Abstract
Gold nanoshells (155 nm in diameter with a coating of polyethylene glycol 5000) were evaluated for preclinical biocompatibility, toxicity, and biodistribution as part of a program to develop an injectable device for use in the photothermal ablation of tumors. The evaluation started with a complete good laboratory practice (GLP) compliant International Organization for Standardization (ISO)-10993 biocompatibility program, including cytotoxicity, pyrogenicity (US Pharmacopeia [USP] method in the rabbit), genotoxicity (bacterial mutagenicity, chromosomal aberration assay in Chinese hamster ovary cells, and in vivo mouse micronucleus), in vitro hemolysis, intracutaneous reactivity in the rabbit, sensitization (in the guinea pig maximization assay), and USP/ISO acute systemic toxicity in the mouse. There was no indication of toxicity in any of the studies. Subsequently, nanoshells were evaluated in vivo by intravenous (iv) infusion using a trehalose/water solution in a series of studies in mice, Sprague-Dawley rats, and Beagle dogs to assess toxicity for time durations of up to 404 days. Over the course of 14 GLP studies, the gold nanoshells were well tolerated and, when injected iv, no toxicities or bioincompatibilities were identified.
Introduction
Gold nanoparticle materials are currently being investigated as promising agents for medical imaging and therapeutics. Overseas trials have been conducted with informed consent and regulatory approval in 4 patients with head and neck tumor at 4.5 mL/kg of 100 optical density (OD) solution (21 mg/kg nanoshells infused). Subsequent patients (4 head and neck, 19 prostate) have been dosed at 7.5 mL/kg of 100 OD solution (35 mg/kg nanoshells infused). No adverse effects related to device exposure have been seen. The unique physical and electromagnetic properties of gold nanoparticles, including ease of functionalization, control of size and shape, optical resonance, and relative inertness of gold in the body, allow for a multitude of potential applications ranging from diagnostic imaging, 1 –4 drug delivery vectors, 4 –10 and exogenous absorbers for thermal ablation of tumors. 11 –19 One of the most promising gold nanoparticle for medical applications, gold nanoshells, is currently being used in clinical investigations of the ablation of solid tumors in head and neck and prostate cancers. Gold nanoshells are solid spherical particles, approximately 155 nm in diameter, with a layer of 5000 molecular weight polyethylene glycol (PEG) covalently bound to the surface to increase circulation time in the blood. 11,14,20 The spherical particle has an approximately 120 nm core of silica upon which a thin gold shell has been deposited. This dielectric core/metal shell structure determines the optical extinction properties of the particle, which is principally at near infrared (NIR) wavelengths. 20,21 The spheres are metabolically inert and thus evaluated as medical devices.
Traditional energy-based tissue ablation techniques (high-powered lasers, radiofrequency, microwave, focused ultrasound) rely upon the transduction of the energy source by the natural components of human tissue, and there is little discrimination between normal and tumored tissue. Accordingly, tumor ablation is dependent upon the placement of the energy applicator. In contrast, nanoparticle-based ablation attempts to use an exogenous material, the nanoparticle, to transduce the energy and direct the ablation. In these nanoparticle-based therapies, the objective is to provide a more specific ablation of the tumor and spare normal tissue. These objectives can be achieved only when the energy source is nonablative and the nanoparticles accumulate in the tumor and not in normal tissue.
The current clinical usage of the nanoshell particles in head and neck and prostate cancers involves a single infusion of the particles directly into the blood stream, followed by application of laser energy 12 to 24 hours postinfusion. The particles have a circulating half-life in the bloodstream of 3 to 6 hours. The nanoshell particles will accumulate in solid tumors through their leaky vasculature, a process known as the “enhanced permeability and retention” (EPR) effect. 22 –24 Previous preclinical efficacy studies have shown that after 12 to 24 hours the particles either accumulated in the tumor or were cleared from the blood stream by the reticuloendothelial system (RES), principally the liver and spleen. These studies also indicated a long-term retention of the particles in the macrophage cells in the liver and spleen, with little evidence of clearance. No evidence of toxicity was observed in these studies. 11,14,25
Following nanoshell accumulation in the tumor, thermal ablation is achieved by illumination of the tumor using NIR laser energy delivery to the tumor site via fiber optics. Due to the large extinction coefficient in the NIR of the nanoshell particles, the incident NIR laser energy is transduced by the particles into heat. By adjusting the power of the incident laser it is possible to control the amount of resultant heat from mild hyperthermia to ablative temperatures. In order to protect the healthy tissues, laser powers are chosen such that the laser energy delivered is insufficient to reach ablative temperatures unless nanoshells are present in the tumor. Through this mechanism, it is the location of the nanoshell particles and not the laser applicator, which is defining the areas of ablation.
The intravenous (iv) delivery of the particles exposes substantially all physiological systems to any particle-associated toxicity. As an initial step, biodistribution studies are essential to identify target organs and tissues, and then acute and chronic toxicity studies are necessary to understand the toxicity profile prior to human use. Prior investigations of infused gold particles of varying sizes have identified the liver, spleen, and kidney as potential target organs, 25 –27 primarily for accumulation. Additionally, the lungs and cardiovascular system may also be exposed to the particles.
As the therapeutic mechanism of action is physical and not biological or chemical, the nanoshell particles are regulated as a medical device. In order to evaluate the toxicity of gold nanoshells, we conducted a series of studies based on the International Organization for Standardization (ISO)-10993 guidance (Biological Evaluation of Medical Devices). In addition to the relevant toxicity tests indicated in the ISO-10993 standard, we performed a series of in vivo studies including a biodistribution/clearance study in mice and acute toxicity in rats. In order to get toxicity information from an additional species, acute and chronic toxicity studies were carried out in Beagle dogs. The studies described herein provide, to the best of our knowledge the most extensive description of the biological fate and safety of gold nanoparticles.
Materials and Methods
Manufacturing and Characterization of the Gold Nanoparticles
Nanoshells were fabricated under clean conditions in a class 1000 clean room. Nanoshell fabrication was based on the method of Oldenburg et al. 20 Briefly, gold colloids 1 to 3 nm in diameter were prepared using the method of Duff and Baiker. 28 Aminated spherical silica particles 120 ± 12 nm in diameter (Precision Colloids, LLC, Cartersville, CA) were exposed to the gold colloid solution forming gold colloid nucleation sites on the silica core. The gold seed sites were then further reacted with hydrogen tetrachloroaurate (HAuCl4) in the presence of formaldehyde. This caused the surface colloid to grow and coalesce, ultimately forming a complete metal shell. Finished particles possessed a 12- to 15-nm-thick shell that resulted in an optical absorption peak between 780 and 800 nm. Thiolated PEG (SH-PEG; Laysan Bio, Arab, AL) was then assembled onto nanoshell surfaces by combining 1 mmol/L SH-PEG and nanoshells in a ratio of 1.5 µL/mL in deionized H2O for 12 hours followed by diafiltration to remove the excess SH-PEG. Particles were then suspended in 10% trehalose solution to create an isoosmotic solution for injection and concentrated by tangential flow filtration to an extinction of 100 ± 5 OD (at 800 nm) to reduce the infused fluid volume. The nanoshell solution was passed through a 0.45-nm filter and filled into iv bags. For the rat and canine in vivo studies, the nanoshell solution was terminally sterilized by e-beam irradiation. The concentration of nanoshells in the 100 OD solution was measured using neutron activation analysis (NAA) to assess the amount of gold in the solution. Particle size and size distribution were measured using transmission electron microscopy (TEM) and dynamic light scattering (DLS) using a Malvern ZetaSizer (Malvern Instruments Ltd., Worcestershire, UK). The presence of bacterial endotoxin was determined by Pyrogent 80 gel clot assay (0.0625 EU/mL sensitivity; Lonza, Allendale, NJ). Osmolarity of the final solution was measured using a Vapro vapor pressure osmometer (Wescor, Logan, UT).
