Abstract
Occidiofungin, a glycolipopeptide obtained from the liquid culture of Burkholderia contaminans MS14, has been identified as a novel fungicide. The present study was designed to initially assess the in vitro toxicity in a rat hepatoma (H4IIE) cell line and acute toxicological effects of occidiofungin using a mouse model. In vitro toxicity was observed in all variables at 5 μmol/L. B6C3F1 mice were given single and repeat doses of occidiofungin up to 20 mg/kg. Key effects were a reduction in body and organ weights. However, no significant decrease in body weight was noted at a dose of 1 mg/kg, which is comparable to the dose level of other cyclic glycopeptide antifungal agents currently approved for human use. Microscopic examination of treated mice did not identify any signs of organ-specific toxicity at the dose levels tested.
Introduction
There has been a growing demand for antifungal agents recently due to the increase in life-threatening fungal infections, which are related to suppression of the immune system caused by medical therapies 1 or the advent of chronic immunosuppressive viral infections like HIV-1. 2 Four major classes of antifungal agents have been developed: polyene antifungals, azole antifungals, allylamine antifungals, and the echinocandins. The first 3 classes mainly target ergosterol or ergosterol synthesis, disrupting the integrity of fungal cell membrane, and the echinocandins target polysaccharide production in the cell wall. 3 –6 These existing antifungals have been widely used to control serious fungal infections. For instance, amphotericin B, a polyene antibiotic, has been used as a “gold standard” for antifungal therapy for decades. 7 However, none of these antifungal drugs satisfy medical needs completely due to problems in spectrum, potency, safety, or pharmacokinetic properties. Also, there is always concern with regard to the development of fungal strains resistant to current drugs, 8 leading to a constant need for the development of new antifungal drugs. Therefore, development of novel antifungals is important.
Occidiofungin is a compound purified from liquid culture of Burkholderia contaminans MS14. 5,9 –11 Structural analyses revealed that this compound is a cyclic glycolipopeptide composed of 8 amino acids. An 18-carbon fatty amino acid with a xylose sugar attached was identified in the compound. Biological activity assays demonstrated its potent antifungal properties against a broad spectrum of plant and animal fungal pathogens. 5 To date, the exact mechanism of activity of occidiofungin is still unknown. However, the strong inhibitory effect of occidiofungin on Pythium spp, which does not express ergosterol in the cell membrane and chitin in the cell wall, suggests that inhibition of ergosterol and chitin biosynthesis is probably not the major mechanism of occidiofungin activity. The inhibitory activity of occidiofungin on Fusarium spp and Cryptococcus neoformans, which are resistant to echinocandins, suggests a new mechanism may be involved. 5 Taken together, these results indicate that occidiofungin is distinctive from the 4 existing classes of antifungal drugs and that occidiofungin’s potential as a therapeutic agent warrants further investigation.
Before testing the efficacy of occidiofungin in an animal model or in humans, it is necessary to determine its toxicological profile. Substantial toxicity at doses expected to be required for antifungal efficacy would preclude further development of this compound as a therapeutic agent. The purpose of the present study is to evaluate the acute toxicity of occidiofungin.
Materials and Methods
Occidiofungin was purified as described previously. 10 Briefly, it was isolated from a bacterial culture supernatant by high-performance liquid chromatography (HPLC). The structure has been characterized 5 and is shown in Figure 1. Phosphate-buffered saline, methylcellulose, and Tween 80 were purchased from Sigma (Sigma-Aldrich, Saint Louis, Missouri).
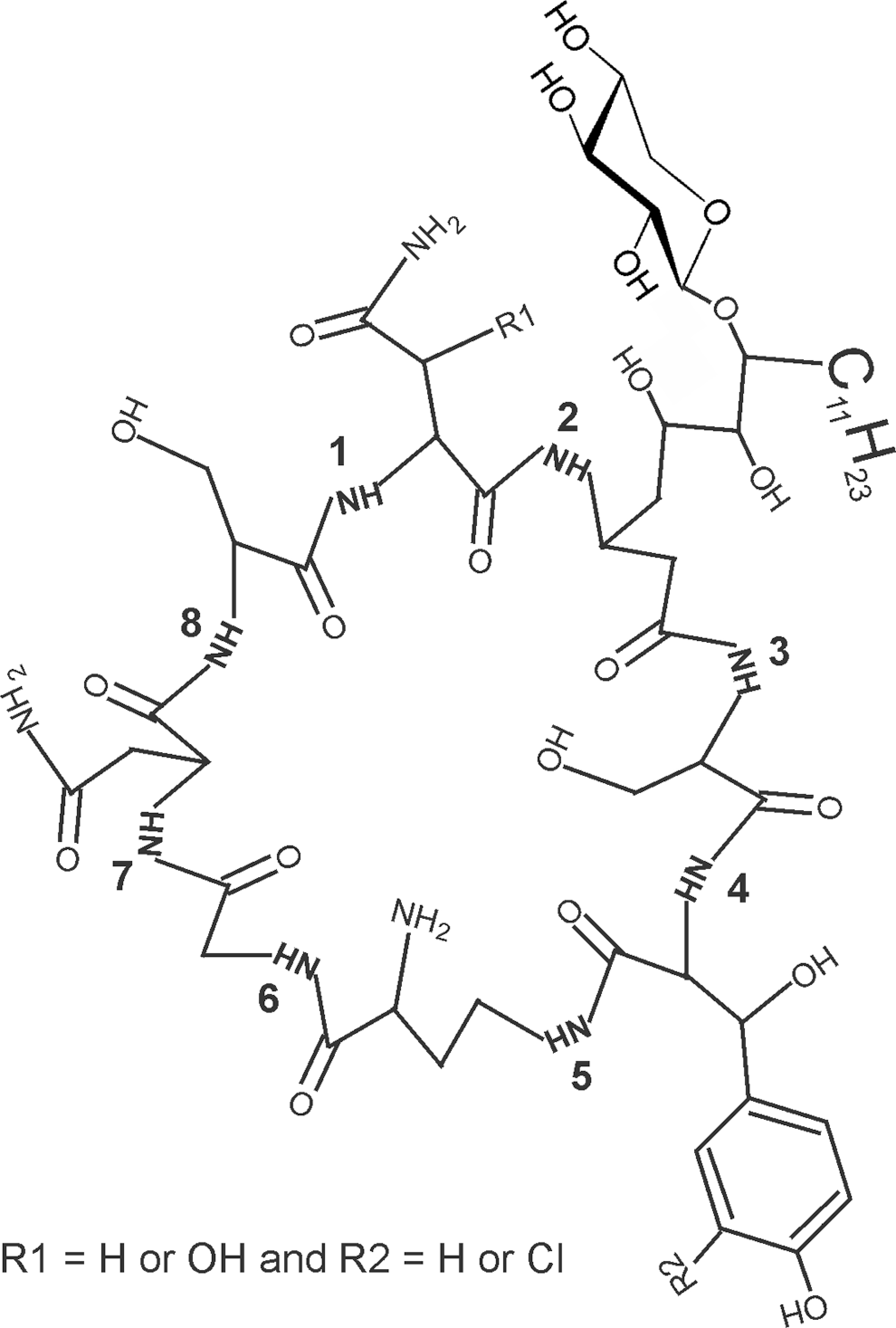
Chemical structure of occidiofungin. Occidiofungin was purified by high-performance liquid chromatography (HPLC) and the structure was deduced as described in a previous study. 5
In Vitro Toxicity Screening
Occidiofungin was prepared in DMSO and the appropriate amounts were then added to the medium containing 10% bovine serum and 10% calf serum at 37oC, as specified by Ceetox Inc (Kalamazoo, Michigan). The samples were evaluated using a light scattering technique and a Nepheloskan instrument (MTX Lab Systems, Inc, Vienna, Virginia). A reading that was greater than or equal to 3 times background was considered the limit of solubility. 12 Occidiofungin solubility limit was determined to be between 10 μmol/L and 50 μmol/L in the medium containing 20% serum. These concentrations are well above the concentrations required for the observed fungicidal activity (0.5-2 µg/mL). 5
An in vitro toxicity screen was performed. Flat bottom 96-well plates were seeded with 10 000 rat hepatoma (H4IIE) cells/well for 48 hours prior to dosing. 12,13 On the morning of the third day after seeding, the test compounds in medium were added to the plates at concentrations of 0, 0.001, 0.01, 0.1, 1, 5, or 10 µmol/L. After 24 hour exposure at 37oC in 5% CO2, the cells and cell supernatant were harvested for biochemical analysis. General cytotoxicity was evaluated by monitoring membrane integrity and mitochondrial function. All in vitro cytotoxicity assays were performed by Ceetox Inc, using a rat hepatoma (H4IIE) cell line following the procedures described below.
