Abstract
Cocamidopropyl betaine (CAPB) and related amidopropyl betaines are zwitterions used mainly as surfactants in cosmetics. These cosmetic ingredients are similar in their chemistry, in particular with respect to the presence of 3,3-dimethylamino-propylamine (DMAPA) and fatty acid amidopropyl dimethylamine (amidoamine) impurities, which are known as sensitizers. The CIR Expert Panel concluded that because these ingredients present no other significant toxicity, when formulated to be nonsensitizing (which may be based on a quantitative risk assessment), these ingredients are safe for use as cosmetic ingredients in the practices of use and concentration of this safety assessment.
Introduction
Cocamidopropyl betaine (CAPB) is a zwitterion used primarily as a surfactant in cosmetic products. A safety assessment for CAPB was published by the Cosmetic Ingredient Review (CIR) in 1991. 1 At that time, the CIR Expert Panel (the Panel) concluded that CAPB is safe for use in rinse off cosmetic products at the current levels of use, and the concentration of use for cosmetic products designed to remain on the skin for prolonged periods of time (leave-on products) should not exceed 3.0%. Because raw material CAPB is commonly supplied to product finishing houses as a 30% preformulation solution, a 3% solution would correspond to a 10% solution of a full-strength CAPB raw material solution. Frequently, these preformulation solutions are described as having an “activity” of the ingredient (eg, typical raw material CAPB has an activity of 30%). Accordingly, to prepare a 3% solution of a CAPB, from a CAPB preformulation solution with 30% activity, the preformulation solution would need to be diluted by a factor of 10.
Based on new published data that described sensitization in patients from use of rinse off products, new uses in aerosol products, and a substantial increase in the number of uses, the Panel reopened the final report on CAPB in 2007. The following report is a compilation of new data and summary data from the original safety assessment on CAPB and related amidopropyl betaines. Because of chemical similarities to CAPB, the available data may be extrapolated to all of the following related aminopropyl betaines, in a process termed read across:
almondamidopropyl betaine,
apricotamidopropyl betaine,
avocadamidopropyl betaine,
babassuamidopropyl betaine,
behenamidopropyl betaine,
canolamidopropyl betaine,
capryl/capramidopropyl betaine,
coco/oleamidopropyl betaine,
coco/sunfloweramidopropyl betaine,
cupuassuamidopropyl betaine,
isostearamidopropyl betaine,
lauramidopropyl betaine,
meadowfoamamidopropyl betaine,
milkamidopropyl betaine,
minkamidopropyl betaine,
myristamidopropyl betaine,
oatamidopropyl betaine,
oleamidopropyl betaine,
olivamidopropyl betaine,
palmamidopropyl betaine,
palmitamidopropyl betaine,
palm kernelamidopropyl betaine,
ricinoleamidopropyl betaine,
sesamidopropyl betaine,
shea butteramidopropyl betaine,
soyamidopropyl betaine,
stearamidopropyl betaine,
tallowamidopropyl betaine,
undecyleneamidopropyl betaine, and
wheat germamidopropyl betaine.
Chemistry
Definition and Structure
The general structure of amidopropyl betaines is as shown in Figure 1, where RCO- represents the fatty acids derived from various oils. 2 For example, for CAPB (CAS No. 61789-40-0), RCO- represents the fatty acids derived from coconut oil. Table 1 presents the definitions and structures of CAPB and related amidopropyl betaine ingredients.
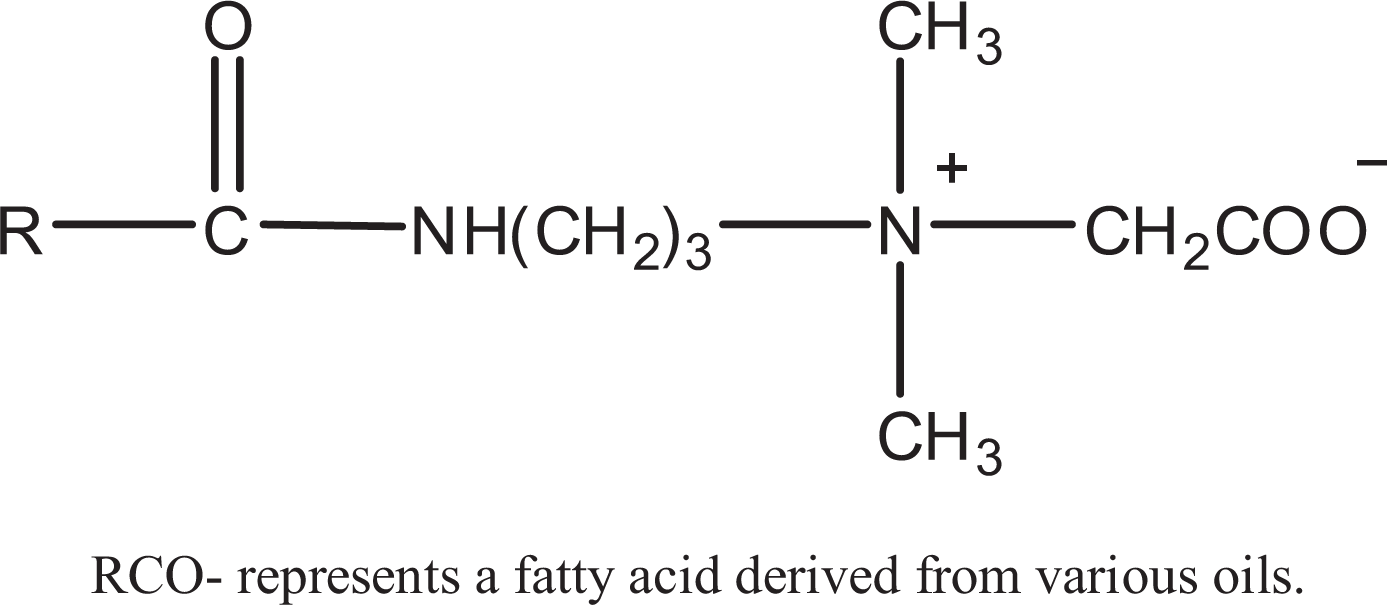
Amidopropyl betaine.
Definitions, Structures, and Functions for CAPB and Related Amidopropyl Betaine Ingredients 2

Technical names for CAPB and its related amidopropyl betaines, as well as the functions these ingredients perform in cosmetics, are found in Table 2. There are numerous trade names and trade name mixtures containing CAPB and its related amidopropyl betaines. 2
Technical Names for CAPB and Related Amidopropyl Betaines 2

Physical and Chemical Properties
The CAPB is a clear, pale yellow liquid of medium viscosity (300-600 cps), with a slight fatty odor.3,4 The CAPB has a boiling point of 230°F, a specific gravity of 1.04 relative to water, and no flash point. 5 The CAPB is soluble in water, ethanol, and isopropanol and insoluble in mineral oil. 3
The CAPB is supplied as a solution in water and with sodium chloride (see Table 3). The concentration of CAPB in such supplied material is described by its activity. 6 The concentration of cosmetic-grade CAPB (active concentration) is what is left in the supplied solution after water (62%-66%) and sodium chloride (4.6%-5.6%) have been accounted for, which is ∼30% of the supplied solution. In this report, unless a concentration has been reported as being active, a concentration of CAPB in solution will be calculated since it is unclear in some cases which is the true concentration that was tested. If, for example, a study reports the use of CAPB at 10% active, the assumption will be made that 10% active was tested. If a study reports use of 10% CAPB, concentrations will be calculated assuming both possibilities: (1) that it was 10% active or (2) it was 10% and only 30% of that was active, yielding 3% active.
Composition, Chemical, and Physical Characteristics of Batches of Cosmetic Grade CAPB 5
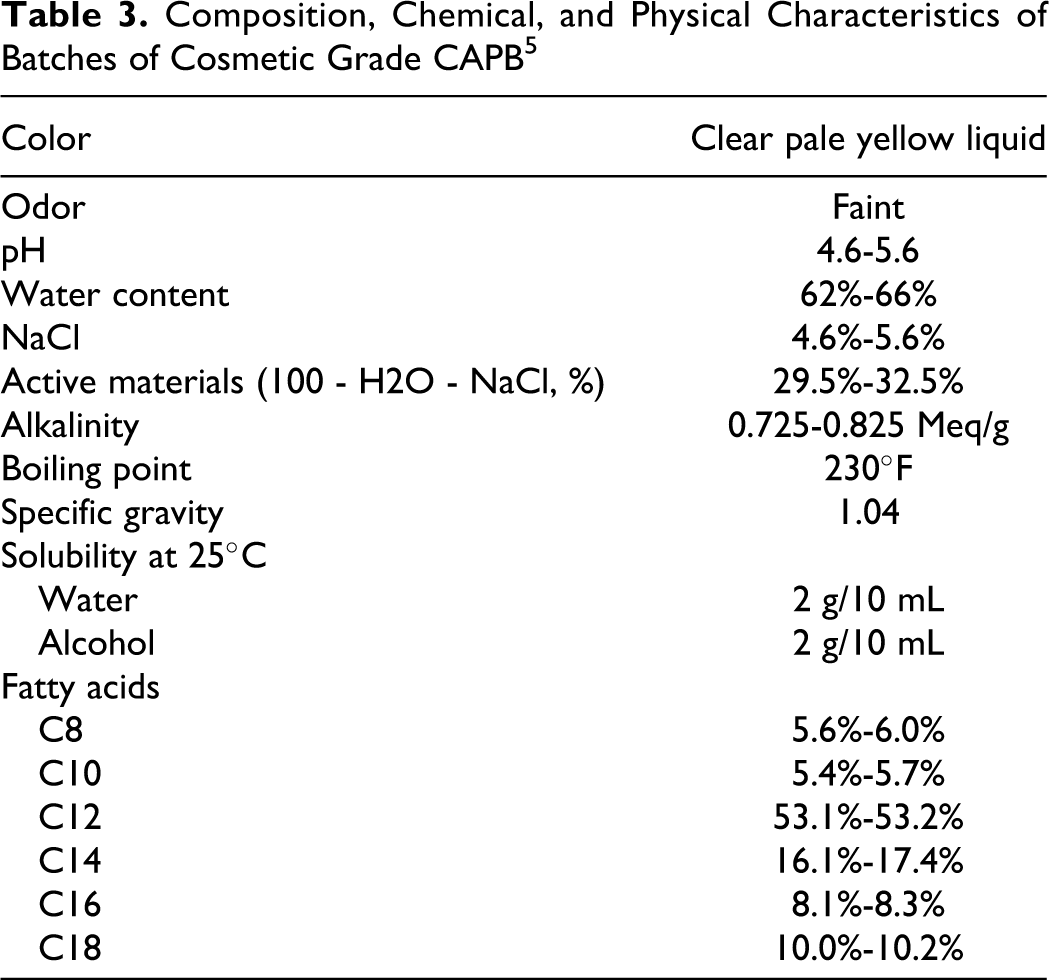
Commercial grades containing concentrations of CAPB greater than 30% may contain solvents, such as propylene glycol. Although most commercial grades contain sodium chloride, low-salt products also are available. The concentration of sodium chloride in cosmetic grade CAPB ranges from 4.0% to 6.0%. Cosmetic grade CAPB may also contain a maximum of 3.0% glycerol. 1
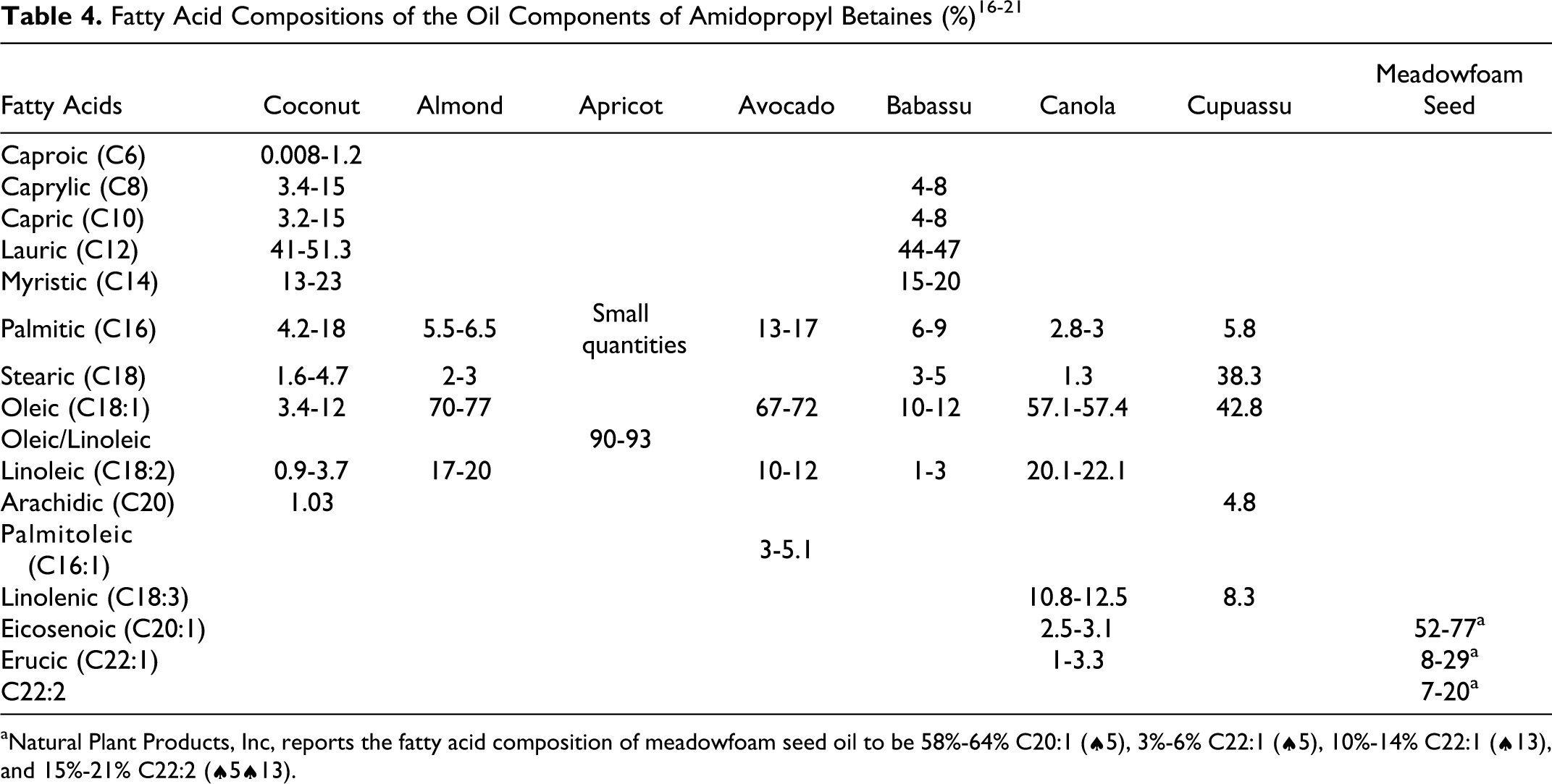
The fatty acid compositions of the oils that are components of the additional amidopropyl betaines described in this report are presented in Table 4.
aNatural Plant Products, Inc, reports the fatty acid composition of meadowfoam seed oil to be 58%-64% C20:1 (♠5), 3%-6% C22:1 (♠5), 10%-14% C22:1 (♠13), and 15%-21% C22:2 (♠5♠13).
