Abstract
BDE-85 is a congener of a class of flame-retardant compounds called polybrominated diphenyl ethers (PBDEs). Although there are some studies on other congeners of PBDEs, there are none on the toxicity potential of this penta-BDE member. This study, therefore, reports the oxidative status and sciatic nerve conduction properties following BDE-85 treatment in rodents. The oxidative stress markers, lipid hydroperoxides, and the activities of antioxidant enzymes, namely superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione-S-transferase (GST), and catalase, in the exposed mice liver and brain tissues showed tissue-specific alterations following intraperitoneal injection of 0.25 mg/kg body weight of BDE-85 for 4 days. The results indicate a significant disruption in the oxidant/antioxidant equilibrium and setting in of oxidative stress. Isolated sciatic nerves of rats exposed to 5 µg/mL or 20 µg/mL of BDE-85 showed a significant reduction in nerve conduction velocity and compound action potential amplitudes, indicating physiological damage to the sciatic nerves.
Keywords
Polybrominated diphenyl ethers (PBDEs) are a class of flame retardants that are added to many commercial and household products such as computers, TVs, foam mattresses, carpets, and so on to inhibit combustion and to prevent fire accidents. Due to their persistent nature, they have become widespread environmental contaminants and about 97% of the American adult population has detectable levels of PBDEs. 1 They are reported to be present in sediments, soil, outdoor and indoor air, household dust, foods, birds, fish, and terrestrial organisms. 2 Despite their ban in many countries, vast amounts of these compounds are found to persist in existing consumer products, potentially contributing to environmental and human burdens for years to come. Humans are exposed to PBDEs through inhalation of household and workplace dust 3 and eating PBDE-contaminated foods. 4
Commercial PBDE mixtures are available in 3 forms, namely, penta-BDEs, octa-BDEs, and deca-BDEs. There are 46 possible congeners of penta-BDE, out of which only 6 are used extensively in commercial products. Most of the studies on penta-BDEs are centered on BDE-47, BDE-99, and BDE-100. 5 –8 However, BDE-85 (Figure 1 ) remains overlooked in spite of its greater potency to bind to aryl hydrocarbon receptor (AhR), which indicates its higher potential for toxicity compared to other PBDEs. 9 Congener-specific distribution and metabolism of PBDEs makes it even more important to consider differential toxicities when assessing the risk to human health. Further, the exact mechanism of toxicity of PBDEs is still to be elucidated. There appeared a school of thought that PBDEs, by reason of their electron affinity, favor free radical formation in various tissues. 10 In the light of the above facts, we have surmised oxidative stress induction following BDE-85 exposure in mice. This is also based on a recent study showing the induction of oxidative stress in rat neurons exposed to BDE-47, another congener of penta-BDE mixture. 11 Therefore, oxidative stress indicators such as lipid hydroperoxides, activities of superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione S-transferase (GST), and catalase were analyzed in the liver and brain tissues of BDE-85-treated mice. The reason for the present choice of tissues stemmed from the fact that liver is a major biotransforming organ and brain the most sensitive target to lipid-soluble toxicants.
Chemical structure of BDE-85.
Another objective of this study is to report the impact of BDE-85 on the peripheral nerve functions. Impact of PBDEs on the nervous system was studied predominately in developing animals, mostly concentrating on their behavior, learning, and memory. 5 –7,12 A report on mice after a single oral dose of PBDE-99 has shown to alter several proteins involved in the neurodegeneration and metabolism. 13 In addition, some in vitro studies on PBDEs report induction of oxidative stress in primary rat hippocampal neurons and human cells. 11,14 If BDE-85 was to cause the generation of reactive oxygen species (ROS) and exerts oxidative stress, then it is imperative to record its effect on the peripheral nervous system as this has an increasing implication on the role of oxidative stress in neurodegeneration. Toward this end, compound action potentials (CAPs), area under CAP amplitudes, and nerve conduction velocity (NCV) were measured in isolated rat sciatic nerves exposed to BDE-85. Although assessment of neurotoxicity in behavioral terms is commonplace, formulating it on the electrophysiological platform is something that is being done for the first time in the study of BDE-85 toxicity.
