Abstract
This study evaluated the antiasthmatic effects of Gleditsia sinensis ethanolic extract (GSEE) and its underlying mechanisms, using an in vivo murine model of asthma. Female BALB/c mice were sensitized, challenged with ovalbumin, and then examined for asthmatic reactions. The results showed that GSEE exerted profound inhibitory effects on the accumulation of eosinophils in the airways and reduced the levels of interleukin (IL)-4 and IL-5 in bronchoalveolar lavage fluid (BALF) and immunoglobulin E (IgE) in BALF and plasma. Gleditsia sinensis ethanolic extract also suppressed the production of reactive oxygen species in BALF and inflammatory infiltration, in a dose-dependent manner, and it inhibited goblet-cell hyperplasia in lung tissue. Thus, GSEE shows antiasthmatic effects in a murine model of allergic asthma, which appeared to be mediated partially by the reduction of oxidative stress and airway inflammation. These results indicate that GSEE could be an effective novel therapeutic agent for the treatment of allergic asthma.
Bronchial asthma is a chronic inflammatory disease of the airways characterized by airway eosinophilia, goblet-cell hyperplasia with mucus hypersecretion, and hyperresponsiveness to inhaled allergens and nonspecific stimuli. 1 The inflammatory response involves the recruitment and activation of inflammatory cells, as well as changes in the structural cells of the airway.2,3 Inflammatory cells, including eosinophils, neutrophils, and macrophages, release oxidants upon activation,4–6 which can overwhelm antioxidant defenses and result in oxidative stress in the lung. 7 Oxidative stress is increased in the airways of asthmatics8,9 with detrimental effects on airway function. 7
Allergic inflammation in asthma is characterized by increased eosinophil levels in the lung tissue and blood, in association with enhanced production of T-helper 2 (Th2) cell cytokines, 10 including interleukin (IL)-4, IL-5, and IL-13. Interleukin4 is also critically important in directing B-cell growth and differentiation, and the secretion of immunoglobulin E (IgE) and IgG1 in the mouse. T-helper 2-derived IL-4 and IL-13 are essential for IgE class switching and production by B cells. Immunoglobulin E promotes subsequent antigen-specific mast cell activation to induce degranulation and secretion of cytokines, including Th2-type cytokines, such as IL-4 and IL-13. Interleukin 5, IL-13, and IL-9 are the key cytokines that promote the accumulation and activation of eosinophils and mast cells, respectively, at the sites of inflammation.11,12 In particular, mast cells (sources of Th2-type cytokines) are significantly involved in the pathogenesis of asthma. Oxidative injury plays an important role in inflammation observed in individuals with asthma or chronic obstructive pulmonary disease (COPD) and corresponding animal models.13–15 Thus, accumulating clinical and experimental evidence suggests the essential roles for reactive oxygen species (ROS) in the pathogenesis of airway inflammation.16–18 Over recent decades, the number of individuals with atopic diseases, such as asthma, has increased dramatically in industrialized countries. However, the majority of therapeutic agents developed to date are relatively inefficient and prolonged use often leads to side effects. Here, we investigate the effects of Gleditsia sinensis Lamarck, with a view to identifying safe lead compounds with minimal side effects.
Gleditsia sinensis is a perennial shrub that is widely distributed throughout China, and which is also found in the Gyeongju city area of Korea. The thorns of the plant, known as “Jo Gak Ja” in Korea, are used in traditional medicine for the treatment of swelling, suppuration, carbuncles, and skin diseases. 19 The crude extract exerts antitumor activity and inhibitory effects on mast cell – dependent anaphylaxis. 20 Moreover, the aqueous crude extract is reported to decrease carrageenan-induced paw edema, adjuvant-induced arthritis, and granuloma formation in vivo.21,22 Recent reports elucidated the biological activity of its chemical constituents, including the antimutagenic activity of stigmasterol, 23 the antimicrobial activity of ellagic acid glycosides 24 and flavonoid, 25 and the anti-HIV activity of lupine acid. 26 However, the effects of a G sinensis thorn ethanolic extract (GSEE) on asthma are yet to be established. Montelukast was developed as a cysteinyl leukotriene (cys-LT) 1 receptor antagonist, 27 and the clinical responses of patients with aspirin-sensitive asthma, nocturnal exacerbations of asthma, and allergic asthma led to montelukast being successfully introduced into the market. 28
The present study investigated the impact of GSEE in an ovalbumin (OVA)-induced airway inflammation model of asthma and further explored the underlying mechanisms of the observed effects.
Materials and Methods
Preparation of GSEE
Gleditsia sinensis thorn was purchased in October 2008 from HMAX (Chungbuk, Republic of Korea). The plant materials were confirmed taxonomically by Professor Je-Hyun Lee of Dongguk University, Gyeongju, Republic of Korea. A voucher specimen was deposited at the Korea Institute of Oriental Medicine (KIOM), Daejeon, Republic of Korea. Fresh G sinensis was washed 3 times with tap water to remove salts, epiphytes, and sand, and stored at −20°C. Frozen samples were lyophilized and homogenized in a grinder before extraction. Extracts were prepared from dried thorns of G sinensis (200 g) with 70% ethyl alcohol ([EtOH] 2 L × 3 times), followed by sonication for 1 hour. The extract solution was filtered through filter paper and evaporated to dryness (7.32 g). The yield of dried extract from crude starting material was 3.6%.
Chromatography System and Chemicals
Analysis was performed using a Shimadzu LC-20A HPLC system (Shimadzu Co, Kyoto, Japan), consisting of a solvent delivery unit, an online degasser, a column oven, an autosampler, and a photodiode array (PDA) detector. The data processor used the LCsolution software (Version 1.24). The analytical column was a Gemini C18 (250 × 4.6 mm; particle size 5 μm; Phenomenex, Torrance, California). The mobile phases were solvent A (aqueous acetic acid, 1.0% volume/volume [v/v]) and solvent B (acetonitrile with 1.0% v/v acetic acid). The gradient flow was as follows: A:B = 95:5 (0 minutes) → A:B = 30:70 (50 minutes) → A:B = 0:100 (55 minutes; hold for 5 minutes) → A:B = 95:5 (65 minutes; hold for 15 minutes). The column temperature was maintained at 40°C. The analysis was carried out at a flow rate of 1.0 mL/min with PDA detection between 190 and 400 nm. The injection volume was 10 μL. (−)-Epicatechin, ethyl gallate, and quercetin (all with purity ≥99.0%) were purchased from ChromaDex (Santa Ana, California). Caffeic acid (purity ≥99.0%) and eriodictyol (purity ≥99.0%) were purchased from Acros Organics (Fairlawn, New Jersey) and Extrasynthese (Genay, France), respectively. High-performance liquid chromatography (HPLC) – grade methanol, acetonitrile, and water were purchased from J.T. Baker (Phillipsburg, New Jersey). Analytical-grade glacial acetic acid was purchased from Junsei (Tokyo, Japan).