Method of Manufacture
Figure 2 depicts the formation of CAPB through the reaction of coconut oil fatty acids (coconut oil or hydrolyzed, glyceryl-free coconut acid) with 3,3-dimethylaminopropylamine (DMAPA), which yields cocamidopropyl dimethylamine (amidoamine or dimethylaminopropyl cococamide). The amidoamine, a tertiary amine, is then reacted with sodium monochloroacetate to produce CAPB. In Figure 2, R represents the coconut fatty acid chain that varies between C-8 and C-18.1 ,3,7–10
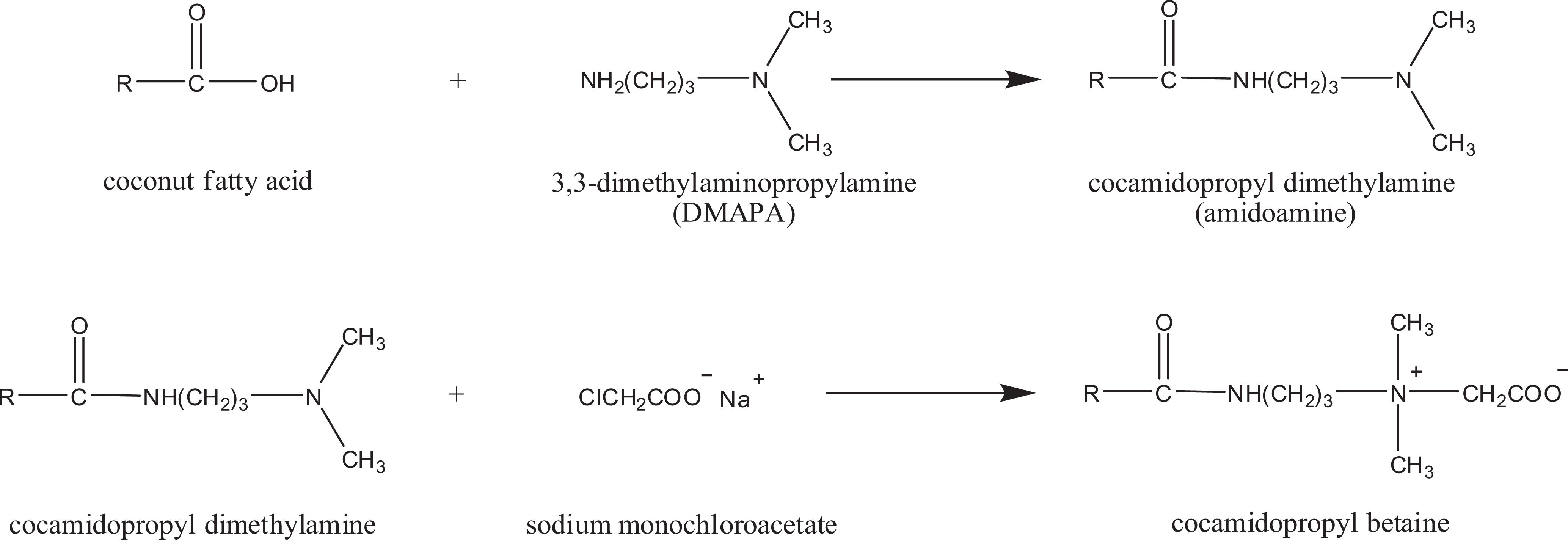
Reaction process of cocamidopropyl betaine (R represents the coconut fatty acid chain that varies between C-8 and C-18).
Supplier information provided to the Personal Care Products Council (the Council) indicated that babassuamidopropyl betaine, coco/sunfloweramidopropyl betaine, cupuassuamidopropyl betaine, isostearamidopropyl betaine, lauramidopropyl betaine, meadowfoamamidopropyl (MF) betaine, oleamidopropyl betaine, ricinoleamidopropyl betaine, and wheat germamidopropyl betaine are manufactured in the same manner as CAPB. 11 Manufacturing data on the remaining amidopropyl betaines were not provided.
In cupuassuamidopropyl betaine, the intermediate is cupuassuamidopropyl dimethylamine, which can be found at a maximum level of 0.2% in the final product. 11 The DMAPA level in final cupuassuamidopropyl betaine product is 0.05%. In MF betaine, the intermediate is MF dimethylamine (MF-DMAPA), which can be found at less than 0.5% in the final product. The manufacturing process for MF betaine exhausts DMAPA. The levels of DMAPA and amidoamine were reported to be below 0.0002% (the detection limit) and <0.5%, respectively, in babassuamidopropyl betaine, coco/sunfloweramidopropyl betaine, isostearamidopropyl betaine, lauramidopropyl betaine, oleamidopropyl betaine, ricinoleamidopropyl betaine, and wheat germamidopropyl betaine.
The CIR accepts the US Food and Drug Administration (FDA) determination (21 CFR 700.27(a)) that tallow derivatives are not prohibited cattle materials.
Impurities
No N-nitroso compounds were detected in samples of commercially supplied CAPB. 12 CAPB samples with and without internal standards of N-nitroso compounds were analyzed using gas chromatography with a thermal energy analyzer (TEA). The CAPB has a secondary amido group that is susceptible to N-nitrosation to form an N-nitrosamide. Although a highly sensitive analytical method failed to detect traces of volatile N-nitrosamines in samples of commercial CAPB, this result does not exclude the possibility that in the presence of N-nitrosating agents CAPB gives rise to reactive and unstable nitrosamides. The TEA method does not detect nitrosamides. 13
Coconut oil impurities may be present in CAPB, depending on the degree of refining to which the coconut oil is subjected, including free fatty acids and low concentrations of sterols, tocopherol, squalene, and lactones. Concentrations of pigments, phosphatides, gums, and other nonglyceride substances are usually low in coconut oil in contrast to other vegetable oils. 14
Impurities associated with CAPB are the reactants and intermediates from production and include amidoamine, sodium monochloroacetate, and DMAPA.7,9,10 Depending on the manufacturer, residual amidoamine and DMAPA can range from 0.3% to 3.0% and from 0.0003% to 0.02%, respectively. 9
In 2007, the Personal Care Products Council surveyed suppliers regarding the levels of DMAPA and amidoamine in CAPB. The limit of detection for DMAPA is 100 ppm in some analytical methods, but some methods may detect this impurity at concentrations as low as 2.5 ppm. Several companies reported DMAPA below the 100 ppm detection limit, with 1 supplier reporting a DMAPA below the limit of detection of 0.0002%. The survey found levels of amidoamine ranged from 0.5% to 5%, with 0.5% the typical value and 1.5% the suggested maximum level. The variability in the amidoamine levels may be due to the differences in analytical methods.11,15
Meadowfoam seed oil has been reported to have a typical value of <1 ppm for the heavy metal iron, copper, lead, mercury, cadmium, selenium, and chromium. The maximum value is 10 ppm. 16
Use
Cosmetic
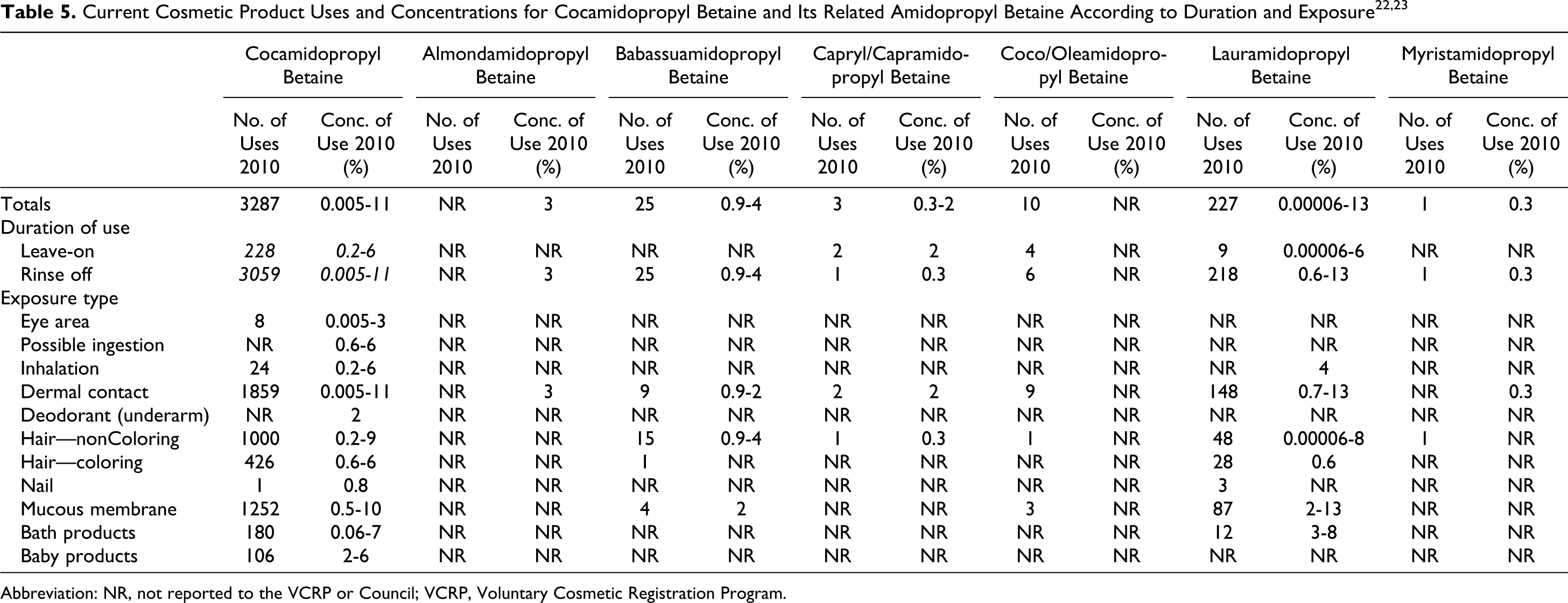
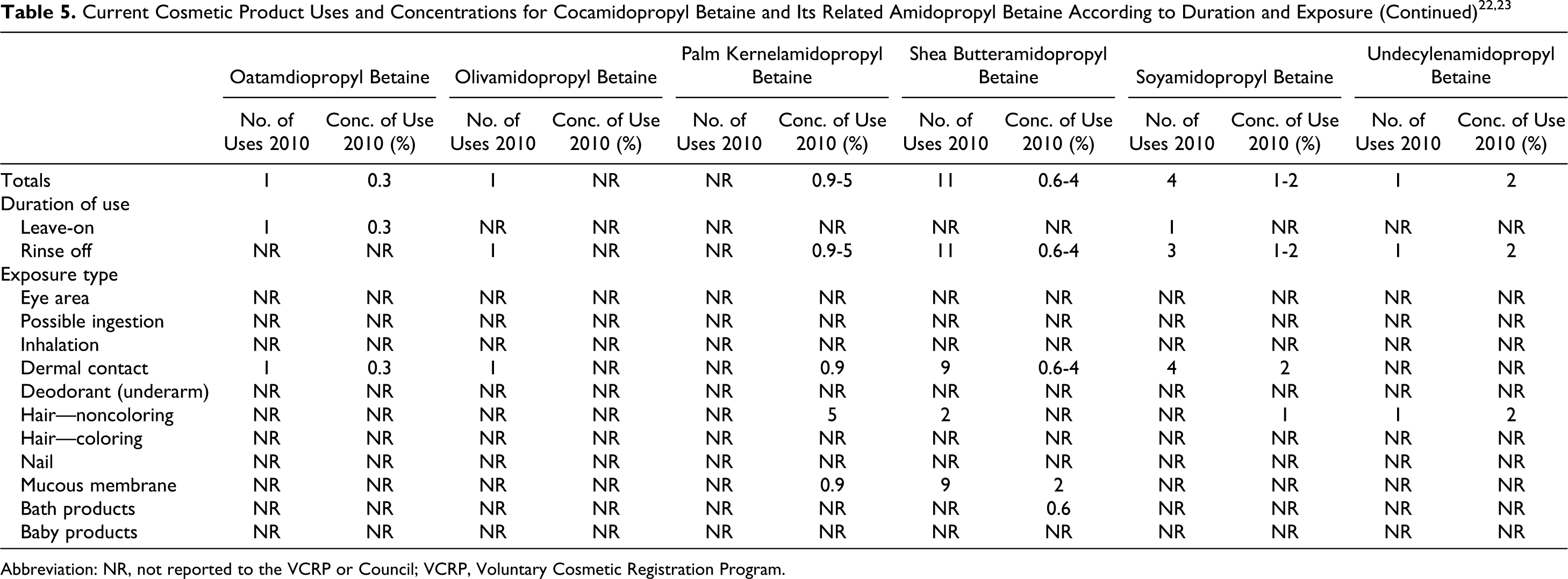
According to information supplied to the FDA by industry as part of the Voluntary Cosmetic Registration Program (VCRP), CAPB is used in a total of 2743 products (Table 5). 22 A use concentration survey conducted by the Council showed CAPB use at concentrations ranging from 0.005% to 11%.23,24
Abbreviation: NR, not reported to the VCRP or Council; VCRP, Voluntary Cosmetic Registration Program.
Abbreviation: NR, not reported to the VCRP or Council; VCRP, Voluntary Cosmetic Registration Program.
The VCRP also reported uses of babassuamidopropyl betaine, capryl/capramidopropyl betaine, coco/oleamidopropyl betaine, lauramidopropyl betaine, oatamidopropyl betaine, olivamidopropyl betaine, soyamidopropyl betaine, and undecylenamidopropy betaine, with the highest total of uses reported for lauramidopropyl betaine at 187. 22 Concentration of use ranges was reported for almondamidopropyl betaine, babassuamidopropyl betaine, capryl/capramidopropyl betaine, lauramidopropyl betaine, myristamidopropyl betaine, oatamidopropyl betaine, palm kernelamidopropyl betaine, shea butteramidopropyl betaine, soyamidopropyl betaine, and undecylenamidopropyl betaine, with the highest concentration of use reported for lauramidopropyl betaine at 13%. 23 For complete information on these ingredients, see Table 5. No uses or concentrations of uses were reported for: apricotamidopropyl betaine, avocadamidopropyl betaine, behenamidopropyl betaine, canolamidopropyl betaine, coco/sunfloweramidopropyl betaine, cupuasuamidopropyl betaine, isostearamidopropyl betaine, MF betaine, milkamidopropyl betaine, minkamidopropyl betaine, oleoamidopropyl betaine, palmamidopropyl betaine, palmitamidopropyl betaine, ricinoleamidopropyl betaine, sesamidopropyl betaine, stearamidopropyl betaine, tallowamidopropyl betaine, and wheat germamidopropyl betaine.
The CAPB is primarily used as a pseudoamphoteric surfactant in hair shampoos. 1 Gottschalck and Bailey described the current functions of CAPB as antistatic agent; hair-conditioning agent; skin-conditioning agent—miscellaneous; surfactant-cleansing agent; surfactant-foam booster; and viscosity increasing agent—aqueous. 2
The CAPB is used in hair sprays and other spray products, and effects on the lungs that may be induced by aerosolized products containing this ingredient are of concern.
There are no specific data for spray products containing CAPB. Jensen and O’Brien reviewed the potential adverse effects of inhaled aerosols, which depend on the specific chemical species, the concentration, the duration of the exposure, and the site of deposition within the respiratory system.
25
The aerosol properties associated with the location of deposition in the respiratory system are particle size and density. The parameter most closely associated with this regional deposition is the aerodynamic diameter, da
, defined as the diameter of a sphere of unit density possessing the same terminal setting velocity as the particle in question. These authors reported a mean aerodynamic diameter of 4.25
Bower reported diameters of anhydrous hair spray particles of 60 to 80 µm and pump hair sprays with particle diameters of
The CAPB was not restricted from use in any way under the rules governing cosmetic products in the European Union. 28
Noncosmetic
The CAPB is used in household cleaning products, including laundry detergents, hand dishwashing liquids, and hard surface cleaners. 29 A 30% active CAPB solution was tested for antibacterial and antimycotic activity using the agar cup plate method. 30 Zones of inhibition were measured for the bacteria and molds around agar cups containing 0.2 mL of the ingredient, which had been diluted with distilled water to 0.5% activity. No inhibition against Escherichia coli or Pseudomonas aeruginosa was observed. Bacteriostatic activity was detected in cultures of Staphylococcus aureus, Streptococcus pyogenes, and Bacillus subtilis. Fungicidal activity was observed in cultures of Candida albicans, Trichophyton mentagrophytes, and Pityrosporum ovale.
Toxicokinetics
No studies were found on the absorption, distribution, metabolism, and excretion of CAPB or other amidopropylbetaines. It is unclear whether the amide bond can be hydrolyzed to yield the fatty acids and 3-aminopropyl betaine. No metabolism data are available on the latter compound.
Toxicological Studies
Single-Dose (Acute) Toxicity
Oral
A full-strength CAPB solution, 30% active, was administered by gastric intubation to groups of 10 CFR mice of the Carworth strain, weighing 18 to 21 g. Mice were observed for 7 days following the administration. The oral LD50 was 6.90 g/kg (calculated from volume per weight dosage units, based on a density of 1.07 g/mL). Confidence range is 6.06 to 7.86 g/kg. 31
Undiluted CAPB, 30% active, with a pH of 5.5, was administered by gavage to groups of 10 (5 female, 5 male) Wistar rats. 32 Dosage groups were 5.00, 6.30, 7.94, and 10.00 mL/kg. The rats were observed for 14 days. The oral LD50 was 7.97 g/kg (calculated from volume per weight dosage units, based on a density of 1.07 g/mL). Confidence range is 6.93 to 9.17 g/kg. Rats in all dosage groups had decreased motor activity, abnormal body posture, coordination disturbance, cyanosis, diarrhea, and decreased body temperature beginning approximately 20 minutes after dosage and persisting for 24 hours. Surviving rats in all groups had body weight gains of 36 to 45 g and were normal in appearance and behavior. Redness of the stomach and intestinal mucous membranes were observed at necropsy.
