Abstract
Propylene glycol is an aliphatic alcohol that functions as a skin conditioning agent, viscosity decreasing agent, solvent, and fragrance ingredient in cosmetics. Tripropylene glycol functions as a humectant, antioxidant, and emulsion stabilizer. Polypropylene glycols (PPGs), including PPG-3, PPG-7, PPG-9, PPG-12, PPG-13, PPG-15, PPG-16, PPG-17, PPG-20, PPG-26, PPG-30, PPG-33, PPG-34, PPG-51, PPG-52, and PPG-69, function primarily as skin conditioning agents, with some solvent use. The majority of the safety and toxicity information presented is for propylene glycol (PG). Propylene glycol is generally nontoxic and is noncarcinogenic. Clinical studies demonstrated an absence of dermal sensitization at use concentrations, although concerns about irritation remained. The CIR Expert Panel determined that the available information support the safety of tripropylene glycol as well as all the PPGs. The Expert Panel concluded that PG, tripropylene glycol, and PPGs ≥3 are safe as used in cosmetic formulations when formulated to be nonirritating.
Introduction
A safety assessment of propylene glycol (PG) and polypropylene glycols (PPGs) was published in 1994. 1 On the basis of the available data, the Cosmetic Ingredient Review (CIR) Expert Panel concluded that these ingredients were safe for use in cosmetic products at concentrations up to 50.0%. In that assessment, the specific PPG chain lengths were not identified, however, concentration of use data were reported for PPG-9, PPG-26, and PPG 425. Currently, the International Cosmetic Ingredient Dictionary and Handbook names PPG-3, PPG-7, PPG-9, PPG-12, PPG-13, PPG-15, PPG-16, PPG-17, PPG-20, PPG-26, PPG-30, PPG-33, PPG-34, PPG-51, PPG-52, and PPG-69. Because new studies published after the 1994 assessment are available, that address the safety of PG and PPGs, the Expert Panel considered these data in support of the safety of these specific PPGs currently listed in the International Cosmetic Ingredient Dictionary as well as all chain lengths that may be added in the future.
This report is an update of the 1994 safety assessment and, as such, it contains information that was published after the 1994 assessment was issued.
Dipropylene glycol is not included in this report since it was previously reviewed in a separate report. In 1985, the Expert Panel determined that dipropylene glycol was safe as used in cosmetics. 1 That conclusion was confirmed in 2006. 2
Tripropylene glycol, which has not been reviewed, is included in this report. Tripropylene glycol is different from PPG-3. The PPG-n designations all acknowledge that these ingredients are produced in a polymerization reaction that can lead to some different chain length compounds, since the process in not end blocked. Tripropylene glycol is an ingredient that contains only the “3” chain length.
Chemistry
Definition and Structure
Propylene glycol (CAS No. 57-55-6) is an aliphatic alcohol that conforms generally to the formula in Figure 1. 3 Tripropylene glycol (CAS No. 24800-44-0) is an organic compound that conforms to the formula in Figure 2. 3 Synonyms for PG and tripropylene glycol are listed in Table 1.
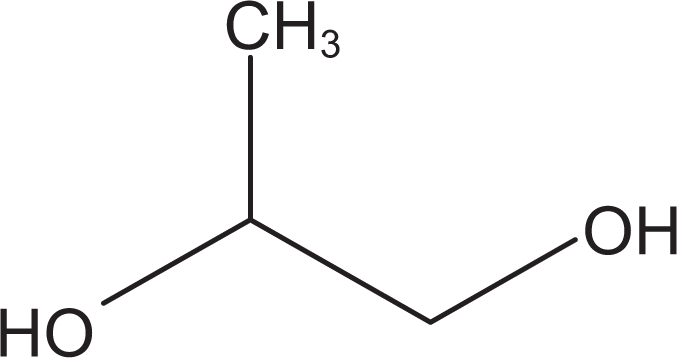
Propylene glycol.
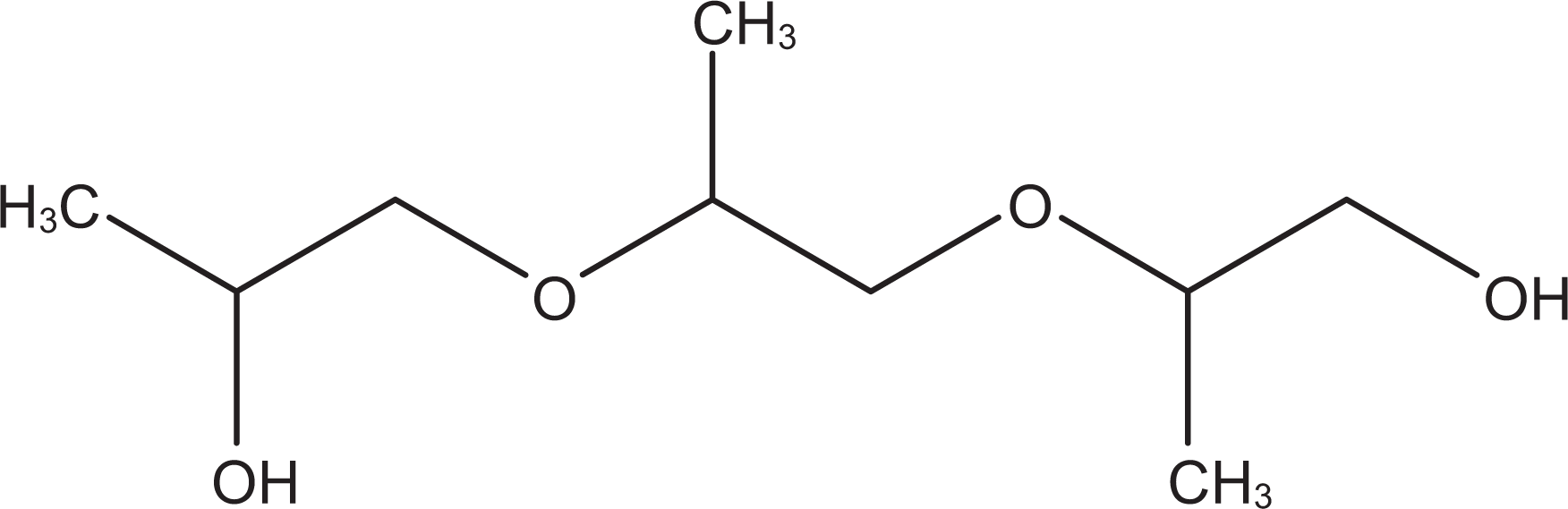
Tripropylene glycol.
Synonyms

Abbreviations: PPG, polypropylene glycol.
The PPGs (generic CAS No. 25322-69-4) are polymers of propylene oxide that conform generally to the formula in Figure 3.
3
According to the International Cosmetic Ingredient Dictionary and Handbook, international nomenclature cosmetic ingredient (INCI) names for the PPGs refer to the average “n” value corresponding to the propylene oxide chain length of the polymer; that is, PPG-3 would have an average chain length of 3. (Synonyms for PPGs are also listed in Table 1.)