Materials and Methods
Chemicals
BDE-85-CS (purity 98%, certified standard) was procured from Cambridge Isotope Laboratories, Inc (Andover, Massachusetts). Assay kits for the determination of lipid hydroperoxides, SOD, GPx, catalase, and GST were purchased from Cayman Chemical Company (Ann Arbor, Michigan). bicinchoninic acid (BCA) protein assay kit was purchased from Pierce Biotechnology Inc (Rockford, Illinois), for the protein assay of the sample. All other chemicals were obtained from Sigma-Aldrich, St. Louis, MO. The protocols followed in all the assays were reviewed and approved by the Biology Department, Gannon University, Erie, Pennsylvania.
Mice
Adult male Swiss Webster mice were obtained from Taconic Inc (Hudson, New York) and used for oxidative stress studies. Mice weighing 33 ± 3 g were maintained at 23°C ± 2°C with 12-hour light/12-hour dark photoperiods. Mice were provided food pellets and water ad libitum and were allowed to acclimatize for 15 days. The guidelines set forth by the institutional animal care and use committee, Gannon University, Erie, Pennsylvania, were followed in the maintenance and use of mice. Mice were grouped into 2 groups randomly (n = 9). Group 1 was injected intraperitoneally with 0.25 mg/kg body weight (bw) of BDE-85 for 4 consecutive days, and group 2 served as control and received the same volume of vehicle solution, corn oil. Oral route of administration was not considered in this study as the primary aim of this work is to report the biochemical alterations induced at this concentration and administering via oral route results in elimination of some fraction of this chemical through feces. The present dosage of BDE-85 was selected based on previous reports on other penta-BDE congeners in rodents. 2,15 Further, the dosage for in vivo experiments in our study also is commensurate with the average levels of PBDEs (except BDE-209) found in household dust. 16 Mice were sacrificed on the fifth day and liver and brain tissues were separated, washed in phosphate-buffered saline (PBS), and weighed. For all the biochemical assays, a 5% tissue homogenate of liver and brain was prepared using appropriate homogenizing buffers.
Biochemical Assays in the Mice Liver and Brain Tissues
The protocols that came with the assay kits were used for all biochemical assays. Briefly, for the determination of lipid hydroperoxides, the tissues were homogenized in nanopure water. The lipid hydroperoxides from the homogenate were extracted into chloroform. The extract was then used for the determination of lipid hydroperoxides directly. For the determination of SOD activity, the tissues were homogenized in 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer containing 1mmol/L ethylene glycol tetraacetic acid (EGTA), 210 mmol/L mannitol, and 70 mmol/L sucrose. The SOD activity was determined from the quantity of tetrazolium salt formed. For the GPx assay, the tissue homogenates were prepared in cold Tris-HCl with 5 mmol/L EDTA and 1 mmol/L dithiothreitol (DTT). The GPx activity was measured indirectly by measuring the rate of oxidation of NADPH into NADP+. For the catalase assay, the tissue homogenates were prepared in cold potassium phosphate buffer containing 1 mmol/L EDTA. The catalase activity was determined from the formaldehyde formed due to its peroxidatic activity. For the GST enzyme assay, tissues were homogenized in cold potassium phosphate containing 2 mmol/L EDTA. Glutathione S-transferase, activity was measured from the quantity of CDNB conjugated to glutathione. The protein content of the enzyme samples was assayed using Pierce BCA Protein Assay Kit. Bovine serum albumin was used as the standard.
