Abstract
Sanguinarine is an alkaloid obtained from the bloodroot plant Sanguinaria canadensis and has beneficial effects on oxidative stress and inflammatory disorders. Previous reports have demonstrated that sanguinarine also exhibit anticancer properties. In the current study, we investigated the effects of sanguinarine on HT-29 human colon cancer cells. It was observed that sanguinarine treatment induces a dose-dependent increase in apoptosis of human colon cancer cells. We also investigated the effects of sanguinarine on the expression of apoptosis-associated proteins, and the results revealed that there was an increase in Bax and a decrease in B-cell lymphoma 2 (Bcl-2) protein levels. Moreover, sanguinarine treatment significantly increases the activation of caspases 3 and 9 that are the key executioners in apoptosis. Our results suggest that sanguinarine induces apoptosis of HT-29 human colon cancer cells and may have a potential therapeutic use in the treatment of human colon cancer.
Introduction
Cancer is characterized by uncontrolled cell growth that presents more than 100 distinct clinical pathologies. 1 Cancer research has produced remarkable advances in our understanding of the genetics and biology of cancer 2 and also has provided the researchers with the spectrum of ideas on the development of new anticancer drugs. Colon cancer is one of the most common cancers and the third most common cause of cancer mortality throughout the world. 3 It is especially difficult to treat because of the incomplete visualization of tumor cells. 4
Sanguinarine (13-methyl-[1,3]benzodioxolo[5,6-c]-1,3-dioxolo-[4,5-i] phenanthridinium chloride) is a benzophenanthridine alkaloid present in the roots of Sanguinaria canadensis and has shown to have anti-inflammatory, antioxidant, and anticancer effects.5–9 It is reported that sanguinarine induces apoptosis in human cancer cells, but the underlying mechanisms are not completely understood. 10
Apoptosis is the major mode of cell death in cancer and therefore should be considered a potential target when developing new anticancer drugs. Several mechanisms by which anticancer drugs induce apoptosis in cancer cells have been reported, including downregulation of the antiapoptotic proteins such as B-cell lymphoma 2 (Bcl-2), upregulation of proapoptotic proteins such as Bax, 11 and activation of caspases. Caspases are the critical executioners of apoptosis induced by different apoptotic stimuli. 12 Caspase activation is often regulated by various kinds of proteins, including Bcl-2 family. 13 The active form 14 of caspase 9 cleaves procaspase 3, generating active form of caspase 3. Active caspase 3 is one of the main executioners of apoptosis, which cleaves many proteins such as other caspases and antiapoptotic protein Bcl-2. 15 In this way apoptosis progresses, leading to morphological changes in cell such as cell shrinkage, chromatin condensation, and DNA fragmentation. 16
The current study aims to extend the knowledge and understanding of the anticancer effects of sanguinarine that is known to induce apoptosis in human cancer cells. To achieve that, we used HT-29 human colon cancer cells as our model cellular system. Our results illustrate that sanguinarine reduces proliferation of human colon cancer cells by inducing apoptotic death in these cells. Furthermore, this study demonstrates for the first time that caspases 3 and 9 were activated in HT-29 human colon cells after treatment with sanguinarine, suggesting that sanguinarine induces apoptosis of HT-29 human colon cells through the caspase-dependent pathway.
Materials and Methods
Cell Culture and Sanguinarine Treatment
HT-29 human colon cancer cells were obtained from American Type Culture Collection (Rockville, Maryland). Cells were grown as described by Lee et al. 17 Briefly, cells were cultured at 37°C in the presence of 5% CO2 in RPMI1640 supplemented with 10% fetal bovine serum (FBS), 200 IU/mL penicillin, 200 μg/mL streptomycin, and 1 mmol/L sodium pyruvate. Sanguinarine was reconstituted in dimethyl sulfoxide (DMSO) and then diluted to the desired concentration in RPMI1640 (final DMSO concentration 0.1% v/v). In the control (untreated) samples, equal amount DMSO was added.
Cell Viability Assay
The effect of sanguinarine on the cellular proliferation and viability was determined using the colorimetric 3-(4,5-dimethylthiazole-2-yl)-2,5-biphenyl tetrazolium bromide (MTT) assay. This method measures mitochondrial activity based on the reductive cleavage of yellow tetrazolium salt to a purple formazan compound by the dehydrogenase activity of intact mitochondria. Briefly, cells (1 × 106 cells/well) were seeded in 24-well microtiter plates (Nunc, Denmark). After exposure to various concentrations of sanguinarine for 24 hours, cells were washed once with phosphate-buffered saline (PBS) before adding 0.5 mL serum-free medium containing 1 mg/mL MTT (Sigma, Missouri) to each well. After incubating for 4 hours, the supernatant was removed and the formazan product obtained was dissolved in 1 mL DMSO; (Sigma). The mixture was stirred for 20 minutes on a microtiter plate shaker and the absorbance was read at 750 nm. HT-29 cells viability was expressed as a percentage of untreated cells that served as the control group and was designated as 100%.
