Abstract
Pelargonic acid and its esters function as skin-conditioning agents in cosmetics. Molecular weight (mw) and octanol–water partition coefficient data suggest that dermal penetration is possible. The biohandling of branched-chain fatty acids is not the same as for straight-chain fatty acids, but the differences are not significant to the conclusion that they all are readily metabolized to nontoxic moieties. Limited data suggested that the penetration of other ingredients may be enhanced if these ingredients are present in the same formulation. These ingredients are not significant oral or dermal toxicants in animal studies. They are not reproductive/developmental toxicants or genotoxic/carcinogenic in animal studies. The available data suggested that product formulations containing these ingredients would be nonirritating and nonsensitizing to human skin, but formulators were cautioned to consider the penetration enhancement potential. The Cosmetic Ingredient Review (CIR) Expert Panel concluded that these ingredients are safe in the present practices of use and concentration.
Keywords
Introduction
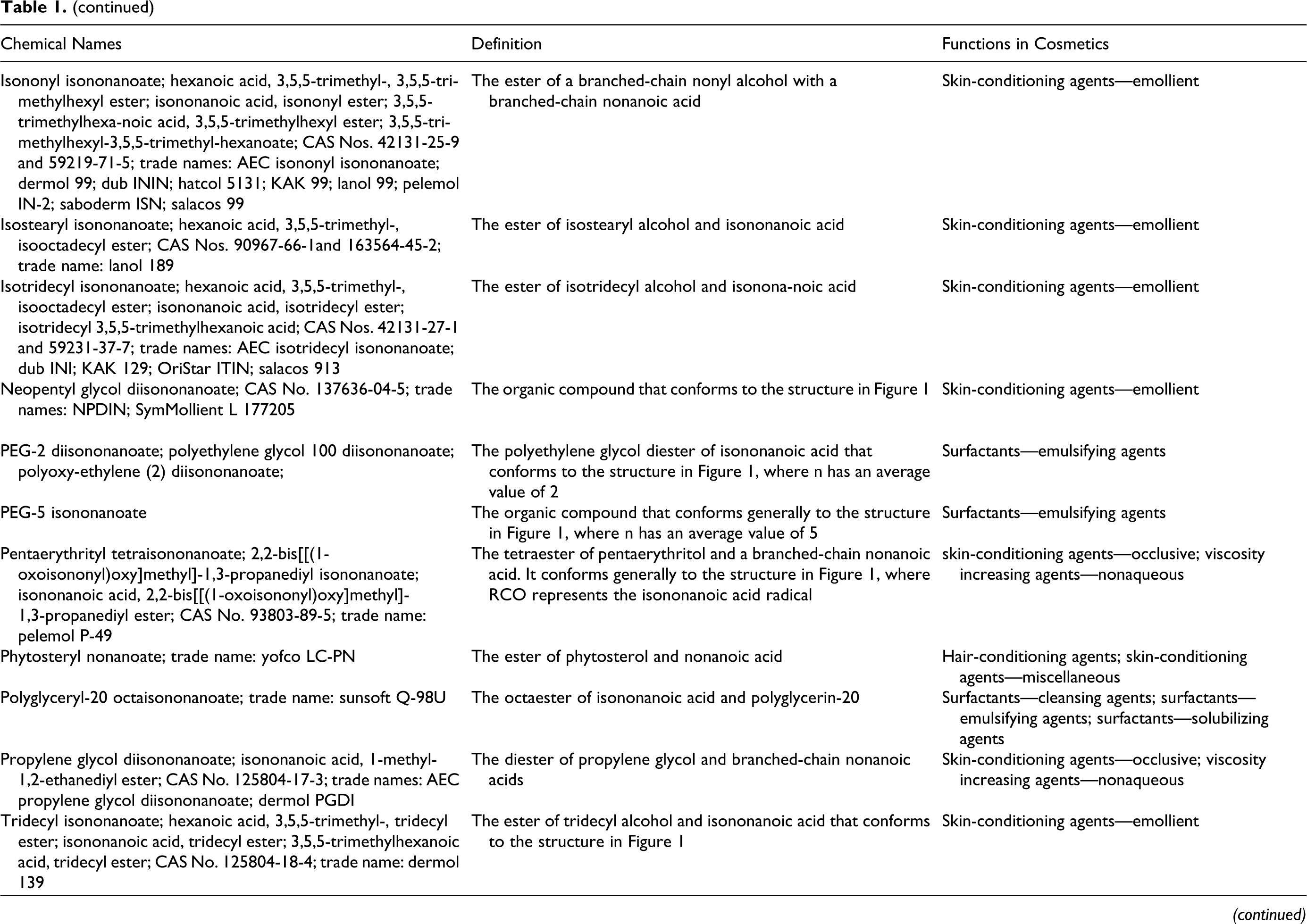
Pelargonic acid (aka nonanoic acid) is a fatty acid that can function as a fragrance ingredient, surfactant-cleansing agent, and surfactant-emulsifying agent in cosmetics. In its soap form, it can function as a surfactant-cleansing agent. Many of the fatty acids that are used in cosmetics and their synthesized derivatives (primarily esters and diesters of the corresponding alcohol and pelargonic acid) have similar additional functions in cosmetics. An idiosyncrasy in the terminology used in the International Cosmetic Ingredient Dictionary and Handbook is that most of these derivatives are termed nonanoates, not pelargonates. This safety assessment includes the following ingredients:
pelargonic acid
butylene glycol diisononanoate
cellobiose octanonanoate
cetearyl isononanoate
cetearyl nonanoate
cetyl isononanoate
cholesteryl nonanoate
diethylene glycol diethylhexanoate/diisononanoate
diethylene glycol diisononanoate
dihydrocholesteryl nonanoate
dipentaerythrityl pentaisononanoate
ethylhexyl isononanoate
glycereth-7 diisononanoate
isodecyl isononanoate
isononyl isononanoate
isostearyl isononanoate
isotridecyl isononanoate
neopentyl glycol diisononanoate
PEG-2 diisononanoate
PEG-5 isononanoate
pentaerythrityl tetraisononanoate
phytosteryl nonanoate
polyglyceryl-20 octaisononanoate
propylene glycol diisononanoate
tridecyl isononanoate
ethylhexyl pelargonate
ethyl pelargonate
isobutyl pelargonate
methyl pelargonate
neopentyl glycol dicaprylate/dipelargonate/dicaprate
pentaerythrityl tetrapelargonate
The nonanoate or pelargonate esters and diesters are straight-chain compounds, whereas the isononanoate esters and diesters are branched-chain compounds. Propylene glycol dipelargonate would have been included in this safety assessment; however, the Cosmetic Ingredient Review (CIR) Expert Panel previously concluded that this ingredient is safe in the present practices of use in cosmetic products. 1
Pelargonic acid is a reactant in the esterification process that yields all of the esters included in this safety assessment. While this fatty acid is not reported to be currently used in cosmetics, much of the data available for review relate to pelargonic acid. The CIR Expert Panel has published safety assessments on the following fatty alcohols and other reactants used to form some of the esters reviewed in this safety assessment, and other fatty acid esters: butylene glycol—safe in present practices of use and concentration (safe), 2,3 cetearyl, cetyl, and isostearyl alcohols—safe, 4,5 cholesterol—safe, 6,3 ethylene glycol—special report on reproductive and developmental toxicity—no conclusion, 7 ethylhexyl palmitate (previously incorrectly named octyl palmitate)—safe, 8,9 PEG-7 glyceryl cocoate—safe as used in rinse off products; safe up to 10% in leave on products, 10 PEGs-2 and -6 dilaurate—safe for use in cosmetics at concentrations up to 25%, 11 octyl stearate and isobutyl stearate—safe, 12,5 isodecyl oleate—safe, 13,14 and propylene glycol dipelargonate—safe, 1 as stated above. These conclusions may contribute to the safety assessment of ingredients in the current review for which little or no data have been identified in the published literature.
Similarly, there are ingredient moieties that have not been reviewed by the Expert Panel, and available data on these chemicals may be useful in the absence of safety test data on some of the esters that are being reviewed. Thus, data on or relevant to the following chemicals are also summarized in the current safety assessment: isononanoic acid, isononyl alcohol, isotridecyl alcohol, neopentyl glycol, isobutyl alcohol, and isodecyl alcohol. Excerpts from the summary and discussion from the CIR Final Safety Assessment on propylene glycol dipelargonate and other propylene glycol esters and diesters are also included, because these data may be useful in evaluating the safety of diesters included in the current review. Excerpts from the CIR Final Safety Assessments on isobutyl stearate and isodecyl oleate are also included, in lieu of data on isobutyl alcohol and isodecyl alcohol, respectively.
Chemistry
Definition and Structure
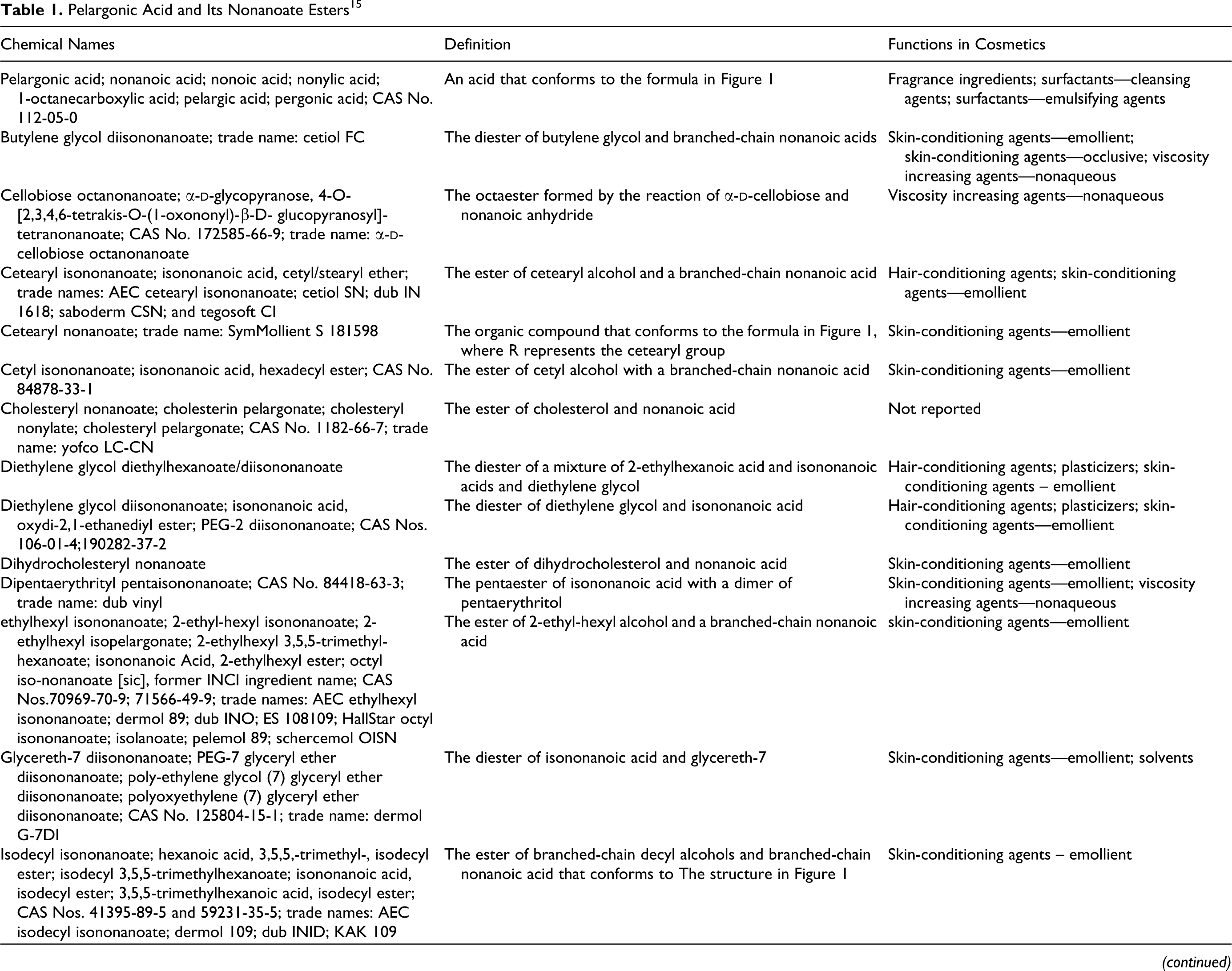
Chemical definitions, other chemical names, and cosmetic ingredient functions for the ingredients reviewed in the safety assessment are included in Table 1. The International Nomenclature Cosmetic Ingredient (INCI) name appears first in each series of chemical names; “iso” in an INCI name denotes methyl branching/substitution and does not necessarily imply substitution on the second to last carbon atom (omega-2 substitution). 15 Chemical structures/formulas are included in Figure 1. The inclusion of [sic] after a technical name or CAS No. in Table 1 denotes those instances wherein the authors of the dictionary associated a specific branched chemical entity other than an omega-2 methyl substituted isomer with an “iso” INCI name. According to the dictionary proviso regarding “iso”-named ingredients, all branched isomers are potentially included by an “iso” INCI ingredient name. For simplicity, only omega-2 isomers are shown in Figure 1. However, in one exception, the [sic] notation is included after “octyl isononanoate,” in the ethylhexyl isononanoate box of Table 1, in that octyl isononanoate is included in the International Cosmetic Ingredient Dictionary and Handbook because it is a former INCI name for this ingredient.
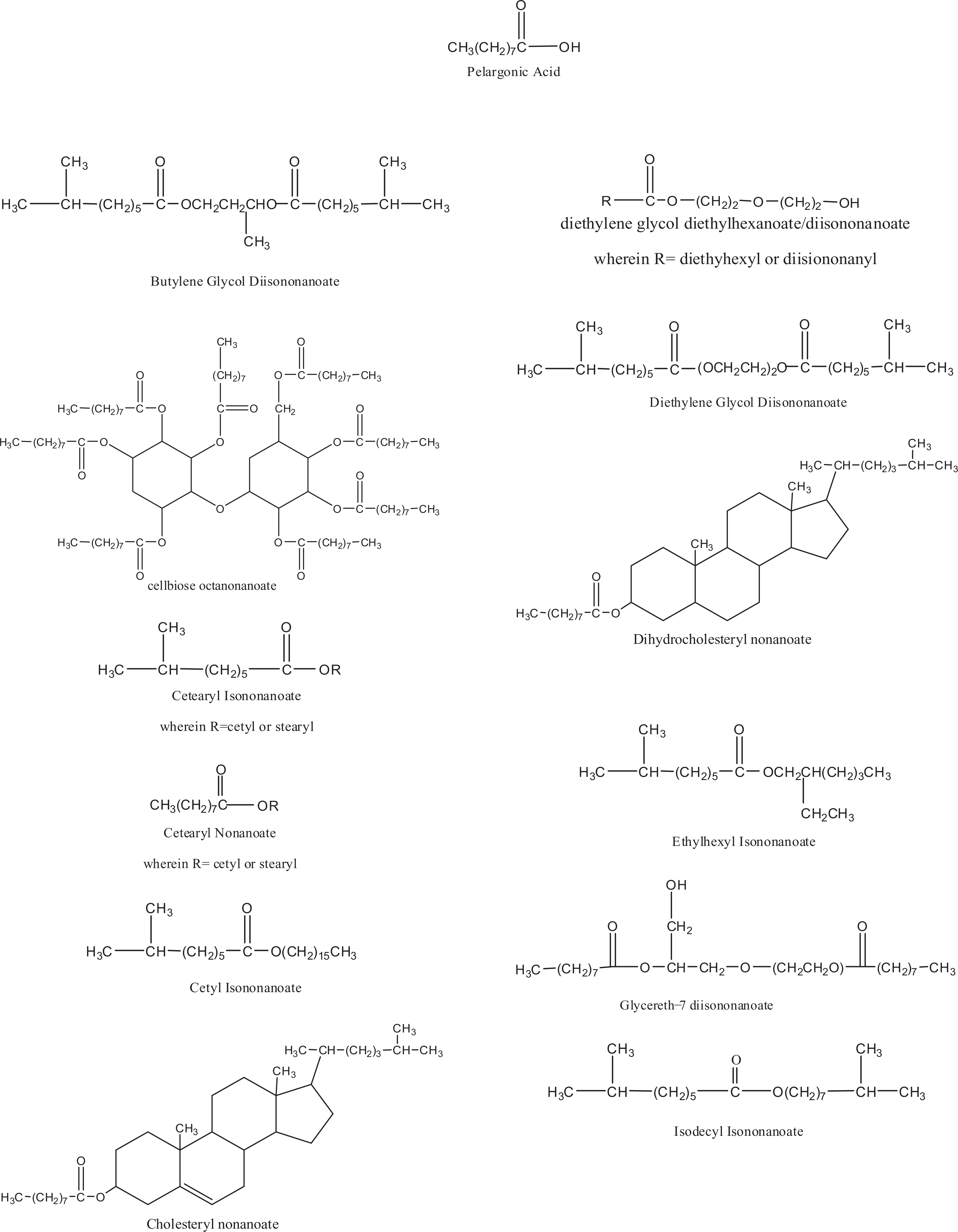
Structures/formulas of nonanoates/pelargonates.
