Abstract
Phthalates are industrial chemicals used in many cosmetics. We evaluated an in vitro model for eye irritancy testing using the human corneal endothelial cell line B4G12. Cell proliferation and toxicity were assessed after exposing to di-n-butyl phthalate (DBP), benzyl butyl phthalate (BBP), di-2-ethylhexyl phthalate (DEHP), diisodecyl phthalate (DIDP), di-n-octyl phthalate (DnOP), and di-isononyl phthalate (DINP). Gene expression and secretion of inflammatory cytokines were evaluated after exposure to DBP. Decreased cell proliferation was observed for the phthalates DBP, BBP, and DEHP, and cell toxicity was observed for DBP and BBP. Upon DBP exposure at nontoxic concentrations, a significant increased gene expression and cytokine cell secretion were observed for interleukin-1β (IL-1β) and IL-8, and also an increased IL-6 secretion was observed. In conclusion, the human corneal endothelial cell line B4G12 may be a potential model for inflammatory eye irritancy testing of phthalates.
Introduction
The ocular surface is a complex system consisting of corneal and conjunctival epithelium, the underlying stroma, and associated cells. Eye irritation is a combination of complex events that possibly impact the cornea, conjunctival and corneal epithelia, and the corneal endothelial cells, which can be predicted using several endpoint observations such as cell proliferation and membrane integrity. The applicability and predictive capacity of 5 nonhuman-related in vitro tests for eye irritation have been previously evaluated and found to have good predictive capacity, reproducibility, and reliability when compared with the Draize eye irritation test in rabbits. 1 Different types of cells including rabbit corneal epithelial cells 2,3 and human corneal endothelial cells 4 have been cultured to assay the eye irritation potential of various compounds.
Mild eye irritants act by disrupting or damaging only the surface cells of the outermost layer of the eye (the corneal epithelium), and therefore corneal epithelial cell cultures are a promising alternative for in vitro eye irritation testing and to predict topical irritation of the eye. However, some cosmetic and personal care components, such as most phthalates, are to some degree lipophilic and might penetrate through the corneal stroma and into the corneal endothelial cell layer. Therefore, the effects on corneal endothelial cells are also relevant when exploring human eye irritation caused by the infiltration of chemicals, such as phthalates, through the epithelial cells and the stroma.
The human corneal endothelial cell line B4G12 is a recently cloned cell line that resembles differentiated human corneal endothelial cells in vivo when cultured in vitro. 5,6 The B4G12 cells were found to have a typical polygonal morphology, form closed monolayers with high cell density, and, like human corneal endothelial cells in vivo, strongly adhere to the culture substratum. 5,6 Furthermore, the B4G12 cells express functional proteins that are typically present in corneal endothelial cells, including the junctional protein ZO-1, Na+, K+-ATPase, and vinculin, a membrane-cytoskeletal protein involved in cell adhesion. 5,6 B4G12 cells can be grown under serum-free conditions. 5,6 This is important because corneal cells in vivo are not in contact with blood serum and also because serum can be inconsistent in content and quality and may be a potential source of immunogens and pathogens.
The assessment of ocular surface irritation is particularly important for personal care and cosmetic products, which often lack early clinical signs of eye irritation and are defined as nonirritants, but may cause eye irritation when applied repeatedly for long periods. Thus, assessment of subtle degrees of eye irritation requires determination of early biological endpoints. Gene expression might be an early biomarker of such irritation response, which might not be detected by looking only at membrane integrity, cell viability, or growth. Inflammation is one of the most important elements involved in irritation of the human eye. 7,8 Thus, gene expression of cytokines involved in the inflammatory reaction could be an indicative of eye irritation. 9
The interleukin-1 (IL-1) gene family is a group of potent cytokines that function as the major mediators of inflammation and immune responses. Another IL family, IL-6, acts both as a proinflammatory and anti-inflammatory cytokine and stimulates the acute-phase reaction, which enhances the innate immune system and protects against tissue damage. 10 Finally, IL-8 is an important inflammatory mediator reported to be a potent chemotactic and cell activator for neutrophils. 11
In order to assess the eye irritating potential of phthalates in vitro, we assessed cell proliferation and cell toxicity using sulforhodamine B (SRB) colorimetric and lactate dehydrogenase (LDH) assays, respectively, using the human corneal endothelial cell line B4G12. Furthermore, gene expression of the inflammatory cytokines IL-1β and IL-8 was determined using real time-polymerase chain reaction (RT-PCR), and cytokine cell secretion of IL-1β, IL-6, and IL-8 was measured with enzyme-linked immunosorbent assay (ELISA). Thus, the present study describes a potential in vitro method for human eye irritation testing.