Intracellular ATP levels. 14
Cellular adenosine triphosphate (ATP) was determined with an assay based on a reaction between ATP +
Tetrazolium dye reduction, 3-[4,5-dimethylthiazol-2-yl] 2,5 diphenyltetrazolium bromide (MTT). 12,15,16
Detailed methodology is described by Berridge et al. 16 After the medium was removed from a plate for α-glutathione S-transferase (α-GST) analysis, the cells remaining in each well were evaluated for their ability to reduce soluble-MTT (yellow) to formazan-MTT (purple). This reaction is performed in the mitochondria, which must be functioning to perform this reduction. Therefore, formation of the formazan is an indicator of cellular metabolic activity and viability. An MTT stock solution was prepared just prior to use and warmed to 37oC in a water bath. Once the medium was removed from all wells, the MTT solution was added to each well and the plate was incubated at 37oC for 3 to 4 hours. Internal method development experiments have demonstrated that color development is linear over this time. After the 3- to 4-hour incubation, all medium was removed and the purple formazan product was extracted using anhydrous isopropanol. Sample absorbance was read at 570 nm and reference absorbance at 650 nm with a Packard Fusion plate reader.
Membrane leakage (α-glutathione S-transferase).12,17
The presence of α-GST was measured in the culture medium using activity enzyme-linked immunosorbent assay (ELISA) for α-GST. α-GST is released into the media when membrane integrity is disrupted and is used to determine membrane damage. Released α-GST was measured using a kit from Argutus Medical (kit BIO64RT, formerly Biotrin Kit #458604). At the end of the exposure period, the medium covering the cells in each well was removed and placed into new 96-well plates with appropriate labeling and then analyzed immediately. Absorbance (α-GST) values were measured with a Packard SpectraCount reader at 450 nm and reference absorbance at 650 nm.
P-glycoprotein binding using the MTT assay.18–20
The H4IIE cells possess high levels of P-glycoprotein (PgP) protein in the cytoplasmic membrane. As a result, they are often used when compounds are evaluated for their protein-binding potential. 21 Cells incubated with and without cyclosporin A (CSA, a PgP inhibitor) were compared for difference in toxicity, as determined by the MTT assay. This method was performed as described in detail previously. 18 –20 It is assumed that compounds with increased toxicity (decreased MTT reduction) in the presence of CSA have a high probability of binding to PgP protein, since the compound is not being sequestered by PgP protein in the membrane. If there is no difference in toxicity, then it is assumed that the compound does not bind to PgP protein.
Mice
Female C57BI/6 × C3H F1 (B6C3F1) mice at age of 6 to 8 weeks were used. These mice were purchased from National Cancer Institute’s Animal Program and allowed to acclimate at least 2 weeks after arrival. They were housed on a 12-hour light–dark cycle in a temperature- and humidity-controlled animal facility that is accredited by the American Association for Accreditation of Laboratory Animal Care. Animal care and use was in accord with National Institutes of Health (NIH) Guidelines and Mississippi State University regulations. Protocols used for the mice models were done in accordance with the methods reported by &Luster et al. 22
Single-Dose Toxicity Study
Solubility of occidiofungin in aqueous buffers is relatively low. To avoid using vehicles such as DMSO that can affect a number of cellular functions, mice (5 mice per group) were given occidiofungin using vehicles that have been used extensively in nonclinical drug testing with little or no indication of meaningful effects, which included phosphate-buffered saline (PBS) or PBS with 0.5% methylcellulose and 0.1% Tween 80. Visual inspection following addition of vehicle to the purified dried drug indicated that a portion of the drug went into solution but the remainder was in the form of a flocculent suspension. Use of a sonicator decreased the size of the suspended particles but did not fully dissolve them, so minimal sonication (10 seconds at 20% power) was used in preparing samples for administering to animals. The suspensions prepared in PBS remained uniformly distributed in the syringe for several minutes and to ensure uniform distribution the syringe was actively rotated along the long and cross-sectional axes constantly between injections. Methylcellulose was included to facilitate the preparation of a homogenous suspension. We have previously reported that the methylcellulose and Tween 80 vehicle decreases the rate of absorption of hydrophobic drugs and decreases peak blood levels. 23 It is not known whether this effect occurs with occidiofungin, but if it did, toxicity associated with a high peak blood concentration might be diminished. Visual inspection indicated that the viscosity of the methylcellulose solution was also effective in maintaining a uniform suspension of the drug. Intraperitoneal (ip) and subcutaneous routes were used in this study, because these routes allow best opportunity for biological availability. Intravenous administration will be evaluated in the future when we have identified an appropriate vehicle to dissolve the material or to prepare a uniform suspension with microscopic particle size. The dose range used in this study was based on reported values for effective doses of structurally related classes of antifungal drugs, the echinocandins (1-2 mg/kg). Doses up to 10 times the expected effective dose were used so that the data reported here can be used to determine the therapeutic index, when efficacy data are obtained.
