Abstract
Alkyl acetates, as well as acetic acid and acetate salts, are widely used cosmetic ingredients, with a wide range of functions as fragrances, solvents, or skin-conditioning agents, depending on the specific ingredient. Available data on alkyl acetates, and acetic acid and the alcohol to which they could be metabolized, were considered adequate to support the safety of the entire group in the present practices of use and concentration in cosmetics.
Keywords
Introduction
The CIR Expert Panel reviewed the available safety information that pertains to methyl acetate, 10 alkyl acetates, acetic acid, 5 acetate salts, and 2 corresponding esterase metabolites, as used in cosmetics. These ingredients include:
methyl acetate
propyl acetate
isopropyl acetate
t-butyl acetate
isobutyl acetate
butoxyethyl acetate
nonyl acetate
myristyl acetate
cetyl acetate
stearyl acetate
isostearyl acetate
acetic acid
sodium acetate
potassium acetate
magnesium acetate
calcium acetate
zinc acetate
propyl alcohol and
isopropyl alcohol.
The CIR Expert Panel previously has reviewed the related ingredients: ethyl acetate, 1 butyl acetate, 1 ethyl alcohol (alcohol denat), 2 and butyl alcohol. 3
The Panel also has reviewed the safety of methyl alcohol (methanol), 4 t-butyl alcohol, 5 butoxyethanol (butoxyethyl alcohol), 6 myristyl alcohol, 7 cetyl alcohol, 7 stearyl alcohol, 8 and isostearyl alcohol. 7
Because alkyl acetate ingredients are esters of acetic acid and the corresponding alcohols and can be metabolized, via hydrolysis by esterases present in skin, back to the parent alcohol and acetic acid, the Panel considered that these previous safety assessments were relevant.
Chemistry
Definition and Structure
The alkyl acetate ingredients are esters of acetic acid and the corresponding alcohol, with the shorter chain alkyl acetates (methyl, propyl, isopropyl, t-butyl, isobutyl and butoxyethyl; molecular weight [MW] range 74-160 g/mol) functioning in cosmetics as fragrance ingredients and solvents, and the longer chain alkyl acetates (nonyl, myristyl, cetyl, stearyl and isostearyl; MW range 186-312) functioning in cosmetics as skin-conditioning agents.
The CAS registry numbers, definitions, functions, and CIR review history of the ingredients under consideration are presented in Tables 1 and 2and the structures are presented in Figure 1. The technical names for these ingredients are listed in Table 3. A map of how the structures and metabolic pathways of these ingredients are related is presented in Figure 2.
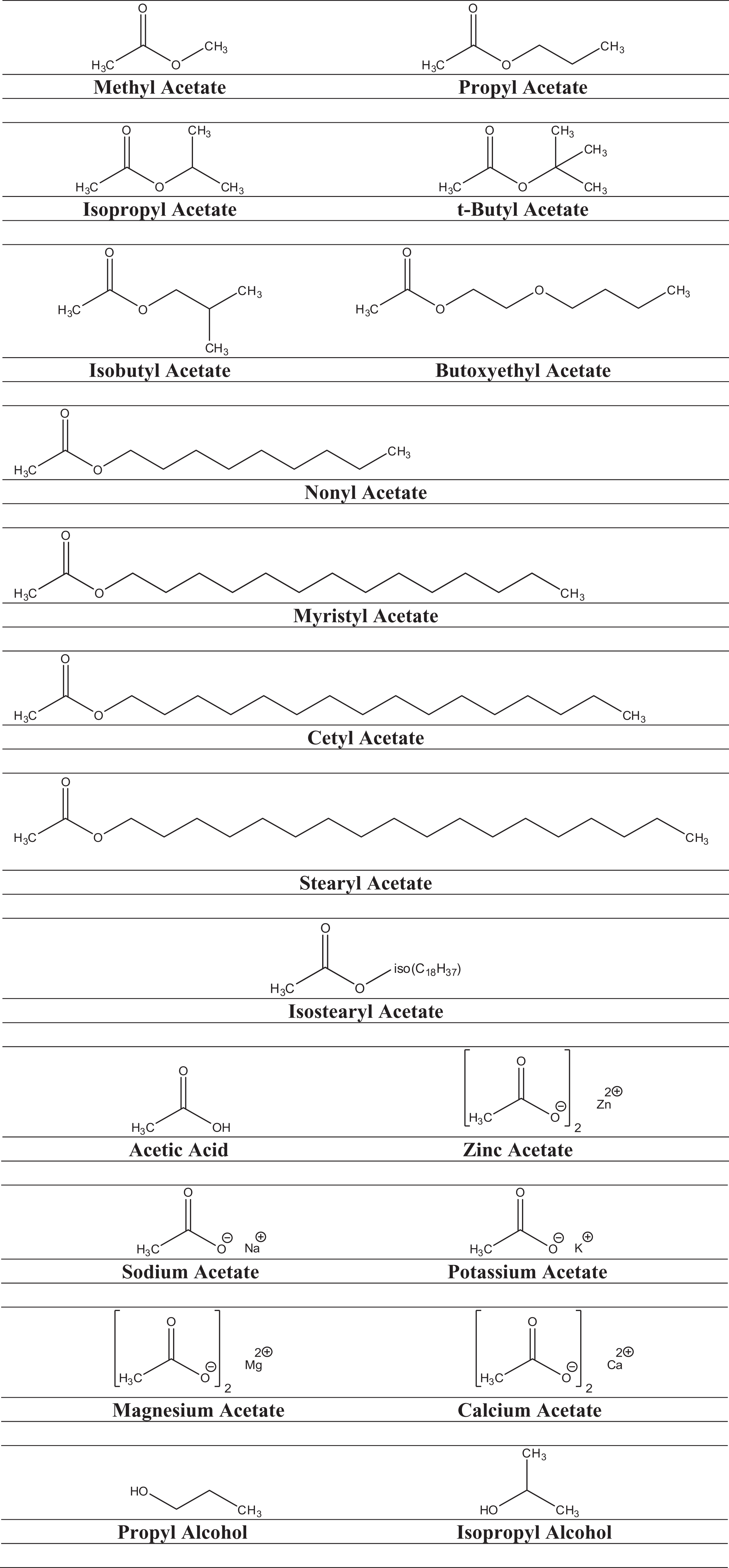
Structures of the ingredients in this assessment.
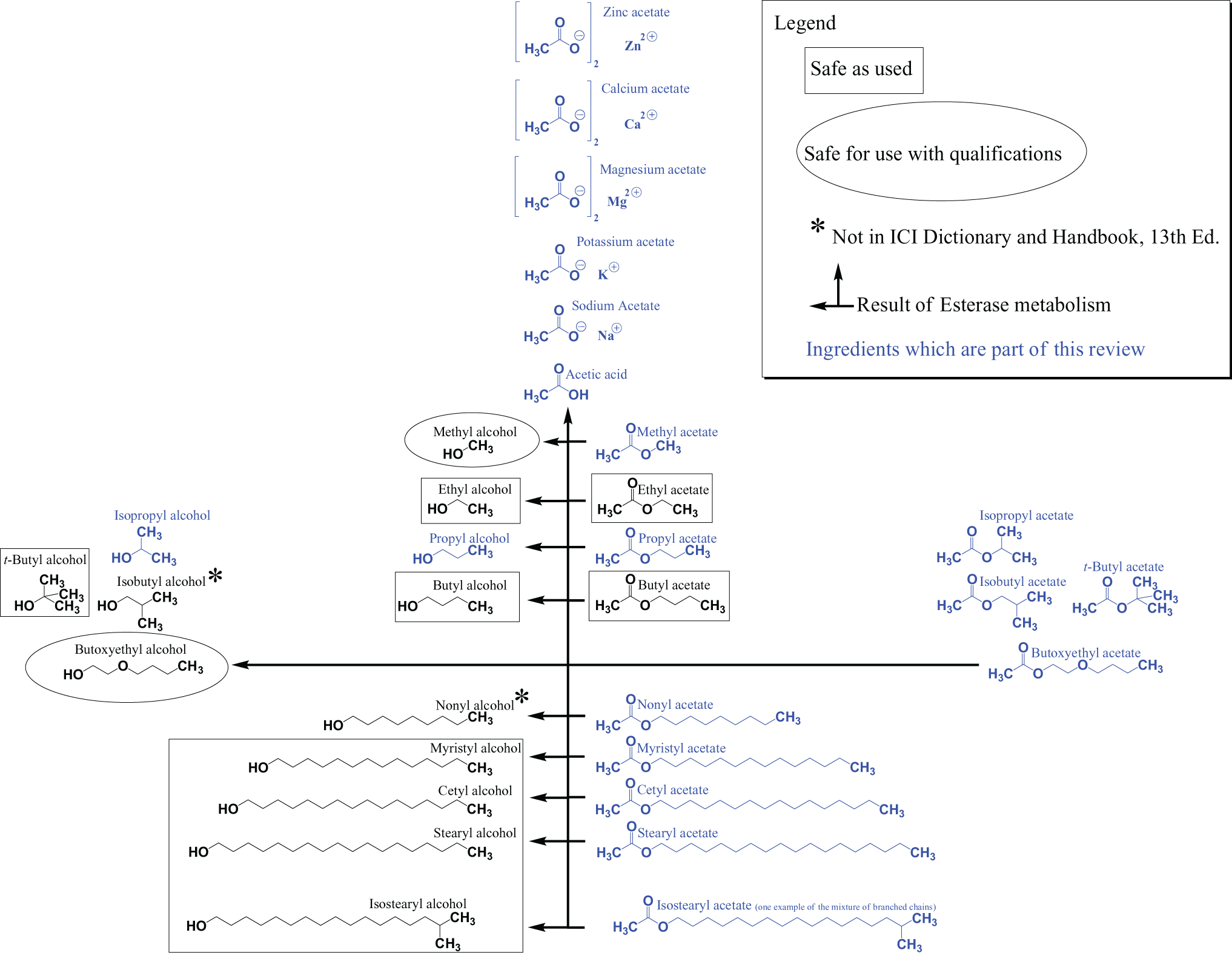
Map of the ingredients in this assessment and the associated esterase metabolites.