Morphological Examination of DNA Condensation
The HT-29 human colon cancer cells were first cultured on 3-chamber slides (Nalgen Nunc International, Illinois). After exposure to various concentrations of sanguinarine for 24 hours, the cells were collected and incubated in 4% paraformaldehyde (PFA) for 30 minutes. After washing in PBS, the cells were incubated in 1 μg/mL 4′,6-diamidino-2-phenylindole (DAPI) in methanol for 30 minutes in the dark. The cells were then observed under a fluorescence microscope (Zeiss, Oberkochen, Germany).
Evaluation of Protein Levels of Apoptosis-Related Signaling Pathway
HT-29 human colon cancer cells were lysed in an ice-cold whole cell lysate buffer containing 50 mmol/L N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (HEPES; pH 7.5), 150 mmol/L NaCl, 10% glycerol, 1% Triton X-100, 1.5 mmol/L MgCl2 hexahydrate, 1 mmol/L ethyleneglycol-bis-(β-aminoethylether)-N,N′-tetraacetic acid (EGTA), 1 mmol/L phenylmethylsulfonyl fluoride (PMSF), 2 μg/mL leupeptin, 1 μg/mL pepstatin, 1 mmol/L sodium orthovanadate, and 100 mmol/L sodium floride. The mixture was incubated at 4°C for 30 minutes. Cell debris was removed by microcentrifugation, followed by quick freezing of the supernatant. The protein concentration was measured using a Bio-Rad colorimetric protein assay kit (Bio-Rad). Thirty micrograms of protein were separated on sodium dodecyl sulfate (SDS)–polyacrylamide gels and transferred onto a nitrocellulose membrane (Schleicher & Schuell GmbH, Dassel, Germany). Anti-α-tubulin, anti-Bcl-2, anti-Bax, anti-caspase-8, anti-caspase-9, anti-caspase-3, and anticytochrome c antibodies (Cell signaling, Massachusetts) were used as the primary antibodies. Horseradish peroxidase-conjugated anti-rabbit and anti-mouse antibodies (Amersham Pharmacia Biotech GmbH, Freiburg, Germany) were used as the secondary antibodies. Band detection was performed using the enhanced chemiluminescence (ECL) detection system.
Caspase Activity Assay
Caspase 3 and 9 activities were measured using specific substrates for caspase 3 (Ac-DEVD-7-amino-4-methylcoumarin) and caspase 9 (Ac-LEHD-AMC, Ac-Leu-Glu-His-Asp-7-amino-4-methylcoumarin, Bachem Bioscience, Pennsylvania). Briefly, HT-29 human colon cancer cells were cultured and treated with 1 μmol/L concentration of sanguinarine and harvested at 0, 1, 3, 6, and 12 hours after the treatment, in lysis buffer (25 mmol/L HEPES [pH 7.5], 5 mmol/L EDTA, 1 mmol/L EGTA, 5 mmol/L MgCl2, 10 mmol/L sucrose, 5 mmol/L dithiothreitol [DTT], 1% [(3-cholamidopropyl)-dimethyl-ammonio]1-propanesulfonate [CHAPS], 10 μL/mL protease inhibitor cocktail, and 1 mmol/L phenylmethylsulfonyl fluoride). Cell lysates were [frozen]/thawed 5 times and centrifuged at 12 000g for 60 minutes. 18 The resultant cell lysates obtained were incubated in a buffer containing 25 mmol/L HEPES, pH 7.5, 10% sucrose, 0.1% CHAPS, and 1 mmol/L DTT supplemented with 50 μmol/L Ac-DEVD-AMC or Ac-LEHD-AMC at 37°C in 96-well Costar plates for 1 hour. The enzyme catalyzed release of the fluorogenic AMC moieties fluorescence was measured in a Wallac Victor III fluorimeter (Perkin Elmer, Wellesley, Massachusetts) using 360 nm excitation and 460 nm emission wavelengths. Caspase activities were expressed as units per microgram of total protein. Induction of caspase 3 and 9 activities in human colon cancer cells was compared with their respective controls.
Statistical Analysis
The results are presented as the mean ± standard deviation. The data were analyzed by 1-way analysis of variance (ANOVA) followed by Duncan post hoc test using SPSS. The differences were considered statistically significant at P < .01.
Results
Sanguinarine Induces Apoptosis in HT-29 Human Colon Cancer Cells
In preliminary studies, we analyzed the effects of sanguinarine treatment on HT-29 human colon cancer cells. As shown in Figure 1, the addition of sanguinarine to the medium reduced the viability of HT-29 human colon cancer cells in a dose-dependent manner (with increasing concentration, 0.125-2 μmol/L). In comparison to the control, incubation of HT-29 human colon cancer cells (treated with DMSO only) with 1 and 2 μmol/L sanguinarine for 24 hours induced 87.5% and 92.8% reductions in cell viability, respectively. This suggests that sanguinarine treatment induces decreased cell viability and/or increased cell death in HT-29 cells.