Pelargonic Acid and Its Nonanoate Esters 15
Chemical and Physical Properties
Available chemical and physical properties are included in Table 2.
Chemical and Physical Properties




UV Absorption
Data provided by a manufacturer 33 indicated that the following chemicals did not absorb significantly in the 250 to 400 nm range: neopentyl glycol diisononanoate, cetyl nonanoate + stearyl nonanoate, trideceth-9 + PEG-5-isononanoate + water, glyceryl triisononanoate + glyceryl diisononanoate, and ethylhexyl isononanoate.
Analytical Methods
Pelargonic acid, cholesteryl nonanoate, ethyl pelargonate, and methyl pelargonate have been analyzed by nuclear magnetic resonance (NMR), infrared (IR), and mass spectrometry; and pentaerythrityl pelargonate has been analyzed by IR spectrometry. 17 Methyl pelargonate and pelargonic acid have also been analyzed by gas chromatography–mass spectrometry, 34 and the same is true for ethyl pelargonate. 35
Methods of Production
In general, the alkyl esters can be produced industrially via the esterification of carboxylic acids with the corresponding alcohols (with or without a metal catalyst). 36 The sources of these carboxylic acids and alcohols are often natural or are derived from natural sources. Alcohols with chains longer than ethanol (C2) are sometimes produced synthetically, but natural sources are more common. Acids and alcohols from natural sources are often mixtures. This is especially true in the case of branched acids and alcohols. Accordingly, the resulting esters are also mixtures. An important method for producing C3-C20 industrial alcohols involves a process known as oxo-synthesis (a process for the production of aldehydes which occurs by the reaction of olefins (which can be natural or petroleum sourced) with carbon monoxide, hydrogen, and a catalyst (typically colbalt based), followed by the hydrogenation of the aldehyde products, to form the alcohols. 37 An industry shift began a couple of years ago toward a green, biocatalytic process developed specifically for the manufacture of esters for use in the formulation of cosmetic and personal care ingredients (ie, for producing cosmetic grade esters). 38
Pelargonic acid is prepared from unsaturated hydrocarbons by the oxo process, or by the oxidation of oleic acid, and from tall oil unsaturated fatty acids or rice bran oil fatty acid. 39 In the oxo process, pelargonic acid is prepared synthetically. Preparation from tall oil unsaturated fatty acids or rice bran oil fatty acid occurs naturally via splitting/separation.
The production methodology for cetearyl isononanoate and isononyl isononanoate includes the use of a typical mineral salt/nonorganic catalyst for ester formation in the reaction; the catalyst is filtered off upon completion of the reaction. 40
Impurities
Specifications on pelargonic acid from a chemical supplier include pelargonic acid (90% minimum), iron (1.0 ppm max), moisture (0.2% max), and the following monoprotic acids: other C9 acids (93%; eg, isononanoic acid) and other length monoprotic acids (2%; eg, octanoic acid). 41
The heavy metal content of both cetearyl isononanoate (Tegosoft CI) and isononyl isononanoate (Tegosoft INI) is described as follows: 20 ppm maximum (copper, lead, tin, platinum, palladium, mercury, arsenic, cadmium, and nickel); <1 ppm (mercury, arsenic, cadmium, and nickel). 24,28
Typical impurities include olefin, acid, and alcohol starting materials; water; and residual metals (from catalysts). Neopentyl glycol formic acid ester and neopentyl glycol isolactic acid ester are impurities that have been detected in neopentyl glycol. 32
Use
Purpose in Cosmetics
The majority of the ingredients reviewed in this safety assessment function as skin-conditioning agents in cosmetics. Cholesteryl nonanoate is the only ingredient for which an ingredient function in cosmetics is not listed in the International Cosmetic Ingredient Dictionary and Handbook. Ingredient functions in cosmetics are included in Table 1. 42
Scope and Extent of Use in Cosmetics
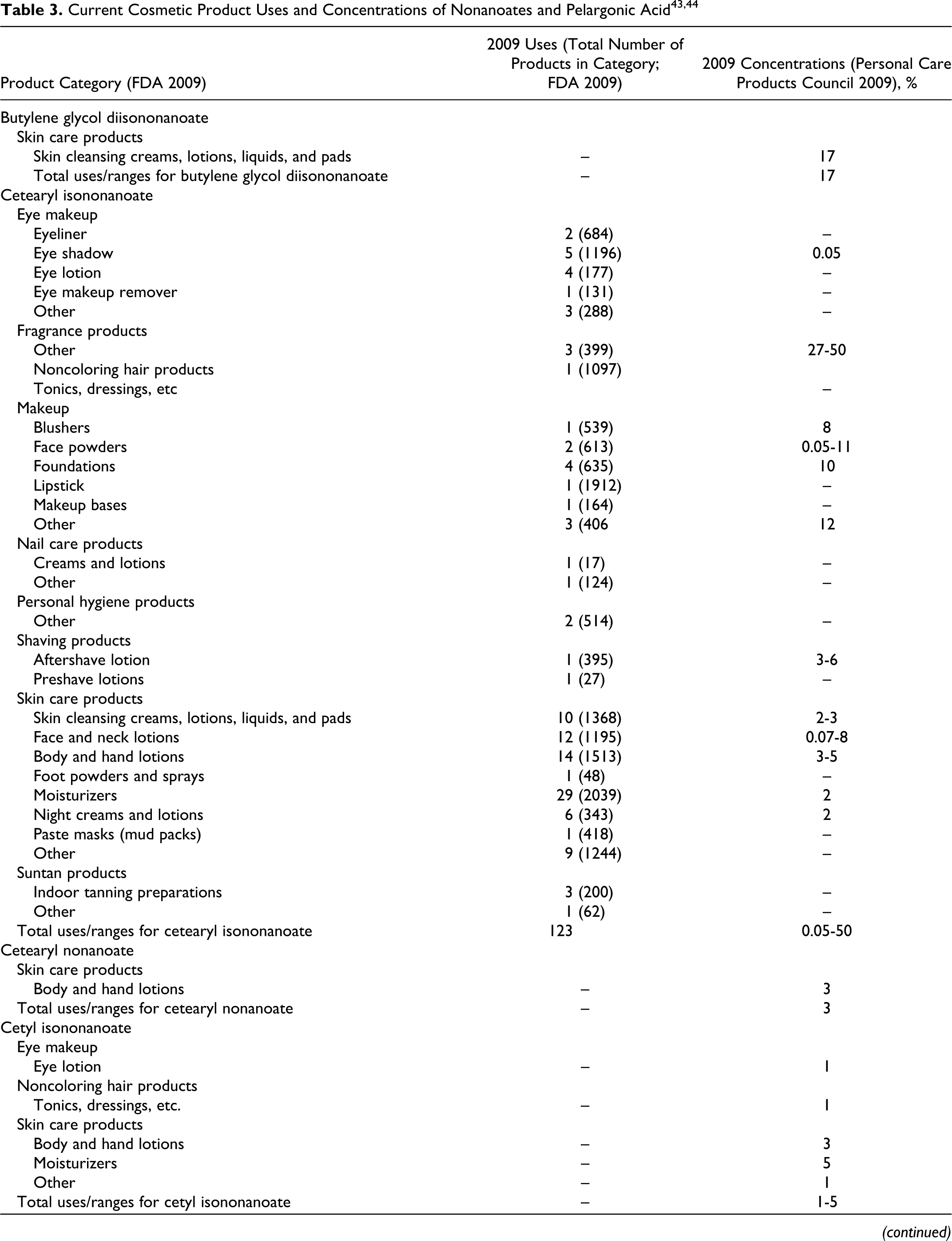
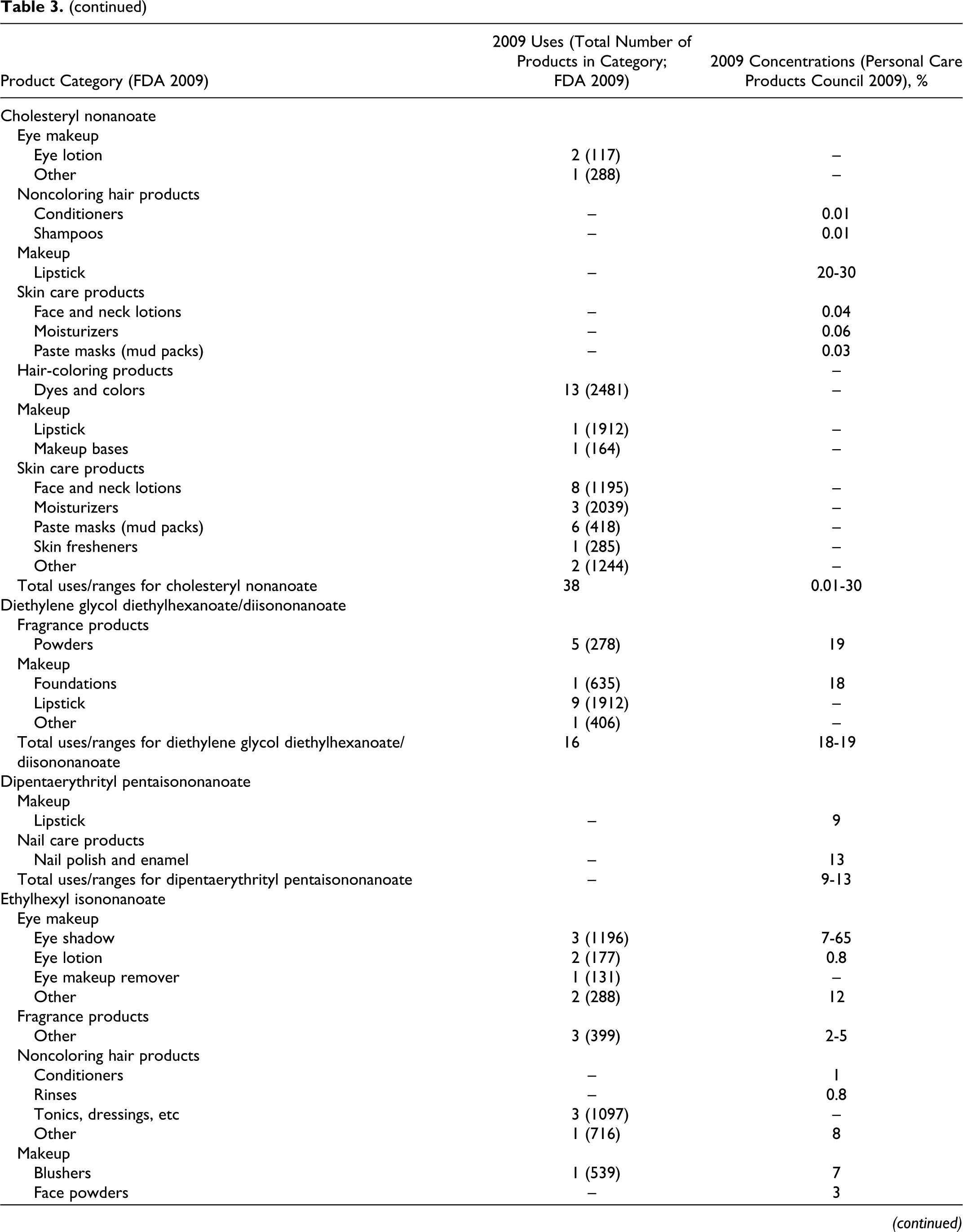
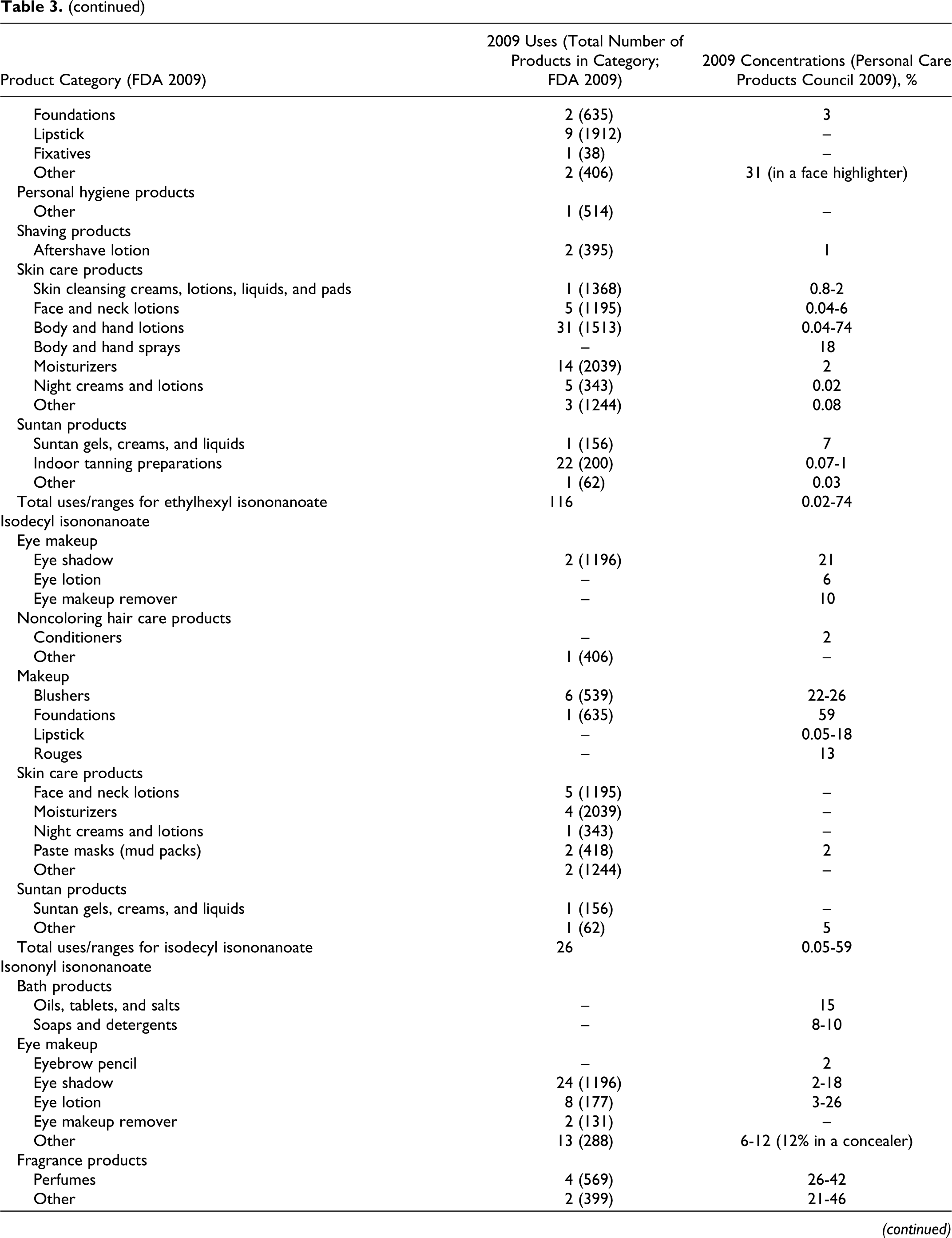
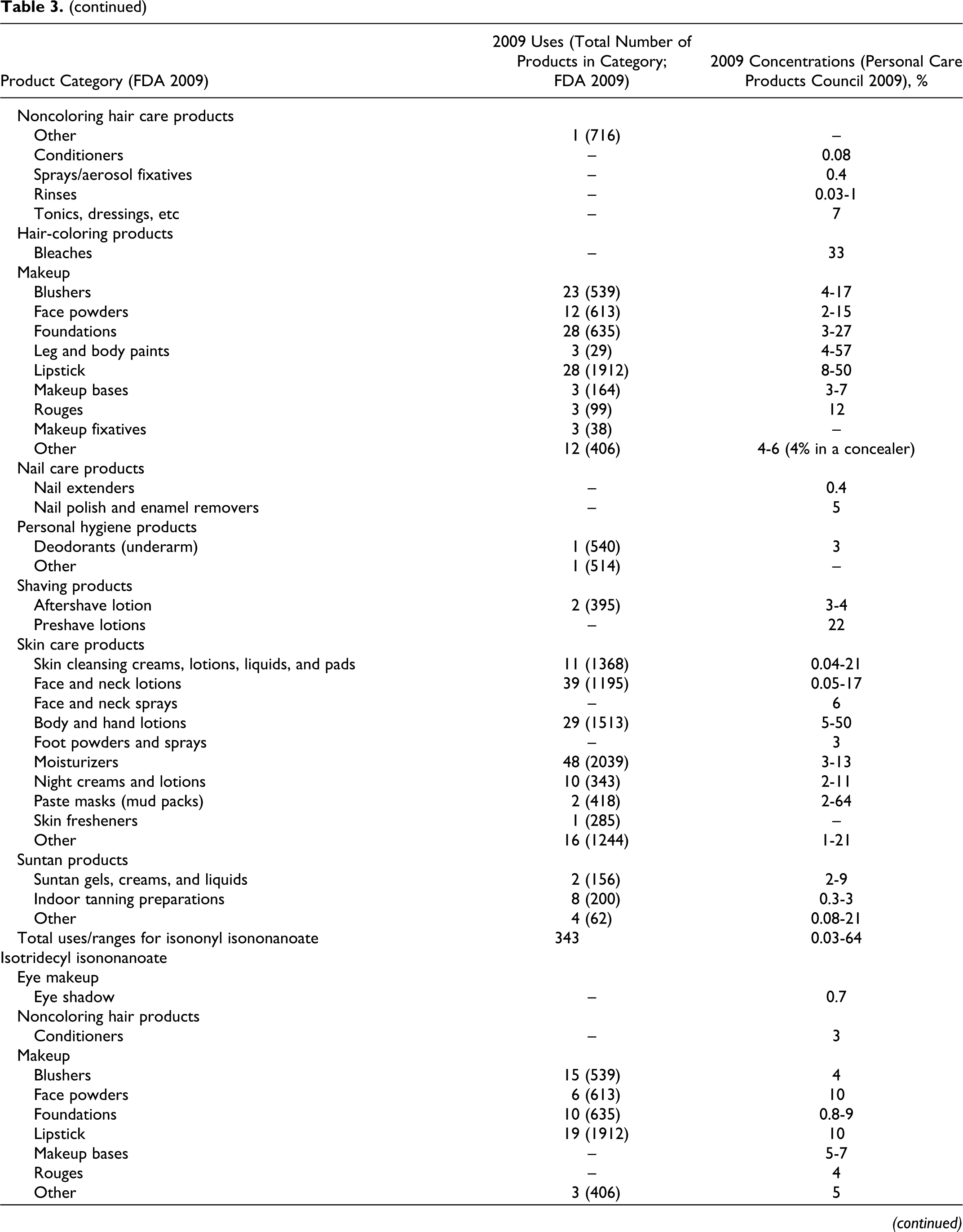
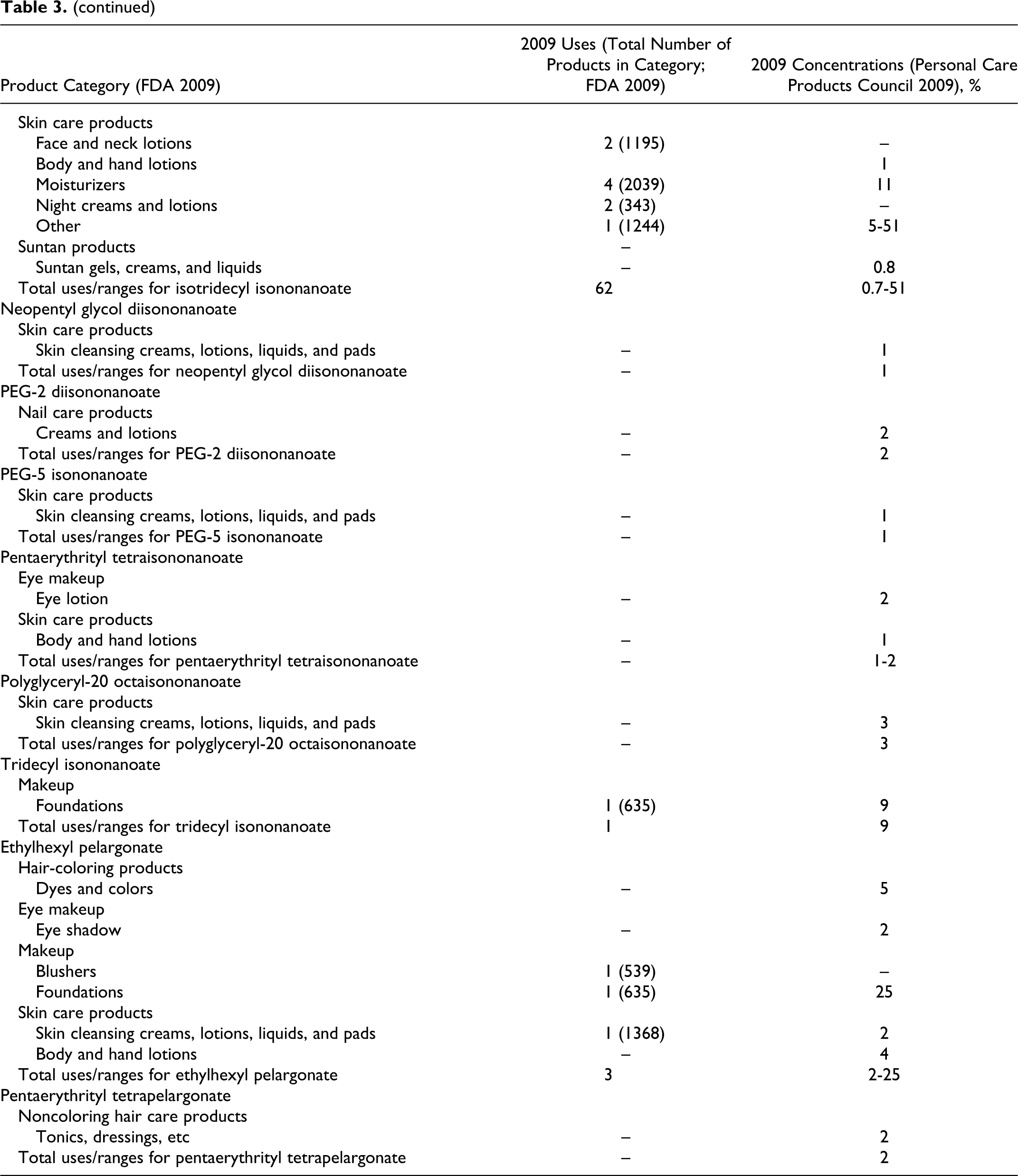
According to information supplied to the Food and Drug Administration (FDA) by industry as part of the Voluntary Cosmetic Registration Program (VCRP) in 2009, 43 the following ingredients reviewed in this safety assessment are being used in cosmetic products: cetearyl isononanoate, cholesteryl nonanoate, diethylene glycol diethylhexanoate/diisononanoate, ethylhexyl isononanoate, isodecyl isononanoate, isononyl isononanoate, isotridecyl isononanoate, tridecyl isononanoate, and ethylhexyl pelargonate. Use data for these ingredients are summarized in Table 3. Independent of these data, the results of a survey of current ingredient use concentrations conducted by the Personal Care Products Council (Council) in 2009 are also summarized in Table 3. 44 Cetearyl isononanoate is used in 5 of the 1196 eye shadow products reported to the VCRP, and results from the Council survey indicate the use of this ingredient at a concentration of 0.05% in these products. In other cases, for example for cetearyl isononanoate, uses are reported to the VCRP, but its use concentration data are not available. Current use concentration data from the Council also indicate that, while not reported to the VCRP, the following ingredients are also being used in cosmetic products: butylene glycol diisononanoate, cetearyl nonanoate, cetyl isononanoate, dipentaerythrityl pentaisononanoate, neopentyl glycol diisononanoate, PEG-2 diisononanoate, PEG-5 isononanoate, pentaerythrityl tetraisononanaote, polyglyceryl-20 octaisononanoate, and pentaerythrityl tetrapelargonate.
Based on the data included in Table 3 (use frequency and use concentration data), there is no indication that the following ingredients are being used in cosmetics: cellobiose octanonanoate, diethylene glycol diisononanoate, dihydrocholesteryl nonanoate, glycereth-7 diisononanoate, isostearyl isononanoate, phytosteryl nonanoate, propylene glycol diisononanoate, ethyl pelargonate, isobutyl pelargonate, methyl pelargonate, neopentyl glycol dicaprylate/dipelargonate/dicaprate, and pelargonic acid.
Cosmetic products containing the in-use ingredients may be applied to the skin or hair, or, incidentally, may come in contact with the eyes, nails, and mucous membranes. Products containing these ingredients may be applied as frequently as several times per day and may remain in contact with the skin/hair for variable periods following application. Daily or occasional use may extend over many years.
Noncosmetic Use
Pelargonic acid is included on the list of food additives (synthetic flavoring substances and adjuvants) permitted for direct addition to food for human consumption, as stated in 21 CFR 172.515. 45 Mixtures containing pelargonic acids may be used in other food preparation or processing uses (21 CFR 173.315, 21CFR 178.1010). 45 Pelargonic acid is registered by the US Environmental Protection Agency (EPA) for use as a blossom thinner and as an herbicide. 46,47 It is exempt from the requirement of a tolerance for pesticide residues in or on all foods, when used as a component of a food contact surface sanitizing solution in food handling establishments. 48
General Biology
Metabolism
Pelargonic acid
The oxidative degradation of fatty acids, such as pelargonic acid, into 2-carbon fragments through enzymatically catalyzed reactions is a well-documented central metabolic pathway in animals and plants. Pelargonic acid, a straight-chain carbon molecule, would be metabolized by β-oxidation to form acetate molecules, which enter the citric acid cycle and are metabolized to carbon dioxide, water, and energy. 46,49
Ethyl pelargonate
Straight-chain aliphatic acid esters are thought to be readily hydrolyzed into their component acids and alcohols, which would then be expected to follow their normal metabolic pathways. 50
