Abstract
Xylene sulfonic acid, toluene sulfonic acid, and alkyl aryl sulfonate hydrotropes used in cosmetics as surfactants, hydrotropes, were reviewed in this safety assessment. The similar structure, properties, functions, and uses of these ingredients enabled grouping them and using the available toxicological data to assess the safety of the entire group. The Cosmetic Ingredient Review Expert Panel reviewed relevant animal and human data related to these ingredients. The panel concluded that xylene sulfonic acid and alkyl aryl sulfonate hydrotropes are safe as cosmetic ingredients in the present practices of use and concentrations as described in this safety assessment, when formulated to be nonirritating.
Introduction
This safety assessment includes a group of alkyl aryl sulfonate hydrotropes that are cosmetic ingredients and the corresponding sulfonic acids from which they are made, including xylene sulfonic acid, ammonium cumenesulfonate, ammonium xylenesulfonate, calcium xylenesulfonate, potassium cumenesulfonate, potassium toluenesulfonate, potassium xylenesulfonate, sodium toluenesulfonate, sodium cumenesulfonate, sodium xylenesulfonate, and toluene sulfonic acid. Cumenesulfonic acid is not included because it is not listed as a cosmetic ingredient. Hydrotropes are amphiphilic substances composed of both hydrophilic and hydrophobic functional groups. They can increase the solubility of less soluble substances. Similar chemical structures, physicochemical properties, and functions and concentrations in cosmetics enable in grouping these ingredients and reading across the available toxicological data to support the safety assessment of the entire group.
All of these ingredients have the same functions (surfactant—hydrotropes) in cosmetics, but only ammonium xylenesulfonate, sodium xylenesulfonate, and sodium cumenesulfonate have currently reported uses in cosmetic products. Xylene sulfonic acid and toluene sulfonic acid are included in this report as they are the immediate precursors to the pH-adjusted materials that are used in cosmetic formulations.
Under the aegis of the Organization for Economic Co-Operation and Development, a summary Information Data Set (SIDS) initial assessment report for the hydrotropes category was prepared. 1 The information contained in that report 1 was the only source for much of the data discussed in this safety assessment specific to the use of hydrotropes in cosmetics.
Chemistry
Definition and Structure
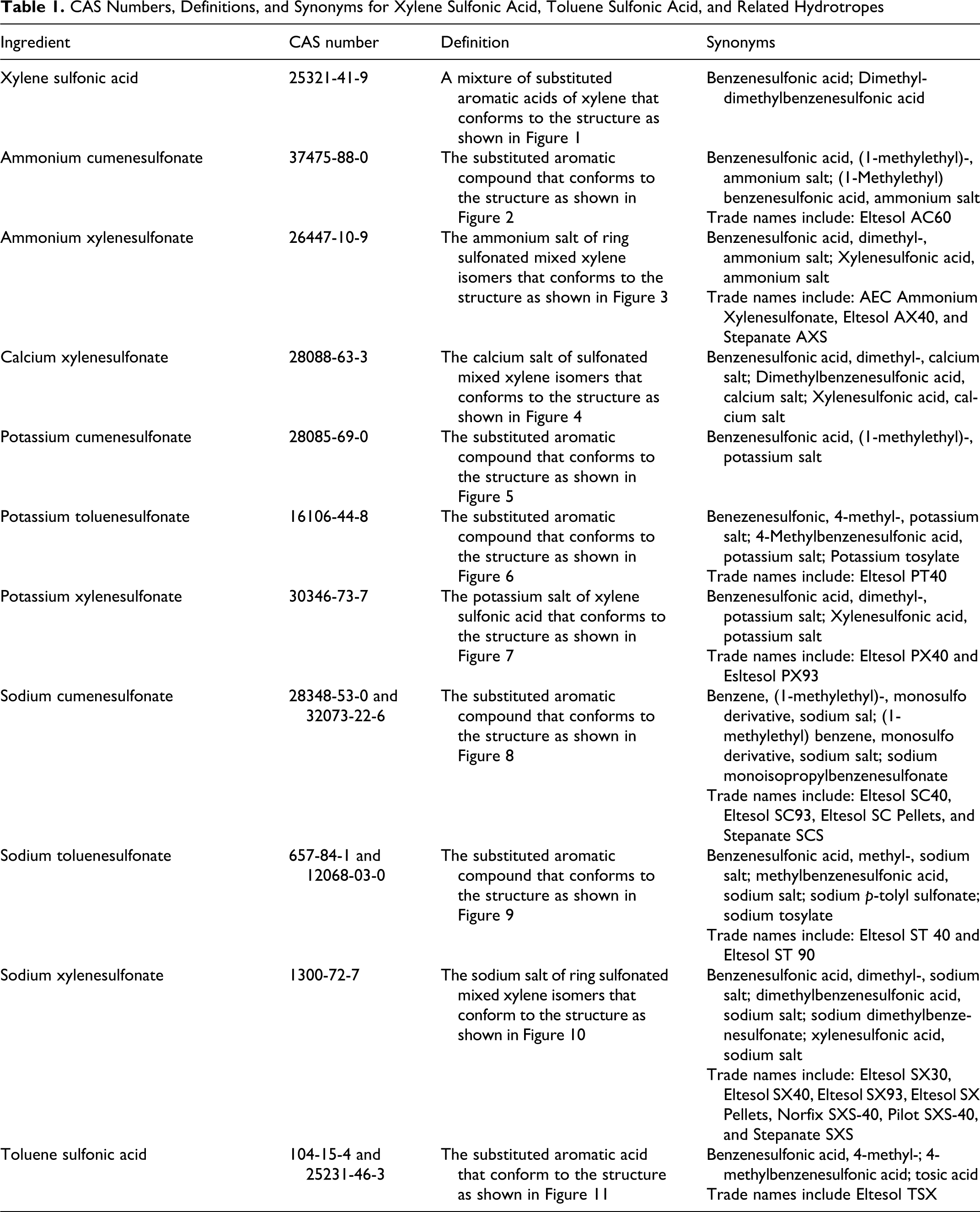
Xylene sulfonic acid, toluene sulfonic acid, and the group of alkyl aryl sulfonate hydrotropes are substituted aromatic acid, compound, and salt, respectively. Their CAS numbers and synonyms are provided in Table 1. Their structures are shown in Figures 1–11.
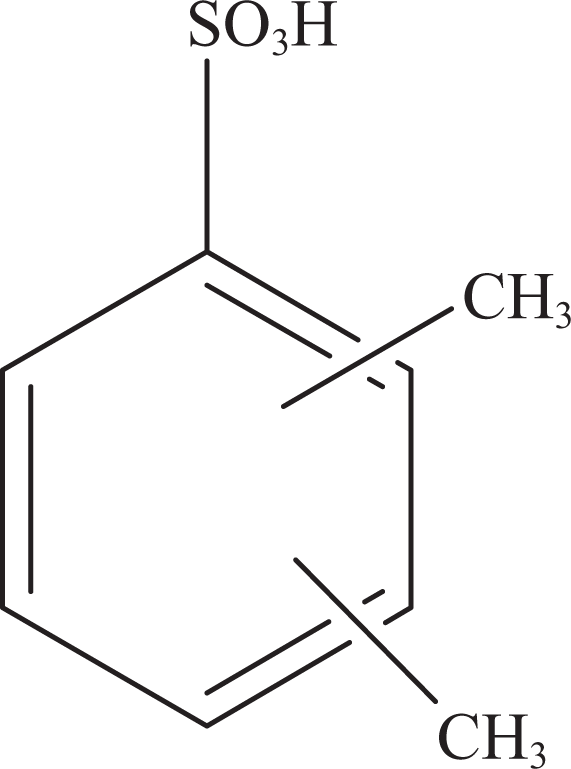
Xylene sulfonic acid.
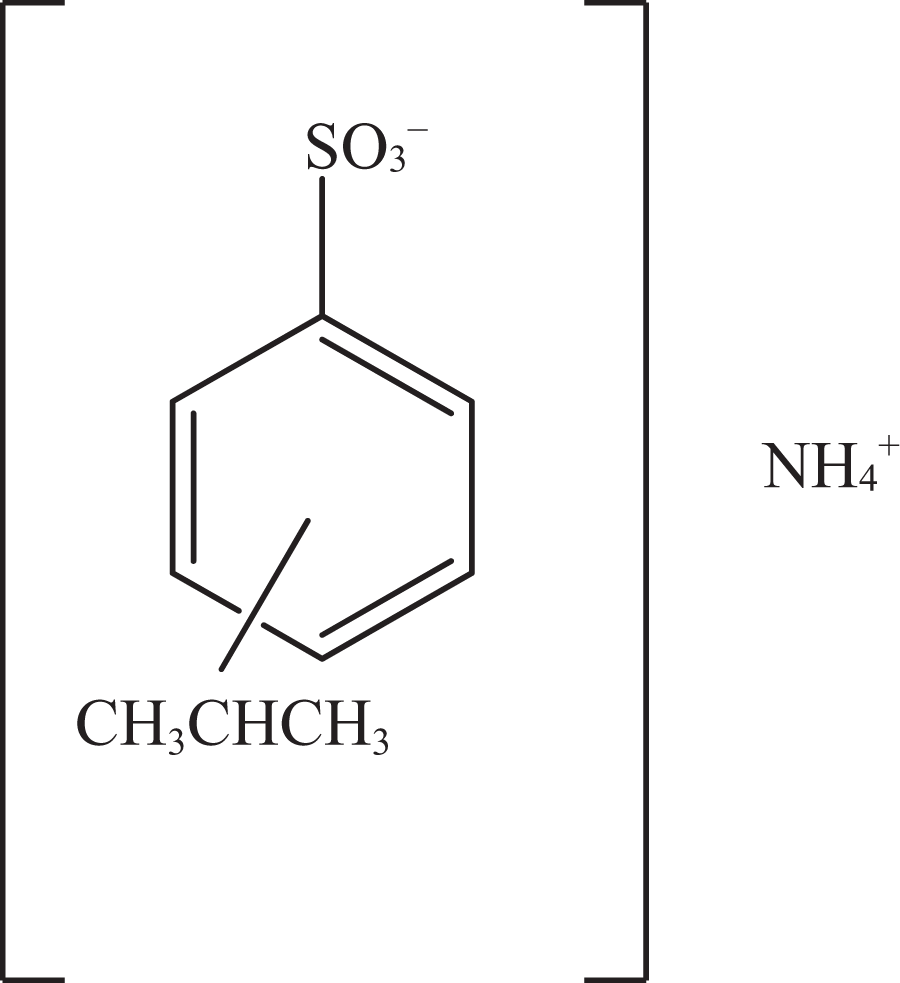
Ammonium cumenesulfonate.
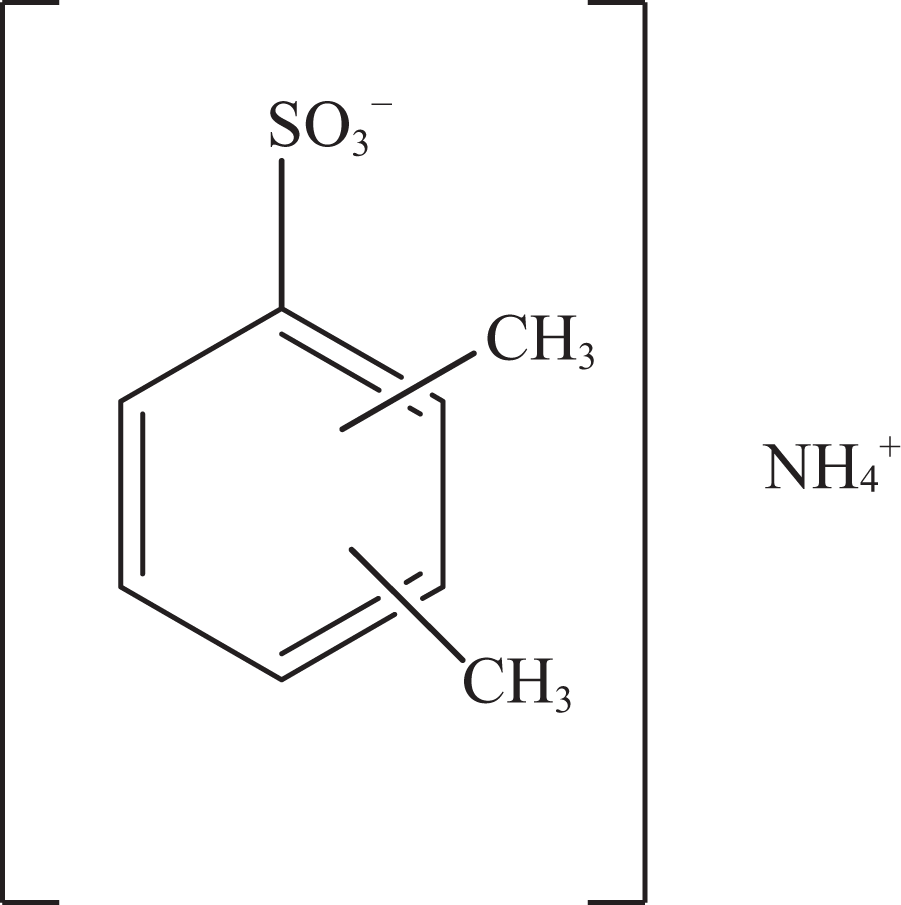
Ammonium xylenesulfonate.