The Name, CAS Registry Number, Definition, Function, and CIR Review History of Metabolites of Alkyl Acetate Ingredients in This Assessment, Which are also Cosmetic Ingredients 8

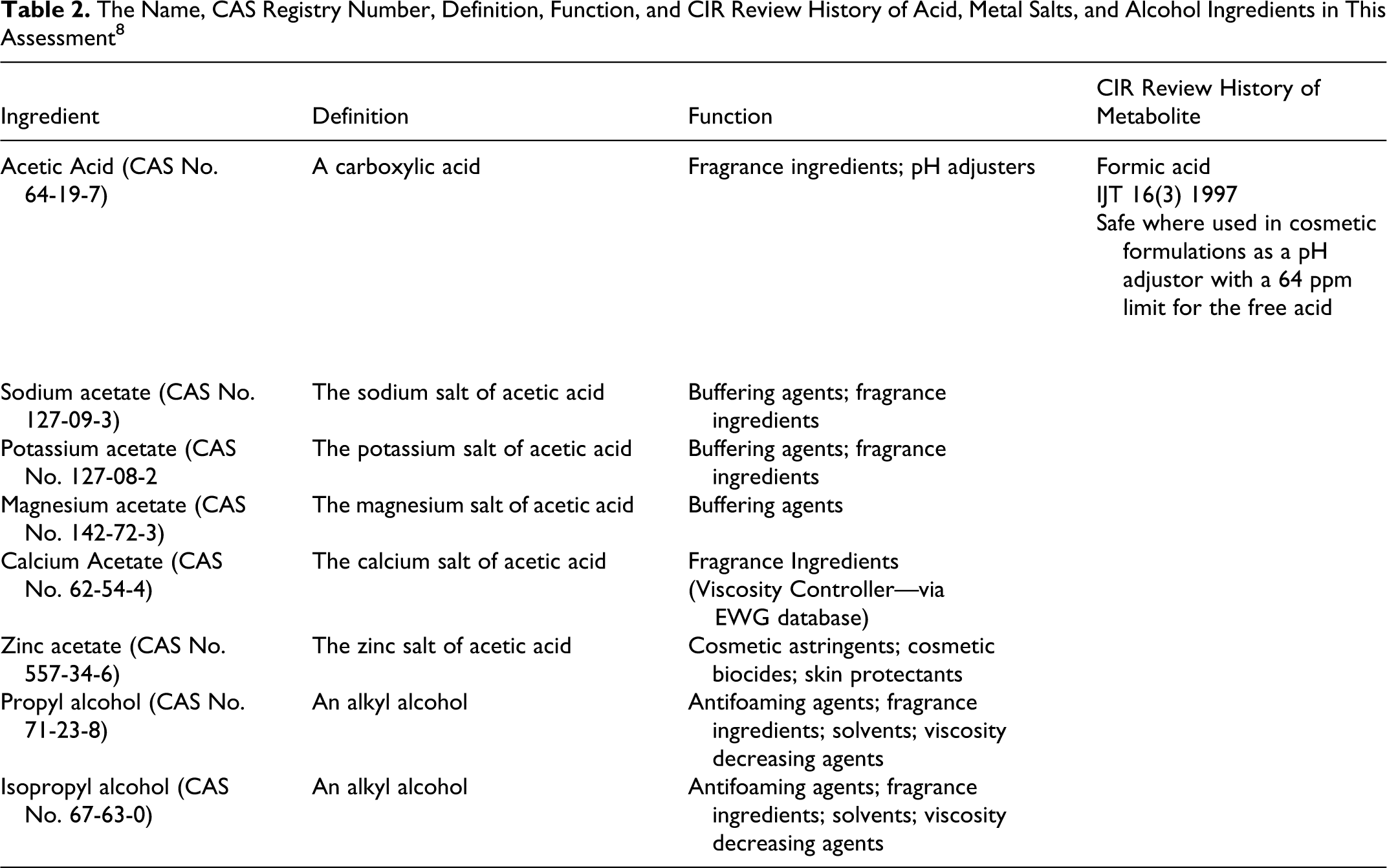
The Name, CAS Registry Number, Definition, Function, and CIR Review History of Acid, Metal Salts, and Alcohol Ingredients in This Assessment 8
Physical and Chemical Properties
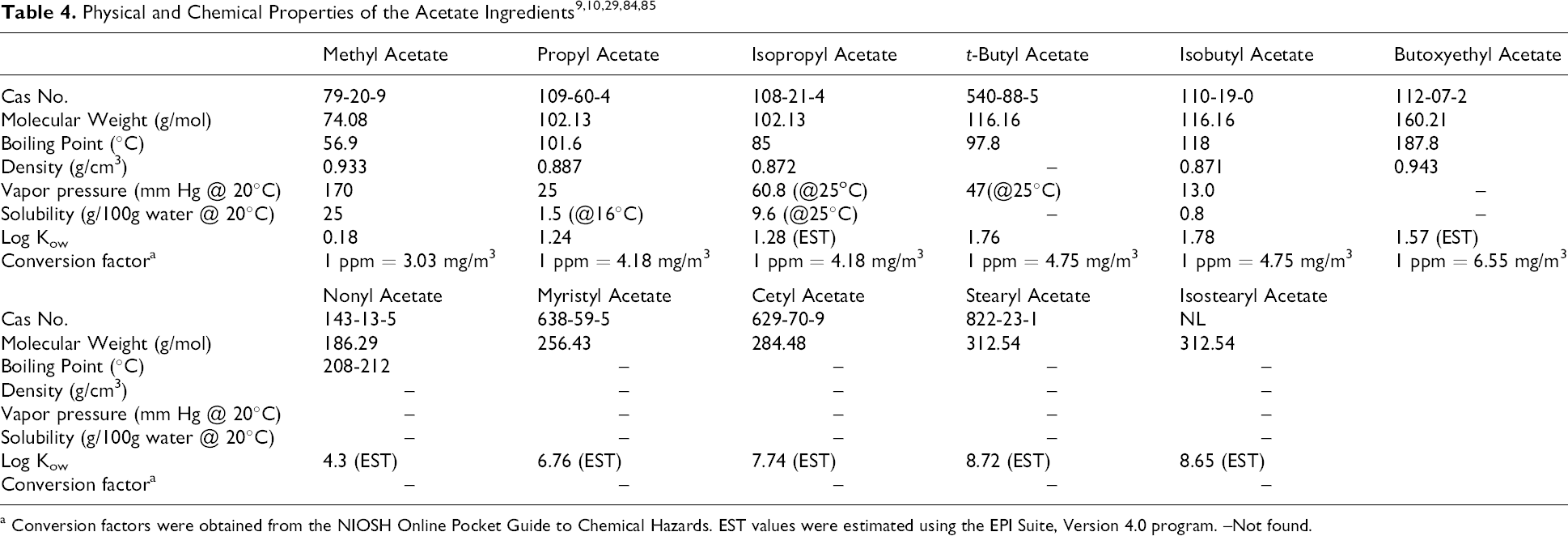
The shorter chain aliphatic esters are colorless and highly volatile liquids. Volatility decreases as the molecular mass (chain length) increases. 9 The physical and chemical properties of the acetates are shown in Table 4. Experimental boiling point, density, vapor pressure, solubility, and log Kow values were available for the shorter alkyl esters while only estimated log Kow values were available for the longer alkyl esters. The shorter alkyl esters (methyl acetate to butoxyethyl acetate) have log Kow values ranging from 0.18 to 1.78 (values for isopropyl acetate and butoxyethyl acetate were estimated using EPI Suite v 4.0), 10 while the longer alkyl esters (nonyl acetate to isostearyl acetate) have estimated log Kow values > 4.
a Conversion factors were obtained from the NIOSH Online Pocket Guide to Chemical Hazards. EST values were estimated using the EPI Suite, Version 4.0 program. –Not found.