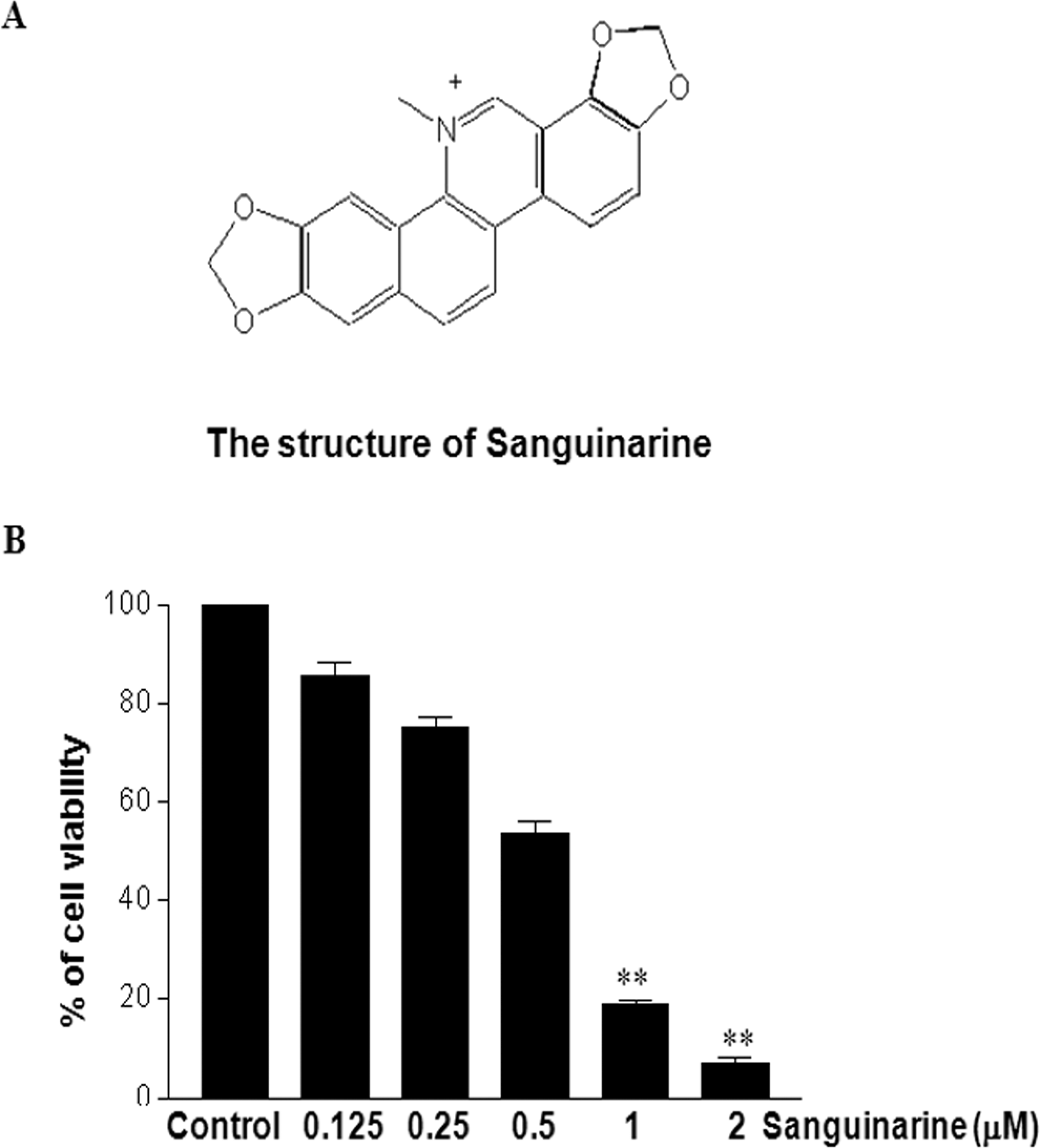
Sanguinarine treatment induces decreased viability and/or increased cell death in HT-29 cells. HT-29 human colon cancer cells were plated in 24-well microplates. Experimental medium (RPMI1640, 0.1% DMSO) containing various concentrations of sanguinarine was added, and viable cell number was assessed by MTT assay after 24 hours of incubation, as described in Materials and Methods (B). The data are reported as the viable cell number of sanguinarine-treated cells expressed as a percentage of control cells that were exposed to 0.1% DMSO. The values for each sanguinarine concentration tested represent the average (±SD) from 3 replicate wells and are representative of 4 separate experiments. The differences were considered statistically significant at **P < .01. DMSO indicates dimethyl sulfoxide; SD, standard deviation.
Sanguinarine Induces the Morphological Changes and Nuclear Condensation in HT-29 Human Colon Cancer Cells
As described in the previous section, we found that sanguinarine significantly inhibits cell viability of HT-29 human colon cancer cells at 0.5, 1, and 2 μmol/L. Therefore, we followed these concentrations for studying major mechanisms underlying sanguinarine-induced apoptosis in HT-29 human colon cancer cells. Condensation and degradation of chromosomal DNA are cardinal features of apoptosis. 19 The nuclear staining assay was used to assess the morphological changes of apoptosis in HT-29 human colon cancer cells. As shown in Figure 2, DAPI staining revealed that the treatment of HT-29 with different concentrations of sanguinarine induces significant nuclear rounding and shrinkage in HT-29 human colon cancer cells compared with the control cells.