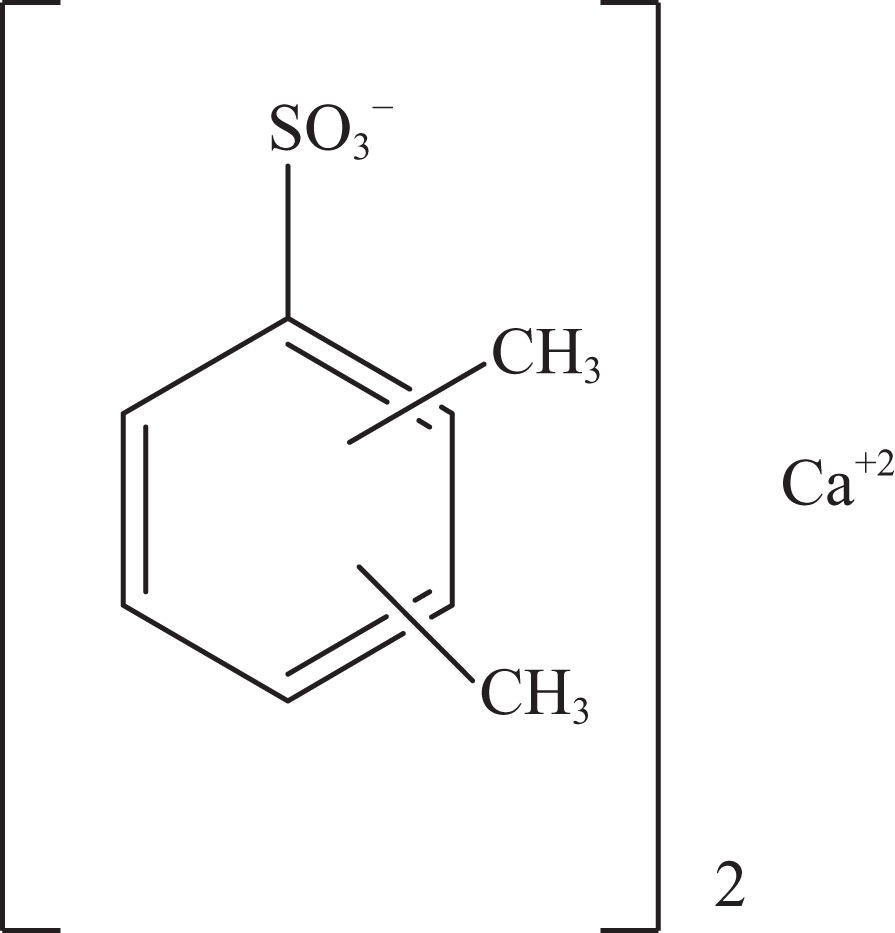
Calcium xylenesulfonate.
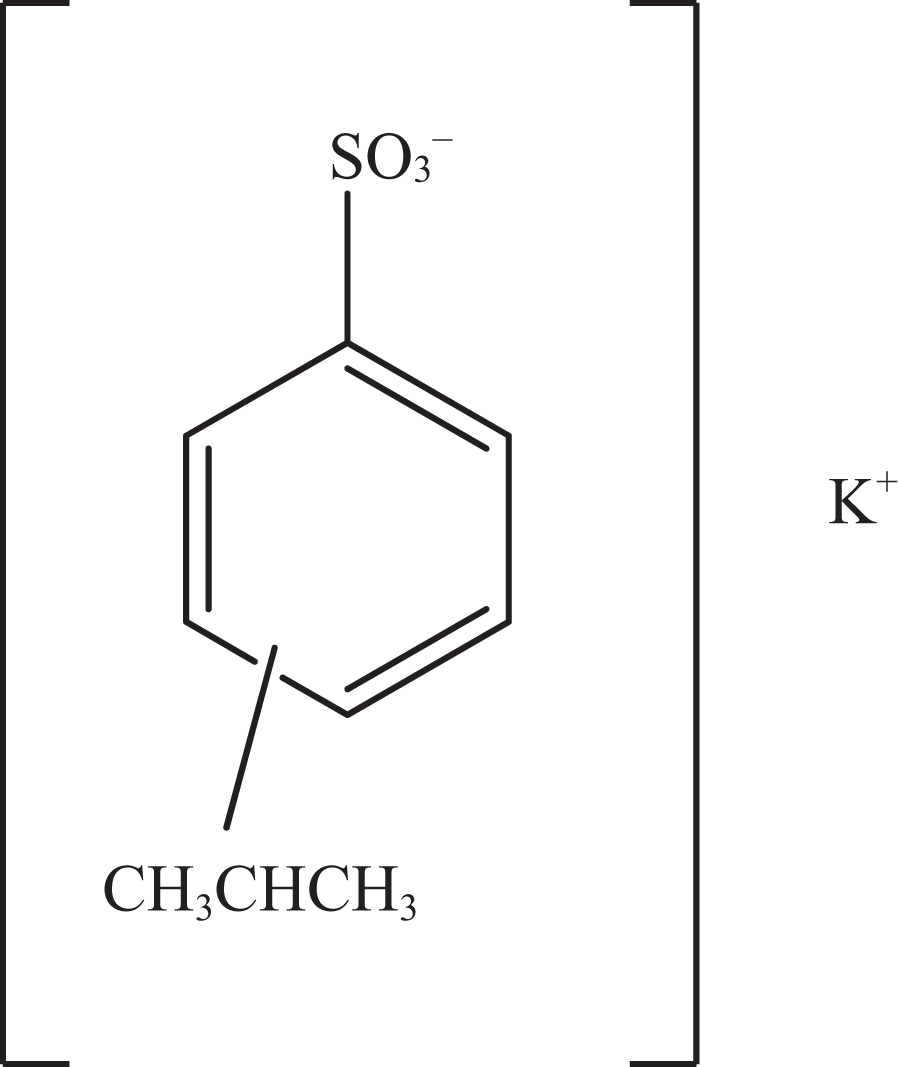
Potassium cumenesulfonate.
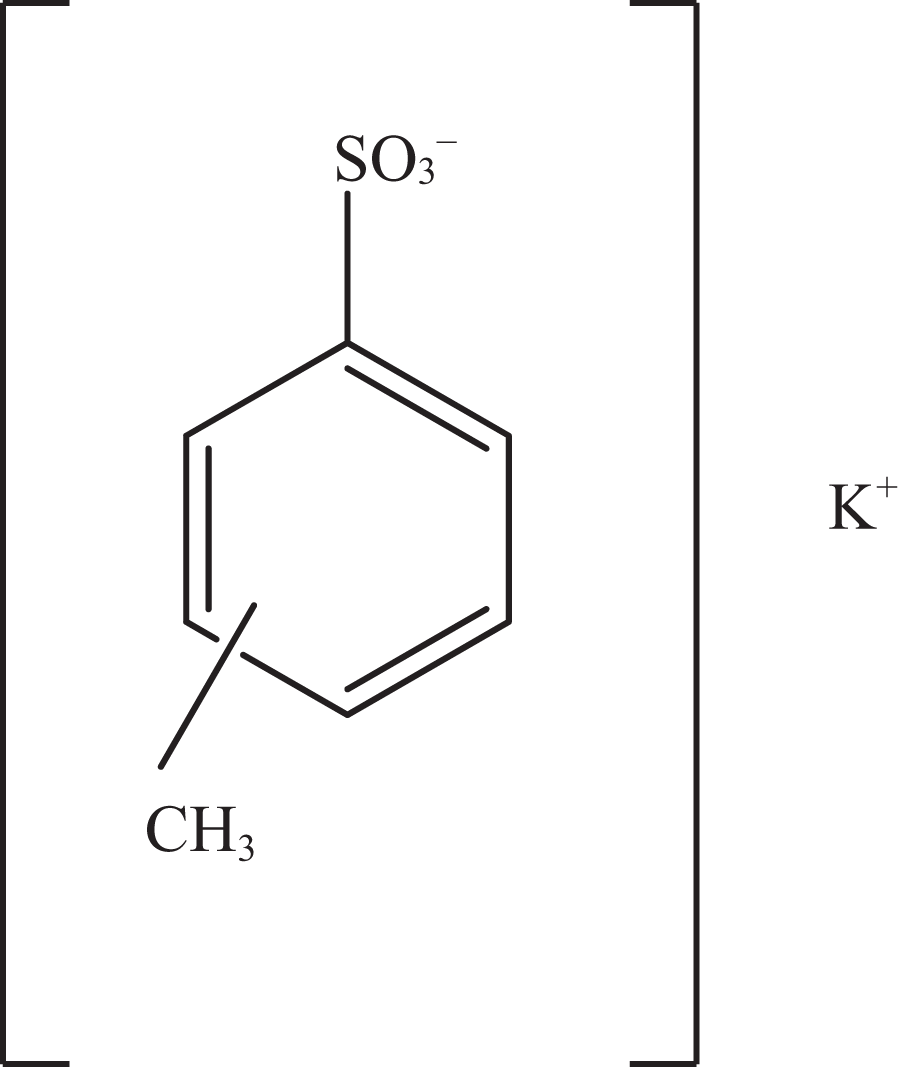
Potassium toluenesulfonate.
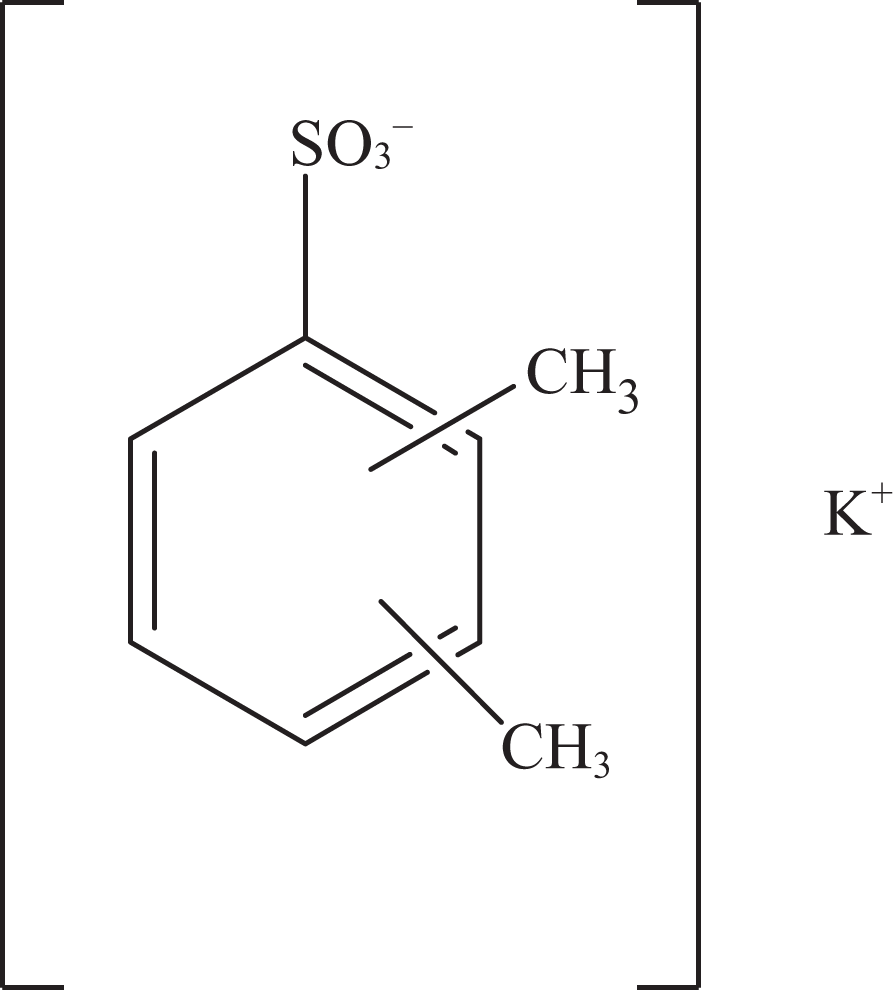
Potassium xylenesulfonate.