For most experiments, occidiofungin was administered by ip injection in a single dose at 1, 5, 10, or 20 mg/kg in PBS of body weight. The compound was also administered in 1 dose at 10 mg/kg in sterilized 0.5% methylcellulose (constituted with 0.1% Tween-80 in PBS) by ip or subcutaneous injection. The excipient control in each experiment matched the vehicle. Control groups received sterile PBS or 0.5% methylcellulose with 0.1% Tween-80 in PBS. Body weight and clinical signs (movement, posture, skin lesions, appearance of fur indicating normal grooming, and behaviors) were recorded daily for 5 days. Necropsies were performed on study day 1 or study day 5 following the administration of occidiofungin.
Measurement of Toxicological Parameters
Blood and tissue samples were collected from animals dosed at 10 mg/kg with occidiofungin in 0.5% methylcellulose. One group of samples was collected on study day 1 following a single dose by ip injection. Another group was collected after 5-day observation with the same single dose through subcutaneous injection. Mice were anesthetized with isofluorane. Blood was then taken from the retroorbital plexus for serum biochemistry assays (alkaline phosphatase, alanine aminotransferase [ALT], aspartate aminotransferase[AST], gamma-glutamyl transferase, creatine kinase, blood urea nitrogen, creatinine) and hematology (white blood cell count and white blood cell differentiation). Body weight was measured immediately after mice were fully anesthetized and organs (spleen, thymus, brain, kidney, lung, and liver) were weighed and fixed in 10% neutral buffered formalin. Histological examination was performed on a portion of each organ by using routine paraffin-embedding technique and staining with hematoxylin and eosin (H & E). 24 All the sections were examined under light microscopy for pathological changes by a coauthor of this article, who is a board-certified veterinary pathologist (J.C.).
Repeat-Dose Toxicity Study
Occidiofungin was evaluated in a 5-day toxicity study by using 5 mice per group. Occidiofungin was dissolved in sterile PBS at dose of 2 mg/kg and administrated by ip injection daily for 5 consecutive days. The control group received sterile PBS. Body weight and clinical signs were recorded daily. Necropsies were performed on day 5 following the first administration of occidiofungin. Clinical chemistry, hematology (the same parameters as in the single-dose study), and histology (the same organs as in the single-dose study, except thymus) were evaluated. Histological sections were reviewed in the same manner as described above.
Body weight, body weight change, organ weight, serum chemistry, and hematology data were analyzed by T test or 2-way analysis of variance (ANOVA) followed by Bonferroni posttest using Prism GraphPad software (San Diego, California). All the analyses were 2-sided, with P < .05 considered statistically significant.
Results
In Vitro Toxicology Screening
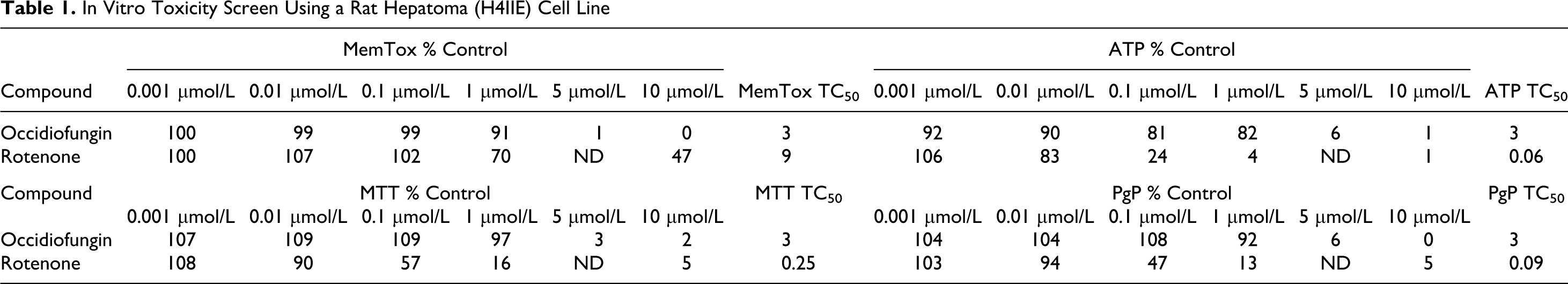
Changes were observed in all cellular toxicity end points with occidiofungin at 5 μmol/L, with a predicted TC50 value of 3 μmol/L (Table 1). The cells remain viable, until a critical concentration threshold of occidiofungin was reached that causes a loss of membrane integrity over the 24-hour period of exposure. This is unlike the control used (rotenone) that is toxic due to inhibition of mitochondrial electron transport, where a dose-dependent response of toxicity was observed. Toxicity did not change when expression of PgP was blocked, suggesting that occidiofungin does not bind to PgP. Given that occidiofungin in vitro toxicity profile was not much different from the profile of other clinically used peptide drugs, further investigation of its toxicity in an animal model was warranted.
In Vitro Toxicity Screen Using a Rat Hepatoma (H4IIE) Cell Line
In Vivo Acute Toxicology
Behavior signs
A consistent behavioral response was observed, in which there was an irritation reaction in mice treated with occidiofungin through ip injection at 10 and 20 mg/kg in PBS. The mice extended their front and rear paws and stretched repeatedly. This behavior occurred intermittently for about 30 minutes after dosing and not associated with more typical rodent behaviors related to severe pain (eg, writhing, vocalization, or lack of spontaneous locomotion). Stretching was not observed in mice treated with occidiofungin through ip injection at 1 and 5 mg/kg in PBS. In addition, a 10-mg/kg dose administered ip in methlycellulose vehicle did not elicit the stretching behavior. Slight lethargy and ruffled fur was observed in the single-dose experiments on the first and second day after a 10 mg/kg or 20 mg/kg treatment in PBS. An inflammatory lesion was found at the location of subcutaneous injection in few mice as well. These issues will need to be investigated further but may be alleviated by altering the dosing route or vehicle. For example, the mice treated subcutaneously with occidiofungin in a methylcellulose/Tween 80 vehicle did not exhibit the stereotypical stretching behavior as noted in the mice which received the same dose ip in PBS. No other behavioral signs were observed.
Body weight
Results shown in Figure 2 indicate the body weight versus time at different doses. The body weight differences ranged from 2% to 12% on the first day after treatment at various doses. At the end of the studies, it was clear that higher dose induced more body weight loss, and this change was dose responsive. However, this effect tended to diminish after the dosing ended, with a consequent regain of body weight during the following days. Daily dosing at 2 mg/kg for 5 days had a similar effect on day 5 as a single 5 mg/kg ip dose.
