Abstract
Information on the effects of propylene glycol (PG) and vegetable glycerin (VG) and on cytotoxicity and subsequent activation of the biological mediators is limited in periodontal diseases. This study analyzes the effect of unflavored PG/VG alone or in combination with nicotine on gingival epithelial cells. The cells were exposed to different PG/VG (± nicotine) concentrations for 24 h and cytotoxicity was evaluated by calorimetric 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromid assay. The expressions of interleukin (IL)-6, IL-8, and matrix metalloproteinases (MMPs)-9 were measured using an enzyme-linked immunosorbent assay and a western blotting. Stimulation with PG/VG mixtures reduced cell viability compared to nonexposed controls (p < 0.05). Adding PG/VG increased the levels of IL-6, IL-8, and MMP-9, and the amount of PG had more biological impact compared to the VG amount. The nicotine augmented this effect compared to its nicotine-free counterparts. In western blotting result, MMP-9 was clearly activated in almost all samples. These findings suggest that the main constituents PG/VG are cytotoxic and able to induce biological response in gingival cells in vitro. Despite being advertised as less harmful than conventional cigarettes, electronic cigarette liquid pose certain risks on periodontal cells. Awareness about the effects of electronic cigarettes on periodontal diseases must be increased.
Introduction
Periodontal disease is a situation in which the supporting structures of the teeth are destroyed. It is a complex disease of the epithelial layer and the surrounding connective tissue, 1 in which the smoking is the most significant and modifiable risk factor. 2 Cigarette smoking and nicotine addiction cause changes to tobacco products because of problems with dental and tobacco-related systemic illnesses. 3 Electronic cigarettes are an alternative delivery device for nicotine intake that is rapidly gaining popularity as a combustion-free alternative. Standard electronic cigarettes feature an aerosol by heating an electronic cigarette liquid, which contains propylene glycol (PG; C3H8O2, propane-1,2-diol), vegetable glycerin (VG; C3H8O3, propane-1,2,3-triol), nicotine (C10H14N2), flavoring agents, and other additives. 4 The electronic cigarette liquid seems to contain fewer known toxins, such as acetaldehyde, toluene, or formaldehydes; however, electronic cigarette liquid mostly contains nicotine as the main addictive agent. 5 In addition to nicotine, the flavoring agents and main constituents PG and VG negatively affect the tissue composition and cause tissue destruction in periodontal diseases via impairment of the immune system. 6 PG and VG are two of the base highest-volume constituents, which keep other constituents in suspension and provide the users with a similar feeling as cigarette smoking. 4 Although the Food and Drug Administration (FDA) has classified PG and VG as safe for oral consumption, 7 the effects of inhalation after repeated use and particularly direct contact on the cells are unclear. Recent studies report that thermal decomposition of PG and VG produces reactive carbonyls, including acrolein, formaldehyde, and acetaldehyde, which are toxic to the respiratory system. 8,9
Electronic cigarette ingredient components, formulations, and emissions have been intensively analyzed. 10 Unfortunately, there is a general lack of ingredient specifications and source of flavors, and commercially available brands cause different changes in cell viability. 11 Similarly, in vitro cytotoxicity tests showed different results for flavored liquids from one brand compared to nonflavored liquids, 12 and the related data become even more different when delivery method was defined using different routes of exposure. 13 However, the lack of data on the direct contact of PG and VG may hide other unexpected harmful effect of electronic cigarettes.
The immediate effect of electronic cigarette liquids is the change in immune response. 6 A network of many cytokines in the immune system is responsible for regulation of the cells. The cell-based immune response is modulated by cytokines, which are essential for initiation and maintenance of inflammatory and immune responses in periodontal tissue destruction. 14 Increased expression of various cytokines and association of smoking status with chronic infectious diseases is one of the most important components of the host defense mechanisms. In the context of periodontal tissue destruction, interleukin (IL)-6 and IL-8 are likely to be important, because they are both involved in the pathologic processes of both soft and hard tissues 15 that support periodontal tissues along with matrix metalloproteinases (MMPs). 16 MMPs are the main extracellular matrix enzymes that are involved in collagen degradation, and they are divided into five subfamilies, including the gelatinases, such as MMP-9. 16 During an inflammatory response, cytokines and MMPs are not isolated from each other. The fine balance between ILs and MMPs orchestrates the pathologic situation. 17 Therefore, because environmental factors are critical influencers of the cellular balance and response, critical questions about the safety of electronic cigarettes are raised. Taking into account the considerable variation among electronic cigarette liquid ingredients, 18 understanding the impact of in vitro exposure to unflavored plain electronic cigarette liquids on gingival epithelial cells will allow us to analyze the functional aspects on periodontal tissues.