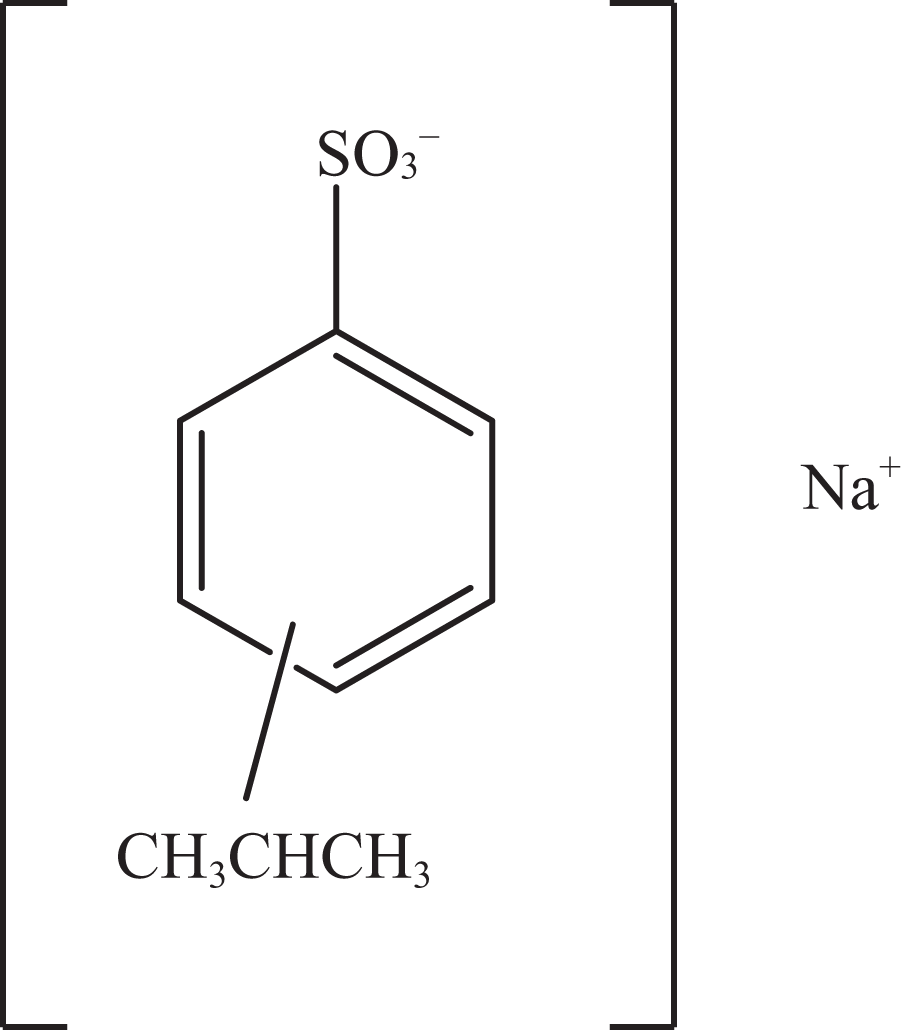
Sodium cumenesulfonate.
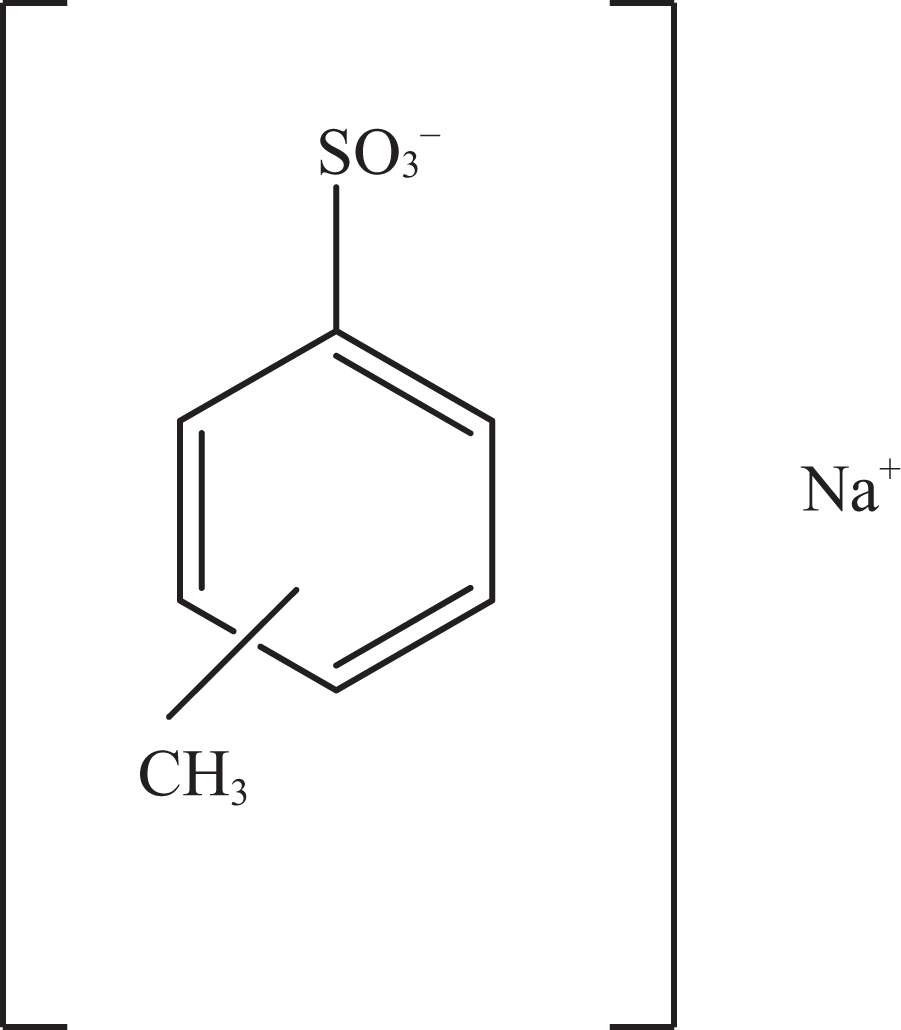
Sodium toluenesulfonate.
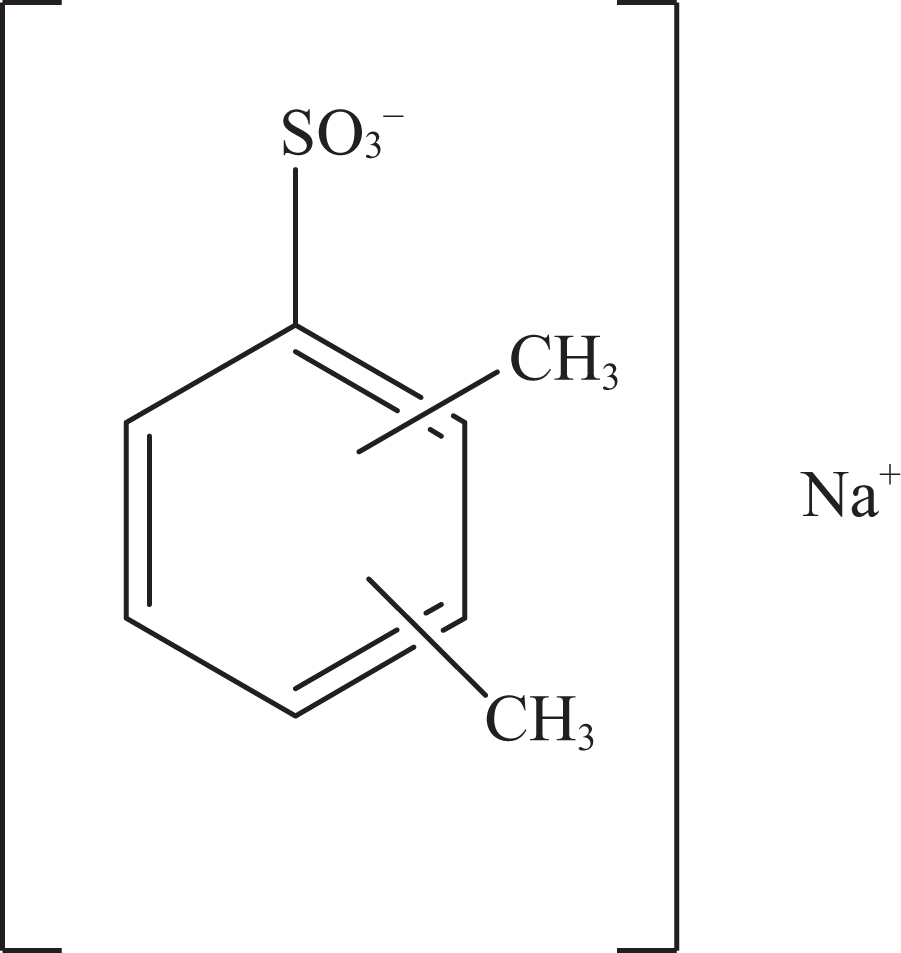
Sodium xylenesulfonate.
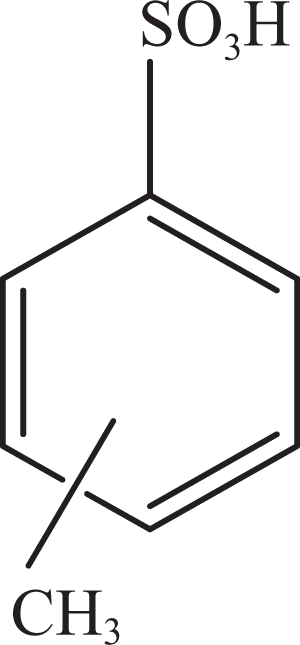
Toluene sulfonic acid.
CAS Numbers, Definitions, and Synonyms for Xylene Sulfonic Acid, Toluene Sulfonic Acid, and Related Hydrotropes
Hydrotropes
Compounds known as hydrotropes are described as amphiphilic substances composed of both hydrophilic and hydrophobic functional groups. 1 The hydrophobic part of the molecule is a benzene substituted apolar segment (ie, methyl [common name: toluene], dimethyl [common name: Xylene], or isopropyl [common name: cumene] apolar segment). The hydrophilic, polar segment is an anionic sulfonate group accompanied by a counter ion (eg, sodium and ammonium). This segment is a comparatively short side chain. Commercial toluene (and cumene) sulfonates consist of mixtures of 3 isomers (ortho-, meta-, and para-). Commercial xylene sulfonic acid consists of mixtures of 6 isomers (ortho,ortho; meta,meta; para,para; ortho,meta; ortho,para; and meta,para).