Methods
Materials
Human endothelial serum free media and cell dissociation buffer were purchased from Invitrogen, Life Technologies (Taastrup, Denmark). Fetal calf serum (FCS) was purchased from Gibco, Life Technologies (Taastrup, Denmark). Basic Fibroblast Growth Factor (bFGF) (human recombinant) was obtained from Sigma-Aldrich (Broendby, Denmark) and used as media supplement. Complete protease inhibitor cocktail, used as trypsin inhibitor, was purchased from Roche (Hvidovre, Denmark). Laminin (mouse) and chondroitin-6 sulphate for coating the plates were purchased from Sigma-Aldrich (Broendby, Denmark). The phthalates (purity ≥98%) used in this study, di-n-butyl phthalate (DBP), benzyl butyl phthalate (BBP), di-2-ethylhexyl phthalate (DEHP), diisodecyl phthalate (DIDP), di-n-octyl phthalate (DnOP), and di-isononyl phthalate (DINP) were all purchased from Sigma-Aldrich (Broendby, Denmark). All phthalates were dissolved in dimethyl sulfoxide (DMSO) as 50 mmol/L stock solution and diluted with media for exposure. A solvent control (SC), consisting of 0.2% DMSO in media, was included in each assay.
Cell Culture
The human corneal endothelial cell line B4G12 was received upon request from Ms Monika Valtink (University of Technology, Dresden, Germany). B4G12 cells were cultured with and without 5% FCS supplement as described elsewhere. 5,6
Cell Culturing for Cell Proliferation and Toxicity Analyses
For the determination of cell proliferation (using the SRB assay, see below) and cell toxicity (using the LDH assay, see below) the B4G12 cells were seeded out in 96-well plates at a density of 20 000 cells/well (for exposure with 5% FCS supplement) and 40 000 cells/well (for exposure without FCS supplement). After cultivating for 24 hours, B4G12 cells were exposed to fresh media with 1 to 200 µmol/L of the different test phthalates as well as the SC for another 24 hours. Three independent assays were performed with 3 determinations at each concentration.
Colorimetric Assay of SRB (Cell Proliferation)
The SRB binds to protein components of cells fixed with trichloroacetic acid (TCA) and is used to determine cell proliferation based on the measurement of cellular protein content. The SRB assay was performed as described. 12 In brief, after exposure to phthalates (1-200 µmol/L) and SC, the media were removed and the cells on the plates were fixed using 10% TCA, followed by SRB staining. The protein-bound dye was dissolved in 10 mmol/L Tris base, and optical density value was determined at 490 nm by an ELx800 absorbance microplate reader (Bio-Tek Instruments Inc, Holm&Halby, Broendby, Denmark). The average inter-coefficient of variation (CV) was below 19%.
Lactate Dehydrogenase Assay (Cell Toxicity)
The LDH is a cytoplasmic enzyme that is rapidly released into the media when the plasma membrane is damaged. The LDH assay is a colorimetric assay and quantifies cell death and cell lysis, based on the measurement of LDH activity. The assay was performed using the LDH cytotoxicity detection kit from Roche (Hvidovre, Denmark), according to the suppliers’ instructions. In brief, after exposure to phthalates (1-200 µmol/L) and SC, the media were removed and the cells incubated with the substrate mixture from the kit. The LDH activity was determined in a coupled enzymatic reaction converting a yellow tetrazolium salt into a red formazan salt. The absorbance was read using the ELx800 absorbance micro plate reader. The average inter-CV was below 18%.
Cell Culturing for Cytokine Gene Expression and Secretion Analyses
Since no difference in cell proliferation and toxicity was observed with and without FCS supplementation at 1 to 10 µmol/L DBP, the gene expression (RT-PCR, see below) and cell secretion (ELISA assay, see below) of the inflammatory cytokines IL-1β, IL-6, and IL-8 were assessed for DBP cell exposure to these nontoxic concentrations with 5% FCS supplementation. The cells were seeded in 12-well plates at a density of 48 000 cells/well (with 5% FCS supplement). After 24 h of culturing, B4G12 cells were exposed to fresh media with 1, 5, or 10 µmol/L DBP as well as the SC for another 24 hours. The media were removed and stored at −80°C until determination of cytokine secretion using ELISA. The cells were harvested and lysed for subsequent RNA isolation (GenElute Mammalian Total RNA miniprep kit) following the instructions from the manufacturer (Sigma-Aldrich, Broendby, Denmark), and the total RNA was stored at −80°C. Two independent cell assays were performed with triple determinations at each concentration.
