Abstract
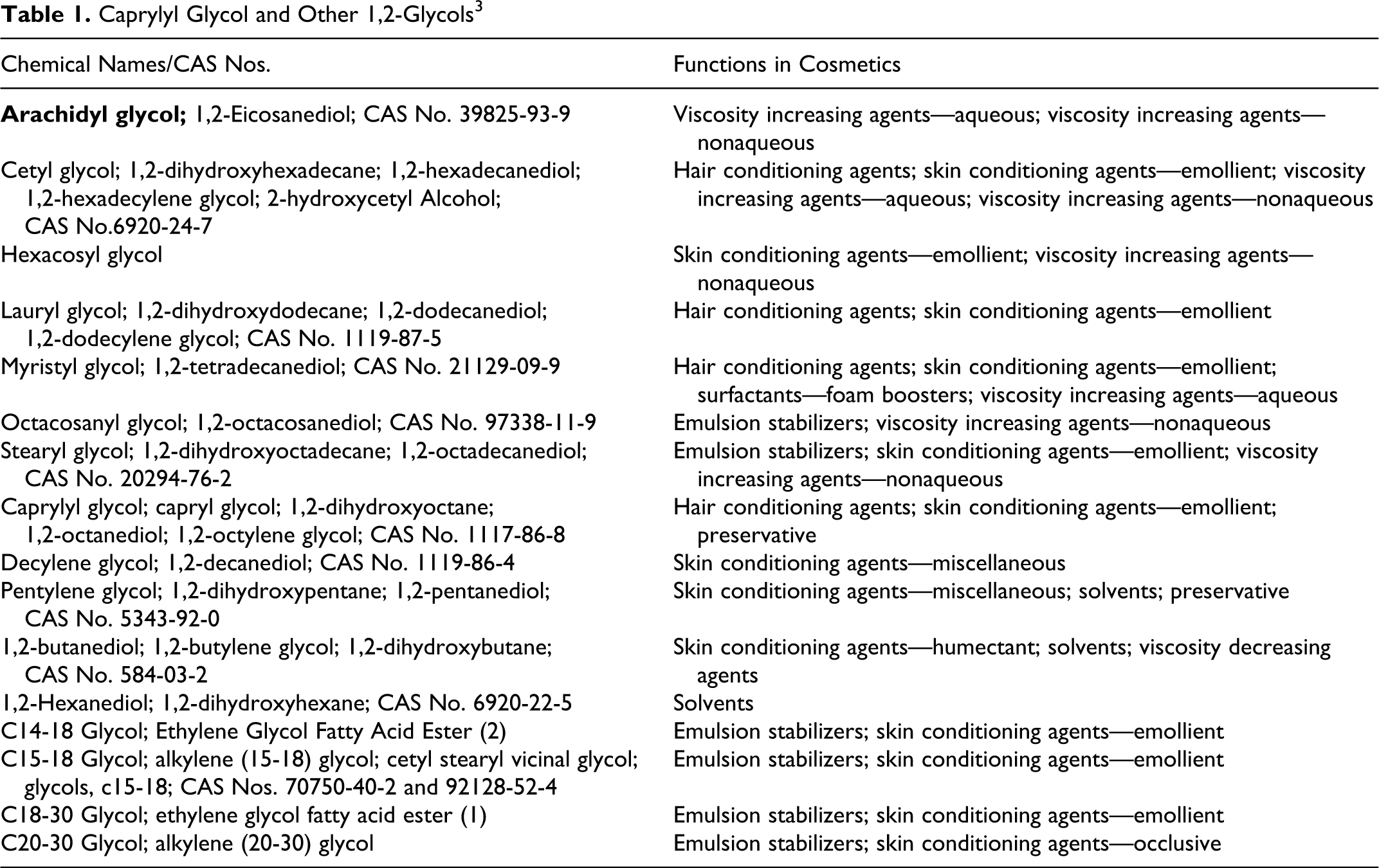
Caprylyl glycol and related 1,2-glycols are used mostly as skin and hair conditioning agents and viscosity agents in cosmetic products, and caprylyl glycol and pentylene glycol also function as cosmetic preservatives. The Cosmetic Ingredient Review (CIR) Expert Panel noted that, while these ingredients are dermally absorbed, modeling data predicted decreased skin penetration of longer chain 1,2-glycols. Because the negative oral toxicity data on shorter chain 1,2-glycols and genotoxicity data support the safety of the 1,2-glycols reviewed in this safety assessment, the Panel concluded that these ingredients are safe in the present practices of use and concentration described in this safety assessment.
Introduction
This safety assessment includes the following short chain 1,2-glycols as used in cosmetic products: caprylyl glycol, arachidyl glycol, cetyl glycol, hexacosyl glycol, lauryl glycol, myristyl glycol, octacosanyl glycol, stearyl glycol, decylene glycol, pentylene glycol, 1,2-butanediol, 1,2-hexanediol, C14-18 glycol, C15-18 glycol, C18-30 glycol, and C20-30 glycol. The 1,2-glycols are used mostly as skin and hair conditioning agents and viscosity increasing agents in cosmetic products. Caprylyl glycol and pentylene glycol are also used as preservatives. Based on the current information, 4 of the16 ingredients reviewed in this safety assessment, are being used in personal care products: caprylyl glycol, pentylene glycol, 1,2-hexanediol, and C15-18 glycol. The remaining 12 ingredients are not reported in the current use.
A Cosmetic Ingredient Review (CIR) final safety assessment on propylene glycol (PG), short-chain 1,2-glycol, and polypropylene glycols (PPGs) was published in 1994. 1 The Expert Panel concluded that PG and PPGs are safe for use in cosmetic products at concentrations up to 50.0%. In 2010, the Expert Panel issued an amended final safety assessment on PG, tripropylene glycol, and PPGs and concluded that PG, tripropylene glycol, PPG-3, -7, -9, -12, -13, -15, -16, -17, 20, -26, -30, -33, -34, -51, -52, -69, and any PPG ≥3, are safe as cosmetic ingredients in the present practices of use and concentration as described in this safety assessment when formulated to be nonirritating. 2 Data on PG from the 1994 and 2010 safety assessments are included to support the safety of 1,2-glycols reviewed this safety assessment.
Chemistry
Definition and Structure
Caprylyl glycol and other 1,2-glycols are generally defined as the compounds that conform to a structure or formula. The fundamental carbon backbone contains a hydroxyl group at the 1 and 2 positions, and the length of the carbon backbone varies from one structure to another. Additional chemical names and cosmetic ingredient functions for the ingredients reviewed in this safety assessment are included in Table 1. 3 Chemical structures are included in Figure 1.
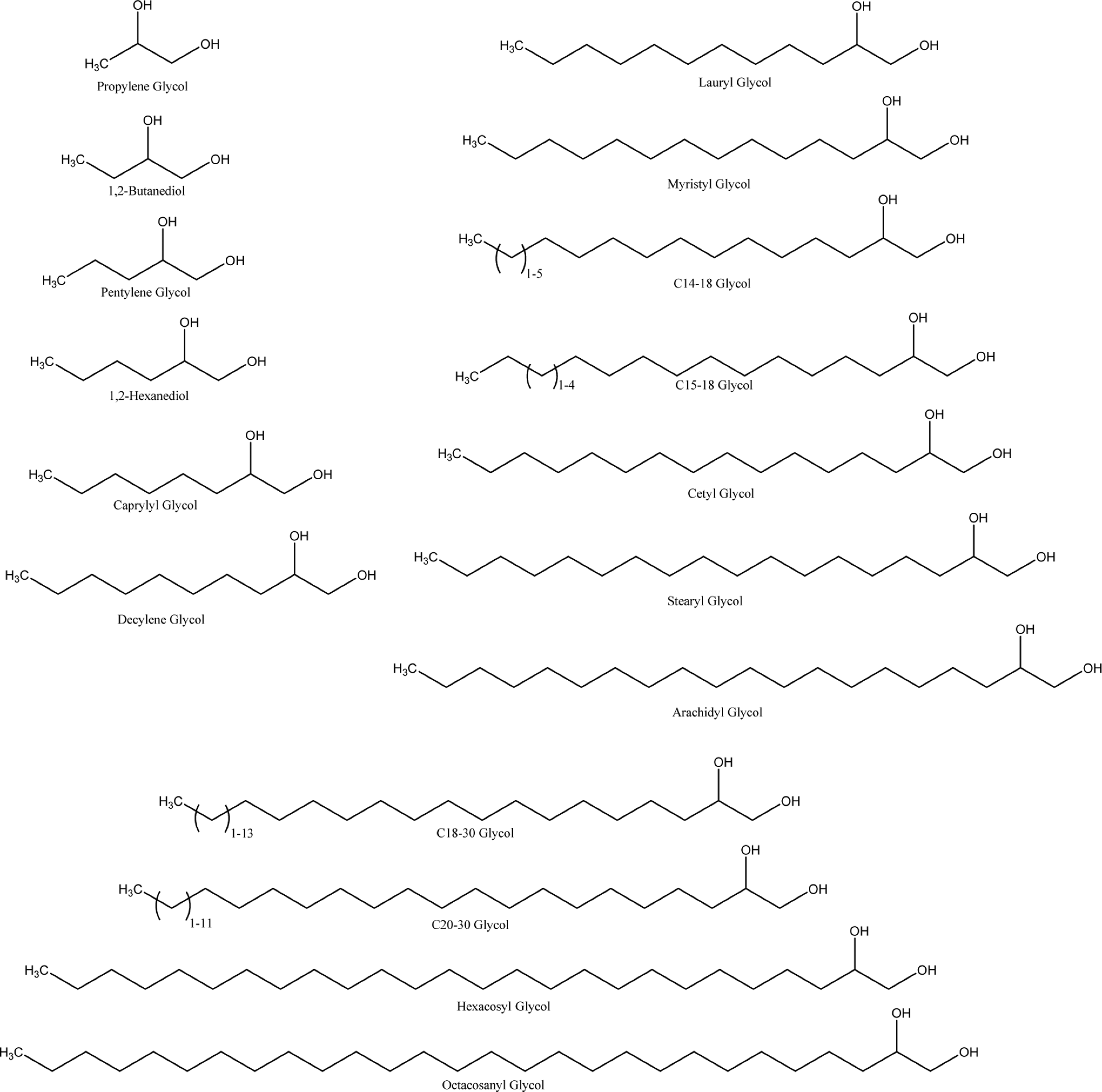
Formulas of 1,2-glycols.
Caprylyl Glycol and Other 1,2-Glycols 3
Chemical and Physical Properties
Data on the properties of caprylyl glycol, arachidyl glycol, cetyl glycol, lauryl glycol, myristyl glycol, octacosanyl glycol, stearyl glycol, decylene glycol, pentylene glycol, 1,2-butanediol, and 1,2-hexanediol are included in Table 2. The solubility of these ingredients in water ranges from highly soluble (1,2-butanediol, octanol/water partition coefficient of −0.8) to poorly soluble (octacosanyl glycol, octanol/water partition coefficient of approximately 11.9; Figure 2).
