Abstract
The biocompatibility/inflammation profile of B2A-coated ceramic granules was evaluated using a panel of standard biocompatibility protocols (International Organization for Standardization-10993) including skin irritation and delayed-type hypersensitivity (Kligman maximization test), as well as acute, subacute, and chronic toxicity. Additionally, the potential of B2A-coated granules to elicit inflammatory reactions was also assessed using in vivo air pouch models, and B2A was evaluated using in vitro models of leukocyte recruitment and endothelial cell activation. Overall, the findings demonstrate that B2A-coated ceramic granules exhibit good biocompatibility profiles in the murine air pouch model and in standard subcutaneous implant models, and B2A did not demonstrate evidence of leukocyte recruitment or endothelial cell activation. These findings suggest that B2A and B2A-coated granules have little, if any, propensity to initiate inflammation reactions based on leukocyte recruitment. Thus, traditional biocompatibility and specially designed inflammation models indicate a high degree of biocompatibility and a low possibility of toxicity, inflammation, or edema following the implant of B2A-coated granules.
Introduction
B2A is a synthetic peptide that acts as a positive receptor modulator to augment osteodifferentiation, thereby promoting bone regeneration. 1 , 2 The B2A is not a recombinant growth factor, hormone, or morphogenetic protein, but its action is consistent with an allosteric activator. At the molecular and cellular level, B2A binds to cell surface receptors in the bone morphogenetic protein (BMP) receptor family, and, in the presence of BMP-2, will augment the differentiation of preosteoblasts and mesenchymal stem cells toward bone tissue. In the absence of BMP-2, B2A does not induce osteodifferentiation.
B2A has been integrated into an investigational, combination medical device (B2A-coated ceramic granules) intended for use in arthrodesis surgery, an orthopedic procedure that fuses the bones that form a joint, essentially replacing the joint with new bridging bone. Arthrodesis is widely used by the orthopedic community in patients whose afflicted joints (in the spine, the wrist, and the foot/ankle) are so severely damaged that usual pain management techniques fail.
The calcium phosphate-based ceramic component of the B2A-coated granules provides a scaffold for cell migration and growth. The B2A, which slowly elutes from the granules, is intended to augment the ongoing, naturally occurring differentiation of preosteoblasts in cooperation with endogenous BMP-2, typically upregulated during bone repair.
Nonclinical safety and effectiveness assessments including experimental arthrodesis studies in animals 3 , 4 supported the use of B2A-coated ceramic granules in pilot clinical studies targeting degenerative disc disease of the lumbar spine (Prefix), 5 and separately, end-stage foot and ankle arthritis (Amplex). 6 The clinical outcomes are supportive of continued investigation in larger, pivotal clinical trials, and no device-related adverse events were identified in the pilot trials.
Here, the biocompatibility/inflammation profile of B2A-coated ceramic granules was evaluated using a panel of standard biocompatibility protocols and using several models targeted to leukocyte recruitment. The concern of inflammation in new orthopedic products has recently been heightened by the reports of recombinant human BMP-2 (rhBMP-2)-induced inflammatory reactions in humans.7–10 In the present study, B2A was evaluated in a number of assays including standard biocompatibility assays and assays specifically designed to evaluate endothelial permeability, adhesion of monocytes to human endothelial cells, leukocyte transendothelial migration, and in vivo recruitment/edema using an air pouch model. 11 Additionally, the effects of B2A on the ligand for receptor activator of nuclear factor κB ligand (RANKL)-induced osteoclastogenesis were examined since RANKL has been established as a key mediator of inflammatory bone loss. The results are consistent with B2A biocompatibility and a low inflammatory profile.
Methods
Test Articles
B2A-coated granules were prepared from a 2-component kit immediately before use. The kit contains a vial of ceramic granules and a vial of lyophilized B2A.
The granules are composed of 20% hydroxyapatite and 80% β-tricalcium phosphate. The diameters of the granules are 1 to 2 mm, and the granule pore diameters are approximately 400 µm. B2A was formulated as a lyophilized powder using mannitol and glycine as excipients. Just prior to use, B2A was hydrated with water for injection (USP) and coated by charge–charge adsorption onto the ceramic granules.
