Abstract
Psychotropics are among the most prescribed medications. There are indications in the literature that fluoxetine (FLX; selective serotonin reuptake inhibitor [SSRI] antidepressant) and methylphenidate (MPH) could have a hormonal mode of action. This study was designed to evaluate the immature rat uterotrophic assay Substitute by (1) if sertraline (SER) and escitalopram (ESC) 2 other SSRI antidepressants would share the estrogenicity described for FLX and (2) MPH for estrogenicity and antiestrogenicity. The 18-day-old Wistar rats with were divided into olive oil, estradiol (0.3 mg/kg), estradiol + tamoxifen (10 mg/kg), SER (5, 15, or 45 mg/kg), ESC (2, 6, or 18 mg/kg), MPH (2.5 or 5 mg/kg), and estradiol + MPH groups. As expected, estradiol increased the weight of uterus, and this effect was counterbalanced by concomitant treatment with tamoxifen. The SER, ESC, and MPH had no effect on the uterus weight. The results suggest that ESC and SER do not share the estrogenicity described for FLX and that MPH does not disrupt estrogenic signaling.
Introduction
Psychotropics are among the most prescribed medications, and their consumption has grown substantially (∼60%) worldwide over the past decade. 1
Among psychotropics, antidepressants were the third most common medication taken by Americans of all ages 2 in 2005 to 2008, and the selective serotonin reuptake inhibitors (SSRIs) is the most prescribed class. The SSRI consist of 6 molecules: citalopram, escitalopram (ESC), fluoxetine (FLX), fluvoxamine, paroxetine, and sertraline (SER).
Another psychotropic medication which use has been increasing lately is methylphenidate (MPH), primarily used to treat attention deficit/hyperactivity disorder in children and adolescents. 3
There are some studies in the literature indicating that both FLX and MPH could have a hormonal mode of action. A list of studies suggesting endocrine effects for FLX in aquatic animals, rodents, and humans may be obtained in Muller et al’s study, 4 who also reported a weak but significant estrogenic activity for FLX both in vivo (uterotrophic assay in rats) and in vitro (Michigan Cancer Foundation 7 [MCF-7] cells). Regarding MPH, delayed vaginal opening, and alteration in estrous cycles were reported in rats after repeated treatment, indicating altered maturation and functioning of the reproductive axis. 5,6
This study was designed with 2 objectives: (1) to evaluate whether other SSRI drugs (SER and ESC) would share the estrogenicity described for FLX and (2) to evaluate MPH for estrogenicity and antiestrogenicity. The evaluation was carried out in the immature rat uterotrophic assay, which is one of the tests listed by the Environmental Protection Agency from USA (US EPA) for the screening of chemicals with estrogen-mediated effect. 7 The SER and ESC were chosen because although all SSRI share the ability to inhibit the serotonin transporter, each molecule is unique regarding their full profile of pharmacological actions: FLX blocks the noradrenaline transporter, inhibits CYP 2D6 and 3A4, and is an antagonist of 5-HT2C receptor; SER blocks the dopamine transporter and is an agonist of the opioidergic σ1 receptor; and ESC is solely described as an inhibitor of the serotonin transporter. 8
Materials and Methods
Drugs and Solutions
Estradiol valerate (Pharma Nostra, Rio de Janeiro, Brazil) and tamoxifen citrate (Pharma Nostra) were dissolved in olive oil and distilled water, respectively. The MPH chlorhydrate (Ritalin; Novartis, Brazil), SER chloride (Valdequímica Produtos Químicos, São Paulo , Brazil), and ESC oxalate (Pharma Nostra) were dissolved in distilled water immediately before the administration, and the doses were calculated based on the free base. The dosing volume for all solutions was 5.0 mL/kg body weight.