Prior reports have extensively shown that nicotine disrupts cell function and their immune response in the short- or long-term manner. However, with varying amounts of PG and VG, the basic components of electronic cigarettes hide their own unique toxic effects and little is known about how they affect the gingival cells. To maintain how PG/VG effect the gingival cells, in this report we investigated the impact of PG/VG in the absence of nicotine or with the costimulation of nicotine. This in vitro study aims to analyze gingival epithelial cell viability upon stimulation with PG + VG ± nicotine, and IL-6, IL-8, and MMP-9 expression levels will be measured in the absence of certain well-known toxic compounds.
Material and methods
Electronic cigarette liquid mixture
The stock solutions of electronic cigarette liquid were prepared using PG (Sigma-Aldrich, St. Louis, Missouri, USA), VG (Sigma-Aldrich), and nicotine (Sigma-Aldrich) as detailed composition in Table 1. Because of the limitless variability in both PG/VG ratio and nicotine content, we set up our experiments in three different PG/VG ratio combinations as 20/80, 50/50, and 80/20, respectively. Experiments were performed by adding a final concentration of 25% electronic cigarette liquid diluted in cell culture medium.
Mixtures of the PG and VG.

PG: propylene glycol; VG: vegetable glycerin; PBS: phosphate-buffered saline.
Cell culture stimulation
An epithelial cell line was a generous gift from former Professor Ismo Virtanen, University of Helsinki, Department of Anatomy, Finland. The cells originally obtained from healthy human gingival biopsy samples. 19 The human gingival epithelial cells were maintained in keratinocyte growth medium 2 (Dulbecco’s modified Eagle’s medium (Gibco, Life Technologies, Paisley, UK) containing the supplement provided by the manufacturer. The medium was changed every 4–5 days and the cells were grown in 10 cm cell culture plates. After the cells were reached into confluence, they were transferred to six-well plates at a concentration of 2 × 106 cells/well and incubated in a 5% CO2 incubator at 37°C. After keeping the cells in serum-free medium for 2 days, the cells were treated with or without electronic cigarette liquids. Control and conditioned media were collected after 24 h and stored at −80°C. Cultures were performed in triplicate in each three biological replicate experiments and the concentrations of IL-6 and IL-8 and MMP-9 in the supernatants were determined with enzyme-linked immunosorbent assay (ELISA) and western blotting.
Cell viability
The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromid (MTT) test was used to analyze the cell viability. The substance used for the MTT test was MTT salt, which turns into a blue formazan product due to the viable mitochondria in active cells. The gingival epithelial cells were seeded at a density of 8 × 104 cells/ml into 96-well plates. The gingival epithelial cells were cultured at 37°C in 5% CO2 for 24 h. After washing the cells, the serum-free medium was added and the cells were kept for 2 days in serum-free medium. The cells were exposed to various concentrations of PG/VG either alone or PG/VG with certain amount of nicotine for 24 h. After removing the medium and washing the cells, 10 µg/ml of MTT (the American Type Culture Collection) solution was added, and the incubation was continued at 37°C in 5% CO2 for 4 h in serum-reduced medium without phenol red. After the incubation medium was removed, 400 µl of isopropanol with 0.04 N HCl was added to each well. The absorbance was measured at 570 nm.
Enzyme-linked immunosorbent assay
The ELISA was performed with commercial colorimetric sandwich ELISA kits in accordance with the manufacturer’s instructions (Quantikine; R&D Systems Inc., Minneapolis, MN, USA). The kit provided the minimum detection limits as 0.7 pg/ml, 1.5 pg/ml, and 0.2 ng/ml for IL-6, IL-8, and MMP-9, respectively. In summary, the provided standards and samples were distributed into the wells, which had been precoated with IL-6, IL-8, and MMP-9 specific antibody (Quantikine). After waiting and washing, an enzyme-linked biotinylated antibody specific for IL-6, IL-8, and MMP-9 were added to the wells. After removing any unbound antibody-enzyme reagent, a substrate solution was added to each well. The substrate solution was prepared by mixing equal amounts of stabilized hydrogen peroxide and tetramethylbenzidine which were provided in the kit. The reaction was terminated by adding stop solution and absorbance was determined in an automated spectrophometer. The intensity of the color was read at 450 nm.