Real Time-Polymerase Chain Reaction (Cytokine Gene Expression)
RNA reverse transcription was performed using the Superscript III first strand synthesis system (Invitrogen, Life Technologies, Taastrup, Denmark) and the resulting complementary DNA (cDNA) was frozen at −80°C until use. RT-PCR for quantification of IL-1β and IL-8 was employed with the on-line detection of the PCR reaction based on fluorescence monitoring (Lightcycler, Roche, Hvidovre, Denmark) as described. 13 Hybridization probes were used to monitor the amount of specific target sequence. Both the primers (Table 1) and probes (Table 2) were designed by TIB MolBiol (Berlin, Germany) and purchased from DNA Technology (Aarhus, Denmark). The expression of the target genes coding for IL-1β and IL-8 was normalized to the steady expression of the housekeeping gene β-2-microglobulin (β2M). The genes were quantified in sequential separate runs.
Sequences of the Forward and Reverse Primers for IL-1β, IL-8, and β2M

Abbreviations: IL, interleukin; β2M, β-2-microglobulin.
Sequences of Fluorescent Labeled Hybridization Probes for IL-1β, IL-8, and β2M
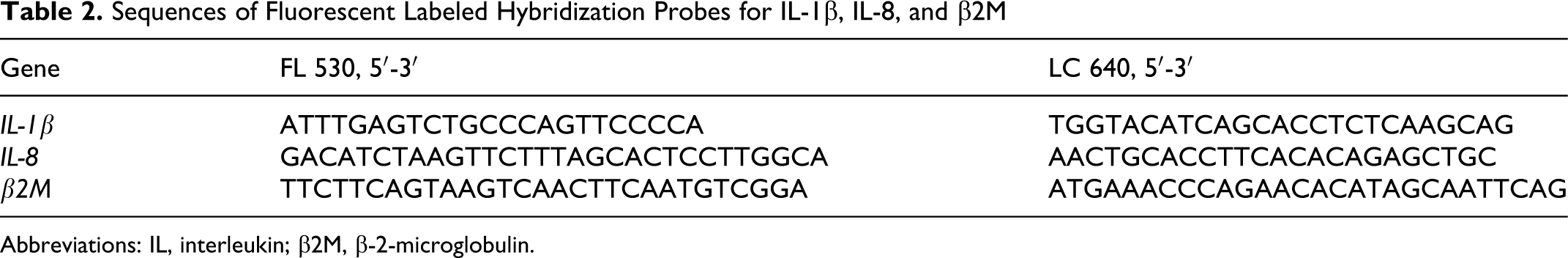
Abbreviations: IL, interleukin; β2M, β-2-microglobulin.
For quantification of the cytokines, plasmids, containing cDNA of the cytokine gene to be analyzed, were prepared as described. 13 In short, the cytokine cDNA fragment were ligated into pGEM-T Easy Vectors according to the protocol and transformed into competent Escherichia coli cells (genotype K12; lac-pro, supE, thi, hsdD5/F’, traD36, proA + B, laci q, and lac Z▵ M15). The plasmids were harvested upon multiplication in the transformed E. coli cells, linearized with the Sca I enzyme, and stocks of 0.01 μg/μl plasmid were stored at −20°C. Before use, this stock was diluted 10 to 106 times for the standard curve.
The signal from the test samples was quantified using the standard curve of the cytokine specific cloned plasmid by the Lightcycler software (version 3.0) by the use of the Fit Points method and proportional baseline adjustment.
Because of inter-run variations and drifting basal levels, the results are expressed relative to the basal levels after normalization against the housekeeping gene. The basal noninduced levels were determined in samples incubated with SC (DMSO). The average inter-CV was below 22%.
Enzyme-Linked Immunosorbent Assay (Cytokine Secretion)
Secretion of the inflammatory cytokines IL-1β, IL-6, and IL-8 was determined using commercially available QuantiGLO kits from R&D Systems (Abingdon, UK) following the manufactures protocol. The ELISA assays employ the quantitative sandwich enzyme immunoassay technique. In short, the cytokine present in the media from exposed cells binds to a specific monoclonal antibody precoated on a microplate. Subsequently, an enzyme-linked polyclonal antibody specific for the cytokine and an enhanced luminol/peroxide substrate solution were added. The LUMIstar luminometer (Ramcon, Birkerød, Denmark) was used to measure the intensity of the light emitted, which is proportional to the amount of cytokine bound in the initial step. The average inter-CV was below 28%.
Statistical Analysis
For cell proliferation and cell toxicity, each phthalate was tested in the range of concentrations in 3 independent experiments. Within each experiment, each concentration was run in triplicate. If one of the values deviated more than 30% from the other 2 values, the mean was calculated from the 2 wells only to ensure a realistic percentage deviation according to the Forensic Toxicology Laboratory Guidelines. 14 Mean values from the independent experiment were used for the statistics (Student t test, see below).
