chlorphenesin. 23 Fifty-five male and female patients (between 27 and 67 years of age) completed the study. Of the original 58 patients, 3 withdrew for reasons unrelated to test material application. During induction, a 1 × 1 in semiocclusive patch containing the test material (0.2 mg/cm2) was applied to the back, between the scapulae, of each patient. Patches were removed at 24 hours and any irritation reaction was scored 24 hours after patch removal. The scoring of reactions was followed by application of a new patch that remained for 24 hours. This cycle was repeated for a total of 9 consecutive patch applications (ie, 3-week induction phase). The 4-day challenge phase was initiated after a 10- to 14-day nontreatment period. A new patch containing 0.2 mL or 0.2 g of the test material was applied (24 hours) to a new test site on the back. Reactions were scored at 48 and 72 hours postapplication. Neither irritation reactions nor sensitization reactions were observed during the study, and it was concluded that the test material did not have dermal irritation or allergic contact sensitization potential.
The skin irritation and sensitization potential of a different test material containing 12% to 17% chlorphenesin was evaluated using 53 male and female patients (between 18 and 66 years of age). 24 Of the original 56 patients, 3 withdrew from the study and it was stated that 1 of the patients withdrew for reasons unrelated to test material application. The test material (0.2 mL or 0.2 g) was applied using a semiocclusive patch according to the test procedure mentioned earlier. In 1 patient, barely perceptible erythema (score = 0.5) was observed on day 19 of induction and mild erythema (score = 1) was observed on day 22. The mild erythema observed was classified as a transitory, weak response that could be considered clinically insignificant skin irritation. There was no evidence of skin sensitization in any of the patients tested.
In a multicenter study, the prevalence of preservative allergy in 584 patients (from 111 hospital dermatology departments in Korea) with cosmetic contact dermatitis symptoms was investigated. 25 From January 2010 to March 2011, the patients were patch tested to identify preservative allergens. An irritancy patch test (30 normal control participants) involving allergens at various test concentrations was also performed. Study results indicated preservative hypersensitivity in 41.1% of the patients and the allergens with the highest rates were as follows: benzalkonium chloride (12.1%), thimerosal (9.9%), and methylchloroisothiazolinone/methylisothiazolinone (5.5%). Results of the irritancy patch tests identified benzalkonium chloride and chlorphenesin as having the highest irritancy rate. At 4 days, 7 of the 30 normal patients had a positive irritant patch test reading to 0.1% benzalkonium chloride and 8 of 30 had the same reaction to 0.5% chlorphenesin in petrolatum. The authors noted that the maximum concentration of chlorphenesin for avoiding skin reactions is less than 0.5%.
Case Reports
A 38-year-old woman developed widespread acute dermatitis after using a proprietary antifungal powder and cream, both containing chlorphenesin. 26 Signs included severe maceration of the toe webs, with severe eczema of the foot. A generalized rash on the legs, forearms, and hands was also observed. Patch testing of individual constituents of the products used revealed a positive response only to 1% chlorphenesin in petrolatum. No reaction to this test concentration was observed in 3 control participants.
A 60-year-old atopic woman developed facial eczema within several hours after applying a foundation (cosmetic) containing chlorphenesin. 27 Patch testing revealed an allergic response (++ reaction) to 1% chlorphenesin in petrolatum. The patient was not patch tested with the foundation. In a second report, a 33-year-old woman who used a proprietary moisturizing cream containing chlorphenesin had a 1-month history of facial eczema. The eczema eventually involved the entire face and spread to the neck, upper chest, and upper arms. The patient had no personal or family history of atopy. Patch test results indicated a + reaction to 1% chlorphenesin in petrolatum and a +++ reaction to the undiluted moisturizer. Both reactions were observed by day 2 and persisted to day 4.
In another case report, a 24-year-old man applied an ointment containing 0.5% chlorphenesin to his feet twice daily to relieve itching. 28 Following 3 days of treatment, a symmetrical vesciculo-bullous eruption was observed on the dorsa of the feet. This reaction extended to the ankles and was accompanied by extensive eczema on the trunk and arms within 24 hours. Patch testing resulted in a ++++ reaction to 0.5% chlorphenesin in white soft paraffin and to the ointment.