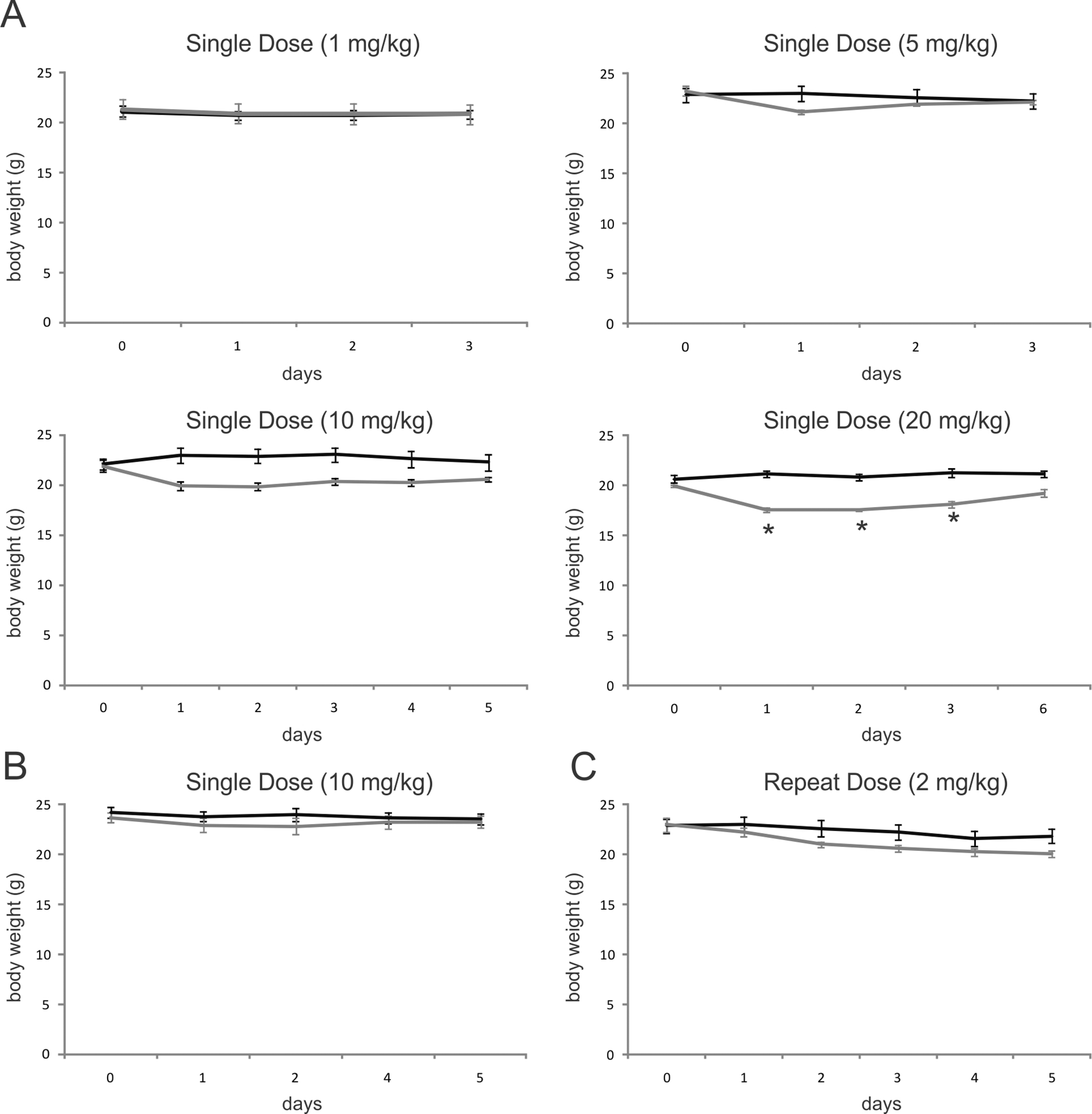
Mean body weights of mice following exposure to occidiofungin. Occidiofungin was reconstituted (A) in PBS for ip administration at 1 mg/kg, 5 mg/kg, 10 mg/kg, and 20 mg/kg for single-dose study; (B) in 0.5% methylcellulose in PBS for subcutaneous administration at 10 mg/kg for single-dose study, (C) in PBS at 2 mg/kg for repeat-dose study. Control animal weights are shown in black and the treated animals are shown in gray. *Signifies significant differences between treated and control group at P < .05.ip indicates intraperitoneal; PBS, phosphate-buffered saline.
Hematology and clinical chemistry
Results shown in Table 2 (panel A) indicate a decrease in absolute total white blood cell count in mice 1 day after ip administration of occidiofungin at 10 mg/kg. There were no statistical differences in hematological or serum chemistry values 5 days after subcutaneous administration of occidiofungin at 10 mg/kg (Table 2 panel B). In the repeat-dose study, white blood cell differential counts showed an increase in neutrophils and decrease in lymphocytes following a 5-day ip administration of occidiofungin at 2 mg/kg (Table 3). Although there were some statistically significant differences in serum clinical chemistry parameters, no consistent dose–response effects were noted, which suggests that the significant effects represented normal biological variation or experimental effects are not directly related to the action of the test compound.
Hematology and Serum Chemistry in Single-dose Experiments
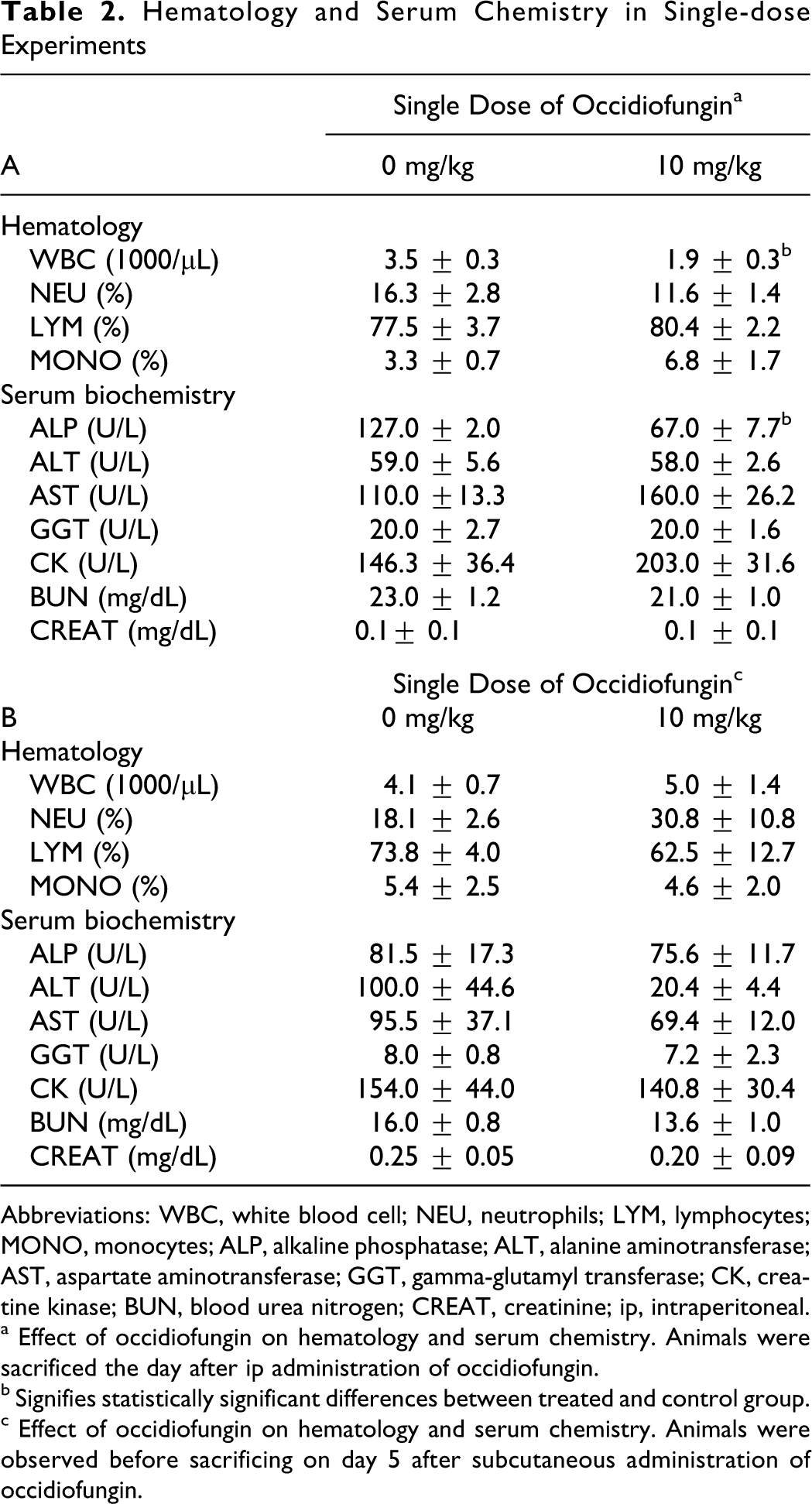
Abbreviations: WBC, white blood cell; NEU, neutrophils; LYM, lymphocytes; MONO, monocytes; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transferase; CK, creatine kinase; BUN, blood urea nitrogen; CREAT, creatinine; ip, intraperitoneal.