For cytokine gene expression and secretion, 2 independent cell assays were performed testing each DBP concentration in triplicate. Subsequent ELISA and RT-PCR assays were performed on each of the 3 cell determinations, and the statistical analysis was done on the obtained values at each concentration from the 2 independent experiments. If one of the values deviated more than 30% from the other values, the statistics was calculated from the other wells only resulting in 4 to 6 quantifications.
Due to the relatively small sets of data, the Student t test in Microsoft Excel was used to test for differences between cells treated with SC and phthalates. The statistical term P < .05 was used to indicate biologically relevant differences.
The Inhibitory Concentration for half maximal response (IC50) values were calculated by fitting dose-response data to a 4-parameter sigmoidal Hill curve using Sigma Plot (SPSS, Chicago, Illinois).
Results
Cell Proliferation
The DBP, BBP, and DEHP exposure significantly decreased B4G12 cell proliferation at 50 to 200 µmol/L both with and without FCS supplementation (Figure 1). The mean IC50 values were significantly lower using the FCS-free conditions compared with the 5% FCS supplementation conditions (Table 3).
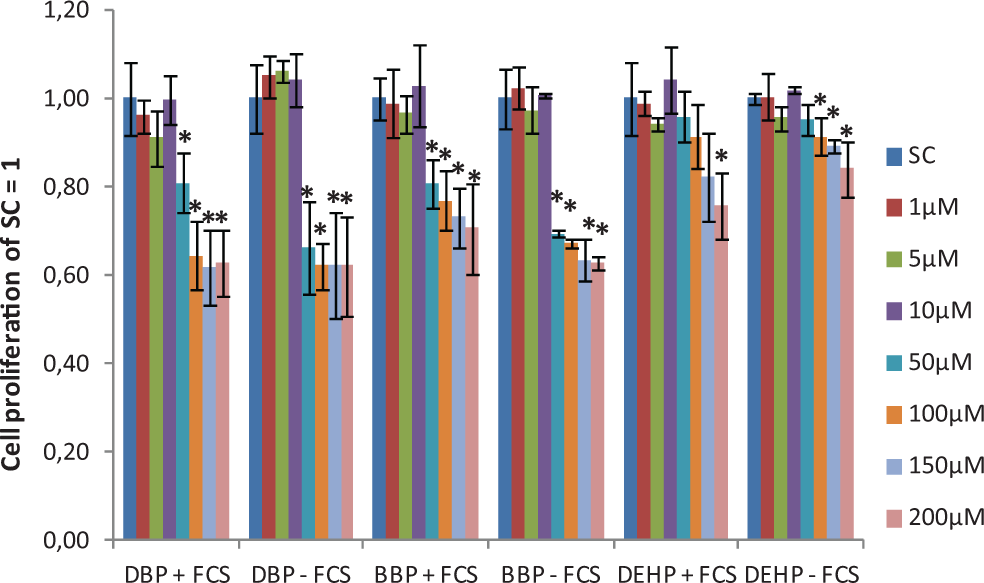
Cell proliferation as examined by the SRB assay after phthalate exposure with and without 5% FCS supplement. Data are expressed as fold over SC and presented as mean ± standard deviation from 3 independent assays. *Significantly different from SC (P ≤ .05). FCS indicates fetal calf serum; SRB, sulforhodamine B; SC, solvent control.
Mean IC50 Value of DBP, BBP, and DEHP Exposure With and Without 5% FCS Supplement a
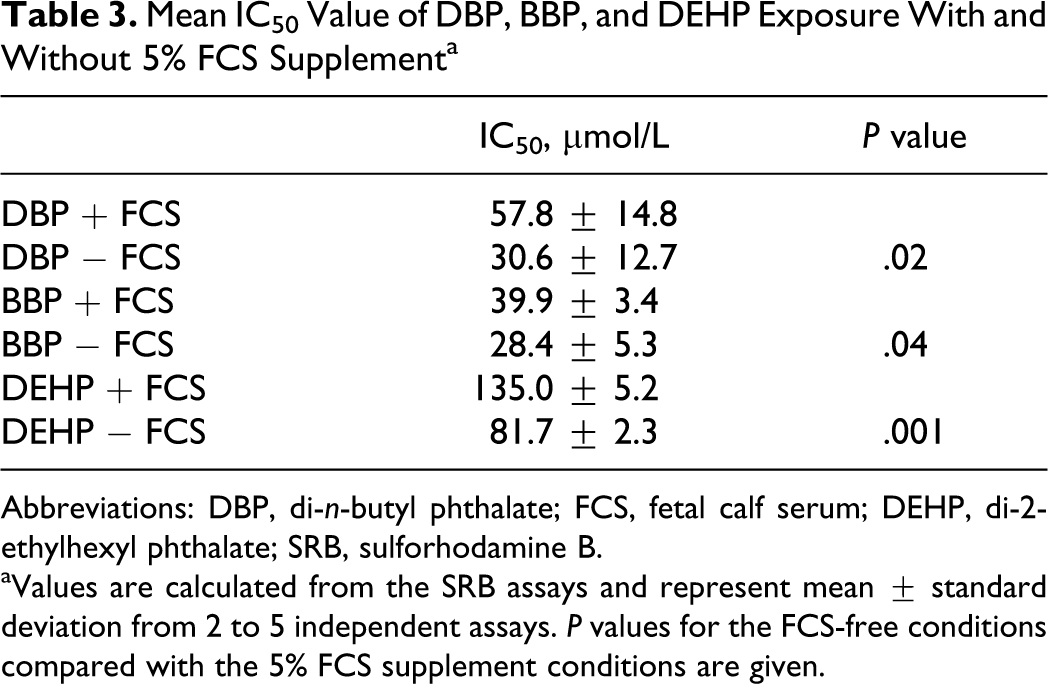
Abbreviations: DBP, di-n-butyl phthalate; FCS, fetal calf serum; DEHP, di-2-ethylhexyl phthalate; SRB, sulforhodamine B.