Neutron Activation Analysis
Neutron activation analysis of gold in tissue, blood, and in solution was carried out at Texas A&M University’s Nuclear Science Center 1 MW Triga research reactor. The irradiation position used in this study has an average neutron flux of approximately 1×1013 n cm−2 s−1. Gamma ray spectroscopy was carried out on all irradiated materials after a delay of 4 to 8 days to allow for decay of matrix activity, primarily 24Na. High-purity germanium detectors with nominal resolutions (full width at half maximum) of 1.74 keV or better and efficiencies of 25% to 47% by industry standard relative measurement were used to quantify the 412 keV gamma-line from 198Au. The Canberra Industries’ OpenVMS alpha processor-based Genie-ESP software was used for acquisition and computation of gold concentrations.
Preliminary Toxicity Studies
Prior to performing acute and chronic toxicity studies, laboratory and preclinical studies were conducted in accordance with the ISO-10993 guidelines. 29 The following tests were conducted: bacterial mutagenicity test—Ames assay (10993-3:2003), in vitro chromosome aberration analysis in Chinese hamster ovary cells (10993-3:2003), in vivo mouse micronucleus assay (10993-3:2003), ISO minimal essential medium elution using L-929 mouse fibroblast cells (10993-5:1999), ISO intracutaneous reactivity test (10993-10:2002), guinea pig maximization sensitization test method for liquid test articles (10993-10:2002), ISO acute systemic injection test (10993-11:2006), and subacute (28 day) iv toxicity in rats(ISO-10993-11:2006). In addition to the ISO toxicity tests listed above, the following tests were also conducted: materials-mediated rabbit pyrogen test (USP 29 2006) and American Society for Testing and Materials (ASTM) hemolysis assay direct contact (ASTM F-756-00). Details of the experimental methods for these studies are described in the Supplementary Material.
Biodistribution and Chronic Toxicity in a Balb/C Mouse
To determine the fate of the gold nanoshells after infusion and to identify target organs as well as tissue-specific toxicity, a nongood laboratory practice single-dose study was performed in mice. Female Balb/c mice (6-7 week, 18 g, Harlan) received a single tail vein injection of 2.34 mg/kg of gold nanoshells (6.18-6.82 mL/kg dose volume). Mice were randomly divided into 3 separate groups for analysis of (i) histopathology; (ii) gold biodistribution among organs by NAA; and (iii) the total mass of gold retained by the animal, also by NAA. Animals were euthanized at 1, 7, 28, 56, 182, and 404 days after particle injection for histopathology, at 1, 7, and 28 days for individual organ gold content analysis, and at 1, 7, 28, 56, 182, and 404 days for gold mass balance analysis (n = 4 for each group and each time point except where indicated). No separate control group was maintained.
Neutron activation analysis was utilized to measure the presence of gold in tissue. Gold represents approximately 95% of the mass of the nanoshell particle, and its presence is indicative of particle accumulation or retention. 25 Organs or tissue were harvested at the specified time points after particle injection, weighed immediately after harvest, and desiccated for evaluation. Radioactivity of the sample was measured and compared to gold standards irradiated at the same time to allow for the determination of total gold content in each sample, which was used to determine the relative gold content (in parts per million of wet weight of the tissue) or total gold present (for total gold retention and excretion analysis).
Histopathology
Mice from the group for histopathology were sacrificed and the major organs and tissues were harvested for histopathology.
Single-Dose Systemic Toxicity in the Beagle Dog Followed by 14 and 28 Days of Observation
The acute systemic toxicity of a single-dose iv infusion of gold nanoshells was evaluated in Beagle dogs. Female Beagle dogs (24 animals ranging from 5.1 to 9.5 kg) were randomly assigned to treatment groups of 8 animals each. The test groups received a single infusion of either 35 mg/kg of gold nanoshells (7.5 mL/kg dose volume) or 70 mg/kg of gold nanoshells (15 mL/kg dose volume), representing the expected clinical dose and double the expected clinical dose, respectively. The control group received an infusion of the vehicle control (7.5 mL/kg of 10% trehalose solution). All infusions were performed at a rate of 2 mL/min. In each group, 4 animals were euthanized on day 14 and the remaining 4 on day 28.
Toxicity assessments included clinical observations; clinical chemistry, hematology, and urinalysis tests; and histopathology of major organs. Blood was collected at baseline, day 2 after dosing, and weekly thereafter. Urine was collected at baseline and at sacrifice. Weight and food consumption were monitored during the study. Animals were sacrificed at either 14 or 28 days postinfusion, necropsies were performed and organs were weighed and prepared for histopathological analysis. Based on the results from the biodistribution study in mice, the following tissues were examined for signs of toxicity from all groups: heart, lungs, liver, spleen, mesenteric lymph nodes; any additional abnormal tissues noted at necropsy were also taken for pathological analysis.
Ten-Month Chronic Systemic Toxicity in the Beagle Dog
The chronic systemic toxicity of infused gold nanoshells was evaluated in the Beagle dog. Male dogs (12 animals ranging from 7.4 to 9.3 kg, age unspecified) were randomly assigned to 2 treatment groups and a control group (4 animals each). All groups received 2 infusions, the first on day 1 and the second on day 8 of the study. The test groups received either a total of 70 mg/kg (35 mg/kg/dose; 7.5 mL/kg/dose) of gold nanoshells (2 infusions of expected clinical dose) or 140 mg/kg (70 mg/kg/dose; 15 mL/kg/dose) of gold nanoshells (2 infusions of twice the expected clinical dose). The control group received vehicle control (10% trehalose solution). All infusions were performed at a rate of 2 mL/min. All animals were euthanized on day 300 of the study.
Toxicity assessments included clinical observations and weights; electrocardiograph (ECG) measurements, ophthalmology examinations, clinical chemistry, including stress hormones (cortical and adrenocorticotropic hormone), hematology and urinalysis tests; and histopathology of major organs. Blood and urine samples were collected at baseline and on days 2, 10, 30, 60, 90, 120, 180, and 300. The ECGs were performed at baseline and on days 10, 30, 180, and 300. Physical and ophthalmologic examinations were performed at baseline and on day 300. On day 300, the animals were sacrificed and necropsies performed. Selected organs were weighed and all major tissues were collected for histopathological analysis including adrenal gland, aorta, bone with bone marrow, brain, kidney, liver, lung, lymph node, pancreas, prostate, skeletal muscle, spleen, and testis.