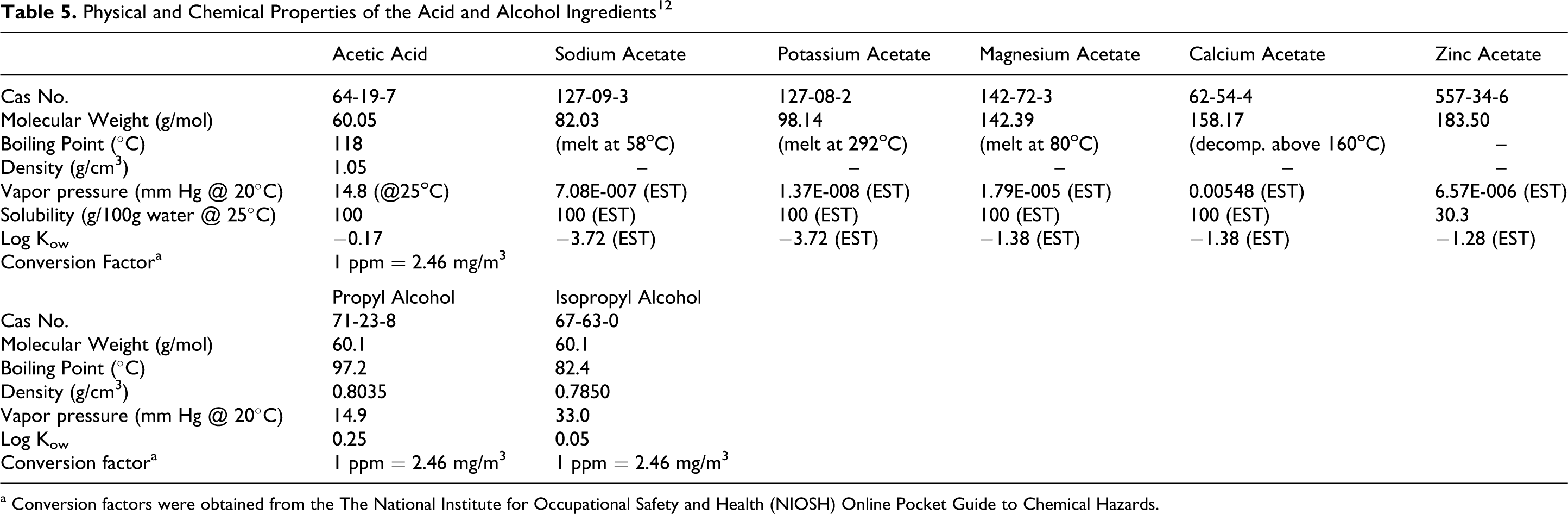
Acetic acid and the methyl acetate salts dissociate readily in water and therefore have negative log octanol/water partioning coefficients and high solubility in water. Propyl alcohol and isopropyl alcohol are volatile liquids. The physical and chemical properties of acetic acid, the acetate salts, and alcohols are shown in Table 5.
Physical and Chemical Properties of the Acid and Alcohol Ingredients 12
a Conversion factors were obtained from the The National Institute for Occupational Safety and Health (NIOSH) Online Pocket Guide to Chemical Hazards.
Manufacture and Production
In general, the alkyl acetates can be produced industrially via esterification of acetic acid. 9 The manufacture of methyl acetate, for example, is traditionally accomplished via a reactive distillation process between acetic acid and methanol. 11 Methanol and ethanol are normally obtained via fermentation of natural sources. However, some sources of alcohols with chains longer than ethanol are often produced synthetically. An important process for producing C3-C20 industrial alcohols involves a process known as oxosynthesis (a process for the production of aldehydes, which occurs by the reaction of olefins [which can be natural or petroleum sourced] with carbon monoxide, hydrogen, and a catalyst [typically cobalt based]), followed by hydrogenation of the aldehyde products, to form the alcohols. 12 More recently, a green, biocatalytic process developed specifically for the manufacture of esters for use in the formulation of cosmetic and personal care ingredients (ie, for producing cosmetic grade esters) has been used. 13
Acetic acid is most commonly manufactured by metal (eg, rhodium or iridium)-catalyzed carbonylation of methanol (via addition of carbon monoxide) or oxidation of ethylene (through an acetaldehyde intermediate and in the presence of manganese acetate, cobalt acetate, or copper acetate), also known as the Wacker process. 14
Impurities
The manufacturing processes of the alkyl esters are typically high yielding (
Analytical Methods
The esters, acids, and alcohols can be analyzed using gas chromatography/mass spectroscopy (GC/MS), nuclear magnetic resonance (NMR) spectroscopy, ultraviolet (UV) spectroscopy, and infrared (IR) spectroscopy. 9,12,16
Ultraviolet Absorption
The ingredients included in this review would not be expected to have any meaningful UV absorption. Except for the acid and ester functional groups, these ingredients do not possess any π bonds or nonbonding electrons. The π bonds and nonbonding electrons in the acid and ester functional groups are not part of any conjugated systems. Accordingly, the likelihood of any of these ingredients to absorb light within the UVA-UVB spectrum, at a detectable molar absorptivity, is extremely low. As such, no UVA-UVB absorption data were found.
Use
Cosmetic
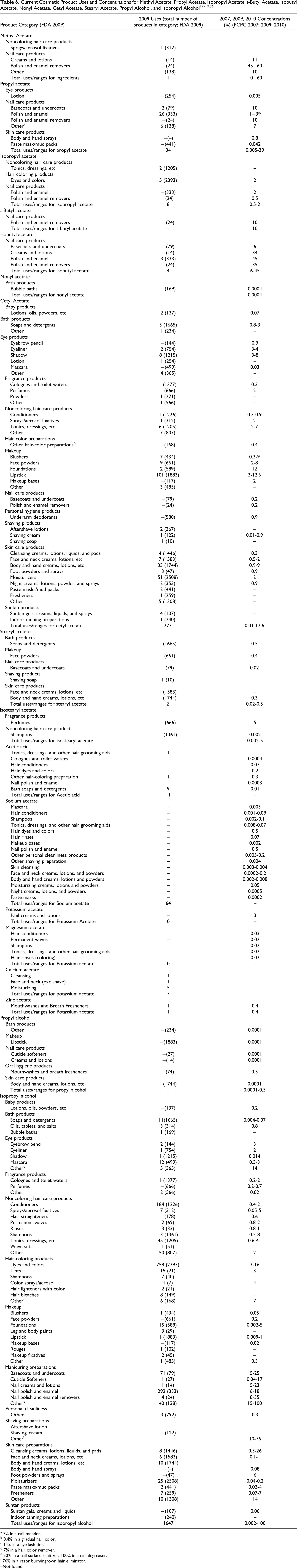
The Voluntary Cosmetic Registration Program (VCRP) administered by the US Food and Drug Administration (FDA) indicates the total number of uses in cosmetic formulations in 2010 for methyl acetate (7), propyl acetate (46), isopropyl acetate (6), isobutyl acetate (4), cetyl acetate (264), stearyl acetate (1), acetic acid (11), sodium acetate (88), calcium acetate (7), zinc acetate (1), propyl alcohol (1), and isopropyl alcohol (1748; Table 6). 17 The use information was not available for t-butyl acetate, nonyl acetate, butoxyethyl acetate, or isostearyl acetate.
a 7% in a nail mender.
b 0.4% in a gradual hair color.
c 14% in a eye lash tint.
d 7% in a hair color remover.
e 50% in a nail surface sanitizer; 100% in a nail degreaser.
f 76% in a razor burn/ingrown hair eliminator.
–Not found.
The main use of methyl acetate is in nail polisher removers. Concentration of use surveys conducted in 2007, 2009, and 2010 by the Personal Care Products Council reported the use percentage ranges for methyl acetate (10-60), propyl acetate (0.005-39), isopropyl acetate (0.5-), t-butyl acetate (10), isobutyl acetate (6-45), nonyl acetate (0.0004), cetyl acetate (0.01-17), stearyl acetate (0.02-0.5), isostearyl acetate (0.002-5), propyl alcohol (0.0001-0.5), isopropyl alcohol (0.002-100), acetic acid (0.0003-0.3), sodium acetate (0.0002-0.5), potassium acetate (3), magnesium acetate (0.02-0.03), and zinc acetate (0.4%). 18,19 Concentration of use survey results for calcium acetate resulted in no reported uses. Neither use nor concentration information was available for butoxyethyl acetate or myristyl acetate.
In the European Union (EU), methanol is allowed only as a denaturant for ethanol and isopropyl alcohol at a concentration of 5%, calculated as a percentage ethanol or percentage isopropyl alcohol. 20 Additionally, the EU limits the amount of zinc acetate in cosmetics to 1% calculated as zinc.