Chronic dermatitis of the axillae was reported for a 29-year-old woman who used a deodorant that contained chlorphenesin. 29 She also had a past history of allergy to metallic jewelry. Patch results for the deodorant were positive at 48 hours (+ reaction) and 96 hours (+ reaction) and patch test results for 1% chlorphenesin in petrolatum were positive at 48 hours (+ reaction) and 96 hours (++ reaction). Positive reactions were not observed in 5 control participants patch tested with 1% chlorphenesin in petrolatum.
A 43-year-old woman experienced burning discomfort and developed a florid eczema after applying a facial moisturizer containing chlorphenesin. 30 The patient had a history of hay fever but no history of medicament or cosmetic intolerance. Patch test reactions were positive (++) for chlorphenesin on days 2 and 4. Positive patch test reactions were also reported for the product on day 2 (++) and day 4 (+).
Photoallergenicity
Eleven patients photoallergic to ketoprofen (a nonsteroidal anti-inflammatory drug) were photo patch tested with chlorphenesin. 31 Testing was initiated on day 0 and the patients were irradiated with UVA light (5 J/cm2) at day 2. Readings were obtained on days 3 and 4. There were no positive reactions in patients photo patch tested with chlorphenesin.
Immunosuppression
The immunosuppressive activity of chlorphenesin was evaluated using groups of 3 to 4 albino rabbits. 32 The groups were immunized with 1 mL of antigen (gram-positive bacteria [CA+] alone or antigen + chlorphenesin). A total of 3 intravenous (iv) injections (1 mL) of each was made on days 0, 3, and 7 according to the following procedure: group 1 (control) received the mixture of 1 part of CA(+) antigen (final dilution of 1:100) and 9 parts of buffer. Group 2 received a mixture of 1 part of antigen and 9 parts of chlorphenesin at concentrations of 0.01, 0.1, 1, or 10 mg/mL. Prior to injection, these mixtures were incubated (37°C) for 30 minutes. Group 3 received the same antigen–chlorphenesin mixtures without prior incubation. The fourth group received antigen and chlorphenesin, albeit separate injections. When tested at a concentration of 1 or 10 mg/mL, but not 0.01 or 0.1 mg/mL, chlorphenesin markedly inhibited the CA (+) hemagglutinin response. It was also noted that injection of the nonincubated mixture and separate administration of the 2 materials into separate ear veins caused an undiminished immune response. The results of additional experiments indicated that chlorphenesin suppressed antibody formation less effectively when larger amounts of antigen were used. With smaller amounts of antigen, chlorphenesin partially inhibited the antibody response, even at a concentration of 0.1 mg/mL.
The immunosuppressive activity of chlorphenesin was studied using a wide variety of in vitro assays for cellular immunity in both humans (25-40 years old) and mice (6-11 months old) of the following strains: BALB/c, C57Bl/6, and BDF1 (C57Bl × DBA) F1 mice. 33 At concentrations of 20 to 50 µg/mL, chlorphenesin inhibited mitogenic responses of B and T cells from mice and humans. Exposure to these doses for 72 hours did not result in death of B or T cells. Mixed lymphocyte reactions in cells from inbred strains of mice and unrelated humans were also inhibited at concentrations of approximately 50 µg/mL. In light of these results, the generation of cytotoxic T cells in cell-mediated lympholysis assays was not inhibited to the same extent as proliferation in mixed lymphocyte reactions. Also, the cytotoxic potential of presensitized mouse T cells for allogeneic targets was totally unaffected. The results of these studies suggest that chlorphenesin may have a broad spectrum of suppressive effects on both B and T lymphocytes and that the predominant inhibition of proliferative responses in these lymphocytes may reduce the expansion of clones of immunocompetent cells in vivo.