Polypropylene glycol.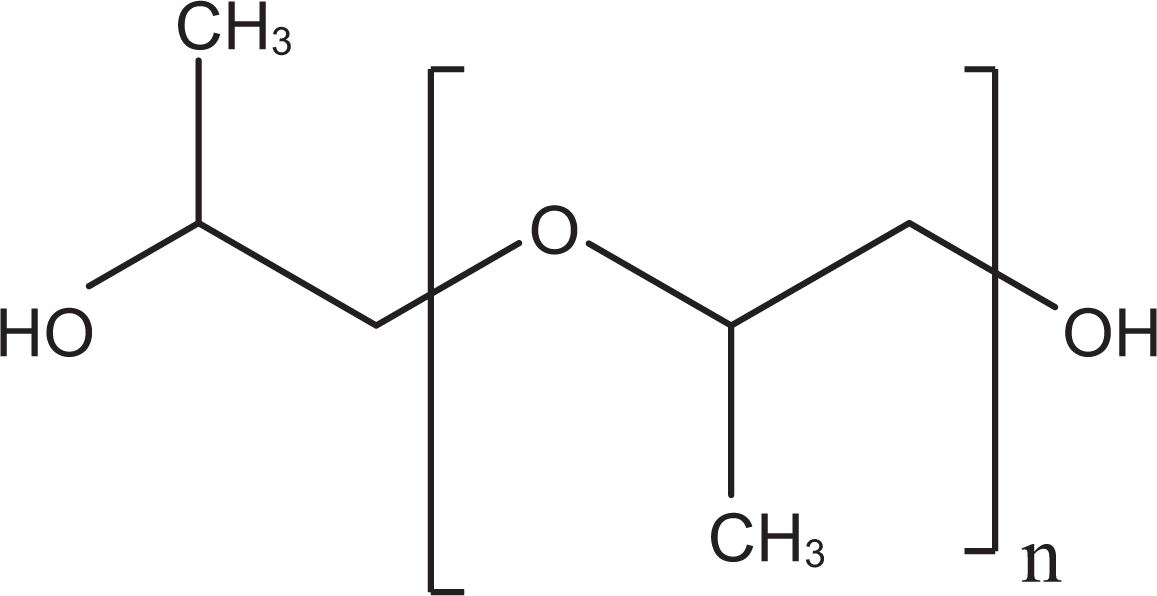
As stated above, the INCI names for cosmetic PPGs refer to the chain length. However, different naming conventions are used in identifying PPGs and the potential for confusion exists. When the official INCI name for each ingredient is used, the name is given as PPG, dash, and then the average number of units, for example, PPG-3. However, the PPGs can also be identified using the average molecular weight as part of the name; this is indicated as PPG, space, average molecular weight, for example, PPG 200. Table 2 gives the INCI name, molecular weight name where available, and calculated molecular weight of the PPGs.
PPG INCI Names, Molecular Weight Names, and Calculated Molecular Weightsa
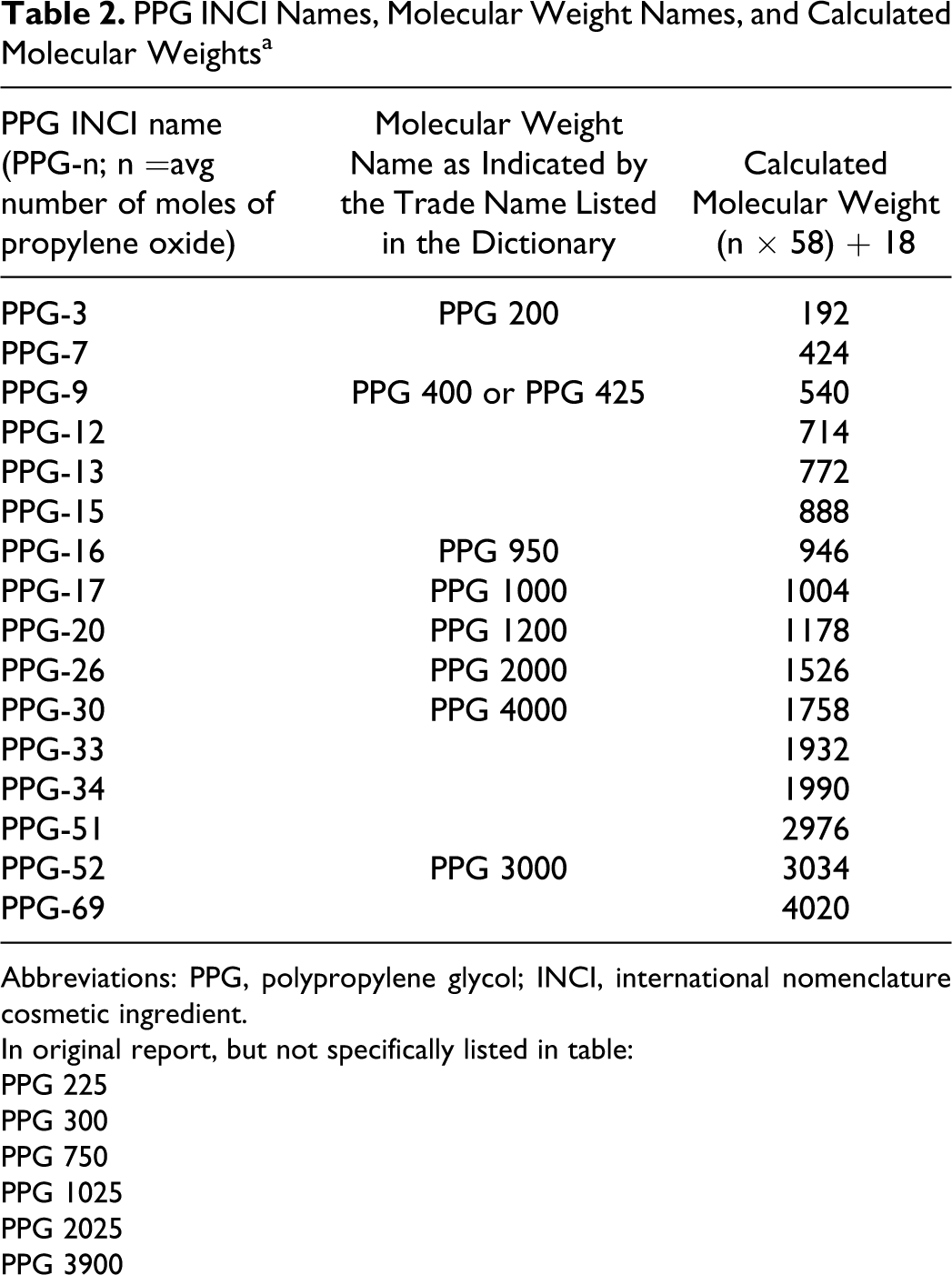
Abbreviations: PPG, polypropylene glycol; INCI, international nomenclature cosmetic ingredient.
In original report, but not specifically listed in table:
PPG 225
PPG 300
PPG 750
PPG 1025
PPG 2025
PPG 3900
Physical and Chemical Properties
The physical and chemical properties of PG, tripropylene glycol, and the PPGs are summarized in Table 3.
Chemical and Physical Properties

Method of Manufacture
Tripropylene glycol (as well as dipropylene glycol) is formed by sequential addition of propylene oxide to PG. 4 The products are formed simultaneously and separated by distillation.
Impurities
In the original safety assessment on PG, Dow Chemical Co recommended that US Pharmacopoeia (USP)-grade PG be used in cosmetics. 5 According to recent information, the USP has set safety limits of diethylene glycol and ethylene glycol content at a maximum of 0.1%. 6 The USP grade PG manufactured by Dow contains diethylene glycol and ethylene glycol at concentrations that are nondetectable (quantification limit of 0.008% wt/wt). Dow also has stated that they meet or exceed all requirements currently found in the European Pharmacopoeia, Japanese Pharmacopoeia, and Food Chemicals Codex. Two companies submitted information regarding the concentration of propylene oxide in PPGs used to make finished products. 7 Both companies report a maximum of 10 ppm propylene oxide.