Noncosmetic
The following ingredients in this report are permitted for direct addition to food for human consumption for flavoring purposes and are generally recognized as safe (GRAS) according to the FDA: methyl acetate, propyl acetate, isopropyl acetate, isobutyl acetate, nonyl acetate, propyl alcohol, isopropyl alcohol, acetic acid, sodium acetate, potassium acetate, magnesium acetate, and calcium acetate. 21 Zinc acetate is an approved ingredient for over-the-counter (OTC) skin protectant drug products. 22
Toxicokinetics
These ingredients can be metabolized via hydrolysis, by esterases present in skin, to the parent alcohol and acetic acid (or a salt). The ability and efficiency of esterases in the skin is often species dependent and may even vary considerably between individuals of the same species. 25 Esterases found in the skin, such as human acetyl cholinesterase (hAchE), are capable of metabolizing branched substrates, including tertiary esters. 26
Shorter chain esters readily penetrate the skin and mucous membranes. Acetic acid esters can be metabolized by esterases (present in the respiratory tract, skin, blood, and gastrointestinal tract 27,28 to the parent alcohols and acetic acid. 9 As such, methyl acetate is metabolized to methyl alcohol, t-butyl acetate is metabolized to t-butyl alcohol, and so forth as illustrated in Figure 2.
The parent alcohols can be oxidized via alcohol dehydrogenases to produce the corresponding aldehydes or ketones. The aldehydes can then further be oxidized via aldehyde dehydrogenases to the corresponding acids.
Acetic acid is a principal metabolite of all of the above alkyl acetates, and in addition to its sodium, potassium, magnesium, calcium, and zinc acetate salts, is a cosmetic ingredient and has been included in this safety assessment.
Isobutyl alcohol and nonyl alcohol are principal metabolites of isobutyl acetate and nonyl acetate, respectively.
t-Butyl alcohol is slowly metabolized by alcohol dehydrogenases and is eliminated in urine as a glucuronide conjugate and acetone. t-Butyl alcohol is also eliminated in exhaled air as acetone and carbon dioxide. 16,29
Propyl alcohol and isopropyl alcohol are principal metabolites of propyl acetate and isopropyl acetate, respectively. Propyl alcohol is metabolized to propanol and propanoic acid, which can be further metabolized to acetaldehyde and acetic acid. 30 Isopropyl alcohol is metabolized to acetone and then to acetate, formate, and ultimately carbon dioxide. 31 The half-life of acetone in humans is 22.5 hours.
Human Studies
Butoxyetanol
Dermal—as reported in the CIR safety assessment of butoxyethanol, 32 5 healthy male volunteers exposed 4 fingers of one hand to undiluted butoxyethanol for 2 hours. The percutaneous uptake of butoxyethanol into the blood varied from 127 to 1891 µmol (7-96 nmol butoxyethanol/min per cm2 of exposed area). A linear regression analysis for all of the experiments suggested that, on the average, 17% of the absorbed dose of butoxyethanol was excreted in the urine.
Inhalation—as reported in the CIR safety assessment of butoxyethanol, 32 2 adult male participants (between 30 and 45 years old) and 1 female participant (24 years old) breathed 200 ppm butoxyethanol during two 4-hour periods, separated by a 30-minute lunch. One male participant and the female participant excreted considerable amounts of butoxyacetic acid in urine, while the other male participant excreted only trace amounts in urine.
Methanol
Methyl acetate is metabolized to methanol and therefore, data on the effects in humans following inhalation exposure are applicable to this assessment. Metabolism of methyl acetate to methanol proceeds at a rate directly proportional to the exposure level. Methanol is further metabolized to formaldehyde and then to formic acid. The CIR Expert Panel concluded that formic acid is safe where used in cosmetic formulations as a pH adjustor with a 64 ppm limit for the free acid. 33 The main toxicological risks in humans are severe metabolic acidosis with increased anion gap, typically following oral exposure resulting in >100 mg/L of formate in the urine. 34 The acidosis and the formic acid metabolites are believed to play a central role in both the central nervous system toxicity and the ocular toxicity.
A study was conducted to determine the formate levels that resulted from exposure of human volunteers to 200 ppm of methanol for 4 hours. Human volunteers (n = 27; age 20-55 years) were exposed to 200 ppm methanol (the Occupational Safety and Health Administration [OSHA] Permissible Exposure Limit) for 4 hours and to water vapor for 4 hours in a double-blind, random study. 35 Urine samples were collected at 0, 4, and 8 hours and blood samples were collected from the participants before they entered the chamber, every 15 minutes for the first hour, every 30 minutes from the first to the third hour and at 4 hours. Urine and serum samples were analyzed for formate (LOD 0.5 mg/L). Twenty-six of 27 enrolled participants completed the study (11 females and 15 males). One volunteer withdrew from the study due to blood drawing intolerance. Urine formate data were excluded for 1 participant due to their consumption of high levels of vitamin C that interfered with the formate assay. The researchers did not find any statistically significant differences in serum or urine formate levels between the 2 exposure conditions at any time point. At the end of the 4-hour methanol exposure, formate concentrations of 14.28 ± 8.90 and 7.14 ± 5.17 mg/L were measured in serum and urine, respectively. Under control conditions, formate concentrations of 12.68 ± 6.43 (P = .38; n = 26) and 6.64 ± 4.26 (P = .59; n = 25) mg/L were measured in serum and urine, respectively. After 8 hours (4 hours of no exposure), the serum concentrations were not statistically different, with 12.38 ± 6.53 mg/L under methanol exposure conditions and 12.95 ± 8.01 (P = .6; n = 26) under control conditions. Urine formate concentrations after 8 hours were 6.08 ± 3.49 and 5.64 ± 3.70 (P = .6; n = 25) in exposed and control conditions, respectively, and were not statistically significantly different.
Methyl acetate
Methyl acetate, 15 mL, was applied to the skin of the forearm with cotton dipped into the solvent and fitted in a plastic vessel of 12.5 cm2, which was fixed to the arm with a rubber band and covered with polyethylene film. 36 The amount of skin absorption was estimated by determining the concentrations of the relevant solvents and their metabolites in blood, expired air, and urine. Blood samples were taken before and immediately after application. All the participants were men and 1 to 4 participants served in each experiment. The concentrations of solvents in blood were determined by gas chromatographic method after separating them by gas–liquid equilibrium method. Blood concentration levels of methyl acetate ranged from 0.9 µg/mL at 1 hour postapplication to as great as 3.8 µg/mL at 2 hours
Animal Studies
Acetic acid
Acetic acid is absorbed from the gastrointestinal tract and through the lungs and is readily, although not completely, oxidized. 37 As noted above, acetic acid can be metabolized and eliminated as carbon dioxide and water.
Propyl acetate
Rats (strain/sex/number not specified) were exposed via inhalation to 2000 ppm (8360 mg/m3) for 90 minutes. 38 Propyl acetate was rapidly hydrolyzed to propyl alcohol. During the 90-minute exposure period, blood levels of propyl alcohol were between 2.6- and 7.7-fold greater than propyl acetate.
Isopropyl acetate
Male Sprague-Dawley rats (n = 6) were placed in a chamber charged with 2000 ppm isopropyl acetate and allowed to inhale for 90 minutes. 39 During this time, the concentration in the chamber decreased and correction for the amount of test compound lost to the chamber and on the surface of the animal was completed. Blood samples were taken at 0, 5, 10, 20, 25, 30, 40, 50, 60, and 90 minutes. Blood levels of isopropyl alcohol exceeded those of isopropyl acetate at 5 minutes into the exposure and at each time point thereafter. At 90 minutes, 245 µmol/L isopropyl alcohol and 24 µmol/L isopropyl acetate were detected in the blood.
t-Butyl acetate
Female Sprague-Dawley rats (n = 5) were exposed via a tracheal cannula to 440 ppm (1900 mg/m3) t-butyl acetate in air for 5 hours. 40 The concentrations of both the acetate and the alcohol increased continuously in the blood over the course of the exposure. By the end of the exposure, the concentration of t-butyl alcohol in the blood (∼340 µmol/L) was greater than that of t-butyl acetate (285 µmol/L). In a second experiment, female Sprague-Dawley rats (n = 5) were exposed via a tracheal cannula to 900 ppm (4275 mg/m3) t-butyl acetate in air for 255 minutes. A similar pattern was observed with concentrations in the blood at the end of the exposure of approximately 400 and 450 µmol/L blood for t-butyl acetate and t-butyl alcohol, respectively.
Isopropyl alcohol
Male rabbits (3/group; strain not specified) were treated by different routes of exposure to compare the absorption and metabolism of isopropyl alcohol. 41 Groups 1 and 2 were treated via gavage with the equivalent of 2 and 4 mL/kg absolute isopropyl alcohol, respectively, as a 35% isopropanol/water solution. Groups 3 and 4 were treated via whole-body inhalation for 4 hours (towels soaked with isopropyl alcohol were place in the inhalation chamber and replenished at ½-hour intervals to maintain a saturated environment; no exact concentration given), with group 3 animals receiving an additional dermal exposure in the form of a towel soaked with 70% isopropyl alcohol applied to the animals’ chests and group 4 animals having plastic barriers on their chests and towels prepared the same way as in group 3 applied on top of the plastic barriers. The alcohol in the towels was replenished at ½-hour intervals throughout the duration of the experiment. Blood samples were taken at 0, 1, 2, 3, and 4 hours. Samples were analyzed for isopropyl alcohol and the metabolite acetone.