A full-strength solution of CAPB, 30% active, was administered by gavage to groups of 5 albino rats at single doses of 2.0, 4.0, 5.0, 6.3, 8.0, and 16.0 g/kg, and the rats were observed for 14 days. 33 Sluggishness, nasal hemorrhaging, diarrhea, and wetness around the hindquarters were observed, increasing in severity with dosage. The oral LD50 for this full strength, 30% active CAPB solution was estimated at 4.9 g/kg, with a 95% confidence limit of 3.7 to 6.5 g/kg.
A full-strength solution of CAPB, 30% active, was administered by gavage to groups of 10 (5 female, 5 male) Sprague-Dawley rats at single doses of 2.0, 2.71, 3.68, 5.0, or 6.78 g/kg, and the rats were observed for 15 days. 34 At necropsy, a blood-like, viscous liquid was found in the intestines. Surviving rats gained an average between 20 and 130 g by day 15. Diarrhea was observed in rats of all treatment groups, and decreased motor activity was observed in rats of all treatment groups, except at the lowest dose. Dried blood around the nose and salivation were observed in male rats of the 5.0 g/kg dosage groups. The acute oral LD50 for this full-strength CAPB, 30% active, was 4.91 g/kg within 95% confidence limits of 4.19 to 5.91 g/kg.
The American Chemistry Council summarized an acute oral toxicity study on 35.61% active CAPB. 35 Fasted Sprague-Dawley rats (5 female, 5 male; 220-294 g) received a single, oral dose via gavage of undiluted test material. The rats were weighed before dosing and at study termination, and they were observed frequently from the day of dosing and for 14 days. Animals that died during the study underwent gross necropsy. All of the female rats died on day 2 of the study. Prior to death, the females exhibited salivation, diarrhea, ataxia, and/or decreased activity. Male rats exhibited similar clinical signs on day 1 (day of dosing) and day 2 but had recovered by day 3. Necropsy data were not reported. The acute oral LD50 for 35.61% active CAPB was >1.8 g/kg for male rats.
The CAPB (31% active) was orally administered to male and female CD rats (5/sex; 110-150 g) at 5.0 g/kg body weight via gavage. Animals were observed daily until 14 days after dosing and were killed on day 15. Individual body weights were recorded on days 1, 8, and 15. Macroscopic postmortem examinations performed. Clinical signs of toxicity included piloerection, increased salivation, hunched posture, and diarrhea. Animals recovered by day 4. Slightly reduced body weight gains were recorded for 4 males and 3 females on day 8, but all animals achieved expected weight gains by day 15. No abnormalities were observed at necropsy. The acute oral LD50 was greater than 5.0 g/kg. 35
In another acute oral toxicity study reported by the American Chemistry Council, fasted Wistar rats (5 rats per dose, sexes combined; 200-300 g) received a single oral gavage dose of CAPB (30% aqueous) at levels of 4.0, 8.0, 10.0, 12.5, 16.0, or 32.0 g/kg. 35 The rats were observed daily for 2 weeks after dosing. No postmortem or histopathology examinations were performed. Clinical signs included slight diarrhea and unkempt coats in the 4.0 g/kg dose group, and lethargy, diarrhea, nasal hemorrhage, and unkempt coats was observed in the dose group of 8.0 g/kg and above, with severity increasing proportionately. The acute oral LD50 was 8.55 g/kg. (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 30% active or 30% aqueous, which equated to 9% active.)
Dermal
The American Chemistry Council summarized an acute dermal toxicity study of CAPB (31% active) using male and female CD rats (5/sex; 200-232 g). 35 The animals received 2.0 g/kg body weight on the clipped surface of the dorsolumbar region. The treated area was occluded. After 24 hours, the dressings were removed and the treated area was washed with warm water and blotted dry. The treated areas were examined daily for 14 days for signs of dermal irritation. The rats were weighed on days 1, 8, and 15. At day 15, the rats were necropsied. No unscheduled deaths occurred and no clinical signs of systemic toxicity were observed. No abnormalities were observed at necropsy. Slight or well-defined erythema was observed on day 2, with well-defined erythema persisting in 3 males and all females on day 3 and completely resolving by day 6. Sloughing or hyperkeratinization affected 6 rats on days 4 and 5 only. The acute lethal dermal dose of CAPB (31% active) was greater than 2.0 g/kg.
Repeated Dose Toxicity
Oral
Male and female Sprague-Dawley rats (8/sex/group) were treated with a full-strength (30.6% active) CAPB solution. 36 Three dose groups (100, 500, and 1000 mg/kg body weight) were given the test material by gavage for at least 28 days. A control group of 16 animals received deionized water. Rats dying during the study and those killed on completion of dosing were necropsied, and tissues were collected for histopathological evaluation.
Mortality was increased in the treated groups as compared to controls, but mortality did not follow a dose–response relationship. The principal necropsy finding in the rats that died was congestion, noted in several tissues, with additional alterations in the lungs of some rats. The death of a high-dose female was ascribed to a dosing accident. It was considered possible that the 1 death of a male of the low-dose group and 1 female of the mid-dose group could be attributed to dosing accidents. The other deaths were related to compound administration. This conclusion was supported by the observation that deaths occurred later (3-4 weeks of study in the mid-dose group, as compared to the high-dose groups: deaths at 1-2 weeks of study). However, doubling of the dose of compound (from 500 to 1000 mg/kg) did not increase mortality, so a dose–response relationship with the mortality was not evident.
Lesions (subacute inflammation and epithelial hyperplasia) of the nonglandular portion of the stomach were suggestive of irritation by CAPB. Lesions were found in 1 of 5 stomachs examined from the high-dose males and in all 7 from high-dose females. The loss of 3 males during the first 2 weeks of dosing prevented adequate evaluation of the response of male rats to the compound. Both males and females of the 100 mg/kg dose group were comparable to concurrent controls.
The American Chemistry Council summarized a 28-day short-term oral toxicity of CAPB (concentration not stated) in Sprague-Dawley rats. 35 Male and female rats received 0, 250, 500, or 1000 mg/kg body weight of the test material once daily via oral gavage on 5 consecutive days per week. The number distribution of the rats per group was not described.
No treatment-related deaths or decreases in feed or water consumption were observed over the course of the study. Hematological evaluations, clinical chemistry, ophthalmic examinations, and absolute and relative organ weights also did not find any treatment-related effects. Head protrusion at the beginning of week 3 and salivation at the beginning of week 4 were observed in the 1000 mg/kg dose group. Compound-related edema of the mucosa of the nonglandular stomach was observed at macroscopic examination in the 1000 mg/kg dose group, which disappeared in the rats in the recovery group. Microscopic examination of the rats in the 1000 mg/kg dose group found acanthosis of the gastric mucosa, inflammatory edema of the submucosa, and multiple ulcerations. Effects were greater in the females than the males. These effects were considered to be the result of the irritating properties of CAPB and not of systemic toxicity, especially since the 1000 mg/kg recovery animals had complete and regular regeneration of the nonglandular mucosa. No other treatment-related effects were observed in the organs. The study concluded that the NOEL was 500 mg/kg per d and the LOEL was 1000 mg/kg per d for exposure to CAPB in this rat study.
Groups of 10 male and 10 female Crl:CF(SD)BR Sprague-Dawley rats received 0, 250, 500, or 1000 mg/kg per d CAPB (concentration not stated) in distilled water once daily via oral gavage at a dose volume of 10 mL/kg per d for 92 days. 35 Clinical signs were recorded daily and body weight and feed consumption were recorded once weekly. Ophthalmic examinations were performed on the control and 1000 mg/kg per d dose groups prior to dosing and to all groups during the final week of treatment. Blood and urine samples were collected from all rats during the final week of treatment. Complete necropsy was performed on surviving rats at study termination. Histopathology was performed on select tissues from the rats in the control group and the 1000 mg/kg per d dose group. Because treatment-related histopathological changes were observed in the stomachs of the 1000 mg/kg per d group, stomachs from the 250 and 500 mg/kg per d groups also were examined microscopically.
No treatment-related deaths or effects were observed during the course of the study for either sex. Necropsy revealed stomach ulcers at the fundic and cardiac regions in 1 male and 1 female in the high-dose group. Microscopic evaluations found nonglandular gastritis in 6 male and 3 female rats in the 1000 mg/kg per d group, and in 2 male and 2 female rats in the 500 mg/kg per d group. This effect was not observed in the 250 mg/kg per d dose group. No other treatment-related effects were observed. The study concluded that the NOEL for this subchronic study of CAPB in rats was 250 mg/kg per d.
Dermal Irritation
Animal
The available data on skin irritation studies are summarized in Table 6. 37–43 These studies demonstrated that, while a full-strength CAPB solution, 30% active, was a mild irritant, a 50% dilution was nonirritating.
Animal Skin Irritation Studies on CAPB
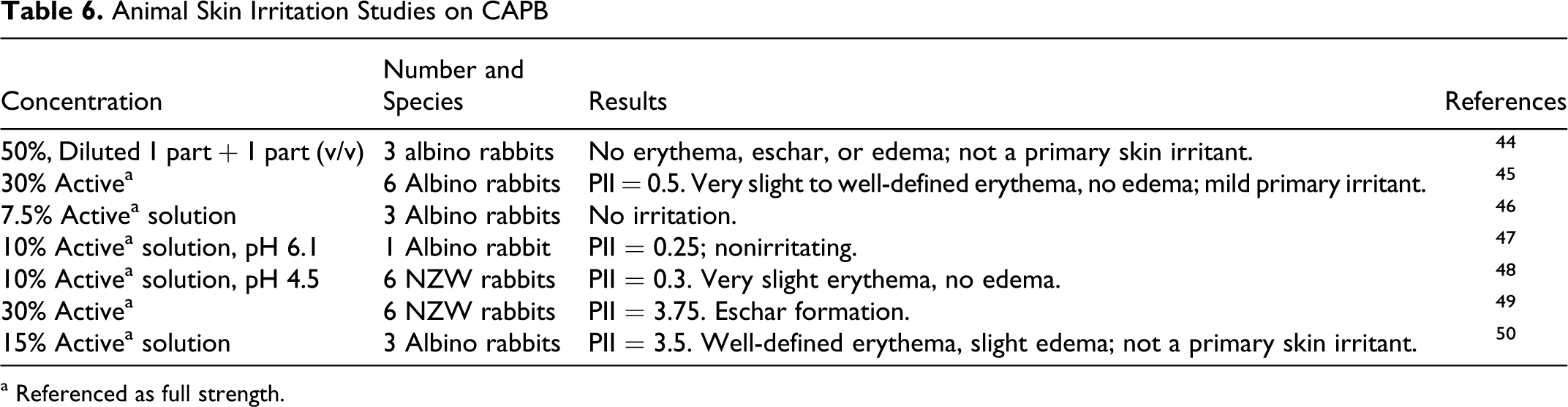
a Referenced as full strength.
Human
Cocamidopropyl betaine
In a study of cumulative irritation, 0.3 mL of 2 soap formulations were applied to skin sites on the backs of 10 panelists using occlusive patches. 37 Each formulation contained 1.9% active CAPB. Daily 23 hour patches were applied for 21 consecutive days. The total irritation scores for all participants for all 21 applications of the 2 formulations were 588 and 581 (max 630), which indicated that these test formulations were primary irritants. The average irritation times for the formulations were 1.48 and 1.69 days, and the median irritation time was 2 days.
The CAPB at 0.06% (1.0% aqueous dilution of a product formulation containing 6.0% active CAPB) was tested for skin irritation using a single insult occlusive patch test and 19 panelists. 1 Fifteen panelists had no irritation and a + score was recorded for 4 panelists. The formulation was considered practically nonirritating.
Daily doses of 0.2 mL of 0.52% CAPB (an 8% aqueous dilution of a liquid soap formulation containing 6.5% active CAPB) were applied via occlusive patches to the forearms of 12 human participants for 5 days. 1 An erythema score of 0.48 (scale 0-4) was calculated.
Wheat germamidopropyl betaine
The irritation potential of 0.005% active wheat germamidopropyl betaine (a 0.5% aqueous solution of 1.0% wheat germamidopropyl betaine in a body polisher) was evaluated against a control shower gel in a single 24-hour insult patch test. Twenty participants completed the study. Two panelists had a ± score and 4 panelists had a 1 score and the primary irritation index (PII) was calculated at 0.25. The control substance elicited a ± score in 4 panelists, a 1 score in 2 panelists, and a + score in 2 panelists, yielding a PII of 0.35. The authors concluded that the test material containing 1.0% wheat germamidopropyl betaine was milder than the reference control. 38
Dermal Sensitization
Animal
Delayed contact hypersensitivity of 15 male Pirbright white guinea pigs (400
Well-defined irritation was observed at all sites receiving intradermal injections of Freund adjuvant. Temporary slight irritation was observed following injections of the 0.5% CAPB sample dilution in all test animals. Topical application of the 60% CAPB sample dilution resulted in slight dermal reactions. The barely perceptible erythema observed on the skin of 2 test animals after 24 hours was considered unrelated to CAPB treatment but was attributed to reactions to the elastic adhesive bandages used for site occlusion. With the exception of slight reactions to the bandages, no reactions were observed in controls throughout the 72-hour observation period. No evidence of delayed contact hypersensitivity was found.
A formulation containing 0.75% active CAPB was tested in a delayed contact hypersensitivity test. 40 Closed patches containing 0.4 mL of the test solution were applied to the shaved area on the left shoulder of 20 albino guinea pigs. After 6 hours, the patch was removed and the area was rinsed with warm water. This procedure was repeated at the same site for the following 2 weeks. The animals were left untreated for 2 weeks before the primary challenge test, which used 0.01875% CAPB (a 2.5% solution of the 0.75% active CAPB) applied to a freshly clipped skin site not previously treated for 6 hours. Responses were graded after 24 and 48 hours. There was no evidence of sensitization following the exposure to the 3 dermal treatments or challenge dose.
A full-strength, 30% active CAPB sample was tested for skin sensitization using a maximization test and a modified Draize test. 41 Albino guinea pigs (20 animals) received intradermal injections of (1) Freund complete adjuvant alone, (2) 0.1% aqueous dilution of the CAPB sample (0.03% active CAPB), and (3) 0.1% aqueous dilution of the CAPB sample (0.03% active CAPB) plus the adjuvant. One week later, a topical 48-hour occlusive induction patch containing the 10% aqueous dilution of the CAPB sample (3% active CAPB) was applied. Animals in the control group received intradermal injections and topical application of water alone. After 3 weeks, single 24-hour occlusive patches were applied to the clipped flanks of all animals. A 10% aqueous dilution of the CAPB sample (3% active CAPB) was applied to the left flank, and water was applied to the right. The lesions at necropsy were erythema and edema in 8 of the 20 test animals after the challenge application. Microscopic findings included epidermal acanthosis, inter- and intracellular edema, and massive infiltration of the superficial layers of the dermis with lymphocytes, monocytes, and a few eosinophils with a tendency to invade the epidermis in 2 of the animals. Less prominent microscopic lesions of acanthosis, mild intracellular edema, and a moderate lymphomononuclear infiltrate in the superficial dermis were found in 4 additional animals. Slight acanthosis was observed in the remaining 2 animals.
This same laboratory also tested 0.15% active CAPB for induction (0.015% for challenge) using the same assay. Slight erythema and edema were observed macroscopically in 6 of the 20 test animals. Slight acanthosis was observed microscopically. Control animals in the maximization and modified Draize tests had no dermatitis-type clinical or histological alterations. A few controls had moderate acanthosis with edema and vasodilation in the subjacent papillary layer of the dermis. The investigators concluded that the commercially supplied CAPB is capable of producing a delayed-type contact sensitization.
Basketter et al reported that CAPB was positive for sensitization in a local lymph node assay (LLNA). 42 The EC3 value was not reported.