Use
Cosmetic
Propylene glycol is used in cosmetic formulations as a skin conditioning agent (humectant or miscellaneous), viscosity decreasing agent, solvent, or fragrance ingredient. 3 The PPGs function primarily as skin conditioning agents, with some functioning as solvents. Tripropylene glycol functions as a humectant, antioxidant, or emulsion stabilizer.
At the time of the original safety assessment, according to information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP), PG was used in 5676 cosmetic formulations at concentrations ranging from 0% to >50%. 5 Both PPG-9 and PPG-26 were used in 6 and 10 cosmetic formulations, respectively, at concentrations of 0.1% to 5%, and PPG 425 (thought to be synonymous with PPG-9) was used in 1 cosmetic formulation at a concentration range of 1% to 5%.
The frequency and concentration of use of PG has increased. Recent VCRP data indicate that PG is used in 9094 cosmetic formulations (out of 34 391 total formulations reported). 8 Polypropylene glycol (chain length not specified) is reported to have 45 uses. Polypropylene glycol-9 is reported to be used in 84 cosmetic formulations, and PPG-12 is used in 3, PPG-15 in 1, PPG-17 in 3, PPG-26 in 2, and PPG-30 in 5 cosmetic formulations. Tripropylene glycol is used in 8 formulations. A survey of current use concentrations conducted by the Personal Care Products Council (the Council) reported that PG is used at concentrations of 0.0008% to 99%. 9 Propylene glycol, which is used in 313 of the 580 deodorant products reported to the VCRP, 8 is used at concentrations of 3% to 73%; this is the greatest leave-on concentration used. 9 The highest concentration of use of PG is 99%, but that use is in products that will be diluted, for example bath oils, tablets, or salts. Additionally, the Council survey results reported that PPG-9 is used at 0.05% to 22%, PPG-12 at 1%, PPG-17 at 1% to 2%, PPG-26 at 0.2%, and PPG-34 at 20%. Tripropylene glycol is used at concentrations up to 22%; the 22% is in an underarm deodorant. Table 4 presents current product formulation data for PG, tripropylene glycol, and the PPGs.
Frequency and Concentration of Use
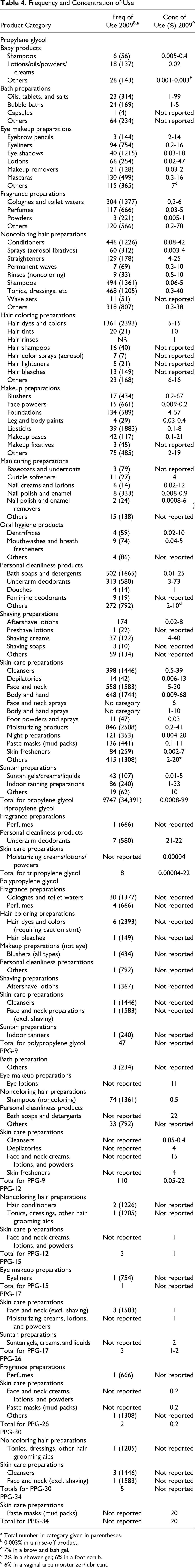
a Total number in category given in parentheses.
b 0.003% in a rinse-off product.
c 7% in a brow and lash gel.
d 2% in a shower gel; 6% in a foot scrub.
e 6% in a vaginal area moisturizer/lubricant.
Propylene glycol is used in hair sprays, and its effects on the lungs that may be induced by aerosolized products containing this ingredient may be of concern. The aerosol properties that determine deposition in the respiratory system are particle size and density. The parameter most closely associated with deposition is the aerodynamic diameter, da , defined as the diameter of a sphere of unit density possessing the same terminal settling velocity as the particle in question. In humans, particles with an aerodynamic diameter of ≤10 µm are respirable. Particles with a da from 0.1 to 10 µm settle in the upper respiratory tract and particles with a da < 0.1 µm settle in the lower respiratory tract. 10,11 Particle diameters of 60 to 80 µm and ≥80 µm have been reported for anhydrous hair sprays and pump hairsprays, respectively. 12 In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range and the mean particle diameter in a typical aerosol spray has been reported as ~38 µm. 13 Therefore, most aerosol particles are deposited in the nasopharyngeal region and are not respirable.
Tripropylene glycol, PG, and PPGs are not included in the list of ingredients that are prohibited for use in the European Union 14 or on the list of ingredients restricted or prohibited for use in Japan. 15
Noncosmetic
Propylene glycol is generally recognized as safe (GRAS) as a direct food additive when used in accordance with good manufacturing practices, and it is approved as a direct and indirect food additive. 16 According to the Joint FAO/WHO Expert Committee on Food Additives (JECFA), the acceptable daily intake (ADI) of PG is 25 mg/kg/bw/d. 17 In Japan, the Ministry of Health, Labour, and Welfare (MHLW) specified that according to the food sanitation law, PG has no potential to cause harm to human health. 18
Propylene glycol is used as an inactive ingredient in a number of FDA-approved drug products. It has been approved at concentrations up to 98.09% in topical drugs and 92% in oral solutions. 19 There is inadequate evidence to establish PG as GRAS and effective in OTC pediculicide drug products.
Propylene glycol has many uses in pharmaceuticals, food, and manufacturing. 20 It is used in organic synthesis, especially for PPG and polyester resins. 21
Polypropylene glycol is approved as a secondary direct food and additive and as an indirect food additive. 16 Polypropylene glycol has many industrial uses. 21
Tripropylene glycol also has many uses in pharmaceuticals, food, and manufacturing. It is used as an intermediate in resins, plasticizers, pharmaceuticals, insecticides, and the production of ethers and esters. 22
Toxicokinetics
Absorption
Propylene glycol
The dermal penetration of [ 14 C]PG through excised female hairless mouse skin from the ternary cosolvent containing 10 mol% oleic acid and 6 mol% dimethyl isosorbide in 84% PG was determined. 23 Over a 24-hour period, the cumulative penetration of PG was 57.1% of the applied amount.
The dermal absorption of PG was determined in the outermost layers of skin using thermal emission decay–Fourier transform infrared (TED-FTIR) spectroscopy. 24 Propylene glycol was applied to the fingertip of one human participant for 30 minutes using PG-soaked cotton wool. The site was wiped and allowed to dry for 1 minute. The thickness of the surface layer of stratum corneum probed was 0.71 µm. Measurements were performed every 25 minutes over a 3-hour period, with 1 measurement taking 15 minutes. The concentration of PG remaining at the surface of the stratum corneum decreased over time. At 12 and 32 minutes, the maximum concentration of PG was found at a depth of <1 µm, while at 107 and 157 minutes, the maximum concentration of PG was found at a depth of 3 to 4 µm. At a depth of 6 µm, the greatest concentration of PG, 0.2%, was seen at 32 minutes. The authors suggested that PG molecules diffuse into stratum corneum only to a depth of 6 to 7 µm, approximately. The researchers also suggested that PG molecules do not reach the dermis.