Hydrotropes are compounds that are used to increase the solubility of less soluble substances (ie, surfactants) in aqueous solution. Hydrotropes have structures somewhat similar to surfactants, but they are not themselves surfactants. The main property of the hydrotropes is related to the minimum hydrotropic concentration, which is defined as the concentration at which hydrotropes begin to aggregate. 2,3
Hydrotropes inhibit the formation of surfactant liquid crystalline phases by forming mixed micellar structures with surfactants. Since the hydrotrope hydrophilic heads are large and their hydrophobic groups are small, they tend to form spherical rather than lamellar or liquid crystalline structures, therefore inhibiting the formation of the latter. This destruction or inhibition of the liquid crystalline phase increases the solubility of the surfactant in the aqueous phase and the capacity of its micellar solution to solubilize material. 3
Physical and Chemical Properties
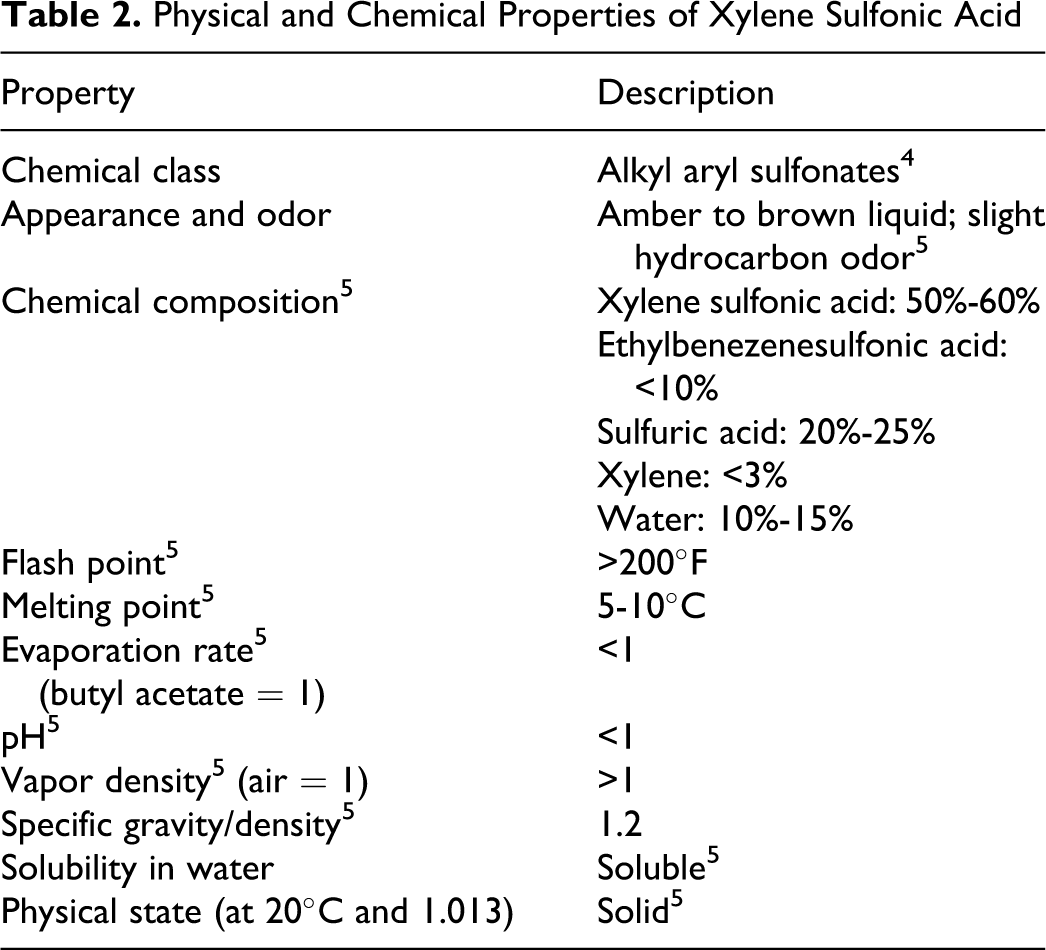
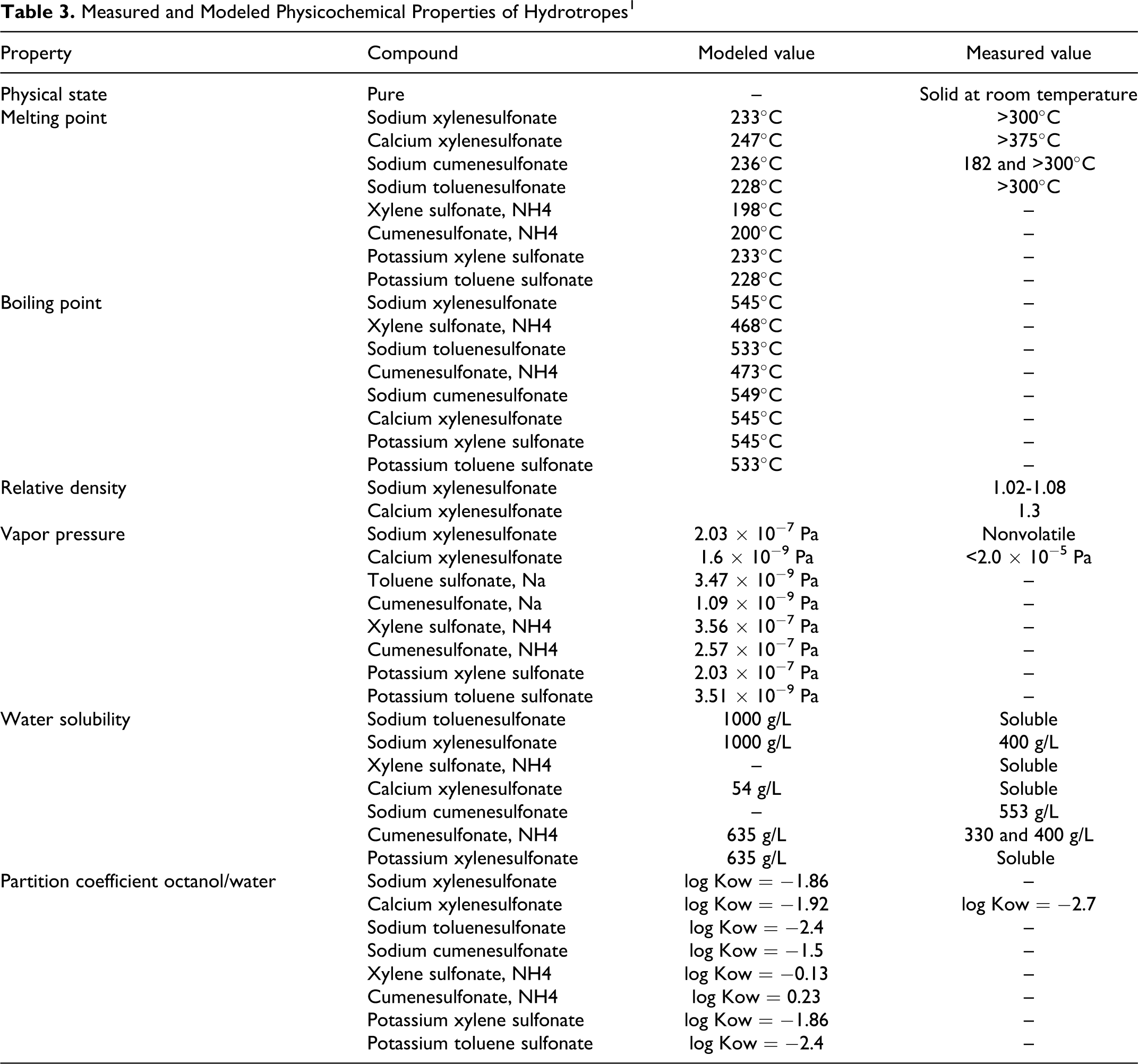
Table 2 presents physical and chemical properties of xylene sulfonic acid. Summary Information Data Set reported the molecular weight range of ammonium xylenesulfonate, calcium xylenesulfonate, potassium xylenesulfonate, potassium toluenesulfonate, sodium toluenesulfonate, sodium xylenesulfonate, and sodium cumenesulfonate to be 194 to 226. Measured and modeled physical and chemical properties of the hydrotropes are given in Table 3. 1
Physical and Chemical Properties of Xylene Sulfonic Acid
Measured and Modeled Physicochemical Properties of Hydrotropes 1
Method of Manufacture
Hydrotropes are produced by sulfonation of an aromatic hydrocarbon solvent (ie, toluene, xylene, or cumene). 1 The resulting aromatic sulfonic acid is neutralized using an appropriate base (eg, sodium hydroxide, ammonium hydroxide, potassium hydroxide, or calcium hydroxide) to produce the sulfonate or hydrotrope. The liquid product is produced in a closed system. According to the Human and Environmental Risk Assessment (HERA), hydrotropes are manufactured for industrial/professional and consumer use and are not used as intermediates/derivatives in further chemical manufacturing processes. 7
Analytical Methods
No data on analytical methods were found.
Impurities
According to SIDS, hydrotropes are produced and transported in either aqueous solutions, typically at a 30% to 60% level of activity. 1 In granular solid form, hydrotropes are typically at 90% to 95% level of activity, with remainder being sulfate salt and water.
Stability and Reactivity
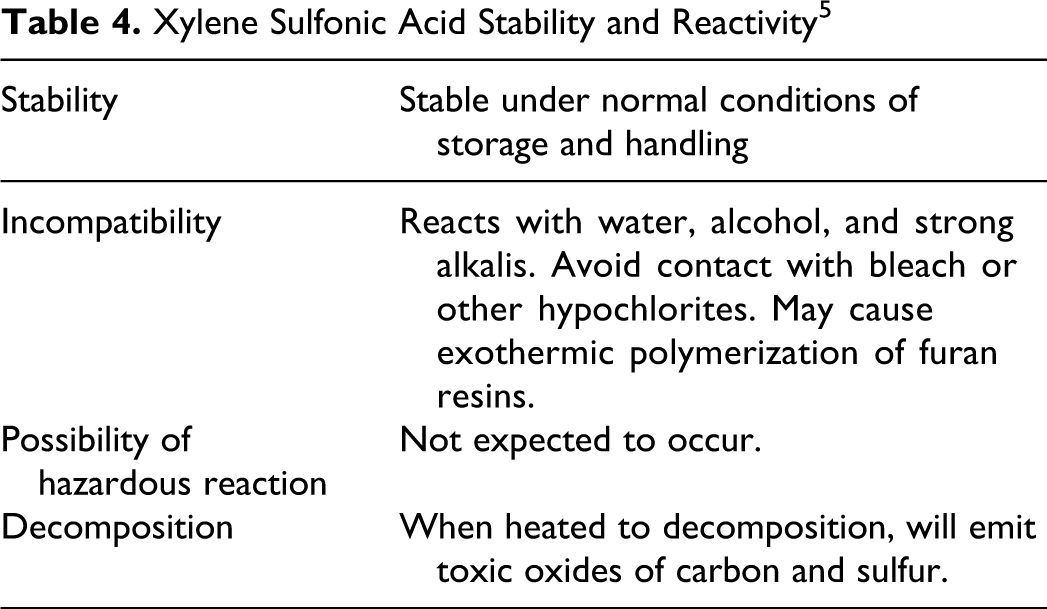
Table 4 includes information regarding the stability and reactivity of xylene sulfonic acid. Summary Information Data Set stated that hydrotrope salts are expected to dissociate completely in water and that hydrotropes are known to be readily biodegradable. 1
Xylene Sulfonic Acid Stability and Reactivity 5
Use
Cosmetic
According to the information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP), there were 75 uses of ammonium xylenesulfonate in cosmetic products, while 74 of them in shampoos. 6 Similarly, 75 uses of sodium xylenesulfonate were reported, of which 58 of were in shampoos. No uses were reported to FDA for the remaining ingredients.
An industry survey was conducted by the Personal Care Products Council (Council) in 2009. 8 Ammonium xylenesulfonate was used at concentrations from 0.008% to 3%, sodium xylenesulfonate was used at concentrations from 0.1% to 15%, and sodium cumenesulfonate was reportedly used at a concentration of 3% in a shampoo product. There were no reportedly used concentrations for the remaining ingredients included in this report. Table 5 presents the currently available product use and concentration of use information reported for ammonium xylenesulfonate, sodium xylenesulfonate, and sodium cumenesulfonate.