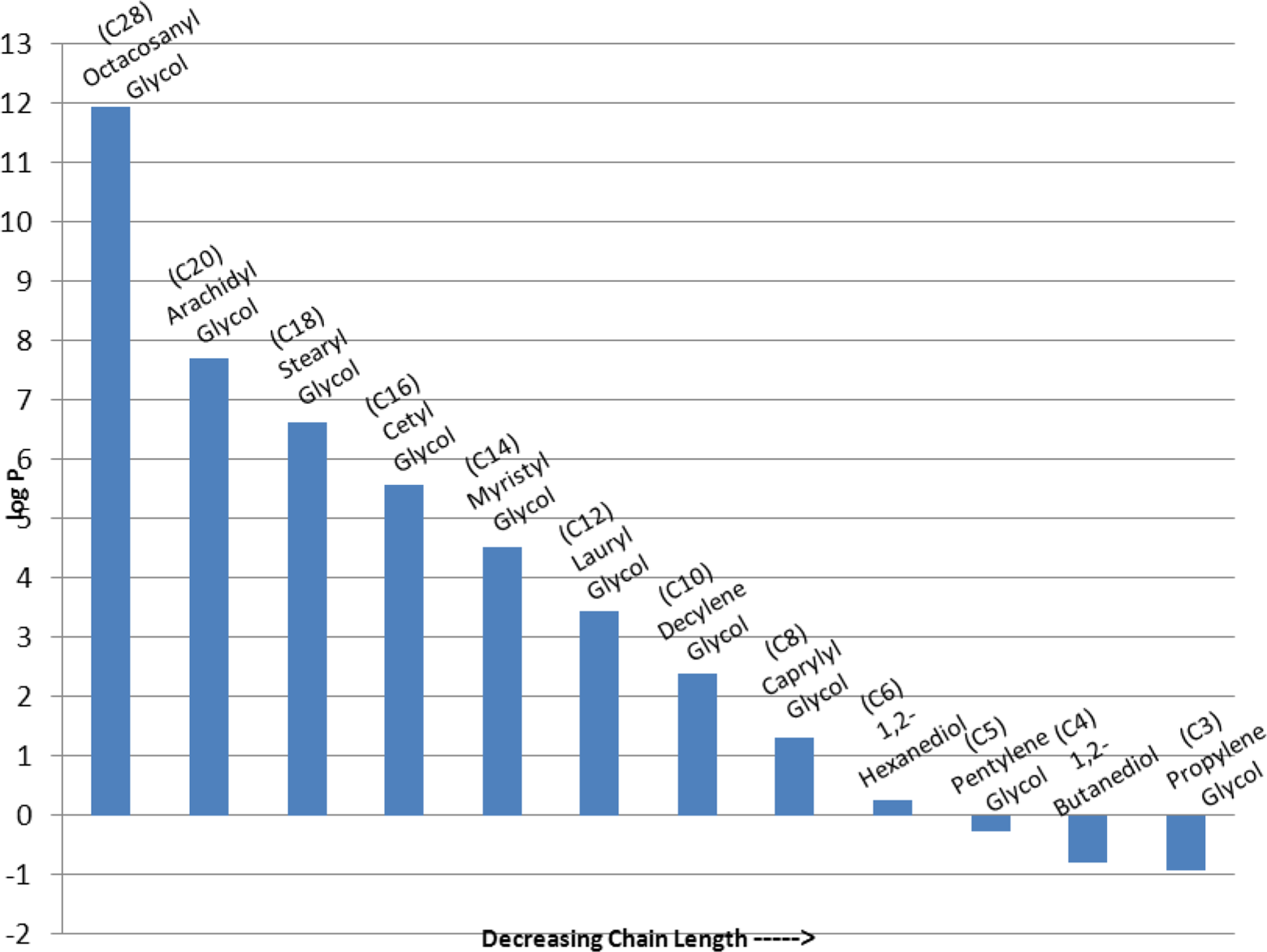
Octanol/water partitioning coefficient (log P).
No information on the chemical and physical properties of C14-18, C15-18, C18-30, and C20-30 glycols were found, but because these ingredients are mixtures of various length glycols, their chemical and physical properties are expected to reflect their individual components.
Methods of Production
Ethylene glycol, the simplest of the 1,2-glycols, is commercially synthesized via a thermal oxidation of ethylene oxide with water. 4 The commercial production of other 1,2-glycols, including those in this assessment, is commonly synthesized via catalytic oxidation of the corresponding alkene oxide or reduction of the corresponding 2-hydroxy acid.
C15-18 Glycol, for example, has been prepared via oxidation of the corresponding C15-C18 1,2-alkylene oxides (and the 1,2-alkylene oxides have been synthesized via epoxidation of the corresponding 1,2-alkenes). 5
Stearyl glycol has been prepared via the reduction of 2-hydroxyoctadecanoic acid with lithium aluminum hydride. 6 This reaction is followed by the quenching of any unchanged lithium aluminum hydride with excess ethyl acetate, filtering of salt, and subsequent drying of the resulting solution.
The production of 1,2-butanediol, much like the synthesis of ethylene glycol, is commonly carried out via a continuous reaction and distillation operation. 7
Composition/Impurities
The heavy metals specification for >98% caprylyl glycol (Dermosoft Octiol) is 5 ppm max (as Pb). 8 Decylene glycol (as SymClariol) contains 98% to 100% decylene glycol. 9 1,2-Butanediol is ≥99% pure and also contains water, 1,4-butanediol, and 1-acetoxy-2-hydroxybutane. 7
Analytical Methods
Cetyl glycol has been analyzed using silica gel thin-layer chromatography and has been identified using infrared (IR) and mass spectrometry (MS). 10,11 Decylene glycol has been analyzed via gas chromatography (GC) and has been identified using MS, IR, and NMR spectroscopy. 11,12 The GC-MS has been used in the analysis of stearyl glycol. 6
Lauryl glycol, myristyl glycol, caprylyl glycol, pentylene glycol, 1,2-butanediol, and 1,2-hexanediol have been identified using mass spectrometry and IR or NMR spectroscopy. 11
The UV absorption data on caprylyl glycol or any of the other 1,2-glycols reviewed in this safety assessment were not provided or found in the published literature. Based on the chemical formulas included in Figure 1, there is no reason to suspect that any UV absorption would be associated with these 1,2-glycols.
Reactivity
At temperatures above 90°C, 1,2-butanediol may form explosive vapor/air mixtures. 13 Additional information on the reactivity of 1,2-butanediol, in relation to the Environmental Protection Agency (EPA)-proposed national rule on the reduction of ozone formation, is included in the section on Noncosmetic Use later in the report text.
Use
Purpose in Cosmetics
Most of the ingredients reviewed in this safety assessment function as skin and hair conditioning agents and viscosity increasing agents in personal care products. 3
Scope and Extent of Use in Cosmetics
According to information supplied to Food and Drug Administration (FDA) in 2011the by industry as part of the Voluntary Cosmetic Registration Program (VCRP), the following ingredients were being used in personal care products: caprylyl glycol, decylene glycol, pentylene glycol, 1,2-hexanediol, and C15-18 glycol. 14 These data are summarized in Table 3. Independent of these data, the results of a survey of ingredient use concentrations conducted by the Personal Care Products Council in 2010, also in Table 3, indicate that three 1,2-glycols were being used at the following concentrations: caprylyl glycol (0.00003%-5%), pentylene glycol (0.001%-5%), and 1,2-hexanediol (0.00005%-10%). 15 According to FDA’s VCRP data, there was no indication that the following remaining ingredients in this safety assessment were being used in cosmetic products in 2011: arachidyl glycol, cetyl glycol, hexacosyl glycol, lauryl glycol, myristyl glycol, octacosanyl glycol, stearyl glycol, 1,2-butanediol, C14-18 glycol, C18-30 glycol, and C20-30 glycol.
Personal care products containing these ingredients may be applied to the skin, nails, or hair, or, incidentally, may come in contact with eyes and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin, nails, or hair for variable periods following application. Daily or occasional use may extend over many years.
Chemical and Physical Properties

Current Frequency and Concentration of Use According to Duration and Type of Exposurea,14,15
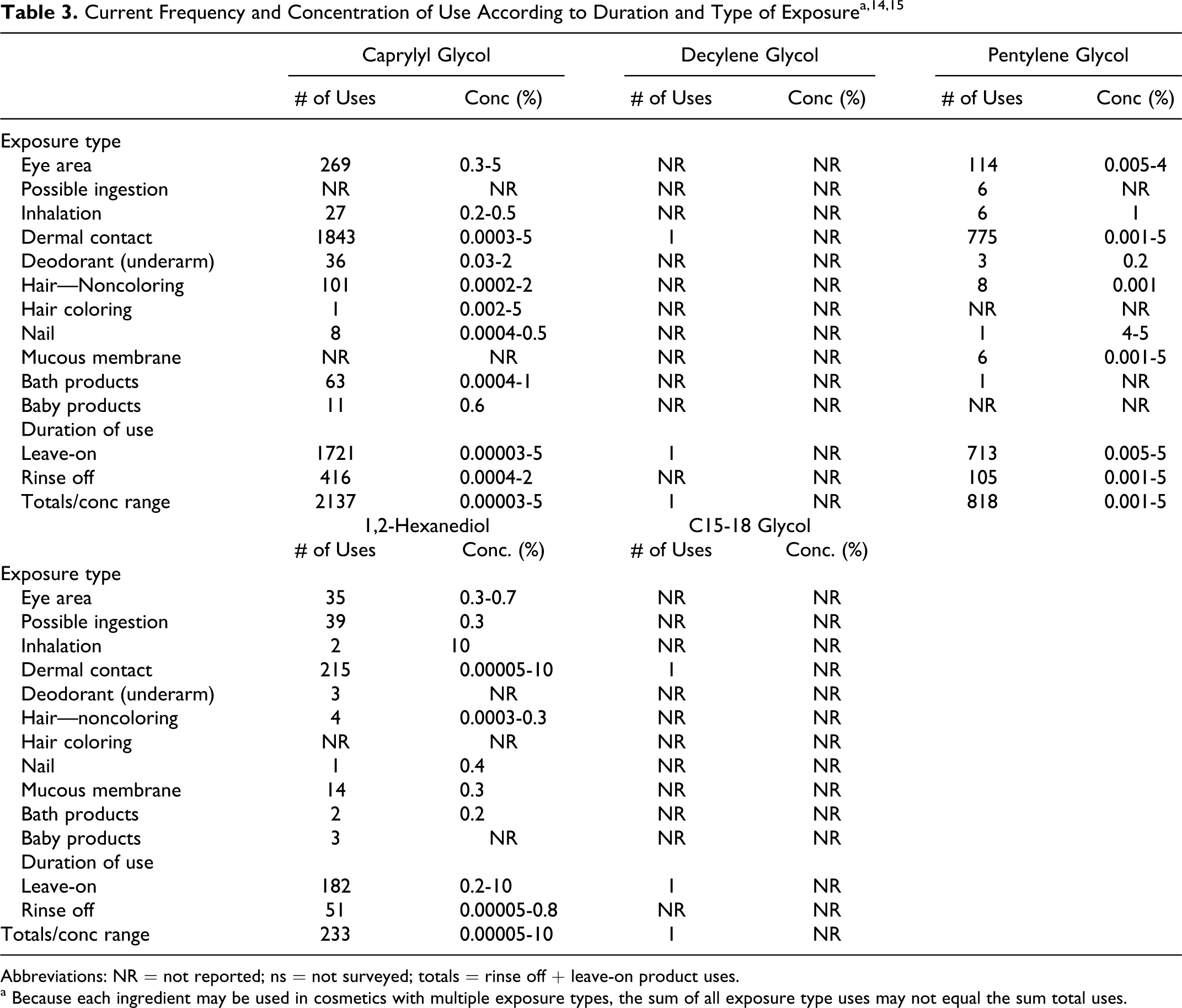
Abbreviations: NR = not reported; ns = not surveyed; totals = rinse off + leave-on product uses.
a Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
Noncosmetic Use
Caprylyl glycol
Results from a 2004 study indicate that treatment of glutaraldehyde-treated tissue with a short-chain alcohol (ethanolic buffered solution) and long-chain alcohol (caprylyl glycol) combination reduce both extractable phospholipids and the propensity for in vivo calcification. The use of glutaraldehyde-treated biological tissue in heart valve substitutes is an important option in the treatment of heart valve disease; however, the durability of these devices is limited, in part, because of tissue calcification. 16
1,2-Butanediol
The EPA lists 1,2-Butanediol as one of the reactive compounds in aerosol coatings (ie, aerosol spray paints), which contribute to ozone (O3) formation. It is listed as having a reactivity factor of 2.21 g O3/g 1,2-butanediol. Reactivity factor is defined as a measure of the change in mass of ozone formed by adding a gram of a volatile organic compound (VOC) to the ambient atmosphere. This listing of compounds, such as 1,2-butanediol, is in keeping with the EPA proposal to amend the aerosol coatings reactivity rule by adding compounds and associated reactivity factors based on petitions that were received. The EPA has concluded that a national rule based on the relative reactivity approach achieves more reduction in ozone formation than would be achieved by a mass-based approach for this specific product category. States have previously promulgated rules for aerosol spray paints based upon reductions of VOC by mass. 17
Cetyl glycol
Some colloidal nanoparticles of Sm-Co alloys are made in octyl ether using samarium acetylacetonate and dicobalt octacarbonyl as precursors in a mixture of 1,2-hexadecanediol (cetyl glycol), oleic acid, and trioctylphospine oxide. 18
Stearyl glycol
Stearyl glycol has been used as a surfactant (in octanol/water microemulsion) in a transdermal delivery system for the drug, 8-methoxypsoralen. 19
Toxicokinetics
Metabolism
Caprylyl glycol, 1,2-hexanediol, decylene glycol, and lauryl glycol
A metabolism assessment for the following 1,2-glycols (C6-C12) was provided by the Personal Care Products Council: caprylyl glycol (1,2-octanediol, C8), 1,2-hexanediol (C6), decylene glycol (1,2-decanediol, C10), and lauryl glycol (1,2-dodecanediol, C12). 20 Because metabolism database searches did not yield information on these 4 compounds, the possible metabolic fates of each were determined based on structural features, a substructure search, and a Meteor (9.0) metabolism prediction. The results of this assessment indicated that it is likely that C-oxidation, C-hydroxylation, glucuronidation, and beta-oxidation may take place to form corresponding metabolites. Furthermore, C-hydroxylation and beta-oxidation are more likely to be favored metabolic pathways for the longer alkyl chain compounds, 1,2-decanediol and 1,2-dodecanediol, than for the shorter alkyl chain length compounds, 1,2-hexanediol and 1,2-octanediol.
1,2-Butanediol
1,2-Butanediol was infused intravenous (iv) into rabbits at a dose of 1 g/kg body weight. Metabolism was described as slow, and 1,2-butanediol was excreted in the urine either as the glucuronide or unchanged. 21 Accumulation in the tissues was not observed. Metabolites were not isolated from the urine of rabbits fed 1,2-butanediol at a dose of 0.2 g/kg body weight.
Propylene glycol
The CIR Expert Panel safety assessment reported that, in mammals, the pathway of PG metabolism is to lactaldehyde and then lactate via hepatic alcohol and aldehyde dehydrogenases. When PG was administered iv to human participants (patients), elimination from the body occurred in a dose-dependent manner.
Percutaneous Absorption
Caprylyl glycol
The dermal absorption and skin penetration of 5% dermosoft octiol in alcoholic solution (5% caprylyl glycol in 70% ethanol/30% PG) in vitro was evaluated using skin from the backs of female pigs (∼130 days old) in Franz diffusion cells. The partition coefficient of caprylyl glycol was estimated using an appropriate computer program (ACD logD-Suite) to be log Pow ≈ 1 (pH 3-7.4). The solution was applied topically to excised pig skin for 24 hours. The investigators used an analytical method that only measured the parent compound, caprylyl glycol, and the total recovery was only 55%.
Approximately 97% of the recovered material was found in the skin within 24 hours post-application, and the following distribution (as percentage of dermal absorbed caprylyl glycol) was reported: ∼10% in stratum corneum, ∼9% in epidermis, and ∼81% in dermis. Caprylyl glycol was not detected in the receptor fluid, and this was likely a result of metabolism in the skin. The authors noted that, normally, the metabolism of caprylyl glycol takes place mainly in the epidermis/dermis. Therefore, undetectable amounts of the unchanged substances (below the detection limit) may penetrate into the receptor fluid. Because size of the sample (N = 2; taken from same pig) was very small and considered nonrepresentative, it was not possible to perform an inductive statistical analysis. Therefore, according to the authors, the descriptive results achieved in this study have to be considered as a trend and interpreted as such. 22
In addition to the dermal penetration study, a study in which caprylyl glycol was incubated with and without the cut pig skin for 24 hours was completed. 22 Compared to the sample without pig skin, 50% of the caprylyl glycol was lost in the presence of skin during the 24-hour incubation. The investigators attributed this loss to chemical or metabolic degradation and suggested that the poor recovery in the dermal penetration study was likely a result of the metabolism.
Caprylyl glycol, 1,2-hexanediol, decylene glycol, and lauryl glycol
Dermal penetration modeling information on the following 1,2-glycols (C6-C12) was provided by the Personal Care Products Council: caprylyl glycol (1,2-octanediol, C8), 1,2-hexanediol (C6), decylene glycol (1,2-decanediol, C10), and lauryl glycol (1,2-dodecanediol, C12). 23 Dermal penetration predictions were made on the basis of J max (maximal flux) values calculated from Kp estimations and calculated water solubility. Based on the calculated J max values, assignment of default percentage absorption values was done, as described by Kroes et al. 24 Utilizing this approach, the default values for percentage dose absorbed per 24 hours were 80% for 1,2-hexanediol and 1,2-octanediol and 40% for 1,2-decanediol and 1,2-dodecanediol.
Propylene glycol
The dermal penetration of [ 14 C]PG through excised female hairless mouse skin from the ternary cosolvent containing 10 mol% oleic acid and 6 mol% dimethyl isosorbide in 84% PG was determined. Over a 24-hour period, the cumulative penetration of PG was 57.1% of the applied amount. 2
Skin Penetration Enhancement
Caprylyl glycol, 1,2-hexanediol, and decylene glycol
Warner et al 12 studied 3 H-corticosterone (CS) and 3 H-triethanolamine flux (TEA) enhancement across full-thickness hairless mouse (SKH-HR1 strain) skin in the presence of 1,2-octanediol (caprylyl glycol), 1,2-decanediol (decylene glycol), and 1,2-hexanediol, each in phosphate-buffered saline (PBS). Permeability experiments were performed using a 2-chamber diffusion cell, and the results are presented in Table 4. Each of the 3 chemicals enhanced the skin penetration of CS and TEA in a concentration-dependent manner.
Corticosterone and TEA Permeability Coefficients in the Presence of Permeation Enhancers 12
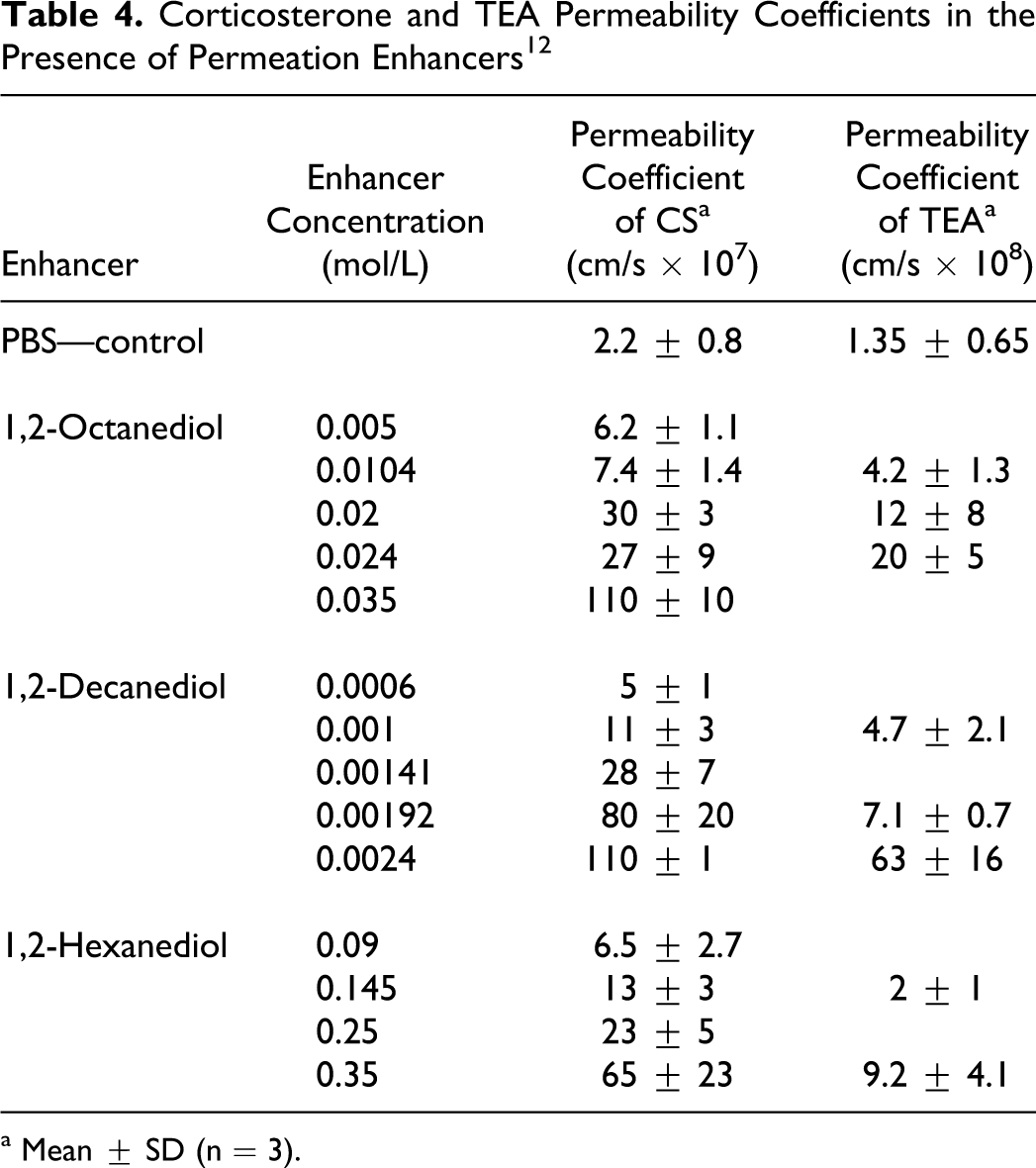
a Mean ± SD (n = 3).
1,2-butanediol and pentylene glycol
In a study by Heuschkel et al, 25 the influence of pentylene glycol and 1,2-butanediol on the skin penetration of the drug dihydrovenavenanthramide D (DHAvD, 0.2% in hydrophilic cream) across full-thickness human skin (from breast, females) was investigated using Franz-type diffusion cells. Relative amounts of DHAvD in different skin compartments (stratum corneum, viable epidermis, and dermis) following penetration from a hydrophilic cream and from a hydrophilic cream containing a 4% pentylene glycol/1,2-butanediol mixture were compared. Within 30 minutes, the amount of DHAvD that penetrated into the viable skin layers doubled in the presence of the glycol mixture. After 300 minutes, 12% of the applied dose was detected in the viable epidermis and dermis after application of DHAvD in hydrophilic cream, compared to 41% after application in the cream with the glycol mixture.
Propylene glycol
Propylene glycol has been described as a penetration enhancer. Proposed mechanisms of penetration enhancement by PG include alteration of barrier function by its effects on a keratin structure or a PG-induced increase in the solution capacity within the stratum corneum. 2
Toxicology
Single-Dose Toxicity
Inhalation
1,2-Butanediol
According to a data summary available from Dow Chemical Company, there were no obvious toxic effects in rats exposed for 7 hours to an atmosphere saturated with 1,2-butanediol. 21 Further details relating to this study were not available.