Following gavage exposure to 2 or 4 mL/kg, maximum blood levels of 147 and 282 mg/dL, respectively, of isopropyl alcohol were measured. Concentrations of acetone rose steadily over the 4-hour period and were 74 and 73 mg/dL following exposure to 2 or 4 mL/kg, respectively. The authors stated that the maximum levels of isopropyl alcohol observed in this experiment correlated with inebriation and near coma in the animals. Following inhalation and dermal exposure, the concentration of isopropyl alcohol in the blood continued to rise and was 112 mg/dL at 4 hours, while the concentration of acetone was 19 at 4 hours. Inhalation exposure with a plastic barrier between the soaked towel and the chest resulted in isopropyl alcohol and acetone blood levels of <10 mg/dL.
The researchers concluded that isopropyl alcohol is absorbed by the dermal route but that prolonged dermal exposure (ie, repeated sponging or soaking for several hours) would be required to produce significant toxicity. 42
Butoxyethanol
The results of 8 studies on the metabolism, distribution, and excretion of butoxyethanol were presented in the CIR expert panel review of butoxyethanol. 43 These data show that butoxyacetic acid is the major metabolite (and toxicant) of butoxyethanol, that the first step of metabolism is mainly by alcohol dehydrogenase in the liver, and that excretion is mainly via urine.
Toxicological Studies
Single-Dose (Acute) Toxicity
Table 7 provides a summary of the available literature on the acute toxicity and LD50 and LC50 data for ingredients in this assessment. 42,44 –49,47, Oral LD50 values were generally >1 g/kg across species and dermal LD50 values were generally higher. Intravenous and intraperitoneal LD50 values were >100 mg/kg. Inhalation LC50 values were > 5000 ppm.
LD50 /LC50 Values Reported in the Literature for Methyl Acetate, Propyl Acetate, Isopropyl Acetate, Isobutyl Acetate, Butoxyethyl Acetate, Nonyl Acetate, Cetyl Acetate, and Isopropyl Alcohol for Various Routes of Exposure and Test Species

Repeated Dose Toxicity
Butoxyethyl acetate
Wistar rats (40 male and female rats divided into groups of 10 males or 10 females) and New Zealand rabbits (4/group) were exposed via inhalation to air vapor mixtures of butoxyethyl acetate (approximately 400 ppm) 4 h/d, 5 d/week for 1 month. 48 No effects were observed on body weight gain, as compared to controls. Red blood cell (RBC) counts and hemoglobin decreased slightly in 2 of 4 rabbits after 3 weeks of treatment. Hemoglobinuria and hematuria were observed in the rabbits but were less pronounced in treated rats. Two rabbits died during the fourth week of treatment and blood-filled kidneys and urinary bladders were observed at necropsy in these 2 animals. No other gross lesions were observed in the other animals killed at either the end of the study or after a 1-week recovery period.
Wistar rats (40 male and female rats divided into groups of 10 males or 10 females) and New Zealand rabbits (2/sex/group) were exposed via inhalation to 100 ppm butoxyethyl acetate (butylglycol acetate [BGA]) 4 h/d, 5 d/week for 10 months. 48 No effects on body weight gain, as compared to controls, and no hematological changes were observed. Upon necropsy, rabbits exhibited very discrete renal lesions including a few areas of tubular nephritis. Additionally, dilation of Henle loop and the distal convoluted tubules was observed to a greater degree than in control animals. Treated and control rats also exhibited discrete renal lesions such as tubular enlargement in males and tubular nephrosis in females.
Acetic acid
Of the 5 rats (sex and strain not specified) exposed via inhalation to 1300 µg/L of acetic acid, 3 showed slight red staining around the nose, with 1 animal showing staining around the mouth. 50 Focal lesions were observed in the respiratory epithelium of the dorsal meatus of level 1 of the nasal cavity in 3 of the 5 rats. Acetic acid also increased spleen and kidney weights/damage at 23 to 31 mg/kg in rats, and induced hyperplasia in both organs at 60 mg/kg. 51
Rats (number, sex, and strain not specified) were exposed to 0.01% to 0.25% solutions, via drinking water, of acetic acid (corresponding to 0.2 mL/kg), with no toxic effects over a period of 2 to 4 months. However, 0.5% solutions (corresponding to 0.33 mL/kg) immediately affected feed consumption and growth. A maximum toleration level of 30 mmol/L (1.8 g/L) daily for 2 weeks was established for rats. 37
Sodium acetate
In contrast to the maximum toleration level recited above for acetic acid, sodium acetate in drinking water was reported to have a maximum toleration level of 80 mmol/L (4.2-4.8 g/L). 37
Isobutyl alcohol
Isobutyl alcohol, the primary metabolite of isobutyl acetate, was evaluated for potential neurotoxicity in Sprague-Dawley rats. Rats (10/sex/group) were exposed via inhalation to isobutyl alcohol vapor concentrations of approximately 0, 770, 3100, or 7700 mg/m3, for 6 h/d, 5 d/week, for 14 weeks. 16 The functional observational battery was conducted along with end points of motor activity, neuropathology, and scheduled-controlled operant behavior. A slight reduction in responsiveness to external stimuli was observed in all treated groups during exposure. This effect resolved upon cessation of exposure to isobutyl alcohol.
Isopropyl alcohol
Fischer 344 rats and CD-1 mice (10/sex/group) were exposed via inhalation to 0, 100, 500, 1500, or 5000 ppm (0, 246, 1230, 3690, or 12 300 mg/m3) isopropyl alcohol for 6 h/d, 5 d/week for 13 weeks. 52 To evaluate the neurobehavioral effects of isopropanol exposure, an additional 15 rats/sex were exposed (via inhalation) to 0, 500, 1500, or 5000 ppm (0, 1230, 3690, or 12 300 mg/m3) for 6 h/d, 5 d/week for 13 weeks.
The NOAEL for rats in this study was reported as 500 ppm and the LOAEL was 1500 ppm, based upon clinical signs and changes in hematology at 6 weeks. 52 Isopropyl alcohol did not produce any changes to the parameters of the functional observations battery which was conducted at 1, 2, 4, 9, and 13 weeks.
The NOAEL for mice in this study was reported as 500 ppm and the LOAEL was 1500 ppm, based on clinical signs and increased liver weights. 52
Methyl Alcohol
As reported in the CIR safety assessment of methyl alcohol, 53 ingestion by humans can result in severe metabolic acidosis, blindness, and death.
Dermal Irritation/Sensitization/Photosensitization
Cetyl Acetate
A lipstick containing 12.6% cetyl acetate caused no dermal sensitization in 99 human test participants. 54 A formulation of 11.7% cetyl acetate did not cause sensitization in humans, in another report. 55
Propyl Acetate
Animal
Male and female rabbits (n = 4; strain not specified) were tested for primary irritation on intact, abraded skin using the Draize method. Undiluted propyl acetate (0.5 mL) was applied to the skin without occlusion and produced only minor irritation with slight erythema in 1 of 4 animals. No edema was observed 72 hours after application. 37
No irritation was observed in 5 rabbits (sex/strain not specified), following a 24-hour nonoccluded treatment with 0.01 mL of undiluted propyl acetate (no further experimental details provided). 37
Erythema and necrosis were observed in rabbits (sex/strain/number not specified) exposed to 20 mL/kg bw (17 756 mg/kg bw) undiluted propyl acetate. 37 Erythema and desquamation were observed in guinea pigs (sex/strain/number not specified) exposed to 10 mL/kg bw (8880 mg/kg bw), undiluted propyl acetate, for 24 hours with occlusion. In the guinea pigs, the skin appeared normal after 14 days.
Human
In a human maximization test pre-screen, 2% propyl acetate in petrolatum was applied to the backs of 25 healthy participants for 48 hours under occlusion. No participant experienced any irritation or sensitization. 55
Butoxyethyl acetate
New Zealand rabbits (6/group/1500 mg/kg) were tested for primary irritation on intact, abraded skin using the Draize method. 48 Butoxyethyl acetate (1500 mg/kg) produced slight erythema (grade 1) in 4 of 6 rabbits at 24 hours. At the 72 hours reading, there was no perceptible irritation; the primary irritation index was reportedly 0.17.
Methyl acetate
Human maximization tests were carried out with 10% methyl acetate in petrolatum on various panels of volunteers. Application was under occlusion to the same site on the forearms or backs of all participants for 5 alternate-day 48-hour periods. 5 Patch sites were pretreated for 24 hours with 2.5% aqueous sodium lauryl sulfate (SLS) under occlusion. Following a 10- to 14-day rest period, challenge patches were applied under occlusion to fresh sites for 48 hours. Challenge applications were preceded by a 60-minute SLS treatment. Reactions were read at patch removal and again at 24 hours thereafter. No sensitization reactions were observed in any of the 25 participants tested.
Zinc acetate
The dermal irritancy of 6 zinc compounds was examined in 3 animal models. 56 In open patch tests involving 5 daily applications, aqueous zinc acetate (20%) was found to be severely irritant in rabbit, guinea-pig, and mouse tests, inducing epidermal hyperplasia and ulceration. Epidermal irritancy in these studies was reportedly related to the interaction of zinc ion with epidermal keratin.