a Effect of occidiofungin on hematology and serum chemistry. Animals were sacrificed the day after ip administration of occidiofungin.
b Signifies statistically significant differences between treated and control group.
c Effect of occidiofungin on hematology and serum chemistry. Animals were observed before sacrificing on day 5 after subcutaneous administration of occidiofungin.
Hematology and Serum Chemistry in 5-Day Repeat-Dose Experiment a
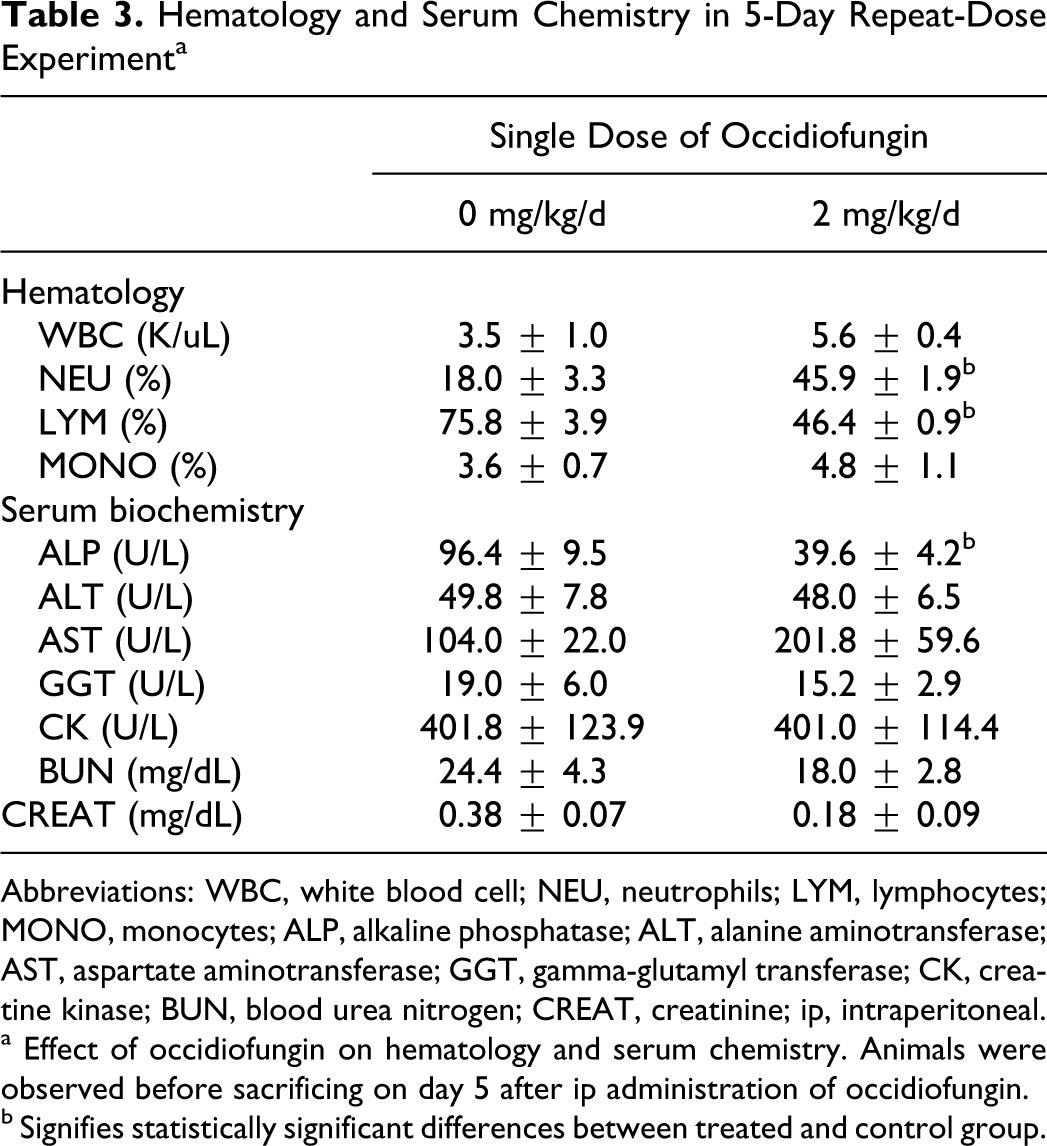
Abbreviations: WBC, white blood cell; NEU, neutrophils; LYM, lymphocytes; MONO, monocytes; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transferase; CK, creatine kinase; BUN, blood urea nitrogen; CREAT, creatinine; ip, intraperitoneal.
a Effect of occidiofungin on hematology and serum chemistry. Animals were observed before sacrificing on day 5 after ip administration of occidiofungin.
b Signifies statistically significant differences between treated and control group.