Frequency of Use and Concentration of Ammonium Xylenesulfonate, Sodium Xylenesulfonate, and Sodium Cumenesulfonate
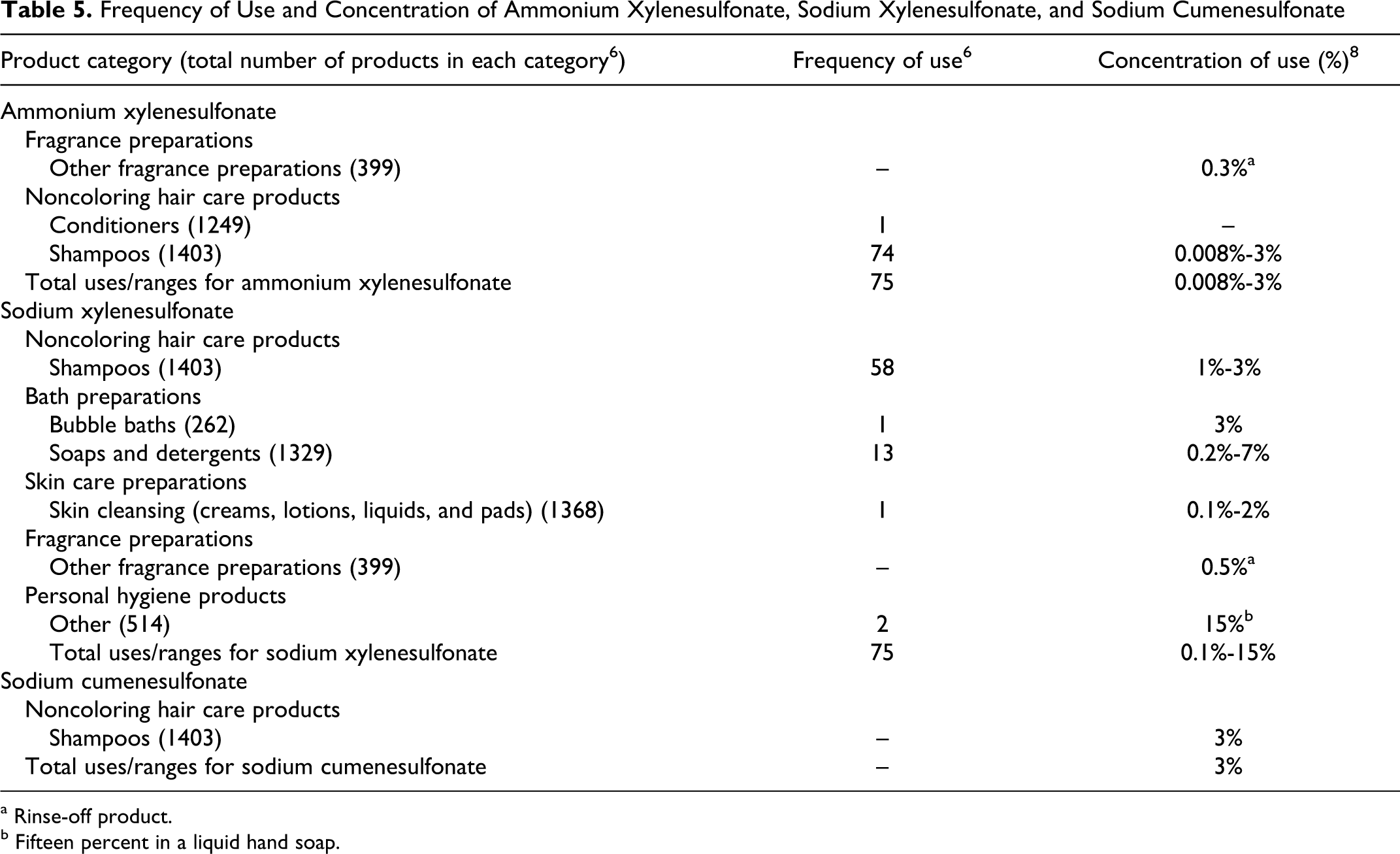
a Rinse-off product.
b Fifteen percent in a liquid hand soap.
The Cosmetic Ingredient Review (CIR) Expert Panel recognized that the product category of use and concentration of use data are incomplete but judged that the information available on the types of products and formulation concentrations was sufficient to define an overall pattern of use. Given the well-understood function of these ingredients in cosmetic products, it was considered that the available data were adequate to anticipate current and future use patterns.
These ingredients function in cosmetics as surfactants, hydrotropes, according to the International Cosmetic Ingredient Dictionary and Handbook. 4 Hydrotropes are used as coupling agents to solubilize the water insoluble and often incompatible functional ingredients of personal care products (body washes, shampoos, hair conditioners, liquid face, and hand soaps). 1 They function to stabilize solutions, modify viscosity and cloud-point, limit low temperature phase separation, and reduce foam formation. Hydrotropes are used as aqueous solutions (30%-60% active substance) or as granular solids containing 90% to 95% active substance.
Noncosmetic
Sodium xylenesulfonate is a component in a variety of liquid household detergents, where it can constitute up to 10% of the total solution. 9 A major use of xylene sulfonic acid is in household/professional cleaning products including liquid and powder laundry detergents, hand dishwashing liquid detergents, machine dishwashing rinse aids, hard surface cleaners, toilet treatments, solvent hand cleaners, carpet cleaners, and optical brightener products. 1 It is estimated that 60% to 70% of the total tonnage is used in dishwashing liquids. There are also some relatively small volume, commercial/professional uses in liquid sulfur textile dyes, acidic recirculation cleaners, wetting agent for tanning, enzymatic recirculation cleaner for dairy and food processing, coolant system conditioner, car wash detergents, cleaners and degreasers, vinyl plastic rubber restorer, and floor strippers.
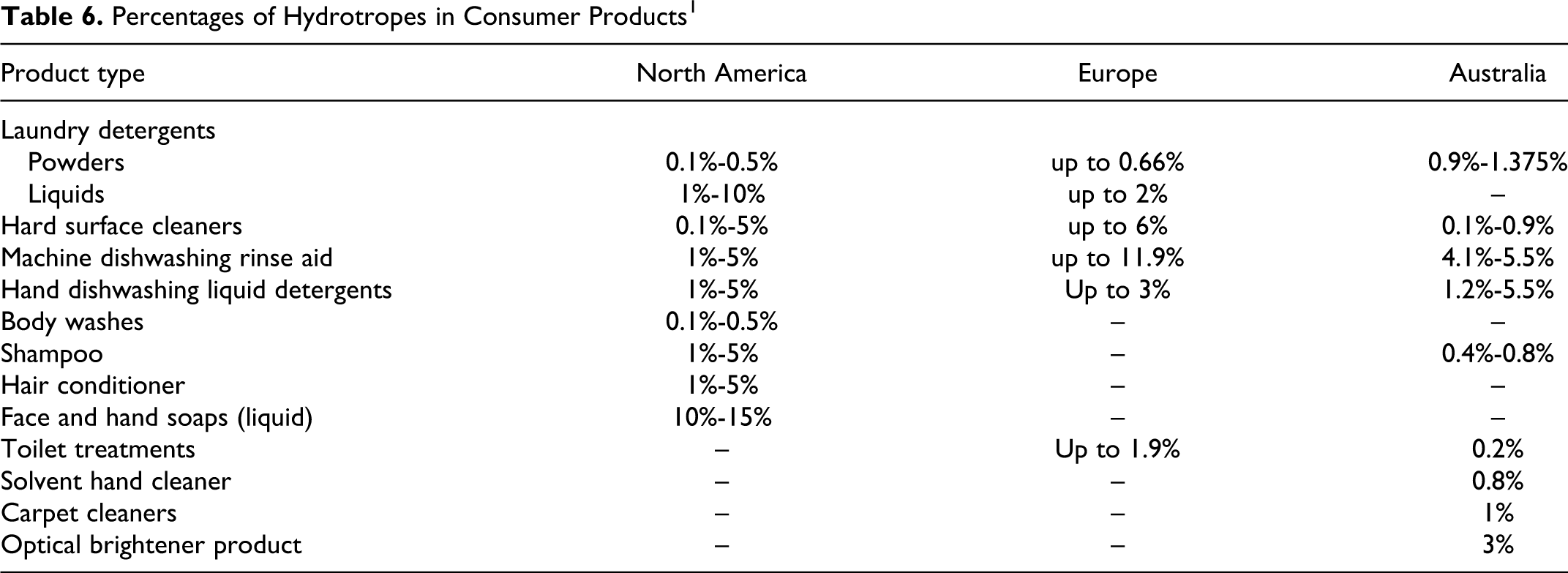
Table 6 lists the percentages of hydrotropes that occur in various types of consumer laundry/cleaning and personal care products in North America, Europe, and Australia. 1
Percentages of Hydrotropes in Consumer Products 1
General Biology
Absorption, Distribution, Metabolism, and Excretion
Using the physical and chemical properties of the hydrotropes and available toxicological data, a general qualitative analysis of absorption was made by SIDS. 1 The molecular weight of these hydrotropes ranges from 194 to 226. Water solubility and log P value of 553 g/L and −2.7, respectively, were used. Based on these properties and the available toxicological studies, significant absorption would be expected to occur following oral administration while absorption following dermal application would be expected to be limited. Regarding the general qualitative analysis of physical and chemical properties used to evaluate absorption, distribution, metabolism, and excretion, molecular weights below 500 were considered, by the authors, to be favorable for absorption from the gastrointestinal tract. Additionally, absorption of very hydrophilic substances, such as the hydrotropes, can occur by passive diffusion and if the molecular weight is low (less than 200), the substance may pass through aqueous pores. In contrast to oral absorption, a molecular weight less than 100 favors dermal absorption.
Animal Toxicology
Acute Toxicity
Intraperitoneal
Xylene sulfonic acid has an LD50 of 500 mg/kg in mice (intraperitoneal [IP]). 5
Oral
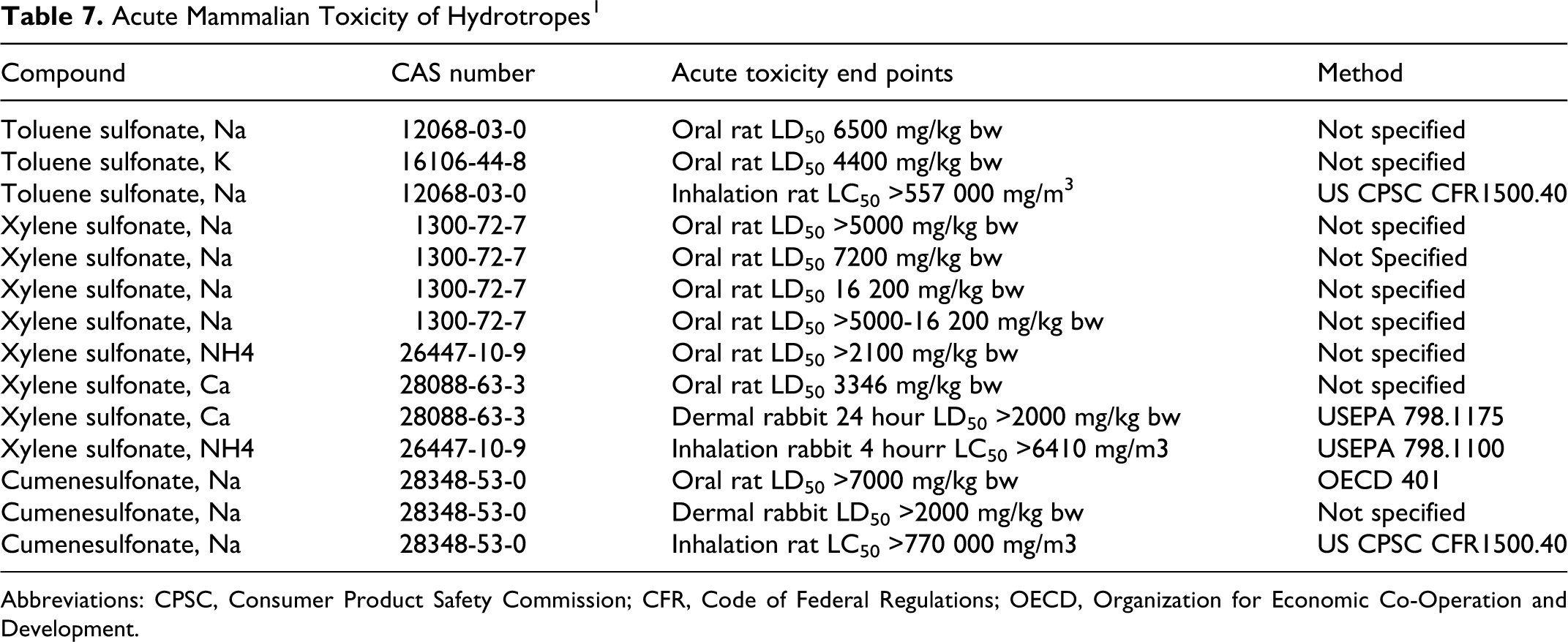
Table 7 provides the available acute toxicity results for toluene, xylene, and cumenesulfonates and their various salts. 1 Overall, these chemicals are not acute oral toxicants. Signs observed at high dose levels in acute oral toxicity studies included decreased activity, weakness, prostration, increased salivation, diarrhea, ptosis, and anogenital staining. Necropsy findings reported in these similar studies included slight pulmonary inflammation, gastrointestinal inflammation or hemorrhage, mild liver changes, congestion (liver, kidneys, adrenal glands, and gastrointestinal tract), and redness of stomach mucosa in animals that died. Slight-to-moderate congestion of the adrenal glands was observed in animals that survived; all other observations were within normal limits.