Animals
A total of 11 male and 21 female Wistar rats from the State University of Londrina colony were mated. The 19 dams gave birth to litters containing at least 8 pups. Litters with more than 12 pups were culled to 12 pups keeping as many females as possible. The day of birth was considered postnatal day (PND) 0, and on PND 18 female pups were weaned, weighed, and randomly assigned to the experimental groups. Littermates were not used in the same experimental group. Weight variation among animals was approximately 15% and 11% for the evaluation of the antidepressants and MPH, respectively. The females were housed in collective polypropylene cages (29 cm × 18 cm × 13 cm) with wood shavings bedding separated by the experimental group. They were kept in a controlled environment with temperature at 21°C ± 2°C; humidity of 55% ± 5%; 12-hour light–dark cycle (lights on at 6:00
All the experiments described subsequently were conducted simultaneously, and even though for the statistical analysis subgroups were selected depending on the hypothesis being tested, it is important to mention that there was only 1 group that received olive oil and only 1 that received estradiol.
This study was approved by the ethical commission for animal use from the State University of Londrina (CEUA 22978.2012.40 and 16381.2012.45).
Uterotrophic Assay
The guideline OPPTS 890.1600 from EPA 7 was followed except that body weights were recorded only on PND 18 and 21 and not daily as recommended in the guideline. Moreover, the 2 groups treated with the lower doses of SER and ESC ended up with 5 animals instead of 6 which is the minimum suggested in the guideline.
The test drugs and the appropriate control chemicals were administered for 3 consecutive days (from 1:00 to 1:30
During the 3-day treatment period, general clinical observation was conducted.
Evaluation of SER and ESC in the Uterotrophic Assay
Considering that FLX was described to present estrogenic activity, SER and ESC were evaluated only for estrogenicity. Rats were divided into the following experimental groups: Olive oil (negative control group): animals were gavaged with olive oil. Estradiol (positive control group): animals were gavaged with 0.3 mg/kg estradiol. SER: animals were gavaged with 5 mg/kg SER (SER 5), 15 mg/kg SER (SER 15), or 45 mg/kg SER (SER 45). ESC: animals were gavaged with 2 mg/kg ESC (ESC 2), 6 mg/kg ESC (ESC 6), or 18 mg/kg ESC (ESC 18).
In humans, SER and ESC prescriptions range from 50 to 150 mg/d and from 10 to 20 mg/d, respectively, which would correspond to approximately 0.83 to 2.5 and 0.17 to 0.33 mg/kg/d, respectively. Considering that the precautionary principle considers animals more resistant than humans, higher doses are usually tested in animals. For comparison purposes, we applied body weight scaling to the 3/4 power (BW3/4), which is considered a general default procedure to extrapolate toxicologically equivalent doses of chronic orally administered agents from laboratory animals to humans. 9 Applying this scaling method, the dose of 50 mg/d of SER in humans (with 60 kg body weight) would correspond to 5 mg/kg/d in rats (with 45 g body weight) and the dose of 20 mg/d of ESC would correspond to 2 mg/kg/d in rats. Based on this exercise and on articles published in the literature, the range of doses employed in this study was chosen. It is interesting to note that the lower dose would be equivalent to the therapeutic dose and then a factor of 3 was applied between the doses to reach a maximum dose that would be, approximately, 10 times higher than the “therapeutic” dose, that is, would account for the intraspecies safety factor.
Evaluation of MPH in the Uterotrophic Assay
Methylphenidate was evaluated for both estrogenicity and antiestrogenicity. For the estrogenicity evaluation, the following experimental groups were evaluated: olive oil and estradiol (as previously described) and MPH-treated groups, which were gavaged with 2.5 mg/kg MPH (MPH 2.5) or 5 mg/kg MPH (MPH 5.0). For the antiestrogenicity evaluation, the rats were divided into the following groups: Estradiol (negative control group). Estradiol + tamoxifen (positive control group for antiestrogenicity): animals were gavaged with estradiol in addition to 10 mg/kg tamoxifen. Estradiol + MPH: animals were gavaged with estradiol in addition to MPH 2.5 and 5 mg/kg.
In children, the effective dose range is 0.3 to 1.0 mg/kg MPH. 10 Applying the BW3/4 scaling, the equivalent doses in rats would be 1.7 to 5.5 mg/kg. The highest dose (5 mg/kg) used in this study would be equivalent to a clinically relevant dose in humans, and higher doses were not tested because pilot studies from our laboratory indicated that 5 mg/kg would already compromise weight gain.