B2A acetate (B2A) is a peptide synthesized by solid-phase peptide chemistry with an empirical formula of C241H418N66O65S2, and a relative molecular mass of 5344.4 (free peptide). B2A contains 42 naturally occurring amino acids and 3 residues of 6-aminohexanoic acid (Ahx), an analog of lysine. All 45 amino acids in B2A are of nonanimal origin. The sequence of B2A is
Ala-Ile-Ser-Met-Leu-Tyr-Leu-Asp-Glu-Asn-Glu-Lys-Val-Val-Leu-Lys-Lys(Ala-Ile-Ser-Met-Leu-Tyr-Leu-Asp-Glu-Asn-Glu-Lys-Val-Val-Leu-Lys)-Ahx-Ahx-Ahx-Arg-Lys-Arg-Leu-Asp-Arg-Ile-Ala-Arg-NH2
In the biocompatibility studies, B2A was coated on the granules at 3 mg/cm3, while in the air pouch model the coating concentration was 500 µg/cm3.
Biocompatibility Studies, ISO-10993
B2A-coated ceramic granules coated at 3 mg/cm3 were evaluated as a medical device (10993-3 2003; 10993-12 2002) for biocompatibility in a standard battery of studies (Toxikon Corporation, Bedford, Massachusetts). Extracts of B2A-coated ceramic granules were tested for allergenic potential or sensitizing capability in guinea pigs (Cavia porcellus) and for their potential to produce irritation after subcutaneous injection in New Zealand white rabbits (Oryctolagus cuniculus). Similarly, extracts were tested for potential toxic effects as a result of single dose and 14-day repeat-dose systemic administration in mice. For these assays, the B2A-coated ceramic granules were extracted in 0.9% NaCl or cottonseed oil at a ratio of 0.2 g/mL at 70°C for 24 hours.
B2A-coated ceramic granules were also evaluated for their potential to induce subchronic toxicity in rats following subcutaneous implant for 13 weeks. In these studies, approximately 0.2 g of the control article or B2A-coated ceramic granules was implanted in 2 sites per animal. The animals were evaluated for local and systemic indications of chronic toxicity including standard observations on body weight, hematology, clinical chemistry, coagulation, urinalysis, organ weight, and microscopic evaluation of implant sites and selected organs among others.
Air Pouch Model
In the murine air pouch model, air pouches were created in the dorsal subcutaneous tissue of BALB/c mice (20-22 g). Initially, 5 mL of filter-sterilized air was injected subcutaneously using a 25-gauge needle, and after 3 days the pouch was reexpanded with an additional 3 mL of sterile air. On day 6, the animals were implanted with either B2A-coated granules (0.5 mg/cm3) or sham-coated granules (vehicle control) when pouches were incised and test material was inserted into the pouch; additional animals were injected with 1 mL of saline (negative control) or 1% I-carrageenan (positive control). On day 7, the animals were euthanized and the pouches were carefully injected with 1 mL saline. The fluid was recovered and immediately put in a test tube on ice. The total white blood count was determined, and then cell smears were prepared and differential counts made following staining with Diff-Quik.
Endothelial Permeation
Human umbilical vein endothelial cells (HUVEC) were obtained commercially (Cascade Biologics) and were used at cell passages not greater than 5 from the original frozen stock. The HUVECs were evaluated using a commercially available kit (In vitro Vascular Permeability Assay 24 well, Millipore Corporation, Billerica, MA) following the manufacturer’s instructions. The HUVECs were cultured on collagen-coated, semipermeable membranes to form confluent monolayers. A high-molecular-weight fluorescent dye, fluorescein isothiocyanate (FITC)-dextran, was added on top of the cells in the presence of a test agent, and after 20 hours, the dye in the lower chamber that permeated through the endothelial cell layer was determined using a fluorescence plate reader (excitation 485 ± 20 nm, emission 528 ± 20 nm). Tumor necrosis factor-α (TNF-α; R&D Systems, Inc, Minneapolis, MN) was used as the positive control. Subsequently, the cells were rinsed, fixed, stained, and examined by bright-field microscopy.
Monocyte Attachment to Endothelial Cells
The HUVECs were cultured in tissue cultureware to form confluent monolayers. Human myeloid human promyelotic leukemia (HL60) cells, used as models of monocytes, were obtained commercially (American Type Culture Collection) and have a predominantly neutrophilic promyelocyte character. 12 The cells were loaded with a vital fluorescent dye (CyQUANT NF, Invitrogen, Life Technologies, Grand Island, NY) and were then added with or without test agent, allowed to attach, and, after rinsing, the attached monocytes were detected as per the instructions of the dye manufacturer. TNF-α was used as the positive control. 9
Transendothelial Migration
Transendothelial migration was evaluated using a commercially available kit following the directions of the manufacturer (QCM leukocyte transendothelial migration assay, Millipore Corporation). The HUVECs were grown to confluency on human fibronectin-coated cell culture inserts. The monolayers were rinsed twice with assay medium. The chemoattractant test compounds were added in serum-low medium to the lower chamber (B2A, 10 µg/mL; TNF-α, 100 ng/mL), and the plates were incubated in a tissue culture incubator for 20 hours. Aliquots of 2 × 105 HL60 cells, prelabeled with a vital fluorescent dye (CellTracker Green CMF DA, Millipore Corporation), were added to the HUVEC monolayers and the plate was further incubated at 37°C for 4 hours. Each group also contained a no-leukocyte-added control. After 4 hours, transmigrated cells were examined by fluorescence microscopy using a FITC filter set. Subsequently, cells in the lower chamber were collected and the cell number was evaluated (CyQUANT Cell Proliferation Assay Kit, Invitrogen) and read in a Synergy HT Multi-Detection Microplate Reader (480 nm and 535 nm excitation and emission, respectively).