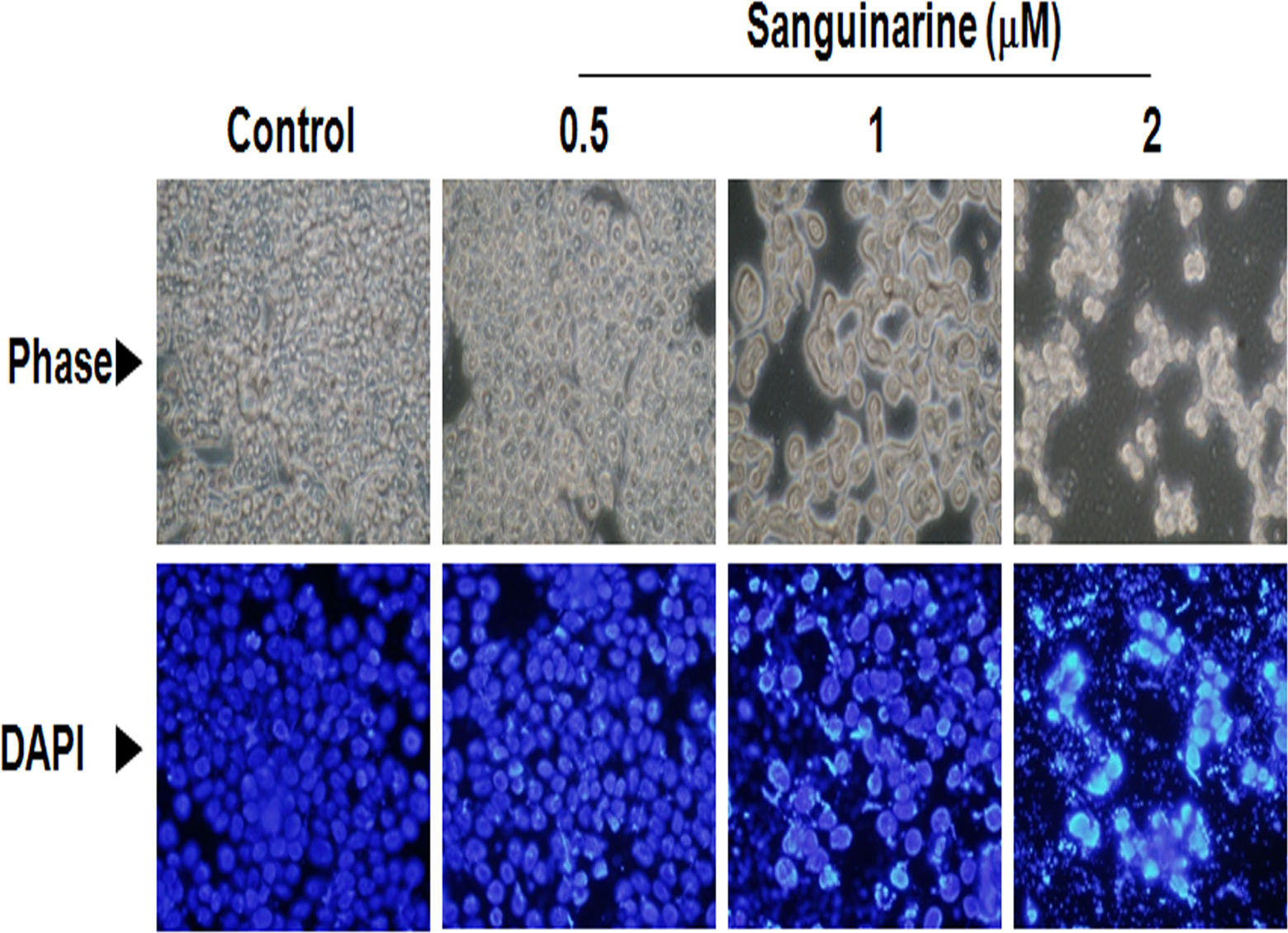
Sanguinarine treatment induces the morphological changes and nuclear condensation in HT-29 human colon cancer cells. HT-29 human colon cancer cells were treated with various concentration of sanguinarine from 0.5 to 2 μmol/L for 24 hours and then DNA condensation was evaluated using DAPI staining, as described in Materials and Methods. The DAPI-stained cells were evaluated using fluorescence microscopy (original magnification, ×600). The upper panels show phase images and the lower panels show the corresponding fields of DAPI fluorescence. DAPI indicates 4′,6-diamidino-2-phenylindole.
The Effects of Sanguinarine on Bax/Bcl-2 Ratio in HT-29 Human Colon Cancer Cells
Levels of these proteins following sanguinarine treatment were examined by Western blotting in order to determine whether sanguinarine induces HT-29 human colon cancer cell death by altering the ratio between Bcl-2 and Bax. The ratio of Bax-to-Bcl-2 protein can determine the susceptibility of the cell to apoptosis. 20 As shown in Figure 3A, treatment of HT-29 human colon cancer cells with sanguinarine significantly induced a dose-dependent decrease in the expression levels of Bcl-2 and increase in the expression levels of Bax. A densitometric analysis of the bands showed that sanguinarine resulted in a dose-dependent increase in the Bax/Bcl-2 ratio (Figure 3B). These results suggest that sanguinarine can induce apoptosis of HT-29 human colon cancer cells by regulating Bax/Bcl-2 ratio.