Acute Mammalian Toxicity of Hydrotropes 1
Abbreviations: CPSC, Consumer Product Safety Commission; CFR, Code of Federal Regulations; OECD, Organization for Economic Co-Operation and Development.
Dermal
Clinical signs observed in acute dermal exposure included erythema with additional desquamation. 1 At necropsy, findings reported were focal or multifocal red discoloration and desquamation of the treated skin.
Inhalation
No effects were reported following inhalation exposures in rats and rabbits (Table 7). 1
Short-Term Toxicity
Dermal
National Toxicology Program (NTP) reported that F344/N rats (number, sex, and dose not specified) were dermally treated with 300 µL of 0, 5, 15, 44, 133, or 400 mg/mL sodium xylenesulfonate in distilled water 5 d/week for 17 days. 9 All rats survived to the end of the study. Final mean body weights and body weight gains of dosed rats were similar to those in the control group. Dermal applications of 300 µL of 5, 15, 44, 133, and 400 mg/mL delivered average daily doses of approximately 10, 30, 90, 260, and 800 mg sodium xylenesulfonate per kg body weight (bw) to males and 13, 40, 120, 330, and 1030 mg/kg to females. Clinical findings generally involved the skin of dosed animals and included tan or brown skin discoloration and crusty white deposits (presumed to be dried chemical) at the application site. Neither of these observations was considered significant. The relative liver weights of 133 and 400 mg/kg male and female rats, respectively, were significantly greater than those of the control groups, but the absolute liver weights were not increased and the biological significance of the relative differences in liver weight was unclear. No chemical-related lesions were observed in any of the animals.
In another 17-day study, B6C3F1 mice (5/sex/group) groups were dermally treated with 100 µL of 0, 5, 15, 44, 133, or 400 mg/mL sodium xylenesulfonate in distilled water, 5 d/week for 17 days. 9 All the mice survived to the end of the study. Final mean body weights and body weight gains of dosed mice were similar to those of the controls. Dermal applications of 5, 15, 44, 133, and 400 mg/mL delivered average daily doses of approximately 20, 60, 190, 540, and 1600 mg sodium xylenesulfonate per kilogram bodyweight to males and 26, 80, 220, 680, and 2000 mg/kg to females. Clinical findings included crusty white deposits (presumed to be dried chemical) at the site of application in two 133 mg/mL males and in all 400 mg/mL males and females. The relative liver weights of 15 and 44 mg/kg male rats and 400 mg/kg males and females were significantly greater than those of the control groups, but the biological significance of these differences was unclear. In males and females, the few lesions observed grossly and microscopically were generally attributed to repeated clipping and were not considered chemical related.
Oral
Rats and mice (species, number, and sex not provided) were given 0%, 0.25%, 0.5%, 1%, 2%, and 4% sodium xylenesulfonate in the diet in a 2-week range finding study. 1 Body weight and food consumption were recorded, and the animals were observed for clinical signs and mortality. There were no clinical signs of toxicity or mortality at any of the doses in mice. Body weight gain was higher than the controls at the 0.25% and 0.5% levels in both sexes of mice. At 1%, 2%, and 4% levels, the body weight gain was reduced by 2%, 2%, and 6% of control values, respectively. In rats, deaths occurred at 2% (2 animals) and 4% (4 animals) in males, and in females, one each at the 0.5%, 1%, 2%, and 4% doses. Body weight gains were decreased by 3% and 19% of the control value at the 1% and 2% dose levels in males, respectively. At the 4% level in females, body weight gain was decreased by 7% of the control values. Palatability issues may have been responsible for the animals developing rough coats and the subsequent weight loss and death of some of the animals.
A second 2-week study was conducted in rats and mice (strain, numbers, and sex not provided) to determine if the mortalities observed in the first 14-day study were reproducible and related to the toxicity of sodium xylenesulfonate. 1 The animals were given 0%, 1%, 2%, and 4% sodium xylenesulfonate in the diet. No mortality was observed at any dose level in either species. Decreased body weight gains were reported at 1%, 2%, and 4% doses in both sexes (5%, 4%, and 17% decreases compared to control values in males, and 2%, 2%, and 5% decreases compared to control values in females, respectively), however, there was no dose–response relationship between test material concentration and body weight gain. Palatability appeared to be an issue in the 4% group; animals were observed scratching feed from the feeders during the last 8 days of the study at this level.
Subchronic Toxicity
Dermal
In a 14-week study by NTP, F344 rats (10/sex/group) were dermally treated with 300 µL of 0, 5, 15, 44, 133, or 400 mg/mL sodium xylenesulfonate in 50% ethanol. 9 These concentrations corresponded to average daily doses of approximately 6, 20, 60, 170, and 500 mg sodium xylenesulfonate per kilogram bodyweight in males and 10, 30, 90, 260, and 800 mg/kg in females. All rats survived to the end of the study. Final mean body weights and body weight gains of dosed male and female rats were similar to those in the control groups. The only notable finding was brown discoloration of the skin at the application site of dosed animals. There were no significant differences in liver weights in female rats. Minimal hyperplasia of the epidermis at the site of application occurred in both male and female rats in the control group as well as in most dosed animals. The incidence of the epidermal hyperplasia in 400 mg/mL males was possibly chemical related.
B6C3F1 mice (10/sex/group) were dermally treated with 100 µL of 0, 5, 15, 44, 133, or 400 mg/mL sodium xylenesulfonate in 50% ethanol for 14 weeks. 9 These concentrations correspond to average daily doses of approximately 17, 40, 140, 440, and 1300 mg sodium xylenesulfonate per kilogram bodyweight in males and 20, 60, 170, 530, and 1630 mg/kg in females. There were no chemical-related deaths and no clinical findings. The mean body weight gain of the 400 mg/mL males was significantly greater than that of the control group. Epidermal hyperplasia occurred in one 44 mg/mL female, two 133 mg/mL males, five 400 mg/mL males, and four 400 mg/mL females. Hyperplasia of the epidermis in 400 mg/mL males and females was probably related to chemical administration. Chronic inflammation of the skin occurred primarily in the control groups of males and females. These lesions consisted of mononuclear inflammatory cells in the dermis. 9
Oral
According to SIDS, 3 subchronic 90-day feeding studies in rats were conducted, 2 with sodium xylenesulfonate and the other with sodium cumenesulfonate. One of the studies also included mice. 1 In the first study, Wistar rats (15/sex/group) were given purified sodium xylenesulfonate at 0%, 0.2%, 1.0%, and 5.0% in the diet. Mean administered doses were 0, 140, 710, and 3800 mg/kg bw for males and 0, 160, 820, and 4400 mg/kg bw for females. The purity of the test substance was stated as at least 93%. Therefore, the doses based on active ingredient (AI) were 130, 660, and 3534 mg AI/kg bw for males and 149, 763, and 4092 mg AI/kg bw for females. A number of organs were examined for histopathological changes. No treatment-related effects other than some sporadic clinical chemistry and hematology changes, which were observed in males at the highest dose (3534 mg AI/kg bw), were observed. A decrease in relative spleen weights in females, along with some clinical chemistry and hematology changes, was observed at the highest dose (4092 mg AI/kg bw). No treatment-related histopathological changes were reported in the spleen or in any other organ examined. The NOAEL from this study is 1% or 763 mg AI/kg bw in females and 5% or 3534 mg AI/kg bw in males.
In the second study, Fischer rats (10/sex/group) and B6C3F1 mice (number/sex not provided) were given sodium xylenesulfonate (purity unknown) at 0%, 0.125%, 0.25%, 0.5%, 1%, and 2% in the diet over a 91-day period. 1 These dietary levels equate to 0, 152, 305, 610, 1220, and 2439 mg/kg bw daily doses for male mice; 0, 154, 308, 617, 1234, and 2467 mg/kg bw for female mice; 0, 89, 179, 357, 715, and 1429 mg/kg bw for male rats; and 0, 98, 195, 390, 781, and 1561 mg/kg bw for female rats. Body weights and feed consumption were recorded. Animals were observed for clinical signs and mortality. No hematology or clinical chemistry tests were done. Gross pathology was recorded when observed and histopathology was performed on all controls and animals exposed to the high dose. There were no significant dose-related treatment effects on feed consumption or body weight in any group for either species. There were also no treatment-related gross or microscopic lesions in either rats or mice. The NOAELs were 2439 and 2467 mg/kg bw/d for male and female mice, respectively, and 1429 and 1561 mg/kg bw/d for male and female rats, respectively.
In the third study, CD rats (20/sex/group) were treated with sodium cumenesulfonate at 0%, 0.005%, 0.05%, and 0.5% in the diet. 1 Mean administered daily doses were 0, 2.6, 26, and 270 mg/kg bw for males and 0, 3.6, 36, and 375 mg/kg bw for females. The test substance was 42.3% pure. Thus, these dietary levels equate to 1.1, 11, and 114 mg AI/kg bw for males and 1.5, 15, and 159 mg AI/kg bw for females, respectively.
End points examined were mortality, body and organ weights, feed consumption, hematology, and histopathology. No treatment-related effects were observed in males at the highest dose (114 mg AI/kg bw). A decrease in body weight gains was reported in females (4%, 5%, and 12% compared to controls at 1.5, 15, and 159 mg AI/kg bw, respectively). The study authors stated that this decrease in body weight gains was within the established ranges for animals of this species and age and was therefore not considered an adverse effect. The feed efficiency of the high-dose females was significantly higher than the controls. The decrease in body weight gains in the high-dose females was not associated with histopathological changes or any other effects. Histopathological studies indicated that severe tubule atrophy and degeneration had occurred in the testes of 1 animal treated at the highest dose. A mild lesion was seen in 1 male treated at 15 mg AI/kg bw. No lesions attributable to the treatment were evident. Prostatitis was observed in 2 animals in the lowest dose group, but no dose–response effect was noted. There was also a slight increase in the number of animals with pulmonary lesions common to the rat (ie, perivasculitis and peribronchitis); however, there was no difference in the incidence between the control and treated rats. Of the 20 rats that were used for the study, 11 had pneumonia (no differentiation was made between the controls and treated). The NOAEL for sodium cumenesulfonate was 114 mg AI/kg bw for males and 159 mg AI/kg bw for females.