Induction of Adhesion Molecules in HUVECs by Cellular ELISA
Induction of E-selectin in HUVECs was measured using a commercially available HUVEC/E-Selectin enzyme-linked immunosorbent assay (ELISA) kit (Astarte Biologics, Redmond, Washington). The HUVECs were seeded in 96-well plates at 15 000 cells/well overnight and then treated with 10 ng/mL TNF-α or 20 μg/mL B2A. After 6 hours the cells were fixed, and the expression of E-selectin was assayed according to the manufacturer’s instructions.
Osteoclast Differentiation and Osteoclast Activation
A commercially available kit (OsteoLyse Bone Resorption Assay Kit, Lonza, Walkersville, Maryland) was used to monitor differentiation of human osteoclast progenitor cells (OPCs; Lonza, Walkersville, Maryland) to osteoclasts, and the subsequent activity of osteoclasts. In these assays, the release of labeled collagen fragments into the osteoclast cell culture supernatant is assayed by time-resolved fluorescence. Briefly, OPCs are seeded in a specially prepared 96-well plate coated with collagen and differentiated in the presence of the growth factors macrophage colony-stimulating factor (M-CSF), soluble RANKL, and/or B2A and assayed following the manufacturer’s instructions. The osteoclasts begin to mature approximately 6 days after seeding and, at that time, acquired the ability to degrade the collagen on the surface of the plate. The release of collagen degradation fragments was measured in a time-resolved, fluorescence-capable fluorometer. Cultures in which the osteoclast progenitors are cultured in the presence of M-CSF but no soluble RANKL serve as undifferentiated controls.
To investigate the differentiation potential of B2A, the soluble RANKL is replaced by B2A at the beginning of the culture. To test whether B2A activates or inhibits the mature osteoclast function, the culture is treated with soluble RANKL and then the test sample is first added with the medium change on day 6.
Results
Biocompatibility Studies
B2A-coated ceramic granules were found to be biocompatible based on series of International Organization for Standardization (ISO)-10993 biocompatibility studies summarized in Table 1. The results of the genotoxicity/cytotoxicity components are reported elsewhere.
Summary of Biocompatibility Studies.
In the single-dose and 14-day repeat-dose systemic studies that evaluated extracts of the B2A-coated granules in mice, the biological response was not significantly greater than controls. The repeat-dose studies included hematologic and histopathologic assessments. Similarly, intracutaneous injection of extracts from B2A-coated granules did not show a significantly greater biological reaction than controls, and there was no erythema or edema.
In the rat chronic systemic toxicity evaluation, there was no mortality or clinical observations that suggested an effect of B2A-coated granules. Additionally, there were no biologically meaningful differences between animals receiving B2A-coated granules and controls in clinical pathology (clinical chemistry, hematology, coagulation studies), urinalysis, and body/organ weights. White blood cell counts and microscopic evaluation of the implant sites showed a mild foreign body response with infiltration around and extending into the test article material and was considered to be mildly reactive (bioreactivity rating = 1.7). However, the level of reactivity is consistent with the nature of the test material. There were no microscopic changes in any of the nonimplanted tissues to suggest an effect of the test article.
Air Pouch Model
The rodent air pouch has been useful model for the evaluation of the response to orthopedic biomaterials13–16 and here was used in follow-on studies to further evaluate inflammatory cellular infiltration.