The effect of chlorphenesin on the immune response in mice, rabbits, and guinea pigs was studied. 34 Male Swiss Webster mice were injected with chlorphenesin mixed with sheep red blood cells or chicken red blood cells or penicillin conjugated with keyhole limpet hemocyanin, iv (volume = 0.1 mL). An assay for localized hemolysis was then performed, in which the degree of hemolysis was determined after 2 hours. Groups of 4 to 8 New Zealand White rabbits were used to determine the presence of circulating antibodies. The antigens were injected into the hind footpads and subcutaneously over each shoulder. The rabbits were bled and tested for antibody titers for up to 21 days postimmunization. Male albino guinea pigs were sensitized with BCG vaccine intradermally and challenged intradermally with tuberculin at 5 weeks postsensitization. In the localized hemolysis assay, partial hemolysis was noted at a chlorphenesin concentration of 10 mg/mL. The joint administration of an antigen with chlorphenesin (50 mg/kg dose) greatly reduced the number of antibody-forming cells in the spleen. The simultaneous administration of antigen with chlorphenesin also resulted in suppression of formation of humoral antibodies in mice and rabbits. Chlorphenesin was effective as an immunosuppressive agent only when administered jointly with an antigen, did not affect existing antibody levels or the secondary response, and did not increase the susceptibility of the animals to infections. If administered at the time of challenge, chlorphenesin (100 and 200 mg/kg doses) affected the bacillus Calmette-Guérin reaction (ie, significantly decreased the reaction to tuberculin) in guinea pigs.
Reproductive and Developmental Toxicity
The effect of chlorphenesin (suspension in 1% methylcellulose) on pregnancy and in utero development of the rat was evaluated using 4 groups of 25 sexually mature, specific pathogen-free female rats of the Crl: CD BR VAF/Plus strain (8 to 10 weeks old). 35 Three groups received oral doses (gavage; 10 mL/kg body weight) of 10, 50, and 100 mg/kg, respectively, once daily on days 6 to 15 postcoitum. The control group was dosed with the vehicle (1% methylcellulose) according to the same procedure. The animals were killed on day 20 and necropsy was carried out to identify any congenital abnormalities or macroscopic pathological changes in maternal organs. Tissues were preserved for microscopic examination. There was no evidence of maternal toxicity at any of the 3 administered doses, and neither maternal body weight gain nor food intake was affected by treatment. Increased fur loss and transient postdosing salivation were observed in the highest dose (100 mg/kg/d) group. Based on necropsy results, it was considered unlikely that fur loss was test substance related. At all doses administered, chlorphenesin had no adverse effect on embryo–fetal survival, growth, or development in utero. The no observed effect level for selective toxicity to the developing fetus was considered to be 100 mg/kg/d.
Genotoxicity
The genotoxicity of chlorphenesin was evaluated in the Ames test (bacterial reverse gene mutation assay) using the following Salmonella typhimurium strains: TA98, TA100, TA1535, TA1537, and TA1538. 36 Test concentrations up to 5000 µg/plate were evaluated with and without metabolic activation . 2-Aminoanthracene served as the positive control for metabolic activation cultures and 2-nitrofluorene, 9-aminoacridine, and N-ethyl-N ′-nitro-N-nitrosoguanidine served as positive controls for nonactivation cultures. Chlorphenesin was not genotoxic with or without metabolic activation over the range of concentrations tested. The positive controls were genotoxic. The same conclusion was reached in another Ames test evaluating the genotoxicity of chlorphenesin in Salmonella typhimurium strain TA102 and Escherichia coli strain WP2 uvrA over the same test concentration range (with and without metabolic activation). 37 Both positive controls (2-aminoanthracene and methyl methane sulfonate [nonactivation]) were genotoxic to both strains.
Chlorphenesin was also evaluated in a forward gene mutation assay using Chinese hamster ovary cells. 38 The test substance was evaluated at concentrations up to 1500 µg/mL with and without metabolic activation. In this assay, forward mutation at the functionally hemizygous hypoxanthine-guanine phosphoribosyl transferase locus is detected by the ability of the cells that have had genetic damage at this locus to form colonies in the presence of 6-thioguanine. Dimethyl sulfoxide served as the vehicle control and ethyl methanesulfonate (without metabolic activation) and 20-methylcholanthrene (20-Mc, with metabolic activation) served as positive controls. Without and with metabolic activation, dose-related cytotoxicity was noted at concentrations >850 µg/mL and >550 µg/mL, respectively. No significant correlation between mutant frequency and increasing dose levels was induced by chlorphenesin either with or without metabolic activation. Neither chlorphenesin nor the vehicle control was genotoxic with or without metabolic activation, whereas the positive controls exhibited the expected genotoxicity.