aValues are calculated from the SRB assays and represent mean ± standard deviation from 2 to 5 independent assays. P values for the FCS-free conditions compared with the 5% FCS supplement conditions are given.
No effect on cell proliferation of B4G12 cells at concentrations up to 200 µmol/L was observed after exposure to DIDP, DnOP, and DINP as determined by the SRB assay (data not shown).
Cell Toxicity
LDH leakage was observed for ≥50 µmol/L DBP and ≥150 µmol/L BBP without FCS supplementation (Figure 2). With FCS supplementation, LDH leakage was only observed for 200 µmol/L DBP (Figure 2).
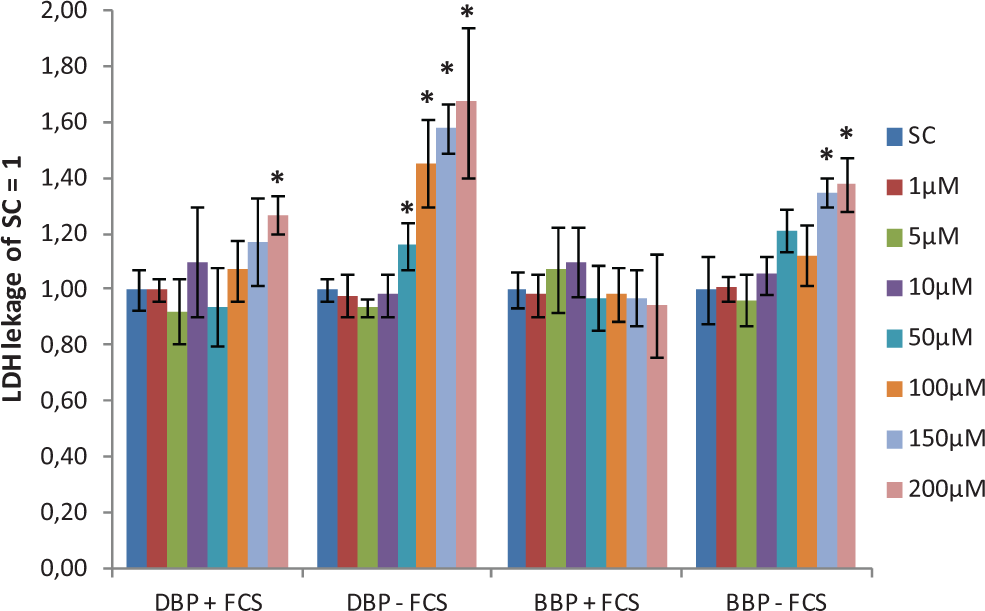
The LDH leakage after phthalate exposure with and without 5% FCS supplement. Data are expressed as fold over SC and presented as mean ± standard deviation from 3 independent assays. *Significantly different from SC (P ≤ .05). FCS indicates fetal calf serum; LDH, lactate dehydrogenase; SC, solvent control.
LDH leakage was not observed for DIDP, DnOP, DINP, and DEHP at any concentration as determined by the LDH assay (data not shown).
Cytokine Gene Expression and Cytokine Secretion
Cytokine messenger RNA (mRNA) gene expression and secretion were only determined after exposure to DBP + 5% FCS at concentrations that did not affect cell proliferation (1-10 µmol/L).
Compared with the SC, the expression of IL-1β mRNA (Figure 3A) was significantly increased at all tested concentrations, and the level of IL-8 mRNA (Figure 3B) was significantly induced at 1 and 10 µmol/L. The gene expression of IL-6 mRNA could not be determined because of primer and probe methodological problems.
