Abstract
In this study, we investigated the effects of polychlorinated biphenyls (PCBs) and organochlorinated pesticides on the serum levels of luteinising hormone (LH), follicle stimulating hormone (FSH) and weights and histomorphometry of uterine tissue in immature female rats using uterotrophic assay. A total of 36 rats were randomly divided into six groups (n = 6 per group) as control, oestradiol (E2, 100 μg/kg), PCB 180, Aroclor 1221, endosulfan and mirex at 10 mg/kg dosage. After 3 days of injections (subcutaneous), animals were decapitated and blood samples were collected. Uteri were dissected, weighed out and then fixed in 10% formaldehyde. They were processed for histomorphometry. The serum levels of LH and FSH were determined by enzyme immunoassay.
Uterine weight was significantly increased by E2 and reduced by mirex (p < 0.001 and p < 0.05, respectively). Total volume of uterus was significantly raised by E2, Aroclor 1221 and endosulfan compared with that of the control group (p < 0.01). The ratio of epithelium was significantly increased by E2, PCBs and pesticides (p < 0.01). The uterine cavity ratio was decreased by aroclor (p < 0.01), PCB 180 and mirex (p < 0.05). The serum levels of LH did not significantly differ among the groups but the levels of FSH were decreased by PCB 180 and endosulfan (p < 0.05 and p < 0.01, respectively). These findings suggest that PCB 180, Aroclor 1221 and endosulfan may be estrogenic in immature uterotrophic assay.
Introduction
Endocrine disrupters are defined as chemicals that interfere with the normal function of hormones, resulting in abnormal development, growth or reproduction. 1 These xenohormones exert their effects mainly by mimicking or antagonising actions of estrogens. 2 Several environmental contaminants have been reported to have “endocrine disrupter” effects including polychlorinated biphenyls (PCBs) and organochlorinated pesticides (OCPs). 3,4
PCBs are the mixtures of different congeners having chlorine atoms of various numbers (1–10) and positions (ortho, meta and para) around biphenyl rings. 5,6 They were widely used as insulators in capacitors and transformers, because of their relative inflammability and insulating properties, as hydraulic fluids and in paints and caulk. Because of their lipophilic nature and persistent properties, they remain global contaminants in both the environment and the human body 7 in spite of the fact that their production was banned in most countries in the late 1970s. 8
PCB mixtures are immunotoxic 9 , carcinogenic 10 , neurotoxic 11 and cause birth defects. 12 However, different PCB congeners have varying patterns of toxicity. Congeners with one or no chlorines in the ortho positions (mono-ortho and non-ortho, respectively) are able to assume a planar configuration, bind to the cytoplasmic aryl hydrocarbon receptor (AHR) and have a pattern of toxicity similar to 2,3,7,8-tetrachlorodibenzo-p-dioxin. 13 However, there is also abundant evidence from in vivo and in vitro studies that the ortho-substituted, non-coplanar, non-dioxin–like congeners have toxic effects on various organ systems. 14,15 2,2′,3,4,4′,5,5′-Heptachlorobiphenyl (PCB 180) is an ortho-substituted non-planar PCB congener that has been reported to bind to the estrogen receptor (ER) in cell culture models. 16 Aroclor 1221 is a technical PCB mixture that comprises many different congeners with a chlorine percentage of approximately 21%. Although this low-chlorinated PCB mixture has been suggested to have estrogenic effect in vivo, 17 aromatase inhibition by Aroclor 1221 has also been reported using in vitro models. 18
OCPs (such as endosulfan and mirex) are persistent environmental pollutants. 19 , 20 They too tend to bio-accumulate both in the environment and living organisms due to their lipophilic features and chemical stability. Residues of OCPs have been found in human tissues all over the World. 19,21,22 Some of these compounds resemble 17β-oestradiol (E2) in terms of chemical structure and thus can bind to ER and mimic the effects of the endogenous ligand. Some xenohormones may display anti-estrogenic actions by binding to AHR and initiating transcription of cytochrome p450 enzymes that metabolise E2. 3
Uterotrophic assay is an internationally recognised screening method to determine estrogenic and anti-estrogenic properties of chemicals in vivo. 23,24 It is based on the development of uterine tissue under the influence of estrogen. We have investigated the effects of PCB 180, Aroclor 1221 and two OCPs (endosulfan and mirex) on the serum levels of luteinising hormone (LH), follicle stimulating hormone (FSH) and weights and histomorphometry of estrogen-sensitive tissues (uterus) in prepubertal female rats using uterotrophic assay.