Branched-chain fatty acids
Mammalian metabolism of some lipids, including 3-methyl (eg, phytanic acid) and 2-methyl (eg, pristanic acid) branched-chain fatty acids, occurs in peroxisomes. 51 Because of the location of a methyl group at the β-carbon of phytanic acid, degradation of the acid via the β-oxidation pathway cannot occur. Instead, the α-methylene group of phytanic acid is oxidatively excised, yielding pristanic acid, which can be metabolized via the β-oxidation pathway.
Percutaneous Absorption
Isononyl alcohol
The results of an acute dermal toxicity study 52 on undiluted isononyl alcohol (rabbits, abraded skin) are summarized later in the report text. When the occlusive binders were observed after 24 hours of contact in this study, percutaneous absorption was evident. Details relating to this finding were not included.
Skin Penetration Enhancement
The skin penetration enhancement effects of fatty acids on p-aminobenzoic acid (PABA) penetration through sheets of human stratum corneum (surgically removed human breast or abdominal skin) were studied. 53 The stratum corneum sheet was pretreated with the penetration enhancer. The permeation of PABA increased with increasing chain length of straight-chain saturated fatty acid, from 6 to 9 carbons (hexanoic, heptanoic, octanoic, and pelargonic acids, respectively). A sharp increase in PABA permeability occurred at fatty acid chain lengths of between 8 and 9 carbons. The mean steady state flux for PABA was 837.84 ± 190.30 µg/cm2 per h in the presence of pelargonic acid, compared to 2.57 ± 0.19 µg/cm2 per h in the presence of water, and 0.28 ± 0.14 µg/cm2 per h in the presence of propylene glycol.
A study on the release profile of melatonin from drug-in-adhesive type transdermal patches (prepared using Eudragit E 100 as adhesive polymer) containing pelargonic acid or other penetration enhancers, and the in vitro penetration of melatonin through hairless rat skin in the presence of the enhancer was conducted. 54 Melatonin release was studied using the US Pharmacopoeia dissolution test apparatus in conjunction with high-performance liquid chromatography. The release profiles of melatonin from the patches with enhancers were similar when compared to the control patch release profile.
In skin penetration experiments, each penetration enhancer was added to the patch at a concentration of 2.5% or 5%. Skin samples from at least 3 rats were used in each experiment, and each mean value for skin penetration represented 3 replicates. The presence of enhancers in the patches resulted in an increase in the permeation of melatonin through hairless rat skin. The mean melatonin flux values for patches containing octanol, pelargonic acid, or myristic acid (each at both concentrations) were higher when compared to the control patch; however, the differences were not statistically significant (P > .05). 54
Endocrine Disruption
According to EPA, 49 it would be unlikely for straight-chain carbon molecules, as in the C9 carbon chain of pelargonic acid, to be associated with a risk of endocrine disruption.
Animal Toxicology
Acute Inhalation Toxicity
Pelargonic acid
EPA 46 placed pelargonic acid in toxicity category III (>0.5 through 2 mg/L), primarily based on the results of the following study. The acute inhalation toxicity of pelargonic acid was evaluated using groups of 10 (5 males, 5 females/group) albino rats. 55 The 4 groups were exposed (4-hour exposure) to aerosol generated from undiluted pelargonic acid, delivering concentrations of 0.510, 0.710, 2.20, and 3.31 mg/L, respectively. The following mortalities were reported: 1 rat (at 0.510 mg/L), 1 rat (at 0.710 mg/L), 8 rats (at 2.20 mg/L), and 10 rats (at 3.31 mg/L). Gross necropsy was performed on animals that died, and the following findings were considered unusual and possibly related to exposure: nasal discharge, polyuria, salivation, and discolored and swollen lungs, and variations thereof. Acute inhalation lethal concentration 50s (LC50s) of 0.87 mg/L (95% confidence limits = 0.50-1.51 mg/L) and 2.10 mg/L (95% confidence limits = 1.71-2.58 mg/L) were reported for males and females, respectively. The overall LC50 was 1.24 mg/L (95% confidence limits undefined).
Isononanoic acid
The respiratory effects of isononanoic acid using groups of 4 specific pathogen-free, male Swiss-Webster mice were evaluated. 56 The animals were exposed to nebulized isononanoic acid (concentration range: 172-755 mg/m3) in a 2.5-L exposure chamber for 180 minutes. Sensory and pulmonary irritation was reported, and recovery immediately post exposure was described as poor. The test substance concentration that was capable of evoking a 50% decrease in the mean respiratory frequency (RD50) was 420 mg/m3. The decreases in respiratory frequency induced by isononanoic acid were described as concentration dependent and due to a combination of sensory and pulmonary irritation.
Isononyl alcohol
The inhalation toxicity of isononyl alcohol using groups of 10 Swiss mice, Wistar rats, and English Short Hair guinea pigs was studied. 52 Each group received a single 6-hour vapor exposure under dynamic conditions; exposure was followed by a 14-day observation period. The concentration of isononyl alcohol in the exposure chamber was calculated to be 21.7 mg/L. None of the animals died during exposure; however, 1 mouse and 2 rats died within the first 14-hour post exposure. Signs of systemic toxicity consisted primarily of central nervous system depression but were not pronounced. Local irritation involving mucous membranes of the eyes and nose was observed to a variable extent, and all animals had recovered within 1 hour after termination of exposure. Histopathological examinations were not performed.
Acute Oral Toxicity
Pelargonic acid
EPA 46 placed pelargonic acid in toxicity category IV (>5000 mg/kg), primarily based on the results of the following study. The acute oral toxicity of nonanoic acid was evaluated using 2 groups of 10 specific pathogen-free Sprague-Dawley ([SD] Crl:CD) rats (5 males, 5 females/group). 57 One group was dosed orally with nonanoic acid in corn oil (dose = 5000 mg/kg). The control group was dosed with vehicle only. None of the animals died and no abnormal clinical signs were noted during the 14-day observation period. There was no evidence of macroscopic abnormalities at necropsy. The LD50 was >5000 mg/kg, and nonanoic acid was considered nontoxic.
Cetearyl nonanoate
The acute oral toxicity of cetearyl nonanoate (97% pure) was evaluated using groups of 4 SD CD rats. 58 One group was dosed orally with 300 mg/kg and the remaining 2 groups were dosed with 2000 mg/kg. None of the animals died. Signs of systemic toxicity were observed in the 300 mg/kg dose group but not in the 2000 mg/kg dose group. Necropsy did not reveal any abnormal findings, and an LD50 of >2000 mg/kg body weight was reported.
Cetearyl isononanoate
According to a manufacturer, 59 an LD50 of >5000 mg/kg was reported for cetearyl isononanoate in a study involving mice. Study details were not provided.
Isononyl isononanoate
According to a manufacturer, 60 an LD50 of >5000 mg/kg was reported for isononyl isononanoate in a study involving rats. Study details were not provided.
Ethyl pelargonate
Acute oral LD50 values of >43 000 mg/kg and 24 190 mg/kg have been reported for rats and guinea pigs, respectively. 50
Ethylhexyl pelargonate
The acute oral toxicity of undiluted ethylhexyl pelargonate was evaluated using 10 albino rats (5 males and 5 females). 61 A single oral dose of 5 g/kg body weight was administered to each animal. Dosing was followed by a 14-day observation period, and gross necropsy was performed on animals that survived. The LD50 was >5 g/kg.
Isononyl alcohol
The acute oral toxicity of isononyl alcohol using 5 fasted, male SD rats was evaluated. 52 An acute oral LD50 of 2.98 g/kg was reported.
Isotridecyl alcohol
The acute oral LD50 for isotridecyl alcohol 62 in rats is 17 000 mg/kg.