Results
Particle Characterization
Figure 1 shows representative TEM analysis for particles prepared using these methods, courtesy of the nanoparticle characterization laboratory of the National Cancer Institute. The TEM analysis (>2000 particles measured) shows that the gold–silica nanoshells are very spherical in shape and have an average particle diameter of 156 ± 15 nm. A representative transmission electron microscope image of the gold nanoshells and the size distribution as measured by TEM is shown in Figure 1.
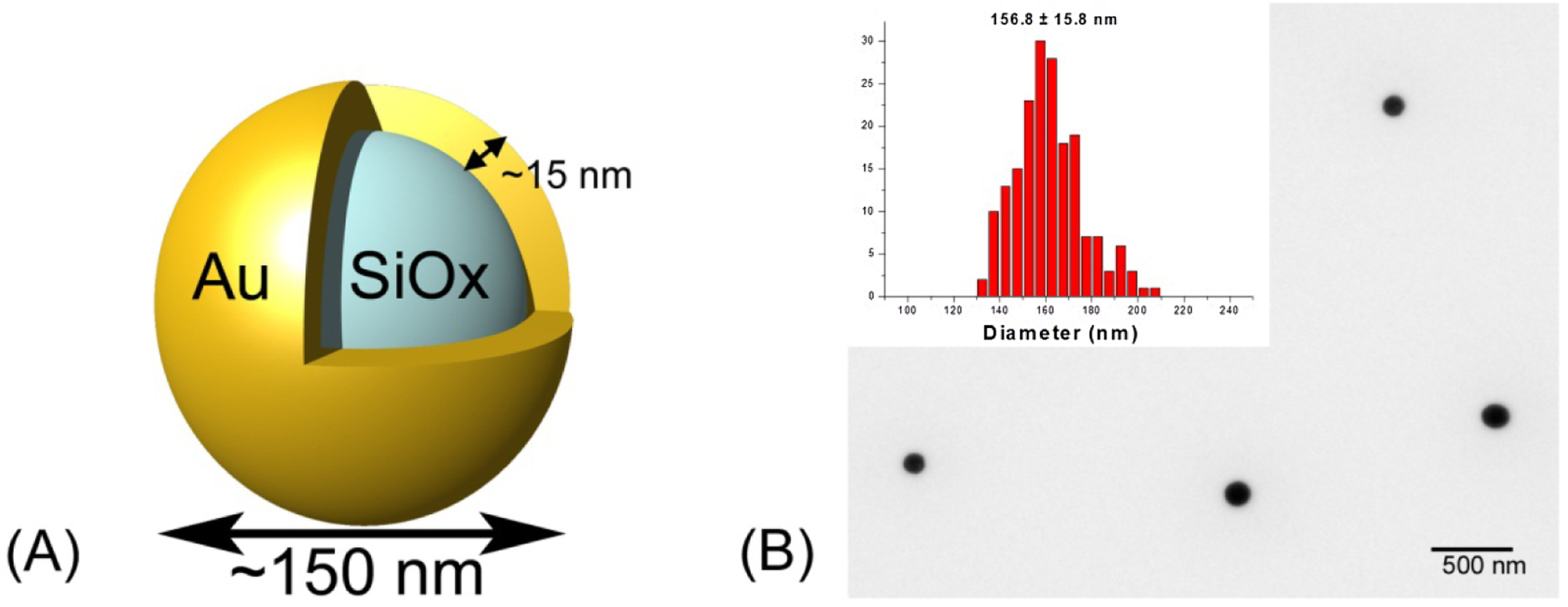
A, Illustration of the nanoshell particle geometry. B, Representative TEM of PEGylated silica-gold nanoshells and particle size distribution as measured by TEM (inset). PEG indicates polyethylene glycol; TEM, transmission electron microscopy.
As the PEG coating of the particles cannot be readily measured by TEM, DLS analysis was used as an indirect measurement of the PEGylation of the particle. Prior to the addition of PEG to the nanoparticle surface, the hydrodynamic radii of the particles measured by DLS was 160.8 ± 1.7 nm. Post-PEGylation, the hydrodynamic radius was measured to be 178.7 ± 1.7 nm. This increase in particle diameter is attributed to the presence of the PEG coating on the nanoparticle surface. Nanoparticle concentration in solution was determined by NAA performed on 100 µL of a nanoshell solution which had an OD of 100 measured at 800 nm. The gold concentration of this solution was determined to be 42.731 μg. The particle concentration (in particles/mL) was calculated from this number to be ∼2.7 × 109 particles/mL (4.67 mg nanoshells/mL). The osmolarity of the solution was measured using a WesCor vapor pressure osmometer and determined to be 278 ± 3 mOsm/L, which compared well with the reference range for blood (275-299 mOsm/L). The nanoshell solution tested negative for bacterial endotoxin using a gel clot assay (Lonza) with a sensitivity of 0.0625 EU/mL.
Summary of ISO In Vitro and In Vivo Toxicity Screen
Prior to the initiation of any in vivo testing, a comprehensive panel of toxicity tests, based on the ISO standard for the biological evaluation of medical devices (ISO-10993), was performed to determine whether the nanoshell particles or the 10% trehalose solution demonstrated any toxic effects in vitro. The ISO testing requirements provide standardized testing methods to evaluate the baseline toxicity of a medical device, including genotoxicity, cytotoxicity, hemocompatibility, tissue tolerance, and pyrogenicity. The results of these tests performed on the nanoshell particle solutions are summarized in Table 1. In all tests listed above, there was no identification of any toxicity for the nanoshell particles. This included the acute systemic toxicity study where the mice received massive doses (∼10× the predicted effective therapeutic dose) of the nanoshell particle solution.
Summary of ISO Toxicity Testing.