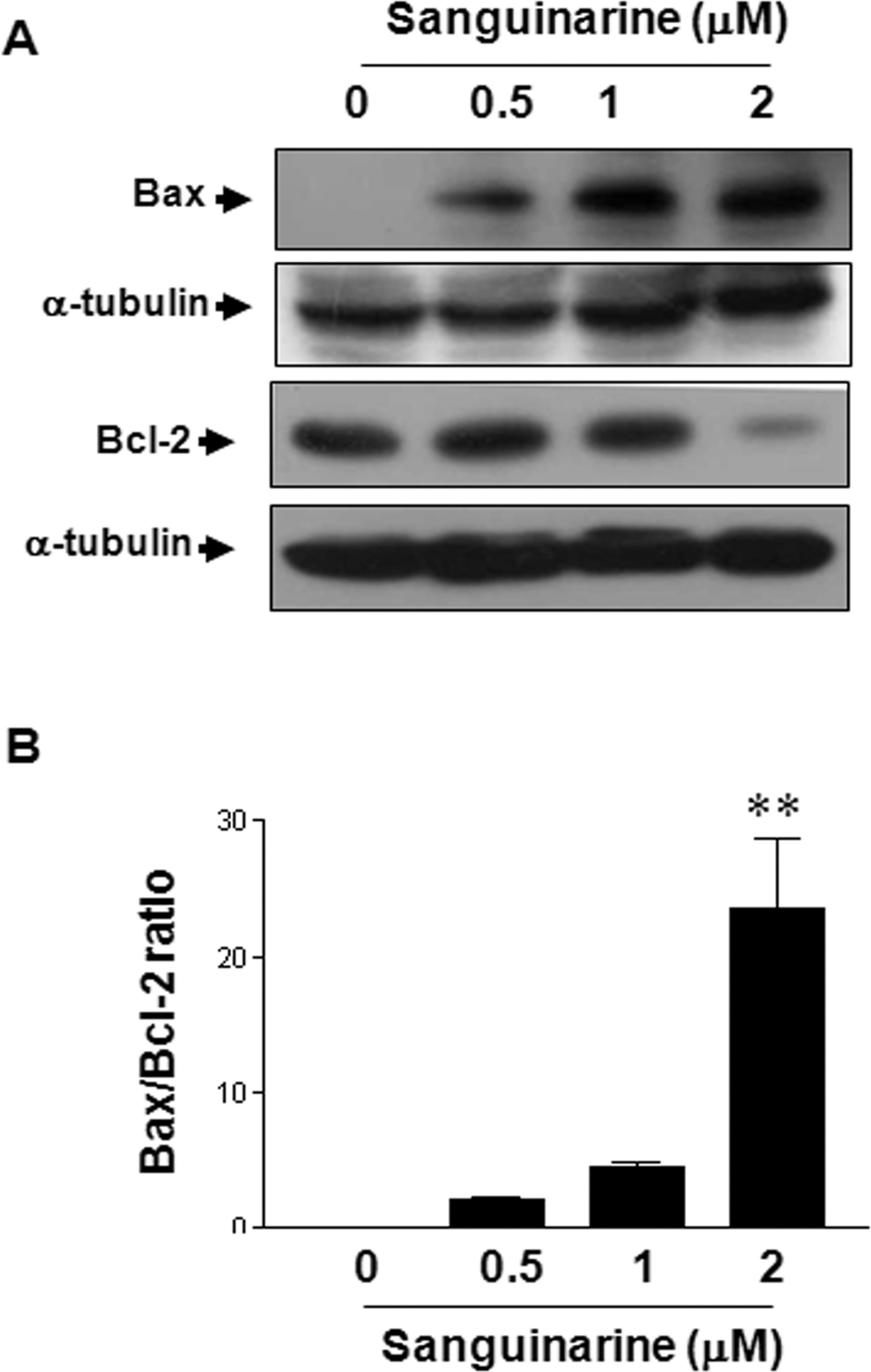
Sanguinarine induces upregulation of Bax and downregulation of Bcl-2 expression. A, Cells were incubated with different sanguinarine concentrations for 24 hours and then subsequently lysed. Equal amounts of proteins were then separated by SDS-PAGE and transferred to nitrocellulose membranes. The membranes were probed with the indicated antibodies (anti-Bax, anti-Bcl-2, and α-tubulin) and detected by ECL. α-Tubulin was used as the internal control. B, Densitometric analysis of the immunoblot data was used to quantify the levels of Bcl-2 and Bax in order to evaluate the effect of sanguinarine on the Bax/Bcl-2 ratio. The results are from one representative experiment of 4 performed that showed similar patterns. The differences were considered statistically significant at **P < .01. SDS-PAGE indicates sodium dodecyl sulfate–polyacrylamide gel electrophoresis; ECL, enhanced chemiluminescence; Bcl-2, B-cell lymphoma 2.