Oral
Caprylyl glycol
The acute oral toxicity of caprylyl glycol was evaluated using male and female rats (number and strain not stated). 26 Doses of ≥464 mg/kg caused sedation and ataxia. Specifically, loss of muscle tone and dyspnea were observed at a dose of 1000 mg/kg, and lateral position, coma, and death were observed at a dose of 1470 mg/kg. Deaths occurred within 2 hours post-administration; at necropsy, pale parenchymal organs were observed in 3160 and 4640 mg/kg dose groups. Surviving animals recovered within 24 hours, and 215 mg/kg was the nontoxic dose in this study. The LD50 values of 2240 (males) and 2200 (females) were reported.
In another study (OECD 423 test procedure) involving rats, the LD50 for caprylyl glycol was >2500 mg/kg. 27,
1,2-Butanediol
An acute oral LD50 of 4192 mg/kg was reported for 1,2-butanediol in a study involving female Swiss albino mice/ICR. 28 Study details were not provided. According to a data summary available from Dow Chemical Company, the acute oral LD50 for 1,2-butanediol in rats was 16 g/kg body weight. 29 Also, high (unspecified) doses caused narcosis in rats (often leading to death in a few hours), dilation of the blood vessels, and kidney damage. 1,2-Butanediol administered orally to rats (ethanol dependent) at a dose of 2.74 g/kg did not induce any overt toxic effects. 21
Pentylene glycol (1,2-pentanediol)
The following acute oral LD50 values have been reported for pentylene glycol: 1.2700 E + 04 mg/kg (rats); 7400 mg/kg (mice); 3700 mg/kg (rabbits); and 5200 mg/kg (guinea pigs). 30
Stearyl glycol
An LD50 of >5000 mg/kg was reported for rats dosed orally with stearyl glycol. 30
C15-18 glycol
The acute oral toxicity of C15-18 glycol was evaluated using adult male Sprague-Dawley rats, and an LD50 of >20.0 g/kg body weight was reported. 5
Propylene glycol
The 24-hour oral LD50 for PG was 22.8 g/kg body weight in a study involving 5 female Fischer rats. Oral LD50 values (rats) of up to 27 g/kg body weight have been reported in other studies. 1
Dermal
1,2-Butanediol
According to a data summary provided by Dow Chemical Company, prolonged application of 1,2-butanediol to the skin of rabbits did not result in overt toxic effects. 21 Details relating to the test procedure were not provided; however, it was presumed that neat material was tested.
Decylene glycol
In an acute dermal toxicity study involving rats, the LD50 for decylene glycol (SymClariol) was >2000 mg/kg. 27
Propylene glycol
The dermal LD50 for PG was >11.2 g/kg in mice and was 13 g/kg in rats. 1
Intraperitoneal
Caprylyl glycol, 1,2-butanediol, and pentylene glycol
In a report by Shoemaker, 31 the intoxicating potency of alcohols, some of which were straight-chain primary alcohols and straight-chain diols, was determined. Data on the following 3 diols reviewed in this safety assessment were included: caprylyl glycol (1,2-octanediol), pentylene glycol (1,2-pentanediol), and 1,2-butanediol. Doses of each alcohol were injected (intraperitoneally [ip]) into male Sprague-Dawley rats, and intoxicating scores were recorded based on the following rating scale: 0 (normal) to 7 (death).
An ED3 value for each chemical was determined. The ED3 was defined as the dose (mmole/kg body weight) required to obtain a score of 3 (ataxia) on the intoxication rating scale (0-7 [death]). The following ED3 values were reported: 1.5 mmole/kg (caprylyl glycol), 256.0 mmole/kg (pentylene glycol), and 32.6 mmole/kg (1,2-butanediol). 31
Groups of 6 adult female, ICR Swiss albino mice were injected ip with increasing doses of 1,2-butanediol (geometric factor of 1.2) in distilled water (injection volume = 0.01 mL/g body weight). Mean LD50 values and 95% confidence limits were calculated from cumulative mortality curves at 24 hours and 144 hours. The following values were reported for 1,2-butanediol: 24-hour LD50 of 66.5 mmol/kg (∼5.99 g/kg) and 144-hour LD50 of 46.5 mmol/kg (∼4.19 mg/kg). 32 Muscle incoordination was observed in rats at an ip dose of ∼2.94 g/kg 1,2-butanediol. 21 An ip TDLo of 3510 mg/kg has been reported for pentylene glycol in rats. 30
Propylene glycol
Following ip dosing with PG (5 mL/kg), none of the 5 female C3H mice died, but peritonitis was observed at necropsy. In other studies, ip LD 50 values up to 13.7 mL/kg (rats) and 11.2 g/kg (mice) have been reported. 1 An acute study was performed in which female ICR mice were dosed ip with 2600, 5200, or 10 400 mg/kg PG. 33 All except the high-dose mice survived 6 days after dosing. Signs of toxicity, such as lethargy and ruffled hair coats, were not observed in the 2600 and 5200 groups. 2
Intravenous
Propylene glycol
Acute iv LD50s of 6.2 mL/kg (rats) and 6.4 mL/kg (mice) have been reported for PG. In other parenteral toxicity studies, acute intramuscular (im) LD50 (20 g/kg rats) and acute subcutaneous (sc) LD50 (18.5 g/kg mice) values have been reported. 1
Repeated-Dose Toxicity
Oral
Caprylyl glycol
In a 28-day oral toxicity study, >98% caprylyl glycol (Dermosoft Octiol) was administered to groups of Wistar rats at doses of 50, 300, and 1000 mg/kg/d, respectively, according to OECD guidelines. 34 The number of animals per group was not stated and the control group was not identified. The authors reported no test substance-related mortalities or toxicologically relevant clinical signs during weeks 1 through 3 or week 4 (functional observational battery). Additionally, there were no differences in feed consumption, body weight, hematological/clinical biochemistry parameters, or macroscopic findings that were considered toxicologically relevant. Test substance-related findings (males and females) included slightly reduced locomotor activity and increased mean absolute and relative kidney weights at the highest dose. Whether or not microscopic changes were observed in the kidneys was not stated.
Systemic effects were not observed at doses up to 300 mg/kg/d. Test substance-related microscopic changes were observed in the stomach of rats in 300 and 1000 mg/kg/d dose groups. These findings were considered indicative of an irritative potential of the test substance on the pars nonglandularis and limiting ridge of the stomach. The authors noted that analogous structures do not exist in humans. Study results indicated a no observed effect level (NOEL) of 50 mg/kg/d, and a no observed adverse effect level (NOAEL) of 300 mg/kg/d for systemic toxicity. The NOAEL was based on the findings (irritation) in the stomach, likely due to local irritation effects. 34
1,2-Butanediol
In an 8-week oral study, groups of rats were fed 1,2-butanediol at concentrations ranging from 5% to 40% in the basic diet (1 dose level per group). 21 A control group only received basic diet. There were no mortalities at the lowest dose (∼2.9 g/kg body weight/d); however, doses ≥10% were classified as fatal. The following signs of toxicity were noted at the highest dose of 22 g/kg/d: weight loss, fatigue, reduced responsiveness, diarrhea, and rapid, shallow breathing. No abnormalities were observed in tissues of major organs from 2 rats at each of the 5 dose levels.
The following study is actually a combined repeated dose/reproductive and developmental toxicity study, and the results relating to reproductive and developmental toxicity appear in that section later in the report text. 35 Groups of Crj-CD(SD) rats (10 males and 10 females) were dosed orally, by gavage, with aqueous 1,2-butanediol at doses of 40, 200, or 1000 mg/kg/d. Males were dosed daily for 42 days, and females were dosed from day 14 before mating to day 3 of lactation. Control rats (10 males and 10 females) were dosed with distilled water.
None of the animals died, and there were no differences in histopathological findings or the following parameters between test and control animals: body weights, feed consumption, hematology parameters, clinical chemistry parameters, and organ weights. However, transient hypolocomotion and hypopnea (slight clinical signs) were observed in females that received 1000 mg/kg doses. No observable effect levels (NOELs) for repeat dose toxicity were 1000 mg/kg/d (males) and 200 mg/kg/d (females). The no observable adverse effect level (NOAEL) was 200 mg/kg body weight/day in this study. 35
According to a summary of data provided by Dow Chemical Company, the administration of large (unspecified) doses of 1,2-butanediol to rats caused irritation of the gastrointestinal tract. 21
Decylene glycol
In a 28-day oral toxicity study, 98% to 100% decylene glycol (SymClariol) was administered to groups of SPF-bred Wistar rats (5 males, 5 females/group) at doses of 100, 300, and 1000 mg/kg/d, respectively, according to OECD guidelines. 36 The vehicle control group received 2.5% ethanol in distilled water. Rats in each group were killed after day 28. Two additional groups (same composition) were untreated and dosed with 1000 mg/kg/d, respectively, for 28 days. The animals in these groups were killed after a 14-day nontreatment period. In all groups, a functional observational battery was performed (week 4) before animals were killed. All of the animals survived the 28-day dosing period, and there were no toxicologically relevant clinical signs during the study. Mean locomotor activity was significantly reduced in males and females in the 1000 mg/kg/d dose group, and this finding was deemed test substance related. Decreased feed consumption was also noted in females at this dose level. Mean body weights of males and females were similar to those of negative control animals.
There were no test substance-related differences in hematological or clinical biochemical parameters that were of toxicological relevance. The presence of ketone in the urine of males and females of the 1000 mg/kg/d dose group was considered likely representative of metabolic adaptation to the test substance. Both absolute and relative organ weights of dosed animals were comparable to those of negative control rats. Toxicologically relevant macroscopic findings were not observed. Squamous epithelial hyperplasia, ulceration, and inflammation of the forestomach were observed at doses of 1000 mg/kg/d, and squamous epithelial hyperplasia of the forestomach was less severe and occurred at a lower incidence in the of 300 mg/kg/d dose group. After a 14-day recovery period, squamous epithelial hyperplasia remained in the animals previously dosed with 1000 mg/kg/d, but the severity and incidence of this finding after the treatment period was largely reversible. Both the NOEL and the NOAEL in this study was 100 mg/kg body weight/d. 36
Pentylene glycol
Pentylene glycol was administered orally to rats, intermittently over a 28-week period. A TDLo of 2450mg/kg was reported. 30
Propylene glycol
A 92- to 97-day study was conducted to assess the safety and tolerability of PG as an alternative formulation vehicle in general toxicology studies in the mouse, rat, dog, and monkey. 37 In Sprague-Dawley (Crl:CD[SD]VAF/Plus) rats (10/sex; 6 ± 1 weeks old) and CD1 (Crl:CD1[Icr]VAF/Plus) mice (10/sex; 6 ± 1 weeks old), the vehicle was administered orally via gavage at dose volumes of 5 mL/kg (rats) and 10 mL/kg (mice) for 92 to 93 days. In Beagle dogs (4/sex; 7 to17 months old) and cynomolgus monkeys (Macaca fascicularis, 4/sex; juvenile to young adult), the vehicle was administered orally by gavage (dose = 1000 mg/kg; dose volume of 5 mL/kg) for 95 to 97 days. Effects on clinical observations, body weight, food consumption parameters, clinical pathology, and histopathology were evaluated across all species. The suitability of formulations containing up to 1000 mg/kg PG for use in preclinical safety studies was confirmed by a lack of effects on all parameters examined. 37
No significant toxicity was observed in oral studies of PG involving dogs and cats. Dogs received 3.0 mL/kg doses of undiluted PG over a 3-day period, and cats received 12% PG in the diet for 5 weeks and 41% PG in the diet for 22 days. Short-term iv dosing with PG resulted in little toxicity in rats. Groups of rats received iv infusions of PG/ethanol/water (5:1:4) over a 2-week period. There was no evidence of toxic effects in an oral toxicity studies in which rats were fed 50 000 ppm PG in the diet for 15 weeks, and dogs received 5% PG in drinking water for 9 months and 10% PG in drinking water for 6 months. Toxic effects were not observed in rats that received up to 50 000 ppm PG in the diet for 104 weeks or in dogs that received 2 g/kg PG in the diet for 104 weeks. 1 Groups of 8 male and 8 female CD-1 mice were given 0.5%, 1.0%, 2.5%, 5.0%, and 10.0% PG in the drinking water for 14 days. Body weight gains of test animals were similar to or greater than controls. No animals died during the study. 2
Inhalation
Propylene glycol
Male and female Sprague-Dawley rats (number per group not given) were exposed to 0.16, 1.0, or 2.2 mg PG/L air for 6 hours/d, 5 days/week, for 13 weeks in a nose-only inhalation study. Relevant differences occurred in some hematological parameters, serum enzyme activities, and lung, spleen, liver, and kidney weights; however these differences were inconsistent and without dose–response trends. 2
Dermal
1,2-Butanediol
According to a data summary provided by Dow Chemical Company, repeated applications of 1,2-butanediol to the skin of rabbits did not result in overt toxic effects. 21 Details relating to the test procedure were not provided.