Dermal Penetration Enhancement
Propylene glycol
Propylene glycol has been described as a penetration enhancer, and penetration enhancers act by various mechanisms to perturb diffusional pathways through the skin. Proposed mechanisms of penetration enhancement by PG include alteration of barrier function by its effects on a keratin structure or a PG-induced increase in the solution capacity within the stratum corneum. 23 Examples of the effect of PG on penetration are summarized in Table 5.
Penetration Enhancement by PG

Toxicology
Cytotoxicity
Propylene glycol
The cytotoxicity of PG was determined in assays that measured inhibition of human foreskin fibroblasts and keratinocytes, inhibition of collagen contraction by fibroblasts, and changes in cell morphology of fibroblasts and keratinocytes. 25 Fibroblast and keratinocyte proliferation was inhibited within 3 days after administration of PG; no significant changes in cell proliferation occurred with a 6-day administration. Propylene glycol was a moderately potent inhibitor, with an IC50 (concentration causing 50% proliferation inhibition) of 280 mmol/L for fibroblasts and 85 mmol/L for keratinocytes. The effect of PG on collagen contraction by fibroblasts was concentration dependent throughout the entire study. The concentration causing 50% contraction inhibition was 180 mmol/L.
The effect of PG on changes in cell morphology also was examined. 25 A gradual detachment of cells from the culture accompanied by changes in cell shape occurred in confluent keratinocyte cultures when the concentration of PG was increased above 5%. After 24 hours, replacing medium containing 5% PG with PG-free medium resulted in almost complete recovery within 48 hours. However, this recovery did not occur with 7% PG. Similar results were observed with fibroblasts, and the concentration inducing irreversible cell damage in both fibroblast and keratinocytes cultures was 660 mmol/L PG.
Single-Dose Toxicity
Oral
Polypropylene glycols
The acute toxicity of PPG 425 was evaluated using 2 groups of 3 rats (strain and gender not specified). 26 The rats were given a single oral dose of 250 or 1000 mg/kg PPG 425 by gavage and observed for 14 days. Animals of the low-dose groups had convulsions and loss of coordination, whereas animals of the high-dose group had convulsions. One high-dose animal died on day 1. All low-dose animals and the remaining 2 high-dose animals survived until study termination.
Parenteral
Propylene glycol
An acute study was performed in which female ICR mice were dosed intraperitoneally (ip) with 2600, 5200, or 10 400 mg/kg PG. 27 All except the high-dose mice survived 6 days after dosing. (The number of high-dose mice that died was not given.) Signs of toxicity, such as lethargy and ruffled hair coats, were not observed in the 2600 and 5200 groups.
Repeated-Dose Toxicity
Oral
Propylene glycol
Groups of 6 inbred male Wistar rats were dosed orally by gavage, daily, with 294.23 mg PG/100 g body wt (as 1 mL 28.4%/100 g) for 10 (group 1), 20 (group 2), or 30 days (group 3), and the effects on a number of intestinal parameters were determined.
28
Control groups received an equal volume of saline for 10, 20, or 30 days. After termination of dosing, animals were fasted overnight and then killed. All animals survived until study termination. Body weight gains were statistically significantly decreased for animals in group 1 and increased for animals in groups 2 and 3. A number of enzyme activities were enhanced; statistically significant increases were seen in sucrase activity in groups 1 and 2 and lactase and γ-glutamyl transpeptidase activity in group 3. Absorptive function was assessed by measuring nutrient uptake. Statistically significant increases of
Nineteen male Han:Wistar rats were given drinking water containing 40 g/L PG for 2 weeks; a control group of 16 rats was given tap water. 29 The animals were placed in metabolism cages during the last 24 hours of dosing and urine was collected. Propylene glycol administration did not have any effect on urinary excretion of oxalic or alkoxyacetic acid, nor did it affect pH or urinary metabolites. Propylene glycol did not cause any renal effects.
Groups of 8 male and 8 female CD-1 mice were given 0.5%, 1.0%, 2.5%, 5.0%, and 10.0% PG in the drinking water for 14 days. 30 Negative controls were given untreated drinking water. Body weight gains of test animals were similar to or greater than controls. No animals died during the study.
Inhalation
Male and female Sprague-Dawley rats (number per group not given) were exposed to 0.16, 1.0, or 2.2 mg PG/L air for 6 hours/d, 5 days/week, for 13 weeks in a nose-only inhalation study. 31 There was no difference in body weights for any of the male dose groups, while mid- and high-dose females had significantly decreased body weights starting on days 64 and 50 of the study, respectively. Feed consumption was decreased for the females starting on days 50 and 43, respectively. Relevant differences occurred in some hematological parameters, serum enzyme activities, and lung, spleen, liver, and kidney weights; however these differences were inconsistent and without dose–response trends. The mid- and high-dose animals had increased goblet cells and increased mucin within these cells.
Ocular Irritation
Propylene glycol
The ocular irritation potential of PG was determined using groups of 6 male and female New Zealand white albino rabbits. 32 First, a single application of 1 drop of PG was instilled into the conjunctival sac of the left eye of each rabbit, and the eye was not rinsed. In the second part of the study, 1 drop of PG was instilled into the conjunctival sac of the left eye every 24 hours for 3 consecutive days. At both times, the contralateral eye was untreated and served as the control. The eyes were examined on days 1, 2, 3, and 7. With the single application, slight-to-moderate conjunctival hyperemia was observed on day 1 and resolved by day 2. The highest total score was 19 of 550, well below the category of marginal irritant (score of 65). Multiple instillations resulted in similar observations, with slight hyperemia lasting up to day 3 in 2 rabbits. The highest total score following multiple installations was 38 of 550, again below the category of marginal irritant.
Dermal Irritation/Sensitization
Propylene glycol
The dermal irritation potential of 100% PG was evaluated with male hairless SKH1 hr/hr mice. 33 Propylene glycol was instilled in polyvinyl chloride cups (vol 0.3 cm3) on the dorsal side of 3 mice. The test substance remained in contact with the skin for 24 hours. At the end of the 24 hours, the animals were killed and a sample of the exposed skin was examined microscopically. Propylene glycol was minimally irritating, with a total score of 7 (maximum score =77).
Reproductive and Developmental Toxicity
Propylene glycol
The reproductive and developmental effects of PG were evaluated using mice, rats, rabbits, and hamsters. 34 Groups of 25 or 28 female albino CD-1 outbred mice were mated and 22, 22, 22, 20, and 23 gravid mice were dosed by oral intubation with 0.0, 16.0, 74.3, 345.0, and 1600.0 mg/kg aq PG on days 6 to 15 of gestation. Groups of 25 to 28 female albino Wistar rats were mated and 22, 23, 22, 20, and 24 were dosed as above, respectively. Positive control groups of 23 mice and 21 rats were given 150.0 or 250.0 mg/kg aspirin, respectively. Body weights were recorded at various intervals and general observations were made daily. Caesarian sections were performed on days 17 and 20 for all mice and rats, respectively. All fetuses were examined macroscopically for visceral or skeletal defects. Administration of PG did not affect maternal or fetal survival in mice or rats, and there were no statistically significant differences in fetal anomalies between test and negative control groups in mice or rats.
Groups of 11, 11, 12, 14, and 13 gravid female Dutch-belted rabbits were dosed by oral intubation with 0, 12.3, 57.1, 267.0, or 1230.0 mg/kg aq PG on days 6 to 18 of gestation, respectively. A positive control group of 10 gravid rabbits was given 2.5 mg/kg 6-aminonicotinamide. Body weights were recorded at various intervals and general observations were made daily. Caesarian sections were performed on day 29. All fetuses were examined macroscopically and kept for 24 hours to evaluate survival. The pups were then examined viscerally and for skeletal defects. Administration of PG did not affect maternal or fetal survival, and there were no statistically significant differences in fetal anomalies between test and negative control group.