Western blotting
Molecular weight of MMP-9 from cell culture supernatants were analyzed by western blotting. Briefly, the samples were boiled for 5 min in sodium dodecyl sulfate gel loading buffer. Electrophoresis was performed in 10% polyacrylamide gel loaded 10 µL supernatant in each lane. Following electrophoresis, the gel was blotted onto nitrocellulose membrane (Bio-Rad Laboratories, Hercules, California, USA). The nitrocellulose membrane was blocked overnight using 3% bovine serum albumin (BSA) in tris-buffered saline. The next day the membrane was washed in a washing buffer containing 0.1% Tween 20, 50 mM Tris-HCl, 0.5 M NaCl, pH 7.5. Following the washes, the membrane was incubated for 1 h in 1:400 rabbit antihuman MMP-9 antibody (Cat# 38898 Abcam, Cambridge, MA, USA) in washing buffer containing 2% BSA. This was followed with washes and incubation for 1 h with the alkaline phosphatase-conjugated goat antirabbit Immunoglobulin G at +22°C (1:5000 in washing buffer containing 2% BSA, Cat# 111-005-003, Jackson Immunoresearch Laboratories). The membrane was washed with washing buffer for 30 min before alkaline phosphatase-binding sites were revealed in color development solution (Alkaline Phosphatase Conjugate Substrate Kit, Bio-Rad Laboratories) containing a mixture of 5-bromo-4- chloro-3-indolyl phosphate and nitroblue tetrazolium chromogen for 30 min. After the reaction was stopped, the evaluation was performed using a Bio-Rad Model GS-700 Imaging Densitometerand Molecular Analyst/PC program (Bio-Rad Laboratories).
Statistical analysis
Statistical significances were calculated by one-way analysis of variance with Bonferroni’s posttests for multiple comparisons. The differences among the mean values of different groups were assessed. Data are expressed as the mean ± standard deviation. Statistical calculations were performed by using GraphPad Prism version 5.00 (GraphPad Software, Inc., La Jolla, California, USA). p Values <0.05 were considered statistically significant. Data are representative of six independent experiments, with technical triplicate cultures per experiment (N = 6, n = 3).
Results
Effect of electronic cigarette liquid on cell viability
The viability of gingival epithelial cells exposed to different PG/VG concentrations were analyzed. All results at 24 h indicate the presence of cytotoxicity upon stimulation with electronic cigarette liquid. The percentages of gingival epithelial cell survival are presented in Figure 1. The addition of any stimulant decreased the viability while indicating that in the nicotine-free PG/VG samples, cytotoxicities were less compared to their counterparts with nicotine (p < 0.001). The addition of nicotine significantly decreased the survival percentage of all epithelial cells. The toxicity level was proportional to PG amounts of the mixture. The highest cell viability decrease was observed at the highest PG amount with nicotine (Figure 1).

Cell viability assay by MTT shows reduction in viability with variable concentrations of PG/VG or plus nicotine in gingival epithelial cell cultures in vitro. Mean ± SDis shown. PG: propylene glycol; VG: vegetable glycerin; Nic.: nicotine; NEG: negative; NS: not significant; ANOVA: analysis of variance; SD: standard deviation; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromid. *p < 0.05; **
Induction of IL-6, IL-8, and MMP-9 in human gingival epithelial cells upon stimulation with PG, VG, and nicotine
The all PG/VG-containing mixtures with nicotine or without nicotine showed significant cytokine and MMP expression compared to nonexposed samples (Tables 2 to 4). Although, the gradually changed PG/VG concentrations showed an increasing tendency for MMP and cytokine productions; the significant differences were seen between 20/80 and 80/20 amounts for nicotine-free samples (p < 0.05). Upon stimulation with PG/VG-containing mixtures, IL-6 expression presented a slight difference between the columns. There was not much difference between the 20/80 and 50/50 column and only a modest increase was observed when compared to the 80/20 column. For IL-8 expression, the effect of PG/VG caused a slight increase between 20/80 and 50/50 column and similarly a small increase between 50/50 and 80/20 columns. Nicotine-containing PG/VG mixtures increased IL-6, IL-8, and MMP-9 compared to their nicotine-free concentrations significantly (p < 0.001; Figures 2 to 4).