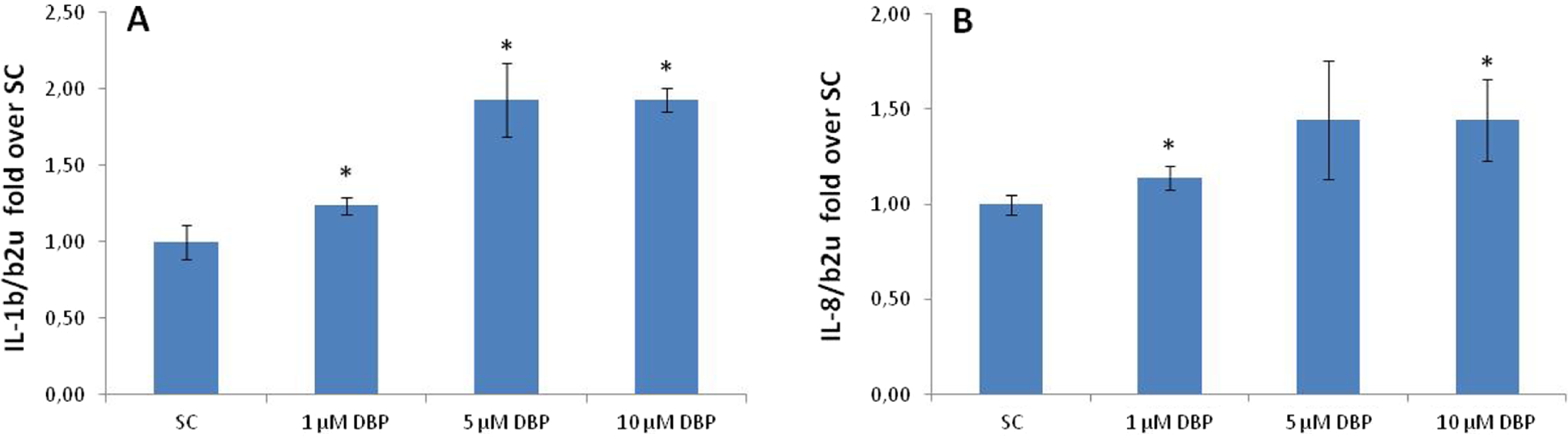
Cytokine gene expression after exposure to DBP + 5% FCS supplement as determined by RT-PCR. The expression of (A) IL-1β and (B) IL-8 was normalized to β-2-microglobulin. Data are expressed as fold over SC and presented as mean ± standard deviation from 4 to 6 quantifications. *Significantly different from SC (P ≤ .05). FCS indicates fetal calf serum; DBP, di-n-butyl phthalate; SC, solvent control; RT-PCR, real time-polymerase chain reaction; IL, interleukin.
A significant increase in secretion of IL-8 protein was observed at 5 and 10 µmol/L DBP (Figure 4B). Since both the basal- and the DBP-induced IL-1β and IL-6 secretion were at a very low level, the quantification of these cytokines is an approximation and conclusions drawn from these data should be made with caution. However, there seemed to be a significant increase in the secretion of IL-1β at 1 µmol/L DBP (Figure 4A) and of IL-6 at 10 µmol/L DBP (Figure 4C).
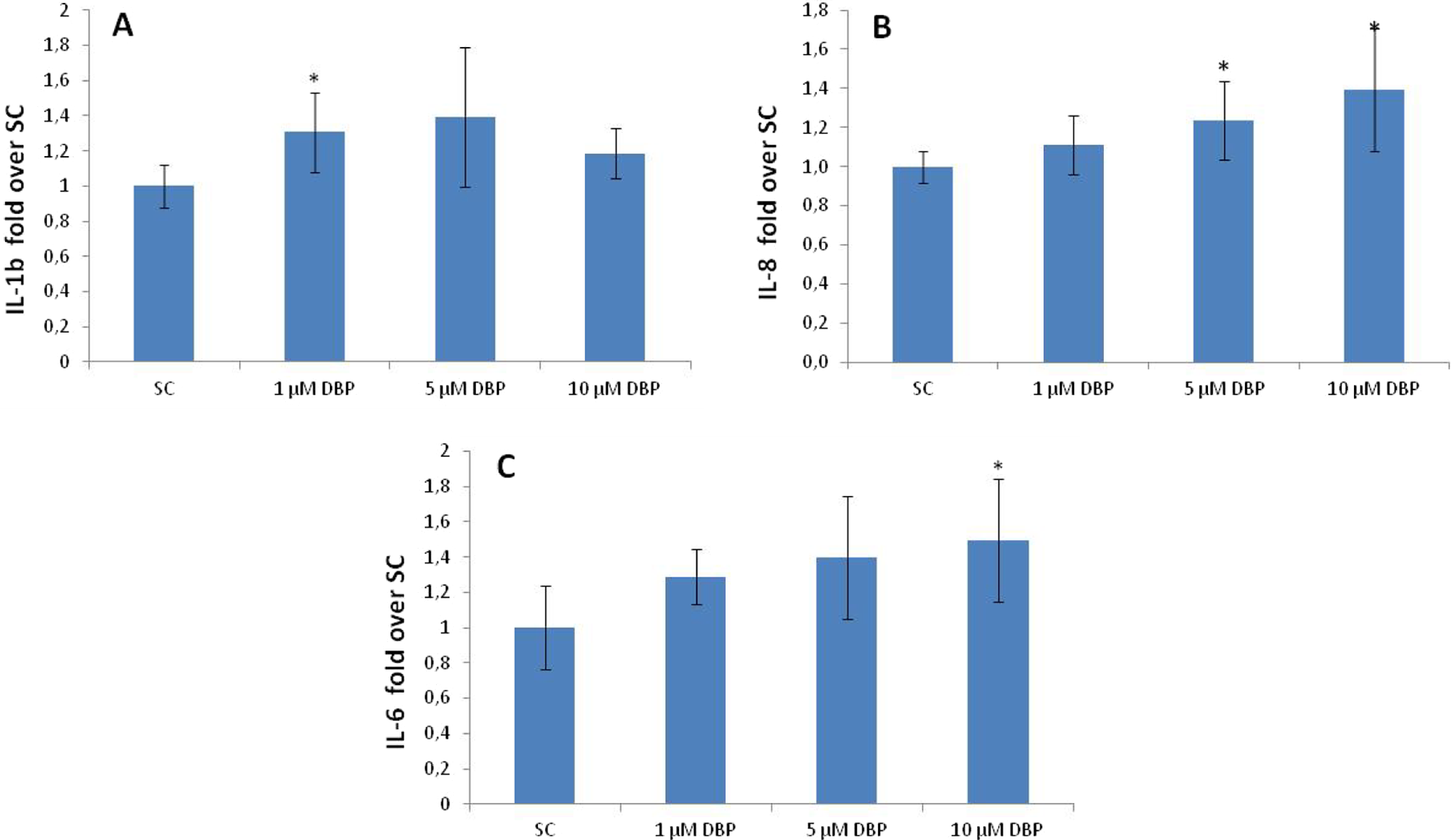
Cytokine (A) IL-1β, (B) IL-8, and (C) IL-6 secretion after exposure to DBP + 5% FCS supplement as determined by ELISA. Data are expressed as fold over SC and presented as mean ± standard deviation from 2 independent assays. *Significantly different from SC (P ≤ .05). FCS indicates fetal calf serum; SC, solvent control; ELISA, enzyme-linked immunosorbent assay; IL, interleukin; DBP, di-n-butyl phthalate.
Discussion
In recent years, many of the potential health hazards of phthalate exposure in humans have been the subject of extensive research. 