Statistical Analysis
Initially, an exploratory analysis was conducted to evaluate normal distribution (Shapiro-Wilk test) and homogeneity of variance (Levene test). In the absence of normal distribution and/or homogeneity of variance, variables were transformed in order to achieve the criteria for parametric analysis. Initial and final body weights were analyzed by repeated measures analysis of variance (ANOVA) complemented with Fisher least significant difference (LSD). The weights of uteri with and without fluid were analyzed by ANOVA complemented with 1-tailed Dunnett and also by analysis of covariance (ANCOVA) using the final body weight as the covariate as suggested in the guideline from EPA. 7 However, since ANCOVA and ANOVA provided similar results, the ANOVA results are being reported. Differences were considered statistically significant if P < 0.05.
Results
All the animals showed good health and no signs of general toxicity.
Evaluation of SER and ESC in the Uterotrophic Assay
Repeated measures analysis of variance indicated an effect of day on weight gain (F 1, 49 = 271.03, P < 0.0005) but lack of interaction day × treatment (F 7, 49 = 0.71, P = 0.66) indicating that all the groups have gained weight in a similar way (Figure 1A).
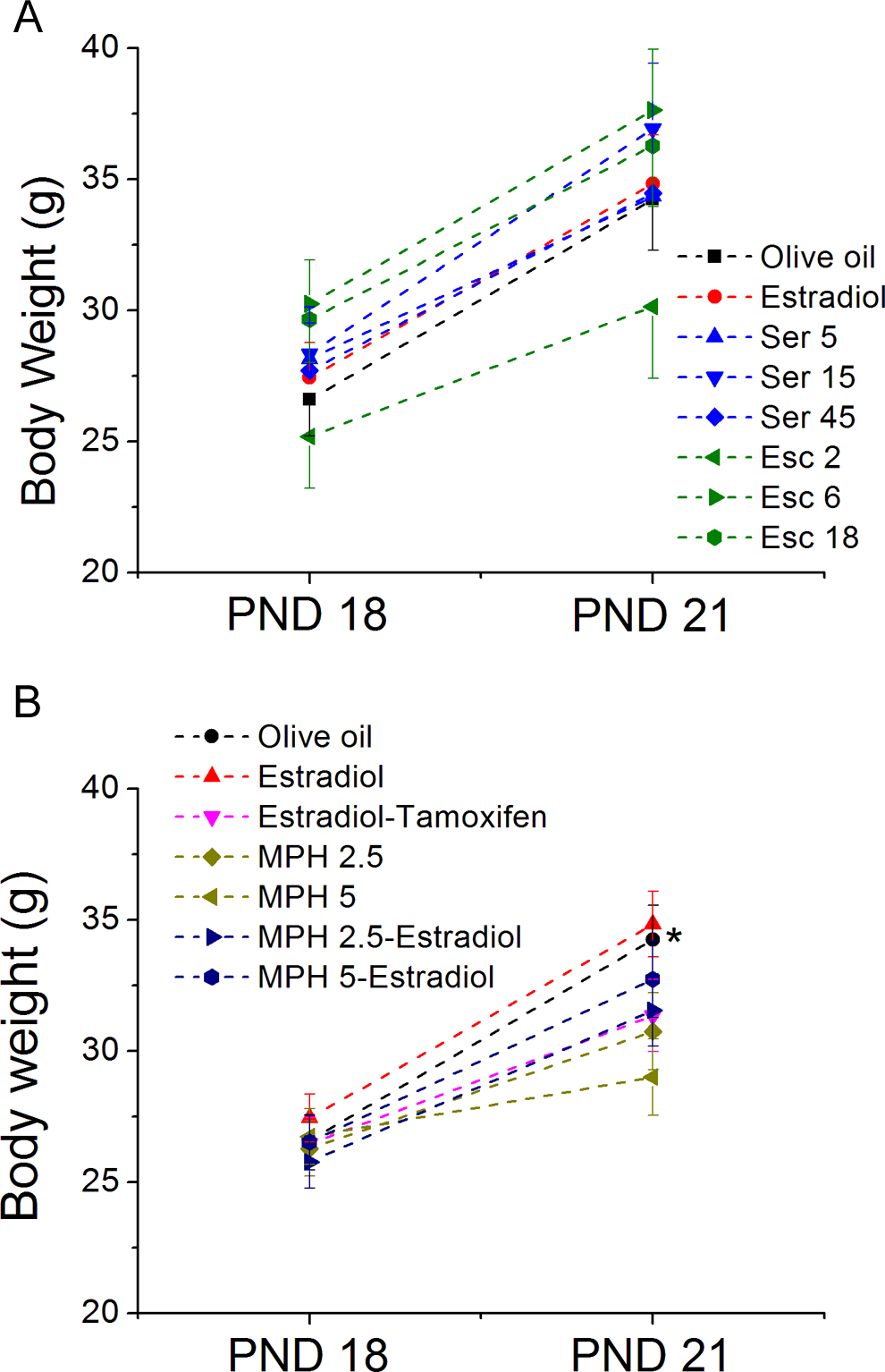
Body weight of animals on postnatal days (PND) 18 and 21. Data are mean ± standard error of the mean (SEM). In (A) RMANOVA indicated a significant effect of day for all the groups but any effect of drug treatment. In (B) RMANOVA indicated a day × treatment interaction, and least significant difference (LSD) multiple comparisons indicated that on PND 21, methylphenidate (MPH) 5 was different from olive oil. RMANOVA indicates repeated measures analysis of variance.
The analysis of the uterus weight with and without fluid (Table 1) indicated a significant effect of treatment (F 7, 57 = 41.1 and 42.4, respectively, P < 0.0005). Dunnett post hoc test showed that, as expected, the weight of the uterus with and without fluid was higher in rats treated with estradiol (positive control for estrogenicity) compared to vehicle (olive oil). The SER and ESC did not influence uterine weight (P > 0.05).
Evaluation of Sertraline (SER) and Escitalopram (ESC) for Estrogenicity in the Uterotrophic Assay Conducted With Rats Sexually Immature.a
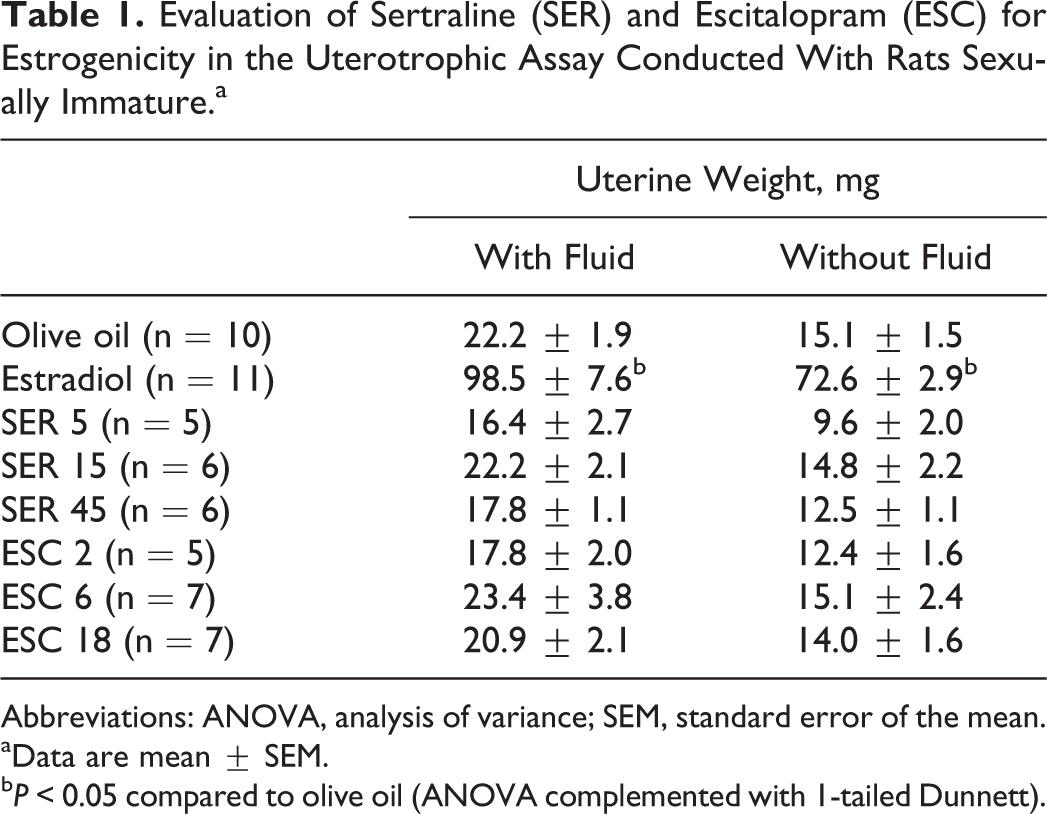