Preparation of Standard and Sample Solutions
Standard stock solutions of caffeic acid, (-)-epicatechin, eriodictyol, ethyl gallate, and quercetin (all 1000 μg/mL) were prepared in methanol and held below 4°C. Working standard solutions were prepared by serial dilution of stock solutions with methanol. The dried thorn of G sinensis (200 g) was extracted 3 times with 70% ethanol by sonication for 60 minutes, and the extract was evaporated to dryness and freeze-dried (yield = 3.7%). Lyophilized extract of G sinensis (10 mg) was dissolved in 70% methanol (10 mL). The solution was filtered through a SmartPor GHP syringe filter (pore size, 0.2 μm; Woongki Science, Seoul, Korea).
Animals
Seven-week-old, specific pathogen-free (SPF) inbred female BALB/c mice that were routinely screened serologically for relevant respiratory pathogens were purchased from Daehan Biolink Co Ltd (Seoul, Korea). Mice were maintained in an animal facility under standard laboratory conditions for 1 week prior to the experiments and provided with water and standard chow ad libitum. The animals were housed in a room that was maintained at a temperature of 22°C ± 3°C and a relative humidity of 50% ± 10%, with artificial lighting from 08:00 to 20:00 and 10 to 20 air changes per hour. All experimental procedures were performed in accordance with the NIH Guidelines for the Care and Use of Laboratory Animals, and animal handling followed the dictates of the National Animal Welfare Law of Korea.
Sensitization and Airway Challenge
Mice were divided into groups that received the following treatments: (1) sham sensitization (phosphate-buffered saline [PBS]), intraperitoneal injection (ip) plus challenge (nebulization [Neb]) with PBS (ipNeb); (2) sensitization plus challenge with OVA (Sigma A5503; Sigma, St. Louis, Missouri; ipNeb); and (3) sensitization with OVA (ip) plus challenge with OVA (Neb) and GSEE (25, 50, or 100 mg/kg), or montelukast (30 mg/kg, per oral [po]). Mice were sensitized and challenged as reported previously 16 but with some modifications. Briefly, mice were sensitized with an intraperitoneal injection of 20 μg OVA emulsified with 2 mg aluminum hydroxide in 200 μL PBS buffer (pH 7.4) on days 0 and 14. Concentrated GSEE was freshly dissolved in PBS prior to use. Before administration of GSEE (on day 16), the IgE levels were measured for false positives. Mice were orally administered GSEE (25, 50, or 100 mg/kg per d) and 30 mg/kg of montelukast every day between days 18 and 23. Montelukast was used as a positive control. Mice were challenged via the airways with OVA (1%, weight/volume [w/v]) in PBS (10 mL of a 10 mg/mL OVA solution) for 30 minutes using an ultrasonic nebulizer (NE-U12; Omron Corp, Tokyo, Japan) on days 21, 22, and 23 after initial sensitization. According to the specifications supplied by the manufacturer, the output of the nebulizer was 0.5 mL/min, and the particles produced had a mean diameter of 3.5 μm. Animals were sacrificed at 48 hours after the final challenge (day 25) before characterizing the inhibitory effects of GSEE. A schematic diagram of the treatment schedule is shown in Figure 1.
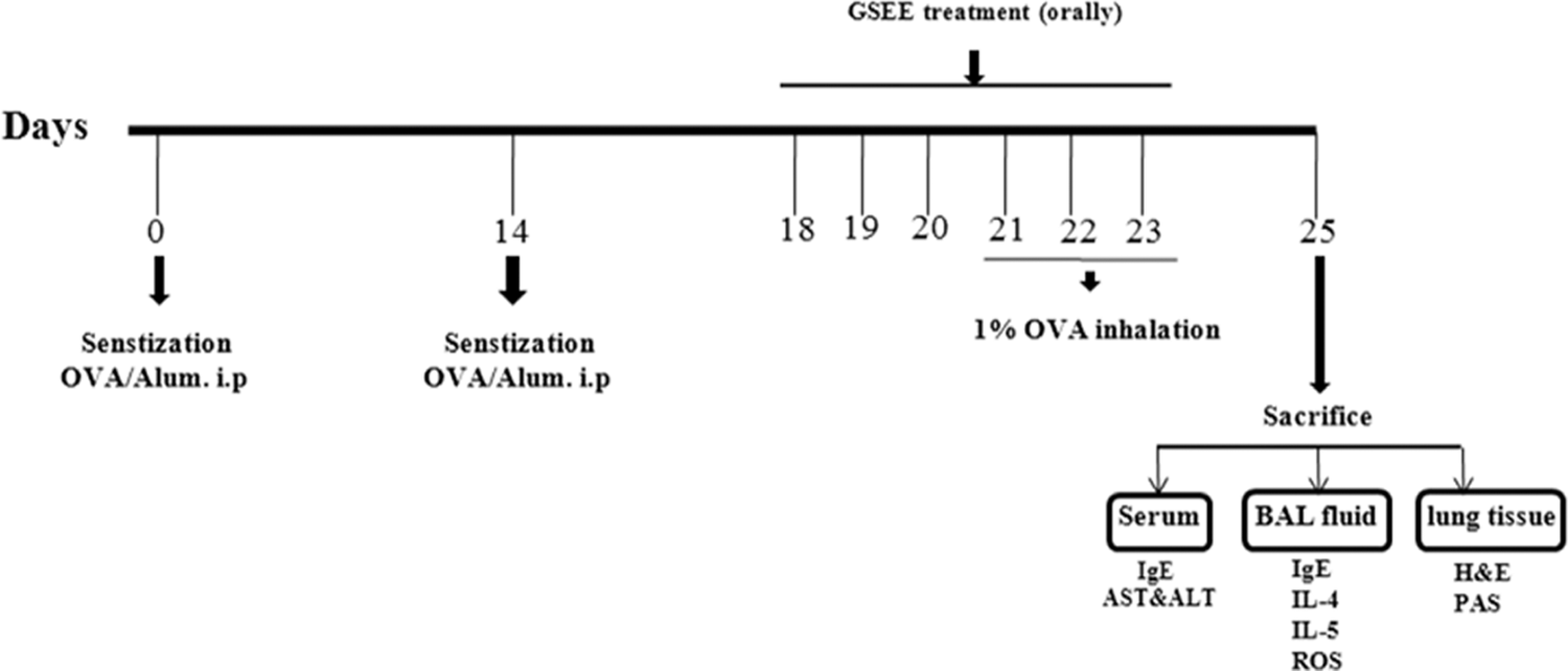
Mouse model of airway inflammation and effects of Gleditsia sinensis Lamarck ethanolic extract (GSEE) treatment.