Dermal Sensitization
Fisher contact dermatitis recommended that patch testing with CAPB should be performed at a concentration of 1% aqueous. 43 Care was advised for patch test readings since mild false-positive irritant reactions may occur.
de Groot, in a review of contact allergy literature, stated that CAPB in rinse off products such as shampoo, shower gel, bath foam, and liquid soap was linked to cosmetic allergy. 7 Because patch testing for sensitization with these products may result in both false-positive and false-negative reactions, the author suggested that CAPB should be tested separately. The author also suggested that CAPB should be included in the hairdresser’s series and the cosmetic series with the knowledge that commercial concentration of CAPB (1% in water, possibly 0.3% active) is a marginal irritant and not all positive patch test reactions indicate contact allergy to CAPB.
Another review of contact allergy literature by Mowad described CAPB as “contact allergen of the year” for 2004. 10 Because impurities in CAPB may be responsible for allergic reactions, the author advised patch testing for amidoamine and DMAPA along with CAPB. The author further suggested that patients that test positive to amidoamine or DMAPA should be advised to avoid products that contain CAPB.
Historically, sensitization study results are reported as positive/negative for a particular concentration of the chemical tested. More recently, the dose per unit area is considered as the relevant parameter. 51 CIR has performed calculations to determine dose per unit area where sufficient information was available.
The available data on clinical sensitization studies are summarized in Table 7.
Clinical Sensitization Studies on CAPB and Related Amidopropyl Betaines.
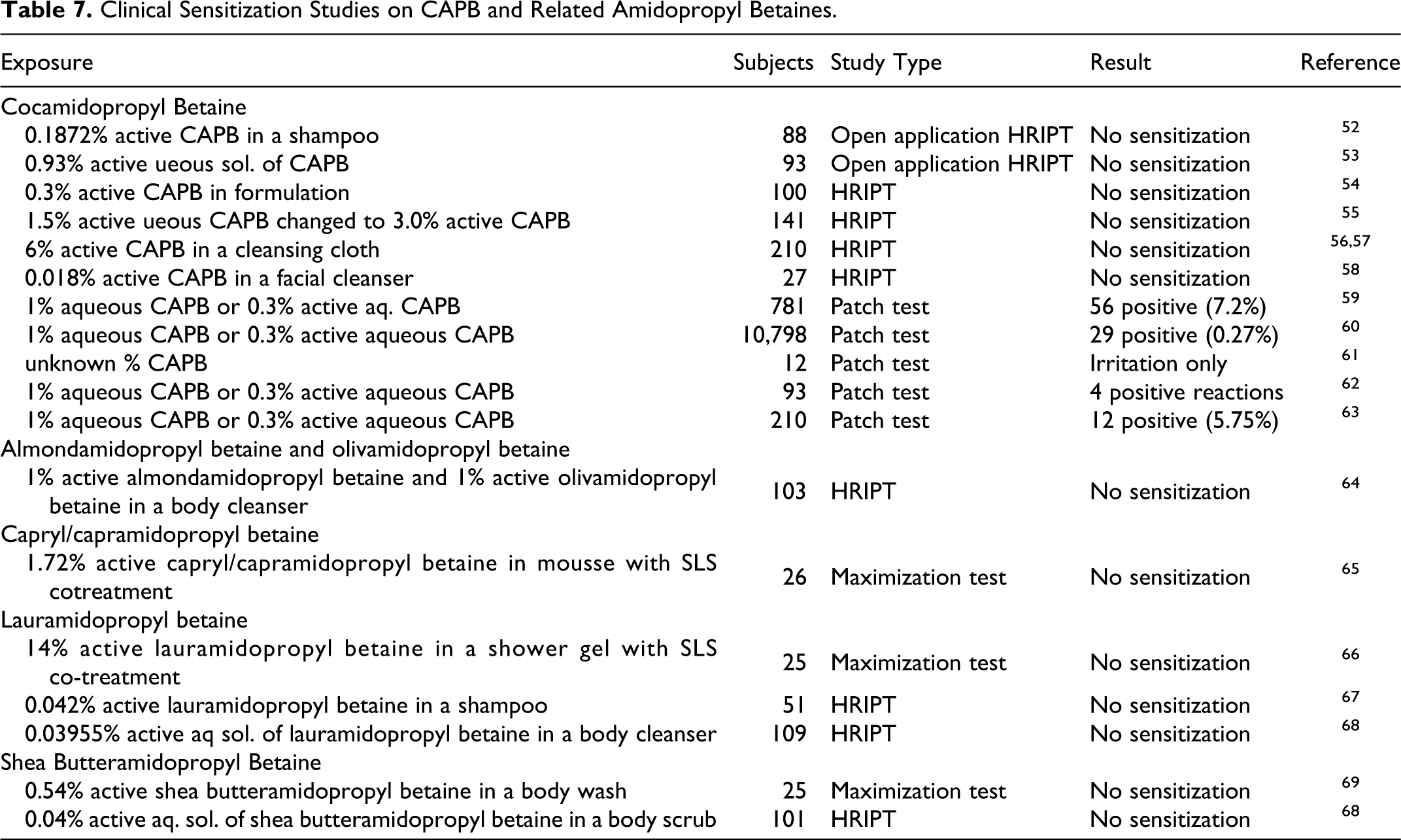
Cocamidopropyl betaine
A repeated open application procedure was performed with 1.872% CAPB (a 10% w/v aqueous dilution of a shampoo containing 18.72% active CAPB), using 88 human volunteers to determine skin sensitization. [Estimated dose/unit area = concentration × amount × density × unit conversion × area−1 = 2.6 × 103 µg/cm2]. The disk was removed after 10 minutes. Induction applications were made 3× a week for 3 weeks. Challenge patch strips were applied simultaneously to both the induction arm and the alternate arm, positioned between the shoulder and elbow, 18 days after the last induction application. The areas were scored 24, 48, and 72 hours following the removal of the patch after a 6-hour period. The same procedures were performed with another test substance containing an identical concentration of CAPB. No sensitization was seen in any of the 88 participants exposed to either of the test materials. 52
Another study was performed with a 0.93% active aqueous solution of CAPB. [Estimated dose/unit area = 7.7 × 102 µg/cm2]. 53 Ninety-three volunteers completed the study. Induction applications were made to the same site unless reactions became so strong that a first or second adjacent site had to be used for complete induction, and the sites were scored following a 48-hour period. An alternate site was used for the challenge test and was scored after 48 and 96 hours. Ten participants had slight responses to the test material. These responses were attributed to primary irritation, rather than sensitization, during both the induction and challenge tests.
In a similar study by Hill Top Research, Inc, a formulation containing 0.3% active CAPB was tested on 100 human volunteers. 54 The study had started out with CAPB at 0.6%, but due to several incidences of mild to moderate skin irritation early in the induction phase, the formulation was diluted. [Estimated dose/unit area = 2.5 × 102 µg/cm2 at 0.3%]. No evidence of sensitization was observed in the formulation at 0.3% active CAPB.
CAPB was studied using 141 human participants. All applications contained a concentration of 1.5% active CAPB in distilled water, until a protocol modification changed the concentration to 3.0% active CAPB. Participants who began the study a week earlier received 2 applications at a concentration of 1.5%, and all other applications of the test material at a concentration of 3.0%. [Estimated dose/unit area = 5.8 × 101 µg/cm2 at 1.5%, 1.2 × 102 µg/cm2 at 3%]. Induction applications were made to the same, previously untreated site on the back 3 times per week for 3 successive weeks. Patches were removed after 24 hours. Following a 10- to 15-day nontreatment period, the challenge application was applied to a previously untreated site for 24 hours, and the site was scored 24 and 72 hours after patch removal. No responses were observed during either the induction or challenge tests. 55
Clinical Research Laboratories, Inc performed an RIPT study on 6% active CAPB in cleansing cloths in 2 groups of participants (in phase I, 104 participants completed the study. In phase II, 106 participants completed the study).56,57 The test area was wiped with 70% isopropyl alcohol and allowed to dry. The test material was cut to a ½ inch square and applied to the upper back under a semioccluded patch for 24 hours. There were a total of 9 induction patches. Induction sites were scored for irritation. Following a 2-week rest period, challenge patches were applied to a virgin site on the back. After 24 hours, the patches were removed and evaluated for dermal reactions. The test sites were scored again at 48 and 72 hours. No reactions were observed in either group of participants. It was concluded that 6% active CAPB in cleansing cloths did not demonstrate a potential for eliciting dermal irritation or sensitization.
In a study by KGL, Inc, 0.018% active CAPB (a 0.5% aqueous dilution of a facial cleanser containing 3.6% active CAPB) was tested on 27 participants to determine skin sensitization. 58 In the induction phase, the participants were pretreated with 0.05 mL of 0.25% aqueous sodium lauryl sulfate (SLS) under an occluded 15 mm Webril disc for 24 hours on the upper outer arm, volar forearm, or back. After 24 hours, the SLS patch was removed and 0.05 mL of the test material was applied to the same site and occluded. The induction patch was left in place for 48 hours and the site was scored for irritation. [Estimated dose/unit area = 5.1 µg/cm2]. If no irritation was present, the SLS patch followed by the test material patch procedure was repeated for a total of 5 induction exposures. If irritation developed at any time during the induction phase, the SLS treatment patch was eliminated and only the test material was reapplied after a 24-hour rest period. Following a 10-day rest period, the participants received 0.05 mL of 5% SLS for 1 hour prior to receiving the challenge patch of the test material to the opposite side of the body. The challenge patch was occluded and left in place for 48 hours. After patch removal, the site was scored 15 to 30 minutes later and again at 24 hours. No reactions were observed during the induction or challenge phases of this maximization study. It was concluded that 0.018% active CAPB in a facial cleanser was not likely to cause contact sensitivity reactions under normal use conditions.
Almondamidopropyl betaine and olivamidopropyl betaine
The irritation/sensitization potential of 0.005% almondamidopropyl betaine and 0.005% olivamidopropyl betaine in a body cleanser (a 0.5% dilution of 1% active almondamidopropyl betaine and 1% active olivamidopropyl betaine) was evaluated in a repeat insult patch test of 103 participants. [Estimated dose/unit area for each betaine = 2.5 µg/cm2]. After the induction phase (3× per week for 3 weeks) and a 2-week rest period, the participants received a single challenge patch. No reactions were observed. It was concluded that a body cleanser containing 0.005% almondamidopropyl betaine and 0.005% olivamidopropyl betaine was not a primary sensitizer or irritant to the skin. 64
Capryl/capramidopropyl betaine
KGL, Inc evaluated the contact-sensitizing potential of a mousse (concentrate) containing 1.72% active capryl/capramidopropyl betaine in a maximization study. 65 Twenty-six adult participants completed the study. During the induction phase, ∼0.05 mL of aqueous SLS (0.25%) was applied to a test sites on the upper outer arm, volar forearm, or the back of each participant. After 24 hours, the SLS patch was removed and 0.05 mL of the test material was applied to the same site and occluded. [Estimated dose/unit area = 4.9 × 102 µg/cm2]. The induction patch was left in place for 48 hours (72 hours if placed over a weekend). After patch removal, the site was examined for irritation. If no irritation was observed, the sequence of patching with SLS followed by patching with the test material was repeated for a total of 5 induction exposures. If irritation was observed during the induction phase, the SLS patch step was eliminated for that participant and only the test material was applied.
At the end of the induction period and a 10-day rest period, a single challenge application of 0.05 mL of the test material was made to a new skin site pretreated with ∼0.05 mL of 5% SLS under occlusion for 1 hour. After 48 hours, the patch was removed and graded on a scale of 0 (not sensitized) to 3 (strong sensitization; large vesiculo-bullous reaction) 1 hour and 24 hours after removal. No adverse or unexpected reactions occurred, and no incidences of contact allergy were recorded. The study concluded that the mousse (concentrate) containing 1.72% capryl/capramidopropyl betaine did not have a detectable contact-sensitizing potential and was not likely to cause contact sensitivity reactions under normal use conditions. 65
Lauramidopropyl betaine
Consumer Product Testing Company performed a repeated insult patch test on a shampoo with 0.042% lauramidopropyl betaine (test material was prepared as a 1% dilution in distilled water of 4.2% active lauramidopropyl betaine). 67 [Estimated dose/unit area = 2.3 × 101 µg/cm2]. Fifty-one participants completed the study. A total of 9 applications were made during the induction phase. Following a 2-week rest period, a challenge patch was applied to a virgin test site on the back. After 24 hours, the patch was removed and the site was scored 24 and 72 hours postapplication. No reactions were observed in any of the participants during the induction or challenge phases of this study. The study concluded that the shampoo containing 4.2% lauramidopropyl betaine, diluted to 1%, did not indicate a potential or dermal irritation or allergic contact sensitization.
In another human repeated insult patch test, the potential of a body cleanser with 0.03955% lauramidopropyl betaine (a 1% dilution of 3.955% active lauramidopropyl betaine) to cause dermal irritation and sensitization was studied. 68 One hundred and nine participants completed the study. Prior to patch application, the test area was wiped with 70% isopropyl alcohol and allowed to dry. The test solution was applied to the upper back and remained in direct skin contact for 24 hours. The induction period was comprised of a total of 9 applications on the same site. The sites were graded for dermal irritation 24 hours after patch removal. Following a 2-week rest period, a challenge patch was applied to a virgin test site on the back. After 24 hours, the patch was removed and evaluated for dermal reactions. The sites were reevaluated at 48 and 72 hours. Several participants had barely perceptible erythema and reactions were observed on 1 or 2 days of induction phase of the study. No incidences of dermal reaction were recorded during the challenge phase. The study concluded that the body cleanser with 3.955% lauramidopropyl betaine, diluted to 1%, did not demonstrate a potential for eliciting dermal irritation or sensitization.
A maximization study to evaluate the contact-sensitizing potential of a shower gel containing 14% active lauramidopropyl betaine was conducted by KGL, Inc. 66 The shower gel was tested as received, namely, 0.5% aqueous. Twenty-five adult volunteers completed the study. The study was conducted in the same manner as the capryl/capramdiopropyl betaine maximization study described above, with the exception that ∼0.1 mL of aqueous SLS (0.25%) and 0.1 mL of the test material were used during the induction and challenge phases. [Estimated dose/unit area = 2.8 × 102 µg/cm2]. No adverse or unexpected reactions occurred, and no incidences of contact allergy were recorded. The study concluded that the shower gel containing 14% lauramidopropyl betaine did not have a detectable contact-sensitizing potential and was not likely to cause contact sensitivity reactions under normal use conditions.
Shea butteramidopropyl betaine
In a human repeated insult patch test, the potential of a body scrub containing 0.04% shea butteramidopropyl betaine (a 1% w/v dilution of 4.0% active shea butteramidopropyl betaine) to cause dermal irritation and sensitization was studied. 70 One hundred and one participants completed the study. The study followed standard RIPT methodology with a total of 9 induction applications of 24 hours in length and a single challenge application following a 2-week rest period. No adverse events were reported and no incidences of dermal reaction were recorded during the challenge phase. The study concluded that the body scrub with 4.0% shea butteramidopropyl betaine, diluted to 1%, was not sensitizing.
A maximization study to evaluate the contact-sensitizing potential of a body wash containing 0.0027% shea butteramidopropyl betaine (a 0.5% dilution of 0.54% active shea butteramidopropyl betaine) was conducted by KGL, Inc [Estimated dose/unit area = 7.6 × 10−1 µg/cm2]. 69 Twenty-five adult volunteers completed this RIPT study. The study was conducted in the same manner as the capryl/capramdiopropyl betaine study described above, with the exception that the patches were made only to the upper outer arm. No adverse or unexpected reactions occurred, and no incidences of contact allergy were recorded. The study concluded that the body wash containing 0.54% shea butteramidopropyl betaine did not have a detectable contact-sensitizing potential and was not likely to cause contact sensitivity reactions under normal use conditions.
Provocative Studies
In 706 patients studied for skin allergy, 93 (83 women and 10 men) were provisionally diagnosed with cosmetic contact dermatitis. 71 Four of the 93 had positive reactions to CAPB 1% aqueous. Two participants had scalp itch and erythema on the forehead, ears, and neck following the use of shampoos with CAPB. The other 2 participants had eczema on the face and/or neck following use of face cleansers that contained CAPB. From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which would equate to 0.3% active.