Groups of 24-27 female golden hamsters were mated and 21, 24, 25, 22, and 22 gravid hamsters were dosed by oral intubation with 0.0, 15.5, 72.0, 334.5, and 1550.0 mg/kg aq PG on days 6 to 10 of gestation, respectively. Positive controls were given 250.0 mg/kg aspirin. Body weights were recorded at various intervals and general observations were made daily. Caesarian sections were performed on day 14. All fetuses were examined macroscopically and for visceral or skeletal defects. Administration of PG did not affect maternal or fetal survival, and there were no statistically significant differences in fetal anomalies between test and negative control groups.
Propylene glycol was used as a vehicle in a reproductive and behavioral development study. 35 It was administered to 15 gravid Sprague-Dawley rats orally by gavage on days 7 to 18 of gestation at a volume of 2 mL/kg. Propylene glycol did not have any effects on reproductive or behavioral development parameters.
Female ICR mice were used to determine whether PG induced cytogenetic aberrations in mouse metaphase II (MII) oocytes that predispose zygotes to aneuploidy. 27 Groups of mice were first given an ip injection of 7.5 IU eCG to augment follicular maturation followed 48 hours later with 5 IU human chorionic gonadotropin (hCG) to induce ovulation. After 3 hours, mice were dosed ip with 1300, 2600, or 5200 mg/kg PG in distilled water. A control group was given distilled water only. For the MII portion of the study, ovulated oocytes were collected from 20 test animals/group and 30 control animals and processed for cytogenetic analysis 16 hours after administration of PG. The number of oocytes collected from test animals was nonstatistically significantly increased compared to controls. A statistically significant change in hyperploidy, hypoploidy, or single chromatids was not observed. An increase in the frequency of PCS at each dose was statistically significant, and the incidence of premature anaphase was statistically significantly greater in the 5200 mg/kg dose group as compared to controls. Neither metaphase I nor diploid oocytes were found.
For the zygote portion of the study, the female mice were paired with undosed males immediately after being given hCG; the females were dosed ip with 1300, 2600, or 5200 mg/kg PG 3 hours after hCG administration. The males were removed 16 hours after dosing with PG. Mated females were given colchine 22 hours after dosing with PG; zygotes were collected 18 hours later. There were 30, 40, 49, and 66 mice in the control, 1300, 2600, and 5200 mg/kg groups, respectively. The increase in hyperploidy was statistically significant in all test groups compared to controls. A statistically significant change was not seen for polyploidy or hypoploidy, and zygotes containing PCS, premature anaphase, or single chromatids were not found. The authors noted that there was no statistically significant difference in the proportion of zygotes collected for each group compared to oocytes. However, the number of zygotes analyzed compared to the number placed on slides was significantly decreased in the test groups; a relatively large portion of these zygotes had clumped chromosomes.
Genotoxicity
Tripropylene glycol
In a preincubation study with tripropylene glycol using Salmonella typhimurium strains TA1535, TA100, TA97, and TA98, the results were negative using concentrations of 0 to 10 000 µg/plate with and without metabolic activation. 36
Clinical Assessment of Safety
Propylene glycol
Propylene glycol dermal penetration was reportedly enhanced by the addition of fatty acids, such as oleic acid. 37 Transepidermal water loss (TEWL) and attenuated total reflectance (ATR)-FTIR were used to evaluate participants exposed to PG and/or oleic acid. 38 The TEWL was determined using 10 participants (number of males and females not specified) with application of occlusive chambers containing nothing, 300 µL PG, or 300 µL 0.16 mol/L oleic acid in PG, for 3 or 24 hours. The fourth site was not treated and not occluded. The TEWL measurements were started 3 hours after chamber removal to reduce volatile solvents on the skin surface in order to avoid interference with the Evaporimeter. The site treated with oleic acid/PG increased water loss for a longer period in comparison to the PG only or empty sites. The 3- and 24-hour applications of PG resulted in an enhanced water loss ratio of 1.1. With oleic acid/PG, these values were 2.0 and 2.1, respectively.
For the ATR-FTIR portion, an occlusion system containing PG or oleic acid/PG was applied to the forearm of each participant; a third site was untreated. The chambers were removed after 3 hours, and ATR-FTIR spectra were recorded. Upon removal at the site where oleic acid/PG was applied, the absorbance at the wavelength measuring free acid indicated the presence of extra free acid, while the absorbance at the wavelength characteristic of esterified ester lipids was similar to untreated and PG-treated sites. The absorbance ratio for these 2 wavelengths leveled off to that of the untreated site 3 hours after removal of the chambers, indicating migration of oleic acid into lower cell layers or lateral spreading within the stratum corneum. The researchers also examined ATR-FTIR when the oleic acid/PG site was tape stripped 5 times, removing 50% of the thickness of the stratum corneum, 2 hours after removal of the application chambers. The results indicated oleic acid accumulates in a deeper layer after the tape stripping.
Dermal Irritation/Sensitization
Propylene glycol
Intradermal injection of 0.02 mL undiluted PG produced a wheal-and-flare reaction within minutes, while the same volume applied epidermally did not produce any reaction. 39 These authors reported that, occasionally, subjective or sensory irritation sometimes occurred in volunteers after application of various concentrations of PG. Some researchers have proposed classifying skin reactions to PG into 4 groups: (1) irritant contact dermatitis; (2) allergic contact dermatitis; (3) nonimmunologic contact urticaria; and (4) subjective or sensory irritation.