Materials and methods
Animal experiments
Immature female Wistar rats aged 20 days were obtained from the Yeditepe University Medical School Experimental Research Center (YUDETAM). The animals were housed three per cage, in a controlled temperature (21 ± 2°C), relative humidity (40–60%) and artificial lighting at 12 h light–12 h dark cycle (lights on from 07.00 to 19.00 h) conditions. Standard rodent diet and water were provided ad libitum. The experimental protocol was approved by the local animal research ethics committee. A modified version of OECD test guideline 440 for the uterotrophic assay was used. 24 The phytoestrogen content of the standard rodent diet used in the present study was not analysed. However, it was confirmed by the manufacturer that they did not use soy products in the diet.
A total of 36 rats were randomly divided into six groups (n = 6 per group) as control (0.1 ml corn oil), oestradiol (E2, 100 μg/kg), PCB 180, Aroclor 1221, endosulfan and mirex at 10 mg/kg dosage. Subcutaneous (s.c.) injections were given for 3 days, and after 24 h, all animals were decapitated (day 24). Blood samples were collected and centrifuged (1500g, 4°C, 10 min). Serum samples were extracted and kept at −20°C until assayed. Body weights of all animals in the beginning and before decapitation were recorded.
Histomorphometry
Uteri were removed and weighed out wet. Uterine blotted weight was not determined and the uteri samples were processed for histomorphometry. A piece (4 mm) from the mid-segment, cut perpendicular to the longitudinal axis of the uterus, was dissected out for morphometric analysis. All specimens were fixed in a 4% formaldehyde solution and stored at room temperature. Finally, the samples were dehydrated in a series of alcohols, embedded in paraffin and cut into serial sections of 10 μm in the horizontal plane using a rotary microtome (HM 325 Microm, Walldrof, Germany). The uterus section sampling factor was 40. The sections were mounted onto a poly-
For image and stereological analyses a semi-automated stereology workstation composed of a CCD digital camera (Optronics Microfire 1600 × 1200P, Goleta, California, USA), an image capture card ATI FireGL (Advance Micro Device, Camberley, UK), a personal computer and computer-controlled motorised specimen stage (Bioprecision, Howthrone, New York, USA), a microcator (Heidenhain, Traunreut, Germany) and a light microscope (Leica DM 4000B, Wetzlar, Germany) were used.
The same sets of consecutive sections of uterus were used for the volume fraction of the epithelium, glands and lumen. The Cavalieri principle was used to estimate the volume of the uterus. A point counting grid (PCG) (i.e. d = 50 μm) was applied for volume estimation to obtain maximum efficiency. The representative areas per point were 2500 μm 2 for each point. After applying the PCG to the sampled sections with different markers in a systematic–random method, we counted the number of points hitting epithelial, glandular cells and luminal area in the uterus. Epithelial, glandular, lumen and total uterus volumes were calculated using a software (StereoInvestigator version 7.5, Microbrightfield, Colchester, Vermont, USA) that uses a point-counting technique based on the Cavalieri principle. 25 The efficiency of sampling and point density for volume estimation were checked by the estimation of coefficient of error as described previously. 26
Enzyme immunoassays for LH and FSH
LH and FSH were analysed according to Pappa et al. 27 with some modifications. Briefly, 96-well immunoplates (Nunc, Roskilde, Denmark) were coated with rat LH or rat FSH. Serum samples or standards were pre-incubated with primary antibodies and were then transferred into coated plates for competition with antigens in the solid phase. Plates were washed and the secondary antibodies conjugated with horseradish peroxidase were added to each well; the colour developed using tetramethylbenzidine as a substrate were read at 450 nm using a plate reader (Tecan Spectra III, Grodig, Austria). Rat LH, rat FSH and primary antibodies (rabbit anti-rat LH and rabbit anti-rat FSH) were obtained from Dr A.F. Parlow (NIDDK, NIH, USA) and secondary antibodies (goat anti-rabbit IgG) conjugated to horseradish peroxidase were purchased from Sigma (Sigma-Aldrich, Taufkirchen, Germany).