Carcinogenicity
Antitumorigenicity
In a study involving groups of Strain A (inbred strain) female mice, immune competence during initiation–promotion carcinogenesis was determined by the length of time required to reject allografts of tail skin and by the incorporation of [ 3 H]thymidine by lymphocytes in culture stimulated with the mitogens phytohemoagglutinin (PHA) and pokeweed mitogen (PWM). 39
During initiation–promotion carcinogenesis, mice were also treated with chlorphenesin, predicated on its reported effects to increase immunological reactivity, particularly cellular immunity. The skin grafting experiment for determining immune competency involved 5 groups of mice. The animals were not dosed with chlorphenesin. Initially, 2.5% croton oil (20 µL) was applied to the intrascapular area twice per week for 30 weeks and mice were then treated with a single application of 7,12-dimethylbenzanthracene (DMBA, 100 µg) 10 days later. The mice were then separated into 2 groups, those with and without tumors. In order to study the effect of the initiating and promoting agents, DMBA (100 µg) was applied to the interscapular area of each animal in the third group at 10 days before grafting. The fourth group was treated only with 2.5% croton oil (20 µL) according to the same procedure, and the fifth group served as the untreated control group. The allografts of the DMBA plus croton oil-treated, tumor-bearing mice were retained significantly longer (P < 0.02) than were the grafts on either the control mice or the DMBA plus croton oil-treated mice that had not developed tumors. The mice that received 1 application of DMBA 10 days before grafting were also inhibited (P < 0.02) from rejecting their skin grafts.
The experiment using lymphocyte cultures also involved 5 groups of mice. Groups 1 and 2 were treated with DMBA and croton oil, respectively (same doses), and group 3 received two 2.5-mg doses of chlorphenesin ip (same day). Group 4 received a dermal application of croton oil and 2 ip doses of chlorphenesin and group 5 served as the untreated control group. The mitogenic response of lymphocytes to PHA and PWM was determined using whole blood lymphocyte cultures. Chlorphenesin inhibited the stimulation of PWM mitogenesis observed in lymphocytes from croton oil-treated mice.
The tumor initiation–promotion experiment involved 2 groups of 30 Swiss mice. In the first group, DMBA (100 µg) was applied to the interscapular area of each animal, and, after 3 weeks, 2.5% croton oil was applied to the skin twice weekly for 20 weeks. Group 2 animals received applications of DMBA and croton oil plus two 2.5-mg injections of chlorphenesin ip (same day) at the same time that croton oil was applied. The animals were necropsied at 20 weeks. The carcinogen DMBA inhibited the cellular immune competence of mice, and lymphocytes from mice treated with croton oil had enhanced PWM response. Chlorphenesin inhibited tumorigenesis in initiation–promotion skin carcinogenesis when injected during promotion. 39
Female Swiss mice were injected ip (day 0) with 0.2 mL of Rauscher murine leukemia virus (RMLV) or Friend murine leukemia virus (FMLV) suspension and distributed randomly into paired groups of 18 to 20 mice each. 40 Chlorphenesin in warm Hank balanced salt solution (HBSS) was then injected ip (dose = 100 mg/kg in 0.5 mL) in the morning and late afternoon on each day of treatment. Chlorphenesin was injected into the RMLV mice on days 1, 2, 3, 4, 7, and 8 and FMLV mice received injections on days 1, 2, 6, 7, 9, 12, and 13. Control mice were injected with HBSS only after virus injection according to the same schedules. Injected virus routinely resulted in 80% mortality in leukemic control groups within 50 to 60 days. Chlorphenesin caused a pronounced sparing effect on mortality due to leukemia after infection with RMLV. Delayed onset of early death in chlorphenesin-treated mice was observed, but the most characteristic finding was the marked sparing effect in later stages of the disease. Mortality in mice dosed with chlorphenesin leveled off at 40%; however, controls continued to die at a nearly constant rate.
Additional experiments evaluating antiviral activity suggested that chlorphenesin was probably acting on malignant cells rather than against the transforming virus. In an effort to confirm this, Leukemia L-1210 in ascites form was implanted sc into B6DF1 mice and results indicated that chlorphenesin had little effect against conventional massive ip doses of this highly malignant cell line. However, when the system was modified using reduced numbers of cells implanted sc, the sparing effect was readily demonstrable. Although all control mice survived the 50-day study period, more than 40% of the treated mice survived until the experiment was terminated at 50 days, at which time there was no visible evidence of residual tumor.