15,16 However, the potential of phthalates to cause eye irritation in humans has not been as thoroughly investigated. Based on the results of a literature search, the eye irritation potential of phthalates has, to our knowledge, only been assessed with Draize rabbit eye irritation test. 17 –22 However, the Draize test has been criticized for many years because of its use of animals, and efforts have been made to replace it. 23 One possible alternative method is to use cultured cells for testing cell function and cytotoxicity after exposure to potential irritants. 23
Thus, for our current investigations of phthalate-induced early-onset eye irritation, we selected as the test system the human corneal endothelial B4G12 cell line, which was established from human eyes and resembles human corneal endothelial cells in vivo. 5,6
We used SRB cell proliferation and LDH cell toxicity assays to assess B4G12 cell viability after in vitro exposure to several phthalates. Furthermore, we determined cytokine mRNA gene expression and cytokine secretion in the B4G12 cells following phthalate exposure. We observed effects of the selected phthalates in all 4 assays. These results in the B4G12 cell test system may reflect human eye response to phthalate exposure.
Some recent work has evaluated the impairment of cell growth by exposure to phthalates. Naarala and Korpi 24 found that DBP and diisobutyl phthalate (DIBP) could significantly decrease the viability of murine macrophages determined by the measurement of DNA content, while DEHP and BBP had no significant effect on cellular viability. In the present study, we also observed decreased B4G12 cell growth after exposure to DBP, but in contrast to Naarala and Korpi, 24 we also observed decreased cell growth after exposure to DEHP and BBP. The use of different cell types and assay setup may explain these diverse observations.