Predictive Testing—Irritation
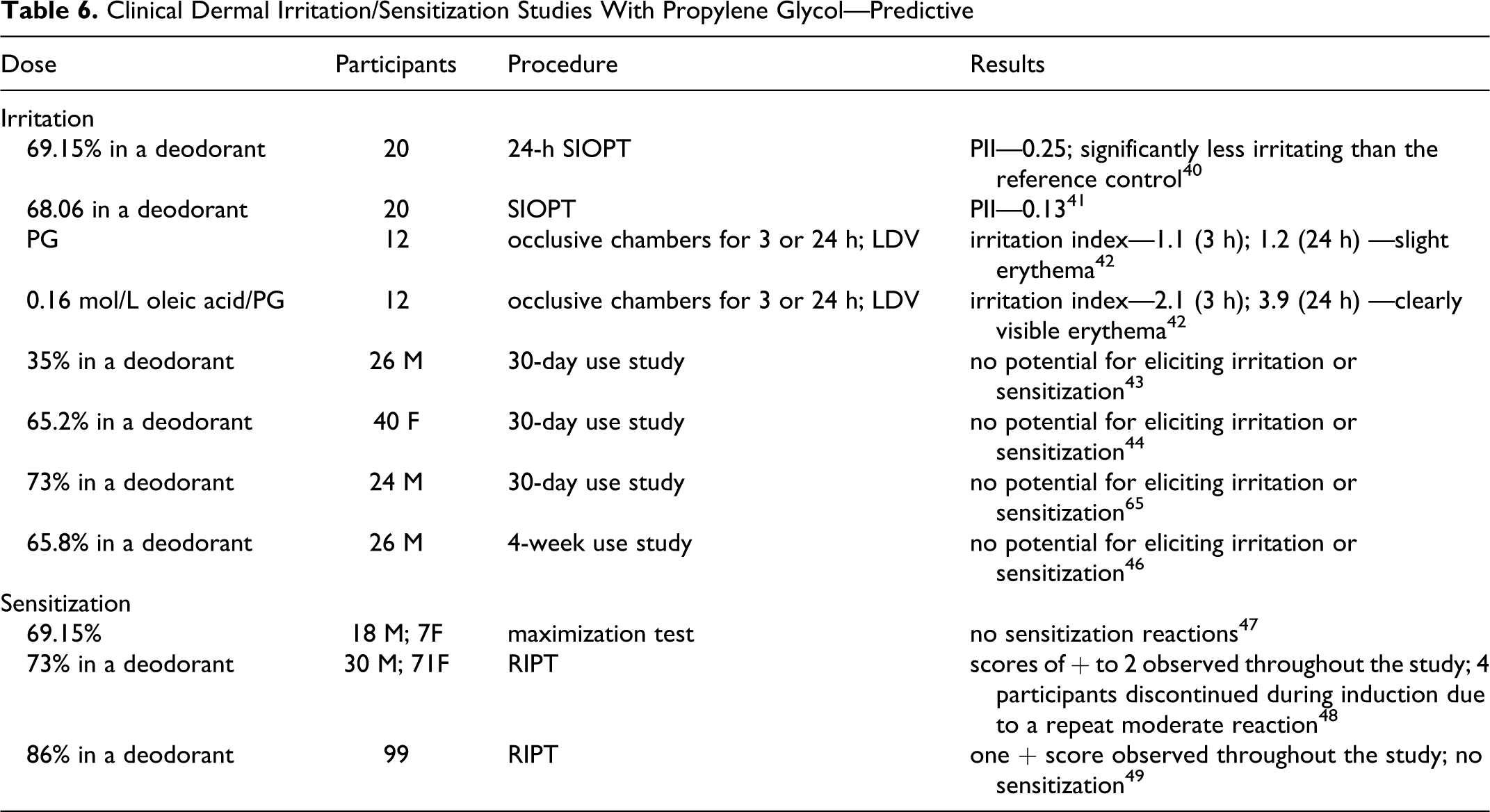
The results of the clinical dermal predictive irritation and sensitization studies on PG described in this section are summarized in Table 6.
Clinical Dermal Irritation/Sensitization Studies With Propylene Glycol—Predictive
Propylene glycol
A 24-hour single insult occlusive patch test (SIOPT) was performed on an undiluted deodorant formulation containing 69.15% PG using 20 participants (gender not specified). 40 A clear stick deodorant was used as a reference control. The test sites were scored on a scale of 0 to 4. With the test formulation, 4 participants had a score of ± (minimal faint uniform or spotty erythema) and 3 participants had a score of 1 (pink-red erythema visibly uniform in the entire contact area.) The primary irritation index (PII) for the deodorant containing 69.15% PG was 0.25. This product was significantly less irritating than the reference control, which had a PII of 0.93 and 17 of 20 participants with scores between ± and 3.
In another SIOPT, a deodorant formulation containing 68.06% PG was tested undiluted using 20 participants (gender not specified) 41 A deodorant currently in use was used as a reference control. Three participants had a score of ± and 1 had a score of 1 to the test formulation. The PII for the test formulation was 0.13, which was not significantly different than the PII of 0.15 for the reference control.
The irritation index for PG and 0.16 mol/L oleic acid/PG was determined using 12 participants (number per gender not specified) by applying occlusive chambers containing these 2 test substance to the volar forearm for 3 or 24 hours. 42 An empty chamber was applied to a third site, and the fourth site was an untreated control. Laser Doppler velocimetry (LDV) was used to measure blood flow upon removal. After 3 and 24 hours, the irritation index for PG was 1.1 (6 participants) and 1.2 (10 participants), respectively, indicating a 1-fold increase in blood flow to the test site. The irritation index for oleic acid/PG was 2.1 (6 participants) and 3.9 (10 participants) after 3 and 24 hours, respectively. Visually, the 24-hour application of PG produced only slight erythema, while the 24-hour application of oleic acid/PG produced clearly visible irritation.
Thirty-day use studies were completed with 26 male, 40 female, and 24 male participants to evaluate the potential for deodorant sticks containing 35%, 43 65.2%, 44 and 73% PG, 45 respectively, to induce dermal irritation and/or sensitization. The participants were instructed to apply the product to the underarm once daily for 30 days. None of the participants had any irritation or sensitization reactions, and the researchers concluded that the deodorant sticks containing 35%, 65.2%, or 73% PG did not demonstrate a potential for eliciting dermal irritation or sensitization. In a 4-week use study completed with 26 male participants following the same procedure, a deodorant stick containing 65.8% PG also did not demonstrate a potential for eliciting dermal irritation or sensitization. 46
Predictive Testing—Sensitization
Propylene glycol
A maximization test was completed with 25 participants, 18 male and 7 female, to determine the sensitization potential of a deodorant containing 69.15% PG. 47 During the induction phase, an occlusive patch containing 0.1 mL of 0.25% aq sodium lauryl sulfate (SLS) was applied for 24 hours to the outer arm, volar forearm, or the back of each participant. That patch was removed and an occlusive patch containing 0.1 mL of the test substance was applied to the same site for 48 to 72 hours, after which time the patch was removed and the site examined. If there was no irritation, the sequence was repeated with the SLS and test article patches for a total of 5 induction exposures. If irritation occurred at any time, the SLS patch was excluded. After a 10-day nontreatment period, a challenge was performed in which a previously unexposed site opposite the test site was first pretreated with an occlusive patch containing 0.1 mL of 5% aq SLS for 1 hour. Then an occlusive patch containing the test substance was applied for 48 hours, and the site was scored 1 hour and 24 hours after removal. All the scores were 0 for all participants following challenge. No sensitization reactions were seen to a deodorant containing 69.15% PG.
An RIPT was completed with 101 participants, 30 male and 71 female, to determine the sensitization potential of a stick deodorant formulation containing 73% PG. 48 During the induction phase, semiocclusive patches containing 0.2 g of the test material were applied to the upper back of each participant for 24 hours, 3 times per week, for a total of 9 applications. The first patch was scored (scale of 0-4) immediately after removal, while all others were scored prior to application of the next patch 24 to 48 hours later. During the induction phase, a score of 2 (moderate reaction) resulted in moving the patch to an adjacent site, while a second score of 2 or scores of 3 to 4 (marked severe) resulted in discontinuation of dosing. The challenge was performed approximately 2 weeks after the final induction patch using the same procedure but at an adjacent previously untested site. Challenge sites were scored 24 and 72 hours after application. Scores of + (barely perceptible or spotty erythema) to 2, with some dryness, were observed throughout the study. Four participants discontinued dosing during the induction phase because of a second moderate reaction. While the authors stated that a stick deodorant formulation containing 73% PG “did not indicate a clinically significant potential for dermal irritation or allergic contact sensitization,” the Expert Panel questioned that conclusion since repeated reactions were observed.
Another RIPT was completed with 99 participants to determine the sensitization potential of a stick antiperspirant formulation containing 86% PG. 49 (Initially, 113 participants were enrolled in the study; withdrawal was not due to adverse effects.) Occlusive patches containing 0.2 g of the test formulation were applied to the infrascapular region of the back 9 times during induction and once during challenge. One “+” reaction was observed during the entire study. There was no evidence of sensitization with an antiperspirant containing 86% PG.