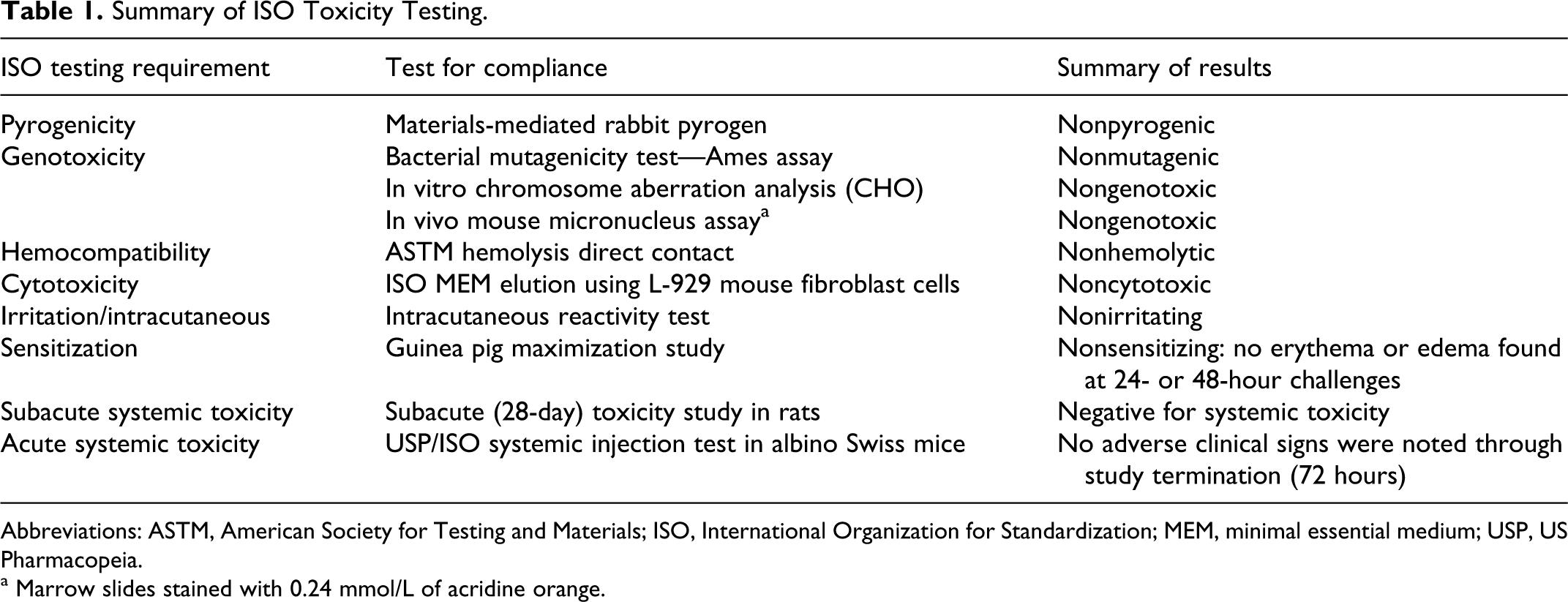
Abbreviations: ASTM, American Society for Testing and Materials; ISO, International Organization for Standardization; MEM, minimal essential medium; USP, US Pharmacopeia.
a Marrow slides stained with 0.24 mmol/L of acridine orange.
Biodistribution and Chronic Toxicity in a Balb/C Mouse
In order to determine the fate of the nanoshells in the body postinfusion, healthy Balb/C mice were administered an infusion of gold nanoshells. The mice were split into 3 groups in order to evaluate the organ biodistribution, the mass balance of gold in the body over time, and histopathological analysis of the major organ systems. The biodistribution and mass balance of nanoshells in the body was determined by measuring gold content in various tissue samples harvested at specific time points postinfusion.
The results of the 1-, 7-, and 28-day time points for organs with significant gold content, with gold content reported in micrograms are shown in Figure 2. It is evident from these data that particles accumulate primarily in the organs of the RES as they are cleared from the bloodstream. High concentrations of particles are seen in the liver and spleen, which account for >85% of the total measured gold. The lack of significant particle accumulation in other tissues would indicate that the gold nanoshells are not able to extravasate into healthy tissue and are cleared by the liver and spleen.
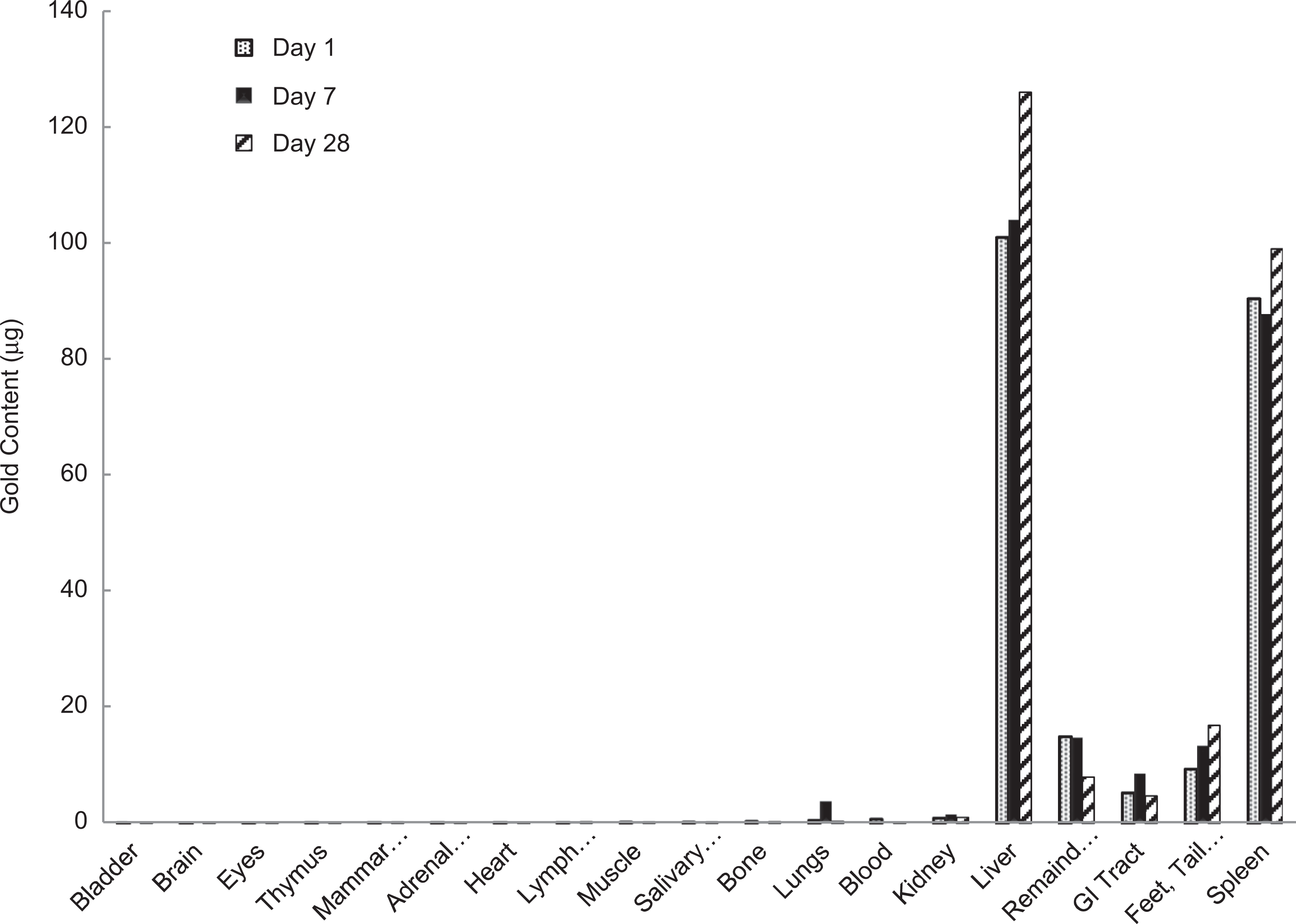
Gold content (µg) in major organ systems after intravenous infusion of nanoshells in Balb/c mice.