Previous work has determined that the morphology of B4G12 cells changed when the cultivation was switched from FCS-free to FCS-supplemented conditions, and that the cells became more analogous to the heterogeneous human corneal endothelial cell population under FCS-supplemented conditions. 5 It is well known that FCS promotes the growth of cells. However, one of the disadvantages of serum (including FCS) is that it contains a mixture of unknown compounds including chemical binding proteins, which may result in an underestimation of cytotoxic effects. Furthermore, corneal cells are not in contact with serum in vivo.
Therefore, in the present study, we used both FCS-free and FCS-supplemented conditions to further evaluate the effects of FCS on the in vitro responses of the B4G12 cells to phthalate exposure. We found that the IC50 values for DBP, BBP, and DEHP were significantly lower in FCS-free cultures compared with the FCS-supplemented cultures, indicating that the cell proliferation was inhibited at lower phthalate concentrations under FCS-free conditions. Furthermore, exposure to BBP and DBP resulted in significantly increased LDH leakage in FCS-free B4G12 cell cultures, while there was no effect in FCS-supplemented cultures, except for DBP at the highest tested concentration. Thus, B4G12 cells seemed to be more sensitive to phthalate exposure in FCS-free conditions compared with FCS-supplemented conditions.
The IL cytokines such as IL-1β, IL-6, and IL-8 are the potent mediators of the inflammatory response in many tissues including the eye. 25 Increased IL-1β is a common biomarker in different ocular allergic diseases, suggesting that IL-1β plays a critical role in ocular allergic inflammation. 26 The IL-1β was found in the epithelium, stroma, and endothelium of the cornea at both the mRNA and protein levels. 27 Elevated IL-6 levels are found in the conjunctival epithelium of patients with Sjögren syndrome and dry eye syndrome, 28,29 in UV-B irradiated human corneal epithelial cells and human conjunctival epithelial cells 30 and in human corneal epithelial cells infected with herpes simplex virus-1. 31 The expression and release of IL-8, which is induced by tumor necrosis factor and IL-1, were observed in many different tissues including corneal endothelial cells 32 and shown to be a useful marker of early signs of human eye irritation. 33,34 In human corneal epithelial cell cultures, an increase in IL-1β secretion was observed after exposure to lipopolysaccharide 35 and increased IL-8 secretion was observed after exposure to make-up removers (10 commercially available and 2 prepared for analysis with the aim to produce itching; no information on the contents were given) and chemically and nonchemically preserved tear substitutes. 33,34 Therefore, gene mRNA expression of ILs involved in the inflammatory reaction could be indicative of eye irritation. In fact, cytokine gene expression and secretion have been investigated in human corneal epithelium cell cultures 30,31,33 –35 but, to our knowledge, have not been studied in corneal endothelial cell cultures.
Thus, in the present study, we evaluated gene mRNA expression and secretion of several IL cytokines after exposure of B4G12 human corneal endothelial cell to phthalates. Significantly induced gene expression of both IL-1β and IL-8 mRNA compared with the SC was observed after exposure to DBP at concentrations that did not affect cell viability. We also observed an increased secretion of IL-8 protein levels associated with exposure to DBP. Both IL-1β and IL-6 were only secreted at low levels upon exposure to DBP, but a tendency toward increased secretion of both proteins was observed. Thus, even though no effect on cell growth was observed at these concentrations, the increased expression of these cytokine genes as well as cytokine secretion may indicate early eye irritation. We also found that the levels of IL-1β and IL-6 secretion from the cells were very low compared with the IL-8 protein levels. This may result from the fact that IL-8 protein persists in the immediate environment of the cells from where it is released because of resistance to inactivation and slow clearance. 11
Animal studies including the Draize rabbit eye irritation test have reported that BBP, DBP, and DEHP caused mild eye irritation, 17,18,19 whereas the phthalates DIDP, DINP, and DnOP only caused minimal eye irritation. 20 –22 Thus, our results using in vitro assays with the human corneal endothelial B4G12 cell line are similar to the in vivo data obtained in rabbits.
In summary, our data showed that the phthalates DBP, BBP, and DEHP adversely affected growth and viability of the B4G12 cells. Furthermore, an increase in gene mRNA expression and secretion of the IL cytokines IL-1β, IL-6, and IL-8 occurred after exposure to DBP at concentrations that did not affect B4G12 cell viability. However, further work is needed to confirm that these B4G12 human corneal endothelial cell in vitro assays can predict early eye irritation resulting from exposure to phthalates and other potential ocular toxicants in humans.
Footnotes
Acknowledgments
The authors thank Ms Monika Valtink (University of Technology, Dresden, Germany) for providing the B4G12 cells.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by the Danish Network In Vitro Toxicology and Alternative Methods; financed by grant no 09-065084 from The Danish Board of Research and Innovation.