Acetic Acid
Animal
Glacial acetic acid (equivalent to 95% acetic acid) caused complete destruction of the skin of guinea pigs on 24 hours contact; 28% acetic acid, however, resulted in only moderate irritation after 24 hours. 57
Human
In a number of independent test facilities, a total of 96 human volunteers (sex not given) were exposed to acetic acid in 4-hour dermal irritation patch test. 58,59 At a concentration of 10% acetic acid, 70% to 94% of volunteers, depending on the facility, reported dermal irritation.
t-Butyl alcohol
As reported in the CIR safety assessment of t-butyl alcohol, 50 one study was conducted using New Zealand white rabbits exposed to a mixture of ethanol and t-butyl alcohol (concentrations unspecified) at abraded and intact skin sites. Only 2 animals had irritant reactions at the intact skin site and only 3 animals had irritant reactions at the abraded site. Overall, the test article was not considered a primary dermal irritant under the conditions of the study. In another study, t-butyl alcohol alone (concentration unspecified) was found to have no irritating effect on the skin of shaved rabbits (strain unspecified) when observed for a period of 1 week. Two case reports were reported with positive patch tests to t-butyl alcohol.
Methyl alcohol
As given in the CIR safety assessment of methyl alcohol, 53 methyl alcohol causes primary irritation to the skin; prolonged and repeated contact with methyl alcohol resulted in defatting and dermatitis. Typical allergic responses observed after contact with alcohols were eczematous eruption and wheal and flare at the exposure sites.
Propyl alcohol
A cumulative irritation study was conducted involving 20 male participants to evaluate the relative irritancy of free fatty acids of different chain lengths using propanol as a vehicle. 60 A control patch containing propanol was applied to each participant. Patches remained in place for 24 hours and reactions were scored 30 minutes after patch removal. This procedure was repeated daily for a total of 10 applications. In both groups of 10 participants, there were no reactions to propanol.
A series of studies 61 –67 of the irritation potential of pelargonic acid were conducted using propanol as a vehicle. In 116 healthy male participants (21-55 years old), 10 females and 6 males (median age of 29.5 years), 42 healthy, nonatopic male volunteers (mean age = 34 years), 16 healthy participants (ages not stated), and 17 healthy volunteers (10 males and 7 females; mean age = 27 years), no positive patch test results were reported at the vehicle control site.
Isopropyl alcohol
A spray concentrate with isopropyl alcohol at 80.74% did not exhibit any potential for dermal sensitization in 9 human participants. 32
In a human repeated insult patch test on 109 test participants, a hair dye base formulation of 2.85% isopropyl alcohol, and 1.95% isopropyl acetate resulted in no dermal sensitization. 68
A study 69 of the irritation potential of pelargonic acid was conducted using isopropanol as a vehicle. In healthy individuals (8 males and 4 males; 18-64 years old), no positive patch test results were reported at the vehicle control site.
Butoxyethyl alcohol
As given in the CIR safety assessment of butoxyethanol, 32 10.0% (vol/vol) aqueous butoxyethanol evaluated using 214 male and female participants between 18 and 76 years of age resulted in no evidence of sensitization.
Cetyl alcohol
As given in the CIR safety assessment of cetyl alcohol, 7 1 out of 80 male participants (21-52 years of age) had etythema, folliculitis, and pustule formation at the application site in a topical tolerance study of a cream base with 11.5% cetyl alcohol. Using the same protocol in 20 participants, the cream base with 6% cetyl alcohol resulted in no irritation. A cream containing 6% cetyl alcohol was a mild irritant in 12 female participants (18-60 years of age). No primary irritation or sensitization was reported in 110 female participants tested with a product containing 8.4% cetyl alcohol. No photosensitization reactions were reported in a test of a lipstick product containing 4.0% cetyl alcohol in 52 participants. In another study, a skin care preparation containing 1.0% cetyl alcohol did not induce photosensitization in the 407 participants tested. In neither case were the experimental details regarding the radiation source given.
Myristyl alcohol
As given in the CIR safety assessment of myristyl alcohol, 7 a moisturizing lotion containing 0.80% myristyl alcohol was applied to the face of each of 53 participants over a period of 4 weeks. None of the participants had signs of skin irritation. In another study, the irritation potential of a moisturizing lotion containing 0.25% myristyl alcohol was evaluated in 51 participants, used daily during a 1-month period. A burning sensation was experienced by 1 of the participants 1 day after the initial use of the product. None of the participants had signs of skin irritation. A moisturizing lotion containing 0.25% myristyl alcohol was applied to the backs of 229 male and female participants via occlusive patches for 24 hours. The product was reapplied to the same sites following a 24-hour nontreatment period and repeated for a total of 10 induction applications. None of the participants had reactions to the product. No photosensitization reactions were reported in a test of a moisturizing lotion containing 0.10% myristyl alcohol evaluated for its photosensitization potential in a study involving 52 participants. Experimental details regarding the radiation source were not given.
Stearyl alcohol
As given in the CIR safety assessment of stearyl alcohol, 70 in a 24-hour single insult occlusive patch tests, mild irritation was produced by 100% stearyl alcohol in 1 participant out of 80. A case report of a contact sensitization to stearyl alcohol in 3 individuals was reported.
Isostearyl alcohol
As given in the CIR safety assessment of isostearyl alcohol, 7 no skin irritation was reported in 19 male and female participants (18-65 years old) at a concentration of 25.0% in petrolatum, or in 3 different lipstick products containing 25.0%, 27.0%, and 28.0% isostearyl alcohol, respectively. Isostearyl alcohol (25% v/v in 95.0% isopropyl alcohol) evaluated in 12 male participants (21-60 years old) resulted in mild irritation in 3 participants but no evidence of sensitization. A pump spray antiperspirant containing 5.0% isostearyl alcohol evaluated using 148 male and female participants resulted in 10 participants with reactions suggestive of sensitization and were rechallenged with the product 2 months later. Six participants had reactions during the rechallenge. In a second study, the same product was applied to 60 male and female participants (same protocol). Five of the participants had positive responses after the first challenge. One of the 5 was rechallenged with 5.0% isostearyl alcohol in ethanol solution, and a positive reaction was observed.
Ocular Irritation
Propyl Acetate
Undiluted propyl acetate (0.5 mL) caused minor corneal injury (grade 2 on a scale of 0-10) in the rabbit eye (n/sex/strain not specified). 38
Butoxyethyl Acetate
Butoxyethyl acetate produced slight conjunctival redness and discharge in 2 of 6 New Zealand rabbits at 24 hours. 48 At 48- and 72-hour observations, no irritation was observed.
Isopropyl Alcohol
Isopropyl alcohol has been labeled a severe ocular irritant based on rabbit ocular irritation tests involving application of 0.1 mL of a 70% solution in water. 31
Acetic Acid
Acetic acid at concentrations greater than 10% caused severe permanent eye injury in rabbits. In contrast, a 5% solution (equivalent to vinegar) caused severe, but reversible (two week recovery), eye injury. 57
Reproductive/Developmental Toxicity
Oral
t-Butyl acetate
Pregnant female Sprague-Dawley rats (22/group) were exposed to 0, 400, 800, or 1600 mg/kg per d t-butyl acetate via gavage on gestational days (GDs) 6 through 19. 71 Dams were monitored for clinical effects, feed consumption, and changes in body weight, and the fetuses examined for body weight, sex, and visceral and skeletal alterations at GD 20. Two dams died after treatment with 1600 mg/kg. Necropsy findings on these animals included liver hypertrophy, stomach expansion, and congestion/hemorrhage of the small intestines. Clinical signs in the 1600 mg/kg group included piloerection, abnormal gait, decreased activity, loss of fur, reddish vaginal discharge, nasal hemorrhage, and coma. There were no deaths and no clinical signs in the 400 and 800 mg/kg groups. A dose-dependent decrease in gestational weight gain was observed during the treatment period, but this was not statistically significant as compared to controls. Feed consumption was significantly decreased on GD 6 and 9 in the 1600 mg/kg treatment group as compared to controls. No effects were observed on the number of corpora lutea, implantations, fetal deaths, litter size, and gender ratios. Male fetal body weight was significantly decreased in the 1600 mg/kg group as compared to controls. Female fetal body weight was also decreased at this exposure level, but the difference was not statistically significant. An increase in the incidence of skeletal variation and a delay in fetal ossification were observed in the 1600 and 800 mg/kg treatment groups, with the changes in the 800 mg/kg treatment group described as minimal by the authors. No evidence of teratogenicity was observed at any tested exposure level. The authors concluded that the observed developmental effects were due to maternal toxicity and determined the NOELs for both maternal and embryo-fetal developmental toxicity in rats of 800 and 400 mg/kg, respectively.