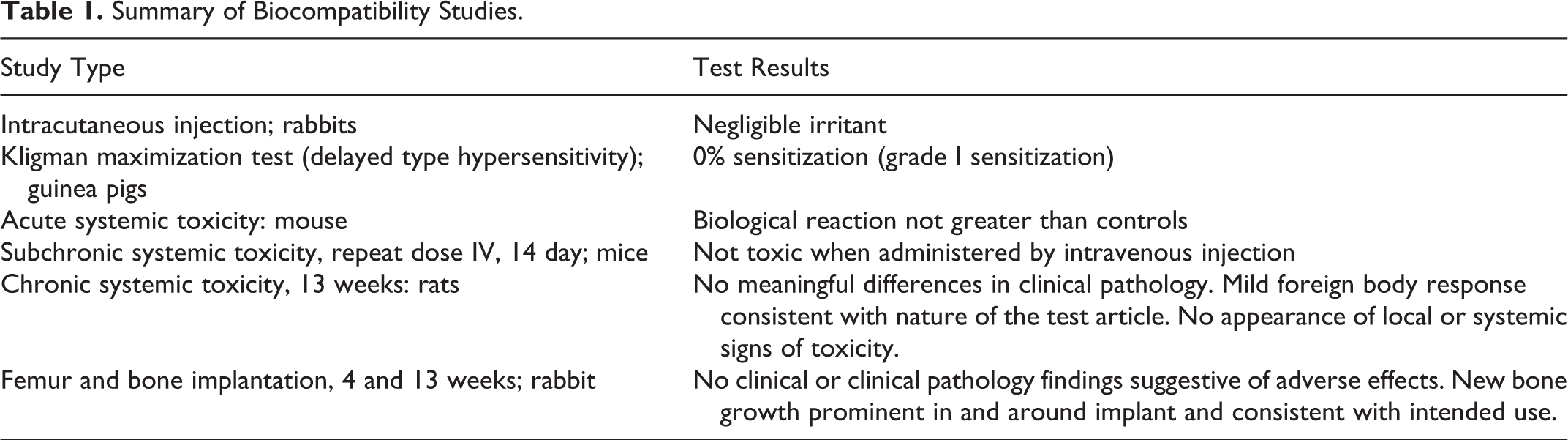
Following subcutaneous preparation of air pouches, 4 treatment groups were used: saline (system negative control), I-carrageenan (system positive control), granules (sham-coated granules, paired control), and B2A-coated granules.
The leukocyte number for each group is shown in Figure 1. There was no significant difference in leukocyte number between animals implanted with B2A-coated granules and sham-coated granules (P = 0.524, paired T test). The leukocyte number of the test-system negative control of saline and positive control of I-carrageenan were, however, significantly different (P ≤ 0.001). Along a similar vein, differential staining of the leukocytes in the exudate indicated that most of the cells from animals receiving saline, sham-coated granules, and B2A-coated granules were agranular leukocytes (lymphocytes and monocytes) averaging approximately 87%, 89%, and 92%, respectively; whereas in the I-carrageenan group, the proportion of agranular leukocytes was 34% and granular leukocytes were predominant (66%).
Concentration of leukocytes in murine air pouch exudates. There was no statistical difference between sham-coated granules and B2A-coated granules (750 μg/cm3 granules). Saline and I-carrageenan were used as negative and positive assay controls, respectively. Data are presented as the mean ± confidence interval (CI) n = 3. Asterisk indicates statistical significance compared to the saline control.
The cell number found in treatments with granules (with or without B2A coating) was somewhat higher than that found with saline (system negative control). It should be pointed out, however, that the granules are a bone void filler and have a rough surface, and therefore would be expected to be mildly abrasive when implanted subcutaneously (rather than in bone), a fact that could contribute to physical injury and mild inflammation.
Models of Human Endothelial Cells Interaction
Inflammation and edema could arise from direct effects on endothelial activation and, ultimately, endothelial permeability. For example, rhBMP-2 has been shown to induce the proinflammatory endothelial phenotype. 17 E-selectin, a leukocyte adhesion molecule expressed on activated endothelium, 18 is induced by TNF-α and other cytokines involved in the pathogenesis of inflammatory diseases including rheumatoid arthritis.