Organ weights
Tables 4 and 5 show the group means of absolute organ weights and organ/body weight values. In the single ip dose of 10 mg/kg experiment, an increase in absolute weight of the liver and liver/body weight values were seen 1 day after dosing (Table 4 panel A). A small but statistically significant decrease in absolute organ weights and organ/body weight values for lung and thymus were noted 5 days after a single subcutaneous dose of 10 mg/kg (Table 4 panel B). Following 5 ip doses of 2 mg/kg per d, only the thymus was found to decrease in absolute organ weights and organ/body percentage weights (Table 5). There was an increase in organ/body weight percentage for the brain and kidney but not an absolute weight gain. The reduction in body weight in this group likely led to increases in relative organ weight values. The consistent finding was a decrease in absolute thymus weight, which may indicate a nonspecific stress response to the compound. This issue is considered further in subsequent sections. Hematology results also were consistent with a stress response (details are provided in the hematology and clinical chemistry section).
Weight Measurements in Single-Dose Experiments
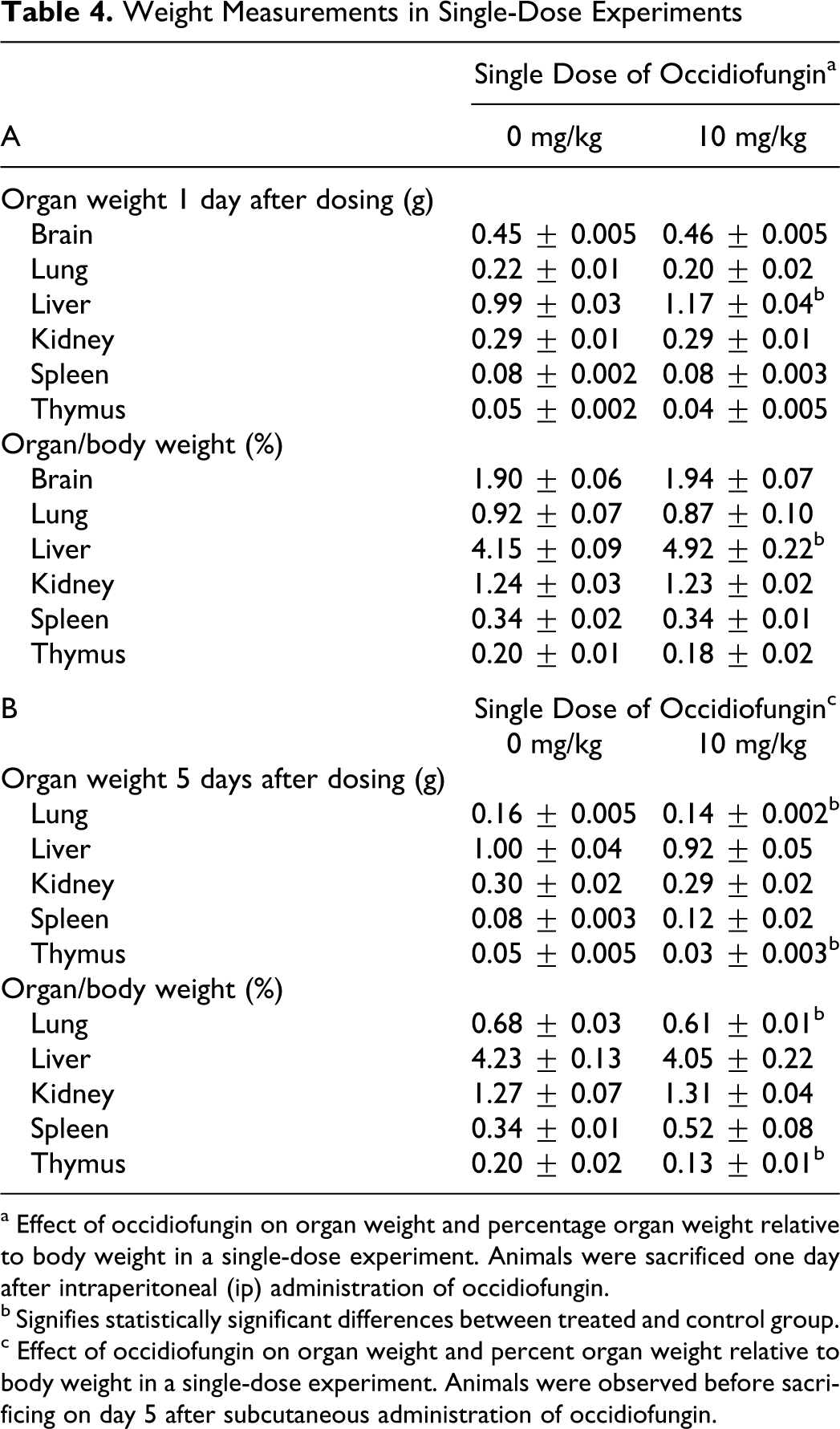
a Effect of occidiofungin on organ weight and percentage organ weight relative to body weight in a single-dose experiment. Animals were sacrificed one day after intraperitoneal (ip) administration of occidiofungin.
b Signifies statistically significant differences between treated and control group.
c Effect of occidiofungin on organ weight and percent organ weight relative to body weight in a single-dose experiment. Animals were observed before sacrificing on day 5 after subcutaneous administration of occidiofungin.
Weight Measurements in a 5-day Dosing Experiment a
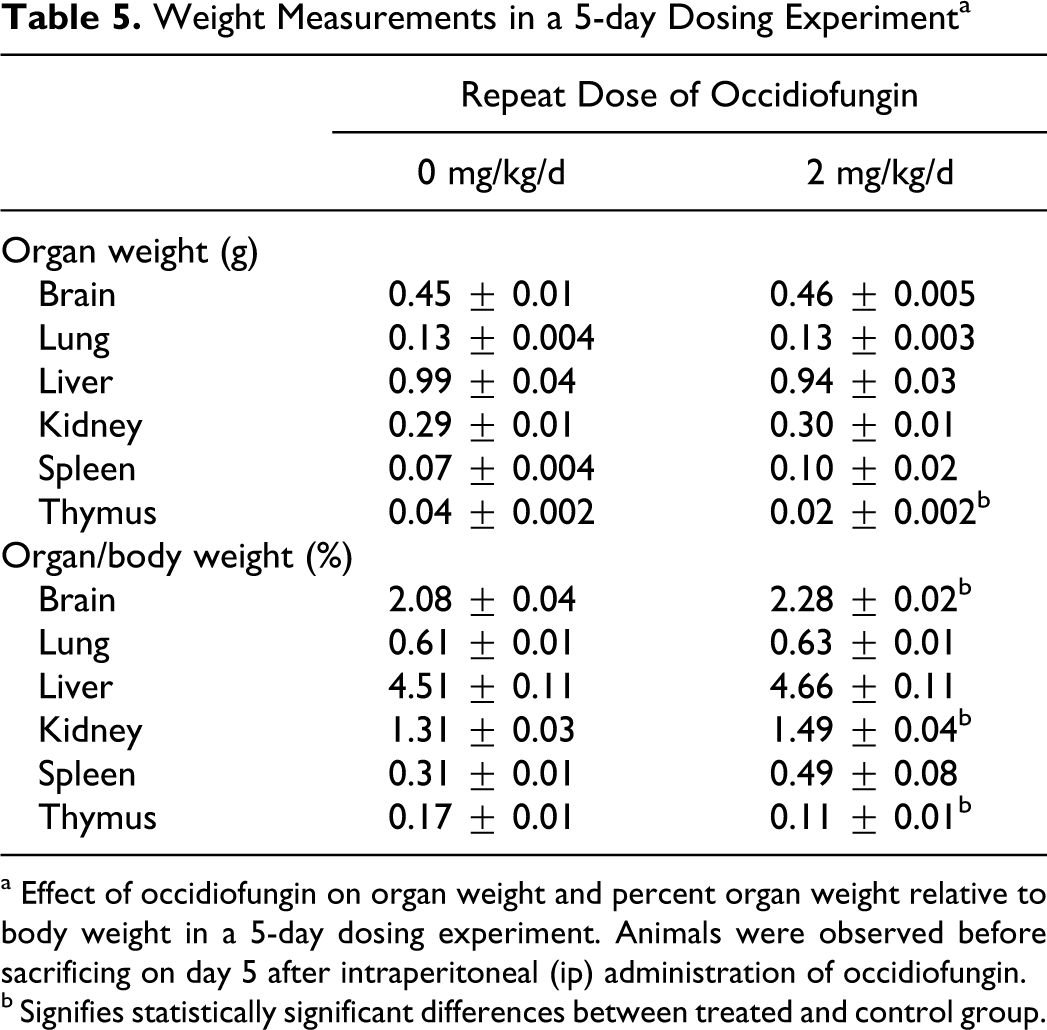
a Effect of occidiofungin on organ weight and percent organ weight relative to body weight in a 5-day dosing experiment. Animals were observed before sacrificing on day 5 after intraperitoneal (ip) administration of occidiofungin.