Sanguinarine Induces the Cytochrome c Release in Cytosol and Activation of Caspase 9 and 3 in HT-29 Human Colon Cancer Cells
The activation of caspases is important in inducing apoptosis and processing, and caspase family can be regulated by molecules such as Bcl-2, Bax, and cytochrome c. 21 Increase in Bax expression may result in altering mitochondria permeability, triggering the release of mitochondrial cytochrome c into cytosol. In cytosol, 22 cytochrome c activates caspase 9, which in turn activates caspase 3. As described previously, sanguinarine-treated HT-29 human colon cancer cells showed lower Bcl-2 protein levels as well as higher Bax protein levels. Therefore, the downstream events of apoptosis in HT-29 human colon cancer cells were characterized by examining the release of cytochrome c. As shown in Figure 4A, the release of cytochrome c and cleavage of capase 9 were markedly increased in the HT-29 human colon cancer cells treated with sanguinarine. We also analyzed the caspase activity by fluorescence peptide substrates. 23 Fluorescence peptide substrates analysis of whole cell lysates, obtained from HT-29 human colon cancer cells treated with 1 μmol/L of sanguinarine, revealed an increase in caspase 9 and 3 activities at a various time points (Figure 4B). These data indicated that apoptosis by sanguinarine may be mediated through the caspase-9-dependant pathway.
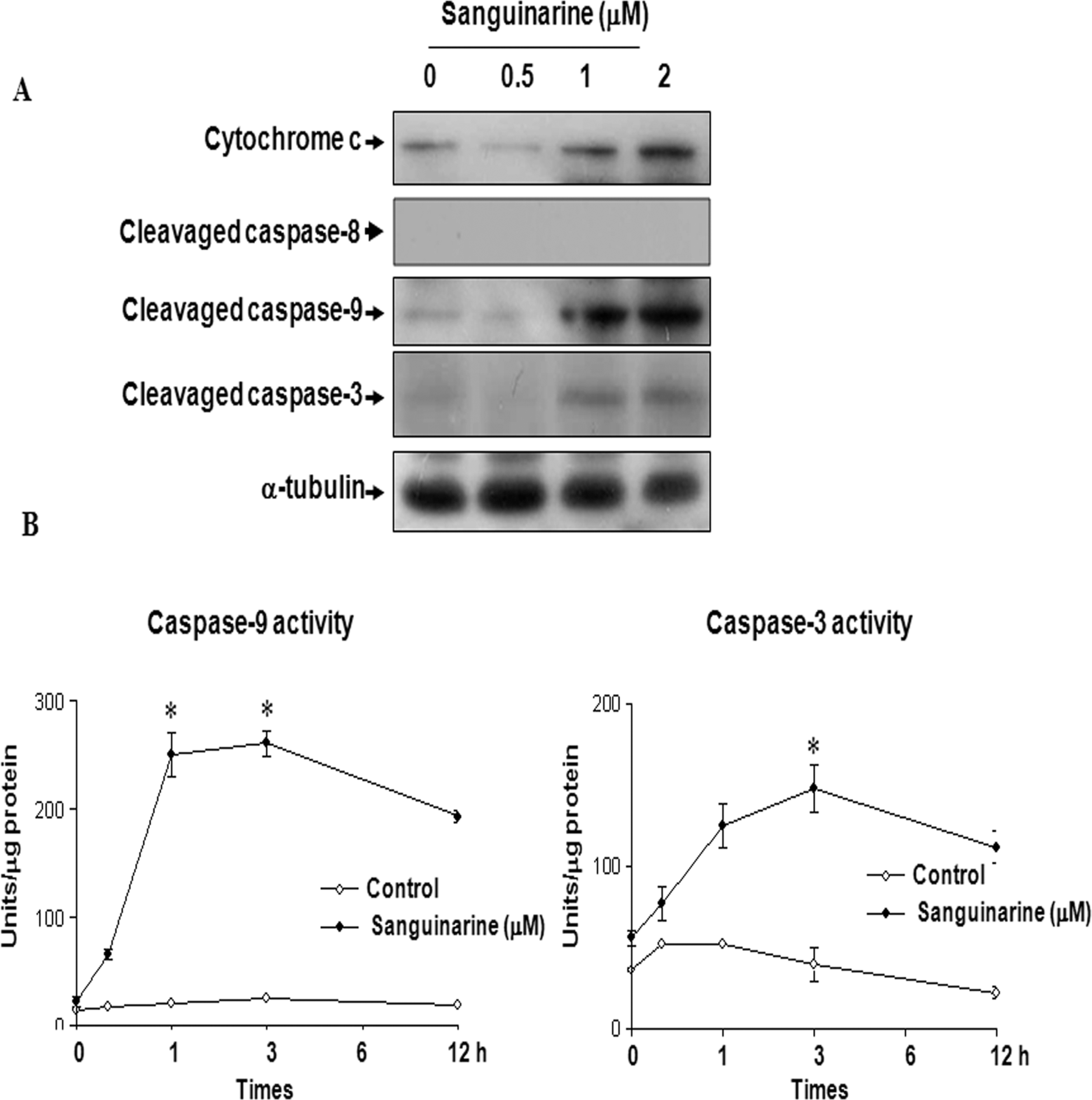
Sanguinarine induces the activation of caspases 9, 3, and the release of cytochrome c in HT-29 human colon cancer cells. A, Cells were incubated with different sanguinarine concentration for 24 hours and subsequently lysed. Equal amounts of proteins were then separated by SDS-PAGE and transferred to nitrocellulose membranes. The membranes were probed with the indicated antibodies (anti-caspase 9, anti-caspase 3, anticytochorme c [cytoplasmic faction], and α-tubulin) and detected by ECL. α-Tubulin was used as the internal control. B, Cells were analyzed for caspase 9 and 3 activities, using the caspase 9 (Ac-IEHD-AMC) and caspase 3 (Ac-DEVD-AMC) substrates. The cell lysates obtained from the untreated and sanguinarine-treated HT-29 cells, with normalized protein content, were incubated with the caspases 9 and 3 substrates. The differences were considered statistically significant at **P < .01. SDS-PAGE indicates sodium dodecyl sulfate–polyacrylamide gel electrophoresis; ECL, enhanced chemiluminescence.