Neopentyl glycol diisononanoate
The acute oral toxicity of undiluted neopentyl glycol diisononanoate was evaluated using groups of 4 SD CD rats. 63 One group was dosed orally with 300 mg/kg and the remaining 2 groups were dosed with 2000 mg/kg. None of the animals died, and there were no signs of systemic toxicity in any of the 3 groups. Necropsy did not reveal any abnormal findings, and an LD50 of >2000 mg/kg body weight was reported.
PEG-5 isononanoate
The acute oral toxicity of PEG-5 isononanoate was evaluated using 2 groups of 3 fasted SD CD rats. 64 Animals of both groups were dosed orally with 2000 mg/kg. None of the animals died and necropsy did not reveal any abnormal findings. An LD50 of >2500 mg/kg body weight was reported.
Neopentyl glycol
The Organisation for Economic Co-operation and Development (OECD) 32 reported an acute oral LD50 of 3200 mg/kg (rats) for neopentyl glycol; others have reported the acute oral LD50 for neopentyl glycol 62 in rats is 3259 mg/kg.
Acute Dermal Toxicity
Pelargonic acid
In a study involving rabbits, 50 the acute dermal LD50 was greater than 5 g/kg (number of animals not stated). Results relating to the skin irritation potential of pelargonic acid in this study are included later in the report text. The US Environmental Protection Agency placed pelargonic acid in toxicity category III (>2000-5000 mg/kg) based on the results of the following acute dermal toxicity studies that were published in a Federal Register notice. 49 The application of pelargonic acid to intact and abraded skin of mice induced moderate-to-severe skin irritation, and an acute dermal LD50 of 9000 mg/kg was reported in this study. An acute dermal LD50 of 5000 mg/kg (rabbits) for undiluted pelargonic acid also has been reported.
The acute dermal toxicity of nonanoic acid was evaluated using 2 groups of 10 specific pathogen-free SD (Crj:CD) rats (5 males, 5 females/group). 65 The test substance, in deionized water, was placed on filter paper that was applied to clipped, shaved skin (4 × 5 cm site) of the back for 24 hours. Deionized water (0.5 mL) was applied to control animals according to the same procedure. None of the animals died during the 21-day observation period. Scales/scabs in the dorsal region (test substance related) were observed only in treated females (days 3-17 post application). Macroscopic abnormalities were not observed in any of the animals (test or controls) at necropsy. It was concluded that the LD50 for nonanoic acid in males and females was >2000 mg/kg.
Cetearyl nonanoate
The acute dermal toxicity of cetearyl nonanoate (97% pure) was evaluated using 10 SD CD strain rats (5 males and 5 females). 58 The test substance was applied to intact skin (24-hour semiocclusive application) at a dose of 2000 mg/kg body weight. None of the animals died and there were no signs of systemic toxicity or dermal irritation. Necropsy findings were not indicative of any abnormalities, and an LD50 of >2000 mg/kg body weight was reported.
Ethyl pelargonate
The acute dermal LD50 in rabbits 50 exceeded 5 g/kg.
Isononyl alcohol
The acute dermal toxicity of undiluted isononyl alcohol was evaluated in a study using 4 rabbits. 52 The test substance was applied (under occlusive binding) to clipped, abraded abdominal skin at the following doses: 0.500, 0.200, 0.794, and 3.16 g/kg. An acute dermal LD50 of 3.2 g/kg was reported. Signs of percutaneous toxicity were not observed.
Neopentyl glycol diisononanoate
The acute dermal toxicity of undiluted neopentyl glycol diisononanoate was evaluated using 10 SD CD strain rats (5 males and 5 females). 63 A dose of 2000 mg/kg body was applied according to the procedure in the preceding section on cetearyl nonanoate. None of the animals died and there were no signs of systemic toxicity or dermal irritation. Necropsy findings were not indicative of any abnormalities, and an LD50 of >2000 mg/kg was reported.
Acute Intravenous Toxicity
The acute intravenous (iv) toxicity of pelargonic acid using 10 mice was studied. 66 An LD50 of 224 ± 6 mg/kg was reported. Similarly, an LD50 of 224 mg/kg was reported for mice dosed iv with undiluted pelargonic acid. 49
Acute Intraperitoneal Toxicity
In a study involving rats, intraperitoneal (ip) dosing with undiluted pelargonic acid resulted in death and the lowest lethal dose (LDLo) was 3200 mg/kg. 49 The dosing (ip) of mice with a 10% solution of pelargonic acid in corn oil resulted in death, and an LDLo of 1600 mg/kg was reported. 49
Short-Term Oral Toxicity
Pelargonic acid
A study was conducted to determine the appropriate dose level of pelargonic acid for a teratology screening study. 67 Groups of 6 cesarean-derived, SD rats (sexually mature; weights = 177-285 g) were used. The test substance was administered (via oral intubation) in corn oil, at 3 dose levels (200, 1000, and 2000 mg/kg per d; 6 inseminated females/dose level) from 6 to 15 days of gestation. The dose volumes corresponding to the administered doses (lowest to highest) were 1.0, 5.0, and 10.0 mL/kg. The control group (6 inseminated females) was dosed with corn oil according to the same procedure. All surviving rats were killed after gestation day 15 and necropsied.
There were no remarkable clinical signs in any of the rats dosed with pelargonic acid, and none of the rats died. Mean feed consumption in the 200 mg/kg dose group was significantly higher, up to gestation day 14, when compared to the control group. Gross pathology findings were observed principally in the lungs, kidneys, or stomach. The numbers of rats with gross lesions included 2 rats at the 200 mg/kg dose level and 1 rat each at the 1000 and 2000 mg/kg levels. It was agreed that the only pelargonic acid-induced effect was on body weight. Study results relating to reproductive and developmental toxicity are included in the Section on Reproductive and Developmental Toxicity later in the report text. 67
In another short-term study, 68 the oral toxicity of pelargonic acid was evaluated using groups of 6-week-old albino rats (Crl:CD (SD) BR strain). Four groups (15 males and 15 females per group) were fed pelargonic acid in the diet at concentrations of 10, 100, 1000, and 5000 ppm, respectively. Ten rats per sex in each group received their respective diet for 28 days and until necropsy; a fifth group received basal diet only. After 28 days of pelargonic acid (in diet) feeding, the remaining rats (5 males and 5 females per group) in the 4 groups were switched to a basal diet, and feeding was continued for an additional 56 days (recovery phase). The fifth group was allowed to continue on the basal diet. Necropsy was also performed at the end of the recovery phase.. Compared to the control group, male rats in the 5000 ppm dietary group had significantly lower (P < .05) group mean body weights during weeks 1 through 4. The same was true for female rats in this group during weeks 3 through 6. Changes in mean body weights, body weight gains, and food consumption in the 100 and 1000 ppm dietary groups were influenced by an approximately 30-hour water deprivation, which occurred during week 3. Reversible changes in clinical pathology variables (blood/urine) following dietary administration of 5000 ppm pelargonic acid were noted.
Treatment-related morphologic changes were noted in the hearts and livers of rats killed after 28 days. Changes in the heart were also observed in male rats during the recovery phase but at a lower incidence and severity. Liver lipid content was greater in female rats on diets containing 100, 1000, and 5000 ppm pelargonic acid. The lower body weights in rats killed after 28 days resulted in greater relative weights in a number of organs. Absolute liver weights were greater in male rats that received 5000 ppm pelargonic acid and in female rats that received 1000 and 5000 ppm pelargonic acid in the diet. All other changes that were observed were considered to have been of no toxicological importance. It was concluded that pelargonic acid appeared to have increased the risk of cardiac changes in treated male and female rats and hepatic changes in female rats that received 5000 ppm in the diet. Changes in the liver were not observed at 56 days posttreatment, while cardiac changes persisted at a reduced intensity.
The lowest observable effect level (LOEL) for pelargonic acid was 100 ppm for antemortem data (lower body weights) and 5000 ppm for clinical pathology in rats of both sexes. Taking into consideration the increased liver weights observed after dosing with pelargonic acid, the LOEL was 5000 ppm for male rats and 100 ppm for female rats; the LOEL for macroscopic effects on the liver was 1000 ppm in rats of both sexes. Regarding both cardiac and hepatic effects, the LOEL for macroscopic changes was 1000 ppm (male rats) and 100 ppm (female rats). However, because histopathology was not performed on livers from lower dose rats from the scheduled sacrifice, the LOEL for microscopic liver changes may actually be lower than these values. 68
A short-term oral toxicity study was conducted using groups of 6 SD albino rats (3 males, 3 females/group). 69 Six groups were fed pelargonic acid in the diet at concentrations ranging from 1500 ppm to 20 000 ppm (1 dietary concentration per group) for 2 full weeks. A seventh group (control) was fed untreated feed. Feeding with pelargonic acid did not induce any adverse effects over the range of concentrations evaluated. Body weight gain and food consumption were normal throughout the study. Other than piloerection (not dose related), no clinical signs were observed. All animals appeared healthy and normal at the time of necropsy. Hematology parameters were all within normal limits, and the same was true for most of the serum clinical chemistry parameters.
Except for the lowest dose group, mean serum alkaline phosphatase (ALP) activity was significantly greater than the control value. Effects on ALP activity were not considered toxicologically significant relative to liver function, taking into consideration the absence of an effect of pelargonic acid on serum alanine aminotransferase (ALT) and serum protein content. Total bilirubin was elevated in some of the groups, controls included high values correlated primarily with the presence of hemolysis in individual blood samples. It was concluded that pelargonic acid did not induce overt signs of toxicity in albino rats, when fed in the diet at concentrations up to 20 000 ppm (2%) for 2 weeks. 69
In another study, 49 8 male rats (weights not stated) were fed 4.17% pelargonic acid in the diet (2100 g/kg per d) for 4 weeks. A slight decrease (4%, not statistically significant) in mean growth was observed. No effects on survival were noted.
Isononyl Isononanoate
The short-term oral toxicity of isononyl isononanoate was evaluated using 4 groups of SD rats (10 males, 10 females/group) of the Caesarian Obtained, Barrier Sustained-Virus Antibody Free (COBS-VAF) strain. 70 One group served as the vehicle control (corn oil) group, and the 3 test groups received the following doses of isononyl isononanoate (in corn oil) by gavage daily for 4 weeks: 100, 300, and 1000 mg/kg per d. Test substance-related mortalities were associated with 1 female in the 300 mg/kg per d dose group and 4 females in the 1000 mg/kg per d dose group. Ptyalism was the only test substance-related clinical sign, and there were no remarkable hematological findings. A correlation between lower body weight gain and lower food consumption was evident only in the highest dose group.
Doses of 300 and 1000 mg/kg per d were associated with higher enzyme activities, namely aspartate aminotransferase (AST), ALT, and/or ALP. Also, compared to controls, the blood urea level was higher in males and females of all dose groups; these changes were attributed to pathological changes in the liver and kidneys, which will be mentioned later. Additionally, it was suggested that the high urine volume associated with animals of the highest dose group was related to kidney damage noted at microscopic examination. Higher absolute and relative liver and kidney weights were noted in animals of all dose groups, and lower absolute and relative spleen and thymus weights were observed in 300 and 1000 mg/kg per d dose groups.
The treatment-related macroscopic findings included enlargement and an accentuated lobular pattern and/or paleness of the liver and a gray/green color of the kidneys in some of the animals from each dose group. Other relevant findings were described as a reduction in size of the spleen and/or thymus in some of the animals from the 300 and 1000 mg/kg per d dose groups. These findings correlated with contracted spleen and lymphoid depletion in the thymus at microscopic examination and were considered secondary to the poor physical condition of several treated animals. The treatment-related microscopic findings were described as follows: hepatocellular hypertrophy of the liver (300 and 1000 mg/kg per d dose groups); liver steatosis (all dose groups); acidophilic globules in cortical tubular epithelium (kidneys), associated with cellular damage in males (all dose groups); vacuolated cortical tubular epithelium in females (300 and 1000 mg/kg per d dose groups); and contracted spleen and thymic lymphoid depletion (1000 mg/kg per d dose group).
Isononyl isononanoate induced mortality at doses of 300 and 1000 mg/kg per d and liver and kidney (target organs) toxicity in rats at all doses administered. Under the conditions of this experiment, it was not possible to establish a no observed effect level (NOEL) for isononyl isononanoate. 70
Comments received on the preceding study suggest that the findings from the study may not be relevant to humans. 71 High-fat diets produce adaptive changes in the liver and kidneys of rodents, and, if maintained on high-fat diets for long periods, these changes may develop into a pathologic condition such as fatty liver or steatosis. Steatosis at an isononyl isononanoate dose of 100 mg/kg per d and higher doses in the preceding study should not be considered a toxicological adverse end point, but an exacerbation of an adaptive response to administration of a fatty material. Regarding changes in the kidneys, the authors suggested that mineral and other oils are likely to induce acidophilic globules in the kidneys (hyaline droplet nephropathy), due to the sex-linked production of α-2-microglobulin, and, understandably, this effect was observed in male rats of all isononyl isononanoate treatment groups. However, because the α-2-microglobulin protein (under androgen control) is absent from man and many species, this sex- and species-specific hyaline droplet nephropathy is not considered relevant to man.
Isononyl alcohol
Isononyl alcohol (in polyethylene glycol 300) was administered by gavage to 5 male rats (Alderly Park Wistar-derived) for 14 days at a dose level that was equivalent to 1 mmol/kg per d. 72 Control animals (10 rats) were dosed with polyethylene glycol 300 (10 mL/kg per d). Livers were removed, weighed, and homogenized for enzyme assays. Testis weights were also determined. No major pathological signs of hepatotoxicity resulted from oral dosing with isononyl alcohol. Minor histological changes consisted of slight centrilobular hypertrophy and fat type vacuolation in control and test animals. No effects on body weight gain or testis weight were noted. Isononyl alcohol also did not induce peroxisome proliferation, hypocholesteremic/hypotriglyceridemic effects, or effects on catalase. However, compared to controls, isononyl alcohol dosing resulted in slight elevation of palmitoyl CoA oxidase (marker enzyme for peroxisome proliferation).
Neopentyl glycol
A combined repeated dose and reproductive/developmental toxicity study on neopentyl glycol was performed using groups of male and female rats of the Slc: SD strain. 32 The test substance, in distilled water, was administered by gavage at doses of 100, 300, or 1000 mg/kg per d. Control rats were dosed with distilled water. Male rats were dosed over a 42-day period, and female rats were dosed from 14 days before mating to day 3 of lactation. There were no dead or abnormal animals with clinical signs related to dosing. Body weight and food consumption data were not indicative of consistent or treatment-related differences between test and control groups. Liver and kidney weights (absolute and relative) were increased in male and female rats of the 300 and 1000 mg/kg dose groups. Necropsy revealed hypertrophy of the liver in 2 males dosed with 1000 mg/kg; definite lesions were not found at microscopic examination. A high incidence of protein casts, hyaline droplets, and basophilic change was reported for renal tubules in males dosed with 1000 mg/kg. The no observed adverse effect level (NOAEL) for this study was of 100 mg/kg.
Short-Term Dermal Toxicity
Pelargonic acid
A 28-day dermal toxicity study 73 was conducted using groups of New Zealand White rabbits (5 males, 5 females/group). Pelargonic acid (25% weight/weight [w/w] mixture in mineral oil) was applied to the skin at doses of 500 mg/kg per d (dose volume = 2 mL/kg) daily for a total of 10 applications. The test substance was applied directly to the skin and spread evenly over the test site; patches were not applied. The skin of half of the rabbits per group (3 males and 2 females) was abraded prior to application. The control group was dosed with mineral oil according to the same procedure. For necropsy, 6 rabbits per group (3 with abraded skin and 3 with intact skin) were killed at 2 weeks and surviving animals were killed at 4 weeks.
Slight weight loss (0.1-0.4 kg) was noted in most of the rabbits dosed with pelargonic acid after 1 and/or 2 weeks of the study. Weight gain was noted in rabbits that were held for a 2-week recovery period. Slight-to-moderate weight gains were also noted in vehicle control rabbits. None of the rabbits dosed with pelargonic acid died. Skin reactions are summarized in the section on Skin Irritation later in the report text. Discoloration of the gastric mucosa was observed in treated animals; other gross morphologic findings in treated and/or control animals were not considered treatment related. Inflammatory changes observed in the kidneys, lungs, and brain and, less frequently, in other organs were not considered treatment related. 73
Undiluted pelargonic acid (25 µL) was applied to both ears (dorsum) of inbred CBA/Ca mice (groups of 4) once per day for 3 consecutive days. 74 None of the animals died.
Following intermittent dermal application of pelargonic acid to the skin of mice over a 3-day period, the 3 mL/kg dose was the lowest dose that caused a toxic effect (TDLo). In a similar study involving mice, a TDLo of 3000 mg/kg was reported. 75
Isononyl isononanoate
The short-term dermal toxicity of isononyl isononanoate was evaluated using 4 groups of SD rats (5 males, 5 females/group) of the COBS-VAF strain with healthy, intact skin. 76 One group served as the vehicle (corn oil) control, and the test groups received cutaneous doses of isononyl isononanoate in corn oil daily for 8 days (860 mg/kg per d dose group) or for 2 weeks (100 and 300 mg/kg per d dose groups). Doses of the test substance or control were applied for 6 hours to a 45 to 50 cm2 area (males) or a 30 to 35 cm2 area (females) on backs that had been clipped free of hair. A constant dose volume of 1 mL/kg per d was used. None of the animals died. Slight cutaneous reactions were observed in the 100 and 300 mg/kg per d dose groups (1 animal/group). However, severe skin irritation and necrosis were observed at the application sites of animals of the 860 mg/kg per d dose group and treatment was discontinued after day 8 of dosing.