b Signifies statistically significant differences between treated and control group.
Macroscopic and microscopic findings
Generally, no macroscopic findings were observed by histological examination (Figure 3). No observable differences were present in the microscopic cell morphology or macroscopic tissue morphology of brain, liver, lung, thymus, or kidney. One of the most interesting findings was a decrease in activated thymocytes in the medulla in the single-dose toxicity study, which was indicated by darker staining in this regions reflecting the lower cytoplasmic volume associated with nonactivated lymphocytes as compared to activated lymphocytes (Figure 3C). This result in conjunction with decreased thymus weight or increased neutrophil percentages (described in the previous section) suggests that occidiofungin causes a nonspecific stress response. A nonspecific stress response may cause a nonspecific activation of the hypothalamic–pituitary–adrenal axis, leading to a sufficient increase in circulating glucocorticoids to cause apoptosis in developing thymocytes. 25 In summary, there was no clear evidence for organ-specific histological effects of occidiofungin.
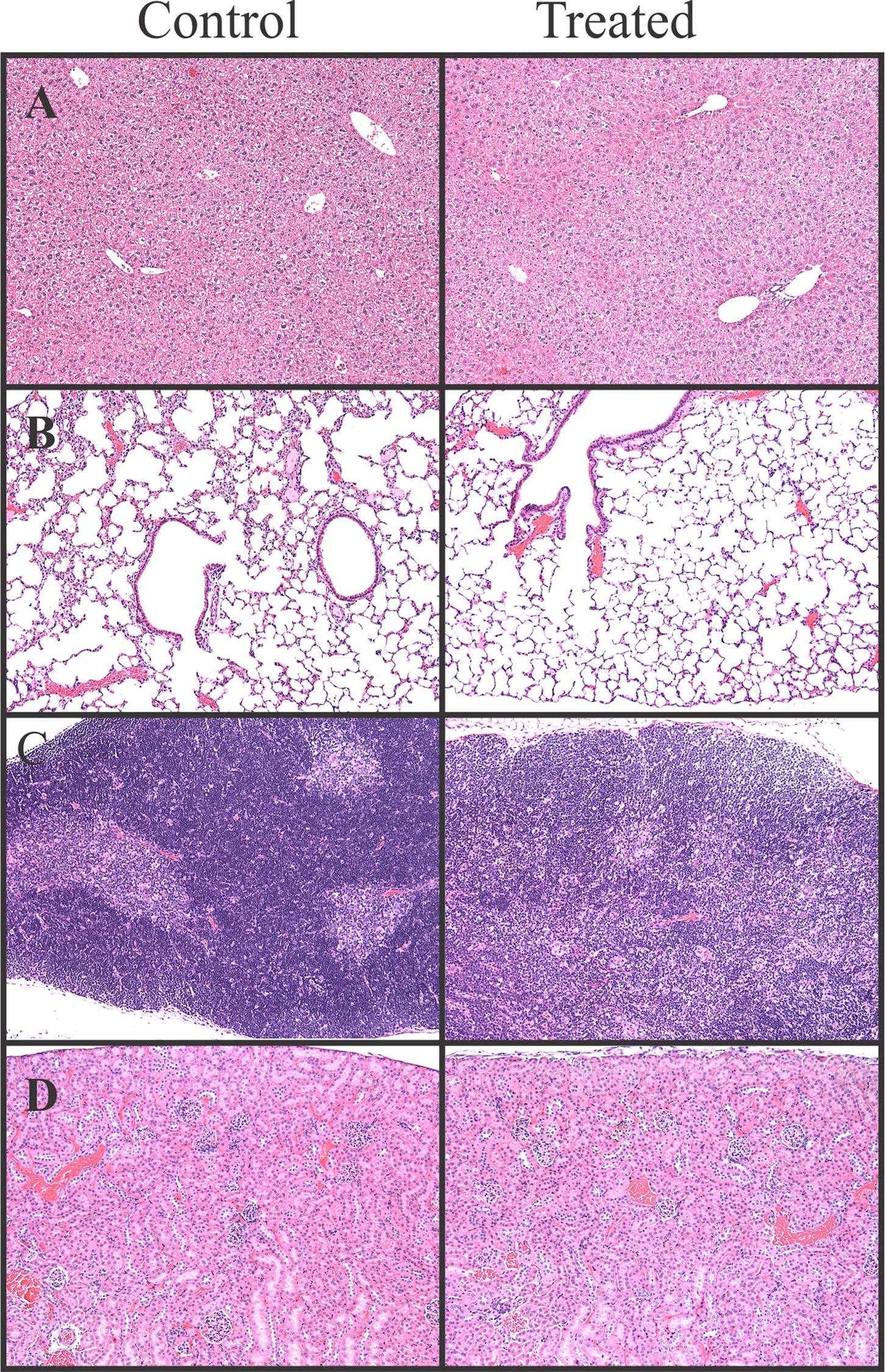
Representative histological sections of control and treated mice. (A) Liver from the 5-day 2 mg/kg repeat-dose experiment (×400 magnification). (B) Lung from 5 days after the 10 mg/kg subcutaneous dose (×100 magnification), (C) Thymus from 5 days after the 10 mg/kg subcutaneous dose (×100 magnification), and (D) Kidney from the 5-day 2 mg/kg repeat-dose experiment (×100 magnification). There were no morphological differences between control and treated mice in these experiments, except for the decreased cellularlity in the thymic medulla.