Clinical trials involving patients with cancer were conducted by the Clinical Screening Group of the European organization for Research on Treatment of Cancer. Patients (31) with a wide range of neoplasms had been treated with chlorphenesin for periods ranging from 1 to 6 weeks. Oral doses ranged from 1 to 6 g daily, with a usual dose of 4 g/day. Treatment with chlorphenesin was ineffective in 16 cases of carcinoma (cervix, uterus, tonsil, esophagus, and lung) and in 4 cases of sarcoma. However, in 9 cases of squamous cell carcinoma of the skin, complete remission was achieved in 1 patient and substantial, though incomplete, remission was achieved in 4 other patients. Also, for 2 patients with basal cell carcinoma, no benefit was observed. 40
Summary
Chlorphenesin, a biocide, is produced by condensing equimolar amounts of p-chlorophenol and glycidol in the presence of a tertiary amine or a quaternary ammonium salt as a catalyst. According to information supplied to the FDA by industry as part of the VCRP in 2012, chlorphenesin was being used in 1386 cosmetic products. Furthermore, results from a survey of ingredient use concentrations provided by the Personal Care Products Council in 2011 indicate that chlorphenesin was being used at concentrations up to 0.32% (rinse-off products) and up to 0.3% (leave-on products). Similarly, the maximum authorized use of this ingredient as a preservative in cosmetic products marketed in the EU is 0.3%.
Some confusion in terminology may result because the drug chlorphenesin carbamate (CAS No. 886-74-8) has also been referred to as chlorphenesin. Chlorphenesin carbamate is a muscle relaxant whereas the cosmetic ingredient chlorphenesin (CAS No. 104-29-0) is not.
The results of a toxicokinetic study (oral dosing) involving rats and dogs indicated that chlorphenesin was rapidly absorbed and excreted mainly in the urine. Urinary end products identified included 3-p-chlorophenoxylactic acid, p-chlorophenoxyacetic acid, and unchanged chlorphenesin. In an in-vivo percutaneous absorption study involving rats, up to 57% of the applied dose was absorbed and practically all of the absorbed dose was excreted in urine over a period of 96 hours.
In an acute oral toxicity study (rats), a mean oral LD50 of 3,000 mg/kg was reported for chlorphenesin. Repeated oral dosing of rats with chlorphenesin for 28 days caused a significant decrease in body weight gain and significantly lower hemoglobin levels in the highest dose group (1,000 mg/kg/day) when compared to controls. Significantly decreased spleen and thymus weights were also reported for this group. The only treatment-related microscopic finding in the study, renal tubular dilatation/necrosis, occurred in one male rat from the highest dose group. A badly groomed appearance and increased salivation were observed in the 100 mg/kg/day dose group. A dose of 10 mg/kg/day was considered the no adverse effect level in this study.
Chlorphenesin was classified as a weak ocular irritant when instilled into the eyes of rabbits at a concentration of 1%. The same test concentration did not induce skin irritation when applied, under an occlusive patch, to rabbits for 24 hours. Negligible dermal irritation was observed in 3 of 25 patients tested with 0.3% chlorphenesin in a 48-hour occlusive patch test. In a facial sensory irritation test involving 16 healthy patients, irritation induced by a formula containing methylparaben, propylparaben, and chlorphenesin (0.4%, in 0.5% aqueous carbopol vehicle) was greater when compared to the same formula without chlorphenesin. In the guinea pig maximization test, chlorphenesin did not induce sensitization at a concentration of 0.5% or 1%. These 2 concentrations were classified as nonirritating in a preliminary test to determine the maximal irritant concentration.
In a human repeated insult patch test (HRIPT) involving 55 patients, a test material containing 5% to 9% chlorphenesin did not exhibit skin irritation or allergic contact sensitization potential. A test material containing 12% to 17% chlorphenesin induced clinically insignificant erythema in 1 of 53 patients in another HRIPT; skin sensitization was not observed in any of the patients. When 11 patients photoallergic to ketoprofen were photo patch tested with chlorphenesin, results were negative. In case reports, positive patch test reactions to 0.5% and 1% chlorphenesin were reported.