Inhibition of Sanguinarine-Induced Apoptosis by Caspase 9 Inhibitor
In order to confirm that the activation of caspase 9 plays a key role in the sanguinarine-induced apoptotic pathway, HT-29 human colon cancer cells were pretreated with caspase 9 inhibitor z-LEHD-fmk, followed by a treatment with 1 μmol/L sanguinarine for 3 hours. Pretreatment with caspase 9 inhibitor significantly blocked caspase 9 and 3 activities (Figure 5A). Interestingly, the blockade of the caspase 9 activity prevented sanguinarine-induced cell death of HT-29 human colon cancer cells (Figure 5B). These results clearly show that sanguinarine-induced apoptosis is associated with caspase 9 activation.
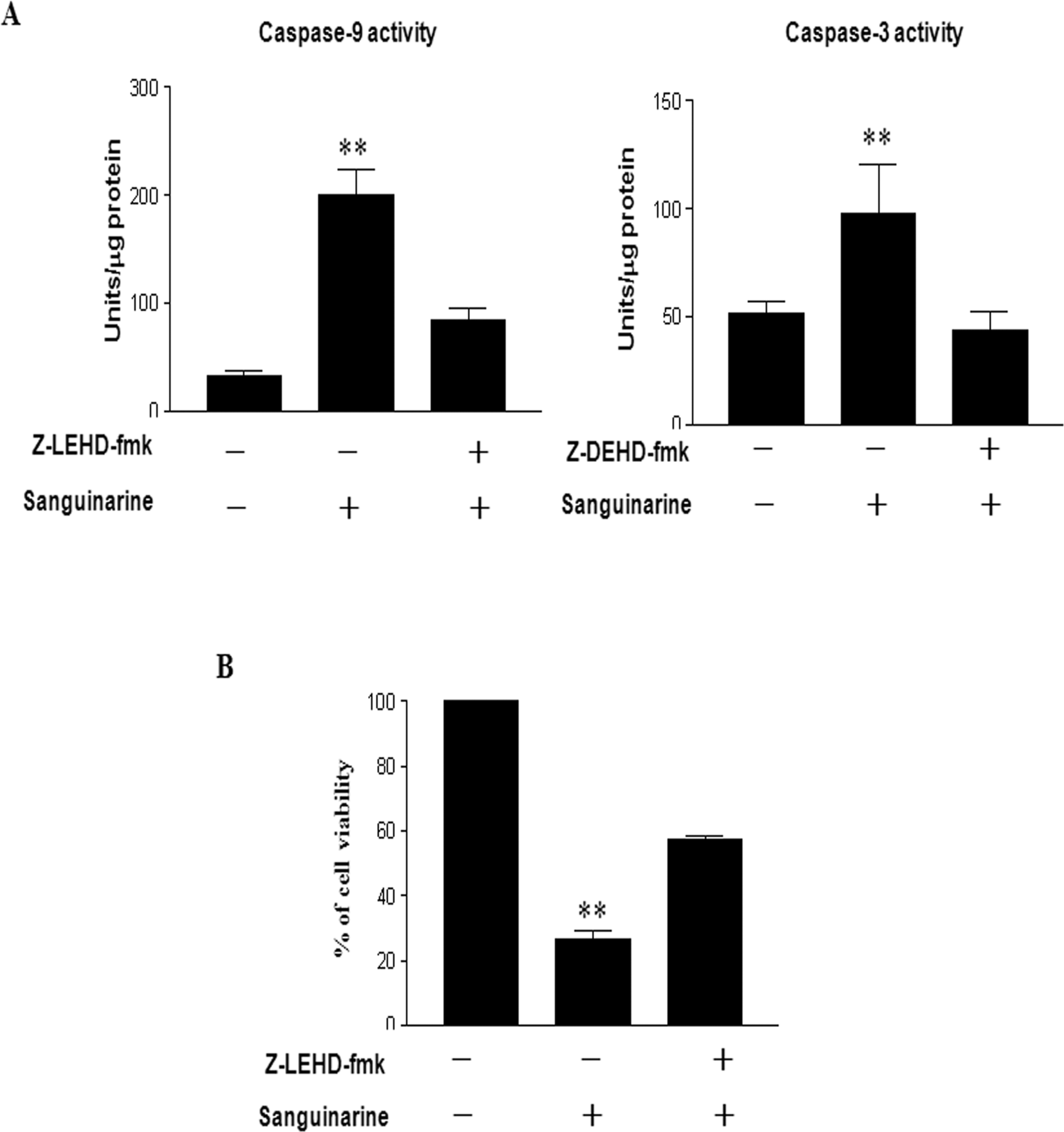
Preincubation of HT-29 human colon cancer cells with specific caspase 9 inhibitor inhibits sanguinarine-induced apoptosis in these cells. Cells were incubated with or without z-LEHD-fmk for 1 hour before being treated with sanguinarine. A, After 3 hours incubation with sanguinarine, the cells were harvested and analyzed for caspase activity with fluorescence peptide substrates, as described in Materials and Methods. B, Cell viability was assessed by MTT assay after 24 hours incubation for each group (control, sanguinarine, and sanguinarine plus caspase 9 inhibitor). Data reported as the mean values from 3 independent experiments and the bars represent the standard deviations. The differences were considered statistically significant at **P < .01. MTT indicates 3-(4,5-dimethylthiazole-2-yl)-2,5-biphenyl tetrazolium bromide.