Statistics
Results were expressed as mean ± SEM. Histomorphometric data were analysed using the independent sample t test. Uterine weight percentages and hormone levels were evaluated using one-way analysis of variance (ANOVA). p < 0.05 was considered to be statistically significant.
Results
The values of pre- and post-treatment body weight and uterine weight are summarised in Table 1. Percentage of uterine weight/body weight was significantly increased by E2 (p < 0.001). Although endosulfan increased and PCB 180 lowered this parameter, the alterations were not significantly different from the control values. The uterine ratio was significantly lowered by mirex (p < 0.05).
Pre- (day 20) and post-treatment (day 24) body weights, uterine weight and ratio of uterine weight (mean ± SEM) to the last body weight in immature female rats.

ANOVA: analysis of variance; PCB: polychlorinated biphenyl.
a p < 0.001 compared with the control group using one-way ANOVA.
b p < 0.05 compared with the control group using one-way ANOVA.
Both PCBs and OCPs produced distinct changes in percentage volumes of total uterus, epithelium, glands and lumen ratio (Figure 1). Histomorphometric parameters are shown in Table 2. Total volume of uterus was significantly raised by E2 compared with that of the control group (p < 0.001). This parameter was also increased by Aroclor 1221 (p < 0.01) and endosulfan (p < 0.05). Although PCB 180 treatment also raised the total uterine volume, this increase was not statistically significant. The ratio of epithelium was significantly increased by E2, Aroclor 1221 and endosulfan (p < 0.001). Growth of epithelium was also stimulated by PCB 180 and mirex (p < 0.01). Percentage of glands did not significantly differ in the E2 and endosulfan groups, but significantly reduced by Aroclor 1221, PCB 180 (p < 0.001) and mirex (p < 0.01). The uterine cavity ratio was significantly decreased by aroclor (p < 0.01), PCB 180 and mirex (p < 0.05).

Histomorphometric evaluation of haematoxylin–eosin stained tissue samples; (a) control, (b) oestradiol, (c) Aroclor 1221, (d) PCB 180, (e) mirex and (f) endosulfan. Scale bars: upper panel (a1, b1, c1, d1, e1 and f1) 110 µm; lower panel (a2, b2, c2, d2, e2 and f2) 27 µm. Epithelial height is demonstrated with (  ).
).
Histomorphometric analyses of uterine tissue from immature rat (mean ± SEM).