Fowler studied 210 patients clinically suspected of having allergic contact dermatitis to cosmetics and toiletries. 63 Patch testing with CAPB (1% aqueous) in addition to the North American Contact Dermatitis Group (NACDG) series (70 allergens total) was performed. Twelve of the participants (5.7%) had positive reaction to CAPB in the patch test. Positive reactions were also observed for formaldehyde or formaldehyde releasers, neomycin, and nickel. All but 2 of the participants had initially reported with head and neck dermatitis. The remaining 2 participants had hand dermatitis. Of the 12 participants, 7 were determined definitely relevant when the reported dermatitis cleared after cessation of use of products with CAPB. Specific case reports for 2 of the participants are detailed in the section on case reports. From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous.
de Groot et al studied 2 groups of patients for CAPB allergy. 59 The first group consisted of 781 patients that were patch tested with the European standard series, hairdresser’s series, cosmetics series, and with other relevant allergens, including the patients’ personal care products, and 1% aqueous CAPB from February 1991 to June 1994. Most of the patients in this group were suspected of having occupational contact dermatitis (217 patients were hairdressers). The second group was studied in approximately the same time period and consisted of 102 patients suspected of having cosmetic dermatitis. The patients were patch tested with 1% aqueous CAPB along with the cosmetic screening series. In both groups, relevance was only declared if the patients used products with CAPB and if their dermatitis cleared upon cessation of use of these products.
In the first test group, 56 patients (7.2%) had positive reactions to CAPB, and of these, 17 were classified as definite and all used shampoos and/or shower gels that contained CAPB. Eight of the 17 were hairdressers and had experienced dermatitis on their hands. In the second test group, only 3 patients (3%) had a positive reaction to CAPB. The patients had been using shower gels, shampoos, and/or body lotions containing CAPB. From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous.
Armstrong et al patch tested patients with suspected contact dermatitis (from January 1991 to September 1998) with a standard series that included 1% aqueous tegobetaine L7 (from 1991 to 1994) or 1% aqueous CAPB (from 1995 to 1998). The authors noted that the latter had significantly lower intermediate and reactant impurities. 60 Of the 10 798 patients tested, 29 (0.27%) had a positive reaction to CAPB (24 reactions to tegobetaine L7). Twenty-three of the 29 cases were deemed relevant and had reported dermatitis on the face, neck, hands, or widespread areas. The authors suggested that higher purity CAPB was linked to a diminished frequency of CAPB sensitization. From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous.
In a double-blind randomized controlled study to evaluate allergenicity to coconut oil derivatives, 10 control participants and 12 participants with previously diagnosed allergy to CAPB were patch tested with 11 coconut-derived surfactants, coconut oil, and lauric acid. 61 Patch testing was performed in random order according to standardized procedures with readings at 48 and 96 hours. Three of the 12 participants had doubtful reactions to CAPB in the patch test and 1 control participant had a doubtful reaction to CAPB. The authors suggested that doubtful reactions to CAPB represent irritant reactions and not allergic reactions.
Photosensitization
An investigation of the potential of a 3.0% active aqueous solution of CAPB to induce contact photoallergy was tested using 30 human participants. The 11 participants who had mild to moderate erythemic responses at the irradiated sites during the induction testing were those that received both UVA and 2 MED of UVB irradiation (source spectrum not reported). These responses were expected from the UVB exposure alone. The CAPB was not a photosensitizer in this study. 55
Case Reports
Numerous case studies of allergic contact dermatitis reported positive patch tests to CAPB at concentration as low as 0.5%.72–84
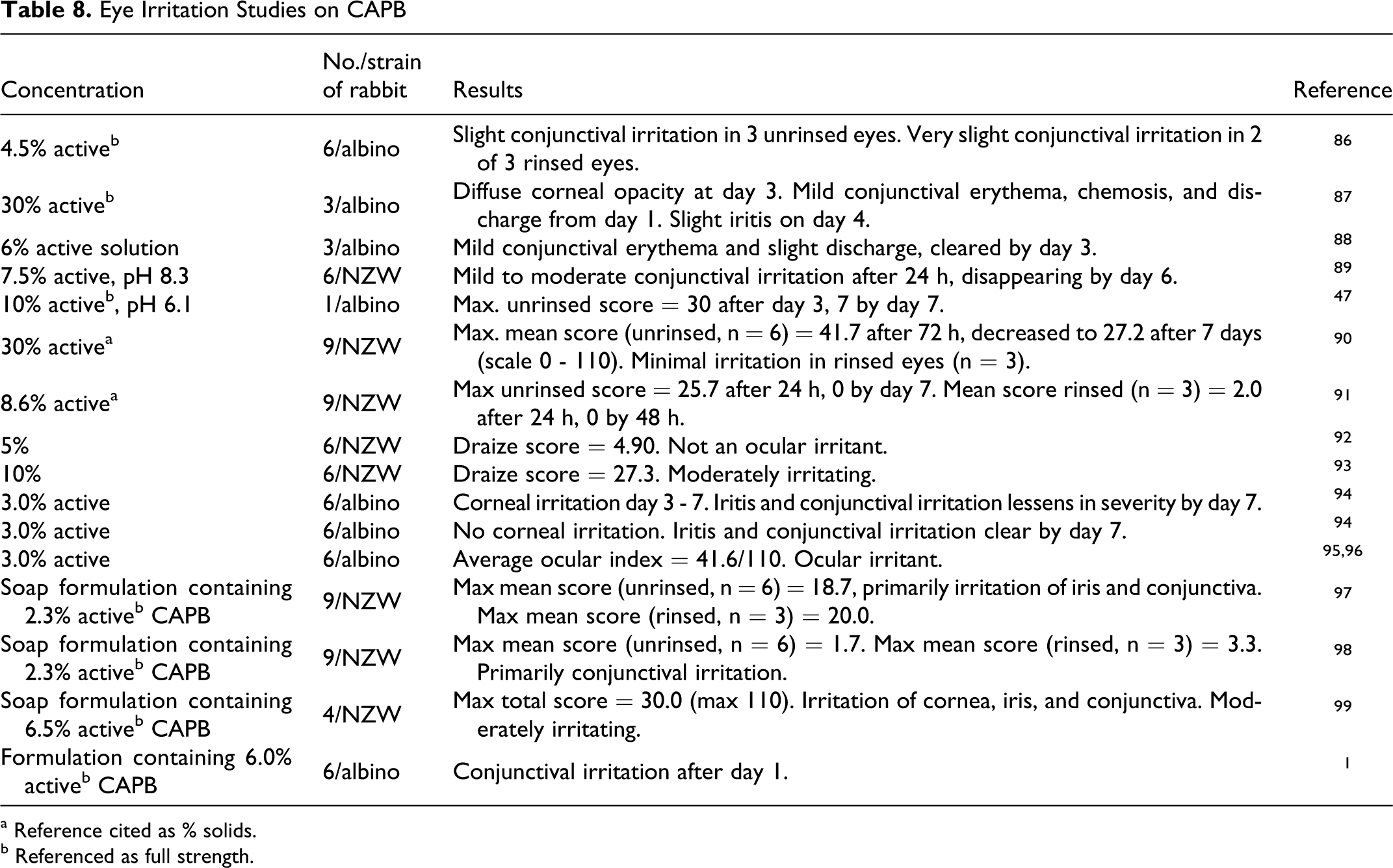
Ocular Irritation
The available data on ocular irritation studies are summarized in Table 8. Two groups of 3 albino rabbits received 0.1 mL instillations of 4.5% active solution of CAPB into the conjunctival sac of 1 eye. 85 Treated eyes of one group were rinsed, but the treated eyes of the other group were not rinsed. Slight conjunctival erythema and chemosis were noted in all treated, unrinsed eyes by day 2 following instillation and subsided by day 7. Slight conjunctival irritation was observed in 2 of the 3 treated, rinsed eyes on the first 2 days of observation. There was no corneal involvement or iris congestion.
Eye Irritation Studies on CAPB
a Reference cited as % solids.
b Referenced as full strength.
The CAPB (30% active) was instilled (0.1 mL) into the conjunctival sac of 1 of the eyes of 3 albino rabbits using the Draize method. 87 Diffuse corneal opacity was observed by day 3 following instillation. Slight iritis was observed by day 4. Mild conjunctival erythema, chemosis, and discharge were noted from day 1.
Three albino rabbits received a 0.1 mL instillation of a 6% active CAPB solution into the conjunctival sac of the right eye. 88 Mild conjunctival erythema and slight discharge were observed in all treated eyes for the first 2 days after instillation, clearing by the third day.
Six NZW rabbits (body weight range 2.4-2.6 kg) received an instillation of 0.1 mL of 7.5% active CAPB with a pH of 8.3 into the conjunctival sac of the left eye. 89 Mild to moderate conjunctival irritation was observed in all treated eyes after 24 hours. The treated eye of 1 rabbit had moderate corneal opacity after the second day. These alterations disappeared by the sixth day after instillation.
One rabbit receiving a 0.1 mL administration of a 10% active CAPB solution (pH 6.1) had Draize scores of 28 after day 1, 25 after day 2, 30 after day 3, 14 after day 4, and 7 after day 7 of the observation period. 47
A full-strength sample of CAPB (30% active) was tested for ocular irritation using 9 NZW rabbits.
90
A volume of 0.1 mL was instilled into the conjunctival sac of one eye of each rabbit. Mean eye irritation scores for treated, unrinsed eyes were 32.5
An instillation of 0.1 mL of a sample of 10% active CAPB was made into the conjunctival sac of 1 of the eyes of 9 NZW rabbits.
91
Mean eye irritation scores for treated, unrinsed eyes were 25.7
In 2 ocular irritation studies by Hazelton Laboratories, 0.1 mL of either 5% or 10% CAPB was instilled into the left eye of groups of 6 NZW rabbits.92,93 The CAPB was not an ocular irritant in the 5% group (Draize score = 4.90) but was considered moderately irritating in the 10% group (Draize score = 27.3).
In a Draize test for ocular irritation, two 3.0% active CAPB samples were instilled into the conjunctival sac of 6 albino rabbits. 94 Scores for corneal irritation were 0 for the first 2 observation days, 1.66 for the third and fourth days, and 4.16 on the seventh day (max score = 80) for 1 of the CAPB samples. No corneal irritation was observed in eyes treated with the other sample. Both samples produced iritis by the first day (scores of 8.33 and 5, respectively, on a scale of 0-10), which decreased in severity by the seventh day (scores of 4.16 and 0, respectively). Both samples produced conjunctival irritation (scores of 15.37 and 14.33, respectively, on a scale of 0-20), which decreased in severity by the seventh day (scores of 6 and 0, respectively).
A 3.0% active CAPB sample was tested for ocular irritation using 6 male albino rabbits.95,96 The average ocular index was 41.6 (max = 110) 24 hours after instillation of 0.1 mL of the sample. The sample was considered an ocular irritant.
A volume of 0.1 mL of a liquid soap formulation containing 2.3% active CAPB was instilled into the conjunctival sac of each of 9 NZW rabbits. 97 An average irritation score of 18.7 (max 110) was calculated for unrinsed eyes, which compared with 20.0 for rinsed eyes. Irritation was observed primarily in the iris and conjunctiva. Under both sets of conditions, the liquid soap formulation was considered moderately irritating.
Another liquid formulation containing 2.3% active CAPB was tested for ocular irritation using 9 NZW rabbits. 98 The maximum average irritation score for the 6 treated, unrinsed eyes was 1.7 (max 110). Slight conjunctival erythema and chemosis were observed in 1 rabbit 2 days after treatment and in the eye of another for the entire 7-day observation period. Slight discharge also was observed in the treated eye of the latter from 72 hours to 7 days following treatment. The formulation was considered minimally irritating to treated, unrinsed eyes of rabbits. The maximum average irritation score for the 3 treated, rinsed eyes was 3.3. Mild conjunctival erythema and chemosis were observed in all tested eyes 1 to 2 days following the instillation. The formulation was considered mildly irritating to treated, rinsed eyes of rabbits.
A liquid soap formulation containing 6.5% active CAPB was tested for ocular irritation by instilling 0.1 mL into the conjunctival sac of one eye of each of 4 NZW rabbits, followed by rinsing. 99 Mean corneal irritation scores were 13.8 after 1 hour, 18.8 after 24 hours, 11.3 after 48 hours, 5 after 72 hours, and 1.3 after 7 days (max 80). Mean iridial irritation scores were 3.8 after 1 hour and 24 hours, decreasing to 0 after 7 days. Mean conjunctival irritation scores were 11 after 1 hour, 7.5 after 24 hours, 4 after 48 hours, 3.5 after 72 hours, and 2 after 7 days. No irritation was observed 14 days after the instillation. With a total mean irritation score of 30.0 (max. total = 110.0), the formulation was considered moderately irritating.
A single 0.1 mL dose of a product formulation containing 6.0% active CAPB was instilled into the conjunctival sac of each of 6 albino rabbits in a Draize test. 1 Conjunctival irritation (mean score of 4; max = 20) was observed in all treated eyes on the first day following instillation, decreasing in severity on the second day. No corneal irritation or iritis was observed.
Mucous Membrane Irritation
Two soap formulations containing 7.5% CAPB were tested for vaginal irritation potential in Beagle dogs (7-10 months old; 8.2-10 kg). The formulations were tested in 3 dogs each. Prior to treatment and again before termination, hematology, clinical chemistry, and urinalysis were performed. A volume of 20 mL of the test material was administered into the vagina via a syringe once a day for 15 days (weekdays only). Vaginas and vulvas were examined 6 hours prior to and after each daily treatment. At termination of the study, the dogs were killed and necropsied. Tissue samples of the liver, kidney, and vulva/vagina were examined. Blood was found in the urine of 5/6 dogs. Gross necropsy revealed discoloration of the lining of the vagina in 5/6 dogs. Diffuse necrosis of vaginal mucosa occurred in 5/6 dogs and focal vaginal necrosis occurred in 1 dog (this dog was in estrus). There was corresponding inflammatory cell infiltration (mainly neutrophils) and often a fibrinopurulent membrane adherent to the injured surface. It was concluded that lesions were the result of test material application. Morphologic changes in the liver and kidneys in all dogs were not considered significant and were within normal parameters.100,101 (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 7.5% active or 7.5% aqueous, which equated to 2.25% active.)
Genotoxicity
Bacterial Assays
A commercial sample of CAPB (31.0% active) was tested using Salmonella typhimurium strains TA98, TA100, TA1535, TA1537, and TA1538, both with and without metabolic activation. The concentrations of CAPB solution tested were 0.004, 0.02, 0.1, 0.2, and 0.4 µL/plate. The CAPB is toxic above 0.3 µL/plate. The test material did not cause a significant increase in mutation frequency in any of the strains tested with or without metabolic activation. 102
CAPB (30% active) was tested using S typhimurium strains TA1535, TA1537, TA1538, TA98, and TA100, with and without metabolic activation. Eight concentrations between 0.001 and 0.300 µL/plate were used, based on CAPB solubility. The CAPB did not produce an increase in mutation frequency, with or without metabolic activation. 103
In a study summarized by the American Chemistry Council, CAPB (28.5-30.5% active) was tested using S typhimurium strains TA98, TA1535, TA1537, and TA1538, both with and without metabolic activation at 0, 50, 150, 500, 1500, or 5000 µg/plate. 35 Positive controls were N-ethyl-N’-nitro-N-nitrosoguanidine (for TA100 and TA1535), 9-aminoacridine (for TA1537), 4-nitro-o-phenylenediamine (for TA1538), 4-nitroquinoline-1-oxide (for TA98), and 2-aminoanthracene (in all strains with metabolic activation only). Cytotoxicity was observed at 150 µL/plate and above. The CAPB in this assay was found to be nonmutagenic.
The American Chemistry Council also summarized the findings of a CAPB (concentration not stated) mutagenicity assay using S typhimurium strains TA1535, TA1537, TA1538, TA98, and TA100, with and without metabolic activation. 35 The test material was tested at 1, 4, 16, 64, or 256 µg/plate without S-9 activation and at 4, 16, 64, 256, and 1024 µg/plate with S-9 activation. The CAPB did not increase the mutation frequency, with or without metabolic activation.
Mammalian Cell Assays
The mutagenic potential of a 30.9% active sample of CAPB was tested in a L5178Y TK
Animal Assays
The American Chemistry Council summarized a mouse micronucleus test that studied CAPB (concentration not stated). 35 Groups of 5 male and 5 female OF1 mice received 2 doses of either 0.02 or 0.2 g/kg of the test material in sterile distilled water via intraperitoneal injection (dose volume 10 g/kg) at 24-hour intervals. Negative and positive controls received sterile distilled water and cyclophosphamide, respectively. The rats were killed 6 hours after the second administration of the test material and bone marrow slides were prepared. One thousand polychromatic erythrocytes (PCEs) per animal were studied for the presence of micronuclei. In both dose groups, the number of micronucleated PCEs was not increased when compared to the negative control. The positive control group yielded expected results. The CAPB was not a mutagen under the conditions of this study.