Provocative Testing—Sensitization
Propylene glycol
Thirty-six patients with chronic venous insufficiency (CVI) were patch tested with 5% PG in petrolatum by application to the back for 2 days. 50 Twelve patients were male; 2, 5, and 5, had first-, second-, and third-degree CVI, respectively. Twenty-four patients were female; 5 and 19 had second- and third-degree CVI, respectively. (Procedural details not provided.) The results were read after 2 and 3 days; doubtful reactions were read after 4 days. The sensitization rate as a percentage of all patients was 8.3%. The sensitization rate of patients with second- and third-degree CVI tested with PG was 10% and 8.3%, respectively. Significant differences were found between males and females; 12.5% of females were sensitized while 0% of males were sensitized.
During the period 2000 to 2004, 308 patients, 111 males and 197 females, with contact dermatitis were patch-tested using the European standard series and some additional chemicals, including PG. 51 Patches were applied to the upper back using Finn chambers that were held in place with Scanpor tape. The patches were removed after 2 days, and the sites were evaluated after 30 minutes and 4 days. Propylene glycol, 5% in petrolatum, did not cause any positive reactions.
Photoallergenicity
Propylene glycol
Over a 2-year period, 30 males and 52 females with photoallergic contact dermatitis were photopatch tested with a standard series of sunscreens as well as some additional chemicals, including PG. 52 (Dose not given.) The allergens were applied in duplicate on the back and covered with opaque tape. After 24 hours, the tape was removed, the test sites evaluated, and one set of test sites was irradiated with an UVA dose of 5 J/cm2 (using a Daavlin UVA cabinet), giving an irradiance of 10.4 mW/cm2; this provided a 320 to 400 nm spectrum. The test sites, which were not covered after irradiation, were evaluated 24 and 72 hours later. While some positive reactions were observed to other test agents, PG did not produce a photoallergenic or contact allergy response.
Enhancement of Irritation Effects
Propylene glycol
The effect of the addition of PG to an isopropanol vehicle on the irritant reaction of benzoic acid was determined in a nonocclusive test using 15 participants, 7 males and 8 females. 53 Benzoic acid in isopropanol was tested at concentrations of 31, 62, 125, and 250 mmol/L without PG as well as with the addition of 1%, 2%, 5%, 10%, and 25% PG. The vehicles were also tested. Visual appearance, laser Doppler flowmetry, and skin color (using a Minolta chromameter) were measured at 20, 40, and 60 minutes after application. Propylene glycol enhanced the strength of the reactions to 125 and 250 mmol/L benzoic acid but not to 31 or 62 mmol/L benzoic acid. (This was observed using all 3 measurement methods.) Enhancement was observed with the addition of 1% PG, and maximal enhancement was attained with 5%. No reaction to application of the vehicles was observed.
Retrospective Analyses
The North America Contact Dermatitis Group (NACDG) performed a number of retrospective analyses on various dermatological conditions, determining that ≤6.0% of patients tested had positive reactions to 30% aq PG. These studies are summarized in Table 7.
Retrospective Analyses With Propylene Glycol
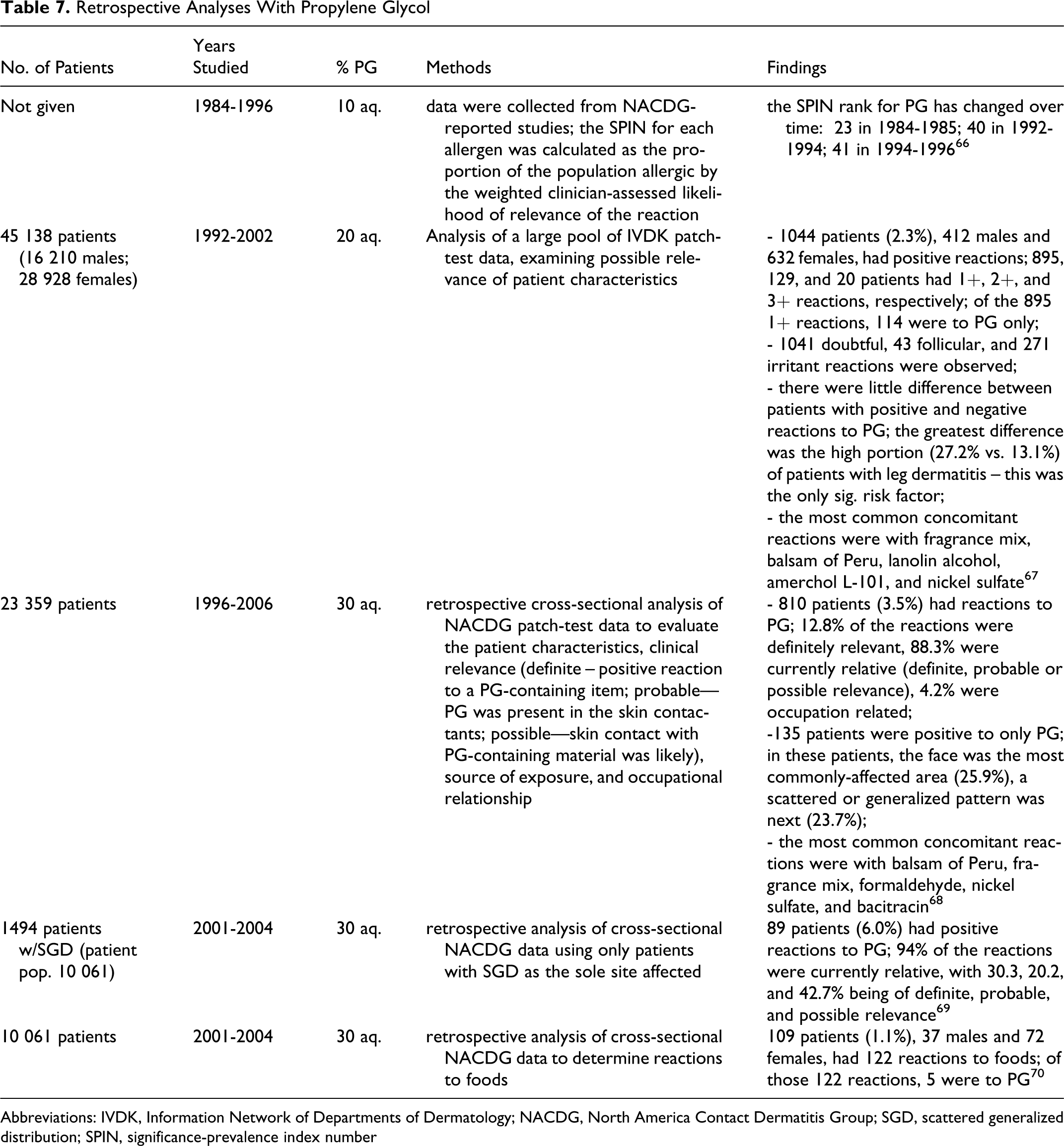
Abbreviations: IVDK, Information Network of Departments of Dermatology; NACDG, North America Contact Dermatitis Group; SGD, scattered generalized distribution; SPIN, significance-prevalence index number
Case Reports
A few case reports have been described concerning PG and hand dermatitis or atopic dermatitis. Patch test results generally had a positive reaction to PG in these case studies. Improvement was seen with the avoidance of PG-containing products. 54,55
Summary
Both PG and PPGs were reviewed by the CIR Expert Panel in 1994, and it was concluded that these ingredients were safe for use in cosmetic products at concentrations up to 50.0%. This rereview was opened to amend the conclusion (the concentration of use of PG is >50%), consider new data, and to add new ingredients so that all of the PPGs identified in the International Cosmetic Ingredient Dictionary and Handbook, that is, PPG-3, PPG-7, PPG-9, PPG-12, PPG-13, PPG-15, PPG-16, PPG-17, PPG-20, PPG-26, PPG-30, PPG-33, PPG-34, PPG-51, PPG-52, and PPG-69, as well as tripropylene glycol, are included.