A correlation between decreased body weight gain and decreased feed consumption was evident only in the highest dose group. Low white blood cell counts were also noted in the highest dose group; however, these changes were considered related to inflammatory reactions and the tissue distribution of inflammatory cells at the application site. Changes in blood biochemistry were noted in each dose group, all of which were treatment related. A high urea level and high ALP enzyme activity were noted in 300 and 860 mg/kg per d dose groups. High AST enzyme activity was noted only in the highest dose group, but neither ALP nor AST activity was high in the lowest dose group.
A gray/green coloration of the kidneys was observed in the mid- and high-dose groups, and this finding was correlated with acidophilic globules in the cortical tubular epithelium of high-dose male rats. An accentuated lobular pattern in the liver was noted in all dose groups, and this finding was correlated with steatosis and hepatocellular hypertrophy noted at microscopic examination and considered related to 300 and 860 mg/kg per d doses. Cortical cell hypertrophy in the adrenal glands ranging in severity from minimal to moderate was observed in the highest dose group. Whether this finding was considered treatment related was not stated specifically. The adrenal glands and the liver were considered target organs for isononyl isononanoate toxicity. Under the conditions of this experiment, it was not possible to establish an NOEL for isononyl isononanoate. 76
Comments on the preceding study were received suggesting that since it is unlikely that isononyl isononanoate penetrates the skin, the steatosis was caused by significant oral exposure secondary to grooming and licking of the application site. 71 As stated above, study results indicate that steatosis was observed in all isononyl isononanoate dose groups and that acidophilic globules were observed in the kidneys of high-dose male rats. Furthermore, some of the effects observed in this dermal study were of a similar order of incidence and severity as those observed at the same dose levels in the short-term oral toxicity study on isononyl isononoate (summarized earlier in report text)..
Subchronic Oral Toxicity
Cetearyl isononanoate
A summary of a 1993 subchronic oral toxicity study on Cetiol SN (cetearyl isononanoate, percentage not stated)) was provided by the Council. 77 Three groups of Wistar rats of both sexes received oral doses of 100, 300, and 1000 mg/kg body weight, respectively, over a period of 90 days. A fourth group served as the untreated control. Reversible fatty alterations of the liver were observed in the 1000 mg/kg dose group and in females of the 300 mg/kg dose group. Based on these results, it was determined that the NOAEL should be 100 mg/kg per d. The authors suggested that branched acids, like isononanoic acid, undergo a specific type of metabolism in rodents, and that the fatty alterations in the liver reflect an adaptive response due to increased metabolic activity. The relevance of these changes in the liver of humans was placed in doubt, and, thus, it was anticipated that the NOAEL for human-relevant effects would be >100 mg/kg body weight.
Ethyl pelargonate
No effects were observed at microscopic examination of the following tissues from rats (5 males and 5 females) fed 1% ethyl pelargonate in the diet for 16 weeks: liver, kidney, heart, spleen, testes, viscera, and hind limb. 50 Terminal hematological examinations and gross pathology and weights of the liver, kidney, heart, spleen, and testes did not differ from the findings in control rats.
Ocular Irritation/Toxicity
Pelargonic acid
The EPA 46 placed pelargonic acid in toxicity category II (corneal involvement or other eye irritation clearing in 8-21 days), based primarily on the results from the following primary ocular irritation study involving 6 Hra: (NZW)SPF adult albino rabbits. 78 The undiluted test substance (0.1 mL) was instilled into the right eye of each animal, and untreated left eyes served as controls. Instillation was followed by a 21-day observation period. Pelargonic acid induced corneal and iridial involvement and severe conjunctival irritation. All reactions had cleared by day 21 postinstillation. The average primary irritation score (5-animal mean) was 40.6 at 1 hour and 0 at day 21.
Pelargonic acid was a mild irritant when instilled into the rabbit eye at a dose of 0.1 mL. 75
Cetearyl nonanoate
The ocular irritation potential of cetearyl nonanoate (97% pure) was evaluated using 3 male New Zealand white rabbits. 58 The test substance (0.1 mL) was instilled into the right eye of each animal and reactions were scored at approximately 1, 24, 48, and 72 hours post instillation. Moderate conjunctival irritation was observed; however, there were no changes in the cornea or iris. All eyes appeared normal at 48 hours post instillation. Cetearyl nonanoate was classified as minimally irritating to the rabbit eye.
Cetearyl isononanoate
A summary of a 1970 study evaluating the ocular irritation potential of Cetiol SN (cetearyl isononanaote, percentage not stated) was provided by the Council. 77 Cetiol SN (10% active matter, 0.05 mL) was instilled into the eyes of 2 rabbits, and reactions were scored for up to 72 hours post instillation. The test substance was classified as a nonirritant. In a 1991 study summary provided by the Council, a homologue, cetyl ethylhexanoate, was applied undiluted (0.1 mL) to the eyes of 3 rabbits and remained for 24 hours. Mild conjunctival reactions (erythema, edema, and lacrimation) were observed, all of which had cleared by 72 hours post instillation. Cetyl ethylhexanoate was classified as slightly irritating to the eyes of rabbits. 77
Isononyl isononanoate
A manufacturer reported that isononyl isononanoate was not irritating to the eyes of rabbits. 60 Study details were not provided.
Ethylhexyl pelargonate
The ocular irritation potential of undiluted ethylhexyl pelargonate was evaluated using 6 New Zealand white rabbits. 61 Eyes were not rinsed following the instillation of the test substance (0.1 mL). Contralateral eyes served as controls. Reactions were scored up to 72 hours post instillation. Ethylhexyl pelargonate was not irritating to the eyes of rabbits.
Neopentyl glycol diisononanoate
A study evaluating the ocular irritation potential of neopentyl glycol diisononanoate in rabbits was conducted according to the procedure in the preceding section on cetearyl nonanoate. 63 Neopentyl glycol diisononanoate (0.1 mL) produced similar results and also was classified as a minimal ocular irritant.
PEG-5 isononanoate
The ocular irritation potential of PEG-5 isononanoate was evaluated using 3 female SPF albino rabbits, 64 according to the procedure in the preceding section on cetearyl nonanoate. Conjunctival redness and edema had cleared within 14 days, and PEG-5 isononanoate was classified as a nonirritant.
Isononyl alcohol
In a study, undiluted isononyl alcohol was instilled (0.1 mL) into the left conjunctival sac of each of 6 rabbits. 52 Untreated eyes served as controls. Ocular irritation reactions were scored using the Draize scale (0-110). Draize median irritation scores of 30 (at 24 hours) and 2 (at day 7) were reported. The ocular irritation induced by isononyl alcohol was classified as marked. The test substance did not produce severe opacity or other corneal effects, such as sloughing or vascularization.
Skin Irritation
Pelargonic acid
Pelargonic acid (concentration not stated) was classified as a strong skin irritant in guinea pigs, and a moderate irritant when applied undiluted (under occlusion) to abraded or intact skin of rabbits 50 for 24 hours.
In a 28-day dermal toxicity study (in the section on short-term dermal toxicity earlier in report text), 73 slight-to-severe erythema and edema without necrosis or eschar formation were observed in most of the rabbits during the first week of the study. Generally, during the second week, necrosis and eschar formation were observed in all rabbits. Atonia, desquamation, fissuring, and exfoliation were also observed. In rabbits held for recovery, dermal responses subsided. At microscopic examination, epidermal necrosis, hyperplasia, and hyperkeratosis were noted at the application site. Diffuse and perifollicular dermal inflammation was also common. The skin application sites in all surviving animals appeared healed by 2 weeks posttreatment.
The EPA 46 placed pelargonic acid in toxicity category II (severe irritation at 72 hours [severe erythema or edema]), based primarily on results from the following skin irritation study involving 6 adult female albino rabbits of the Hra: (NZW) SPF strain. 79 The test substance (undiluted) was applied to intact skin of the back (0.5 mL, exposure area ≈6.25 cm2), and the site was covered with a semiocclusive patch for 4 hours. Reactions were scored up to day 21 post removal. Skin irritation was observed in all animals; reactions ranged from moderate-to-severe erythema and edema. The average of the 4-, 24-, 48-, and 72-hour scores was 5.6 (severely irritating). The following observations were also made at application sites: subcutaneous hemorrhaging, blanching, desquamation, fissuring, possible necrotic areas, denuded areas, and possible scar tissue. With the exception of a denuded area in 1 animal, all irritation reactions had cleared by day 21.
In a study, undiluted pelargonic acid (25 µL) was applied to both ears (dorsum) of inbred CBA/Ca mice (groups of 4) once per day for 3 consecutive days. 74 Skin irritation (erythema and edema) was not observed at the test sites. LLNA results are included in the section on skin irritation and sensitization later in the report text.
According to Scientific and Technical Information Network (STN), 75 pelargonic acid (500 mg dose) was moderately irritating, following application to rabbit skin for 24 hours. Undiluted pelargonic acid was classified as a severe irritant, following application to guinea pig skin.
Cetearyl nonanoate
The skin irritation potential of cetearyl nonanoate (97% pure) was evaluated using 3 male New Zealand white rabbits. 58 The test substance (0.5 mL) was applied to skin clipped free of hair and the application site was covered with a semiocclusive patch for 4 hours. Reactions were scored 1 hour after patch removal and 24, 48, and 72 hours later. Cetearyl nonanoate was classified as a nonirritant.
Cetearyl isononanoate
A manufacturer reported that cetearyl isononanoate was not irritating to the skin of rabbits. 59 Study details were not provided. Repeated applications of undiluted Cetiol SN (cetearyl isononanoate, percentage not stated) were made to the skin of 5 hairless mice for a total of 5 days. It was concluded that the test substance was slightly irritating to the skin. 77 A homologue, cetyl ethylhexanoate, was applied undiluted (0.5 mL) to dorsal shaved skin of 3 rabbits under semiocclusive conditions for 4 hours. Slight-to-moderate erythema and edema were observed for up to 72 hours post application, and eschar was observed at 1 week post application. All reactions cleared within 14 days, and cetyl ethylhexanoate was classified as a slight-to-moderate skin irritant. 77
Isononyl isononanoate
A manufacturer reported that isononyl isononanoate 60 was slightly irritating to the skin of rabbits tested according to OECD method 404. Additional study details were not provided.
PEG-5 isononanoate
The skin irritation potential of undiluted PEG-5 isononanoate was evaluated using 3 male New Zealand white rabbits. 64 The test substance (0.5 mL) was applied to skin clipped free of hair and the application site was covered with a semiocclusive patch for 4 hours. Reactions were scored 1 hour after patch removal and 24, 48, and 72 hours later. The test substance induced well-defined erythema and very slight edema; no corrosive effects were observed. PEG-5 isononanoate was classified as a mild irritant (primary irritation index [PII] = 2).
Ethyl pelargonate
Moderate skin irritation was observed after undiluted ethyl pelargonate was applied, under occlusion, to intact or abraded skin of rabbits for 24 hours. 50
Ethylhexyl pelargonate
The skin irritation potential of undiluted ethylhexyl pelargonate was evaluated using 6 New Zealand white rabbits. 61 The test substance (0.5 mL) was applied to intact and abraded skin sites that remained occluded for 24 hours. Reactions were scored at 24 and 72 hours postapplication. Ethylhexyl pelargonate did not induce skin irritation in any of the rabbits (PII = 0.40).
Isononyl alcohol
The results of an acute dermal toxicity study 52 on undiluted isononyl alcohol (4 rabbits, abraded skin) are summarized earlier in the report text. The test substance was applied under an occlusive binding for 24 hours, and the doses administered ranged from 0.500 to 3.16 g/kg. In this study, dermal irritation (erythema and edema) was classified as marked overall. Both the intensity and duration of skin irritation were dose related. Atonia and desquamation, with some necrosis or eschar, were persistent findings. Some of the irritation observed was associated with the trapping of liquid under the occlusive binder at a point where the binder was bound to the animal. These areas of intimate contact and pressure gave rise to some of the reported necrosis and eschar.
Inflammation
A study to examine the tissue response of pelargonic acid in the buccal mucosa of the rat was conducted. 80 Both the methyl ester and propyl ester of pelargonic acid (both in acetone) were tested using groups of 6 SD rats (3 months old). The protocol used consisted of sensitization (dorsal skin) with 2% pelargonic acid (both solutions; dose volume = 100 µL) and challenge in the buccal mucosa (dose volume = 50 µl) with different concentrations of the sensitizing solution (0.2% and 2.0%). The area of the application site (cm2) was not stated. Allergenic potential, as evidenced by the tissue response in the buccal mucosa, was investigated using a skin-sensitization procedure and elicitation with 2% or 0.2% solutions. Control rats were exposed to acetone only. The animals were killed 48 hours after the last application, and the right buccal mucosa was excised and prepared for microscopic examination. Cellular infiltrates in the buccal mucosa were recorded and compared to normal rat buccal mucosal.
Both test substances (at both concentrations) caused increased cellularity, mainly of the mononuclear cell type. The low concentration of the methyl ester of pelargonic acid (0.2%) induced stronger inflammatory reactions than the high concentration (2.0%). This finding was the opposite of that reported for the propyl ester of pelargonic acid. Both substances were said to have shown a sensitization tendency. Repeated applications of the propyl ester of pelargonic acid (2%) decreased the inflammatory response, when compared to 1 application. However, for the methyl ester of pelargonic acid, a clear irritative potential was noted with repeated applications. Pre-exposure of dorsal skin prior to buccal painting resulted in an enhanced reaction to pelargonic acid in methyl ester and pelargonic acid in propyl ester. 80
Comedogenicity
Cetearyl isononanoate
Repeated applications of Cetiol SN (cetearyl isononanoate, percentage not stated) to the rabbit ear at concentrations ranging from 10% to 100% did not cause any alterations or produce structures typical of comedogenicity in the infrainfundibulum of hair follicles. 77 The positive control, isopropyl myristate, was comedogenic at a concentration of 10% and non-comedogenic at a concentration of 2%.
Skin Irritation and Sensitization
Pelargonic acid
The skin sensitization potential of pelargonic acid was evaluated in a repeated insult patch test using 24 male albino guinea pigs. 81 The test group consisted of 10 animals, and negative (corn oil) and positive (2,4-dinitrochlorobenzene [DNCB]) control groups contained 10 and 4 animals, respectively. During induction, pelargonic acid (50% weight/volume [w/v] mixture in corn oil, 0.5 mL) was placed on an adhesive patch (Hill Top Chamber, 25-mm diameter) that was applied to shaved skin of the anterior left flank for 6 hours per application. Following a nontreatment period, a 6-hour challenge application of the test mixture (0.4 mL) was made to the anterior right flank of each test animal and corn oil was also applied to a new site on the anterior left flank. The 10 negative control animals were not treated during induction but received challenge applications of the test mixture and corn oil alone. The positive control was applied during induction and challenge phases.
Pelargonic acid (50% w/v mixture in corn oil) induced moderate-to-strong dermal reactions (erythema/edema) in all 10 guinea pigs during the induction phase. Dermal reactions to the mixture were not observed in the negative control animals during the challenge phase. The test mixture also did not elicit any dermal reactions in test animals during the challenge phase and was considered a nonsensitizer in guinea pigs. The positive control induced sensitization. 81
Three female BALB/c mice or female CBA/J mice (6 weeks old; Harlan) were treated with pelargonic acid for 3 consecutive days. 82 The test substance was applied topically (25 µL total/ear; application area not stated) to both ears at concentrations ranging from 20% to 80% pelargonic acid in 1-propanol (volume/volume [v/v]) and an LLNA was performed. 83
Compared to the vehicle control, pelargonic acid produced slight increases in the percenatge of B220+ lymphocytes at all doses. These findings were not dose related. Even at high concentrations, the cell number per node and the percentage of B220+ cells never approached the values that were associated with allergens such as 1-chloro-2,4,6-trinitrobenzene (TNCB) and DNCB. 82
Pelargonic acid, neat or in dimethylformamide, in the LLNA
Pelargonic acid showed a dose–response relationship and positive results when tested at concentrations of ≥50% and was classified as a potential sensitizer. 74
Pelargonic acid was administered to the dorsal and ventral surfaces of each ear of groups of 5 female B6C3F1 mice (C57BL/6 × C3HHeN at concentrations ranging from 5% to 60% in acetone) for 4 consecutive days. 85 Compared to the vehicle control, pelargonic acid produced a dose-dependent and statistically significant increase in lymph node cell proliferation at concentrations of 20%, 40%, and 60%. The no-effect-level was at a concentration of 10%. Known sensitizers (ie, oxazolone, 2,4-dinitrofluorobenzene, and toluene diisocyanate) evaluated in the assay produced marked lymph node cell proliferation.