Compound Action Potentials and NCV in Rat Sciatic Nerves
Adult male Holtzman rats were purchased from Harlan Laboratories (Madison, Wisconsin) and were maintained and acclimatized at the same conditions as mentioned for mice. For this study, rats were used instead of mice due to larger size of their sciatic nerves. Guidelines set by the institutional animal care and use committee, Gannon University, Erie, Pennsylvania, were followed. Rats weighing about 200 ± 20 g were sacrificed and their sciatic nerves of about 2.2 to 2.6 cm long were excised and stabilized in continuously aerated mammalian Ringer solution for 2 to 4 minutes prior to experimentation. The isolated nerves were then placed in glass containers (10 mL capacity) with 3 mL of mammalian Ringer solution containing 5 µg/mL or 20 µg/mL of BDE-85 with constant aeration for 30 minutes. These dosages and duration of exposure were based on a previous report. 17 Control nerves were placed in Ringer solution for 30 minutes with constant aeration. After 30 minutes, the nerves were transferred to the nerve chambers for CAP and NCV measurements using the PowerLab data acquisition systems interfaced to a computer via LabChart 7.0 software both from ADInstruments, Colorado. The CAP amplitude for varying stimulus strength and NCV were measured in the 9 control sciatic nerves and 18 exposed nerves (of which 9 were exposed to 5 µg/mL of BDE-85 and the remaining 9 were exposed to 20 µg/mL). The experimental setup is shown in Figure 2 . The nerve segment placed in the nerve chamber was excited using the stimulating electrodes and the CAPs were recorded with recording electrodes placed on the other end. From the actual nerve length between these 2 pairs of electrodes and the transit time taken by the stimulus to reach the recording electrodes, NCV was determined in a straightforward way.
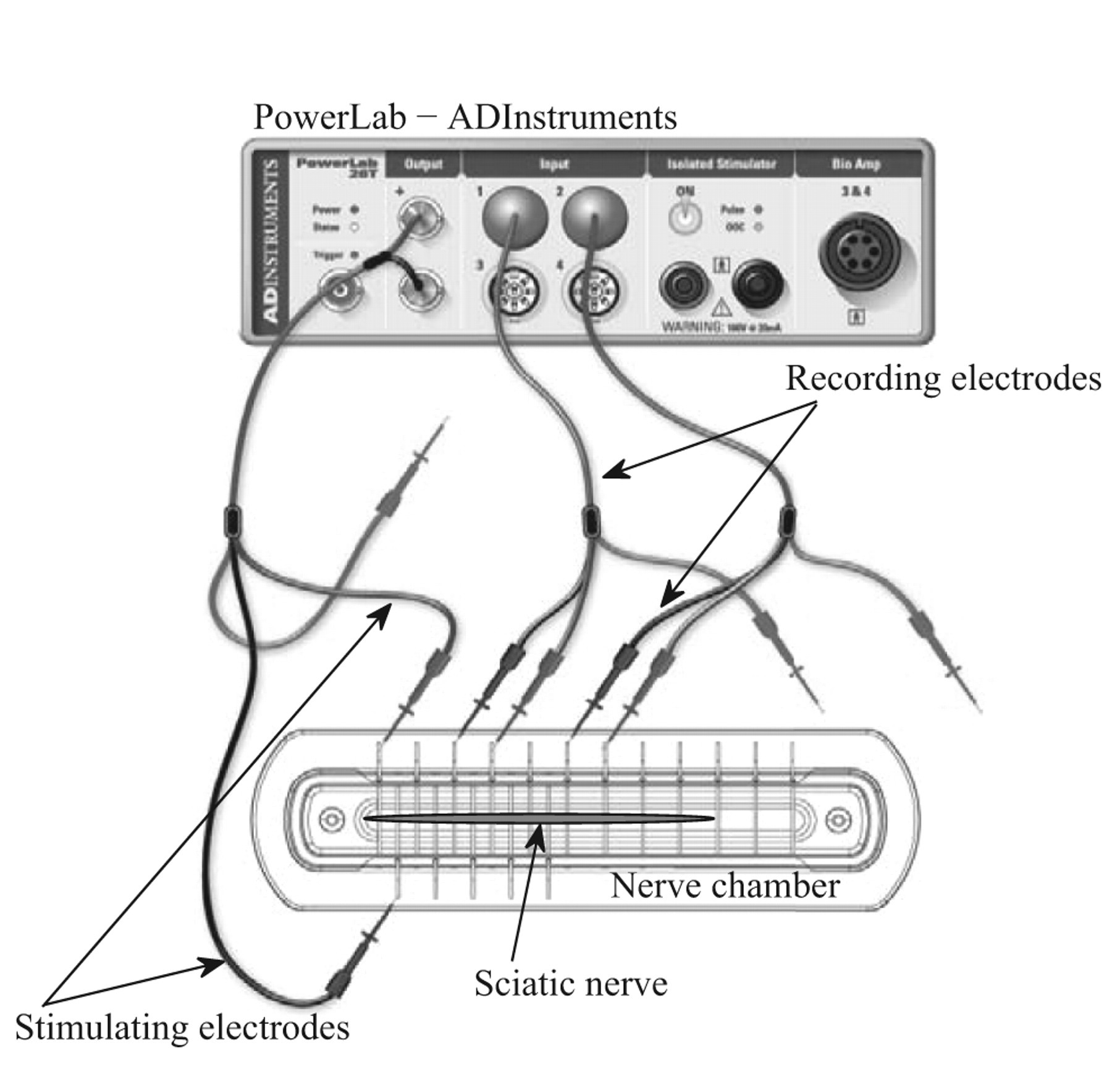
Experimental setup showing the nerve chamber, the nerve, and the stimulating and recording electrodes used in the CAP and NCV measurements. CAP indicates compound action potential; NCV, nerve conduction velocity.
Statistical Treatment of the Data
Data presented in Figure 3
(A-E) are mean ± standard deviation (SD) of 9 animals per group. The data were subjected to analysis of variance (ANOVA) and when means were significantly different they were further subjected to Tukey test. The percentage variation from controls was calculated and presented in the following results section. A value of
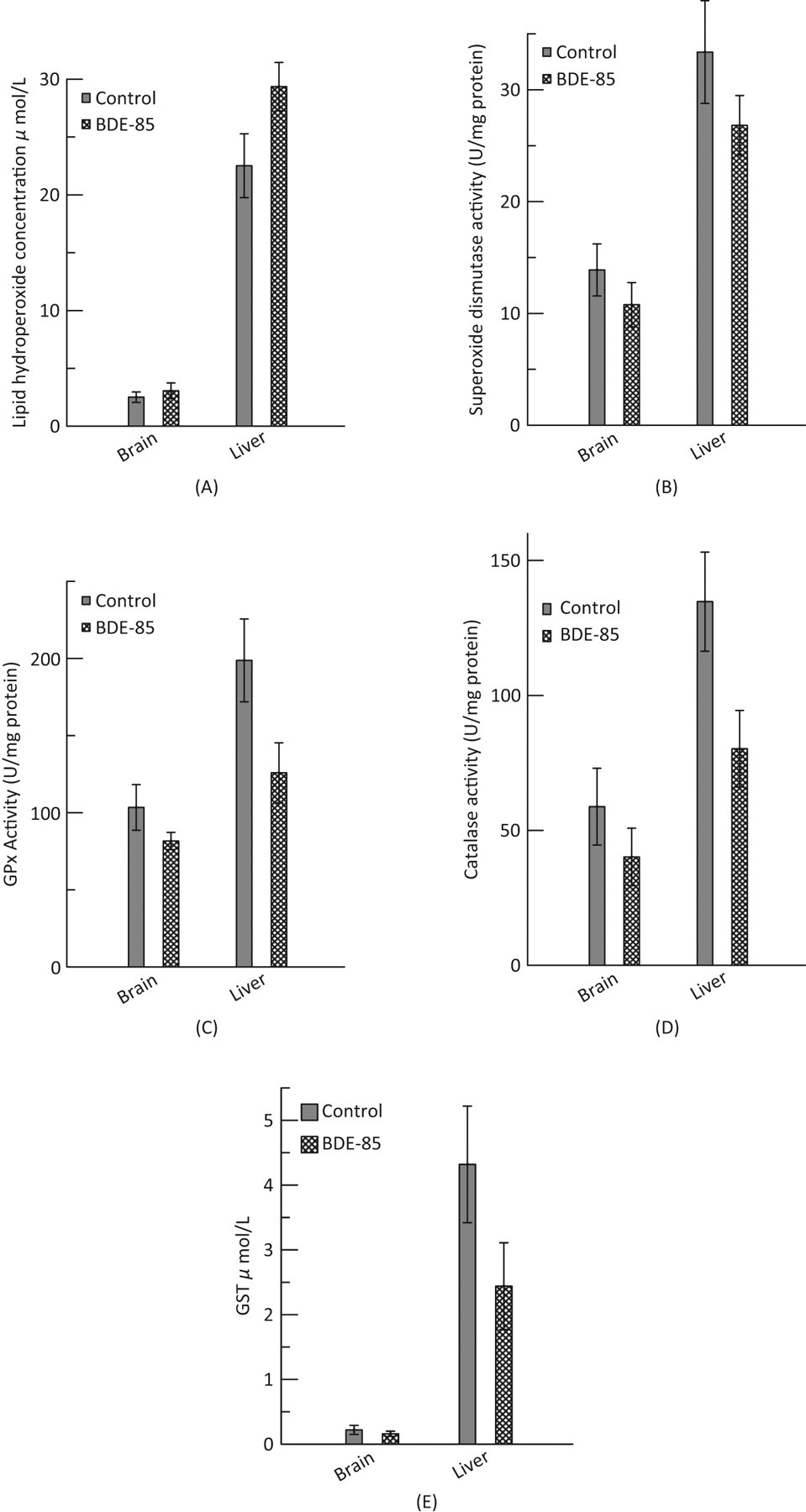
Oxidative stress markers following BDE-85 treatment. A, Lipid hydroperoxides: values are mean ± SD. Liver
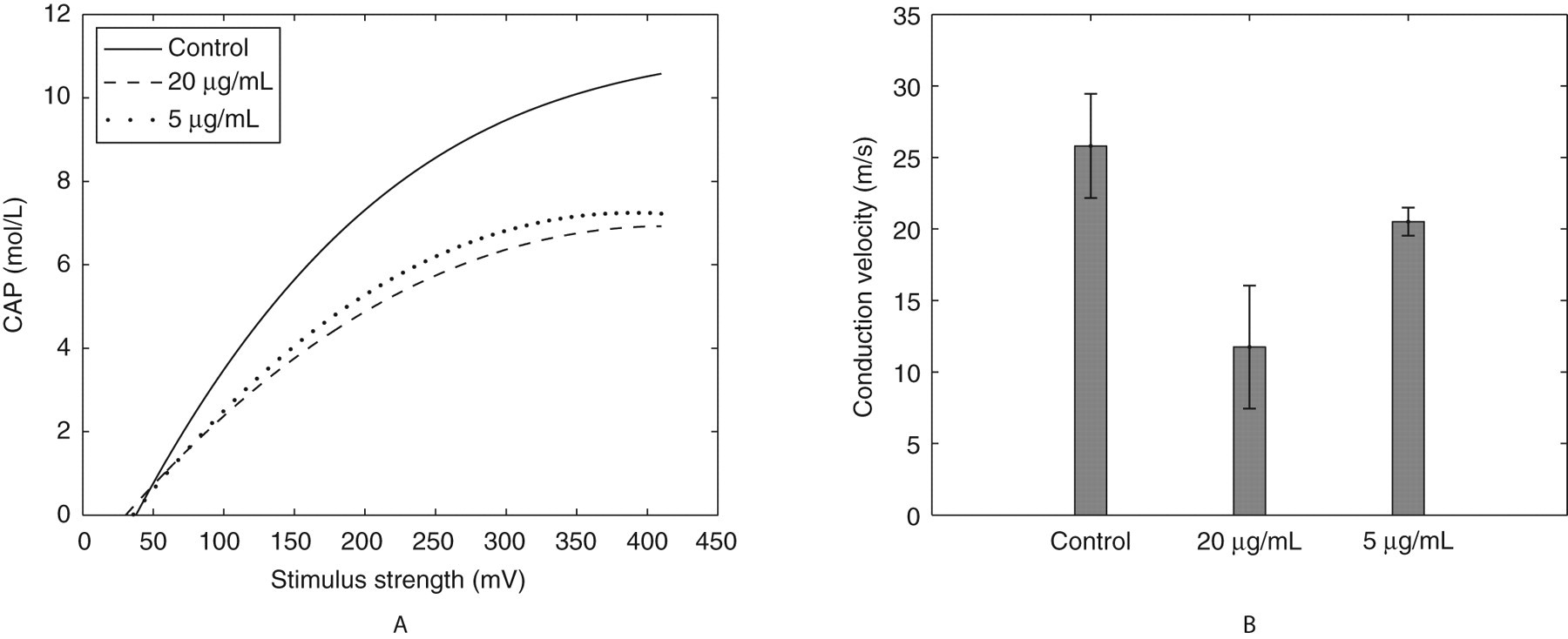