Chronic Toxicity
Dermal
National Toxicology Program 9 reported on a 2-year study in rats. F344 rats (50/sex/group) were treated with 0, 60, 120, or 240 mg sodium xylenesulfonate per kilogram bodyweight in 50% ethanol daily for 104 weeks. In males, there were no treatment-related effects noted. In females, irritation at the application site was observed in 1, 4, and 2 females with 0, 120, and 240 mg/kg, respectively. There were no neoplasms at any site (including the skin) that were considered treatment related. Low incidences of hyperplasia of the epidermis at the application site occurred in males in the 60, 120, and 240 mg/kg groups. Low (number not provided) incidences of hyperplasia of the epidermis at the application site also occurred in females in the 120 and 240 mg/kg groups and they occurred with a significant positive trend. Low incidences of hyperplasia of the sebaceous gland occurred in control and 60 mg/kg males and in control, 120 mg/kg, and 240 mg/kg females.
B6C3F1 mice (50/sex/dose) were dermally treated with 0, 182, 364, or 727 mg sodium xylenesulfonate per kilogram bodyweight in 50% ethanol for 104 to 105 weeks. With the exception of irritation at the application site in one 364 mg/kg female, there were no clinical findings related to sodium xylenesulfonate administration. 9
Ocular and/or Mucosal Irritation
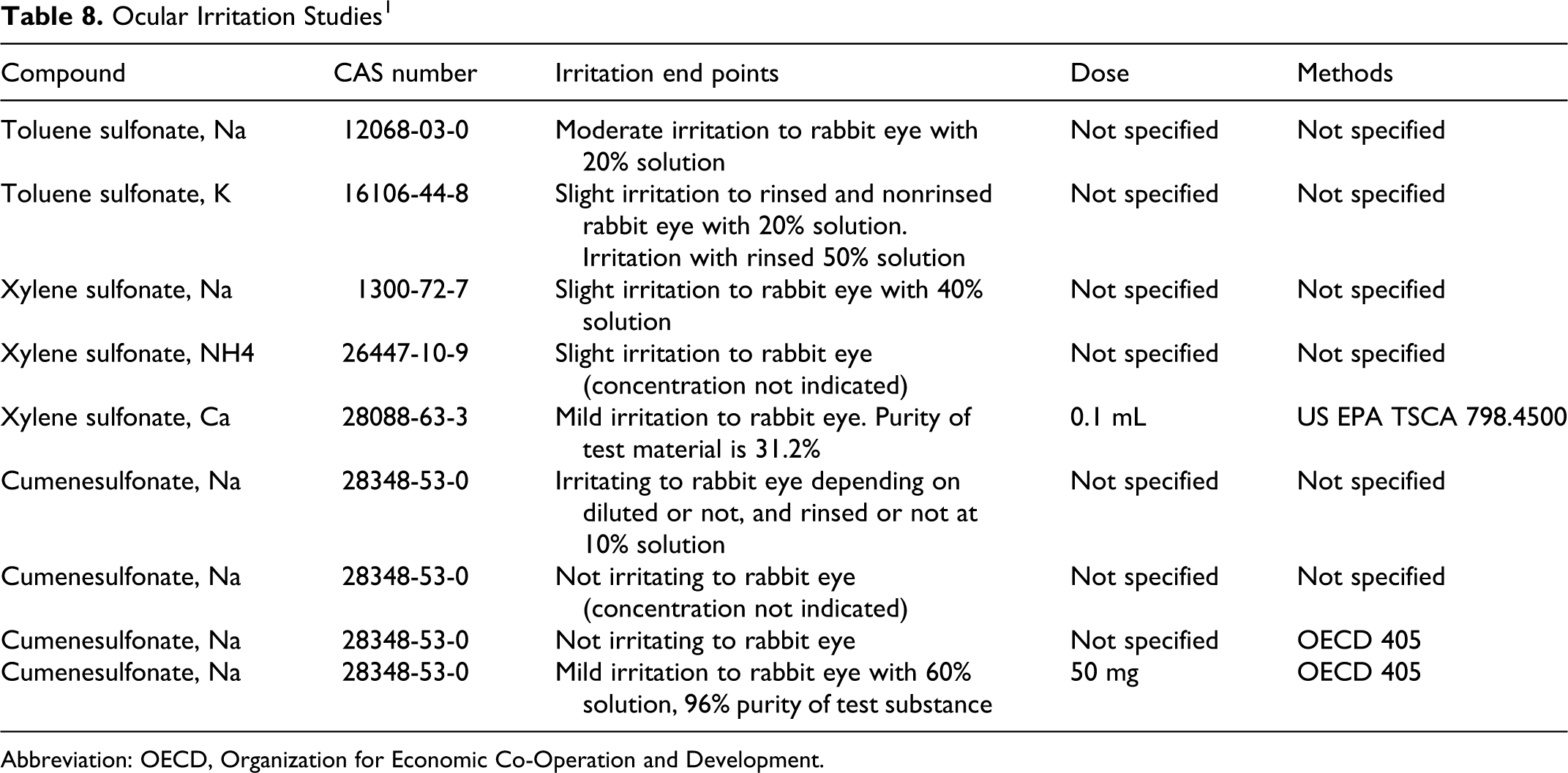
Table 8 summarizes eye irritation studies for sodium toluenesulfonate, potassium toluenesulfonate, sodium xylenesulfonate, ammonium xylenesulfonate, calcium xylenesulfonate, and sodium cumenesulfonate. 1 At a variety of concentrations and exposure conditions, slight-to-moderate irritation was observed following exposure to the hydrotropes as presented in the table. Doses were not provided for some of these studies.
Ocular Irritation Studies 1
Abbreviation: OECD, Organization for Economic Co-Operation and Development.
Dermal Irritation
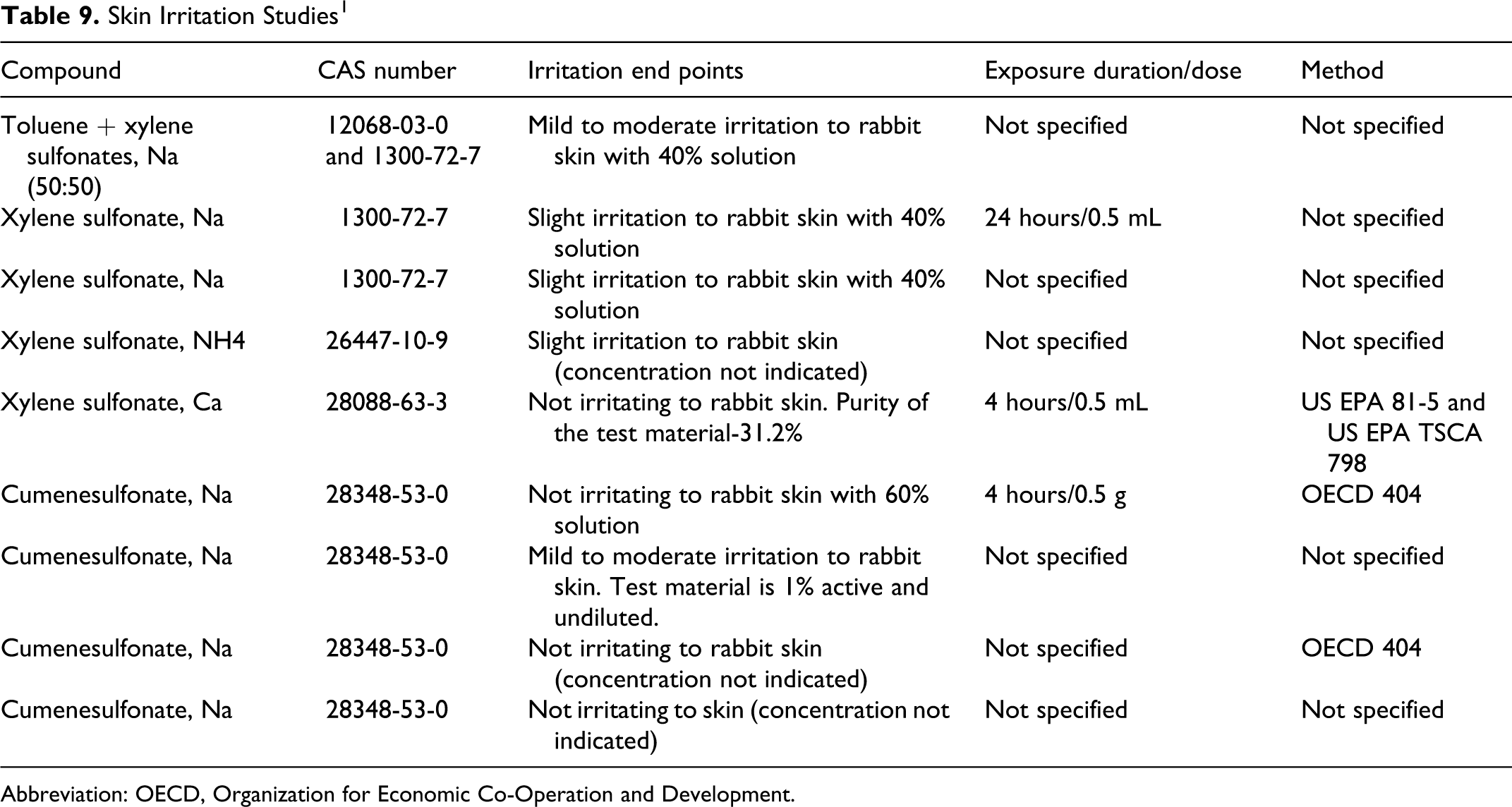
Table 9 lists skin irritation studies for sodium toluenesulfonate, potassium toluenesulfonate, sodium xylenesulfonate, ammonium xylenesulfonate, calcium xylenesulfonate, and sodium cumenesulfonate. 1 In dermal irritation studies, mild-to-moderate skin irritation was observed in rabbits treated dermally with solutions containing xylene sulfonates, toluene sulfonates, or cumenesulfonates in concentrations ranging from 1% to 60%. Doses were not provided for some of these studies.
Skin Irritation Studies 1
Abbreviation: OECD, Organization for Economic Co-Operation and Development.
Dermal Sensitization
As reported by SIDS, a guinea pig study (number of animals not specified) reported no evidence of skin sensitization following dermal, occlusive exposure to a 42.8% solution (deionized water) of sodium toluenesulfonate. 1 No irritation was observed during the study (neither at induction nor at challenge). Under the conditions of this study, the authors concluded that there was no indication of skin sensitization potential for hydrotropes.
Phototoxicity
No data on phototoxicity were available.
Reproductive and Developmental Toxicity
Calcium xylenesulfonate (31% AI) was administered via gavage to female rats (30 per group; species not specified) at 0, 150, 1500, or 3000 mg/kg bw in water on days 6 to 15 of gestation. 1 Clinical signs were noted daily from day 6 to 20. Body weight gain and feed consumption were recorded on day 0, 6, 9, 12, 16, and 20. All females were macroscopically examined on day 20 (or on day of death). The uteri were removed, weighed, and examined for the number of implantation sites and number and location of fetuses and resorptions. Corpora lutea were counted. Fetuses were inspected on total number, sex, weight, and external, visceral (one half), and skeletal (one half) defects. One animal died during the treatment dose (mid-dose). No treatment-related effects were observed. An increase in feed intake observed at the highest dose was considered within ranges of biological variation for this species. The NOAEL for maternal and fetal toxicity was the highest dose tested, 3000 mg/kg bw/d, which corresponds to 936 mg AI/kg bw/d.
Genotoxicity
In Vitro
The following in vitro genotoxicity studies of xylene sulfonates were reported by SIDS: 1 sodium xylenesulfonate, calcium xylenesulfonate, and sodium cumenesulfonate were found nonmutagenic, with or without metabolic activation when tested at concentrations up to 10 000, 5000, and 2000 μg AI/plate, respectively, in TA98, TA100, TA1535, TA1537, and TA1538. Cytotoxicity data were not reported. There was no mention of the negative controls. Positive controls for sodium xylenesulfonate and calcium xylenesulfonate were reported to give results that confirmed the validity of the test.