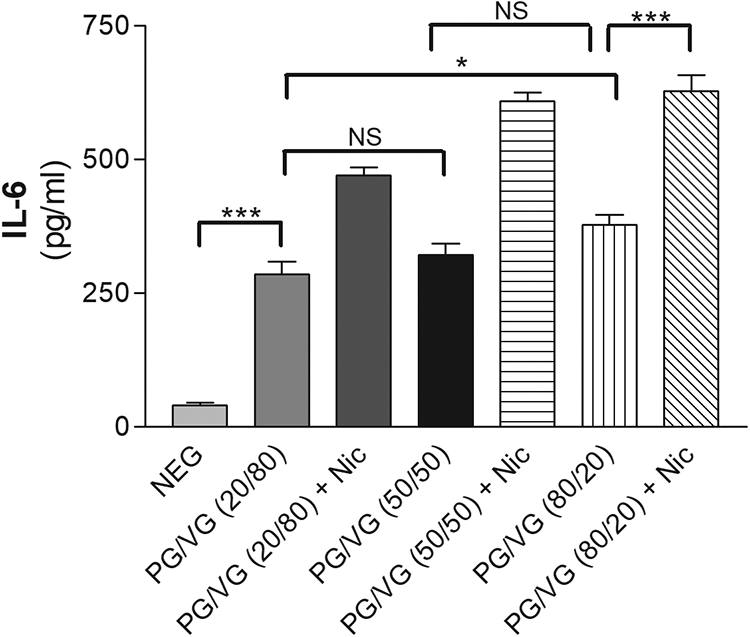
Effect of PG/VG or plus nicotine on induction of IL-6 concentration in gingival epithelial cell cultures in vitro by ELISA. Mean ± SD is shown. PG: propylene glycol; VG: vegetable glycerin; Nic.: nicotine; NEG: negative; NS: not significant; IL: interleukin; SD: standard deviation; ELISA: enzyme-linked immunosorbent assay; ANOVA: analysis of variance; . *p < 0.05; **p < 0.01; ***p < 0.001, analyzes by ANOVA.

Effect of PG/VG or plus nicotine on induction of IL-8 concentration in gingival epithelial cell cultures in vitro by ELISA. Mean ± SD is shown. PG: propylene glycol; VG: vegetable glycerin; Nic.: nicotine; NEG: negative; NS: not significant; IL: interleukin; SD: standard deviation; ELISA: enzyme-linked immunosorbent assay; ANOVA: analysis of variance. *p < 0.05; **p < 0.01; ***p < 0.001, analyzes by ANOVA.

Effect of PG/VG or plus nicotine on induction of MMP-9 concentration in gingival epithelial cell cultures in vitro by ELISA. Mean ± SD is shown. PG: propylene glycol; VG: vegetable glycerin; Nic.: nicotine; NEG: negative; NS: not significant; SD: standard deviation; MMP: matrix metalloproteinase; ELISA: enzyme-linked immunosorbent assay; ANOVA: analysis of variance. *p < 0.05; **p < 0.01; ***p < 0.001, analyzes by ANOVA.
Effect of PG/VG or plus nicotine on induction of IL-6 concentration in gingival epithelial cell cultures in vitro by ELISA.

PG: propylene glycol; VG: vegetable glycerin; IL: interleukin; SD: standard deviation; ELISA: enzyme-linked immunosorbent assay; Nic.: nicotine; NEG: negative.
Effect of PG/VG or plus nicotine on induction of IL-8 concentration in gingival epithelial cell cultures in vitro by ELISA.

PG: propylene glycol; VG: vegetable glycerin; SD: standard deviation; ELISA: enzyme-linked immunosorbent assay; IL: interleukin; Nic.: nicotine; NEG: negative.
Effect of PG/VG or plus nicotine on induction of MMP-9 concentration in gingival epithelial cell cultures in vitro by ELISA.