The mice in the mass balance group were euthanized, their entire body homogenized and weighed, and representative samples of the total body were analyzed by NAA to measure total gold content in the animal over time. The NAA results are summarized in Figure 3. As indicated, the total gold measured at each time point was approximately the same as the injected quantity, indicating substantial retention of the injected particles by the animals. Small measurement errors in the injected volume of the particle solution may account for the differences. From the mass balance data, it is apparent that even at >1 year postinfusion, the amount of gold in the body has not decreased significantly. This indicates that the body is unable to break down and excrete the gold nanoshells. From these results it is evident that the gold nanoshells persist in the body, primarily in the liver and spleen for extended periods of time.
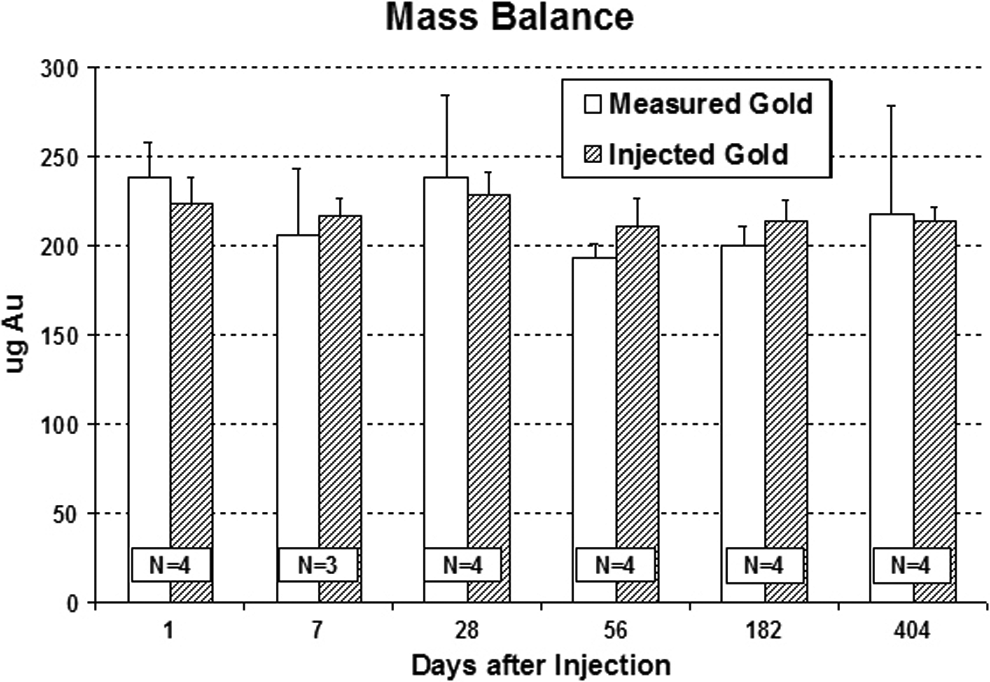
Neutron activation analysis (NAA) results for gold content of mice receiving a single injection of gold nanoshell particles and sacrificed at the time points indicated. Amounts reported as total gold content present in animal at sacrifice date (measured gold) compared to gold injected (injected gold) at the beginning of the experiment.
The NAA analysis was corroborated by histopathology, where the finding of a small, grainy pigment was noted in occasional regional macrophages. Histopathology was negative for other significant lesions including no evidence of histiocytosis. Based on this study, the target organs after iv infusion or injection were considered the liver and spleen, with a potential concern also for kidney and adrenal gland accumulation.
Single-Dose Systemic Toxicity in the Beagle Dog Followed by 14 and 28 Days of Observation
A 28-day acute systemic toxicity of a single dose in the beagle dog study was performed to determine whether there was any change in the safety profile in a different species. The dog is one of the species on which human hazard can be based. All animals tolerated the infusion well, and there were no signs of infusion-related reactions (including hypersensitivity) during and after infusion. No findings related to the test or control articles were noted during physical examinations. Postinfusion behavioral monitoring was also unremarkable.
There were occasional statistically significant differences (P < .10) among groups regarding serum chemistry and hematology values, but none were considered toxicologically significant and there was no dose-related trend. No dose-related effects were noted in urinalysis.
Histopathology revealed the presence of “black pigment” within phagocytic cells of the liver and spleen. This observation is consistent with the previous mouse study and other preclinical studies and is believed to represent accumulations of gold nanoshells cleared from the blood.
There was also mild brown pigmentation noted within splenic macrophages in 7 of 16 treated dogs (and none of the controls) consistent with elevated erythrocyte turnover. The differences in incidence or severity of the brown pigment were insufficient to determine a dose-dependent relationship. However, hematology tests showed normal to elevated values for hemoglobin and hematocrit at any time points postinfusion. No changes in RBC counts were observed that would be considered biologically significant. A previous study evaluating blood compatibility, the ASTM standard test for hemolysis was negative, indicating that gold nanoshells were nonhemolytic. This mild level of brown pigmentation was not considered biologically significant.
There were no other significant histopathological observations in the heart, lung, liver, spleen, or mesenteric lymph nodes of the treated groups.
Ten-Month Chronic Systemic Toxicity in the Beagle Dog
Due to the long residence time of the particles in the body as determined in the mass balance study, a study to investigate any chronic toxicity related to the particle infusions was undertaken. In this 10-month chronic systemic toxicity in the beagle dog study there were no noted clinical observations. The study consisted of 3 groups, a 10% trehalose control group, a group receiving 2 doses at 7.5 mL/kg body mass, and a group receiving 2 doses at 15 mL/kg body mass. After the infusion of the control solution or the nanoshell solution, blood chemistry, hematology, urinalysis, ECG ophthalmology, body weights, and general health were measured over the course of the 10-month study. The ECG and ophthalmology examinations were normal. Some signs of anxiety (shivering, panting, and salivation) were seen during the postdosing observations. There were no significant differences in body weight changes among groups. The 2 nanoshell groups consumed significantly less food than the control, and the high nanoshell dose group consumed significantly less than the low-dose group. Findings during weekly clinical observations included occasional erythema in the ear, flaky skin, red or irritated eyes, clear ocular discharge, alopecia, and gingivitis. Hyperactivity and/or excitability were also noted. Occasional decreased appetite, salivation, vomit, and red eyes were seen during the twice-daily general health observations. Since these were found in both the test and vehicle control groups, none were considered dose related. Mean body weights for all groups are summarized in Figure 4. On day 30, a decrease in mean body weight was seen in both groups treated with the nanoshells. The decrease was not dose dependent and was transient in nature, returning to normal by day 37. Furthermore, there were no additional variations in the mean body weights for any of the groups compared to the control. For these reasons, the single reported, transient decrease was not considered to be biologically relevant.