Cytotoxicity
Pentylene glycol
Anselmi et al 38 conducted an in vitro DNA fragmentation assay (human promyelocytic leukemia cell line [HL60]) to investigate the apoptosis- and necrosis-inducing potential of brief, 10-minute applications of the preservative, pentylene glycol (between 0.01% and 5% [usual concentration as a preservative]). Cells treated with phosphate-buffered saline served as controls. The percentage of apoptotic cells was quantified by analysis of DNA content. Pentylene glycol induced apoptosis only at a concentration of 5%. Externalization of phosphatidyl serine, a hallmark of apoptosis, was concomitant with the subdiploid DNA peak in HL60 cells treated with pentylene glycol.
Lauryl glycol
Osorio e Castro et al 39 studied hemolysis rates (at 37°C) of human erythrocytes induced by C2 and C8-C14 straight chain 1-alkanols, 1,2-alkanediols, and the corresponding benzilidene derivatives (benzaldehyde acetals). The most active compound was 1-dodecanol (50% hemolysis at 15 µmol/L), followed by 1,2-dodecanedol (lauryl glycol, 50% hemolysis at 99 µmol/L) and the C10 benzylidene acetal (50% hemolysis at 151 µmol/L).
Cetyl glycol
In an antitumor activity test, 1,2-hexadecanediol (cetyl glycol) was injected intraperitoneally (ip) into 8 inbred C57BL/6 mice in which Ehrlich ascites carcinoma (EAC) cells had been implanted. Doses of 80/mg/kg/d were injected for 10 consecutive days. The survival of mice was monitored over a 2-month period. Compared to the control mice, dosing with cetyl glycol prolonged the life span of animals more than 2.7-fold. Antitumor effects were described as marked, in that 4 of 8 mice injected were alive, with scarce tumor proliferation, at 60 days. Cetyl glycol (130 µg/mL) was found to have a cytocidal effect (irreversible cell degeneration) on cultured EAC cells. 40
Propylene glycol
PG was found to be cytotoxic in assays that measured inhibition of human foreskin fibroblasts and keratinocytes, inhibition of collagen contraction by fibroblasts, and changes in cell morphology of fibroblasts and keratinocytes. Changes in morphology included detachment of cells from the culture and changes in cell shape. 2
Ocular Irritation
Caprylyl glycol
In an in vitro assay (hen’s egg test on the chorioallantoic membrane [HET-CAM]) for evaluating ocular irritation potential, caprylyl glycol was classified as a nonirritant at test concentrations of 1% and 3% in neutral oil. 41
Caprylyl glycol and 1,2-hexanediol
A 50:50 (w/w) mixture of 1,2-hexanediol and caprylyl glycol (Symdiol 68) was also tested in the HET-CAM assay. The mixture was classified as a severe eye irritant at a test concentration of 1% aqueous (effective concentration per ingredient = 0.5%). 42
1,2-Butanediol
According to a summary of data provided by Dow Chemical Company, undiluted 1,2-butanediol was irritating to the eyes of rabbits but was a nonirritant when tested as a 10% aqueous solution. 21
Pentylene glycol
The ocular irritation potential of a lash gel serum containing 3% pentylene glycol was evaluated using the following in vitro assays: neutral red release (NRR) assay using rabbit cornea fibroblasts, HET-CAM, and the reconstituted human epithelial culture (REC) assay. 43 In the NPR assay, the undiluted product and dilutions (in hydrophilic or lipophilic substance) ranging from 0.1% to 60% were tested. Sodium dodecyl sulfate served as the positive control. The test product concentration that gave rise to the release of 50% neutral red dye (NR50) was used as an end point to reflect cytotoxicity. Data were expressed as a percentage of cytotoxicity, compared to the negative control (dilution 0%), and the NR50 was calculated by interpolation from the curve representing the percentage of viability versus the concentration of test product. An NR50 of >50% (slightly cytotoxic) was reported for the lash gel serum.
In the HET-CAM assay, the undiluted product (0.3 mL) was applied to the CAM and classified as moderately irritating. In the REC assay, the product (neat or diluted) was applied to the apical surface of the epithelial culture. Hexadecylpyridinum bromide solution in saline and saline solution served as positive and negative controls, respectively. Results were expressed as a percentage of cytoxicity, compared to the negative control. The product was classified as slightly cytotoxic. Together, the results for the 3 in vitro assays indicate that the lash gel serum might be a slight ocular irritant, with a Draize score that might range from 0 to 15. The conclusion for this study (slight ocular irritant) is from a global assessment conducted by the International Research and Development Center that was based on results of the 3 methods used, because no single alternative method can predict ocular irritation with a sufficient level of safety. 43
Decylene glycol
In an ocular irritation study (OECD 405 protocol) involving rabbits, decylene glycol (SymClariol) induced corrosion when tested at a concentration of 100%. Additionally, the ocular irritation potential of 1% SymClariol in neutral oil was evaluated in the HET-CAM assay, and the results were negative. 27
Lauryl glycol
According to Worth and Cronin, 44 the European Union has classified 1,2-dodecanediol (lauryl glycol) as a severe ocular irritant. The European classification system has allowed 2 classes of acute eye toxicity, R36 for moderate irritants and R41 for severe irritants, and the Draize eye test has been used for the identification of R41 chemicals. Actual Draize test results for lauryl glycol were not included. This classification of lauryl glycol as a severe ocular irritant is included in a study by the preceding authors to explore the possibility of distinguishing between eye irritants and nonirritants by using in vitro end points of the HET-CAM assay and the neutral red uptake (NRU) test.
According to one of the prediction models for eye irritation potential, a chemical is more likely to be an eye irritant if its log (TH10) value is low (ie, if a 10% solution of the chemical produces rapid hemorrhaging of the chorioallantoic membrane) and if its log (IC 50) value is low (i.e., if the chemical is cytotoxic to 3T3 cells). TH10 is defined as the mean detection time (units not stated) for hemorrhage in the vascularized chorioallantoic membrane of embryonated chicken eggs. The IC50 is defined as the concentration of test chemical (mg/ml) resulting in 50% inhibition of NRU in 3T3 cells. The TH10 and IC50 values for lauryl glycol were 171.0 and 0.02, respectively. 44 Using a logarithm calculator, log 0.02 = −1.70 and log 171.0 = 2.23.
Propylene glycol
PG (0.1 mL, pH 8.8) was a slight ocular irritant in rabbits in 1 study, but PG (0.1 mL, pH unknown) did not induce ocular irritation in another study involving rabbits. 1 The ocular irritation potential of PG was determined using groups of 6 male and female New Zealand white albino rabbits. Following instillation of a single drop and multiple instillations, slight-to-moderate conjunctival hyperemia was observed and all reactions had cleared by day 3. 2
Skin Irritation and Sensitization
Caprylyl glycol
The skin sensitization potential of caprylyl glycol was evaluated in the guinea pig maximization test (OECD 406 protocol) using 20 animals. During intradermal and topical induction, caprylyl glycol was applied at concentrations of 5% (in peanut oil) and 50% (in petrolatum). The challenge concentration was 50% in petrolatum. Sensitization was not observed in any of the animals tested. 41
1,2-Butanediol
According to a summary of data provided by Dow Chemical Company, 1,2-butanediol did not induce skin irritation in rabbits following prolonged and repeated application. 21 Details regarding the test procedure were not provided; however, it was presumed that neat material was used.
1,2-Hexanediol
The sensitization potential of 1,2-hexanediol was evaluated at concentrations of 10%, 50%, and 100% in acetone/olive (3:1) using the mouse local lymph node assay (OECD 429 protocol). Study results were negative for skin sensitization. 45
Decylene glycol
In a skin irritation study (OECD 404 protocol) involving rabbits, 100% decylene glycol (SymClariol) was classified as a moderate skin irritant (PII = 3.2). SymClariol was evaluated at the following concentrations in the guinea pig maximization test: 1% in arachis oil (intradermal induction), 5% in arachis oil (topical induction), and 2% and 5% in arachis oil (challenge). Sensitization was not observed in any of the 19 guinea pigs tested. 27
The skin sensitization potential of SymClariol was also evaluated at the following test concentrations in the mouse local lymph node assay: 5%, 10%, 25%, and 50% in acetone/olive oil (4:1). Sensitization was not recorded at any of the concentrations tested. 27
Propylene glycol
In nude mice, hypertrophy, dermal inflammation, and proliferation were observed with 50% PG, but undiluted PG was, at most, a mild dermal irritant in a Draize test using rabbits with intact and abraded skin. No reactions to undiluted PG were observed with guinea pigs, rabbits, or Gottingen swine. PG (concentrations not given) was negative in a number of sensitization/allergenicity assays using guinea pigs, but, in another study, PG (0.5 mL) was a weak sensitizer in guinea pigs. 1
The dermal irritation potential of 100% PG was evaluated using male hairless SKH1 hr/hr mice. The PG was minimally irritating, with a total score of 7 (maximum score =77). 2
Reproductive and Developmental Toxicity
1,2-Butanediol
The test procedure for the combined repeated dose and reproductive/developmental toxicity study (Crj-CD(SD) rats) and results relating to oral toxicity are included in the Short-Term Oral Toxicity section earlier in the reported text. All of the animals were killed on day 4 of lactation. Neither effects on reproduction (copulation, implantation, pregnancy, parturition, or lactation) nor developmental toxicity effects on offspring were observed. The NOAEL was 1000 mg/kg for parental animals and the F1 generation. 35 The estimated dose of low concern (EDCL) for this study was calculated as 10 mg/kg/d, using an NOAEL of 1000 mg/kg/d and a reproductive toxicity uncertainty factor of 100. 7
1,2-Hexanediol
The developmental toxicity of Hydrolite-6 (99% 1,2-hexanediol) was evaluated using groups of 24 mated Sprague-Dawley rats of the Crl:CD strain. 46 Three groups received oral doses (gavage) of 30, 100, and 300 mg/kg/d, respectively, between days 5 and 19 of gestation. The negative control group received vehicle (not stated) only. Pregnant females were killed on day 20 of gestation and subjected to macroscopic necropsy. Doses up to 300 mg/kg/d were well tolerated and did not induce any effects on clinical condition, body weight, body weight change, food intake, or necropsy observations. There were also no effects on embryo-fetal survival, growth, or development at doses up to 300 mg/kg/d. It was concluded that Hydrolite-6 at doses up to 300 mg/kg/d was not associated with any adverse effect on the pregnant rat or the developing conceptus. The Hydrolite-6 (1,2-hexanediol) NOEL for the pregnant female and for embryo-fetal survival, growth, and development was considered to be 300 mg/kg/d.