Abbreviations: ANOVA, analysis of variance; SEM, standard error of the mean.
aData are mean ± SEM.
b P < 0.05 compared to olive oil (ANOVA complemented with 1-tailed Dunnett).
On PND 21, vaginal opening was observed only in 1 female from the estradiol group.
Evaluation of MPH in the Uterotrophic Assay
Repeated measures analysis of variance indicated a day × treatment interaction (F 1, 56 = 7.1, P < 0.0005) on the body weight among the experimental groups. Multiple comparisons conducted with LSD indicated that even though the initial weight was similar among groups, on PND 21, MPH 5 presented decreased body weight when compared to animals treated with olive oil.
Results from the study designed to evaluate MPH for estrogenicity are presented in Table 2. The ANOVA complemented with Dunnett demonstrated that estradiol increased the uterus weight with and without fluid (F 3, 37 = 65.8 and 98.2, respectively, P < 0.0005), whereas MPH lacked effects.
Evaluation of Methylphenidate (MPH) for Estrogenicity in the Uterotrophic Assay Conducted With Rats Sexually Immature.a
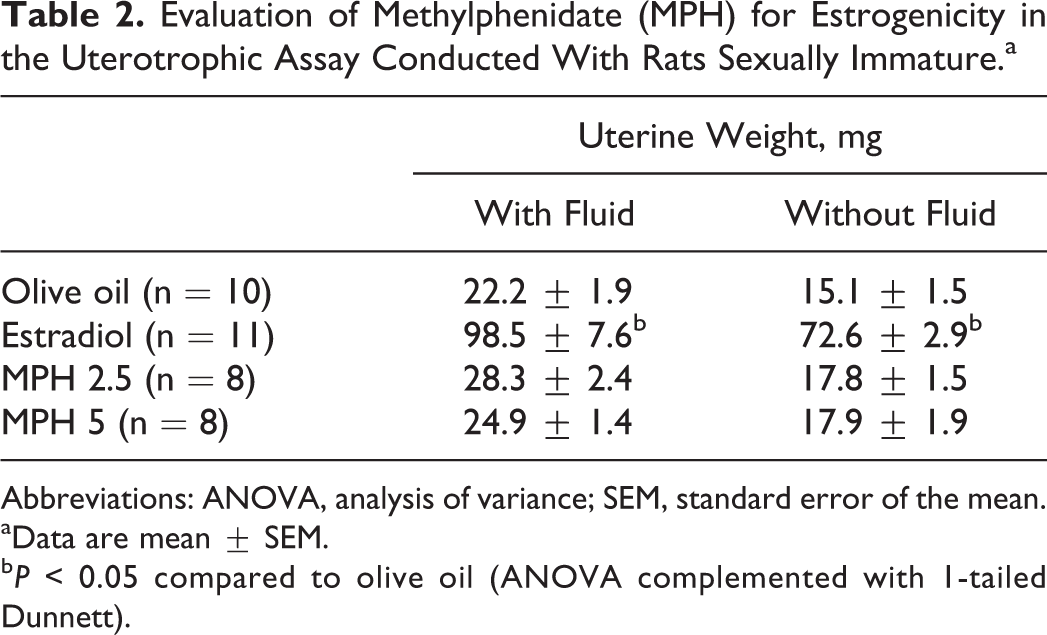
Abbreviations: ANOVA, analysis of variance; SEM, standard error of the mean.