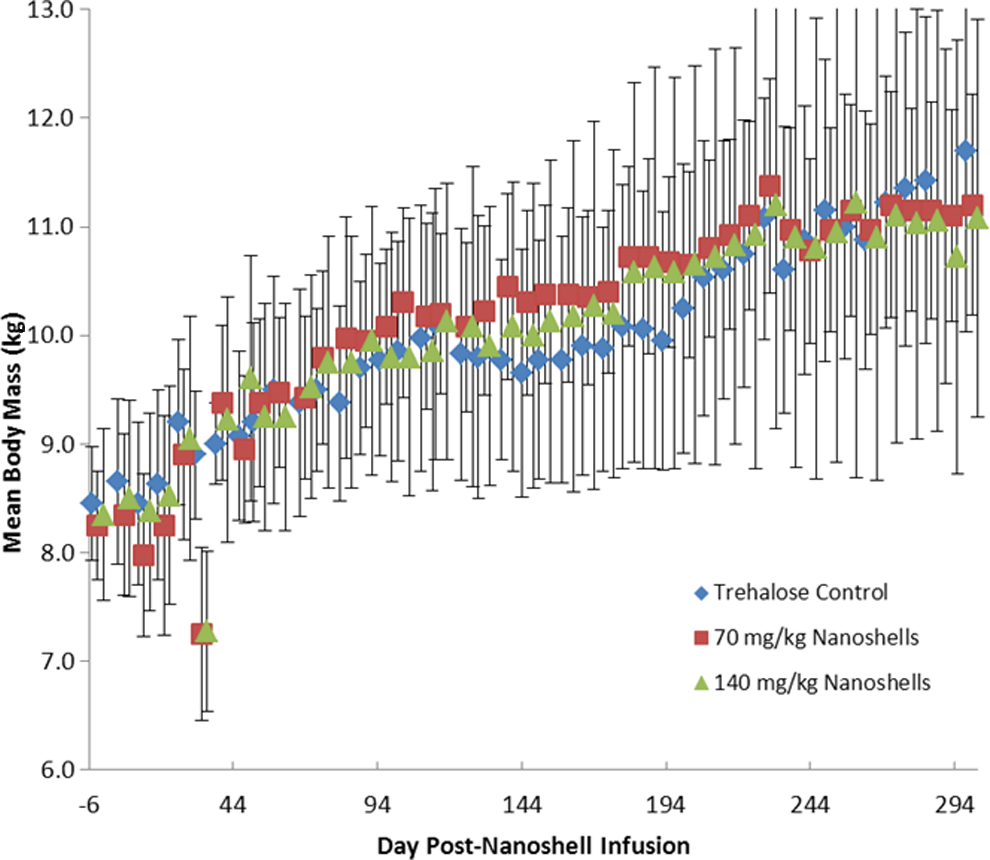
Mean body mass (±standard deviation [SD]) for all test groups. Over the course of the 10-month study, no treatment-related variations in body weights were seen for the test groups compared to the control.
Blood chemistry and hematology analysis showed no significant results for either test groups with the exception of the following: AST values were significantly less for the nanoshell test groups than the control values, on days 9 and 119. The hemoglobin level in low-dose nanoshell group was significantly greater than the control group on days 91 and 191, and hematocrit was significantly greater than control on day 191. None of these results were considered to be clinically significant. The mean values for each test group for all of the serum chemistry and hematology assays are included in the Supplementary Material.
At the end of the 10-month study, the animals were sacrificed and the major organ systems including heart, lung, liver, spleen, kidney, bladder, testes, and brain were harvested for histopathological analysis. The relative organ mass for the test and control groups showed no significant change in organ weight for any group when compared to control (Figure 5). There were no other significant pathological findings during the necropsy.
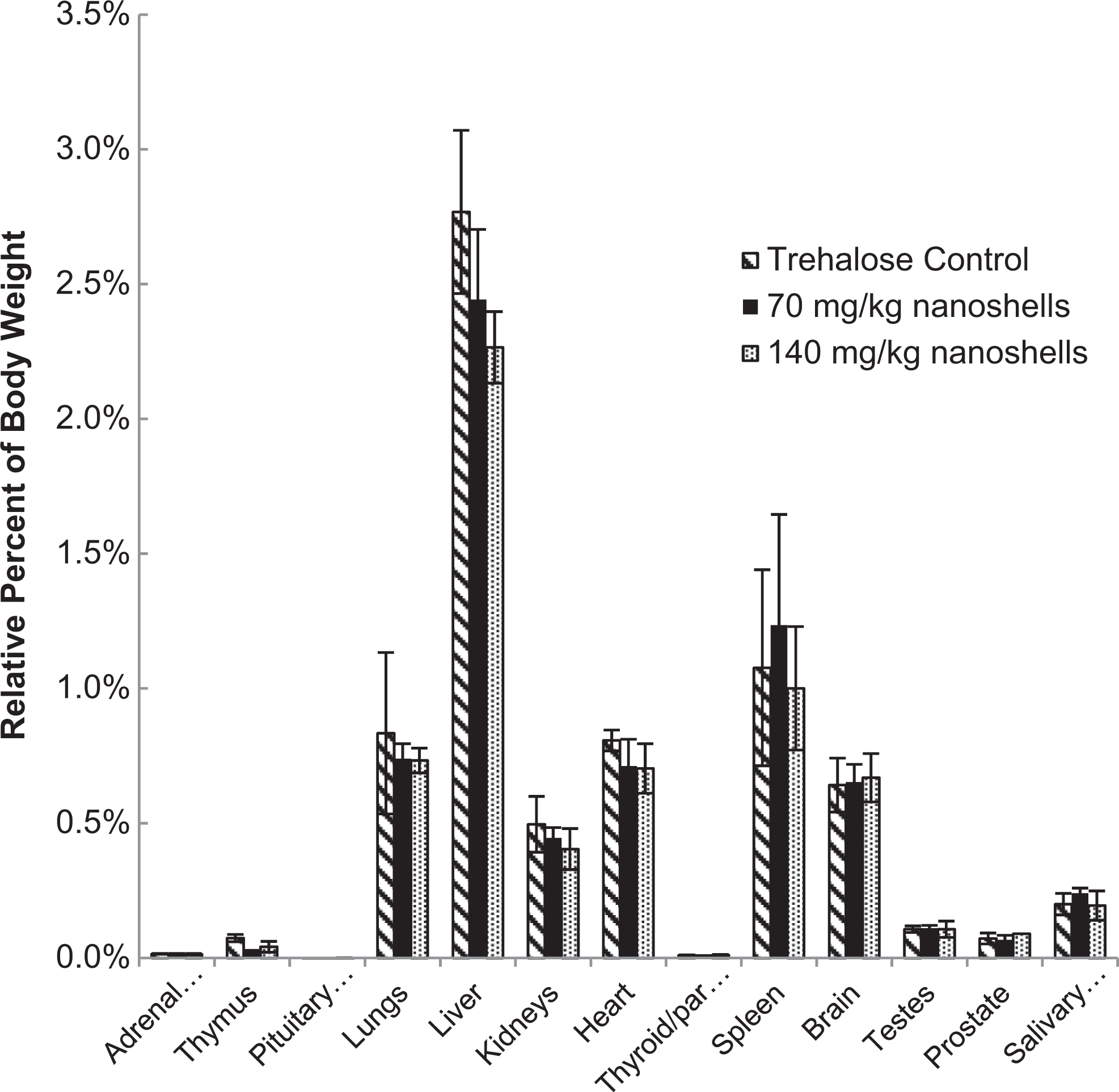
Relative organ mass at 10 months postinfusion for all test groups.