Inflammatory Cell Counts in Bronchoalveolar Lavage Fluid
Mice were sacrificed with an overdose (50 mg/kg) of pentobarbital at 48 hours after the last challenge and tracheostomy was performed. The trachea was cannulated and the left bronchi were tied for histological experiments. Following instillation of ice-cold PBS (0.6 mL) into the lungs, bronchoalveolar lavage fluid (BALF) was obtained using 3 aspirations (total volume, 1.8 mL) via tracheal cannulation. Bronchoalveolar lavage fluid was centrifuged and the supernatant fractions were collected and stored at −70°C. Total numbers of eosinophils, macrophages, lymphocytes, and neutrophils were assessed by counting cells in at least 5 squares of a hemocytometer, after exclusion of dead cells via trypan blue staining. Bronchoalveolar lavage fluid (100 μL) was pipetted onto a slide and centrifuged (200g, 4°C, 10 minutes) to fix cells using a cytospin machine (Hanil Science Industrial, Seoul, Korea). Cell pellets were suspended in 0.5 mL PBS, and 100 μL of each solution was spun onto a slide. After the slides were dried, the cells were fixed and stained using Diff-Quik staining reagent (B4132-1A; Dade Behring Inc, Deerfield, Illinois), according to the manufacturer’s instructions. Differential cell counts were performed, according to the microscopic morphology.
2′,7′-Dichlorofluorescein Fluorescence Assay
Induction of oxidative stress was monitored using 2′,7′-dichlorofluorescein diacetate (DCF-DA; Molecular Probes, Eugene, Oregon), which is converted into highly fluorescent DCF by cellular peroxides, including hydrogen peroxide. In brief, the BALF cells were washed with PBS and total cells (5 × 103) were counted. Cells were treated with 25 μmol/L DCF-DA for 10 minutes at 37°C. Reactive oxygen species activity was measured by determining fluorescence at 488 nm excitation and 525 nm emission using a fluorescence plate reader (PerkinElmer, Waltham, Massachusets).
Measurement of IgE Levels in BALF and Plasma
Bronchoalveolar lavage fluid and plasma were collected and stored at −70°C after centrifugation (300g, 10 minutes). Total IgE levels were measured using an enzyme-linked immunosorbent assay (ELISA). Complementary capture and detection antibody pairs for mouse IgE were purchased from Pharmingen (Serotec, Oxford, UK). The IgE ELISA was performed according to the manufacturer’s protocol. Duplicate samples were diluted 1:100 in plasma. Immunoglobulin E levels in each sample were measured using optical density readings at 450 nm, and specific concentrations were calculated from a standard curve generated using recombinant IgE (5-2000 ng/mL).
Measurement of Th2 Cytokine Levels in BALF
The amounts of IL-4 and IL-5 present in BALF were measured with a specific mouse ELISA kit (BioSource International, Camarillo, California). Enzyme-linked immunosorbent assay experiments were performed according to the manufacturer’s instructions. The assay detection limit was 250 pg/mL.
Lung Tissue Histopathology
Before the lungs were removed, the left lungs were filled intratracheally with a fixative (0.8% formalin and 4% acetic acid) using a ligature around the trachea. Lungs were fixed in 10% v/v neutral buffered formalin for 24 hours at 4°C. Tissues were embedded in paraffin, sectioned at 4 μm thickness, and stained with hematoxylin and eosin ([H&E] solution; hematoxylin, Sigma MHS-16, and eosin, Sigma HT110-1-32) and periodic acid-Schiff ([PAS] IMEB Inc, San Marcos, California) to estimate mucus production. Tissues were then mounted and coverslipped using Dako mounting medium (Dakocytomation, Glostrup, Denmark). The degree of cell infiltration in the airway was scored in a double-blind screening by 2 independent investigators. 29 Peribronchiolic and perivascular inflammation was evaluated using a scoring of 0 to 5, as follows: 0, no cells; 1, a few cells; 2, a ring of cells 1 cell layer deep; 3, a ring of cells 2-4 cells deep; 4, a ring of cells; and 5, deep cells. Goblet-cell hyperplasia in the airway epithelium was quantified based on a 5-point system: 0, no goblet cells; 1, <25% of the epithelium; 2, 25%-50% of the epithelium; 3, 50%-75% of the epithelium; and 4, >75% of the epithelium. For each mouse, 5 airway sections randomly distributed throughout the left lung were analyzed and their average score was calculated. Quantitative analysis of mucus production was performed using an image analyzer (Leica Microsystem Imaging Solution Ltd, Cambridge, UK).
Image Capture and Photomicrography
Photomicrographs were obtained using a Photometric Quantix digital camera running a Windows program, and montages were assembled in Adobe Photoshop 7.0. Images were cropped and corrected for brightness and contrast but not otherwise manipulated.
Statistical Analysis
Data are expressed as means ± SD (standard deviation about the mean). Statistical comparisons were made by 1-way analysis of variance (ANOVA) using SPSS 16.0, followed by Duncan’s new multiple range test, using P < .05 as the level of significance.
Results
High-Performance Liquid Chromatography Analysis
The chromatogram was obtained using an HPLC-PDA detector. Under optimized chromatography conditions, 5 components were eluted within 35 minutes of sample analysis using mobile phases consisting of solvent A (1.0% v/v, aqueous acetic acid) and solvent B (acetonitrile with 1.0% v/v acetic acid). Figure 2 shows chromatograms of reference compounds and GSEE. The retention times of compounds were 15.0 minutes ((-)-epicatechin), 15.5 minutes (caffeic acid), 18.5 minutes (ethyl gallate), 27.0 (eriodictyol), and 28.0 minutes (quercetin).