Propylene glycol
A continuous breeding reproductive study was conducted using COBS Crl:CD-1 (ICR)BR outbred Swiss albino mice (6 weeks old). The 3 experimental groups received the following doses (in feed or water), respectively, during a 7-day premating period: 1.0% PG (daily dose of 1.82 g/kg), 2.5% PG (daily dose of 4.80 g/kg), and 5.0% PG (daily dose of 10.10 g/kg). The PG was not a reproductive toxicant in this study. 2 The reproductive and developmental effects of PG were evaluated using mice, rats, rabbits, and hamsters. Groups of 25 or 28 female albino CD-1 outbred mice were mated and 22, 22, 22, 20, and 23 gravid mice were dosed by oral intubation with 0.0, 16.0, 74.3, 345.0, and 1600.0 mg/kg aq PG on 6 to 15 days of gestation. Groups of 25 to 28 female albino Wistar rats were mated and 22, 23, 22, 20, and 24 were dosed as above, respectively. The PG was not a reproductive or developmental toxicant in this study. 2
Groups of 11, 11, 12, 14, and 13 gravid female Dutch-belted rabbits were dosed by oral intubation with 0, 12.3, 57.1, 267.0, or 1230.0 mg/kg aq PG on days 6 to 18 of gestation, respectively. Administration of PG did not cause reproductive or developmental toxicity. 2
Groups of 24 to 27 female golden hamsters were mated and 21, 24, 25, 22, and 22 gravid hamsters were dosed by oral intubation with 0.0, 15.5, 72.0, 334.5, and 1550.0 mg/kg aq PG on days 6 to 10 of gestation, respectively. The PG was not a reproductive or developmental toxicant in this study. 2
The PG was used as a vehicle in a reproductive and behavioral development study. It was administered to 15 gravid Sprague-Dawley rats orally by gavage on days 7 to 18 of gestation at a volume of 2 mL/kg. The PG did not have any effects on reproductive or behavioral development parameters. 2
Embryonic development was reduced or inhibited completely in cultures of mouse zygotes exposed to 3.0 or 6.0 mol/L PG, respectively. 1
A study was performed to determine whether PG induced cytogenetic aberrations in mouse metaphase II (MII) oocytes that predispose zygotes to aneuploidy. In the MII portion of the study, female ICR mice were dosed ip with 1300, 2600, or 5200 mg/kg PG in distilled water after dosing with human chorionic gonadotropin (hCG). A statistically significant change in hyperploidy, hypoploidy, or single chromatids was not observed. An increase in the frequency of PCS at each dose was statistically significant, and the incidence of premature anaphase was significantly greater in the 5200 mg/kg dose group as compared to controls. In the zygote portion of the study, female mice were dosed ip with 1300, 2600, or 5200 mg/kg PG 3 hours after hCG administration. There were 30, 40, 49, and 66 mice in the control, 1300, 2600, and 5200 mg/kg groups, respectively. The increase in hyperploidy was statistically significant in all test groups compared to controls. A statistically significant change was not seen for polyploidy or hypoploidy, and zygotes containing PCS, premature anaphase, or single chromatids were not found. There was not a statistically significant difference in the proportion of zygotes collected for each group compared to oocytes. However, the number of zygotes analyzed compared to the number placed on slides was significantly decreased in the test groups; a relatively large portion of these zygotes had clumped chromosomes. 2
Genotoxicity
Caprylyl Glycol
The genotoxicity of >98% caprylyl glycol (Dermosoft Octiol) was evaluated in a gene mutation assay involving Chinese hamster V79 cells in vitro according to OECD and European Commission guidelines. 47 Test concentrations up to 1480 µg/mL were evaluated. The first experiment (with and without metabolic activation) involved a 4-hour treatment period, whereas the second experiment involved 4-hour and 24-hour treatment periods (without activation). A substantial or reproducible dose-dependent increase in the mutation frequency was not observed in either of the 2 experiments. Appropriate reference mutagens (positive controls, unnamed) induced a distinct increase in mutant colonies. Negative control cultures were not described. Caprylyl glycol, >98% (Dermosoft Octiol) did not induce gene mutations under the experimental conditions reported and, therefore, was considered nonmutagenic.
The genotoxicity of >98% caprylyl glycol (ADEKA NOL OG) was evaluated in the chromosome aberrations assay using Chinese hamster lung (CHL/IU) cells in vitro according to Ministry of Health and Welfare (Japan) genotoxicity test guidelines. 48 Short-term treatment of cultures (with and without metabolic activation) involved concentrations up to 700 µg/mL and continuous treatment involved concentrations up to 180 µg/mL, both with and without metabolic activation. Negative and positive control cultures were not identified. In all test cultures, the number of structural and numerical chromosomal aberrations was not increased when compared to negative control cultures. The positive control was genotoxic. The test substance did not induce chromosomal aberrations with or without metabolic activation.
1,2-Butanediol
1,2-Butanediol was not mutagenic to Salmonella typhimurium strains TA100, TA98, TA97, and TA102 at doses up to 5,000 µg/plate with or without metabolic activation. The test substance also induced neither chromosomal aberrations nor polyploidy in Chinese hamster CHL cells at doses up to 0.9 mg/mL either with or without metabolic activation. 49
Decylene Glycol
In the Ames test (OECD 471 protocol), decylene glycol (SymClariol) was classified as nonmutagenic. Test concentrations were not stated.
Propylene Glycol
Propylene glycol (≤10 000 µg/plate) was not mutagenic in Ames tests with or without metabolic activation. Propylene glycol, tested at concentrations of 3.8 to 22.8 mg/mL, was a weak, but potential, inducer of sister chromatid exchanges (SCEs), causing a dose-dependent increase in SCEs in a Chinese hamster cell line. However, in another SCE assay using human cultured fibroblasts and Chinese hamster cells with and without metabolic activation, PG was not mutagenic. Propylene glycol, 32 mg/mL, induced chromosomal aberrations in a Chinese hamster fibroblast line but not in human embryonic cells. Propylene glycol was not mutagenic in mitotic recombination or base pair substitution assays, or in a micronucleus test or a hamster embryo cell transformation assay (concentration used not specified). 1
Carcinogenicity
Propylene Glycol
Propylene glycol was noncarcinogenic in a 2-year bioassay in which rats were given ≤50 000 ppm PG in the diet (feeding schedule not included). The dermal application of undiluted PG (volume not stated) to Swiss mice in a lifetime study was noncarcinogenic. Propylene glycol was noncarcinogenic in other oral, dermal, and subcutaneous studies. 1
Clinical Assessment of Safety
Predictive Testing—Irritation and Sensitization
Caprylyl glycol and 1,2-hexanediol
A lipstick containing 0.5% caprylyl glycol was evaluated in an RIPT using 105 healthy participants (males and females). The product was applied to the upper back of each participant and the application sites were covered with a semiocclusive patch for 24 hours. It was concluded that the product did not demonstrate a potential for eliciting skin irritation or sensitization. 50
Levy et al 51 studied the potential for delayed type IV dermal sensitivity following exposure to a new preservative containing 1,2-hexanediol and caprylyl glycol. In a repeat insult patch test, a 15% mixture of 1,2-hexanediol and caprylyl glycol (equal parts of the 2 ingredients) in carbomer gel (total volume = 20 µL) was applied to each of 205 participants (163 females, 42 males; 18-70 years old). The mixture was applied under 48 hours of occlusive patches (Finn chambers) during induction and challenge phases. Challenge application involved a new test site and the reactions were scored at 48 and 72 hours post-application according to the following scale: + (definite erythema without edema) to +++ (definite erythema, edema, and vesiculation). One of the participants had a D reaction (damage to the epidermis: oozing, crusting, and/or superficial erosions) to the mixture; however, no reactions were observed in a subsequent 4-day repeat open application test. The reaction observed was indicative of irritation.
A cosmetic formulation containing the same preservative (gel vehicle) at an actual use concentration (0.5%) was evaluated in an additional group of 224 participants (176 females, 48 males; 19-70 years old) according to the same test procedure. None of the participants had a delayed type IV dermal reaction. 51
A 50:50 (w/w) mixture of 1,2-hexanediol and caprylyl glycol (Symdiol 68) was evaluated in an RIPT involving 56 participants. At a test concentration of 20% in gel (effective concentration per ingredient = 10%), the mixture did not induce skin sensitization in any of the participants tested. 42
A leg and foot gel containing 0.5% 1,2-hexanediol was applied to the upper back of each of 101 healthy participants (males and females) in an RIPT. Each site was covered with a semiocclusive patch that remained in place for 24 hours. The product did not induce skin irritation or sensitization in this study. 52
In an in-use safety evaluation for skin irritation and sensitization potential, 28 participants (males and females) were instructed to use a body wash containing 0.15% 1,2-hexanediol for a minimum of 3 times per week over a 30-day period. There was no evidence of erythema, edema, or dryness of application sites in any of the participants, and it was concluded that the product did not demonstrate a potential for eliciting skin irritation or sensitization. 53
Pentylene glycol
A foundation containing 0.112% pentylene glycol was evaluated in an RIPT using 101 participants (males and females). A 1" × 1" semiocclusive patch containing 0.2 g of the product was applied repeatedly (24 hours of applications) to the upper back. It was concluded that the product did not have a potential for inducing skin irritation or allergic contact sensitization. 54
Decylene glycol
The skin irritation potential of decylene glycol (SymClariol) was evaluated using 52 participants in a 48-hour semioccluded patch test. At a concentration of 20% in petrolatum, the test substance did not induce skin irritation. SymClariol (1% in neutral oil) had low skin irritation potential when applied to scarified skin sites on 10 participants. In an HRIPT, SymClariol (20% in petrolatum) did not induce skin sensitization in any of the 55 participants tested. 27