aData are mean ± SEM.
b P < 0.05 compared to olive oil (ANOVA complemented with 1-tailed Dunnett).
Table 3 shows the results from the study designed to evaluate a possible antiestrogenicity of MPH. The ANOVA complemented with Dunnett revealed that cotreatment with tamoxifen prevented estradiol-induced increase in uterine weight with and without fluid (F 3, 37 = 5.3 and 9.2, respectively, P < 0.0005). This same effect was not observed when females were cotreated with estradiol and MPH, indicating that MPH did not act as an antiestrogenic compound in the uterotrophic assay.
Evaluation of Methylphenidate (MPH) for Antiestrogenicity in the Uterotrophic Assay Conducted With Rats Sexually Immature.a
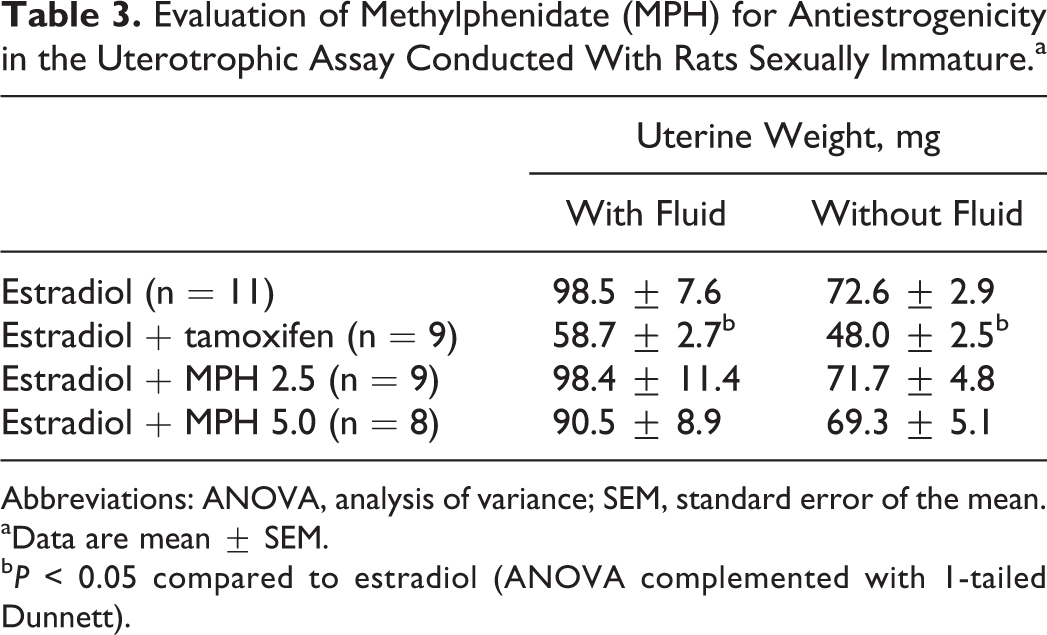
Abbreviations: ANOVA, analysis of variance; SEM, standard error of the mean.
aData are mean ± SEM.
b P < 0.05 compared to estradiol (ANOVA complemented with 1-tailed Dunnett).
On PND 21, vaginal opening was observed in 1 female from the estradiol group, 1 female from the estradiol + MPH 2.5, and 2 females from the estradiol + MPH 5 group.
Discussion
One of the aims of this study was to evaluate whether SER and ESC would share the estrogenicity described for FLX in the uterotrophic assay. 4 The present results suggest that this action is not common to the 2 SSRI molecules evaluated.