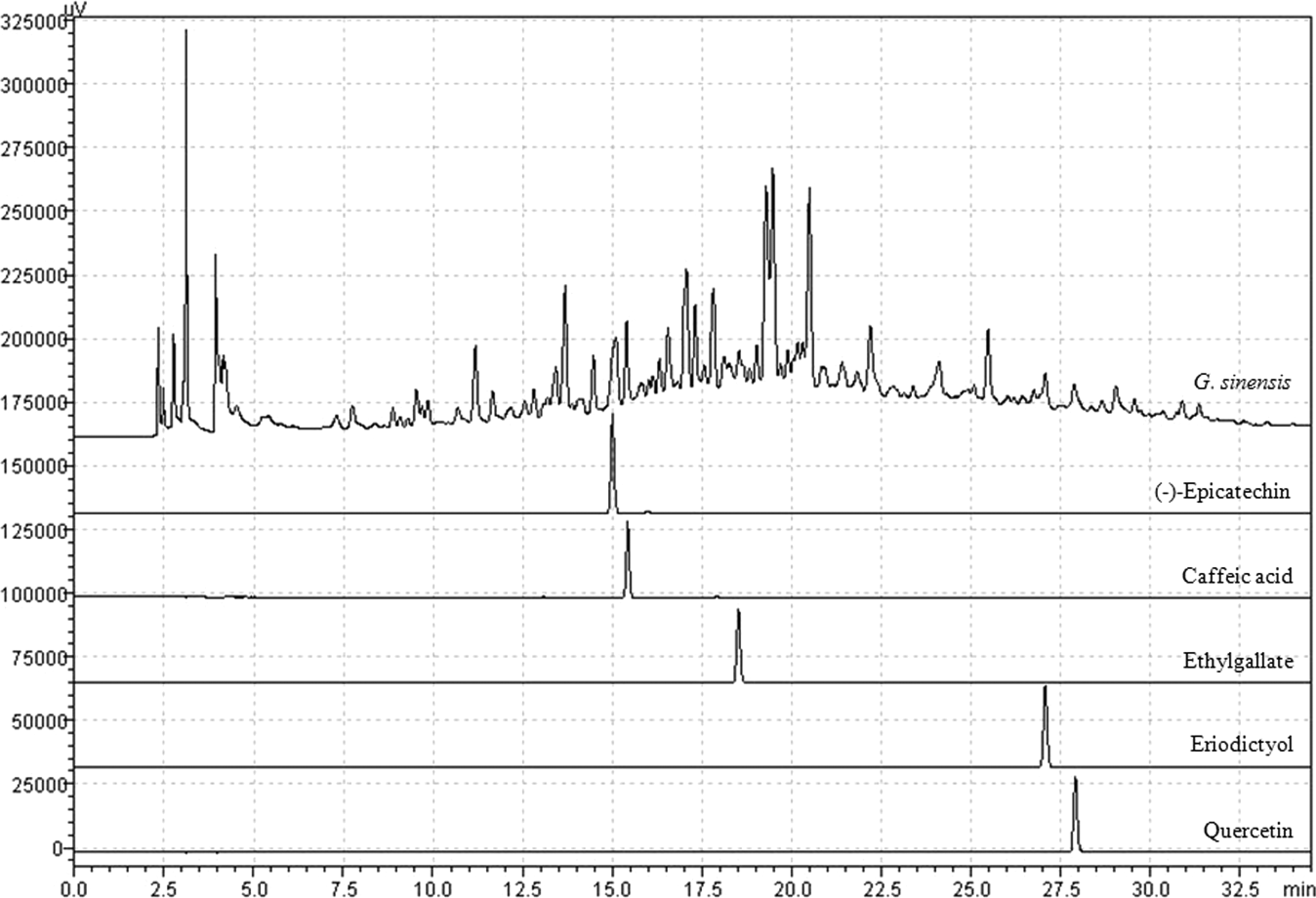
Chromatogram of GSEE extract by HPLC-PDA. The 70% ethanol extract of GSEE and its 5 components were characterized by HPLC-PDA analysis. A, Phenomenex Gemini C18 (250 × 4.6 mm) column was eluted with solvent A (water containing 1.0% volume/volume (v/v) acetic acid) and B, acetonitrile containing 1.0% v/v acetic acid at flow rate of 1.0 mL/min. GSEE indicates Gleditsia sinensis Lamarck ethanolic extract; HPLC, high-performance liquid chromatography; PDA, photodiode array.
Effects of GSEE on OVA-Induced Eosinophilia and ROS Generation in BALF
To determine whether GSEE had a protective effect against airway inflammation, total cell and eosinophil numbers in the BALF were measured in OVA-challenged mice that were either untreated or treated with GSEE. Ovalbumin-challenged mice displayed marked increases in both total cell and eosinophil numbers in the BALF. However, in GSEE-treated mice, cell influx to the airspace was significantly (P < .05) attenuated in terms of eosinophils and total cells, at 50 and 100 mg/kg, compared with the OVA-induced group without GSEE exposure (Figure 3A). The decreased leukocyte infiltration was attributable mainly to a decrease in macrophage and eosinophil infiltration.