Discussion
Occidiofungin exhibits antifungal activities for a broad range of fungal pathogens and has a potential as a new type of antifungal agent for both agriculture and clinical therapies. 5 A candidate compound for these goals should be of equal or greater safety than the currently marketed compounds. To date, no nonclinical safety assessment of occidiofungin has been reported. The present study evaluated in vitro toxicity in a rat hepatoma (H4IIE) cell line and acute toxicity in mice administered occidiofungin through ip and subcutaneous routes.
In the in vitro study, a 24-hour exposure of 5 μmol/L occidiofungin resulted in a rapid decline in viable rat hepatoma cells, while lower concentrations had no observed toxicity (Table 1). The toxicity profile is not much different than what has been described for the natural products used to synthesize micafungin, caspofungin, and anidulafungin, which are used in the clinic for treating fungal infections, 26 –28 as well as what has been described for the natural product daptomycin which is used in the clinic for treating gram-positive infections. 29 –32 All of these lipopeptides are chemically modified to reduce toxicity, while occidiofungin is a natural, unmodified product. Although the results reported here are consistent with an effect on the cell membrane as a possible cause of cellular toxicity in mammalian cells, this is probably not the mechanism of toxicity for fungi. 5
In the animal studies, body weight decreases occurred in mice treated with some but not all dose levels of occidiofungin. However, this body weight loss was transient and recovered rapidly once the administration stopped. Body weight loss was not significant following administration of occidiofungin at 1 mg/kg, a dose level similar to the dosage used in humans for echinocandin antifungal drugs. 33 Dose levels up to 10 mg/kg did not consistently cause statistically significant decreases in body weight. Food consumption is related to body weight change, and change in food intake was seen with other antifungal drugs. 34,35 It is possible that occidiofungin acts similarly. Organ injury indicated by changes in clinical chemistry and microscopic evaluation did not seem severe enough to cause weight loss, so alteration of appetite may have occurred, which will be evaluated in future studies.
Specific effects on organs induced by occidiofungin were not detected by microscopic examination, although changes in either absolute or relative weights of several organs were found. The reason for these changes is not clear. An increase in liver weight without histological changes or clinical chemistry changes was only seen in mice after 1 day treatment at 10 mg/kg. This may suggest an adaptive response due to minor liver injury. However, clinical chemistry parameters that indicate liver injury (eg, ALT and AST) were not significantly elevated, even after 5 days of dosing. Overall, these results suggest a transient and mild effect of occidiofungin on the liver. Several reports indicate that oral antifungals can cause mild hepatotoxicity, 34,36,37 an indication of minor changes in the liver which seem to decrease following multiple doses, would not necessarily preclude the clinical use of occidiofungin. However, it is clear that further evaluation using different administration methods and dosing for longer periods of time will be needed to determine the extent of side-effects on liver function.
The differential WBC count in hematological samples from mice given multiple doses of occidiofungin at 2 mg/kg showed a tendency of augmented neutrophil and reduced lymphocyte percentages. This pattern along with decreased thymus weight is a typical sign of nonspecific stress responses. 38 –41 Harmonized international guidelines for preclinical testing indicate that stress-mediated immunological changes in rodents are not generally the cause for concern with regard to future assessment of the drug in humans. 42
The potency of antifungal activity of occidiofungin seems similar to or better than the echinocandin antifungals, which are also circularized lipopeptides. 5,9 Typical dosages of the echinocandin antifungals when used therapeutically in humans are 1 to 2 mg/kg per d, which yields peak blood levels of ~10 µg/mL, a concentration higher than the lethal concentration for most pathogenic fungi. 33 Results from the present study indicate that a dose level of 2 mg/kg is not associated with specific organ toxicity, although a body weight decrement was observed. A dose level of 1 mg/kg did not cause a significant body weight decrement or distress in the treated mice. Furthermore, the observed toxicity at higher doses may be mitigated by altering the route of administration.
It is important to point out that dose scaling for many drugs is more effective when dosages are expressed as mg/m2 of body surface area rather than mg/kg of body weight. 43 Accordingly, a dose level of 2 mg/kg in mice would yield similar biological effects (including toxicity) as a dosage of 0.17 mg/kg in humans. Echinocandins with similar antifungal activity in vitro as occidiofungin exhibit efficacy in antifungal therapy in mice at dose levels below 1 mg/kg, 44 a dose level at which occidiofungin did not cause a body weight decrease. It should be noted that occidiofungin is an unaltered natural product, whereas the clinically used echinocandins are semisynthetic molecules modified to decrease toxicity. 6 Assessment of the efficacy of occidiofungin and possible derivatives of this compound in animal models is clearly needed and is being planned.
Recently, antifungal peptides have gained attention due to their advantages in potential multifunctional mechanisms of action, less possibility to raise resistance and acceptable toxicity profiles. 45,46 Many antifungal peptides found abundant in plants and animals have been described as “nature’s antibiotics,” which play roles in innate immune response. 47 With the development of protein engineering and solid-phase synthesis, therapeutic application of antifungal peptides in the clinic becomes promising. However, many problems potentially induced by these peptides have to be overcome before clinical development, such as toxicity to eukaryotic cells and development of allergies. 48 Generally, 4 main targets are examined for the assessment of oral antifungal safety: the liver, the endocrine system, serum cholesterol, and the developing embryo. 49 As a demonstrated antifungal candidate, occidiofungin exhibits a broad antimycological spectrum and relatively low acute toxicity in the previous and present studies. When it is considered that occidiofungin is a raw natural product and that the toxicity of related products has been decreased by chemical modification, it would seem worthwhile to begin efforts to produce chemically modified forms and to continue the toxicological assessment before potential clinical use, such as chronic toxicity evaluation to determine maximum tolerated dose (MTD), which is defined as the dose that causes approximately a 10% decrease in body weight 50 and is part of the profile in the nonclinical safety assessment. 42 In any case, the results presented here did not identify organ-specific toxicities that would preclude the use of this compound, particularly if 1 mg/kg proves to be an efficacious dose in an animal model.
In summary, changes on weight of body and organs induced by occidiofungin administration at the doses present here is not associated with any negative gross or microscopic findings. Hematology and serum biochemistry tests reveal that occidiofungin does not significantly alter the functions of organs. Consequently, on the basis of above studies, occidiofungin does not cause observable adverse effect at doses evaluated in this study. It is conceivable that the observed weight changes can be mitigated if the weight loss is due to changes in feeding. Given that occidiofungin is an unaltered natural product and has a relatively minor toxicological effect in vivo, further investigational studies to assess the in vivo activity of the compounds are merited. The work presented in this study helps lay the foundation for these investigations.
Footnotes
Jim Cooley and Leif Smith contributed equally to this paper.
Acknowledgment
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Funding for this work was provided by the College of Veterinary Medicine, Mississippi State University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