Carcinogenicity
An aqueous preparation of a nonoxidative hair dye formulation containing an unspecified grade of CAPB at a concentration of 0.09% active CAPB was tested for carcinogenicity using groups of 60 male and female random-bred Swiss Webster mice from the Eppley colony. 105 The formulation also contained 5% propylene glycol, 4% benzyl alcohol, 0.6% kelzan (xanthan gum), 0.9% lactic acid, 0.04% fragrance, and less than 0.1% each of the disperse brown, red, yellow, and blue dyes. A dose of 0.05 mL per mouse was applied 3 times weekly for 20 months to interscapular skin that was clipped free of hair and shaved. Mortality, behavior, and physical appearance of the mice were observed daily. Dermal changes in particular were noted. Body weights were recorded weekly. Ten males and 10 females from each group were killed at 9 months for a hematological study, urinalysis, and necropsy. At termination, all mice were necropsied, and the tissues were examined microscopically. No adverse effects were noted on average body weight gains, survival, hematological or urinalysis values in any group. Varying degrees of chronic inflammation of the skin were seen in all groups, including controls. Other lesions occurred but were considered unrelated to hair dye treatment. The incidence of neoplasms in treated animals did not differ significantly from control groups.
Irritation/Sensitization Studies With Amidoamine, DMAPA, and Related Amines
Amidoamine is a term used for fatty acid esters of amidopropyl dimethylamine, intermediates in the synthesis of the amidopropyl betaines; DMAPA is also an intermediate in the synthesis of the amidopropyl betaines. These compounds can exist as impurities in cosmetic formulations containing amidopropyl betaines.
Animal Studies
Hill Top Research, Inc performed a delayed contact hypersensitivity study of stearamidopropyl dimethylamine in guinea pigs. 106 A pre-induction primary irritation study was conducted to determine the concentration for the induction phase of the study. Twenty Hartley outbred guinea pigs were treated with 1.0% w/v stearamidopropyl dimethylamine in 80% ethanol/20% distilled water. The test material was applied for 6 hours at a dose volume of 0.3 mL using 25 mm diameter occluded Hill Top chambers on clipped, intact skin on the left shoulder. [Estimated dose/unit area = 6.1 × 102 µg/cm2]. The exposure sites were rinsed after removal of chambers and re-exposed once a week for a total of 3 exposures. A control group of 10 guinea pigs received the vehicle alone. After a 2-week rest period, the animals received primary challenge patches of 0.25% w/v stearamidopropyl dimethylamine in acetone on naive skin. [Estimated dose/unit area = 1.5 × 102 µg/cm2]. One guinea pig had delayed contact hypersensitivity to the test material. The control animals had no reactions. A rechallenge was conducted in 6 guinea pigs 13 days after the primary challenge with 0.25%, 0.125%, and 0.0625% w/v stearamidopropyl dimethylamine. An additional 5 animals were used as controls. One guinea pig had a positive response to the test material at 0.25%. No other reactions were observed.
Palmityl/stearylamidopropyl dimethylamine at a concentration of 25% active in 8.95% phosphoric acid and 66.05% water was studied for delayed contact hypersensitivity using albino Dunkin/Hartley guinea pigs. 107 A preliminary irritation test was conducted to determine the maximum concentration for the induction and challenge phases of the study. In the induction phase, 10 male and 10 female animals received 0.4 mL of test material on a 4 cm2 patch on the clipped skin of the left shoulder for a period of 6 hours. [Estimated dose/unit area = 2.5 × 104 µg/cm2]. The patches were occluded. An additional 5 male and 5 female animals were left untreated as the controls. A total of 3 induction patches were applied, once weekly, for 3 weeks. Following a 2-week rest period, all animals received primary challenge patches of 0.4 mL of test material on the right flank for 6 hours. The test sites were scored at 24 and 48 hours postapplication. All but 3 of the 20 guinea pigs had patchy to severe erythema at the 24- and 48-hour observation periods. Four control animals had slight to moderate patchy erythema during the observation periods. Rechallenges were conducted on 0.25% active and 0.5% active palmityl/stearylamidopropyl dimethylamine. No sensitization was observed with the 0.25% active material, but 0.5% active material elicited reactions in sensitized animals. The study concluded that palmityl/stearylamidopropyl dimethylamine had the potential to cause delayed contact hypersensitivity in guinea pigs.
Two guinea pig maximization studies to assess the skin sensitization potential of amidoamine were evaluated. 71 In the first study, preliminary tests determined the maximum concentrations of intradermal injections, topical induction, and challenge applications. Ten albino Dunkin/Hartley guinea pigs (6 females and 4 males) received two 0.1 mL injections of 50% Freund complete adjuvant at the first pair of sites, two 0.1 mL injections of 0.1% amidoamine at the second pair of sites, and two 0.1 mL injections of amidoamine in DOBS/saline vehicle and Freund complete adjuvant (50/50 ratio) to yield a final concentration of 0.1% amidoamine at the third pair of sites. One week following the injections, a single occlusive 48-hour induction patch (2 × 4 cm) of 0.2 to 0.3 mL amidoamine 5% in acetone/PEG400 vehicle was applied to the same shaved area. Four male control animals received intradermal injections and induction patches using only the vehicles. Two weeks after the induction patch, all animals received a single occlusive 24-hour challenge patch (8 mm diameter patch in a Finn chamber) saturated with 0.5% amidoamine in acetone/PEG 400 on a clipped and shaved flank. The treatment sites were examined 24 and 48 hours after patch removal. Two more challenges were made 1 and 2 weeks after the first challenge. Reactions were scored on a scale of 0 (no reaction) to 3 (severe erythema and edema).
At the first challenge, 7 animals had a reaction score of
The second maximization study was conducted in the same manner as the first with the only changes being that 0.025% amidoamine was used in the intradermal injections instead of 0.1%, 1% amidoamine was used in the topical induction, only 2 challenges were made, and 4 female guinea pigs were used as controls.
At the first challenge, 3 animals had a reaction score of
Wright et al reported on the results of an LLNA study performed on 4 chemicals that are recognized human contact allergens, including DMAPA (99.0+ % pure). 72 The chemicals were tested in 7 different vehicles: acetone, olive oil (4:1), dimethylsulfoxide, methethylketone, dimethyl formamide, propylene glycol, and 50:50 and 90:10 mixtures of ethanol and water. Groups of 4 female CBA/Ca mice were exposed topically on the dorsum of both ears to 25 µL of 0.5%, 1.0%, 2.5%, 5.0%, or 10.0% of the test material, or to an equal volume of the appropriate vehicle alone, daily for 3 consecutive days. Five days after the initial topical treatment, all animals were injected intravenously with 20 µCi of [ 3 H] methyl thymidine. Approximately 5 hours after injection, the animals were killed and the auricular lymph nodes were excised. Single-cell suspensions were prepared from pooled lymph nodes, with the cells precipitated by trichloroacetic acid (TCA), and the radioactivity measured by liquid scintillation. The stimulation indices (SIs) were calculated, and at 10.0% DMAPA ranged from 2.2 in propylene glycol to 15.7 in dimethyl formamide. The estimated concentrations for a SI of 3 (EC3) ranged from 1.7% (in dimethyl formamide) to >10% (in propylene glycol).
An LLNA study was performed using stearamidopropyl dimethylamine (TEGO AMID S 18).
108
A certificate of analysis reported that the DMAPA level conformed to the
No deaths occurred during the treatment period in any dose group. No clinical signs of toxicity were observed during treatment in the control group or in the 0.1% and 0.5% dose groups. Slight to moderate ear erythema was observed after the second or third application at both dosing sites in all mice in the 1%, 2.5%, and the 5% dose groups. This persisted for 2 days in the 1% dose group and until treatment end in the 2.5% and 5% dose groups. Body weight development was not affected in any of the animals. The SIs were1.4, 2.1, 2.1, 5.8, and 3.9 for the 0.1%, 0.5%, 1%, 2.5%, and 5% dose groups, respectively. The EC3 was calculated at 1.4%. The positive control group had expected results and validated the study. The study concluded that steramidopropyl dimethylamine (TEGO AMID S 18) was a potential skin sensitizer in this LLNA test. 108
Calvert Laboratories, Inc performed an LLNA study using amidoamine (∼99% C12-C18). 110 A preliminary dose range study was performed. In the main study, groups of 5 mice received 0%, 0.1%, 0.5%, 1%, 2.5%, or 5% of the test material in ethanol/water, 7:3 (v/v) neutralized to pH 6.0 with citric acid monohydrate. An additional 5 mice received the positive control, 35% hexylcinnamaldehyde. The mice were treated on the dorsal surface of both ears (25 µL/ear) once daily for 3 days. On day 6, the mice were injected intravenously (iv) with 20 µCi of 3 H-thymidine. Five hours later, the mice were killed and the draining auricular lymph nodes were removed, processed, and assessed for lymphocyte proliferation. No mortality or adverse effects were observed throughout the study. Very slight erythema was observed on day 3 and very slight erythema and edema were observed on days 4 to 6 of the 2.5% dose group. In the 5% dose group, 4 of the 5 mice treated had very slight erythema and very slight edema on day 2. On days 3 to 6, mice in this dose group had well-defined erythema and slight edema. The SIs were 1.8, 1.0, 3.1, 24.5, and 60.6 for the 0.1%, 0.5%, 1%, 2.5%, or 5% dose groups, respectively. The EC3 for amidoamine was calculated at 0.98%. The positive control group had expected results and validated the study. This LLNA study concluded that amidoamine has skin-sensitizing activity.
Human Studies
Hill Top Research, Inc performed an investigation of the potential of stearamidopropyl dimethylamine to induce skin sensitization in 112 human participants. 73 Applications contained a concentration of 0.25% w/v of the test material in undiluted mineral oil. Induction applications of 0.3 mL were made to the same site, with a Webril patch for a total of 9 applications. Challenge applications were made to naive alternate sites. Frequent incidences of slight to moderate irritation, including erythema, some edema, papules, glazing, and cracking, were observed during the induction period but were considered transient. Five participants had a reaction of grade 1 or greater during the challenge phase. The responses to stearamidopropyl dimethylamine were indicative of primary irritation rather than contact sensitization.
In a study by Inveresk Research International, the sensitization potential of a 4% aqueous liquid fabric softener formulation containing 0.5% stearyl/palmitylamidopropyl dimethylamine was investigated using 77 participants. 74 During the induction phase, the test material was applied at a dose volume of 0.5 mL with a ¾ inch square Webril pad to the dorsal surface of the upper arm. [Estimated dose/unit area = 6.9 × 102 µg/cm2]. Patches were applied for a duration of 24 hours, 9 times over a period of 3 weeks. The test material caused some degree of irritation in most volunteers. After a rest period of 2 weeks, the participants received challenge patches with the same concentration of test material on both arms. Patch sites were graded 48 and 96 hours after patching. Eight participants reacted at challenge, and 7 submitted to rechallenge with 4% and 0.4% aqueous formulations. No reactions indicative of sensitization occurred at rechallenge. The test formulation containing stearyl/palmitylamidopropyl dimethylamine had no significant sensitization potential.
Foti et al patch tested 285 consecutive dermatitis patients with the European standard series supplemented with oleamidopropyl dimethylamine (0.5% aqueous), CAPB (1% aqueous), and DMAPA (1% aqueous). 75 The standard patching technique was employed and test sites were scored on days 2, 3, 4, and 7. Twenty-three patients (8%) had allergic responses to DMAPA, 14 patients (4.9%) had allergic responses to DMAPA and oleamidopropyl dimethylamine, and 8 patients (2.8%) had allergic responses to all 3 of the supplemental chemicals. Analyses by thin-layer chromatography (TLC) of the oleamidopropyl dimethyl amine sample revealed contamination by DMAPA (6 ppm or 0.12% of the sample) and indicated that the allergic responses in the last group were not due to cross-reaction. (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
In a 2-year study by Pigatto et al, 1190 patients with eczema were patch tested with 1% aqueous CAPB using standard technique and grading according to the European Contact Dermatitis Group (ECDG). 76 From this patch test, 17 patients were diagnosed with allergic contact dermatitis to CAPB. Relevance was established with an additional positive patch test of 2+ or more to at least 1 personal care product containing CAPB used by the patients. Fifteen patients were further tested with CAPB 0.01%, 0.5%, 1% (from 2 different manufactures), and 2% in water; and DMAPA at 0.05%, 0.1%, and 1% in petrolatum; and, if possible, the patients’ reported cosmetics diluted in water at 1:10, 1:100, and 1:1000.
In 12 patients tested with their own personal cosmetics, 9 had positive reactions to at least 1 dilution and 5 had irritant reactions. All except 3 patients, who were not tested, had 2 or 3+ reaction to DMAPA at concentrations as low as 0.05%. Only 1 patient had a positive reaction to CAPB. The presence of DMAPA was investigated via TLC in the personal cosmetics of 4 of the patients that had positive reactions. These positive reactions from DMAPA suggest that the positive reaction to CAPB-containing products was likely due to a certain concentration of DMAPA that was an impurity. The DMAPA was measured in the products at 50 to 150 ppm. The concentration of DMAPA was also measured in the 2 CAPB types: one had a concentration of DMAPA at 200 ppm and DMAPA was below detection level (level not reported) in the other type. The authors stated that the sensitizing agent in CAPB allergy is DMAPA, although their findings did not exclude the role of CAPB itself from causing allergic dermatitis. 76 (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
A study of sensitization to commercially available CAPB in patients with dermatitis was performed by Angelini et al. 77 Twelve hundred consecutive patients with dermatitis of various types were patch tested with the European standard series and CAPB 1% aqueous (30% active ingredient). Some of the patients that had allergic or irritant reactions to CAPB were then patch tested with the chemicals that were intermediates or reactants in the synthesis of CAPB (amidoamine, DMAPA, and monochloroacetic acid) along with a sample of CAPB of greater purity and Tego 103 G 1% aqueous.
Positive allergic reactions to CAPB were observed in 46 participants (3.8%), while irritant reactions were recorded in 15 participants (1.25%). Of these 46 participants, 30 had positive reactions to DMAPA 1% aqueous. In these 30 participants, 3 and 16 were positive to the purer grade of CAPB 0.5% aqueous and CAPB 1% aqueous, respectively. Patients with irritant reactions had negative reactions to the synthetic materials and to the purer grade of CAPB. No allergic or irritant reactions to DMAPA were observed in 50 healthy controls. No positive reactions to amidoamine 0.05% were observed. The authors concluded that the results suggested that DMAPA impurity was responsible for CAPB allergy. 77 (From the study documentation, it was not possible to determine whether the administered CAPB concentrations were 0.5% active and 1% active or 0.5% aqueous and 1% aqueous, which equated to 0.15% active and 0.3% active, respectively.)
A further study by Angelini et al was performed to determine whether CAPB or an impurity of CAPB was responsible for cases of contact dermatitis. 78 In this study, TLC was employed to analyze a sample of CAPB (Tego Betaine F 30% solution) and isolate and identify unknown impurities other than DMAPA, chloroacetic acid, and amidoamine found in the CAPB solution. An infrared spectrum analysis was used to confirm the presence of the sodium salt of N,N-dimethyl-propylene-diaminotriacetic acid.
Upon identifying the impurity, 30 patients with a history of contact allergy to 1% aqueous CAPB and 1% DMAPA were patch tested with pure CAPB and a blend containing sodium chloride and N,N-dimethyl-propylene-diaminotriacetic acid (both at 1%). None of the participants reacted to any of the chemicals. The authors suggested that pure CAPB, chloroacetic acid, amidoamine, and N,N-dimethyl-propylene-diaminotriacetic acid were not the components responsible for CAPB sensitivity and the involvement of DMAPA cannot be ruled out. 78 (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
In another study by Angelini et al, DMAPA was tested at varying concentrations with other tensioactive chemicals to determine whether they enhanced sensitivity to DMAPA. 79 Thirty-four participants with confirmed contact allergy to 1% aqueous DMAPA were patch tested with DMAPA in water, DMAPA in a SLES 2% aqueous solution, and DMAPA in a polysorbate 20 2% aqueous solution, all in decreasing concentrations from 0.1% to 0.00005%. The participants were also patch tested with CAPB and a series of 10 substances chemically related to DMAPA. Test sites were occluded for 2 days and the sites were measured for reactions on days 2, 3, 4, and 7.