PG: propylene glycol; VG: vegetable glycerin; SD: standard deviation; MMP: matrix metalloproteinase; ELISA: enzyme-linked immunosorbent assay; Nic.: nicotine; NEG: negative.
Proteinase in gingival epithelial cells
The effect of electronic cigarette liquid mixture is shown in Figure 5. Western blotting analysis revealed that all supernatant samples produced MMP-9 in its latent 92 kDa proform. Upon stimulation with PG/VG mixtures, 86-kDa-molecular-weight proteolytically processed active form was started to be seen in exposed samples (Figure 5).

Western blot analysis of MMP-9. Characterization of latent (92 kDa) and active (86 kDa) molecular forms of MMP-9 in gingival epithelial cell cultures in vitro. MMP: matrix metalloproteinase.
Discussion
The cytotoxicity effect of PG and VG has been studied in the context of electronic cigarette liquids containing different proportions of flavoring agents. In this study, high-quality pure PG and VG mixtures were used to expose the gingival epithelial cells, leading to an analysis without confounding factors. 20 All PG and VG amounts were shown to be enough to cause cytotoxicity in gingival epithelial cells and the addition of nicotine increased this effect.
Despite the large and increasing safety concerns of electronic cigarettes, no objective results have determined the effect of electronic cigarettes on oral tissues. According to a recent study, electronic cigarette aerosols increase the adhesion of plaque bacteria, which, in turn, promotes the biofilm formation. 21 Furthermore, increased gingival bleeding and periodontal pockets were found to be positively related with the use of electronic cigarettes. 22 There are thousands of different electronic cigarette formulations with over 7700 available electronic cigarette liquid flavors and it is not possible to test all of the liquids. 18 Electronic cigarette liquids are customized by varying amounts of nicotine, PG, and VG and flavoring ingredients. Due to the easily available substances, electronic cigarettes increasingly commonly are prepared individually in a large number of different compositions as well. In this research, we validated a simple and basic system that demonstrated dose-dependent cytotoxicity of PG and VG with or without nicotine. Although PG and VG are categorized on the FDA’s Generally Recognized As Safe list, 7 consistent with earlier research, 23 the cytotoxicity profile of PG and VG that have direct contact with gingival epithelial cells seems to exert a different cytotoxicity profile in combination with nicotine.
This cytotoxicity eventually leads to the release of the cytokines IL-6 and IL-8, which is, therefore, relevant to the development of periodontal tissue destruction. 24 Notably, cell viability results showed that exposure to 80/20 (PG/VG) plus nicotine led to significant, and the highest, cell death in this experiment. Thus, recognition of a higher cytotoxicity level with an increase in the PG content confirms that PG is more cytotoxic to gingival epithelial cells and that addition of nicotine increases the cytotoxicity of all mixtures. 23 The results are consistent with those of previous studies where an increase in inflammatory mediators (e.g. IL-8) was seen after exposure of gingival tissues to PG/VG. 25
Both PG and VG belong to the alcohol chemical class. In electronic cigarette liquids, VG is commonly used to provide a thick sensation to the vapor, whereas PG provides a similar sensation experience as tobacco smoking. Thus, it is not possible to standardize a certain VG or PG that will be mostly preferred by the users. 7 The groups in this study contain different proportions of PG/VG and/or nicotine because the electronic cigarette liquids may or may not contain nicotine. 26 In the literature, many studies have reported the effects of nicotine. However, there is only limited data on how nicotine exposure promotes cell proliferation or survival, whereas conversely, many researchers have claimed that nicotine inhibits cell proliferation. 27,28 These inconsistent findings result from the different parameters that were used in the studies, such as cell type, exposure type, or presence of nicotinic receptors for specific cell types. 29,30 However, based on previous results, the cytotoxic effect of PG and VG most probably results from the high osmolarity because gene expression analysis in VG/PG mixtures showed fluctuations in the osmotic stress and apoptosis networks. 23 Similar results were seen in organotypic human bronchial epithelial cells, in which the aerosol solution of a PG/VG mixture perturbed the osmotic stress network with or without nicotine. 31 The current study revealed that, although nicotine is the critical driver of cytotoxicity in gingival epithelial cells, pure nonvaped PG/VG mixture adversely affects cell viability. Because electronic cigarette vapor condensate is even more toxic than nonvaped electronic cigarette liquids, 32 there will be a dramatic increase in cytotoxicity in this situation if the experiments were performed under vaped conditions. Furthermore, it is certain that serum starvation changes the cell activity. 33 However, to obtain more consistent results between the experiments, we used serum-free medium. 34 Both unstimulated cells and stimulated cells were grown in the same medium. We wanted to check how the cells in the same medium react differently to the electronic cigarette liquid between the groups. The specific cause of the cell viability was not determined in this study. Furthermore, as a control of the study, the cell morphology was monitored under the microscope, which we did not realize any visible change compared to cells growing in medium with serum.