Cetearyl nonanoate
The skin sensitization potential of cetearyl nonanoate (89% pure) in sesame oil was evaluated in a maximization test using 15 male guinea pigs. 58 Of the 15, 5 comprised the vehicle control group. Because topical induction with 50% cetearyl nonanoate in sesame oil did not induce skin irritation in a preliminary experiment, the skin was treated with sodium lauryl sulfate (SLS) in order to induce local irritation. Cetearyl nonanoate (10% in sesame oil) was administered during the first (intracutaneous) induction stage, and reactions were evaluated after 24 and 48 hours. During the second (topical) induction stage, cetearyl nonanoate (50% in sesame oil, 2 mL) was applied and reactions were scored 48 and 72 hours after the initiation of exposure. At day 21, the animals were challenged with 10% cetearyl nonanoate in sesame oil (2 mL). Skin changes were not observed following intracutaneous induction or during the challenge phase, and the same was true for the vehicle control. Cetearyl nonanoate was classified as a nonsensitizer.
Cetearyl isononanoate
Anon-GLP-sensitization study was conducted on Cetiol SN (cetearyl isononanoate, percentage not stated). 77 During induction and challenge phases, the test substance (25%) was injected intracutaneously (10 injections within 14 days) into 5 male guinea pigs of the Pirbright White strain. The skin reactions observed in test animals did not differ from those observed in the control group.
Neopentyl glycol diisononanoate
A maximization test on neopentyl glycol diisononanoate was performed according to a slight modification of the preceding test procedure. 63 Undiluted test material was applied during the second induction and challenge phase. Initially, the skin was treated with SLS because topical induction with undiluted neopentyl glycol diisononanoate did not induce skin irritation in a preliminary experiment. Neopentyl glycol diisononanoate was classified as a nonsensitizer.
PEG-5 isononanoate
The skin sensitization potential of PEG-5 isononanoate in CBA/Ca mice (groups of 4) following topical application was evaluated in the local lymph node assay. 64 The undiluted test substance and concentrations of 25% and 50% in acetone/olive oil were applied to the dorsal surface of the ear. The control group was treated with vehicle only. The stimulation index (SI) was expressed as the mean radioactive incorporation for each treatment group divided by the mean radioactive incorporation of the vehicle control group. The SI values of 1.70 (25% concentration), 2.42 (50%), and 1.85 (100%) were reported, and PEG-5 isononanoate was classified as a nonsensitizer.
Reproductive and Developmental Toxicity
Pelargonic Acid
A study was conducted to determine the appropriate dose level of pelargonic acid for a teratology screening study involving cesarean-derived, SD rats. 49 Details relating to the conduct of this study are included in the section on short-term oral toxicity earlier in the report text. The number of corpora lutea per ovary and the number and placement of uterine implantations, resorptions, and live and dead fetuses were recorded. Mean ovarian and uterine weight data were comparable between treated and control groups. No treatment-related reproductive effects were noted over the range of administered doses (1.0-10.0 mL/kg).
A study to evaluate the embryo/fetal toxicity and teratogenic potential of pelargonic acid was conducted using groups of 22 mated female Crl:COBS, CD (SD)BR rats (14 weeks old). 86 Females of the test group were dosed orally (by gavage) with pelargonic acid (in corn oil; dose = 1500 mg/kg) on gestation days 6 through 15. The control group received corn oil according to the same procedure. Pregnant females were killed on day 20 and fetuses were delivered by cesarean section. Neither test substance-related maternal toxicity nor effects on food and water consumption were observed in the test or control group. Additionally, there was no definitive evidence of teratogenic effects in the test or control group.
The EPA 46 reported the results of a developmental toxicity study involving rats. Treatment of the animals with pelargonic acid had no adverse effects on clinical signs, body weight gain, or food/water consumption. Fetal toxicity was not observed in treated rats or in untreated control rats, and the following parameters were comparable between treated and control rats: mean number of viable fetuses, early or late resorptions, implantation sites, corpora lutea, pre- and postimplantation losses, sex ratios, and fetal body weights. The NOEL for maternal and developmental toxicity was 1500 mg/kg per d, and the LOEL was greater than 1500 mg/kg per d.
Cetearyl Isononanoate
In a teratogenicity study on Cetiol SN (cetearyl isononanoate, percentage not stated), 3 groups of pregnant CD rats received oral doses (gavage; dose volume = 10 mL/kg) of 100, 300, and 1000 mg/kg body weight, respectively, from day 6 to 15 of gestation. 77 A fourth group served as the untreated control. None of the animals died and maternal body weight gain was not affected by treatment. All of the females had viable fetuses, and preimplantation loss and mean numbers of resorptions were not affected by treatment. Nondose-related postimplantation loss was observed in treatment groups. All parameters were said to have been comparable to those of the control group. The results of skeletal and visceral examinations did not provide evidence of any treatment-related malformations. The NOAEL for maternal toxicity and embryotoxicity/fetotoxicity was 1000 mg/kg body weight.
Isononyl Isononanoate
The developmental toxicity of isononyl isononanoate (in corn oil) was evaluated using groups of 10 mated female SD rats. 87 The 3 test groups received doses of 30, 100, and 300 mg/kg per d, respectively, by gavage on day 6 to day 17 post coitum. The control group was dosed with corn oil. There was no evidence of treatment-related, macroscopic postmortem findings in any of the females, and none of the animals died. There was also no evidence of total resorption or abortion. The number of implantation sites and corpora lutea per female was similar in all dose groups, and, compared to controls, the number of resorptions (early and late) and postimplantation loss per female in either dose group were similar. Additionally, there were no differences in the number of live fetuses in either dose group when compared to controls, and neither external anomalies nor malformations were observed. It was concluded that isononyl isononanoate did not induce direct embryotoxicity or fetotoxicity at doses up to 300 mg/kg per d.
Isononyl Alcohol
The developmental toxicity of isononyl alcohol using sexually mature, virgin Wistar rats of outbred strain Chbb/THOM was studied. 88 The 2 types of isononyl alcohol, both identified as CAS No. 68515-81-1, tested were isononanol type 1 (purity ≥99%) of commercial origin, consisting of roughly equivalent amounts of 3,4-, 4,6-, 3,6-, 3,5-, 4,5-, 5,6-dimethylheptanol-1; and Isononanol type 2 (purity ≥99%) produced at BASF with 4,5-diemthylheptanol-1 (∼23%), 4-methyloctanol-1 (29%), 3-ethylheptanol-1 (3%), 6-methyloctanol-1 (15%), and 3-ethyl-4-methylhexanol (1%) as main components. (Note: Based on these chemical composition data, there is reason to believe that neither chemical [type 1 or 2) is isononanol. However, it is evident that both are branched-chain nonanols.). The test substances were diluted (twice-distilled water, employing ∼0.005% Cremophor EL [PEG-35 Castor Oil] as emulsifier) to a standard dose volume of 5 mL/kg body weight. Each test substance was administered by gavage (doses ranging from 1 to 10 mmol/kg per d) to pregnant females (10/group) on days 6 to 15 post coitum. Two control groups were treated with either double-distilled water alone (control group 1) or water plus ∼0.005% Cremophor EL (control group 2).
Both isononanols (types 1 and 2) exhibited a marked degree of maternal and fetal toxicity at daily doses of 7.5 and 10 mmol/kg per d, and slight fetal effects at 5 mmol/kg per d doses. Of the fetal findings (malformations, variations, or retardations), the only ones that were significantly different from controls were the number of fetuses with skeletal retardations in the 5 mmol/kg per d dose group (p < .1, both control groups) and the number with skeletal variations in this group (p < .05; 1 control group). Dosing at 1 mmol/kg per d did not cause adverse effects. When pregnant females were dosed with 7.5 mmol/kg per d (isononanol type 1) in a supplementary experiment, the incidence of malformations (mainly related to the heart) was statistically significantly increased (P < .01; 1 control group). Resorptions and postimplantation loss were also significantly increased (P < .01; 1 control group) at this dose level. For isononanol type 2, the only significant fetal findings (7.5 mmol/kg per d doses) were the number of fetuses with skeletal retardations (P < .01, .05, 1 control group—both values), number of fetuses with skeletal variations (P < .051, 1 control group), number of fetuses with variations (P < .05, 1 control group), and number of fetuses with malformations (P < .05, 1 control group). Resorptions were also significantly increased (P < .05; both control groups).
Neopentyl Glycol
A combined repeated dose and reproductive/developmental toxicity study on neopentyl glycol was performed using groups of male and female rats of the Slc: SD strain. 32 The test substance, in distilled water, was administered by gavage at doses of 100, 300, or 1000 mg/kg per d. Control rats were dosed with distilled water. Male rats were dosed over a 42-day period and female rats were dosed from 14 days before mating to day 3 of lactation.
There were no test substance-related effects on copulation, fertility, or the estrous cycle of rats, and the same was true during the lactation period. With the exception of 1 control rat, delivery was normal for all dams. There were no test substance-related abnormal findings in any of the pups delivered. The body weight gain of pups was normal up to day 4 of lactation. Test substance-related, abnormal gross findings were not reported for stillborn, dead pups, or pups killed at day 4 of lactation. Additionally, no developmental toxic effects were associated with test substance administration. The NOAEL for neopentyl glycol (P and F1 generations) was 1000 mg/kg.
Genotoxicity
Pelargonic Acid
In an Ames test, 89 the mutagenicity of pelargonic acid was evaluated with and without metabolic activation using Salmonella typhimurium strains TA98, TA100, TA1535, TA1537, and TA1538. The test substance (in dimethyl sulfoxide [DMSO]) was evaluated at doses ranging from 100 to 5000 µg/plate in this reverse mutation assay. Pelargonic acid did not cause a positive increase in the number of histidine revertants per plate in any of the tester strains, with or without metabolic activation, and, therefore, was nonmutagenic.
The mutagenicity of pelargonic acid was evaluated in a forward mutation assay, using the L5178Y mouse lymphoma cell line. 90 In preliminary cytotoxicity assays, pelargonic acid (in DMSO) induced dose-related cytotoxicity and was completely toxic at a concentration of 4000 µg/mL (without metabolic activation) and at a concentration of 2000 µg/mL (with metabolic activation). In forward mutation assays without metabolic activation (concentrations of 150-1600 µg/mL), pelargonic acid did not induce repeatable increases in the mutant frequency. In assays with metabolic activation, dose-related increases in the mutation frequency that exceeded the minimum criterion for a positive response were observed at concentrations ranging from 50 to 600 µg/mL. These increases were primarily due to increases in small colonies considered to reflect gross chromosomal changes rather than small changes within a gene. Results for pelargonic acid were positive with metabolic activation but negative without metabolic activation.
The mutagenicity of pelargonic acid was evaluated in the in vivo micronucleus assay. 91 Three groups of ICR mice (5 males, 5 females/group) received pelargonic acid, in corn oil, at oral doses of 1250, 2500, and 5000 mg/kg, respectively. After dosing, the animals were killed and bone marrow was extracted. Pelargonic acid did not induce a significant increase in micronuclei in bone marrow polychromatic erythrocytes and was considered nonmutagenic in this assay.
The National Toxicology Program 92 evaluated the mutagenicity of pelargonic acid (in dimethylsulfoxide) in Salmonella typhimurium strains TA 98 and TA 100 using the Ames preincubation assay. Pelargonic acid was tested at doses up to 5000 µg/plate with and without metabolic activation, and results were negative in each strain.
Cetearyl Nonanoate
The mutagenicity of cetearyl nonanoate (in acetone; doses up to 5000 µg/plate) was evaluated in the Ames test using Salmonella typhimurium strains TA1535, TA1537, TA102, TA98, and TA100. 58 Results were negative with and without metabolic activation.
Cetearyl Isononanoate
A genotoxicity study evaluated Cetiol SN (cetearyl isononanoate, percentage not stated) at concentrations up to 5000 µg/plate with and without metabolic activation. 77 Neither toxicity nor reverse mutations were observed over the range of concentrations tested, and Cetiol SN was classified as nonmutagenic.
Ethylhexyl Isononanoate
The mutagencity of ethylhexyl isononanoate (in acetone; doses up to 5000 µg/plate) was evaluated in the Ames test using the bacterial strains stated immediately above. 93 Results were negative with and without metabolic activation.
Isononyl Isononanoate
The mutagenicity of isononyl isononanoate (in ethanol) was evaluated using the following Salmonella typhimurium strains with and without metabolic activation: TA1535, TA1537, TA98, TA100, and TA102. 94 Anthramine served as the positive control for assays with metabolic activation and the following chemicals served as positive controls for nonactivation assays: sodium azide, 9-aminoacridine, 2-nitrofluorene, and mitomycin C. Isononyl isononante was not mutagenic at doses up to 5000 µg/plate with or without metabolic activation. All positive controls were mutagenic.
Neopentyl Glycol Diisononanoate
The Ames test was also used to evaluate the mutagenicity of neopentyl glycol diisononanoate (in acetone; doses up to 5000 µg/plate) in the Salmonella typhimurium strains mentioned in the preceding study.
PEG-5 Isononanoate
A battery of mutagenicity tests on PEG-5 isononanoate was performed. 64 Ames test results (doses up to 5000 µg/plate ± metabolic activation) were negative in the Salmonella typhimurium strains mentioned in the preceding section on cetearyl nonanoate. The mutagenicity of a formulation containing water, trideceth-9, and 29% PEG-5 isononanoate was evaluated in a chromosomal aberration assay involving human lymphocytes in vitro. The highest test concentration of the formulation was 5000 µg formulation/mL (1450 µL PEG-5 isononanoate/mL). Dose-dependent increases in chromosomal aberrations, with metabolic activation, were within the range of the laboratory’s historical control data and, thus, considered biologically irrelevant. Clastogenicity was not observed with or without metabolic activation. The mutagenicity of the same formulation and maximum test concentration was evaluated in the mammalian cell gene mutation test (mouse lymphoma assay) using the L5178Y/TK+/− cell line with and without metabolic activation. Results were negative with and without metabolic activation.
Neopentyl Glycol
The mutagenicity of neopentyl glycol was evaluated using Salmonella typhimurium strains TA100, TA 1535, TA98, and TA1537 and Escherichia coli strain WP2 uvrA. 32 Mutagenicity was evaluated at doses up to 5000 µg/plate with and without metabolic activation. The minimum dose at which toxicity to bacteria was observed, with and without metabolic activation, was >5000 µg/plate. Results for neopentyl glycol were classified as negative in this assay. The mutagenicity of neopentyl glycol was also evaluated in an assay involving Chinese hamster CHL cells. Test substance (in distilled water) doses up to 1.0 mg/mL were evaluated and results were classified as negative.
An Ames test and a chromosomal aberration test, using Chinese hamster lung (CHL/IU) cells, on neopentyl glycol were conducted on neopentyl glycol. 95 In the latter assay, proliferating cells were treated with neopentyl glycol for 6 hours (short term) with and without metabolic activation. These cells were also treated with neopentyl glycol for 24 and 48 hours continuously without metabolic activation. Ames test results were negative. In the chromosomal aberrations test, results for neopentyl glycol were negative at doses manifesting 50% or <50% cytotoxicity (or at 5 mg/mL or 10 mmol/L). Negative chromosomal aberration test results (with and without metabolic activation) were associated with short-term as well as continuous treatment assays.
Methyl Pelargonate
The anticlastogenic potential of methyl esters of fatty acids was evaluated in vivo in the chromosomal aberration assay using Chinese hamster bone marrow cells. 96 Chinese hamsters of both sexes were gavaged (single oral dose) with the methyl ester of pelargonic acid, followed immediately by dosing with the mutagenic alkylating agent, busulfan (1,4- butandiolbismethane sulphonate). The chromosome-breaking activity of bisulfan was not modulated by the methyl ester of pelargonic acid (C9) and other short-chain fatty acids. However, the methyl esters of fatty acids ranging from lauric acid (C12) up to nonadecanoic acid (C19) reduced the rate of aberrant metaphases from 9.4% to ∼3% at doses of 100 mg/kg and less.
Carcinogenicity
The dermal carcinogenicity of undiluted pelargonic acid was evaluated using groups of 50 male C3H/HeJ mice. 97 Pelargonic acid (dose = 50 mg/kg) was applied twice weekly to interscapular skin (clipped free of hair) on the back for 80 weeks or until a neoplasm was grossly diagnosed as an advanced tumor. All surviving mice were killed between 80 and 83 weeks. Other groups included in the study were an untreated control group, a group treated with mineral oil, and a positive control group (0.05% benzo(a)pyrene in mineral oil). Sixty-six percent of the mice in the pelargonic acid-treated group survived to week 78, compared to 52% and 64% for untreated and mineral oil controls, respectively. None of the positive control mice receiving applications of 0.05% benzo(a)pyrene survived to week 78. Forty-two mice treated with pelargonic acid lived long enough to have sufficient exposure to develop a tumor within the average latent period.