The uterotrophic assay is sensitive to both direct-acting (via the estrogenic receptors) and indirect-acting (eg, effects on steroidogenesis, aromatization, and degradation) agents.
Although hormone-dependent effects have been described for SSRI, specially for FLX, which is the oldest and most studied molecule from this group, the mode of action for these hormone-dependent end points is still speculative, and there are at least 3 possible explanations for these effects. First, considering the known modulation of the hypothalamus–pituitary–gonad (HPG) axis by serotonergic innervation, 11,12 increased serotonin availability after SSRI administration could influence HPG homeostasis. Second, a direct agonistic action in the estrogenic receptor was described for FLX in breast cancer cells (MCF-7) using the gene reporter assay. 4 Third, SSRI are being described to influence steroidogenesis through altering aromatase expression (which converts androgens into estrogens) or modulating the activities of 5α-reductase (which reduces progesterone to 5α-dihydroprogesterone) and 3α-hydroxysteroiddehydrogenase (which reduces α-dihydroprogesterone to allopregnanolone). 13,14 It is interesting to mention that different SSRIs (FLX, SER, and paroxetine) may impact enzymes involved in steroidogenesis with different affinities. 13 Moreover, in a study conducted with male fish, exposure to FLX but not to SER resulted in plasma vitellogenin induction, which is a marker for estrogenicity. 15
Based on the above-mentioned considerations, our result supports the idea that even though SSRI molecules have similar modes of action as psychotherapeutic drugs in humans, their different molecular structures make them unique when considering hormonal end points. Whether these differences are mainly driven by pharmacodynamic or pharmacokinetic profile remains to be determined.
Regarding MPH, there are many studies evaluating its toxicity, but little is known about its effects on the reproductive axis. Chatterjee-Chakrabarty et al 5 reported poor vaginal opening and alteration in estrous cycle and folliculogenesis in females receiving 450 µg/d MPH through a subcutaneous pump from PNDs 28 to 56. Delayed vaginal opening and alteration in estrous cycle have also been reported when females (5-7 or 21-23 days old) were treated subcutaneously twice daily with 35 or 100 mg/kg MPH for 21 or 30 days, respectively. 6 The reported end points may be influenced by estrogenic signaling, but other hormones, such as gonadotrophins, are also involved. The lack of MPH effect in the uterotrophic assay observed in the present study suggests that the described reproductive effects possibly do not result from direct alterations in the estrogenic signaling. Moreover, considering that in both the studies 5,6 MPH treatment induced significant reduction in body weight gain, the role that general toxicity may play in the reproductive effects observed remains to be elucidated.
Weaknesses of this work include the lack of information regarding phytoestrogens levels in the laboratory chow, water, and bedding material employed. However, EPA guideline reports that the uterotrophic assay may be compromised if high levels of phytoestrogens are present in the laboratory chow, which may also result in early puberty. In this work, early puberty (vaginal opening) was observed only in 4 females, and all of them have been treated with estradiol. In addition, EPA guideline recommends analysis of phytoestrogen content in the diet in the case of high uterine control weight relative to historic controls or an inadequate response to the reference estrogen, which did not occur in the present study. Finally, the relation mean uterus weight (mg)/mean body weight (g) in the control group of this work was 0.64, whereas in studies conducted with immature Wistar rats a relation of 0.67 to 0.82 was present, 16 –19 supporting that the rats from our laboratory do not seem to be under exogenous estrogenic influence.
In conclusion, results from the uterotrophic bioassay conducted with immature female rats suggest that ESC and SER do not share the estrogenicity described for FLX and that MPH does not disrupt estrogenic signaling.
Footnotes
Authors’ Note
B. Montagnini and S. Bortolan received Master and PhD fellowships, respectively, from Coordenadoria de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). B. D. dos Santos and A. P. Moreno received junior research fellowships from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and SETI/Fundação Araucária, respectively. E.G. Moreira received senior research fellowship from SETI/Fundação Araucária. BM and SB equally contributed to the data collection. EGM and DCCG share senior authorship of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