In the nicotine-free electronic cigarette liquid experiments, side effects such as the release of inflammatory markers, gene expression, and DNA damage have been detected in various cell types, 20 but the effect of PG and VG on gingival epithelial cells has not been studied as much. Therefore, in this study, the observed effect of PG/VG ± nicotine and the resulting higher release IL-6, IL-8, and MMP-9 indicate a direct or indirect role of electronic cigarette liquids during gingival inflammation, because MMPs and cytokines are important signal communication molecules that promote turnover of extracellular matrix molecules in the periodontal supporting tissues. 16 Smoking causes activation and recruitment of inflammatory cells, which releases IL-6 and IL-8 from the epithelial cells. 35 Because the epithelial cells are the first barrier for external stimuli, 16 electronic cigarette-triggered IL-6 and IL-8 expression is critical for the innate defense system in the periodontal gingiva. 24 IL-6 and IL-8 are two important markers for cellular activation, which participate actively in the local inflammatory process. 36 IL-6 protects against tissue damage via mediating the innate immune system. 37 Thus, IL-6 consequently stimulates the production of IL-8, which functions as a local chemoattractant for polymorphonuclear leukocytes. 38 Overall, IL-6 and IL-8 modulate MMP-9 production by the gingival epithelium. 16 The role of these cytokines as potent inducers of MMP-9 was reflected in both ELISA and western blot results that showed a significant positive correlation of MMP-9 expression with IL-6 and IL-8 expression in gingival epithelial cells, which resulted from the electronic cigarette liquid component cytotoxicity. The correlation suggests that, in epithelial cells, increased IL-6 and IL-8 expression in the gingival epithelium is caused by the PG/VG, which stimulates MMP-9 expression in the epithelial cells. This further contributes to gingival tissue degradation. 16
IL-6 and IL-8 present slight differences between columns; however, from the point of upregulated MMP-9, it is possible to conclude the simultaneous activation of the epithelial cells under the effect of PG and VG. One possible reason for the cellular response may be increased local and extrinsic stimulation activity. Indeed, the consequence of inflammatory and infectious events such as osmotic pressure-activated epithelial cells released enough IL-6 and IL-8, which in an autocrine and paracrine manner stimulated and upregulated MMP-9. This indicates that in the presence of an additional effect like osmotic pressure, epithelial cells responded to slight high levels of IL-6 and IL-8 with a different release pattern of MMP-9 which leads to challenge the protective functions. In electronic cigarettes, PG has a capacity to solubilize nicotine and other ingredients, which consequently lead to affect osmotic pressure activity of the cell environment. Due to the concentration changes, the gradient pressure exists across the epithelial cells. It is thus seen that the more amount of PG is added, the more osmotic pressure acts at cell membrane. In a similar way, in a recent study, the emulsifying properties of PG were justified to initiate the structural perturbations, which led to produce the disruption in lipoprotein homeostasis in alveolar macrophages and epithelial cells. 39 Together, although there is a small increase between the groups, with effects like increased pressure or disruption of structural homeostasis, MMP-9 expression is released indicating simultaneous activation of the epithelial cells, since MMP-9 expression is inducible by IL-6 and IL-8, and MMP-9 positivity indicates an activated state of the cell. In an earlier study, following stimulation with periopathogenic bacteria, IL-6 secretion was induced earlier than the IL-8 secretion in epithelial cells. 40 Such kind of comment cannot be substantiated for the current study. However, after exposure to electronic cigarette liquid, higher amounts of IL-6 and IL-8 were analyzed than those in the nonexposed controls and showed an increasing tendency with the addition of PG. It can be speculated that the secretion of IL-6 and IL-8 and finally MMP-9 with an increasing tendency hint that electronic cigarette has a dose-dependent effect on gingival epithelial cells.