A, Compound action potential (CAP) amplitudes in control and BDE-85 (20 and 5 µg/mL)-treated rat sciatic nerves. Each curve is a best-fit cubic polynomial obtained from mean data of 9 nerves in each case. The coefficients of the polynomials and area under each curve are furnished in Table 1. The reduction in the area under the experimental CAP curves for both concentrations is significant at
Results
BDE-85–induced alterations in the lipid hydroperoxides and antioxidant enzymes of mice liver and brain tissue are shown in Figure 3 (A-E). The sciatic nerve function, in terms of CAPs and NCV after BDE-85 exposure, is shown in Figure 4 (A-B). The levels of lipid hydroperoxides in the exposed liver tissue were significantly (
The CAP amplitude as a function of stimulus strength is measured in the control and exposed sciatic nerves and the average values were used to fit a cubic polynomial: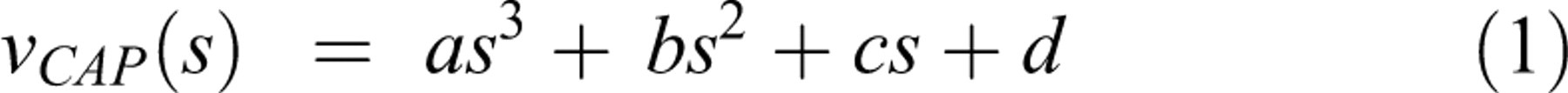
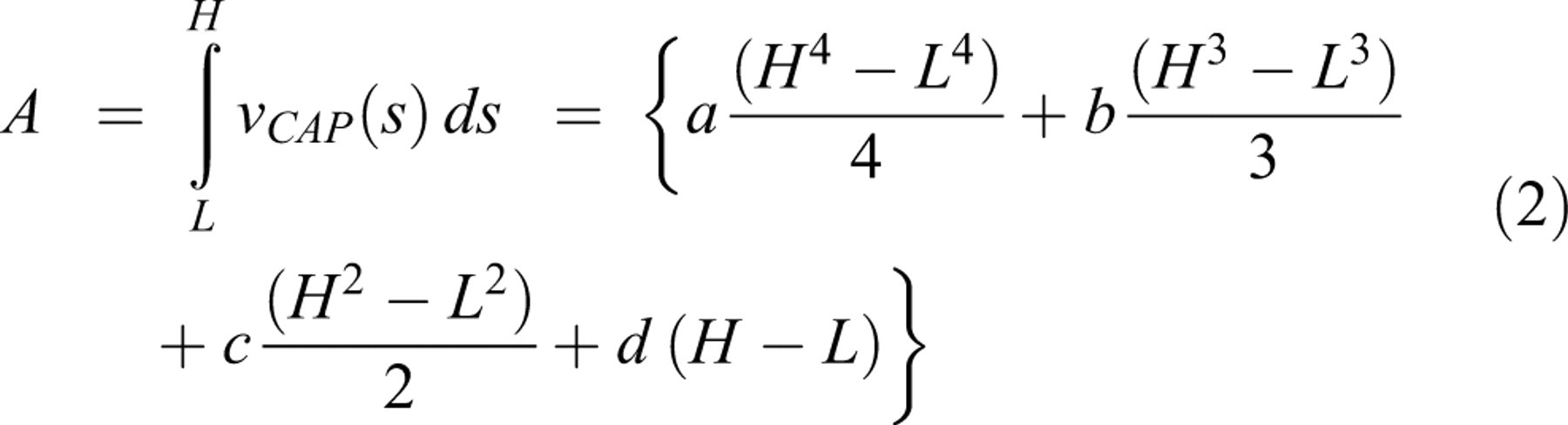
Parameters of the Cubic Polynomials Used to Fit the CAP Curves and the Area Underneath Over the Interval of 20-410 mV
Discussion
The results of this study indicate a clear susceptibility of mice tissues to BDE-85-induced oxidative damage. Tissue-specific alterations in the levels of lipid hydroperoxides were observed following BDE-85 treatment. Liver showed significantly elevated