Eighteen participants had positive reaction to DMAPA in water at 0.1%. No positive reactions were noted for DMAPA in water at 0.01% to 0.00005%. Positive reactions were observed in DMAPA in SLES, with 27 participants positive at the highest concentration, 10 participants positive at 0.01%, 5 participants positive at 0.005%, and 1 participant positive at 0.0001%. Positive reactions were also observed in DMAPA in polysorbate 20 in 21 participants at 0.1% and 4 participants at 0.01%. Patch tests for the chemically related structures were positive in 28 participants for N,N-dimethyl-2-ethylenediamine 1% aqueous, 12 participants for cocamidopropylamine oxide 1% aqueous (35% active material), and 18 participants for CAPB 1% aqueous (30% active material). No other reactions occurred. The authors concluded that tensioactives such as SLES and polysorbate 20 may enhance the risk of sensitization to DMAPA at low concentrations. They also concluded that the primary amine and the tertiary amine groups (dimethyl substituted) are the sensitizing chemical structures in DMAPA and related molecules when they are separated by 2 or 3 carbon atoms. 79
In another study by Angelini et al, 20 patients (ages 17-51 years, 13 females and 7 males) with confirmed contact allergy to DMAPA (1% aqueous) and CAPB (1% aqueous) were tested. 80 All the patients had intolerance to detergents and shampoos and none were sensitized through an occupation. The patients were patch tested using serial dilutions of DMAPA (100 ppm) in surfactant solutions (1% or 2% w/w surfacatants) that included purified CAPB (DMAPA <1 ppm), SLES, polysorbate 20 (Tween 20), lauryl polyglucoside (APG), SLES/CAPB 3:1 (w/w), and APG/CAPB 3:2 (w/w). The test sites were scored on days 2, 3, 4, and 7. (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
Positive reactions were observed in serial dilutions of DMAPA in 1% CAPB at 1 ppm and higher (1 reaction each to 1 ppm and 5 ppm DMAPA, 3 reactions to 10 ppm DMAPA, and 4 reactions to 50 ppm DMAPA). Similar positive observations were made in serial dilutions of DMAPA in 1% SLES/CAPB 3:1. No positive reactions were observed when DMAPA (100 ppm) was tested in water, but 7 positive reactions were recorded when the material was tested in 2% CAPB. A greater number of reactions were observed when 100 ppm DMAPA was mixed with 2% SLES/CAPB (5 reactions) than when mixed with 2% APG/CAPB (2 reactions). The authors noted that CAPB and SLES/CAPB 3:1 act as carriers for DMAPA when applied under occlusion at 1%, and that surface activity in more concentrated surfactant solutions may be responsible for allergic reactions by DMAPA. The authors concluded that the concentration limit for DMAPA in 1% CAPB or 1% SLES/CAPB 3:1 should be 0.5 ppm (corresponding to 15 ppm and 60 ppm, respectively) and that betaine should be blended with nonionic surfactants to reduce allergy risks. 80 (From the study documentation, it was not possible to determine whether the administered CAPB concentrations were 1% active and 2% active or 1% aqueous and 2% aqueous, which equated to 0.3% active and 0.6%, respectively.)
Uter studied 80 participants (mainly hairdressers) with dermatitis from 1996 to 1999. 81 During this period, the participants were patch tested with the hairdresser’s series supplemented with DMAPA (1% pet and 1% aq Uter). The hairdresser’s series contained CAPB (1% aqueous) that had a maximum residual DMAPA of <15 ppm. Of the 80 participants, 6 had + to +++ reactions to CAPB, but none of the 6 had reactions to DMAPA. A housewife with scalp and neck dermatitis had a + reaction to DMAPA 1% aqueous and a +? reaction to DMAPA 1% pet. This participant had no positive reaction to CAPB. (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
McFadden et al studied 7 participants that had relevant dermatitis to CAPB. 82 The dermatitis occurred after use of liquid soaps, and in one case an eye makeup remover that contained CAPB. Four of the 7 participants were patch tested with partially purified CAPB (1% aqueous) containing <0.5% cocamidopropylamine and 0.1% and 0.01% cocamidopropylamine. The patch sites were read at day 2 and day 4 after the initial patching. One participant had a positive reaction that appeared only with cocamidopropylamine. Another had a reaction only with CAPB; however irritancy could not be ruled out since the participant’s patch sites were only read on day 2. The other 2 patients had positive reactions to cocamidopropylamine and CAPB. Control participants had negative patch results.
Six out of the 7 original participants with dermatitis were patched tested with DMAPA along with controls on normal and tape-stripped skin at 0 ppm to 10 000 ppm. The participants were also tested with DMAPA in the presence of 0.2% aqueous, SLS, or in the presence of 1.0% pure CAPB (<0.3% cocamidopropylamine, <10 ppm DMAPA). The patch sites were again read on day 2 and day 4 after the patch applications. One of the 6 participants reacted to DMAPA on normal and tape-stripped skin at concentrations >1000 ppm. Three of the 6 participants reacted to DMAPA in the presence of SLS (1 at 10 000 ppm, 1 at 1000 to 10 000 ppm, and 1 at 100 to 10 000 ppm). None of the participants reacted to the 1.0% pure CAPB. The authors concluded that the sensitization experienced by the participants to the CAPB products was likely due to the residual intermediates from the CAPB production, with reaction to cocamidopropylamine more likely than DMAPA. 82 (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
The impurities DMAPA and amidoamine in CAPB were further analyzed for sensitization potential in 10 participants with CAPB allergy. 83 The participants that had all tested positive to CAPB 1% aqueous (Firma type) were patch tested with CAPB 1% aqueous (Chemotechnique type), DMAPA 1% aqueous, and purified amidoamine at 0.5%, 0.25%, and 0.1% aqueous. All the participants had ++ reactions to DMAPA at 1% and purified amidoamine at 0.5%. Most participants also had ++ reactions to purified amidoamine at 0.25% and the remaining had + reactions to this concentration. Four patients had positive reactions (++) to the purified amidoamine at 0.1%. No reactions were observed to the CAPB from Chemotechnique, which was suggested to have a higher purity by the authors. Control patches in 20 volunteers were negative for amidoamine. The authors concluded that cross-reactivity between DMAPA and amidoamine causes CAPB allergy. They also suggested that DMAPA is the true sensitizing material and amidoamine aids in the trans-epidermal penetration of DMAPA. (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
Brey and Fowler performed a retrospective study of patients that had positive patch test results to 1.0% aqueous CAPB and/or 1.0% amidoamine in the year 2001. 84 Reactions to other allergens were also recorded. Out of 957 patients patch tested in 2001, 49 had positive reactions to CAPB, amidoamine, or both. A follow-up evaluation in 35 patients was performed to establish relevance of reactions to CAPB and amidoamine with the use of products containing these chemicals. Fifteen patients (42.9%) reacted to CAPB, 12 patients (34.3%) reacted to amidoamine, and 8 patients (22.8%) reacted to both. Of the 35 patients, 29 (83%) could identify products containing CAPB at home. (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
Fowler et al performed a retrospective study of patients with CAPB and/or amidoamine contact allergy in 2001. 111 Out of 975 patients, 15 had a positive patch test reaction to 1.0% CAPB only, 25 had a positive patch test reaction to 0.1% amidoamine only, and 18 had positive reactions to both (58 patients total). Definite and probable relevance (known exposure to CAPB) was determined in 16 patients that tested positive for amidoamine and in 16 that tested positive for CAPB. This study also evaluated formaldehyde allergy. Of the 58 patients, 12.7% were also allergic to formaldehyde. This was compared to the 10.1% of the total 975 patients that had formaldehyde allergy. The authors suggested that there is no significant relationship between CAPB or amidoamine allergy and formaldehyde allergy. (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
The NACDG evaluated 4913 patients for allergic contact dermatitis with an extended screening series of 65 allergens from January 1, 2001 to December 31, 2002. CAPB (1% aqueous) and the by-product of CAPB production, amidoamine (0.1% aqueous), were both included in this screening series. Positive results for CAPB were observed in 2.8% of the patients, while 2.3% were positive for amidoamine. The relevance of the CAPB and amidoamine reactions (present and past) was 90.9% and 85%, respectively. 112 (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
In a study by Li to determine the sensitization rate of CAPB in China and to analyze the relationship between CAPB and DMAPA, 429 patients (105 male, 324 female; 9-81 years old) with suspected contact allergy were patch tested with 1% aqueous CAPB (purified) and 1% aqueous DMAPA. 113 The patients were also tested with the European standard series.
Of the 429 participants tested, 9 had irritant reactions, 12 had questionable reactions, and 42 had + reactions to CAPB. No reactions to CAPB greater than ++ were observed. Also of the 429 patients, 76 were diagnosed with cosmetic allergic contact dermatitis. Twenty-seven of these participants and 15 (out of 353) of the participants with cosmetic allergic contact dermatitis had positive reactions to CAPB (P < .05). Only 25 of the former and none of the latter had relevant reactions. Ten of the 429 patients had positive reactions to DMAPA, 8 of which were considered relevant. Six of the 10 patients also had positive reactions to CAPB. Because the participants of this study had positive reactions to both CAPB (purified) and DMAPA, the authors recommended that patch tests in cases of suspected cosmetic allergic contact dermatitis contain both CAPB and DMAPA. 113 (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
Provocative Use Studies
A provocative use study of products containing CAPB was performed by Fowler et al. 114 Ten participants were identified through positive reactions to 1% aqueous CAPB in routine patch testing. Ten control participants negative to CAPB were also enrolled. The provocative use test was divided into 3 phases, with 3 different test products (shampoo, liquid hand soap, and body wash) used in each phase. The products were specially formulated with CAPB-F grade (active level of CAPB in shampoo was 5.0%; active level in hand soap and body wash was 5.2%). Phase I was a forearm wash test with the shampoo diluted to 10% in tap water. If no allergic reaction occurred in Phase I, participants then entered Phase II of the study: daily use of shampoo as hair cleanser. Participants proceeded to phase III of the study if no allergic reactions to the shampoo occurred. In phase III, the participants used the shampoo, body wash, and hand soap for 3 weeks.
At least 2 months after the product use tests, the participants were patch tested with CAPB grades F and S (both 1% aqueous), DMAPA (0.1% pet), amidoamine (0.1% aqueous), sodium monochloroacetate (0.1% aqueous), a proprietary mixture of preservatives for CAPB, and other potential allergens (perfumes and preservatives) that were in the test product formulations. Control participants were patched with 1% CAPB.
Three participants completed the product use phases without experiencing an allergic reaction. Seven participants had erythema, scaling, and pruritus on the arms, face, and/or neck in either phase I or II of the study. One participant that experienced a positive reaction in the first phase was asked to repeat the forearm use test with the CAPB-containing shampoo on the left arm and with a CAPB-absent shampoo on the right arm. The participant experienced a positive reaction on both arms, which was likely caused by the preservatives in the shampoo products (as shown through patch testing). In phase III, 3 participants had scalp, face, and/or neck and body dermatitis.
Patch testing was performed in 9 of the 10 participants, with 6 participants reacting to 0.1% amidoamine. Five of these 6 participants had positive reactions during the product use phases. Two participants had reactions to the CAPB-F grade with preservative, 3 had reactions to CAPB-F grade without preservative, 1 reacted to the CAPB-S grade, and 1 reacted to the proprietary preservative mixture. Two participants had questionable reactions to DMAPA. No other adverse reactions were noted in the participants. (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
A follow-up patch test with 7 of the participants was performed using purified CAPB (containing only 1 ppm amidoamine), CAPB-F grade (with approximately 3000 ppm amidoamine), and 2 concentrations of amidoamine (0.1% and 0.01% aqueous). Two participants had questionable reactions to the purified CAPB, while there were 3 positive reactions to the CAPB-F grade, 4 positive reactions to the higher concentration of amidoamine, and 2 positive reactions to the lower concentration of amidoamine. The authors concluded that the impurity amidoamine may be the causative allergen in CAPB sensitivity and they recommend that cosmetics and personal care products should be formulated to minimize contamination with this impurity. In addition, the authors could not rule out the possibility that CAPB alone was not an allergen to presensitized individuals.114,115
Another provocative use test was conducted by Fartasch et al. 116 Participants with eczema were tested for CAPB allergy while undergoing patch testing for the standard allergen series. Out of 1063 patients, 13 were identified with a positive patch reaction; however, relevance could only be established in 4 of the participants. Another 6 patients were referred to the study for eczematous eruptions of the scalp and/or hand dermatitis and had positive 1% aqueous CAPB patch test reactions. Twenty volunteers served as controls for the study.
The product use study consisted of 3 phases. In phase I, a 0.1 mL test sample of shower gel containing CAPB (25% dilution; DMAPA below 1 ppm) was applied, lathered for 1 minute, and rinsed on the participants’ forearms twice daily for 7 days. The second phase of the study consisted of patch testing in order to differentiate irritant reactions from allergic reactions and to reconfirm the sensitivity to CAPB and DMAPA. The participants were patch tested with 0.1%, 0.3%, and 1.0% dilutions of CKKB (Tegobetaine CKKB5; 1.1 ppm DMAPA) and DMAPA, respectively. Patch sites were read on days 2, 3, and 4 following application. Participants that had no allergic reactions in phase I participated in phase III. In this phase, the participants used the shower gel as they would in normal daily hygiene practices for 4 weeks.
No skin irritation was observed in phase I of the study. One participant with a history of atopic dermatitis was removed from the study due to a flare. Another participant had an immediate “wheal like reaction” on days 3 and 6 that cleared within minutes. This participant continued the forearm test an extra week and had no further effect. In phase II, 1 control had an irritating reaction to 1% CAPB. In the study group, 5 out of the 10 participants had a positive reaction to 1% CAPB and another 3 had marginal and/or irritant reactions. One participant had a positive reaction to DMAPA but had no clear reaction to CAPB. Another participant that had a positive reaction to CAPB had a doubtful reaction to 1% DMAPA. Eight participants did not react to DMAPA. Only 7 participants participated in phase III of the study (the other 2 were not available), and no adverse reactions were observed in these participants. The authors concluded that CAPB as tested may be used safely in individuals with CAPB sensitivity. 116 (From the study documentation, it was not possible to determine whether the administered CAPB concentration was 1% active or 1% aqueous, which equated to 0.3% active.)
Case Reports
Several case studies of allergic contact dermatitis reported positive patch tests to amidoamine and DMAPA, with 1 study reporting DMAPA elicited reaction at concentrations of 0.1% and greater.9,123–127
Quantitative Risk Assessment
The Personal Care Products Council’s Task Force on Sensitization Risk from CAPB Impurities used a quantitative risk assessment (QRA) approach developed by Api et al. 51 and the Research Institute for Fragrance Materials (RIFM) 117 to determine the levels of DMAPA and amidoamine impurities for which no sensitization should occur. 118 Based on the findings of LLNA and human sensitization studies on DMAPA and amidoamine described in this report, the Council’s task force determined the conservative weight of evidence no expected sensitization induction levels (WoE NESIL) for DMAPA and amidoamine to be 425 µg/cm2 and 180 µg/cm2, respectively. When the level of impurities in raw CAPB materials is determined for product exposure (based on a typical exposure of 0.5% for amidoamine and 0.01% for DMAPA and estimated dose per unit area), a level of acceptable risk can be calculated for each cosmetic product category. These values are calculated based on sensitization assessment factors (SAFs), acceptable exposure levels (AELs = WoE NESIL × SAF−1), and consumer exposure level (CEL) that are appropriate for each product category. According to the QRA method, the ratio of AEL × CEL−1 must be equal to or greater than 1 to ensure no sensitization to consumers. See Tables 9 and 10 for the breakdown of the values used in the calculations for this QRA. The QRA found that all of the product categories had acceptable levels of risk for exposure to DMAPA.
a Assumptions in table above: AA @ 0.5% of CAPB; AA NESIL = 180 µg/cm2.
b Shaded rows indicate the ratio of AEL × CEL−1 is less than 1.
c These data are derived from RIFM. It is advisable that formulators use experimentally determined exposure data when available.
d Note that these product categories may be diluted prior to application, such that maximum CAPB activity in finished product is 3%.