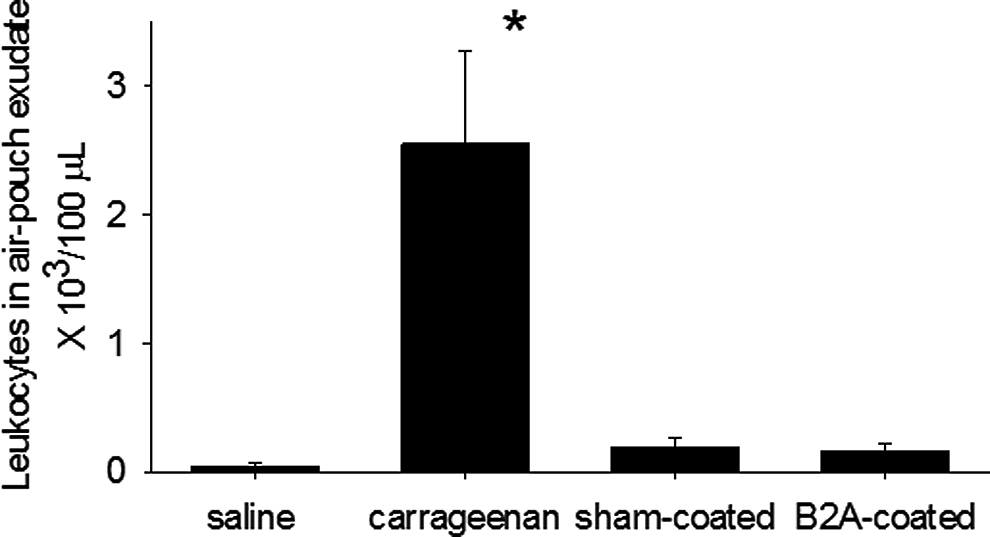
In the current studies, the potential for B2A to affect activation of the human endothelial cell, HUVEC, was evaluated by culturing cells in the presence of B2A at concentrations up to 10 µg/mL (Figure 2, right panel), and subsequently monitoring E-selectin, a marker of activated endothelium. 6 B2A did not induce expression of E-selectin, whereas the positive control compound TNF-α did. In corollary evaluations, B2A did not increase the proliferation of either HUVEC or bovine aorta endothelial cells or induce tube formation in an in vitro model of angiogenesis (data not presented).
Effect of B2A on human endothelial cells. B2A was used at 10 µg/mL. Tumor necrosis factor α (TNF-α) was used as a positive control at 100 ng/mL. The panel on the right illustrates that only TNF- α, the positive control, statistically increases the expression of E-selectin. Data are presented as the mean of the optical density at 405 nm ± SD. The panel on the left similarly illustrates that only TNF- α statistically increases fluorescent dye permeability. Data are presented as the mean of fluorescent units ± standard deviation (SD; n = 3).
A permeability analysis of HUVEC monolayers treated with B2A was also conducted. No impact on endothelial permeability was detected using B2A at either 5 or 10 µg/mL as measured with fluorescent dextran (Figure 2, left panel). On the other hand, treatment with TNF-α at 100 ng/mL did result in a statistically significant increase in endothelial cell permeability. Consistent with those results were microscopic observations of HUVEC cultures that revealed that B2A did not disrupt cell-to-cell contacts, whereas TNF-α treatment profoundly did so.
Models of Human Leukocyte–Endothelium Interaction
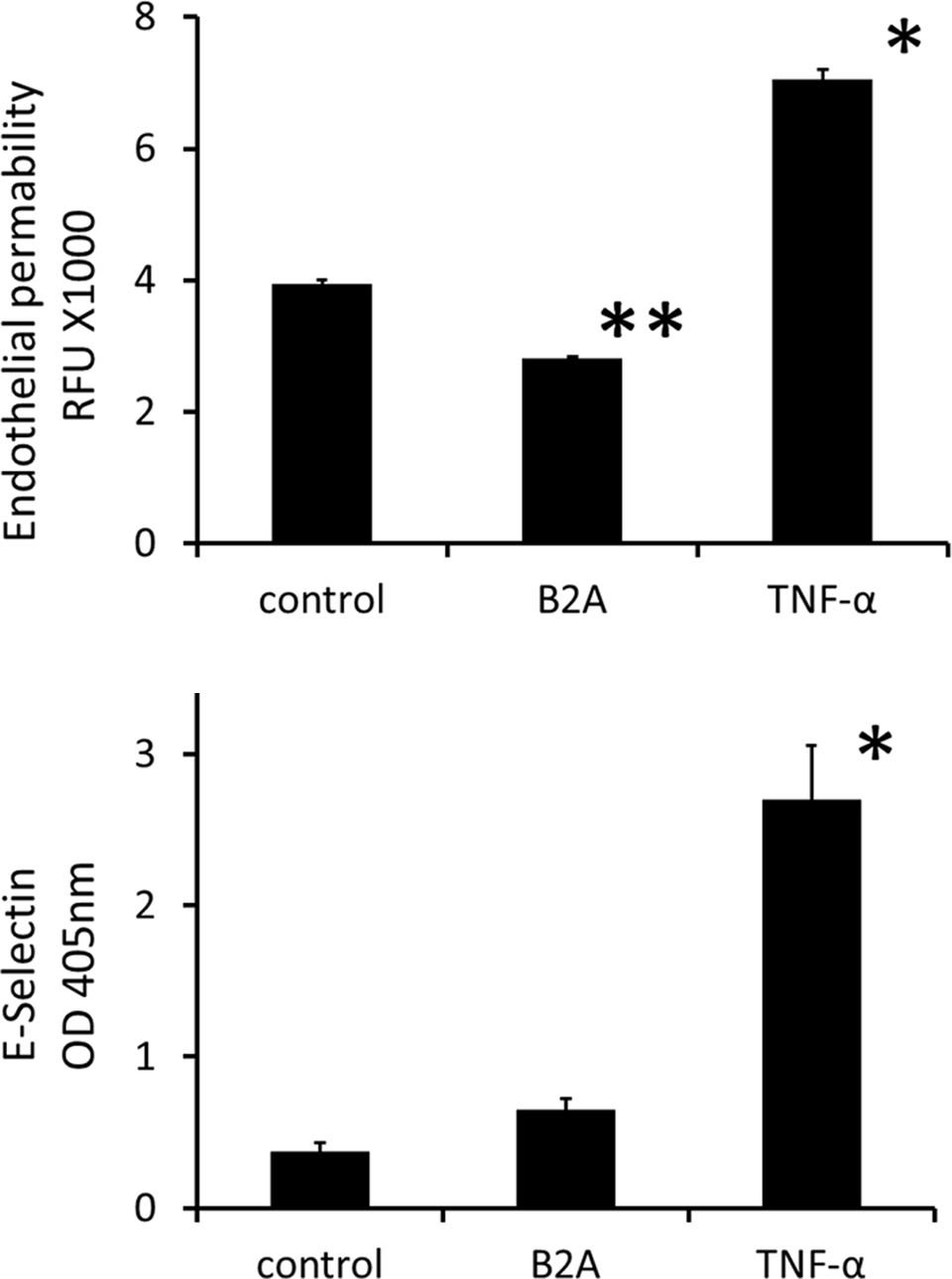