In a study evaluating the immunosuppressive activity of chlorphenesin in albino rabbits, marked inhibition of the CA (+) hemagglutinin response was observed at test concentrations of 1 or 10 mg/mL but not 0.01 or 0.1 mg/mL. In other animal studies, the simultaneous administration of antigen with chlorphenesin resulted in suppression of formation of antibodies in mice and rabbits. When the immunosuppressive activity of chlorphenesin was studied using a wide variety of in vitro assays for cellular immunity in both human and mouse test systems, the results suggested that it may have a broad spectrum of suppressive effects on both B and T lymphocytes. However, dosing with chlorphenesin did not increase the susceptibility of animals to infections in vivo.
Chlorphenesin had no adverse effect on embryo–fetal survival, growth, or development in utero when administered orally to rats at doses up to 100 mg/kg/d on days 6 to 15 postcoitum. In the Ames test, chlorphenesin was not genotoxic to the following bacterial strains when tested at concentrations up to 5000 µg/plate, with or without metabolic activation: S typhimurium strains TA98, TA100, TA102, TA1535, TA1537, and TA1538 and E coli strain WP2 uvrA. Chlorphenesin was also not genotoxic, with or without metabolic activation, in a forward mutation assay using Chinese hamster ovary cells.
In an initiation–promotion experiment designed to assess the antitumorigenic activity of chlorphenesin, DMBA (100 µg) was applied to the interscapular area of each of 30 mice and, after 3 weeks, 2.5% croton oil was applied to the skin twice weekly for 20 weeks. A second group of 30 mice received applications of DMBA and croton oil plus two 2.5 mg injections of chlorphenesin ip (same day) at the same time that croton oil was applied. Chlorphenesin inhibited tumorigenesis when injected during promotion. In another study, mice previously injected with murine leukemia virus (RMLV or FMLV) were injected ip with 100 mg/kg chlorphenesin for up to 7 days. Chlorphenesin caused a pronounced sparing effect on mortality due to leukemia after infection with RMLV. Thirty-one patients with cancer received chlorphenesin orally at a usual daily dose of 4 g/day for 1 to 6 weeks. Treatment was ineffective in 16 cases of carcinoma (cervix, uterus, tonsil, esophagus, and lung) and in 4 cases of sarcoma. However, in 9 cases of squamous cell carcinoma of the skin, complete remission was achieved in 1 patient and substantial, though incomplete, remission was achieved in 4 other patients.
Discussion
The CIR Panel noted that the drug chlorphenesin carbamate (CAS No. 886-74-8), sometimes referred to as chlorphenesin, has muscle relaxant effects not expected for the cosmetic ingredient, chlorphenesin (CAS No. 104-29-0).
Chlorphenesin induced low acute oral toxicity in rats, exhibited a no observable adverse effect level of 10 mg/kg/d in a 28-day repeated oral toxicity study involving rats, and elicited minimal ocular irritation potential in rabbits. Chlorphenesin was not a dermal irritant, sensitizer, or photosensitizer in animals or humans, except in a very small number of case reports. Chlorphenesin is not genotoxic in bacterial or mammalian assays. Oral and other carcinogenicity studies suggested antitumor activity. The ingredient was not an oral reproductive or developmental toxicant. When applied to the skin, chlorphenesin was well absorbed.
The Panel acknowledged the potential immunosuppressive activity of chlorphenesin, based on in vitro assay results. However, after considering that dosing with chlorphenesin did not increase the susceptibility of animals to infections or act as a tumor promoter in in vivo studies, it was agreed that there would be very little to no safety concern relating to the immunosuppressive activity of chlorphenesin as an ingredient under current conditions of use in cosmetic products.
The Panel considered the study in which chlorphenesin was reported to increase the sensory irritation potential of some creams, especially when used concomitantly with parabens + phenoxyethanol. The Panel had evaluated such sensory irritation potential when it considered AHA ingredients and determined that the sensitivity of tissue around the area of the eye to sensory irritation was such that AHA-containing products intended for use near the eye be formulated in such a way to reduce stinging and burning reactions. The AHA ingredients, however, were also known dermal irritants, whereas chlorphenesin is not. Concerns about sensory irritation may be more relevant for baby products, for example, diaper creams. Chlorphenesin, however, is not reported to be used in baby products.
Conclusion
The CIR Expert Panel concluded that chlorphenesin is safe in the present practices of use and concentration described in this safety assessment.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L St. NW Suite 1200 Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