Discussion
Apoptosis is a highly regulated process that involves a series of molecular events that lead to cell death. It is characterized by chromatin condensation, changes in cellular morphology, alteration in the expression of proapoptotic and antiapoptotic proteins, activation of caspases, and the appearance of apoptotic bodies, which are associated with DNA fragmentation and ladder formation. 24
Cell death is controlled by several cascade proteins, especially by the members of the Bax and Bcl-2 family. 25 Bcl-2 itself is an antiapoptotic protein and has the ability to block various apoptotic signals. On the other hand, Bax is another member of the Bcl-2 family but exerts a proapoptotic effect. 26 B-cell lymphoma 2 is an antiapoptotic protein and its function is modulated by its ability to heterodimerize with other members of the Bcl-2 family, predominantly with Bax. Bax is a proapoptotic protein and can trigger the activation of caspases by mitochondrial dysfunction, which results in the release of apoptosis-promoting factors into the cytoplasm, 27 leading to cell death. Susceptibility to undergo apoptosis is therefore dependent on the ratio between Bcl-2 and Bax.
Sanguinarine is a benzophenanthridine alkaloid present in the roots of S canadensis, and it is known to induce apoptosis in various human cancer cells via the activation of caspases, NF-κB activation, cell cycle arrest, and mitochondrial damage, individually.10,28–32
In this study, we conducted a detailed investigation of the effects of sanguinarine on HT-29 human colon cancer cells. Sanguinarine inhibited HT-29 human colon cancer cell growth in a dose-dependent manner (Figure 1). After 24 hours of incubation, at the concentrations of 0.5, 1, and 2 μmol/L sanguinarine significantly suppressed the viability of HT-29 human colon cancer cells to 42%, 87.5%, and 92.8%, respectively, in comparison to the control (untreated) cells. After establishing that sanguinarine reduces proliferation of human colon cancer cells and induces apoptosis in these cells, we investigated the mechanism underlying this effect. Our data showed that sanguinarine treatment of HT-29 human colon cancer cells induces an increase in Bax expression and a decrease in Bcl-2 expression. This alteration in the expression of Bax and Bcl-2 leads to high Bax/Bcl-2 ratio (Figure 3) which is an important factor in determining the cell’s vulnerability to apoptosis; high Bax/Bcl-2 ratios lead to greater apoptotic activity. Thus, current data suggest that sanguinarine-induced apoptosis is controlled by a balanced expression between these apoptosis-inducing and apoptosis-suppressing molecules. This inducible expression of Bax may result in altering mitochondria permeability, triggering the release of mitochondrial cytochrome c into cytosol. We found that treatment of sanguinarine induced the expression of Bax in human colon cancer cells (Figure 3A). This result suggested that sanguinarine-induced Bax expression is a key regulator of mitochondria damage of human colon cancer cell death.
In cytosol, cytochrome c activates caspase 9, which in turn activates22,33 caspases 3, 6, and 7. To illustrate the effect of sanguinarine treatment on activities of different caspases, we performed caspase activity assays using fluorescence peptide substrates. We found that sanguinarine treatment (1 μmol/L, 3 hours) increases the activities of caspases 3 and 9 about 6- to 7-fold (Figure 4). Additionally, HT-29 human colon cancer cells were incubated with specific caspase 9 inhibitor (z-LEHD-fmk) before sanguinarine treatment, and then the caspase 3/9 activities were analyzed in these samples. Results showed that preincubation of HT-29 human colon cancer cells with z-LEHD-fmk effectively inhibited the caspase 9 activity and cell death (Figure 5B). These results suggest that the activation of caspase 9 is essential for sanguinarine-induced apoptosis of HT-29 human colon cancer cells.
Conclusions
In conclusion, sanguinarine treatment reduces proliferation and induces apoptotic death in HT-29, a human colon cancer cell line. Additionally, it was found that sanguinarine induces apoptosis via regulation of Bax/Bcl-2 ratio and caspase-9-dependent pathway. The identification of apoptotic properties of sanguinarine toward human colon cancer cells indicates that sanguinarine may have a prospective therapeutic use in the prevention and/or treatment of human colon cancer.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