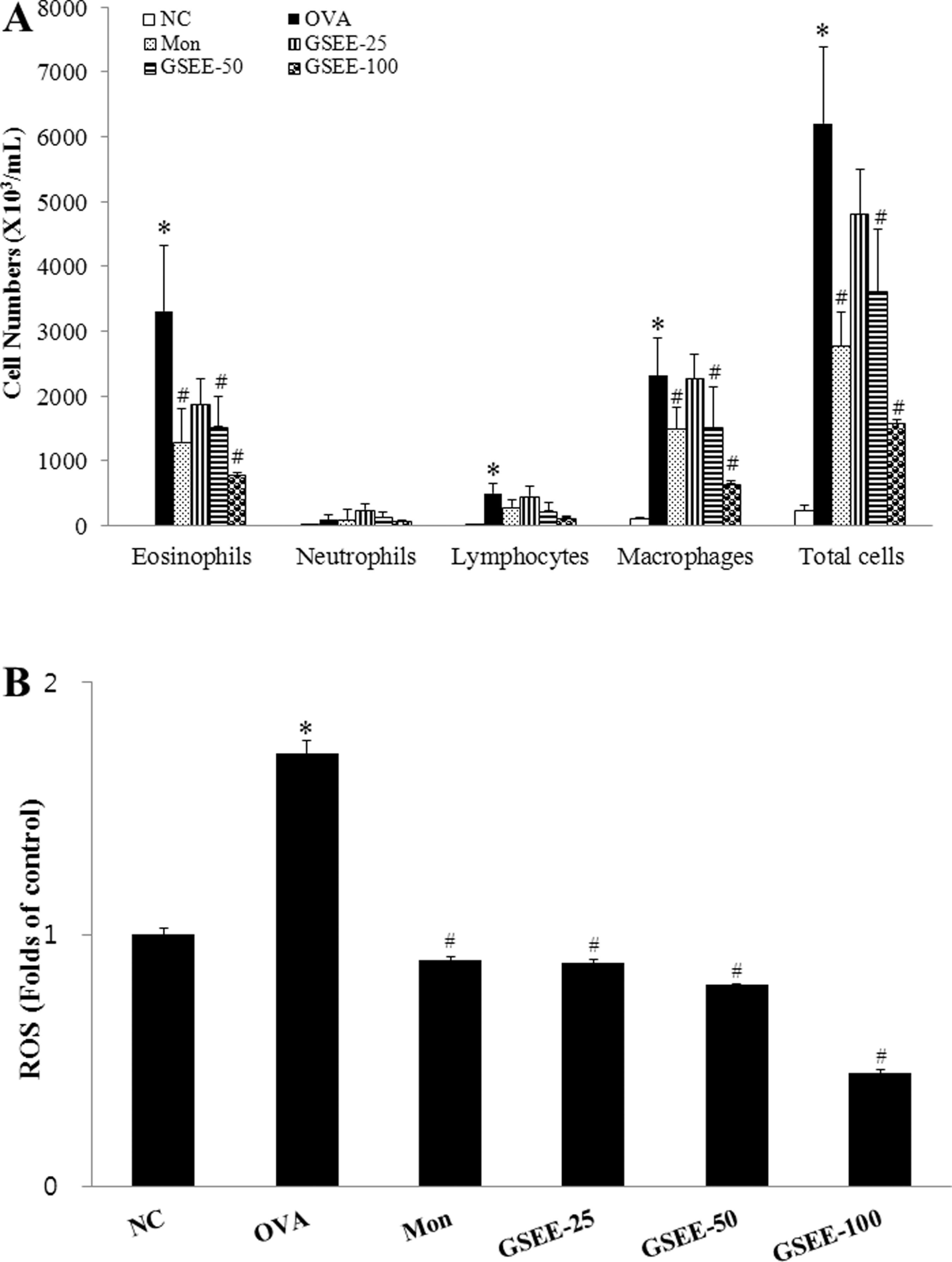
Effects of GSEE on recruitment of inflammatory cells and ROS in BALF cells of mice 48 hours after the final OVA challenge. A, Cell numbers of eosinophils, macrophages, lymphocytes, and neutrophils. B, Oxidative stress. Values represent relative fluorescence units per milligram of protein. NC indicates negative control (phosphate-buffered saline [PBS] only); OVA, ovalbumin-sensitized/challenged mice; GSEE-25, GSEE (25 mg/kg) + OVA-sensitized/challenged mice; GSEE-50, GSEE (50 mg/kg) + OVA-sensitized/challenged mice; GSEE-100, GSEE (100 mg/kg) + OVA-sensitized/challenged mice; ROS, reactive oxygen species; BALF, bronchoalveolar lavage fluid; SD, standard deviation. Gleditsia sinensis ethanolic extract or montelukast treatment was performed 1 hour before challenge. Values are expressed as means ± SD (n = 6 per group). *Significantly different from NC, P < .05; #significantly different from OVA, P < .05.
Reactive oxygen species levels in BALF cells were markedly increased (1.72-fold, compared with controls) in OVA-challenged mice, as shown in Figure 2B. However, preadministration of GSEE led to a significant dose-dependent decrease (GSEE-25, 0.89-fold; GSEE-50, 0.80-fold; and GSEE-100, 0.45-fold; P < .05 vs OVA-induced group) in the ROS level compared with the OVA-induced group (Figure 3B).
Effects of GSEE on Cytokine Levels in BALF
The effects of GSEE on cytokine release in the OVA-induced asthmatic mouse model were assessed by ELISA to measure the BALF cytokine (IL-4 and IL-5) levels 48 hours after the final challenge. Ovalbumin challenge led to significantly increased IL-4 and IL-5 levels in BALF compared with the control group. In the GSEE-treated group, cytokine elevation was suppressed markedly in a dose-dependent manner relative to the results with the OVA-challenged group. Montelukast (positive control) also induced significant (P < .05) decreases in IL-4 and IL-5 levels (Figure 4A and B). Clearly, GSEE suppressed IL-4 and IL-5 expression in the BALF of the asthmatic mouse model, as did montelukast.