The following nonneoplastic skin lesions were observed in pelargonic acid-treated mice: ulcer (7 mice), skin pigmentation (41 mice), fibrosis (48 mice), scar formation (14 mice), acanthosis (48 mice), and hyperkeratosis (40 mice). The authors noted that hyperplasia of the dermis (fibrosis), acanthosis, and hyperkeratosis are common findings in areas of mouse skin that have been clipped free of hair. There was no evidence of gross skin tumors in pelargonic acid-treated mice or in the 2 control groups. Gross skin tumors were reported for 46 positive control mice. The incidence of hepatocarcinomas in the test group was at least as high as that in the negative control groups after 80 weeks. 97
Clinical Assessment of Safety
Skin Irritation and Sensitization
Data from predictive and provocative human skin irritation and sensitization testing on pelargonic acid and related ingredients are summarized in Table 4.
Skin Irritation and Sensitization Studies on Pelargonic Acid, Nonanoate Esters, and Related Chemicals
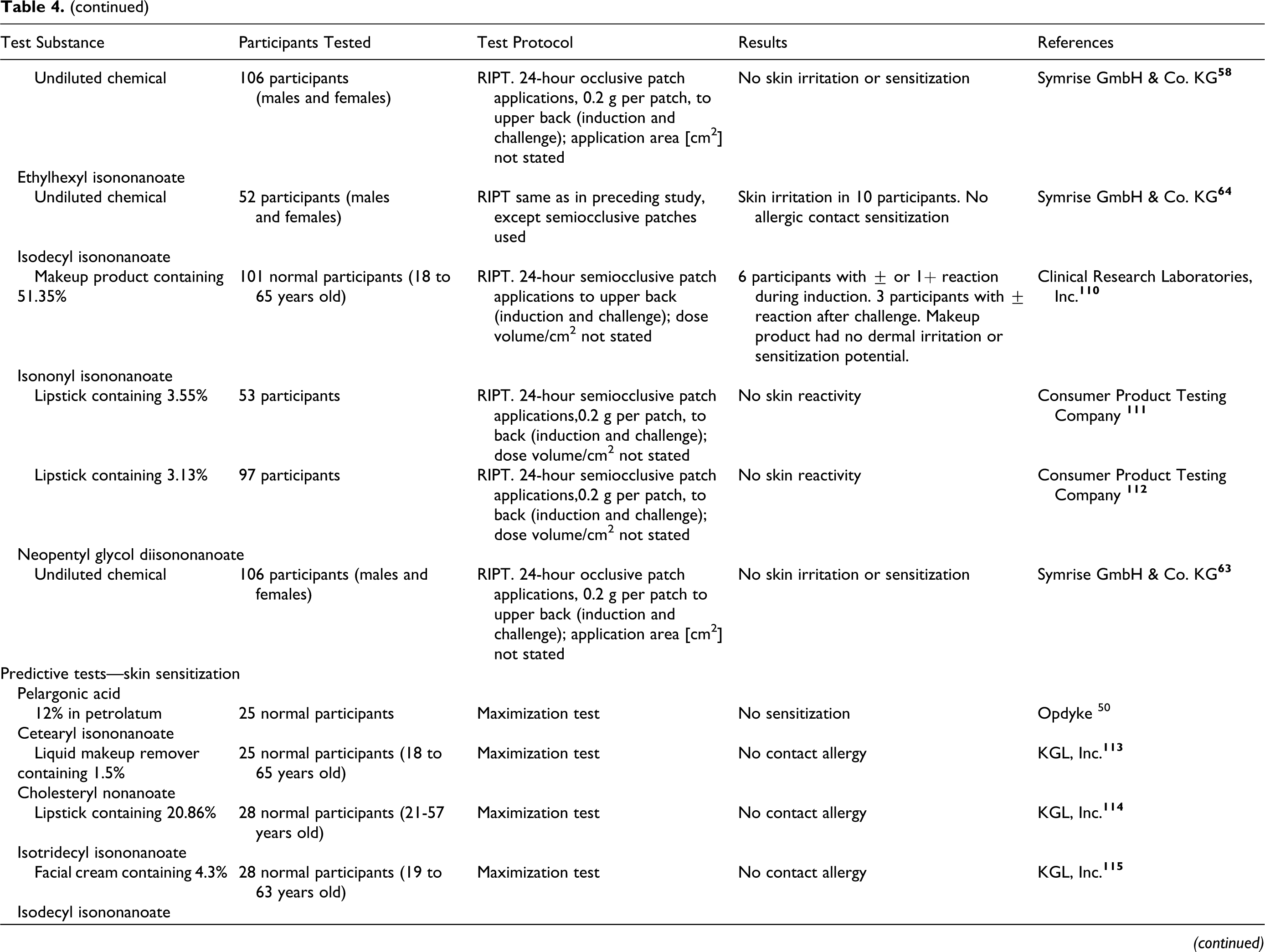
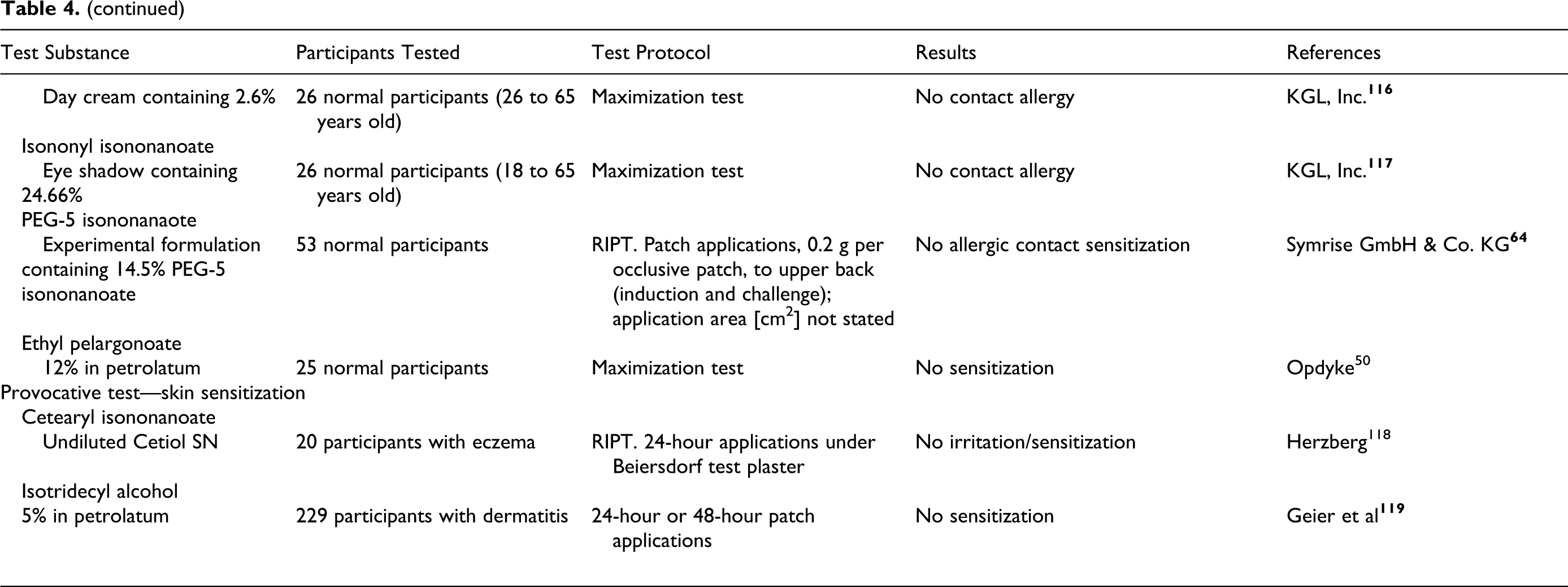
Pelargonic acid is a known skin irritant, based on the results of both predictive and provocative human skin irritation studies. In predictive tests, pelargonic acid-induced skin irritation at concentrations ranging from 5% to 80%; ethyl pelargonate was a skin irritant at a concentration of 20% but not 12%. Predictive human skin irritation test results for undiluted cetearyl nonanoate, Cetiol SN (cetearyl isononanoate, percentage not stated) at a concentration of 20%, and undiluted neopentyl glycol diisononanoate were negative, and the same was true for predictive human skin irritation and sensitization studies on cetearyl nonanoate, ethylhexyl isononanoate, and neopentyl glycol diisononanoate, all undiluted, and product formulations containing isodecyl isononanoate (51.35%) and isononyl isononanoate (3.552%). Similarly, predictive human skin sensitization studies on 12% pelargonic acid, 12% ethyl pelargonate, and formulations containing the following pelargonic acid esters were negative: cetearyl isononanoate (1.5%), cholesteryl nonanoate (20.86%), isotridecyl isononanoate (4.3%), isodecyl isononanoate (2.6%), isononyl isononanoate (24.66%), and PEG-5 isononanoate (14.5%). Results were negative for undiluted Cetiol SN (cetearyl isononanoate, percentage not stated) and 5% isotridecyl alcohol in provocative human skin sensitization studies.
Epidermal Proliferation and Apoptosis
Alterations in the proliferative capacity of human epidermis following topical exposure to pelargonic acid (80% [v/v] in propan-1-ol) were investigated 120 . Finn chambers containing the test substance were applied to the volar aspect of the forearm of each of 10 healthy, nonatopic male participants for 48 hours. Punch biopsies were removed from each application site. Samples of normal skin were also obtained. Compared to the vehicle control, pelargonic acid induced a statistically significant (P < .05) increase in the density of proliferating keratinocytes (ie, increase in mitotic activity).
The effect of 80% pelargonic acid (in propan-1-ol) on Langerhans cells and on epidermal proliferation and apoptosis was studied. 121 Punch biopsies were obtained from the volar forearm of 46 participants with irritant contact dermatitis (25 males and 21 females) and 10 healthy participants, following application of the test substance (Finn chambers) and vehicle control to the skin for up to 48 hours. A higher number of Langerhans cells/mm basement membrane in the patients, compared to controls, was reported. However, there was no difference in the number of dendrites/Langerhans cell or in dendrite length. Pelargonic acid caused a decrease in keratinocyte proliferation after 24 hours of exposure, but a return to basal levels was observed after 48 hours. Pelargonic acid induced epidermal cell apoptosis after only 6 hours of exposure and dramatically decreased the Langerhans cell number after 24 and 48 hours of exposure. Apoptosis was induced in over half of the Langerhans cells that were present after 24 and 48 hours.
Effect on CD1a and Intercellular Adhesion Molecule 1 Expression
The possibility of differences in the interaction between different irritants (pelargonic acid and SLS) and immunological parameters in the epidermis were investigated in 9 healthy participants. 122 The reactions were evaluated by immunohistochemistry using monoclonal antibodies directed against CD1a, CD3, and intercellular adhesion molecule 1 (ICAM-1) molecules. Initially, occlusive patch tests (Finn chambers) involving the following 3 groups were conducted. In group 1 (2 males and 1 female), 2% SLS and 4% SLS in distilled water (w/v) and distilled water alone were applied under occlusion for 24 hours, and biopsies were obtained at 48 hours. In group 2 (3 males), 20% pelargonic acid and 80% pelargonic acid in isopropanol (v/v), and isopropanol alone were applied under occlusion for 24 hours, and biopsies were obtained at 48 hours. In group 3 (3 males), 4% SLS in water and 80% pelargonic acid in isopropanol were applied under occlusion for 24 hours; biopsies were obtained immediately after Finn chamber removal.
At 48 hours (groups 1 and 2), marked edema was observed at the 4% SLS site; this reaction was greater in severity when compared to those induced by 20% and 80% pelargonic acid. At 24 hours (group 3), the reactions to 4% SLS and 80% pelargonic acid were similar. Reactions were not induced by the distilled water or isopropanol vehicle. At both 24 and 48 hours, an exposure-related (SLS and pelargonic acid) increase in the number of CD3+ cells in the upper part of the dermis was observed. A minor increase in CD3+ cells following exposure to distilled water, but not isopropanol, was also observed at 48 hours. Few CD3+ cells were also identified in the epidermis following SLS and pelargonic acid exposure.
Differences in ICAM-1 expression in the epidermis following SLS and pelargonic acid exposure were observed. Following SLS exposure, an increase in ICAM-1+ keratinocytes at 24 and 48 hours was noted. A tendency toward increased numbers of CD1a+ cells was noted at 48 hours after treatment with 4% SLS. A definite decrease in the number of CD1a+ cells was observed following exposure to 80% pelargonic acid. Reactivity of ICAM-1 was not detected in the epidermis following exposure to 20% or 80% pelargonic acid. Increased levels of ICAM-1 expression were observed in the epidermis following exposure to both water and isopropanol controls. In the control biopsies, 3 of 9 specimens had ICAM-1 reactivity (single cells or few keratinocytes). It was concluded that different irritants applied to the skin surface may induce different responses in epidermis measured with markers for immunological components, although the clinical picture indicates primary irritancy. 122
Case Reports
Cetearyl Isononanoate
In a case report, 123 a 23-year-old female with a history of allergic contact dermatitis developed acute dermatitis after application of a urea-based moisturizing cream containing cetearyl isononanoate. The participant was patch tested (repeated open application test) with ingredients of the cream, each diluted to a concentration identical to that in the product. Allergic reactions to the cream (day 2: ++; day 4: ++) and cetearyl isononanoate (day 2: negative; day 4: ++), both diluted to concentration of 4% in liquid mineral oil were reported. Patch test results for 4% cetearyl isononanoate were negative in 10 voluntary control participants on days 2, 3, and 4.
Isononyl Isononanoate
In a case report, 124 a 40-year-old, nonatopic female presented with contact cheilitis following application of a lipstick product containing isononyl isononanoate in 2002. In 2007, she presented with severe contact dermatitis on the eyelids following application of a new lipstick product containing isononyl isononanoate to the eyelids. Patch test results for isononyl isononanoate were positive. Additional patch testing (patch test chambers on forearm) was performed, and reactions were scored according to International Contact Dermatitis Research Group criteria on days 2 and 4 and later. Strong (vesicular) positive reactions were observed at all tested ethanolic dilutions of isononyl isononanoate, from 20% (actual use concentration in product) to 1%. The patient developed severe edema of the entire test site. Patch test results for isononyl isononanoate (5% in ethanol) were negative in 20 control participants. Further patch testing in 2008 to identify possible cross-reactions did not yield further positive reactions.
Summary of Information From Earlier CIR Safety Assessments
Propylene Glycol Esters and Diesters—Including Propylene Glycol Dipelargonate
There were limited data on many of the propylene glycol esters and diesters. There were data indicating that propylene glycol dicaprylate/dicaprate was a minimal dermal irritant and was not comedogenic. In the discussion, it was noted that the caprylic (8-carbon chain)/capric (10-carbon chain) moiety is similar to the dipelargonate (9-carbon chain) moiety. Propylene glycol dipelargonate enhanced the skin penetration of caffeine and testosterone through human stratum corneum in vitro.
Overall, the Panel relied substantially on the prior reviews of the following ingredients previously reviewed to demonstrate overall safety of this group:
propylene stearate for mutagenicity, chronic toxicity, and skin sensitization;
caprylic/capric triglyceride for reproductive toxicity, chronic toxicity, and skin sensitization;
coconut acid for chronic toxicity, tumor promotion, skin sensitization, phototoxicity, and photosensitization;
isostearic acid for skin sensitization, photosensitization, and phototoxicity;
lauric, myristic, and oleic acids for reproductive toxicity, carcinogenicity, skin sensitization, and photosensitization;
In the original safety assessment of isostearic acid, two reports that discussed metabolism were noted. One study concluded that rat liver homogenate acyl coenzyme A synthetase was found to activate isostearic acid. In another study that focused specifically on metabolism of iso-fatty acids versus straight-chain fatty acids, it was reported that metabolism is similar by the mitochondrial and microsomal fractions of rat liver homogenate. The straight-chain fatty acids are successively oxidized at the β carbon to yield 2-carbon fractions. The iso-fatty acids also follow that path but, in addition, are oxidized at the ω carbon to ultimately form 3-carbon dicarboxylic acids. The enzymes catalyzing the ω-hydroxylation are present in the mitochondrial and microsomal fractions, whereas the enzymes catalyzing further oxidation into carboxylic acids are in the soluble fractions of rat liver homogenate.
In the discussion, the Panel did note a concern about dermal penetration enhancement with propylene glycol dipelargonate. It has been shown that propylene glycol dipelargonate enhances the skin penetration of caffeine through human stratum corneum in vitro.