The potential effect of B2A on leukocyte attachment to endothelial cells and leukocyte transendothelial migration was evaluated using the human cell line HL60 and human endothelial cells, HUVEC. The HL60 cells are widely used to characterize leukocyte–endothelial interaction, including selectin-mediated adhesion. 22 In these evaluations, TNF-α but not B2A resulted in statistically significant increases in HL60 binding to HUVEC. B2A treatment (5 or 10 µg/mL) of cocultures of HL60 and HUEVC resulted in essentially no increase in HL60 cell attachment compared to control (Figure 3, left panel). The TNF-α (100 ng/mL) did, however, result in a statistically significant (P = 0.0001) increase. In evaluations of leukocyte transendothelial migration, B2A treatment did not increase the number of migrating leukocytes (Figure 3, right panel). On the other hand, TNF-α treated HUVEC clearly displayed significantly enhanced transmigration over the control value (P < 0.0001).
Human leukocyte–endothelium interaction is not influenced by B2A. Panel on the left illustrates the adhesion of monocytes (human promyelotic leukemia [HL60] cells) to human umbilical vein endothelial cells (HUVECs) in the presence of 10 µg/mL B2A or 0.1 µg/mL TNF-α as determined using fluorochrome-loaded cells. Panel on the right illustrates the transmigration of monocytes (HL60 cells) through monolayers of HUVECs in the presence of 10 µg/mL B2A or 0.1 µg/mL TNF-α as determined using fluorochrome-loaded cells. Data are presented as the mean of relative fluorescent units (RFUs) ± standard deviation (SD). Asterisks indicate statistical significance relative to controls (P < .05), n = 3.
Osteoclasts and RANKL
In the current studies, the potential for B2A to affect the RANKL pathway was evaluated using human osteoclast precursor cells (OCPs). Treatment of OCPs with soluble RANKL in the presence of M-CSF induces differentiation into osteoclasts, and that differentiation can be monitored by determining the amount of collagen released to the culture medium (Figure 4, left panel). When treated with B2A, the OCPs did not release collagen and the OCPs remained undifferentiated. This indicated that B2A was not able to induce osteoclast differentiation or substitute for RANKL.