In a facial stinging test, SymClariol was classified as having very slight stinging potential when applied at concentrations of 1% and 2% (in neutral oil) in a group of 10 participants. 27
Propylene glycol
Propylene glycol induced skin irritation reactions in normal participants. Reactions were observed at concentrations as low as 10% in predictive tests. Use studies of deodorants containing 35% to 73% PG did not report any potential for eliciting irritation or sensitization. Propylene glycol generally did not induce sensitization reactions, including maximization tests, when tested at 12% to 86%. In a modified Draize sensitization study with 203 participants, PG (0.2 mL; concentration not stated) induced 19 cutaneous reactions at challenge. 1,2
Provocative Testing—Irritation and Sensitization
Propylene glycol
Reactions were observed at concentrations as low as 2% in provocative tests. 1 During the period 2000 to 2004, 308 patients, 111 males and 197 females, with contact dermatitis were patch tested using the European standard series and some additional chemicals, including PG. Propylene glycol, 5% in petrolatum, did not cause any positive reactions. The North America Contact Dermatitis Group (NACDG) performed a number of retrospective analyses on various dermatological conditions, and data on the relevance of positive reactions to PG were presented. These studies are summarized in Table 5. 2
Retrospective Analyses With Propylene Glycol
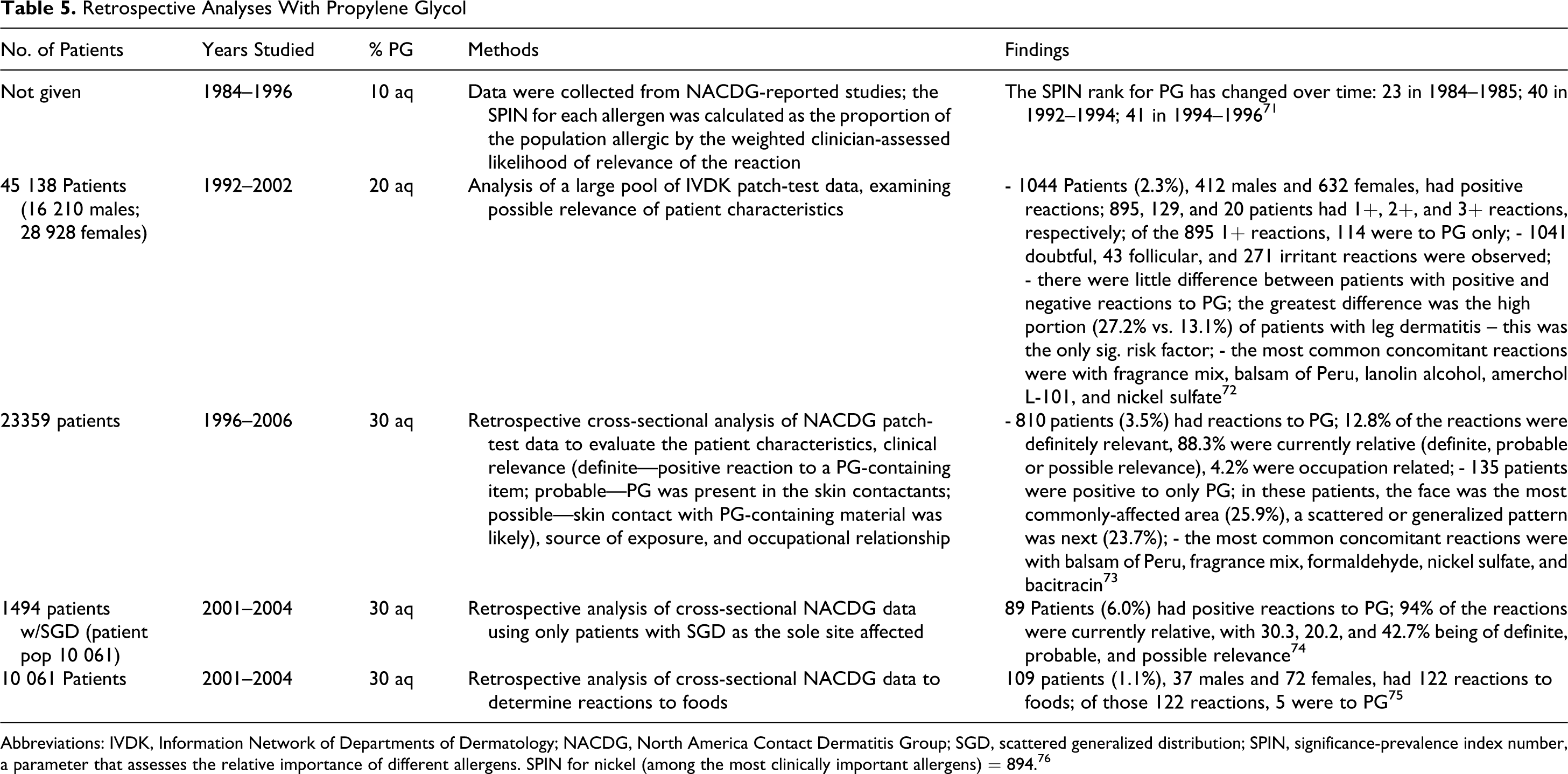
Abbreviations: IVDK, Information Network of Departments of Dermatology; NACDG, North America Contact Dermatitis Group; SGD, scattered generalized distribution; SPIN, significance-prevalence index number, a parameter that assesses the relative importance of different allergens. SPIN for nickel (among the most clinically important allergens) = 894. 76
Photoallergenicity
Propylene glycol
Over a 2-year period, 30 males and 52 females with photoallergic contact dermatitis were photopatch tested with a standard series of sunscreens as well as some additional chemicals, including PG (dose not given). The allergens were applied in duplicate on the back and covered with opaque tape for 24 hours. One set of test sites was irradiated with a UVA (320-400 nm) dose of 5 J/cm2. Propylene glycol did not produce a photoallergenic or contact allergy response. 2
Case Reports
Pentylene glycol (1,2-pentanediol)
A 68-year-old, nonatopic female developed facial dermatitis after using an eye cream that contained pentylene glycol (1,2-pentanediol), and patch test results were positive. Positive patch test reactions (+1) to 0.5% and 5% aqueous pentylene glycol were also reported. Except for 1 control participant with a follicular reaction to 5% pentylene glycol, reactions to 0.5% and 5.0% aqueous pentylene glycol were negative in a control group of 29 participants. 55
Propylene glycol
A few case reports have been described concerning PG and hand dermatitis or atopic dermatitis. The cases generally had positive patch test reactions to PG. Improvement was seen with the avoidance of PG-containing products. 2
Summary
The sixteen 1,2-glycols included in this safety assessment function primarily as skin and hair conditioning agents and viscosity increasing agents in personal care products, although caprylyl glycol and pentylene glycol also function as preservatives. The following five 1,2-glycols were reported to FDA as being used: caprylyl glycol, decylene glycol, pentylene glycol, 1,2-hexanediol, and C15-18 glycol. The results of a Personal Care Products Council industry survey indicate that ingredient use concentrations have range from 0.00003% (caprylyl glycol) to 10% (1,2-hexanediol). Use concentrations of pentylene glycol (up to 5%) were also included in this survey. The C15-18 glycol was included in this survey, but no uses or use concentrations were reported.
Safety test data from the CIR safety assessment on PG have been reviewed and are relevant to the safety assessment of other 1,2-glycols included in this report, based on structural similarities.
The Environmental Protection Agency (EPA) lists 1,2-butanediol as one of the reactive compounds in aerosol coatings (ie, aerosol spray paints) that contributes to ozone (O3) formation.
Stearyl glycol is prepared via the reaction of 2-hydroxyoctadecanoic acid with lithium aluminum hydride in dry tetrahydrofuran, and the production of 1,2-butanediol is via a continuous reaction and distillation operation. The available impurities data indicate that 1,2-butanediol is ≥99% pure and also contains water, 1,4-butanediol, and 1-acetoxy-2-hydroxybutane.
Information on the metabolism, distribution, and excretion of 1,2-butanediol following iv dosing indicate that, in rabbits, this chemical is metabolized slowly and excreted in the urine either as the glucuronide or unchanged; there was no evidence of tissue accumulation. Metabolites were not isolated from the urine of rabbits fed 1,2-butanediol in the diet. Based on metabolism modeling information on caprylyl glycol, 1,2-hexanediol, decylene glycol, and lauryl glycol, it is likely that C-oxidation, C-hydroxylation, glucuronidation, and beta-oxidation may take place to form corresponding metabolites. C-hydroxylation and beta-oxidation are more likely to be the favored metabolic pathways for the longer alkyl chain compounds, 1,2-decanediol and 1,2-dodecanediol, compared to the shorter alkyl chain length compounds, 1,2-hexanediol and 1,2-octanediol.
Following topical application of 5% caprylyl glycol in 70% ethanol/30% PG (5% Dermosoft Octiol in alcoholic solution) to female pig skin in vitro, approximately 97% of the test solution was dermally absorbed within 24 hours post-application. Based on dermal penetration modeling information on caprylyl glycol, 1,2-hexanediol, decylene glycol, and lauryl glycol, the default values for percentage dose absorbed per 24 hours were 80% for 1,2-hexanediol and 1,2-octanediol and 40% for 1,2-decanediol and 1,2-dodecanediol.
A skin penetration enhancement effect for caprylyl glycol, decylene glycol, pentylene glycol, 1,2-butanediol, and 1,2-hexanediol has been demonstrated in vitro.
There were no significant toxic effects in rats exposed for 7 hours to an atmosphere saturated with 1,2-butanediol. Acute oral toxicity data on caprylyl glycol and other 1,2-glycols for which data are available suggest that death would occur at relatively high doses (LD50 range: 2200 to >20 000 mg/kg). Reportedly, high (unspecified) oral doses of 1,2-butanediol caused narcosis, dilation of the blood vessels, and kidney damage in rats. Overt toxic effects were not observed in ethanol-dependent rats dosed orally with 2.74 g/kg 1,2-butanediol.
The available data suggest that 1,2-butanediol (LD50s up to 5.99 g/kg) and pentylene glycol (TDLo = 3.51 g/kg) are not significant acute ip toxicants. However, muscle incoordination was observed in rats at an ip dose of ∼2.94 g/kg. In an ip dosing study in which ED3 values for caprylyl glycol (1,2-octanediol), pentylene glycol (1,2-pentanediol), and 1,2-butanediol were compared, caprylyl glycol had the lowest ED3 value (1.5 mmole/kg), suggesting that its intoxication potency (ie, ability to induce ataxia) was greatest. In an acute dermal toxicity study involving rats, the LD50 for decylene glycol (SymClariol) was >2000 mg/kg. Prolonged application or repeated applications of 1,2-butanediol to the skin of rabbits did not result in overt toxic effects.
An NOEL of 50 mg/kg/d and anNOAEL of 300 mg/kg/d for systemic toxicity in rats were reported in a 28-day oral toxicity study on >98% caprylyl glycol (Dermosoft Octiol). The NOAEL was based on the findings of irritation on the pars nonglandularis and limiting ridge of the stomach; analogous structures do not exist in man. An NOAEL of 100 mg/kg/d was reported for rats in a 28-day oral toxicity study on decylene glycol (SymClariol). Short-term oral administration of 1,2-butanediol to rats yielded an NOAEL of 200 mg/kg/d. Reportedly, in another repeated dose study, the administration of large (unspecified) doses of 1,2-butanediol to rats caused irritation of the gastrointestinal tract. Signs of toxicity were noted at the highest dose of 22 g/kg/d in rats receiving 1,2-butanediol in the diet for up to 8 weeks; abnormalities were not observed in tissues from major organs. Intermittent oral administration of pentylene glycol to rats over a 28-week period yielded a TDLo of 2450mg/kg. In a 92- to 97-day oral toxicity study involving mice, rats, dogs, and monkeys dosed with a formulation containing PG (dose = 1000 mg/kg), there were no adverse effects on body weight, feed consumption, clinical pathology, histopathology, or adverse clinical observations.
Cetyl glycol (130 µg/mL) had a cytocidal effect on EAC cells, lauryl glycol (99 µmol/L) had a hemolytic effect on human erythrocytes, and pentylene glycol (5%) induced apoptosis in a human promyelocytic leukemia cell line in vitro.
Based on Draize test results, lauryl glycol has been classified as a severe ocular irritant. Undiluted 1,2-butanediol, but not 10% aqueous, induced ocular irritation in rabbits. Undiluted decylene glycol (SymClariol) induced corrosion when instilled into the eyes of rabbits. In an in vitro ocular irritation assay (HET-CAM), 1% SymClariol in neutral oil and caprylyl glycol (1% and 3%) in neutral oil were classified as nonirritants; however, a 50:50 (w/w) mixture of caprylyl glycol and 1,2-hexanediol was classified as a severe ocular irritant when evaluated at a concentration of 1% aqueous (effective concentration per ingredient = 0.5%) in the same assay. Together, the results of an NRR assay, the HET-CAM assay, and the REC assay indicated that a lash gel serum containing 3% pentylene glycol might be a slight ocular irritant.
In the guinea pig maximization test, results were negative for caprylyl glycol at a challenge concentration of 50% in petrolatum. Undiluted decylene glycol (SymClariol) was classified as a moderate skin irritant in rabbits but did not induce sensitization in the guinea pig maximization test at challenge concentrations of 2% and 5% in arachis oil or in the mouse local lymph node assay at concentrations of 5% to 50% in acetone/olive oil (4:1). Repeated applications of 1,2-butylene glycol to the skin of rabbits did not result in skin irritation, and results were negative for 1,2-hexanediol (10% to 100%) in the mouse local lymph node assay for evaluating sensitization potential.