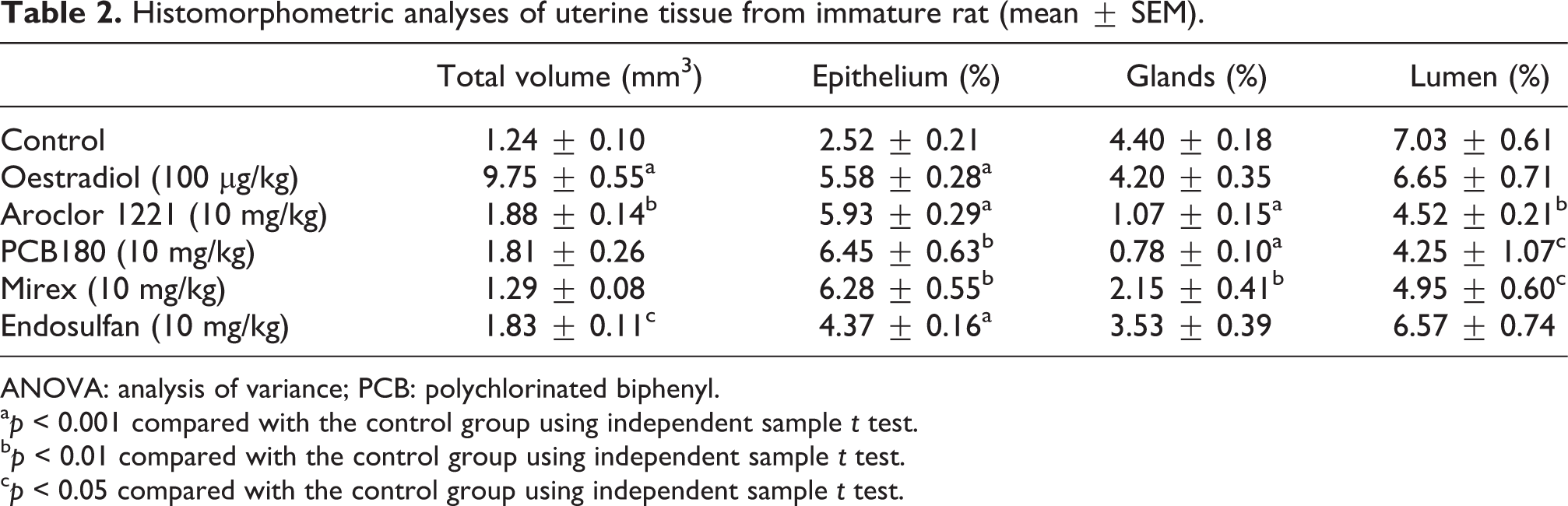
ANOVA: analysis of variance; PCB: polychlorinated biphenyl.
a p < 0.001 compared with the control group using independent sample t test.
b p < 0.01 compared with the control group using independent sample t test.
c p < 0.05 compared with the control group using independent sample t test.
The serum levels of LH did not significantly differ among the treatment groups, although they were lower in the E2, Aroclor 1221 and endosulfan groups compared with those of the control values (Figure 2). The serum levels of FSH were significantly lower in the E2 (p < 0.05), PCB 180 (p < 0.05) and endosulfan (p < 0.01) groups compared with those of the control group. Changes in FSH in the Aroclor 1221 and mirex groups did not significantly differ from the control group.

Serum levels of LH (a) and FSH (b) (ng/ml, mean ± SEM) in immature female rats following exposure to oestradiol, PCBs and pesticides. a: p < 0.05 and b: p < 0.01 compared with the control group using one-way analysis of variance (ANOVA).
Discussion
In the present study, administration of E2 to immature female rats produced uterotrophic effects. It dramatically increased the uterine wet weight and ratio to body weight when compared with the control group. Histomorphometric analysis has also shown that E2 caused significant increases in total uterine volume and epithelium. Although treatment with PCBs (PCB 180 and Aroclor 1221) and pesticides (mirex and endosulfan) caused distinct changes in uterus histomorphometry, uterine weight or ratio to body weight was not significantly affected (except in the mirex group). To the best of our knowledge, this is the first study examining histomorphometric changes in uterine tissue following the PCB and OCP treatment.