Isopropyl alcohol
Female Sprague-Dawley rats (25/group) received 0, 400, 800, or 1200 mg/kg per d isopropyl alcohol via gavage on GD 6 through 15. 72 Female New Zealand white rabbits (15/group) were exposed to 0, 120, 240, or 480 mg/kg per d isopropyl alcohol via gavage on GD 6 through 18. Animals were observed for body weight, clinical effects, and feed consumption and the fetuses examined for body weight, sex, and visceral and skeletal alterations at GD 20 for rats and GD 30 for rabbits. In the rat study, 2 dams died at the 1200 mg/kg dose and 1 dam died at the 800 mg/kg dose. Maternal gestational weight gain was reduced at the highest dose tested. No other effects were observed on maternal reproductive health. Fetal body weights at the 2 highest doses were decreased statistically. No evidence of teratogenicity was observed at any dose. In the rabbit study, 4 does died at the 480 mg/kg dose. Treatment-related clinical signs of toxicity were observed at the 480 mg/kg dose, which included cyanosis, lethargy, labored respiration, and diarrhea. No treatment-related findings were observed at GD 30. Decreased feed consumption and maternal body weights, at 480 mg/kg, were statistically significant. No other effects were observed on maternal reproductive health. No evidence of teratogenicity was observed in the rabbits at any dose. The authors determined NOELs for both maternal and developmental toxicity of 400 mg/kg, each, in rats and 240 and 480 mg/kg, respectively, in rabbits.
Acetic acid
An assessment plan for acetic acid and salts submitted as part of the high production volume chemical challenge program reported that no effects were observed on nidation or on maternal or fetal survival in mice, rats, and rabbits at oral doses (intubation/dosed day 6 of gestation) up to 1600 mg/kg bw/d of acetic acid. 51
Sodium acetate
No maternal or neonatal effects were observed in mice exposed (gavage/dosed daily on days 8-12 of gestation) to 1000 mg/kg of sodium acetate. 51 Sodium acetate was also determined to be nonteratogenic to chick embryos (10 mg/egg).
Inhalation
Propyl alcohol
The effects of propyl alcohol on fertility were investigated by exposing male Sprague-Dawley rats (18/group) to 0, 3500, or 7000 ppm (0, 8.61, or 17.2 mg/L) propyl alcohol vapor via inhalation 7 h/d, 7 d/week for 62 days, prior to mating with unexposed virgin females. 53 Female Sprague-Dawley rats (15/group) were similarly exposed and mated with unexposed males. Following parturition, litters were culled to 4/sex and the pups fostered by unexposed dams. The pups were weaned on postnatal day (PND) 25 and weighed on PNDs 7, 14, 21, 28, and 35. Male rats exposed to 7000 ppm exhibited a decrease in mating success with 2 of 16 producing a litter (1 male died as a result of a cage fight and 1 male did not mate). Mating success was not affected in 3500 ppm exposed males or in females. Six males from the 7000 ppm group were retained to determine whether this effect was reversible. All 6 males successfully mated 15 weeks after exposure. The authors reported that weight gain was not affected in females exposed to 7000 ppm (data not shown) but the feed intake was decreased in this treatment group. Crooked tails were observed in 2 to 3 offspring in 2 of 15 litters from the 7000 ppm maternally exposed group. No other effects on female fertility were reported. No significant differences resulted between offspring of the 7000 ppm group and controls on several behavioral toxicology measures including the Ascent test, Rotorod test, Open Field test, activity test, running wheel activity, avoidance conditioning, and operant conditioning. Activity measures were significantly different between offspring of the 3500 ppm exposure group and controls.
Genotoxicity
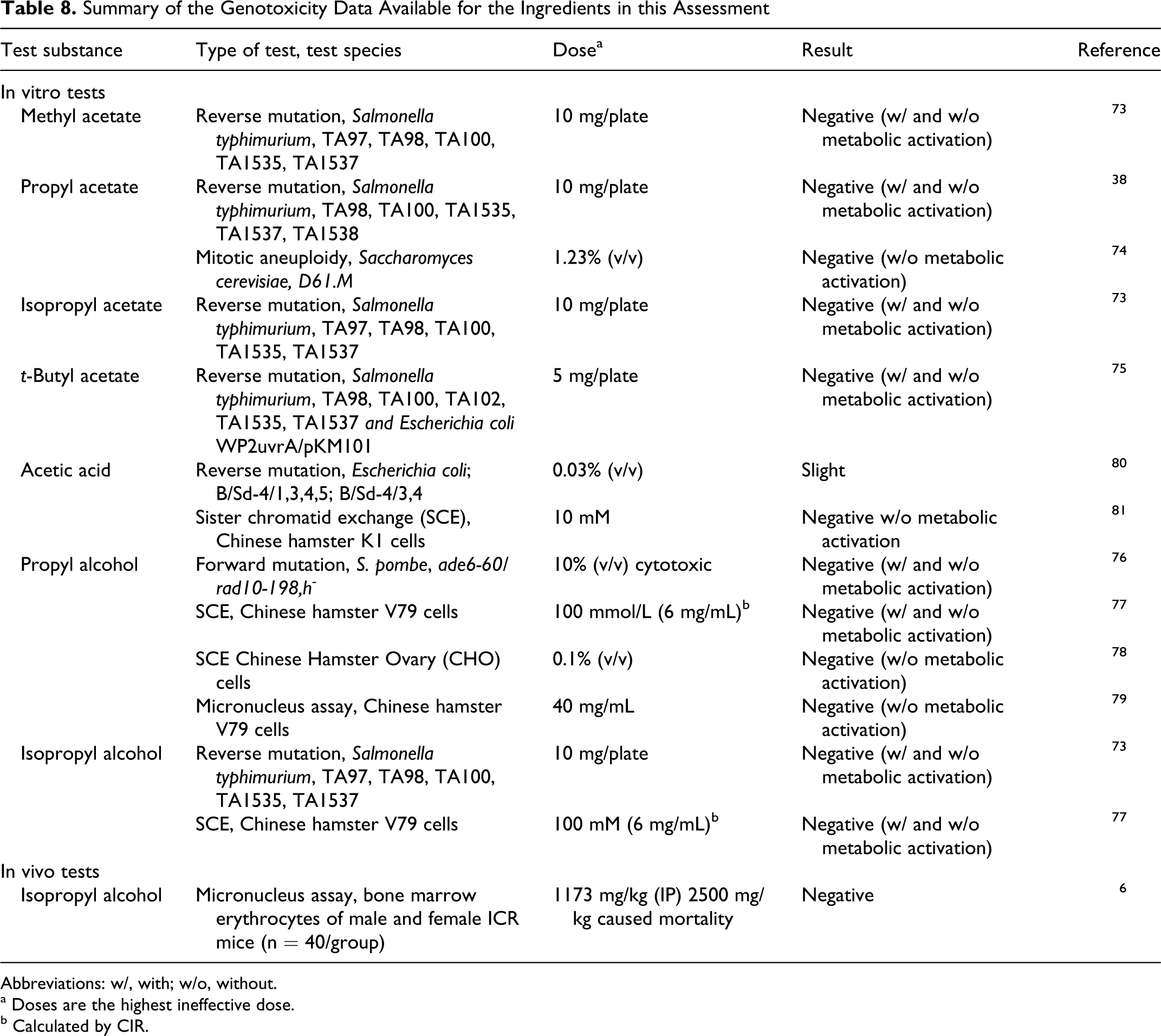
A summary of in vitro and in vivo genotoxicity studies is given in Table 8.
Summary of the Genotoxicity Data Available for the Ingredients in this Assessment
Abbreviations: w/, with; w/o, without.
aDoses are the highest ineffective dose.
bCalculated by CIR.
Methyl acetate, propyl acetate, isopropyl acetate, t-butyl acetate, propyl alcohol, and isopropyl alcohol
Methyl acetate, propyl acetate, isopropyl acetate, t-butyl acetate, propyl alcohol, and isopropyl alcohol were not mutagenic in in vitro bacterial and mammalian cell assays. 38,73 –79 Isopropyl alcohol was not genotoxic in an in vivo micronucleus assay. 6
Acetic acid
Acetic acid was reported to be slightly mutagenic to Escherichia coli and mammalian cells, but, a more recent mammalian assay suggests that acetic acid is not mutagenic and that previous results were an aberration due strictly to low pH, and not the identity of the pH reducer. 80,81
Carcinogenicity
Inhalation
Isopropyl alcohol
Fischer 344 rats and CD-1 mice (65/rats/sex/group; 55/mice/sex/group) were treated via inhalation with 0, 500, 2500, or 5000 ppm (0, 1230, 6150, or 12 300 mg/m3) isopropyl alcohol for 6 h/d, 5 d/week for 104 weeks in rats and 78 weeks in mice. 82 An additional 10/animals/sex/species were treated with these same concentrations of isopropyl alcohol for 6 h/d, 5 days/week for 72 weeks in rats and 54 weeks in mice and underwent an interim evaluation. Another 10 mice/sex/group were treated according to the paradigm described above for 54 weeks and then allowed to recover before being killed at 78 weeks. Animals were observed and evaluated for body and organ weights, ophthalmology, and clinical and anatomic pathology.