An NOAEL of 1000 mg/kg for reproductive/developmental toxicity has been reported for 1,2-butanediol in rats dosed orally. In a prenatal developmental toxicity study involving rats, an NOEL of 300 mg/kg was reported for 1,2-hexanediol.
Caprylyl glycol >98% (Dermosoft Octiol) did not induce gene mutations in Chinese hamster V79 cells (concentrations up to 1480 µg/mL) and >98% caprylyl glycol (ADEKA NOL OG) did not induce chromosomal aberrations in Chinese hamster lung cells (concentrations up to 700 µg/mL) in vitro. Decylene glycol (SymClariol) was nongenotoxic in the Ames test, and 1,2-butanediol was not genotoxic in assays involving bacterial cells (doses up to 5000µg/plate) or mammalian cells (doses up to 0.9 mg/mL). Marked antitumor effects of cetyl glycol were observed in mice in vivo following ip doses of 80 mg/kg/d. Cetyl glycol (130 µg/mL) was found to have a cytocidal effect (irreversible cell degeneration) on cultured EAC cells.
Results were negative for skin irritation and sensitization potential in RIPTs in which 105 participants were patch tested with a lipstick containing 0.5% caprylyl glycol and 101 participants were patch tested with a leg and foot gel containing 0.5% 1,2-hexanediol. An in-use test of a body wash containing 0.15% 1,2-hexanediol did not result in skin irritation or sensitization reactions in 28 participants. 1,2-hexanediol/caprylyl glycol mixture (in preservative system) was nonsensitizing at a concentration of 0.5% or 15% in an RIPT involving 205 human participants. Skin sensitization also was not observed in another RIPT in which 56 participants were tested with a 50:50 (w/w) mixture of 1,2-hexanediol and caprylyl glycol (Symdiol 68; effective concentration per ingredient = 10%). Decylene glycol (SymClariol) did not induce skin irritation in 52 participants or sensitization (RIPT) in 55 participants patch tested at a concentration of 20% in petrolatum. However, SymClariol (1% in neutral oil) had low skin irritation potential when applied to scarified skin in a group of 10 participants, and very slight stinging potential when tested at concentrations of 1% and 2% in neutral oil in 10 participants. A foundation containing 0.112% pentylene glycol did not induce skin irritation or sensitization in an RIPT involving 101 participants. Positive reactions were observed in a patient patch tested with 0.5% and 5% 1,2-pentylene glycol but not in the control group.
Propylene Glycol
In mammals, the major pathway of PG metabolism is to lactaldehyde and then lactate via hepatic alcohol and aldehyde dehydrogenases. When PG was administered iv to human participants (patients), elimination from the body occurred in a dose-dependent manner.
Dermal penetration of PG from a ternary cosolvent solution through hairless mouse skin was 57% over a 24-hour period. Using thermal emission decay (TED)-Fourier transform infrared (FTIR) spectroscopy, it appeared that PG did not reach the dermis.
Propylene glycol is a penetration enhancer for some chemicals and, under some conditions, in human participants, and can act synergistically with other enhancers. The mechanism by which PG enhances penetration has not been identified.
Based on the 1994 safety assessment and more recent information, few toxic effects were seen in dosing with PG. The oral LD50 of PG was >21 g/kg for rats. The dermal LD50 of PG was >11.2 g/kg for mice and was 13 g/kg for rats. Mortalities were observed in mice at the highest ip dose of PG (10 400 mg/kg). All mice survived in a short-term study in which mice were given 10% PG in drinking water for 14 days, and all rats and mongrel dogs survived oral dosing with up to 3.0 mL 100% PG, 3 times per day, for 3 days. In a subchronic study, a dose of ≤50 000 ppm PG given in the feed for 15 weeks did not produce any lesions. Subchronic inhalation data reported some effects in rats due to PG exposure of 2.2 mg/L air for 6 hours/day, 5 days/week, for 13 weeks, but these effects were inconsistent and without dose-response trends. In the 1994 safety assessment, no toxic effects were reported in chronic studies when rats or dogs were given feed containing 50 g/kg or 5 g/kg, respectively, PG.
Undiluted PG was, at most, a slight ocular irritant. Dermal irritation studies were reported in the 1994 CIR final safety assessment and in the amended final safety assessment. In one study using nude mice, 50% PG may have caused skin irritation, while in another study, 100% PG was minimally irritating to hairless mice. Hypertrophy, dermal inflammation, and proliferation were also observed with 50% PG in nude mice. These effects were not seen in hairless mice with undiluted PG. Undiluted PG was at most a mild dermal irritant in a Draize test using rabbits with intact and abraded skin. No reactions to undiluted PG were observed with guinea pigs, rabbits, or Gottingen swine. Propylene glycol (concentrations not given) was negative in a number of sensitization assays using guinea pigs. In a study using guinea pigs, 0.5 mL PG was a weak sensitizer.
Oral administration of PG did not have any adverse reproductive or developmental effects when evaluated in mice at concentrations of ≤5%, rats at doses of ≤1600 mg/kg, rabbits at doses of ≤1230 mg/kg, or hamsters at doses of ≤1550 mg/kg. Embryonic development was reduced or inhibited completely in cultures of mouse zygotes exposed to 3.0 or 6.0 mol/L PG, respectively. A study examining induction of cytogenetic aberrations in mice reported an increase in the frequency of premature centrosphere separation with 1300 to 5200 mg/kg PG. In zygotes from PG-dosed mice, hyperploidy was increased.
Propylene glycol, ≤10 000 µg/plate, was not mutagenic in Ames tests with or without metabolic activation. Propylene glycol, tested at concentrations of 3.8 to 22.8 mg/mL, was a weak but potential inducer of SCEs, causing a dose-dependent increase in SCEs in a Chinese hamster cell line. However, in another SCE assay using human cultured fibroblasts and Chinese hamster cells with and without metabolic activation, PG was not mutagenic. Propylene glycol, 32 mg/mL, induced chromosomal aberrations in a Chinese hamster fibroblast line but not in human embryonic cells. Propylene glycol was not mutagenic in mitotic recombination or base pair substitution assays, or in a micronucleus test or a hamster embryo cell transformation assay.
Propylene glycol was not carcinogenic in a 2-year chronic study in which rats were given ≤50 000 ppm PG in the diet. Dermal application of undiluted PG to Swiss mice in a lifetime study produced no significant carcinogenic effects. Propylene glycol was not carcinogenic in other oral, dermal, and subcutaneous studies.
Combined exposure to PG and oleic acid synergistically enhanced the dermal penetration of both compounds. Addition of PG to an isopropanol vehicle enhanced the irritant reactions of benzoic acid; maximal enhancement was seen with 5% PG.
Propylene glycol–induced skin irritation reactions in normal participants and in patients. Reactions were observed at concentrations as low as 10% in predictive tests and 2% in provocative tests. Use studies of deodorants containing 35% to 73% PG did not report any potential for eliciting irritation or sensitization. Propylene glycol generally did not induce sensitization reactions when tested at 12% to 86%, although results were questionable in an RIPT of a deodorant containing 73% PG. Additionally, in a modified Draize sensitization study with 203 participants, PG (0.2 mL, concentration not stated) induced 19 cutaneous reactions at challenge. Propylene glycol did not produce a photoallergic response in a provocative photopatch test. Retrospective analysis of pools of patient patch test data indicated that ≤6.0% of patients tested had positive reactions to 30% aq PG. A few case reports concerning PG and hand dermatitis or atopic dermatitis have been described, and positive reactions were reported. Patients with diseased skin may be at risk with respect to developing irritation/sensitization reactions to PG.
Discussion
The available safety test data for 1,2-glycols indicate that they are not significant acute toxicants, are not significantly genotoxic, are noncarcinogenic, and are not significant dermal irritants, sensitizers, or photosensitizers. Data on the following 1,2-glycols were reviewed: caprylyl glycol, lauryl glycol, stearyl glycol, decylene glycol, pentylene glycol, 1,2-butanediol, 1,2-hexanediol, C15-18 glycol, and PG. Many of the studies included in this safety assessment are on PG. However, because increasing the chain length of the carbon backbone likely will not increase the potential for toxicity of longer chain 1,2-glycols, data on PG may be used to support the safety of all 1,2-glycols reviewed in this safety assessment.
Results from an in vitro skin penetration study on 5% caprylyl glycol in 70% ethanol/30% PG (5% Dermosoft Octiol) using female pig skin indicated significant percutaneous absorption of caprylyl glycol. Dermal penetration modeling data on caprylyl glycol (C8), 1,2-hexanediol (C6), decylene glycol (C10), and lauryl glycol (C12) predicted that skin penetration would decrease with increasing chain length. Acknowledging the dermal absorption of these compounds, the Expert Panel determined that evaluation of reproductive/developmental toxicity data would be key to determining a safe level. The results of oral reproductive/developmental toxicity studies on PG (C3), 1,2-butanediol (C4), and 1,2-hexanediol (C6) were negative, and there was no evidence of systemic toxicity in other oral repeated dose toxicity studies involving caprylyl glycol (C8), PG (C3), 1,2-butanediol (C4), pentylene glycol (C5), and decylene glycol (C10). Additionally, the available repeated dose toxicity data included some 28-day oral toxicity studies, but no 28-day dermal toxicity data, and dermal reproductive/developmental toxicity data also were not available. However, the Expert Panel agreed that these oral toxicity data could be used to evaluate the safety of 1,2-glycols in products applied to the skin in the absence of dermal studies, because 1,2-glycol blood levels following oral exposure would be higher when compared to dermal exposure and systemic toxicity was absent in the oral studies.
Dermal absorption modeling data predicted that skin penetration decreases with increasing chain length, significant dermal penetration of the longer chain 1,2-glycols may occur. Metabolism modeling data on caprylyl glycol, 1,2-hexandiol, decylene glycol, and lauryl glycol predicted that C-oxidation, C-hydroxylation, glucuronidation, and beta-oxidation may take place to form corresponding metabolites. The Expert Panel agreed that the negative oral reproductive/developmental toxicity (up to C6) and other negative oral repeated dose toxicity data (up to C10) may be extrapolated to longer chain 1,2-glycols. The negative results of bacterial/mammalian genotoxicity assays on caprylyl glycol, 1,2-butanediol, and decylene glycol were also considered, and the Expert Panel agreed that these data can also be extrapolated to longer chain 1,2-glycols as well. Thus, the modeling data predictions of decreased skin penetration of longer chain 1,2-glycols and those relating to their metabolic fate, together with the negative oral toxicity data on shorter chain 1,2-glycols and genotoxicity data, support the safety of all of the 1,2-glycols reviewed in this safety assessment in products applied to the skin.
The Expert Panel noted the potential for caprylyl glycol, decylene glycol, pentylene glycol, 1,2-butanediol, and 1,2-hexanediol to be penetration enhancers. Some cosmetic ingredients have been regarded as safe based on the fact that they do not penetrate the skin. If caprylyl glycol, decylene glycol, pentylene glycol, 1,2-butanediol, and 1,2-hexanediol enhance the penetration of such ingredients, then industry is advised to consider the impact of the penetration enhancing activity of these ingredients on the safety of other ingredients in formulation.
Conclusion
The CIR Expert Panel concluded that the following cosmetic ingredients are safe in the present practices of use and concentration described in this safety assessment:
caprylyl glycol
arachidyl glycol*
cetyl glycol*
hexacosyl glycol*
lauryl glycol*
myristyl glycol*
octacosanyl glycol*
stearyl glycol*
decylene glycol
pentylene glycol
1,2-butanediol*
1,2-hexanediol
C14-18 glycol*
C15-18 glycol
C18-30 glycol*
C20-30 glycol*
*Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in the group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of no financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