Although MMP-9 is particularly produced from polymorphonuclear leucocyte, gingival epithelial cells also produce MMP-9 that destroys the periodontal tissue. 41 Therefore, the direct and strong correlation of IL-6 and IL-8 with MMP-9 suggests that electronic cigarette liquid PG, VG, and nicotine trigger the initiation and progression of extracellular matrix destruction. As shown by western blotting, the MMP-9 latent form was upregulated and secreted in all samples, and it was subsequently activated by proteolytic cleavage of an inhibitory domain upon stimulation with an electronic cigarette liquid. 42 After stimulation with PG/VG (nicotine), an increase in the active form of the enzyme indicated the enzymatic activation of MMP-9. Thus, although physiological activity was the reason for induction of the MMP-9 protein, administration of all electronic cigarette liquid contents seemed to be the reason for conversion to activated MMP-9. 2 This is supported by the clearly observed active MMP-9 bands in all samples except PG/VG (20/80), which may result from the rather low secretion. This PG/VG concentration showed the lowest effect of IL-6 and IL-8, which correlates with less disease severity in periodontal diseases. Although western blotting and ELISA are both based on immunodetection, the advantages and disadvantages of these methods largely depend the purpose of interest and one of the advantages of western blotting is that it is less likely to give false-positive results. In our study, both ELISA and western blotting were used to detect the active and the proform of MMP-9 in which both gave similar results. This verified result clearly presents that the electronic cigarette liquid substances make different MMP-9 expression compared to the same matched unstimulated cells. In further studies, the addition of zymography will reveal the amount of the active enzyme for MMP-9.
Currently, the pathobiological effects of electronic cigarette liquid constituents on tissue destruction are not fully understood. It is critical to explain the degradation of the extracellular matrix. A large amount of data regarding possible downstream regulation exists. Drawing a definitive conclusion about the mechanism of nicotine is not easy, but a comprehensive analysis of the abundant nicotinic acetylcholine receptor-α7 subunit (α7 nAChR) expression in oral epithelial cells has been performed. 43 Activation of α7nAChR leads to downregulation of pro-inflammatory cytokine expression, which causes different downstream regulation. 44 Inflammatory cytokines such as IL-8 and IL-1β are produced via the α7nAChR/nuclear factor kappa B (NF-κB) pathway in periodontal ligament cells upon stimulation with nicotine. 45 Additionally, in a human leukemia cell line, nicotine increases MMP-9 release in a dose- and α7nAChR-dependent manner. 46 Similarly, NF-κB is a critical regulator of inflammatory factors such as IL-6 and IL-8 expression in periodontal diseases through nucleotide-binding oligomerization domain-mediated NF-κB. 47 Based on these previous results and the current experimental results, we can speculate that nicotine in electronic cigarettes might increase MMP-9 expression in gingival epithelial cells, and this effect may occur through the α7nAChR and NF-κB pathways.
The primary objective of the present study was to provide information on the combination of different electronic cigarette liquid components. It is noteworthy concern that uniqueness of the liquid compositions in each brand along with the inconsistency of electronic cigarette machines and the way of vapor is hard to evaluate the final concentrations of PG, VG, and nicotine in real-life exposure. Considering the present huge critical information gap, it now appears that much of the health effects of electronic cigarettes are still unknown, which indicates a strong need of the certain standards for electronic cigarettes. Future studies are needed to identify how electronic cigarette liquid ingredients can activate molecular pathways leading to MMP activation. There are numerous types of electronic cigarette products and an enormous amount of aerosols, but a less harmful alternative to conventional cigarettes remains unknown.
Conclusion
Although the use of electronic cigarette is promoted as a safer alternative to conventional cigarettes, the present study reports the cytotoxic effect of the basic components of electronic cigarette liquid in gingival epithelial cells. Furthermore, upon exposure to PG/VG or plus nicotine, the gingival epithelial cells responded by producing IL-6, IL-8, and MMP-9. This study indicates that electronic cigarette liquid engage the local epithelial defense system as the amount of certain immune response molecules increases in the cells exposed to liquid.
Footnotes
Authors’ contributions
AB and DU proposed the research questions and the overall framework. AB performed the lab work and both authors have contributed equally to writing, analyzing, discussing, and revising the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the authors.