Table 2 summarizes the results of histopathology for organs with findings. Consistent with prior studies, histopathology noted the presence of a black pigmentation in the RES cells of the liver (Kupffer cells) and in macrophages of the spleen and in some lymph nodes. In the liver, the pigment was in the cytoplasm of Kupffer cells located in sinusoids. The pigmented Kupffer cells were often in aggregates (4-6 cells) especially near or in portal triads and appeared as microgranulomas. The degree of pigmentation of Kupffer cells was graded mostly moderate in the higher dose animals and graded mostly mild in the lower dose animals. There were no changes observed in hepatic cells or other structures of the liver, which suggests that the nanoshells are inert in the liver.
Histopathological Findings From 10-Month Chronic Dog Study.
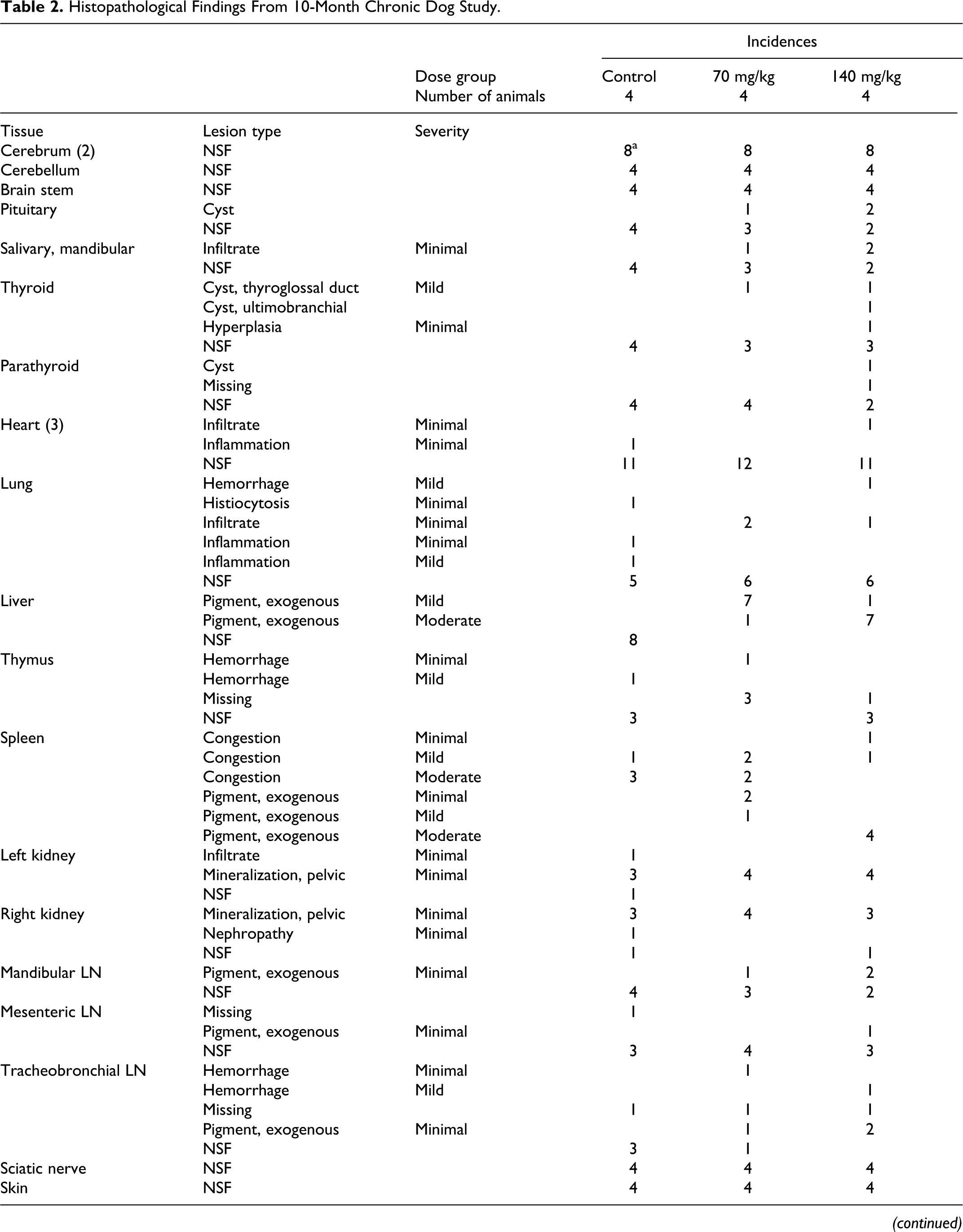
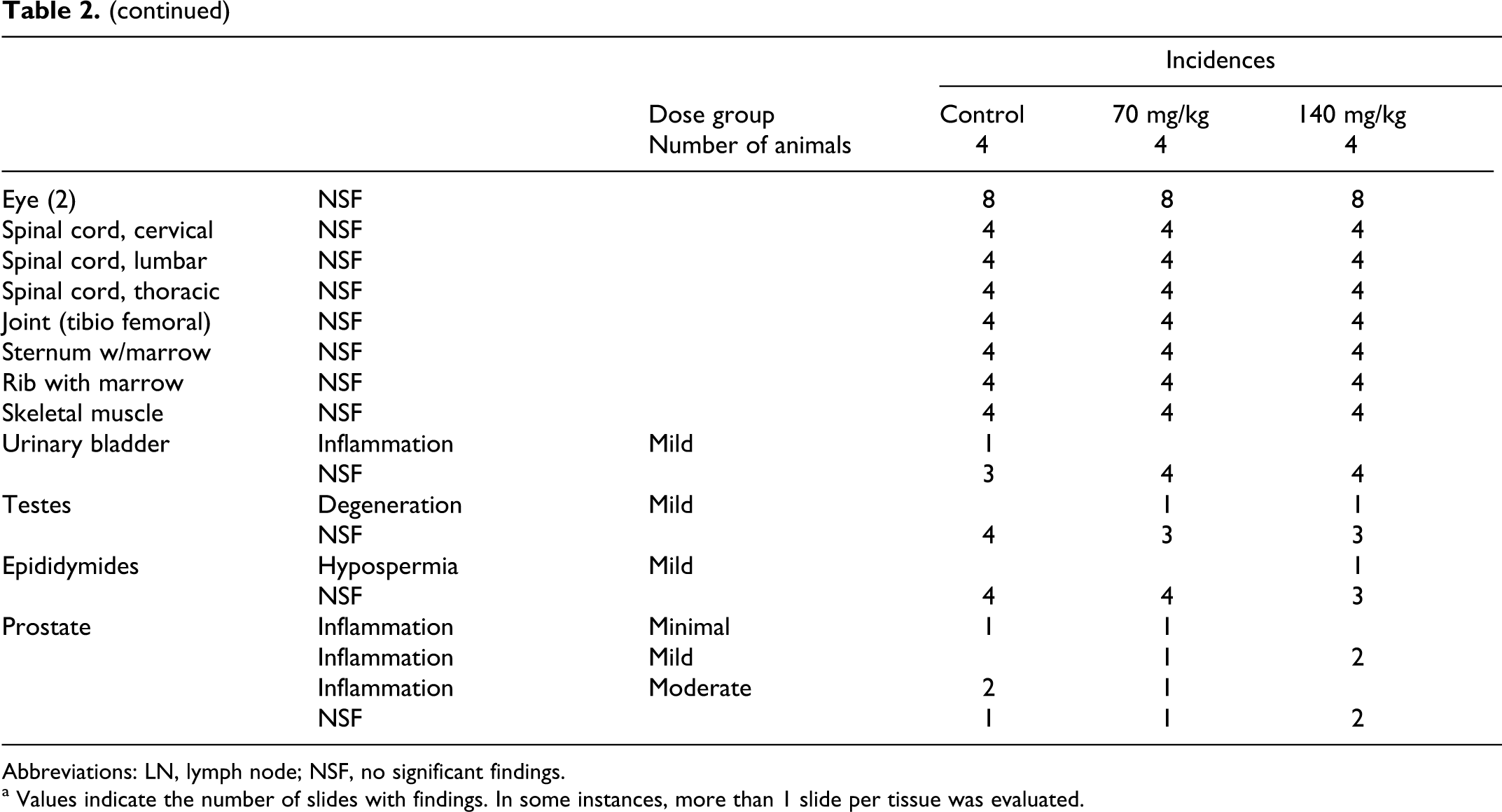
Abbreviations: LN, lymph node; NSF, no significant findings.