Propylene glycol is an aliphatic alcohol that is manufactured as a reaction product of propylene oxide and water. Tripropylene glycol is manufactured by sequential addition of propylene oxide to PG and is only of 3 chain length. Polypropylene glycols are manufactured by the addition of propylene oxide to dipropylene glycol and have average chain lengths of their “n” value; for example, PPG-3 would have an average chain length of 3. The USP grade PG (used in cosmetics) manufactured by Dow contains diethylene glycol and ethylene glycol at concentrations that are nondetectable (quantification limit of 0.008% wt/wt). Two companies reported that the concentration of propylene oxide in PPGs used to make finished products is ≤10 ppm propylene oxide.
In 1984, PG was reported to the FDA as being used in 5676 cosmetic formulations at concentrations of 0% to >50%. As of 2009, the use of PG has increased significantly, and PG was reported to FDA as being used in 9747 cosmetic formulations. Concentration of use has also increased, with bath oil/tablet/salt preparations containing up to 99% PG and leave-on formulations, including deodorants, containing up to 73% PG. The PPGs are not as widely used as PG, and the maximum reported concentration is 22%. Tripropylene glycol is used in 8 formulations, 7 of which are deodorants, at up to 22%.
Dermal penetration of PG from a ternary cosolvent solution through hairless mouse skin was 57% over a 24-hour period. Using TED-FTIR spectroscopy, it appeared that PG molecules did not reach the dermis in human skin.
Propylene glycol can act as a penetration enhancer for some chemicals and under some conditions. Often, it works synergistically with other enhancers. The mechanism by which PG enhances penetration has not been definitively identified.
Few toxic effects were seen in dosing with PG or PPGs. In an acute oral toxicity study, 3 of 3 rats dosed with 250 mg/kg and 2 of 3 rat dosed with 1000 mg/kg PPG 425 survived. All mice survived in a repeated-dose oral toxicity study in which mice were given 10% PG in drinking water for 14 days. Repeated-dose inhalation data reported some effects in rats due to PG exposure of 2.2 mg/L air for 6 hours/d, 5 days/week, for 13 weeks, but these effects were inconsistent and without dose–response trends.
Undiluted PG was less than marginally irritating to rabbit eyes In a dermal irritation study in mice, undiluted PG was minimally irritating.
Oral administration of PG did not have any adverse reproductive or developmental effects when evaluated in mice and rats at doses of ≤1600 mg/kg, rabbits at doses of ≤1230 mg/kg, or hamsters at doses of ≤1550 mg/kg. A study examining induction of cytogenetic aberrations in mice reported an increase in the frequency of premature centrosphere separation with 1300 to 5200 mg/kg PG. In zygotes from PG-dosed mice, hyperploidy was increased.
Tripropylene glycol, ≤10 000 µg/plate, was not mutagenic in an Ames assay.
Combined exposure to PG and oleic acid synergistically enhanced the dermal penetration of both compounds. Addition of PG to an isopropanol vehicle enhanced the irritant reactions of benzoic acid; maximal enhancement was seen with 5% PG.
The dermal irritation potentials of deodorant formulations containing 68.06% or 69.15% PG were evaluated in an SIOPT and compared to a reference in-use control formulation; the formulations containing PG were no more irritating or even less irritating than the reference control. Use studies of deodorant formulations containing 35% to 73% PG did not report any potential for eliciting irritation or sensitization. Deodorant formulations containing 69.15% or 86% PG did not induce sensitization reactions; however, questionable results were obtained in an RIPT of a deodorant containing 73% PG.
In a provocative study in which the sensitization potential of PG was evaluated in patients with contact dermatitis, 5% PG in petrolatum did not cause any positive reactions in a patch test. Retrospective analysis of pools of patient patch test data, mostly NACDG data, indicated that, 6.0% or less of patients tested had positive reactions to 30% aq PG. Additionally, PG (concentration not specified) did not produce a photoallergic response in a provocative photopatch test.
Discussion
The CIR Expert Panel reopened the 1994 safety assessment of PG and PPGs to address the safety of current high-use-concentrations of PG as well as to add all the PPGs currently listed in the International Cosmetic Ingredient Dictionary and Handbook. This report is intended to also address the safety of similar PPGs that may be used as cosmetic ingredients in the future.
Since tripropylene glycol is similar to PG and the PPGs, its safety can be supported by the existing data and, therefore, the Panel included tripropylene glycol in this safety assessment.
Propylene oxide is used in the manufacture of PPGs but should not appear in cosmetic formulations because of safety concerns. The Panel expects that PPGs contain ≤10 ppm propylene oxide, ensuring the safety of formulations in which PPGs are used.
Both PG and PPGs were not considered to be acute or chronic toxicants in oral or dermal studies, were not genotoxic or carcinogenic, and were not reproductive or developmental toxicants, supporting that their use in cosmetics would be safe in regard to these end points.
At the time of the original safety assessment, a concentration limit of 50% PG and PPGs was established based on the results of existing irritation and sensitization studies. The potential for skin irritation was especially of concern under occlusive conditions, and this potential could be concentration dependent. An RIPT performed using a stick antiperspirant containing 86% PG produced no evidence of sensitization. Additionally, use studies of deodorant sticks containing 35% to 73% PG did not demonstrate a potential for eliciting dermal irritation or sensitization. Therefore, the Panel determined that PG would not present a sensitization risk at the concentrations currently in use.
The Expert Panel did note that PG may act as a penetration enhancer. Some cosmetic ingredients have been regarded as safe based on the fact that they do not penetrate the skin. If PG enhances penetration of such ingredients, then they should not exist together in formulation.
Additionally, PG is used in aerosols. The potential adverse effects of inhaled aerosols depend on the specific chemical species, the concentration and the duration of the exposure, and their site of deposition within the respiratory system. In practice, aerosols should have at least 99% of their particle diameters in the 10 to 110 µm range and the mean particle diameter in a typical aerosol spray has been reported as ~38 µm. Particles with an aerodynamic diameter of ≤10 µm are respirable. In the absence of significant inhalation toxicity data, the Panel determined that PG can be used safely in hair sprays because the product particle size is not respirable.
The CIR Expert Panel, as noted earlier, considers that the available data for PPG-3 through PPG-69 would extend to any PPG-n to be used in cosmetics in the future. There are no concerns regarding residual monomers in PPGs. If the “n” in PPG-n is 32, for example, ample evidence suggests that its toxicity would be no different from PPG-30 or PPG-33. If the “n” is 120, the ingredient would be sufficiently large so that no dermal penetration would be possible.
Conclusion
The CIR Expert Panel concluded that PG, tripropylene glycol, PPG-3, -7, -9, -12, -13, -15, -16, -17, 20, -26, -30, -33, -34, -51, -52, -69, and any PPG ≥3 are safe as cosmetic ingredients in the present practices of use and concentration as described in this safety assessment when formulated to be nonirritating. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in the group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