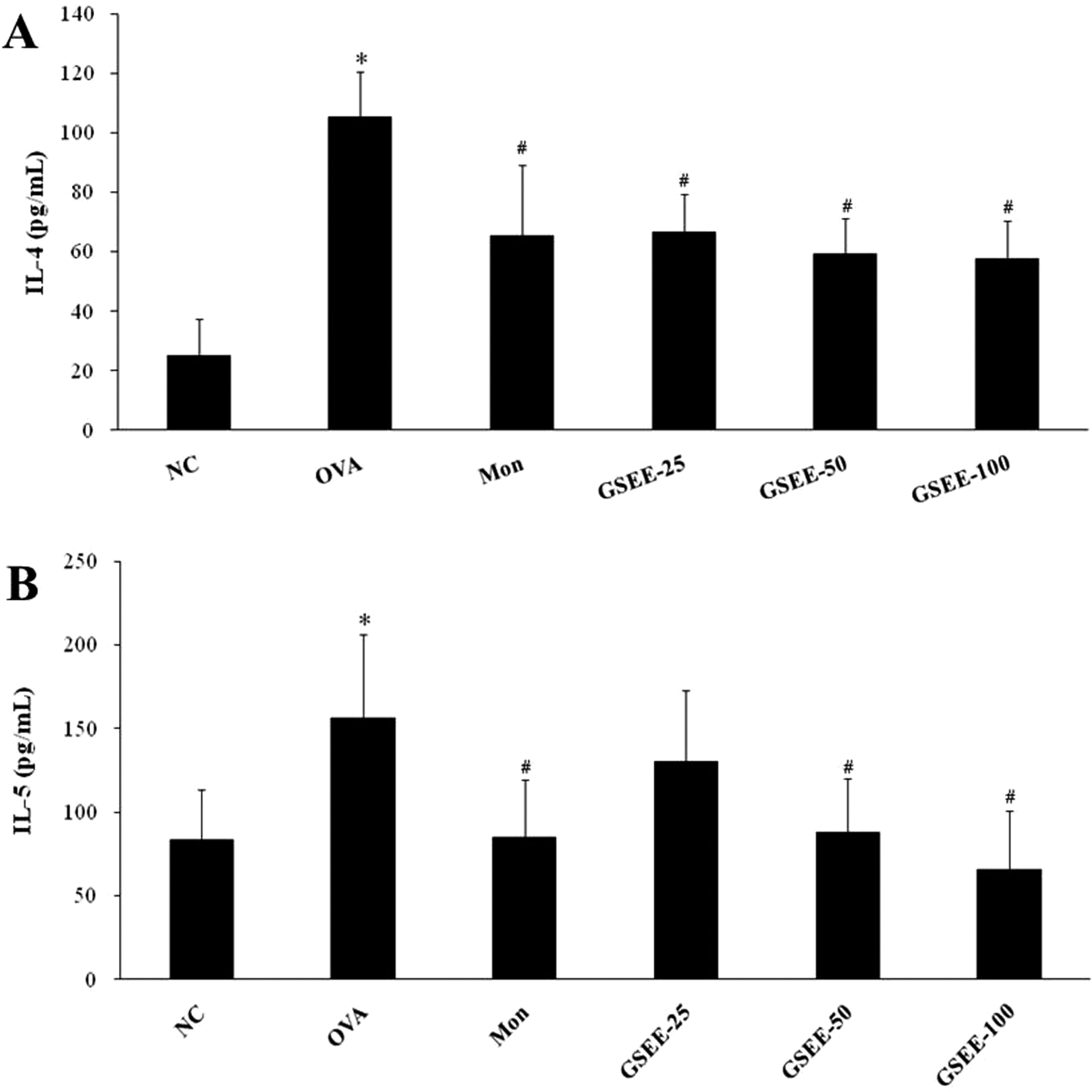
Effects of GSEE on cytokine levels in BALF. A, IL-4 level in BALF. B, IL-5 level in BALF. NC indicates negative control (phosphate-buffered saline [PBS] only); OVA, ovalbumin-sensitized/challenged mice; GSEE-25, GSEE (25 mg/kg) + OVA-sensitized/challenged mice; GSEE-50, GSEE (50 mg/kg) + OVA-sensitized/challenged mice; GSEE-100, GSEE (100 mg/kg) + OVA-sensitized/challenged mice; BALF, bronchoalveolar lavage fluid; IL, interleukin; SD, standard deviation. Gleditsia sinensis ethanolic extract or montelukast treatment was performed 1 hour before challenge. Values are expressed as means ± SD (n = 7 per group). *Significantly different from NC, P < .05; #significantly different from OVA, P < .05.
Effects of GSEE on Airway Eosinophilia and Mucus Production in Lung Tissue
The anti-inflammatory effects of GSEE were further confirmed by histological examination. Inflammatory cells were not detected in peribronchial or peribronchiolar lung areas of PBS-challenged mice. In contrast, leukocytes infiltrated peribronchiolar and perivascular connective tissue in OVA-challenged lung tissue. The majority of inflammatory cells found infiltrating were eosinophils. Gleditsia sinensis ethanolic extract (50 and 100 mg/kg) dose-dependently attenuated the eosinophil-rich leukocyte infiltration, compared with the level seen in OVA-challenged mice (Figure 5A and C).
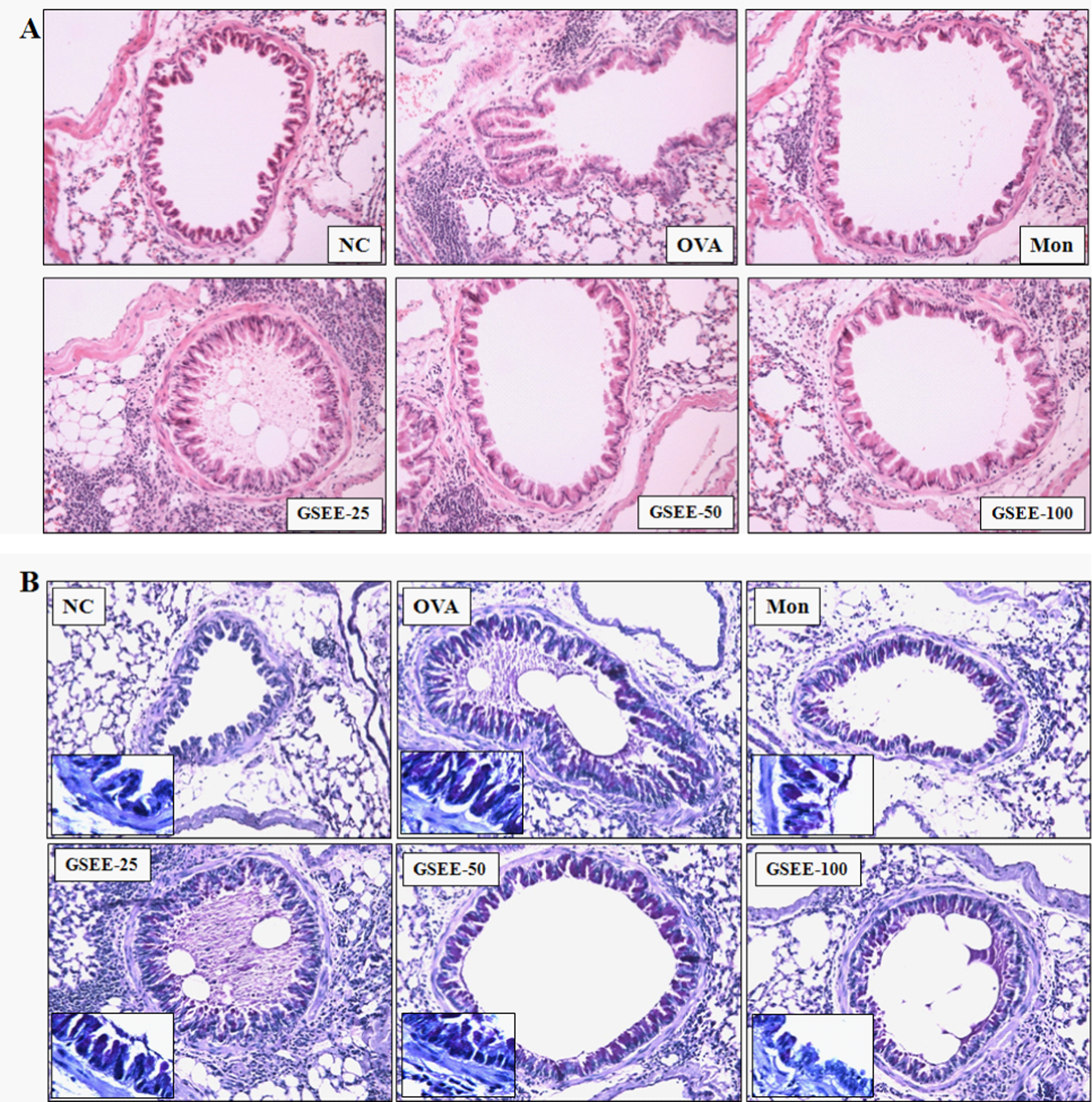
Effects of GSEE on recruitment of leukocytes and mucus production in lung tissue. A, H&E staining (original magnification, ×200). B, Periodic acid-Schiff (PAS) staining. (original magnification, ×200 [inset, ×400]). C, Scoring of the extent of inflammation by quantitative analysis of inflammatory cell infiltration in lung sections. D, Scoring of mucus production in lung sections. NC indicates normal control mice (phosphate-buffered saline [PBS] only); OVA, ovalbumin-sensitized/challenged mice; GSEE-25, GSEE (25 mg/kg) + OVA-sensitized/challenged mice; GSEE-50, GSEE (50 mg/kg) + OVA-sensitized/challenged mice; GSEE-100, GSEE (100 mg/kg) + OVA-sensitized/challenged mice; Mon, montelukast (30 mg/kg) + OVA-sensitized/challenged mice. Gleditsia sinensis ethanolic extract or montelukast treatment was performed 1 hour before challenge. Values are expressed as means ± SD (n = 6 per group). *Significantly different from NC, P < .05; #significantly different from OVA, P < .05.
Mucus hypersecretion is characteristic of the airway response to inflammation. To assess possible GSEE-induced suppression of mucus overproduction caused by goblet-cell hyperplasia, we stained lung sections with PAS. The mucus area was scored as 3.39 ± 0.01 in the OVA-treated mice, but this decreased significantly (P < .05) in the GSEE 50 and 100 mg/kg groups, and levels in the GSEE groups were also lower than those found in the positive reference, montelukast (Figure 5B and D). This is consistent with the above results and clearly supports the finding that GSEE inhibited airway inflammation in asthma.
Effects of GSEE on Total IgE Release Into BALF and Plasma
Total IgE levels were evaluated by ELISA in each experimental group 48 hours after the final OVA challenge. Total IgE levels in BALF and plasma were markedly increased in OVA-challenged mice, compared with negative controls. In contrast, GSEE treatment led to a reduction in total IgE levels in both the BALF and the plasma of OVA-challenged mice (Table 1).
Effects of GSEE on Total IgE Levels in BALF and Plasma
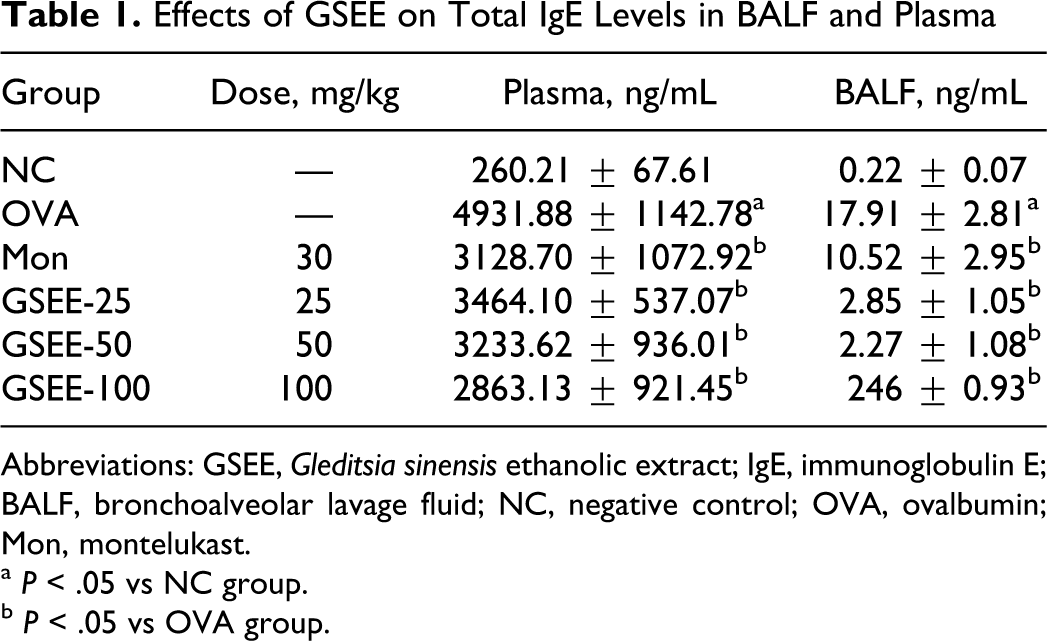
Abbreviations: GSEE, Gleditsia sinensis ethanolic extract; IgE, immunoglobulin E; BALF, bronchoalveolar lavage fluid; NC, negative control; OVA, ovalbumin; Mon, montelukast.
a P < .05 vs NC group.
b P < .05 vs OVA group.