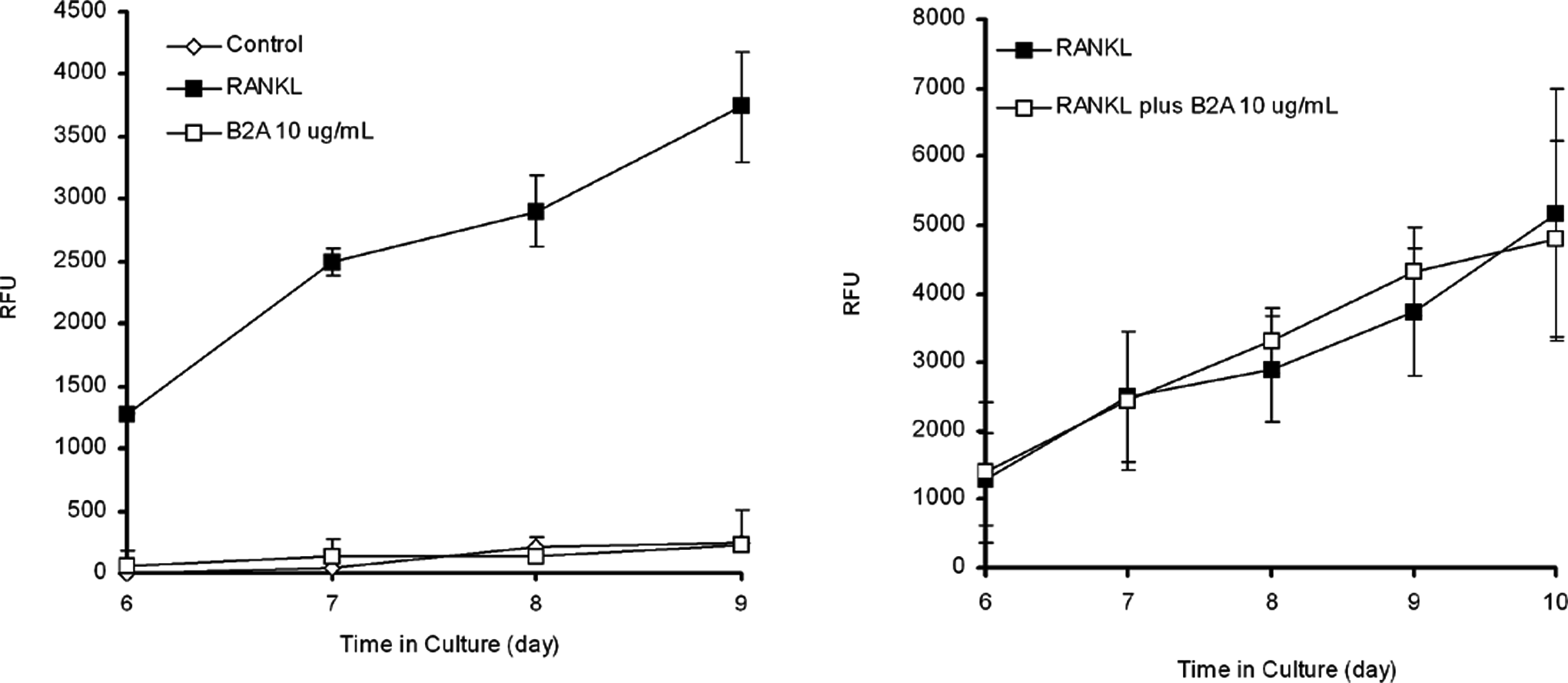
B2A does not affect osteoclast precursor cells (OCPs) osteoclast differentiation. Panel on the left illustrates that B2A does not induce osteoclast differentiation. Primary human osteoclast precursors were seeded onto an OsteoAssay plate at 10 000 cells/well and cultured in medium containing macrophage colony-stimulating factor (M-CSF) and soluble receptor activator of nuclear factor κB (RANK) ligand or B2A. After 6 days of culture, samples of medium at indicated time points were removed and counted for collagen degradation. (Data were presented as mean ± standard deviation [SD], n = 4). Panel on the right illustrates that B2A does not change the activity of osteoclasts as measured by collagen degradation. Primary human osteoclast precursors were seeded onto an OsteoAssay plate at 10 000 cells/well and cultured in medium containing M-CSF and soluble RANK ligand for 6 days. After 6 days of culture, the culture medium was changed to M-CSF and soluble RANK ligand with or without B2A. Samples of medium at indicated time points were removed and counted for collagen degradation. (Data were presented as mean ± standard deviation [SD], n = 4).
To investigate whether B2A could upregulate the resorptive activity of human osteoclasts, OCPs were stimulated by M-CSF and soluble RANKL for 6 days to generate osteoclasts. Thereafter, B2A was added to the medium, and medium samples were subsequently tested for collagen degradation (day 6-day 10). Figure 4, right panel, illustrates that treatment of osteoclasts with B2A did not affect collagen degradation, and B2A did not upregulate the resorptive activity of osteoclasts.
Discussion
In the present study, B2A and B2A-coated ceramic granules were evaluated with a number of assays including standard biocompatibility assays and assays specifically designed to evaluate endothelial permeability, adhesion of monocytes to human endothelial cells, leukocyte transendothelial migration, and in vivo recruitment/edema using an air pouch model. 11 The results suggest that B2A would be biocompatible and have an acceptable inflammatory profile in clinical use as a bone-graft material.