PCB 180 is a wide-spread environmental contaminant and has been detected in sea foods, human serum and breast milk. 28 –30 Based on the chemical structure, it has been suggested that ortho-substituted PCB congeners may elicit estrogenic responses. 31 Indeed, PCB 180 has been proposed to have estrogenic effects using in vitro yeast reporter gene assay. 30 Recently, weak anti-estrogenic action of this non-planar PCB congener has also been reported using an in vitro model (MCF-7 cells). 32 It appears that PCB 180 exerts both estrogenic and anti-estrogenic effects, which is not unusual for endocrine-active substances. 33 Aroclor 1221, a lower chlorinated PCB mixture, has also caused environmental contamination. 8,34 Neither PCB 180 nor Aroclor 1221 has been studied in terms of its uterotrophic effects using an in vivo model. The present results have shown that PCB 180 or Aroclor 1221 had no significant effect on the uterine weight or ratio. However, they both increased total uterine volume and ratio of epithelium, and reduced the ratio of glands and lumen in the uterus tissue. Our findings suggest that PCB 180 and Aroclor 1221 may have estrogenic effects in immature uterotrophic assay. Another technical PCB mixture with higher percentage of chlorination (Aroclor 1242) has also been reported to cause uterotrophic effects in the immature rat model. 35 A review by Weiderpass et al. 36 reported no evidence for the hypothesis that organochlorine exposure increases the risk of endometrial cancer. However, a recent study has reported a correlation between increased levels of xenoestrogens and endometrial stromal sarcomas in patients. 37
Endosulfan has been suggested to be estrogenic in various in vitro models. 38,39 It has been documented previously that endosulfan had no significant uterotrophic effect in mice 23 and up to 3 mg/kg is given orally combined with deltamethrin in rats. 40 We also observed the lack of effect of this OCP on uterine weight ratio. However, in the present study, s.c. injection of endosulfan at 10 mg/kg dose increased total uterine volume and surface epithelial ratio. These findings suggest that endosulfan produces estrogen-like effects at a high dose (10 mg/kg s.c.) and may be carcinogenic for the uterus tissue. Although mirex has been shown to cause benign and malignant liver tumours, benign adrenal tumours and transitional-cell papillomas in the kidney, 41 it has not been tested in uterotrophic assay before. In our study, administration of mirex to immature female rats significantly reduced the uterine weight ratio. It also reduced gland and lumen percentages. However, mirex treatment increased epithelial volume fraction without significantly changing the total uterine volume. Our findings suggest that these OCPs may be the risk factors for epithelial neoplasm formation.
Stimulation of epithelial growth was accompanied by decrease in lumen ratio caused by PCBs. Mirex also reduced this histomorphometric parameter, although it had no significant effect on the uterine epithelial ratio. PCB 180, Aroclor 1221 and mirex-induced decreases in gland ratio, which may be due to general toxicity of these chemicals. However, reduction in gland ratio in response to organochlorinated substances remains to be further elucidated.
It has been reported that Aroclor 1221 and OCPs (methoxychlor and chlorpyrifos) stimulate gonadotropin-releasing hormone (GnRH) release in GT1-7 cells. 4 These effects were observed to be similar to those exerted by E2. In our study, the serum levels of gonadotropin were measured in immature (24 day old) female rats. The serum levels of LH did not significantly differ among the groups. The levels of FSH were selectively reduced by PCB 180 and endosulfan. Interestingly, we did not observe a significant change in LH as would be expected in view of the findings reported by Walker and Gore, 42 who reported that GnRH neurons are specifically sensitive to PCBs and pesticides. In the present study, mirex treatment had no significant effect on gonadotropin secretion in immature prepubertal female rats. Similarly, it has been reported previously that mirex had no adverse effect on GnRH-stimulated LH or FSH secretion in rat pituitary cell cultures. 43 The effect of endosulfan on gonadotropins has been controversial. Caride et al. 44 have shown that long-term (45 days) exposure to endosulfan resulted in elevated levels of LH in pubertal male rats. In contrast, Singh and Pandey 45 reported a reduction and Wade et al. 46 reported no change in the serum levels of gonadotropin in male rats. In our study, a short duration (3 days) of administration of endosulfan had no significant effect on serum LH in immature female rats. The present findings of PCB 180 and endosulfan causing a reduction in FSH secretion provide further evidence that persistent environmental toxicants may exert adverse neuroendocrine effects at the hypothalamic–pituitary axis level in addition to those on the uterine tissue.
In conclusion, the present findings suggest that PCB 180, Aroclor 1221 and endosulfan may be estrogenic substances in immature uterotrophic assay. Stimulation of uterine epithelial tissue was not reflected in the macroscopic findings (uterine weight ratio). This may be because of short duration of treatment in immature uterotrophic assay. Our findings confirm that PCB 180, Aroclor 1221 and endosulfan have endocrine disruptive effects at a dose of 10 mg/kg in immature female rats.
Footnotes
Conflict of Interest
The authors declared no conflicts of interest.
Funding
This study was supported by TUBITAK (project: 104-T-240).