a Values indicate the number of slides with findings. In some instances, more than 1 slide per tissue was evaluated.
The same black pigment was observed in macrophages in the red pulp of the spleen. Many spleens had moderate blood congestion, which is common in canines that have been euthanized with a barbiturate agent. This congestion masked (obstructed) the pigmented macrophages in the red pulp. In spleens with less congestion, macrophages with brown pigment (normal hemosiderin) were observed in the red pulp. The spleens of test animals had both macrophages with brown pigment and macrophages with black pigment. The degree of black pigment observed varied considerably (minimal to mild) due to the variation in congestion, but the black pigment was only in treated dogs.
The same black pigment was observed in macrophages in the medulla of several lymph nodes. The degree of pigmentation, when present, varied as a result of size of medulla present in the section of lymph node on the slide.
No other test article–related changes were considered present in this study. A number of spontaneous lesion commonly observed in dogs were diagnosed. None was considered to be test article related. Chronic inflammation in the prostate, which occurred in both test and control animals, was observed at a slightly higher incidence than usual.
Discussion
The biocompatibility of gold and its use in medical applications has previously been established. Gold salts (eg, sodium aurothiomalate) have been used for the chronic treatment of rheumatoid arthritis without significant toxicity. 30 While gold salts are a reactive, ionic form of gold and have substantial differences from zero-valent nanoparticles, the recent use of gold-based nanoparticles requires a complete reevaluation of toxicity. In particular, potential effects on liver and kidney function must be evaluated, given the infrequent but potentially significant effects of the gold salts.
The biological fate of nanoparticles in the body is strongly affected by the shape and size of the nanoparticle as well as its surface coating. As a result, any toxicological analysis of a nanoparticle agent must include detailed information on the physical properties of the particles and the solution in which the particles were suspended.
As the complete array of testing presented herein took place over the course of multiple years several individual batches of particles were prepared using our standard methods and batch records. In order to be able to make reasonable comparisons for the various toxicity studies carried out on different batches of particles, all of the particles were manufactured following current good manufacturing practices. Written batch records detailing all raw material specifications and acceptance criteria were retained for all particle batches. All particle batches underwent extensive physical characterization prior to use in any study. The result of this physical characterization is to remove any significant variability in the physical properties of the particles, which may confuse the analysis of the resulting tests. The acceptance specifications for all particle lots were identical to our “clinical grade” material with respect to particle size, peak optical resonance, particle concentration, osmotic strength, and endotoxin levels. For all of the in vivo tests, the material was also tested for sterility prior to use.
The intended clinical use of the particles involves a systemic injection of the particles, which will allow for accumulation of the particles in the tumors to be treated through the EPR effect. As a result, essentially every organ system in the body has some exposure to the nanoshells prior to clearance from the bloodstream. This necessitates a comprehensive toxicity study to determine whether the nanoparticles are safe for use. The battery of tests based on the ISO-10993 standards is designed to determine the toxicity of any medical device prior to any clinical use. These tests look a broad range of potential toxic effects ranging from mutagenicity to sensitization. The nanoshell particles showed no significant signs of toxicity in vitro or in vivo at dose levels greater than or equal to the doses expected for clinical usage.
After finding no evidence of toxicity in the ISO standard tests, a more comprehensive biodistribution study looking at the biological fate of the particles after infusion in healthy animals was performed. In this study, it was determined that the particles accumulate in the liver and spleen of the animals, which is consistent with the clearance of the particles from the blood stream by the RES. The mass balance study indicates that the particles remain in the liver and spleen for extended periods of time, perhaps indefinitely. The presence of the particles in the liver and spleen, in quantities similar to the initial infused dose, indicates that the body is unable to break down the gold shell of the particle. As the particle size is too large to be cleared through standard excretion pathways, the particles reside in the liver and spleen for an extended period of time, perhaps permanently. Histopathology performed on animals >1 year after infusion of particles showed no signs of toxic effects in the liver and spleen. As such, while the particles may remain in the liver and spleen for the life of a treated patient, there does not appear to be any evidence of any toxic side effects from the particles. As the selective accumulation of particles in tumor versus normal tissue is the primary advantage to thermal therapy using nanoshell particles, the accumulation in the liver and spleen may make diseases of those organs poor targets for this therapy.
The long-term presence of particles in the body places increased importance on the determination of any effect the particles may have on the various organ systems in which they reside. In order to verify that there is no significant toxicity related to nanoshell accumulations in the liver and spleen, a 10-month chronic toxicity study in Beagle dogs was undertaken specifically to address this question. The use of dogs provides a second species on which toxicity can be studied. As the nanoshell particles are already in clinical trials for treatment of prostate cancer in patients scheduled for radical prostatectomies, in this study we chose to use male dogs, which was a variation from the previous (acute) study in female dogs. This change was made in order to determine whether there was any significant particle accumulation in healthy prostates, whether there is nanoshell accumulation, and whether there is any resultant toxicity. The lack of significant toxicity in the 10-month study, and in the hematology and blood serum chemistry taken throughout the term of the study in particular, demonstrates that the nanoshell particles are not associated with any toxic effects.
Conclusion
While there are reports in the literature of special risks associated with nanoparticles, in aggregate these have generally been associated with smaller particles and with higher surface charge densities. Due to the wide variety of nanoparticle compositions, surface chemistries, manufacturing methods, and applications, it is important to treat each nanoparticle application individually to determine the potential benefits and hazards to patient health. The thorough evaluation conducted on the gold nanoshell at the extensive exposure levels evaluated here identified no indication of toxicity, lack of tolerance, or immunological effects.
Supplementary Material
Experimental Details for ISO Toxicity Screen
Footnotes
Acknowledgments
We wish to thank the Nanoparticle Characterization Lab of the National Cancer Institute for their assistance in physical characterization of the particles and Dennis James of Texas A&M for the development of NAA methodologies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: in part by Contract 70NANB4H3040 of the Advanced Technology Program.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