In arthrodesis surgery, cartilage in a joint is typically removed and replaced with bone graft material. Historically, morselized bone graft harvested from the iliac crest has been used as graft material as it provides a combination of osteoconduction (bone growth on a substrate), osteoinduction (stimulation of stem cells to become active osteoblasts), and living bone. However, due to pain and morbidity associated with the graft harvest, 19 there has been a shift away from its use in recent years and toward the introduction of synthetic bone grafts as alternatives, particularly those active agents to encourage differentiation or cell growth.
Both rhBMP-2 and rh PDGF-BB are active agents that have been incorporated in synthetic bone grafts. Recombinant human BMP-2 is approved for use in certain spinal fusion settings and rh-PDGF-BB/synthetic bone graft has been used in a randomized control trial to support a marketing application in the United States. The rhBMP-2 that is used solely in orthopedics has been associated with a number of adverse events resulting from inflammation/edema and osteolysis among others,7–10 , 20–22 many occurring in off-label uses. Indeed, the fact that BMP-2 induces inflammation is thought to be indisputable. 23 On the other hand, rh PDGF-BB, marketed in the United States for treatment of diabetic foot ulcers, does not appear to have adverse events stemming from inflammation; however, it can be associated with a potential risk of malignancy in certain applications. 24
Principally, in light of the clinical experience with rhBMP-2, a series of additional studies were used to evaluate the inflammatory/edema potential of B2A-coated ceramic granules in addition to the standard biocompatibility assays.
One of these additional studies involved the use of the rodent air pouch model of acute local inflammation. That model has been used in the evaluation of a number of drugs 25 , 26 and orthopedic biomaterials13–16 and here was applied to B2A-coated granules.
Mechanistically, inflammation and edema could arise from direct effects on endothelial activation and, ultimately, endothelial permeability. Endothelial activation refers to specific changes in endothelial phenotype that are pivotal in inflammation including an increase in endothelial–leukocyte interactions and permeability. During endothelial activation by TNF-α, for example, E-selectin, a leukocyte adhesion molecule, is expressed and therefore E-selectin has been used as a marker of activated endothelium. 27 However, B2A did not appear to induce endothelial activation or E-selectin expression.
E-selectin recognizes and binds to ligands on the surface of leukocytes associated with acute and chronic inflammatory such as neutrophils, monocytes, and eosinophils among others. Typically, for most inflammatory responses, neutrophils and then mononuclear cells must attach to and then pass through the endothelial barrier to access sites of inflammation.
Substances eluting from implanted materials could differentially activate leukocytes to increase their endothelial attachment or impact both leukocytes and endothelium. For example, rhBMP-2 reportedly elicits endothelial dysfunction and increases monocyte attachment. 17
Here, the potential effect of B2A on leukocyte attachment to endothelial cells and leukocyte transendothelial migration was evaluated using the human cell line HL60 and human endothelial cells, HUVEC. The HL60 cells are widely used to characterize leukocyte–endothelial interaction, including E-selectin-mediated adhesion and transmigration. 28 B2A did not increase HL60 binding to HUVEC. Similarly, in evaluations of leukocyte transendothelial migration, B2A treatment did not increase the number of migrating leukocytes. Additionally, B2A-coated granules did not increase the inflammatory cellular infiltration compared to uncoated granules in a rodent air pouch model.
If inflammatory cells in bone produce proinflammatory cytokines such as interleukin 1 (IL-1) and TNF-α osteoclastic osteolysis can arise as an outcome of the stimulation of RANKL production. 29 The RANKL is a potent osteoclastogenic factor, and both TNF-α and RANKL are abundant in sites of inflammatory bone erosions. During osteolysis, proinflammatory cytokines such as IL-1 and TNF-α (originating from cells of the immune system) induce bone resorption via the induction of RANKL on osteoblasts. RANKL in turn stimulates osteoclast maturation and potentially unbalanced bone erosion. In this study, B2A did not upregulate the resorptive activity of osteoclasts and B2A was not able to induce osteoclast differentiation or substitute for RANKL.
In conclusion, the results of this study were found to be consistent with a good biocompatibility profile for B2A-coated ceramic granules. The in vivo air pouch model demonstrated B2A-coated ceramic granules had little propensity to initiate inflammation and were supported by additional in vitro studies that followed the inflammation cascade.
Footnotes
Acknowledgments
Editorial support was provided by M. C. Schneider. The work presented here was done in part in association with Brent Atkinson, PhD, formerly of BioSurface Engineering Technologies, Inc.
Authors’ Note
The authors Zamora, Liu, Guo, and Lin were employees of BioSurface Engineering Technologies, Inc. at the time the studies were conducted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclose receipt of the following financial support for the research, authorship, and/or publication of this article: funded by BioSurface Engineering Technologies, Inc.