Discussion
The thorns of G sinensis have been used in the prevention and treatment of carbuncles, scabies, and suppurative skin diseases to provide anti-inflammatory effects, 30 but the molecular mechanism responsible for its antiasthma effect has not been fully elucidated. In this study, we examined the therapeutic effects of orally administrated GSEE in an OVA-induced murine airway inflammation model. Allergic airway inflammation is a central feature of several diseases and many long-term effects are attributable to the actions of inflammatory cells recruited to the airway. 31 Before investigating the effects of GSEE (25, 50, and 100 mg/kg) in a mouse model of allergic asthma, we made a preliminary study of GSEE dose effects (100 and 200 mg/kg) on plasma IgE to determine the optimal dosage and dose-dependent effects of GSEE (data not shown). Both doses reduced IgE levels, so the lower dosage was chosen for the main study. Asthma is a common pulmonary disease and its prevalence has increased substantially in the last decades. 32 This increase is related to various environmental changes. According to the recent studies, dietary changes, including reduced antioxidant intake, contribute to the increase in prevalence of asthma. 33 The increased air pollution caused by industrialization generates allergic chemicals and increases ozone levels to induce the onset of allergic asthma. 34 Western housing styles also promote greater exposure to indoor or outdoor allergens such as dust mites, pollen, animal dander, and mold, and much research has focused on increased exposure to these allergens as a cause of the rise in asthma.35,36 Here, we showed that GSEE suppressed eosinophilic infiltration (50 and 100 mg/kg) and mucus production in the lungs and airways, and inhibited systemic eosinophil recruitment and Th2 cytokines in the BALF at 50 and 100 mg/kg.
Reactive oxygen species may contribute to tissue injury and inflammatory reactions. Accumulating clinical and experimental evidence indicates that ROS plays an essential role in the pathogenesis of airway inflammation. 16 - 18 Eosinophils are known to act as the primary effector cells in the pathogenesis of asthma through the release of ROS and specific granule proteins. 37 Many substances, including allergens, gaseous pollutants, chemicals, drugs, bacteria, and viruses, can cause recruitment and activation of inflammatory cells in asthmatic airways. The activated inflammatory cells generate ROS, which they release into the surrounding cells. When ROS levels overwhelm host antioxidant defenses, the oxidative stress causes many detrimental effects on airway functions, including airway smooth muscle contraction, induction of airway hyperresponsiveness (AHR), mucus hypersecretion, epithelial shedding, and vascular exudation. 7 Therefore, reduction in intracellular ROS levels during inflammation could potentially be valuable in the treatment of inflammatory lung diseases. Consistent with these findings, we demonstrated that GSEE has potent antioxidant effects in an OVA-induced murine asthma model, as measured by DCF fluorescence (25, 50, and 100 mg/kg). This was principally attributed to a reduction in the level of inflammatory cell recruitment, which was significantly elevated in OVA-treated mice. Gleditsia sinensis ethanolic extract induced a marked suppression of ROS levels in BALF cells. Inflammatory cells recruited to asthmatic airways have a heightened ability to produce ROS. Gleditsia sinensis ethanolic extract administration led to a reduction in eosinophils in BALF. However, we observed no changes in the numbers of other leukocytes, including neutrophils and lymphocytes. The specific reduction in eosinophils observed in the present study may be attributable to decreased IL-5-dependent eosinophil recruitment to the lungs. Histological analysis revealed that OVA-treated mice expressed peribronchial and perivascular infiltrations composed mainly of eosinophils. However, GSEE inhibits the pulmonary accumulation of eosinophils upon injection in mice, in parallel with a decrease in the IL-5 level in BALF. The results suggest that GSEE plays a key role in blocking recruitment of eosinophils in the lung via partial inhibition of IL-4- and IL-5-dependent pathways. Goblet cells are normally absent from small airways, 38 but they are observed in these airways in patients with chronic airway inflammatory conditions, where they contribute to bronchiolar obstruction. 39 In the present study, the percentage of goblet cells and mucus hypersecretion in the distal conducting airways was also dramatically reduced by GSEE treatment (50 and 100 mg/kg). Our findings implied that hyperplasia of goblet cells and mucus hypersecretion was attenuated by the suppressive effects of GSEE on the cytokine production and eosinophilia. We suggest that the reduction in the level of PAS-stained goblet cells in smaller airways by GSEE might be critical to the relief of asthmatic symptoms. Romagnani and colleagues 40 proposed a central role of Th2 cytokines in the pathogenesis of asthma. Both Th1 and Th2 responses appear to be reciprocally regulated in vivo, so modulation of the Th1/Th2 balance should be a valuable therapeutic strategy for asthma treatment. 41 In the present study, we showed that the elevation of Th2-type cytokines (such as IL-4 and IL-5) in BALF is suppressed by GSEE. Interleukin 4 is essential for the early expansion phase of the allergic response and acts in synergy with IL-5 to promote eosinophilic inflammation. 10 Therefore, GSEE-induced suppression of IL-4 (25, 50, and 100 mg/kg) may also contribute to a decreased level of lung eosinophils. Interleukin 4 additionally promotes IgE isotype switching and upregulation of MHC-II in B cells, which is associated with Th2 allergic responses. 42 In our experiments, oral administration of GSEE inhibited IgE production in BALF and plasma (25, 50, and 100 mg/kg). Gleditsia sinensis ethanolic extract induced a significant reduction in total ROS levels, the numbers of airway inflammatory cells, and IgE, IL-4, and IL-5 concentrations in BALF and plasma in the OVA-induced asthmatic mouse model. These findings provide further evidence that oral administration of GSEE might be an effective therapeutic candidate for the treatment of allergic diseases such as asthma. We analyzed 5 GSEE components by HPLC, that is, caffeic acid, (-)-epicatechin, eriodictyol, ethyl gallate, and quercetin. We confirmed the protective effect of GSEE on allergic asthma in the present study but could not determine the components that were responsible for its protective effects on allergic asthma. Therefore, further detailed experiments will be needed to determine the component responsible for the protective effects and to explore its mechanism.
Footnotes
Acknowledgments
This work was supported by grants from the Korea Research Council of Fundamental Science & Technology of the Republic of Korea.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by project, ‘Development of herbal materials for treatment and prevention of allergic disease,’ which was funded by the Korea Research Council of Fundamental Science and Technology.