In rats, increased mortality due to chronic renal disease was observed at 5000 ppm (both sexes) and at 2500 ppm (males only). 82 Hypoactivity and lack of startle reflex were observed in rats treated with 2500 ppm, and hypoactivity, lack of startle reflex, and narcosis were observed in rats treated with 5000 ppm. With the exception of the ataxia, the clinical signs were transient and ceased when the exposure ended. Increases in body weight, body weight gain, and liver weights were observed in rats treated with 2500 and 5000 ppm. Chronic renal disease was exacerbated in rats treated with isopropyl alcohol. Male rats had a concentration-related increase in absolute and relative (B.W.) testes weights. At the interim euthanasia (after 72 weeks), male rats treated with 5000 ppm had an increased frequency of testicular seminiferous tubule atrophy upon microscopic evaluation. At the terminal euthanasia (104 weeks), male rats had a concentration-dependent increase in the incidence of interstitial (Leydig) cell adenomas of the testes at all administered doses. No other tumor types were increased in rats under these treatment conditions as compared to controls.
In mice, no differences in mortality were observed between control and treated animals. 82 Hypoactivity, lack of a startle reflex, narcosis, ataxia, and prostration were observed in mice treated with 5000 ppm . Hypoactivity, lack of startle reflex, and narcosis were observed in mice treated with 2500 ppm. Increases in body weight, body weight gain, and liver weights were observed in mice treated with 2500 and 5000 ppm. Male mice in all treatment groups had a decrease in relative weights of testes, and female mice exposed to 5000 ppm isopropyl alcohol exhibited decreases in absolute and relative (BW) brain weights. At the terminal euthanasia (78 weeks), an increased incidence of minimal-to-mild renal tubular proteinosis was observed in males and females in all treatment groups. Male mice exposed to 2500 and 5000 ppm exhibited an increased incidence of dilation of the seminal vesicles. No neoplastic lesions were observed in male or female mice. The authors reported a NOAEL for toxic effects of 500 ppm for both rats and mice based on kidney and testicular effects.
The International Agency for Research on Cancer has determined that isopropyl alcohol is not classifiable as to its carcinogenicity to humans (group 3). 69
Oral
t-Butyl alcohol
F-344 rats (n = 60/group, males and females) were exposed orally, via drinking water, to t-butyl alcohol at various doses for 15 months to 103 weeks. At 420 mg/kg per d in males and 650 mg/kg per d in females, there was a decreased survival. 4 Additionally, a dose-related decrease in body weight gain was observed. All treated females had nephropathy and a dose-related increase in kidney weight. Males also demonstrated kidney weight gain but not at all doses. 4
After 24 months of exposure, a combined incidence for adenoma and carcinoma of the renal tubules was found in 8/50, 13/50, 19/50, and 13/50 of the control, low-, mid-, and high-dose males (not females), respectively. 4
Summary
The ingredients methyl acetate, propyl acetate, isopropyl acetate, t-butyl acetate, isobutyl acetate, butoxyethyl acetate, nonyl acetate, myristyl acetate, cetyl acetate, stearyl acetate, and isostearyl acetate are alkyl esters that function in cosmetics as fragrance ingredients, solvents, and skin-conditioning agents. The ingredients acetic acid, sodium acetate, potassium acetate, magnesium acetate, calcium acetate, and zinc acetate represent the acetic metabolite of the above alkyl acetates. Acetic acid and its salts function as one or more of the following agents: pH adjusters, buffering agents, viscosity controllers, cosmetic astringents, cosmetic biocides, skin protectants, and fragrance ingredients. The ingredients propyl alcohol and isopropyl alcohol are also included because they are metabolites of propyl acetate and isopropyl acetate, respectively, and they function in cosmetics as antifoaming agents, fragrance ingredients, solvents, and viscosity decreasing agents.
Exposure to these ingredients is expected to occur mostly by the inhalation and dermal routes, although some oral or ocular exposure could occur depending on the types of products in which they are used. Shorter chain acetic esters readily penetrate the skin and mucous membranes and are metabolized via esterases to the parent alcohol and acetic acid. The alcohols are further metabolized to the corresponding aldehyde or ketone and then to the corresponding acid. The LD50 values, for those ingredients in this assessment with acute toxicity data, are greater than 1 g/kg.
Alkyl acetates
Central nervous system depression and narcotic-like effects have been documented in animals for the shorter alkyl chain acetates at doses much larger than can reasonably be attained from cosmetic product exposures. 83 The alkyl acetate ingredients have been labeled as the minor skin and eye irritants in animal studies. Those alkyl acetate ingredients that have been tested have been found to be negative for mutagenicity, in vitro.
A formulation of 1.95% isopropyl acetate did not cause sensitization in humans in 1 report. A formulation of 11.7% cetyl acetate did not cause sensitization in humans in one report, or in a formulation of 12.6% cetyl acetate in humans in another report. A formulation of 10% methyl acetate did not cause sensitization in humans in 1 report. A formulation of 2% propyl acetate did not cause sensitization in humans in 1 report. The NOAELs for reproductive toxicity for t-butyl acetate were greater than or equal to 400 mg/kg in rats. Ethyl acetate and butyl acetate have been found to be safe as used by the CIR Expert Panel.
Acetic acid/salts
Central nervous system depression has been documented in animals exposed to acetic acid. Acetic acid has been labeled as a minor skin irritant, at low concentrations, in animal and human studies, and a severe ocular irritant in a rabbit ocular irritation test. The sodium salt of acetic acid has a more than 2-fold higher toleration level than the pure free acid, and acetic acid is not mutagenic when buffered to physiological pH.
Alcohols
The alcohol metabolites ethyl alcohol, butyl alcohol, t-butyl alcohol, butoxyethyl alcohol (with qualifications), myristyl alcohol, cetyl alcohol, stearyl alcohol, and isostearyl alcohol have been found to be safe as used by the CIR Expert Panel.
Isopropyl alcohol has been labeled a severe ocular irritant in a rabbit ocular irritation test. Isopropyl alcohol was negative in an in vivo micronuclei assay. A formulation of 2.85% isopropyl alcohol did not cause sensitization in humans, in 1 report, and a spray concentrate of 80.74% did not exhibit any potential for dermal sensitization in humans, in another report. Central nervous system depression and behavioral effects have been documented in humans for isopropyl alcohol at relatively high concentrations unlikely to result from cosmetic product exposures. Reproductive toxicity NOELs for isopropyl alcohol were reported for maternal and developmental toxicity of 400 mg/kg each in rats and 240 and 480 mg/kg in rabbits, respectively. In an inhalation carcinogenicity study of isopropyl alcohol, rats exhibited an exacerbation of chronic renal disease and a concentration-dependent increase in interstitial cell adenomas of the testes. Male mice exhibited dilation of the seminal vesicles at 2500 ppm, but no neoplastic lesions were observed.
Exposure to 3500 ppm of n-propyl alcohol resulted in significantly unfavorable offspring behavioral toxicology measures as compared to controls.
Inhalation exposure to isobutyl alcohol induced a slight reduction in responsiveness to external stimuli in rats.
Long-term oral exposure of t-butyl alcohol to rats, at relatively high concentrations unlikely to result from cosmetic product exposures, resulted in more combined adenoma and carcinoma of the renal tubules than in controls (13-19/50 vs 8/50).
Discussion
An unpublished maximization study of a lipstick formulation containing cetyl acetate showed no sensitization at the highest reported concentration of use (12.6%). Additionally, the sensitization data received supported the lack of potential for human dermal sensitization to methyl acetate, propyl acetate, and isopropyl acetate. The Panel determined that the ingredients assessed herein are safe in present practices of use and concentration.
The Panel recognized that butoxyethanol, a metabolite of butoxyethyl acetate, was previously determined to be safe as used with the qualification that it may be safely used up to 10% in hair and nail products and 50% in nail polish removers. However, the Panel determined that the concentration of butoxyethyl acetate that would be required to generate appreciable quantities of butoxyethanol through metabolic pathways is well above the present use concentrations of related alkyl acetates in cosmetics (butoxyethyl acetate is not currently being used in cosmetics). Furthermore, the LD50 values reported for butoxyethyl acetate are relatively high. Accordingly, the Panel has determined that butoxyethyl acetate should be included in the safe as used assessment of this report.
The CIR Expert Panel recognizes that there are data gaps regarding the use and concentration of these ingredients. However, the overall information available on the types of products in which these ingredients are used and at what concentrations indicate a pattern of use, which was considered by the Expert Panel in assessing safety.
Conclusion
The CIR Expert Panel concluded that methyl acetate, propyl acetate, isopropyl acetate, t-butyl acetate, isobutyl acetate, butoxyethyl acetate*, nonyl acetate, myristyl acetate*, cetyl acetate, stearyl acetate, isostearyl acetate, acetic acid, sodium acetate, potassium acetate, magnesium acetate, calcium acetate, zinc acetate, propyl alcohol, and isopropyl alcohol are safe in the present practices of use and concentration. Were ingredients in this group not in current use (identified by *) to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group (eg, butoxyethyl acetate would be safe if used in product categories and at concentrations comparable to the alkyl acetates in this assessment).
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.