Quantitative Risk Assessment of 3,3-Dimethylaminopropylamine (DMAPA) in Cosmetic Products Containing CAPBa,118
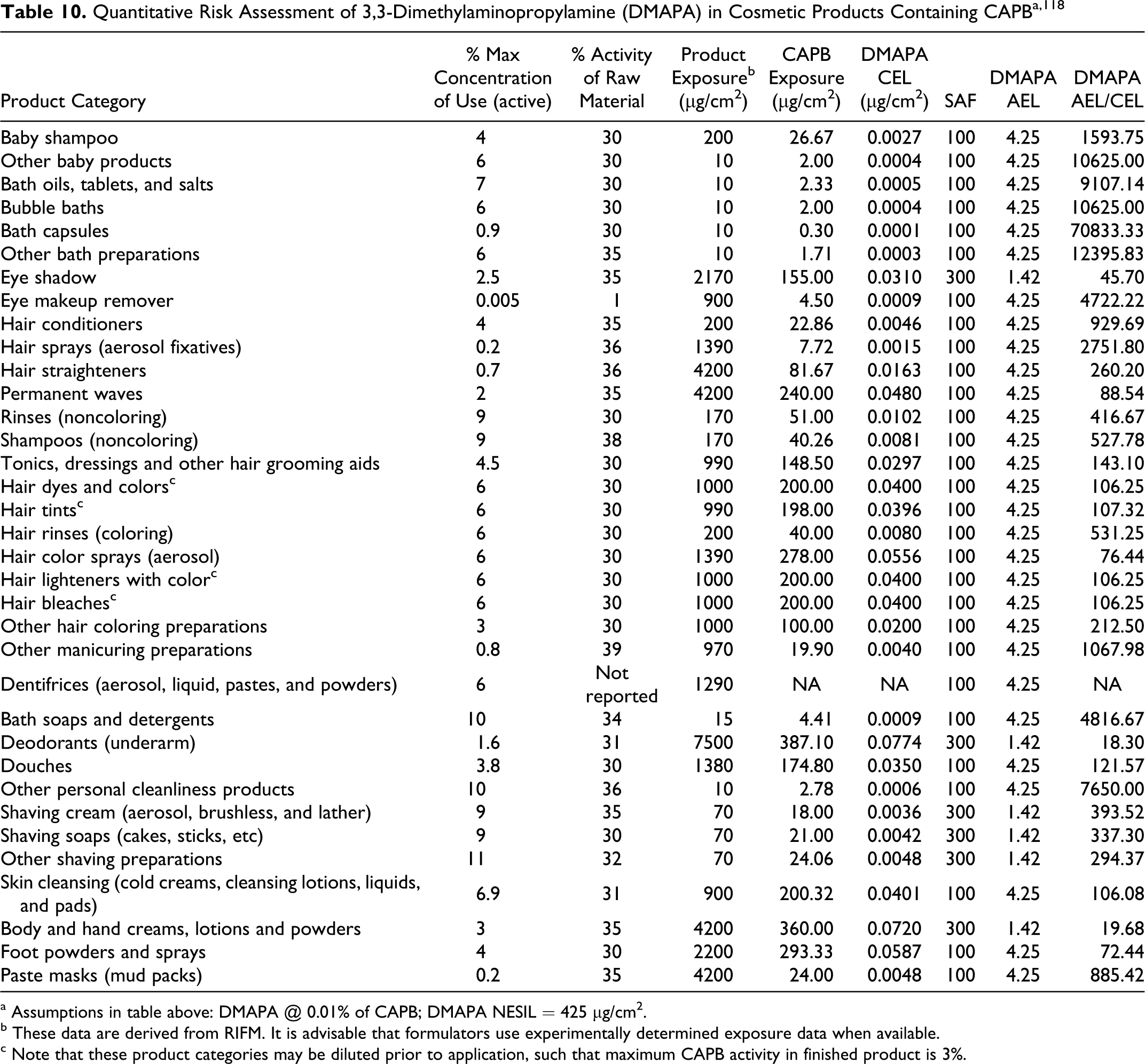
a Assumptions in table above: DMAPA @ 0.01% of CAPB; DMAPA NESIL = 425 µg/cm2.
b These data are derived from RIFM. It is advisable that formulators use experimentally determined exposure data when available.
c Note that these product categories may be diluted prior to application, such that maximum CAPB activity in finished product is 3%.
Using this approach, a ratio of less than 1 may result using the parameters given above, for example, with amidoamine in underarm deodorants (AEL × CEL−1 = 0.15). Such a finding could be addressed for such particular product applications by reducing the concentration of CAPB raw material in these finished products or choosing CAPB of higher purity when producing these products.
Summary
Cocamidopropyl betaine is a zwitterionic ammonium compound containing a moiety of either a saturated or unsaturated fatty acid ranging in length from 6 to 18 carbons in amide linkage with aminopropyl betaine. The source of these fatty acids, predominately lauric acid, is coconut oil. Other related ingredients are amidopropyl betaines with attached fatty acid moieties unique to the source, for example, sesame oil for sesamidopropyl betaine.
Cosmetic grade CAPB, an aqueous solution, normally contains 35% solids. The NaCl content of these solids ranges from 4.5% to 5.6%. The concentration, when expressed as activity, is determined by subtracting the percentage NaCl from the percentage total solids. Because of uncertainty in whether concentrations given are active or dilutions of an active cosmetic grade material, in some cases the actual concentration of CAPB or other tested material is not known, but it appears that any uncertainty would not be greater than a factor of 3. No N-nitroso compounds were detected in samples of commercially supplied CAPB analyzed by gas chromatography–thermal energy analysis.
CAPB is used primarily as an amphoteric surfactant in shampoos, conditioners, and other cleansing preparations. It was listed as an ingredient in 2460 cosmetic formulations voluntarily reported to FDA. Reported use concentrations range from 0.2% to 25%.
The oral LD50 of full-strength commercial samples of 30% active CAPB was 4.91 g/kg in CFR mice and 7.45 mL/kg in Wistar rats. Another study of 30% active CAPB in Wistar rats found the acute oral LD50 to be 8.55 g/kg. The oral LD50 of 30% active CAPB in albino rats of an unspecified strain was 4.9 g/kg. The acute oral LD50 for 35.61% active CAPB was >1.8 g/kg for male Sprague-Dawley rats. All female rats in this study died before study end. The acute oral LD50 was greater than 5.0 g/kg and the acute lethal dermal dose was greater than 2.0 g/kg in studies of CAPB (31% active) with CD rats.
In a 28-day short-term study in which groups of 8 male and female animals received 0, 100, 500, or 1000 mg/kg of 30% active CAPB, treatment-induced lesions were produced in the nonglandular portion of the stomach in the high-dose groups. Both males and females of the low-dose (100 mg/kg) group were comparable to concurrent controls.
In another 28-day oral toxicity study, rats received 0, 250, 500, or 1000 mg/kg of an unknown concentration of CAPB. In the 1000 mg/kg dose group, compound-related edema of the mucosa of the nonglandular stomach was observed at macroscopic examination and acanthosis of the mucosa, inflammatory edema of the submucosa, and multiple ulcerations were observed during microscopic examination. These effects were thought to be the result of the irritating properties of CAPB and not of systemic toxicity. The NOEL and LOEL for this study were 500 and 1000 mg/kg per d, respectively.
A subchronic oral toxicity study of an unknown concentration of CAPB rats that received 0, 250, 500, or 1000 mg/kg per d CAPB concluded that the NOEL was 250 mg/kg per d. Gastritis of the forestomach was observed in rats in the 500 and 1000 mg/kg per d dose groups.
Topical administration of varying commercial grades of CAPB (7.5%-30% activity) in single insult occlusive patch tests involving rabbits resulted in PIIs ranging from 0 to 3.75 (maximum score = 8). Slight edema was observed with CAPB with a 10% activity but not with CAPB with a 7.5% activity.
No evidence of delayed contact hypersensitivity was found in Pirbright white guinea pigs topically administered solutions of 10% active CAPB in a Magnusson-Kligman maximization test. Microscopic changes in the treated skin of albino guinea pigs indicated slight delayed-type contact sensitization by a 3.0% active CAPB solution in a maximization test and modified Draize test.
Maximum mean irritation scores for eyes of rabbits treated with 30% active CAPB and left unrinsed ranged from 26 to 42 (maximum score = 110). Score for rinsed eyes ranged from 2 to 10. Irritation was observed primarily in the conjunctivae of treated eyes. At 4.5% active CAPB, there was slight conjunctival irritation in unrinsed eyes and very slight irritation in rinsed eyes. Scores for product formulations containing 2.2% to 6.3% active CAPB ranged from 4 to 30 in unrinsed, treated eyes of rabbits and were 3.3 and 20.0 in rinsed, treated eyes of rabbits.
The mutagenic potential of 30.9% and 31.0% active CAPB formulations was tested in the Salmonella/mammalian microsome mutagenicity assay and the L5178Y TK +/− mouse lymphoma assay. CAPB was nonmutagenic in these assays. CAPB was not mutagenic to the S typhimurium indicator organisms in Ames Salmonella/microsome reverse mutation assays and in a mouse micronucleus assay.
In a single insult occlusive patch test of a 1.0% aqueous dilution of a product formulation containing 6.3% active CAPB, no skin irritation was observed in 15 of 19 human participants; 4 of the participants had slight irritation. Slight erythema was observed after occlusive patching of 12 participants with an 8% aqueous dilution of a soap formulation containing 2.0% active CAPB daily for 5 days. Two soap formulations containing 2.25% active CAPB were considered primary irritants after a 21-day consecutive occlusive patch study.
A formulation containing almondamidopropyl betaine and olivamidopropyl betaine (both at 0.005% active concentration) was not a primary skin sensitizer or skin irritant in 103 participants. A formulation containing capryl/capramidopropyl betaine at 1.72% active concentration was not a skin sensitizer in 26 participants. No dermal irritation or allergic contact sensitization was reported in studies of formulations containing 0.42%, 0.7%, or 0.03955% active lauramidopropyl betaine. Formulations containing shea butteramidopropyl betaine were not sensitizing in studies of 0.04% or 0.54% active concentration.
An additional study investigated the potential of a 3.0% active solution of CAPB to induce contact photoallergy. There was no response to the challenge tests except for those exposed to both UVA and UVB radiation, who had mild to moderate erythemic responses that were not uncommon and were said to have resulted from the sunburn derived from UVB exposure.
CAPB was not a skin sensitizer at 1% in a study of 100 volunteers or in another study at 1.5% in 141 volunteers. Clinical sensitization studies and case studies show that persons already sensitized to CAPB react to concentrations of 1.0% of the material in water. Several case reports have found patients reporting contact allergy to multiple types of personal care products, including shampoos, contact lens solutions, eye makeup remover, bath gels, and toothpaste. Researchers have included the CAPB impurities, DMAPA and amidoamine, in the scope of sensitization and case studies and have found that one or both of the impurities may be the responsible agent for contact allergy to CAPB. QRAs of these impurities may be performed to ensure acceptable levels of risk in consumers.
Discussion
While very few toxicity studies were identified specifically for the additional amidopropyl betaines (with R groups representing fatty acids derived from a source other that coconut oil) that were added to this safety assessment, there is no reason to expect these ingredients to differ in toxicity from CAPB. The amidopropyl betaines appear to be manufactured in the same manner as CAPB, with the difference only being in the fatty acid composition of the oil that is the source of the R group. Some of these fatty acid compounds have already been reviewed by the Panel and have been found to be safe for use in cosmetic ingredients. The Panel noted gaps in the available safety data for some of the amidopropyl betaines in this safety assessment. The available data on many of the ingredients are sufficient, however, and similarity between structural activity relationships and biologic functions in cosmetic concentrations of use and can be extrapolated to support the safety of the entire group. Therefore, the Panel determined that the toxicity data on CAPB could be read across to include: almondamidopropyl betaine, apricotamidopropyl betaine, avocadamidopropyl betaine, abassuamidopropyl betaine, behenamidopropyl betaine, canolamidopropyl betaine, capryl/capramidopropyl betaine, coco/oleamidopropyl betaine, coco/sunfloweramidopropyl betaine, cupuassuaidopropyl betaine, isostearmidopropyl betaine, lauramidopropyl betaine, meadowfoamamidopropyl betaine, milkamidopropyl betaine, minkamidopropyl betaine, myristamidopropyl betaine, oatamidopropyl betaine, oleamidopropyl betaine, olivamidopropyl betaine, palmamidopropyl betaine, palmitamidopropyl betaine, palm kernelamiodpropyl betaine, ricinoleamidopropyl betaine, sesamidopropyl betaine, shea butteramidopropyl betaine, soyamidopropyl betaine, stearamidopropyl betaine, tallowamidopropyl betaine, undecyleneamidopropyl betaine, and wheat germamidopropyl betaine.
In reviewing studies involving CAPB and related ingredients, often the percentage of active material in the test material was clearly stated; but in other cases, it was not clear whether the test material was active material or a dilution of active material. Because the difference, at most, would be a factor of 3, the uncertainty was factored into the review process.
The Panel considered that the available acute, short-term, and subchronic animal toxicity studies were supportive of the safety of CAPB. In vitro genotoxicity studies supported the absence of mutagenic activity. The Panel noted the absence of reproductive and developmental toxicity and absorption data but also noted that CAPB did not produce systemic toxicity in a 92-day oral toxicity study in rats. Because these ingredients are very large molecular weight structures and water soluble, the Panel concluded that they would not be readily absorbed into the skin.
In the absence of inhalation toxicity data, the Panel determined that CAPB can be used safely in hair sprays, because the product particle size was not respirable. The Panel reasoned that the particle size of aerosol hair sprays (∼38 µm) and pump hair sprays (>80 µm) was large compared to respirable particulate sizes (
In past ingredient safety assessments, the Panel had expressed concern over N-nitrosation reactions in ingredients containing amine groups. CAPB, and the other betaine ingredients in this assessment, contain secondary amides that may serve as substrates for N-nitrosation. Additionally, these ingredients may contain secondary amine impurities which may serve as substrates for N-nitrosation. Therefore, the Panel recommended that these ingredients should not be included in cosmetic formulations containing N-nitrosating agents.
The Panel expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use the necessary procedures to limit these impurities in the ingredient before blending into cosmetic formulation.
The Panel considered the dangers inherent in using animal-derived ingredients, namely the transmission of infectious agents. While tallow may be used in the manufacture of some ingredients in this safety assessment and is clearly animal derived, the Panel noted that tallow is highly processed and tallow derivatives even more so. The Panel agreed with determinations by the FDA that tallow derivatives are not risk materials for transmission of infectious agents.
While CAPB and the related amidopropyl betaines were noted to be dermal irritants, the primary concern was related to the presence of impurities that were found to be dermal sensitizers. The Panel recognized that these ingredients can have the potential to induce skin sensitization, most likely due to the impurities DMAPA and fatty acid amidopropyl dimethylamine (amidoamine). Thirteen studies of CAPB and related amidopropyl betaines on normal human skin at use concentrations indicated no sensitization induced by these cosmetic ingredients. A QRA on DMAPA at a concentration of 0.01% in raw CAPB indicated no sensitization in finished cosmetic products; amidoamine at a concentration of 0.5% in raw CAPB may cause sensitization in certain finished cosmetic products. The Panel concluded that skin sensitization is not a concern with the use of CAPB and related amidopropyl betaines as currently used in cosmetic products when a QRA is performed to demonstrate that concentration, product type, and product usage will not produce exposures that could induce sensitization. The Panel advises industry to continue minimizing the concentrations of the sensitizing impurities.
Conclusion
The CIR Expert Panel concluded that the following ingredients are safe in cosmetics as long as they are formulated to be nonsensitizing, which may be based on a QRA cocamidopropyl betaine, almondamidopropyl betaine, apricotamidopropyl betaine*, avocadamidopropyl betaine*, babassuamidopropyl betaine, behenamidopropyl betaine*, canolamidopropyl betaine*, capryl/capramidopropyl betaine, coco/oleamidopropyl betaine, coco/sunfloweramidopropyl betaine*, cupuassuamidopropyl betaine*, isostearamidopropyl betaine*, lauramidopropyl betaine, meadowfoamamidopropyl betaine*, milkamidopropyl betaine*, minkamidopropyl betaine*, myristamidopropyl betaine, oatamidopropyl betaine, oleamidopropyl betaine*, olivamidopropyl betaine, palmamidopropyl betaine*, palmitamidopropyl betaine*, palm kernelamidopropyl betaine, ricinoleamidopropyl betaine*, sesamidopropyl betaine*, shea butteramidopropyl betaine, soyamidopropyl betaine, stearamidopropyl betaine*, tallowamidopropyl betaine*, undecyleneamidopropyl betaine, and wheat germamidopropyl betaine*.
Were ingredients in this group not in current use (identified with an *) to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.