Butyl, Cetyl, Isobutyl, Isocetyl, Isopropyl, Myristyl, and Octyl (Now Ethyhexyl) Stearate
Few data were available that demonstrated the metabolic fate of the iso forms compared to straight-chain forms beyond noting that aliphatic esters are hydrolyzed to the corresponding alcohol and fatty acid and further metabolized. One study was provided in which isopropyl stearate, diluted in 9,10- 3 H2-labeled oleic acid, was given by gavage to thoracic duct fistula rats. Radiolabel was found in lymph lipids, suggesting a dietary origin. Less than 10% of the recovered radiolabel was in the form of the isopropyl ester and, conversely, over 95% of the radiolabel was in triglycerides, leading to the suggestion that the isopropyl ester is hydrolyzed in the intestine and that the fatty acids thus liberated are reesterified before distribution to lymph lipids.
Safety test data indicated low acute oral toxicity, no reproductive toxicity, and minimal skin irritation but no skin sensitization, phototoxicity, or photosensitization. The discussion noted the absence of data on comedogenicity.
Decyl and Isodecyl Oleate
These fatty acid esters have low acute oral toxicities, but few other toxicity data were available. They can be minimal-to-moderate dermal irritants but are not sensitizers in human and animal tests. No metabolic fate information relevant to the esters was available.
Summary
Pelargonic acid and nonanoate esters are cosmetic ingredients that function as skin-conditioning agents in cosmetics. The following ingredients are reported as being used: butylene glycol diisononanoate, cetearyl isononanoate, cetearyl nonanoate, cetyl isononanoate, cholesteryl nonanoate, diethylene glycol diethylhexanoate/diisononanoate, dipentaerythrityl pentaisononanoate, ethylhexyl isononanoate, isodecyl isononanoate, isononyl isononanoate, isotridecyl isononanoate, tridecyl isononanoate, ethylhexyl pelargonate, neopentyl glycol diisononanoate, cetearyl nonanoate, cetyl isononanoate, dipentaerythrityl pentaisononanoate, neopentyl glycol diisononanoate, PEG-2 diisononanoate, PEG-5 isononanoate, pentaerythrityl tetraisononanaote, polyglyceryl-20 octaisononanoate, and pentaerythrityl tetrapelargonate. Current ingredient use concentrations range from 0.01% (cholesteryl nonanoate) to 74% (ethylhexyl isononanoate).
The following chemicals do not absorb significantly in the 250 to 400 nm range: neopentyl glycol diisononanoate, cetyl nonanoate + stearyl nonanoate, trideceth-9 + PEG-5-isononanoate + water, glyceryl triisononnoate + glyceryl diisononanoate, and ethylhexyl isononanoate.
Straight-chain pelargonic acid esters are likely hydrolyzed to component alcohols and pelargonic acid, which is further metabolized by β-oxidation. Iso-fatty acids and straight-chain fatty acids both are metabolized at the β-carbon to yield 2-carbon fractions by mitochondrial and microsomal fractions of rat liver homogenate. Additionally, iso-fatty acids are oxidized at the ω carbon to ultimately form 3-carbon dicarboxylic acids. The enzymes catalyzing the ω-hydroxylation are present in the mitochondrial and microsomal fractions, whereas the enzymes catalyzing further oxidation into carboxylic acids are in the soluble fractions of rat liver homogenate. With the exception of pelargonic acid and ethyl pelargonate, specific information relating to the metabolism of the remaining ingredients reviewed in this safety assessment was not identified in the published literature. Branched-chain fatty acid metabolism involves initial α-oxidation, which is followed by the β-oxidation pathway.
Octanol–water partition coefficient (logP) and mw data included in the safety assessment may be used to predict the skin penetration potential of pelargonic acid and its esters/ester moieties. Most of the ingredients reviewed in this safety assessment have a logP of >5 and a mw of <500. Compounds with a logP of >5 and a mw of >500 are less likely to penetrate the skin. For example, cholesteryl nonanoate has a logP of 10 and a mw of >500, suggesting that dermal absorption is unlikely. The skin penetration enhancement effect of pelargonic acid on other chemicals has been demonstrated in vitro using human stratum corneum and hairless rat skin. The percutaneous absorption of isononyl alcohol was reported in an acute dermal toxicity study using rabbits with abraded skin and occlusion of the site.
An acute inhalation LC50 of 1.34 mg/L was reported for pelargonic acid in a study involving rats. Inhalation exposure to isononanoic acid caused a concentration-dependent decrease in respiratory frequency in mice, and an RD50 of 420 mg/m3 was reported. Few and no deaths were reported for mice/rats and guinea pigs, respectively, following inhalation exposure to isononyl alcohol at a concentration of 21.7 mg/L.
Pelargonic acid and the esters for which data are available are not significant acute oral toxicants (LD50s > 1 g/kg) or acute dermal toxicants (lowest LD50 reported = 5 g/kg).
Short-term oral dosing with pelargonic acid (in diet) yielded LOELs of 5000 ppm (for clinical pathology) and 100 ppm (for antemortem data) in rats. Overt signs of toxicity were not associated with higher doses in other short-term oral toxicity studies. Short-term oral dosing with isononyl isononanoate induced liver and kidney toxicity in rats at doses up to 1000 mg/kg per d. Limited numbers of rats given isononyl alcohol at 1 mmol/kg per d had no overt signs of toxicity. The NOAEL for neopentyl glycol in rats was 100 mg/kg. Subchronic oral dosing with 1% ethyl pelargonate (in diet) did not result in any remarkable gross or microscopic findings in rats. However, subchronic oral dosing with Cetiol SN (cetearyl isononanoate, percentage not stated) induced reversible fatty alterations in the liver of rats, and an NOAEL of 100 mg/kg per d was reported.
Repeated applications of pelargonic acid (25% in mineral oil) to the skin of rabbits in a 28-day study did not cause death, and the random inflammatory changes observed in various organs were described as spontaneous. In other studies, dermal application of pelargonic acid to mice over a 3-day period did not cause death and a TDLo of 3000 mg/kg was reported. Short-term cutaneous dosing with isononyl isononanoate induced liver and adrenal toxicity in rats at doses up to 860 mg/kg per d.
The ocular changes observed in a 28-day oral toxicity study (rats) on pelargonic acid were considered sporadic and unrelated to treatment. In studies involving rabbits, pelargonic acid (0.1 mL) was severely irritating or mildly irritating. Neither isononyl isononanoate, ethylhexyl pelargonate (0.1 mL), nor Cetiol SN (cetearyl isononanoate, percenatge not stated) at a concentration of 10% (0.5 mL) induced ocular irritation in rabbits; however, both cetearyl nonanoate and neopentyl glycol diisononanoate (0.1 mL) induced minimal ocular irritation, and isononyl alcohol (0.1 mL) induced marked ocular irritation. PEG-5 isononanoate (0.1 mL) induced transient ocular reactions but was not classified as an ocular irritant.
Undiluted pelargonic acid was a mild-to-severe skin irritant in rabbits and a severe skin irritant in guinea pigs but was not irritating to the skin of mice. However, undiluted cetearyl isononanoate was nonirritating to the skin of rabbits, but, as undiluted Cetiol SN (cetearyl isononanoate, percentage not stated), was slightly irritating to the skin of mice. The remaining studies involved rabbits only. Isononyl isononanoate (unknown concentration) and undiluted PEG-5 isononanoate were slightly/mildly irritating, and undiluted ethyl pelargonate was moderately irritating to the skin; however, undiluted isononyl alcohol induced marked skin irritation. Skin irritation was not observed following the application of undiluted cetearyl nonanoate, cetearyl isononanoate, or undiluted ethylhexyl pelargonate. Repeated applications of Cetiol SN (cetearyl isononanoate, percentage not stated) to the rabbit ear at concentrations ranging from 10% to 100% did not cause any alterations or produce structures typical of comedogenicity in the infrainfundibulum of hair follicles.
In an RIPT, pelargonic acid (50% in corn oil) induced skin irritation, but not sensitization, in guinea pigs. Sensitization test results for guinea pigs injected intracutaneously with Cetiol SN (cetearyl isononanoate, percentage not stated) at a concentration of 25% did not differ from those of control guinea pigs. In guinea pig maximization tests, cetearyl nonanoate (10% in sesame oil) and undiluted neopentyl glycol diisononanoate were nonsensitizers; results for 50% cetearyl nonanoate in sesame oil and undiluted neopentyl glycol diisononanoate (for induction) were negative in preliminary skin irritation tests. Both the methyl ester and propyl ester of pelargonic acid (both at 0.2% and 2.0%) were said to have shown a sensitization tendency in a study examining the tissue response to pelargonic acid in the buccal mucosa of the rat. In mouse LLNAs (for sensitization potential), results were positive at pelargonic acid concentrations of ≥50% and ≥20% (no effect level = 10%) and negative for PEG-5 isononanoate at concentrations up to 100%.
Daily doses of pelargonic acid up to 1500 mg/kg per d did not induce reproductive effects in inseminated female rats. Results from other studies support pelargonic acid daily doses of 1500 mg/kg per d as the NOEL for maternal/developmental toxicity in rats, and isononyl isononanoate daily doses of 300 mg/kg per d as the NOEL for developmental toxicity in rats. In a teratogenicity study on Cetiol SN (cetearyl isononanoate, percenatge not stated), the NOAEL for maternal toxicity and embryotoxicity/fetotoxicity was 1000 mg/kg body weight. Two branched-chain nonanols (perhaps incorrectly identified as isononanol) caused a marked degree of maternal and fetal toxicity in rats at daily doses of 7.5 and 10 mmol/kg per d and slight fetal effects at 5 mg/kg per d doses. Neopentyl glycol at oral doses up to 1000 mg/kg per d did not induce reproductive effects.
While 1 mammalian cell assay of pelargonic acid was positive with metabolic activation (doses up to 600 µg/mL), it was negative without metabolic activation. All other bacterial, mammalian cell, and in vivo assays for pelargonic acid (doses up to 5000 µg/plate or 5000 mg/kg) and the following pelargonic acid esters (doses up to 5000 µg/plate) were negative: Cetiol SN (cetearyl isononanoate, percentage not stated), cetearyl nonanoate, ethylhexyl isononanoate, isononyl isononanoate, neopentyl glycol diisononanoate, and PEG-5 isononanoate. Negative results were also reported for neopentyl glycol at doses up to 5000 µg/plate (bacterial cells) and up to 5 mg/mL (mammalian cells). There was no evidence of gross skin tumors in mice dosed with undiluted pelargonic acid in a dermal carcinogenicity study.
Pelargonic acid is a known skin irritant, based on the results of both predictive and provocative human skin irritation studies. In predictive tests, pelargonic acid induced skin irritation at concentrations ranging from 5% to 80%; ethyl pelargonate was a skin irritant at a concentration of 20% but not 12%. Predictive human skin irritation test results for undiluted cetearyl nonanoate, Cetiol SN (cetearyl isononanoate, percentage not stated) at a concentration of 20%, and undiluted neopentyl glycol diisononanoate were negative, and the same was true for predictive human skin irritation and sensitization studies on cetearyl nonanoate, ethylhexyl isononanoate, and neopentyl glycol diisononanoate, all undiluted, and product formulations containing isodecyl isononanoate (51.35%) and isononyl isononanoate (3.552%). Similarly, predictive human skin sensitization studies on 12% pelargonic acid, 12% ethyl pelargonate, and formulations containing the following pelargonic acid esters were negative: cetearyl isononanoate (1.5%), cholesteryl nonanoate (20.86%), isotridecyl isononanoate (4.3%), isodecyl isononanoate (2.6%), isononyl isononanoate (24.66%), and PEG-5 isononanoate (14.5%). Results were negative for undiluted Cetiol SN (cetearyl isononanoate, percenatge not stated) and 5% isotridecyl alcohol in provocative human skin sensitization studies.
In other human studies, pelargonic acid (80%) increased the density of proliferating keratinocytes and caused epidermal cell apoptosis and a transient decrease in keratinocyte proliferation.
In case reports, a moisturizing cream containing cetearyl isononanoate induced contact dermatitis and a lipstick containing 20% isononyl isononanoate induced contact cheilitis. Patch test results for 4% cetearyl isononanoate and 5% isononyl isononanoate were negative in healthy control participants in these reports.
Discussion
The CIR Expert Panel recognizes that the pelargonic acid branched esters included in this safety assessment are not pure substances but are always mixtures. The INCI name for each pelargonic acid branched ester actually refers to a mixture with general properties that, in many instances, cannot be fully characterized. Regardless, the material, as supplied to the industry for use in cosmetics, is the same material that is used in safety testing. While specific data on the metabolic fate of the branched esters included in this safety assessment were not found, data on phytanic acid, a 20-carbon branched fatty acid, indicate that the α-methylene group is oxidatively excised (α-oxidation) in mammalian microsomes, yielding pristanic acid. Pristanic acid is then metabolized via the β-oxidation pathway. These data were deemed representative of the metabolic pathways for branched fatty acids in general.
The Expert Panel noted the availability of acute inhalation toxicity data on pelargonic acid, isononanoic acid, and isononyl alcohol, but not on any other ingredients that are being reviewed in this safety assessment. However, in the absence of these data, the Panel determined that the ingredients included in this review can be used safely in hair sprays, because the product particle size is not respirable. The Panel reasoned that the particle size of aerosol hair sprays (around 38 µm) and pump hair sprays (>80 µm) is large compared to respirable particle sizes (≤10 µm). The Panel expressed concern over the findings of liver steatosis and acidophilic globules in the renal cortical tubules of male rats in both short-term oral and dermal toxicity studies on isononyl isononanoate but also acknowledged industry comments on the effect that high-fat diets produce liver steatosis. This sex- and species-specific hyaline droplet nephropathy was not considered relevant to man because the alpha-2-microglobulin protein is absent from man as well as many species.
Because animal sources of cetearyl isononanoate, cetyl isononanoate, cholesteryl nonanoate, dihydrocholesteryl nonanoate, and isostearyl isononanoate have been reported, the Panel was also concerned with the dangers inherent in using animal-derived ingredients, namely the transmission of infectious agents. The CIR Expert Panel stressed that the preceding ingredients must be free of detectable pathogenic viruses or infectious agents. Suppliers and users of these ingredients must accept responsibility for assuring that these ingredients are risk free. Tests to assure the absence of a pathogenic agent in the ingredients, or controls to assure derivation from pathogen-free sources are two approaches that should be considered.
The Expert Panel also recognized that pelargonic acid and related ingredients, because of the skin penetration enhancement property of pelargonic acid in the presence of PABA, could enhance the penetration of similar chemicals, possibly cosmetic ingredients, through the skin. The Panel cautioned that care should be taken in formulating cosmetic products that may contain these ingredients in combination with any ingredients whose safety was based on their lack of dermal absorption data, or when dermal absorption was a concern.
Panel deliberations on the safety of pelargonic acid esters focused on current ingredient use concentration data from industry in relation to human RIPT data on cosmetic products containing these ingredients. Current ingredient use concentrations ranged from 0.01% (cholesteryl nonanoate) to 74% (ethylhexyl isononanoate), and ethylhexyl isononanoate was a mild skin irritant, but a nonsensitizer, when tested undiluted. Similarly, cetearyl isononanoate, cetearyl nonanoate, and neopentyl glycol diisononanoate, each undiluted, did not induce sensitization in human RIPTs. Other data indicate that the highest ester concentration, undiluted excluded, evaluated in a human RIPT was 51.35% isodecyl isononanoate in a makeup product (nonirritant and nonsensitizer), which approaches the maximum use concentration of 59% for this ingredient. Results from a non-good laboratory practice (GLP) sensitization study on Cetiol SN (cetearyl isononanoate, percentage not stated) at a test concentration of 25% were also considered only because the skin reactions of test animals did not differ from those of controls. Based on negative results for sensitization at high ingredient test concentrations, the Expert Panel reasoned that is not likely that the pelargonic acid esters reviewed in this safety assessment would induce sensitization in the present practices of use.
Conclusion
The CIR Expert Panel concluded that butylene glycol diisononanoate, cetearyl isononanoate, cetearyl nonanoate, cetyl isononanoate, cholesteryl nonanoate, diethylene glycol diethyl-hexanoate/diisononanoate, dipentaerythrityl pentaisononanoate, ethylhexyl isononanoate, isodecyl isononanoate, isononyl isononanoate, isotridecyl isononanoate, neopentyl glycol diisononanoate, PEG-2 diisononanoate, PEG-5 isononanoate, pentaerythrityl tetraisononanaote, polyglyceryl-20 octaisononanoate, tridecyl isononanoate, ethylhexyl pelargonate, pentaerythrityl tetrapelargonate, cellobiose octanonanoate, diethylene glycol diisononanoate, dihydrocholesteryl nonanoate, glycereth-7 diisononanoate, isostearyl isononanoate, phytosteryl nonanoate, propylene glycol diisononanoate, ethyl pelargonate, isobutyl pelargonate, methyl pelargonate, neopentyl glycol dicaprylate/dipelargonate/dicaprate, and pelargonic acid are safe as cosmetic ingredients in the present practices of use and concentration described in this safety assessment (note 1).
Footnotes
Acknowledgments
F. Alan Andersen, PhD, Bart Heldreth, PhD, and Wilbur Johnson are employed by Cosmetic Ingredient Review.
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St., Suite 412, Washington, DC 20036, USA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review program is financially supported by the Personal Care Products Council.