Technical grade (65% AI) sodium xylenesulfonate was tested for mutagenicity potential in L5178Y mouse lymphoma cells up to 5000 μg/mL with and without metabolic activation using supplemented Fischer medium vehicle and at 2500 μg/mL without metabolic activation using DMSO vehicle. Test concentrations were reported to be selected based on cytotoxicity. There was no mutagenic activity without metabolic activation and an equivocal result was reported with activation. Positive results were seen at the highest doses, where cytotoxicity was also reported.
Technical grade (65% AI) sodium xylenesulfonate (at 500 to 5000 μg/mL) was tested in Chinese hamster ovary cells with and without metabolic activation. Cytotoxicity was reported at 2513 to 5000 μg/mL without metabolic activation. No clastogenic activity was recorded with metabolic activation. A significant increase in SCEs was observed without metabolic activity, but only at dose levels that were reported to produce cell cycle delay. Positive controls produced clear increases in SCEs.
Technical grade (65% AI) sodium xylenesulfonate was tested in Chinese hamster ovary cells with and without metabolic activation. Test concentrations were 2513, 3750, and 5000 μg/mL. There was no clastogenic activity with and without metabolic activation. Positive controls gave results that confirmed the validity of the test.
In Vivo
Three mouse micronucleus cytogenetic assays were reported by SIDS. One study with calcium xylenesulfonate used a single IP injection of 0, 145, 290, or 580 mg AI/kg bw (5/sex/group). Doses were selected from a preliminary dose range-finding study. Two oral (gavage) studies are available with sodium cumenesulfonate. One study used a single administration of 0 or 4467 mg AI/kg bw (5/sex/group) with the dose selected from a preliminary dose ranging study. The other doses (total of 9) of 400, 2000, and 4000 mg AI/kg bw were delivered in 2 equal applications 24 hours apart (7/sex/group). One male and 1 female died at the highest dose in this repeated dose study. Negative results were obtained in all 3 studies. In all 3 assays, the positive controls gave results that confirmed the validity of the test.
Carcinogenicity
According to Lewis et al, a series of 30 NTP chemicals was evaluated prospectively for rodent carcinogenicity and overt toxicity by Computer Optimized Molecular Parametric Analysis for Chemical Toxicity (COMPACT: CYP1A and CYP2E1). 10 The COMPACT predicts the potential of a compound to act as a substrate for one of the cytochromes P450. Evaluations were also made by Hazard expert and for metal ion redox potentials; these, together with COMPACT, were compared with results from the Ames test for mutagenicity in the Salmonella micronucleus test and 90-day subchronic rodent pathology. Of the 30 chemicals, 7 (nitromethane, chloroprene, xylene sulfonic acid, furfuryl alcohol, anthraquinone, emodin, and cinnamaldehyde) were positive for potential carcinogenicity in the COMPACT evaluation; xylene sulfonic acid and furfuryl alcohol were only equivocally positive.
A 2-year NTP study of sodium xylenesulfonate was performed in F344 rats (50/sex/group) and B6C3F1 mice (50/sex/group) using the dermal route of exposure. No carcinogenic activity was observed in either rats or mice. The chronic effects from this study, and the methodology, are discussed in the chronic toxicity section.
Clinical Assessment of Safety
Dermal Irritation
No clinical data on dermal irritation were found.
Dermal Sensitization
According to SIDS, there was no evidence of skin sensitization reported in a human repeat insult patch test of 0.5% aqueous sodium cumenesulfonate in a 0.1% aqueous solution of granular laundry detergent. Limited study details were provided by the authors.
Margin of Exposure Estimations
The Human and Environmental Risk Assessment project, in its reports on ingredients of European household cleaning products and guidance document, determined a margin of exposure (MOE). They calculated the MOE by dividing the local effects NOEL (local epidermal hyperplasia—440 mg/kg bw/d) by the estimated external exposure to products having direct skin contact (eg, using hard surface cleaners) and indirect skin contact (eg, residue on clothing from laundering). The direct and indirect skin contact exposure scenarios assumed 1% percutaneous absorption. These calculations are shown in Table 10. The MOE for indirect exposure was approximately 6 × 103 and for direct exposure ranged from 4.4 × 104 to 4.9 × 105, suggesting that the use of these products containing hydrotropes could not present a dermal risk.
Margin of Exposure for Hydrotropes 7

Abbreviation: MOE, margin of exposure.
Summary
While not used in cosmetics, xylene sulfonic acid is listed as a cosmetic ingredient and is used in the synthesis of a series of hydrotropes that are used in cosmetics, including ammonium xylenesulfonate, calcium xylenesulfonate, potassium xylenesulfonate, and sodium xylenesulfonate. Other sulfonic acids and their corresponding hydrotropes are also cosmetic ingredients, including ammonium cumenesulfonate, potassium cumenesulfonate, potassium toluenesulfonate, sodium toluenesulfonate, sodium cumenesulfonate, and toluene sulfonic acid. Hydrotropes are amphiphilic substances composed of both hydrophilic and hydrophobic functional groups. They can increase the solubility of less soluble substances.
Hydrotropes are produced by sulfonation of an aromatic hydrocarbon solvent (ie, toluene, xylene, or cumene). 1 The resulting aromatic sulfonic acid is neutralized using an appropriate base (eg, sodium hydroxide, ammonium hydroxide, potassium hydroxide, or calcium hydroxide) to produce the sulfonate or hydrotrope.
Information supplied to the FDA by industry as part of the VCRP states that there were 76 and 75 reported uses for ammonium xylenesulfonate and sodium xylenesulfonate, respectively. No FDA uses were reported for the remaining hydrotropes. In an industry survey, ammonium xylenesulfonate, sodium xylenesulfonate, and sodium cumenesulfonate were reportedly used in cosmetics up to concentrations of 3%, 15%, and 3%, respectively.
No absorption, distribution, metabolism, or excretion studies for the hydrotropes were identified, but the molecular weight, water solubility, and octanol/water partition coefficient (Log P) values suggest that while significant absorption may occur following oral administration, absorption following dermal application would be limited.
Acute toxicity studies for xylene sulfonic acid reported LD50 values of 2100 to 16 200 mg/kg bw (dermal), 2000 mg/kg bw (oral), and 557 000 to 770 000 mg/m3 (inhalation). An IP study using mice reported an LD50 of 500 mg/kg bw. Repeated dose dermal toxicity studies using both rats and mice with up to 2% technical grade sodium xylenesulfonate in water (17 day) and ethanol (90 day) reported no treatment-related deaths.
Two 2-week range-finding studies in both mice and rats were performed using up to 4% sodium xylenesulfonate in the diet uncovered no clinical signs of toxicity or mortality at any of the doses in mice. Decreased body weight gains were reported, but with no dose–response relationship between test material concentration and body weight gain. In rats, deaths occurred at 2% and 4% doses in the first study, but no deaths were reported in the second study.
In a 90-day feeding study, no treatment-related effects were observed in rats administered sodium xylenesulfonate at up to 5.0% in the diet. A dose–response effect was not observed at the 3 doses. There were no treatment-related histopathologic alterations in the livers. No treatment-related effects were observed in the 90-day study for sodium xylenesulfonate in female mice. There was some epidermal hyperplasia observed in male and female mice at the highest doses. From the 90-day studies, the NOAEL for local effects, based on epidermal hyperplasia at the site of application, was 440 mg AI/kg bw for male mice and 540 mg AI/kg bw for female mice.
Other 90-day feeding studies using rats and mice resulted in no treatment-related effects were observed in males and a loss of relative spleen weight in females, along with some clinical chemistry and hematology changes, but no treatment-related gross or microscopic lesions noted at necropsy in either rats or mice. Rats exposed to sodium cumenesulfonate at up to 0.5% in the diet exhibited no treatment-related effects. Rats dermally treated with up to 240 mg sodium xylenesulfonate per kilogram bodyweight in 50% ethanol for 104 weeks resulted in no treatment-related effects in males and only to irritation at the application site in females.
Slight-to-moderate ocular irritation was observed in rabbits treated with a solution containing xylene sulfonates, toluene sulfonates, and cumenesulfonates in solutions ranging from 20% to 60%. There was mild-to-moderate skin irritation observed in rabbits treated dermally with solutions containing xylene sulfonates, toluene sulfonates, and cumenesulfonates in solutions ranging from 1% to 60%.
In a guinea pig study, there was no evidence of skin sensitization following dermal, occlusive exposure to a 42.8% solution (deionized water) of sodium toluenesulfonate. No irritation was observed during the study (neither at induction nor at challenge).
Calcium xylenesulfonate administered via gavage to female rats at doses up to 3000 mg/kg bw in water on days 6 to 15 of gestation produced no treatment-related effects. The NOAEL for maternal and fetal toxicity was the highest dose tested, 3000 mg/kg bw/d that corresponded to 936 mg AI/kg bw/d.
There was no evidence of mutagenic potential of sodium xylenesulfonate, calcium xylenesulfonate, and sodium cumenesulfonate in an Ames test using strains TA98, TA100, TA1535, TA1537, and TA1538 with and without metabolic activation. There was some evidence of sodium xylenesulfonate mutagenic potential in L5178Y mouse lymphoma cells at 5000 μg/mL with and without metabolic activation. No clastogenic activity was found for sodium xylenesulfonate using Chinese hamster ovary cells. Three mouse micronucleus cytogenetic assays using calcium xylenesulfonate were negative.
Xylene sulfonic acid was equivocally positive for potential carcinogenicity in the COMPACT evaluation. There was no evidence of carcinogenic activity in F344/N rats (highest dose 240 mg AI/kg bw/d) and B6C3F1 mice (highest dose 727 mg AI/kg bw/d) that received dermal application to clipped skin 5 d/week of technical grade sodium xylenesulfonate for 2 years. There was no evidence of skin sensitization reported in a human repeat insult patch test of 0.5% aqueous sodium cumenesulfonate in a 0.1% aqueous solution of granular laundry detergent.
Margins of exposure calculated by dividing the local effects NOEL by the estimated external exposure to products having direct skin contact (eg, using hard surface cleaners) and indirect skin contact (eg, residue on clothing from laundering) resulted in values of 6 × 103 for indirect exposure and for direct exposure, 4.9 × 105, suggesting that the use of these products containing hydrotropes could not present a dermal risk.
Discussion
The ingredients in this safety assessment include alkyl aryl sulfonates that function in cosmetics as hydrotropes. Xylene sulfonic acid and toluene sulfonic acid, although not actually used as cosmetic ingredients, are used as intermediates in the production of the salts. The CIR Expert Panel recognized that there are data gaps regarding use and concentration of some of these ingredients. But, there is a pattern of use and, were ingredients not currently used to be used in the future, there is an expectation that they would be used in similar product types at similar use concentrations. The Panel noted the widespread use of alkyl aryl sulfonates in detergents; yet, there is an absence of reports of allergic reactions to these compounds.
Based on molecular weight, water solubility, and octanol–water partition coefficient, the Expert Panel accepted that the significant absorption could occur following oral administration, but considered that absorption following dermal application would be limited.
In single and repeated dose toxicological studies, including genotoxicity, reproductive and developmental toxicity, and limited carcinogenicity, data on all end points indicate a low toxicity for these compounds. Dermal irritation studies indicated that toluene sulfonate, xylene sulfonate, and cumenesulfonate were not irritating at a 15% use concentration. The Panel noted the absence of adverse reactions to hydrotropes in clinical experience, despite their widespread use, as well as the absence of sensitization reports in the literature.
Although there are data gaps, the similar chemical structures, physicochemical properties, and functions and concentrations in cosmetics allow grouping these ingredients together and interpolating the available toxicological data to support the safety of the entire group.
Conclusion
Xylene sulfonic acid, ammonium cumenesulfonate, ammonium xylenesulfonate, calcium xylenesulfonate, potassium cumenesulfonate, potassium toluenesulfonate, potassium xylenesulfonate, sodium toluenesulfonate, sodium cumenesulfonate, sodium xylenesulfonate, and toluene sulfonic acid are safe as cosmetic ingredients in the present practices of use and concentrations as described in this safety assessment, when formulated to be nonirritating. 1
Footnotes
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St., Suite 412, Washington, DC 20036, USA.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. F. Alan Andersen is employed by the Cosmetic Ingredient Review.
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review program is financially supported by the Personal Care Products Council.