levels of lipid hydroperoxides, while the brain did not exhibit any significant changes (Figure 3A). Observation of elevated lipid hydroperoxide levels in liver substantiates higher lipid peroxidation and oxidative stress, which could be due to an immediate perturbation in metabolizing enzymes in liver. 18 The degree to which this can happen remains open for further research. Moreover, liver hydrolyzes PBDEs in its attempt to eliminate them from the body. In this process, the toxic metabolites formed might damage the liver. 19 Similarly, increased ROS production in liver during BDE-99 treatment 19 in rats supports our observation. The brain tissue, on the other hand, did not show significant increase in lipid hydroperoxides levels (Figure 3A). The differences in responses in liver and brain tissues could be due to their intrinsic biochemical and functional differences. The dosage used in this study could have resulted in this response in the brain tissue. A study 2 on the adult rat brains exposed to 2 different concentrations of BDE-99 showed contrasting results on TBARS (indicative of oxidative stress) levels. At lower dosage of exposure, the levels of TBARS were found to be reduced and at higher dosage they were increased. The exact mechanism underlying ROS generation during PBDEs is not clear, but it is suggested that they act as electron acceptors in biological systems under standard conditions. 10 Reports on PBDE exposure and their involvement in the release of [3H] arachidonic acid, protein kinase-C translocation, and disruption in calcium homeostasis 20,21 could further explicate the mechanism of ROS formation. 11
Another important observation of this study is that BDE-85 at 0.25 mg/kg bw for 4 days has resulted in an overall reduction in the activities of SOD, GPx, catalase, and GST (Figure 3B-E). Diminished activities of SOD, GPx, and catalase enzymes in both the liver and brain tissues indicate that these tissues are not able to quench ROS and oxidative stress. Reduced catalase activity reflects an inability of these tissues to eliminate hydrogen peroxide. Catalase and GPx enzymes exhibit a greater degree of cooperativity in their actions. 22 During increased hydroperoxide formation catalase activity depends on GPx activity from being inactivated (Figure 5 ). In the face of decreased GPx activity in BDE-85 exposed tissue, the catalase might be inactivated by higher concentration of hydroperoxides. Glutathione-S-transferase is another antioxidant enzyme whose activity showed a tissue-specific response during BDE-85 exposure. Liver GST was reduced, but brain GST did not show any change in the activity. Similar response in GST activity was observed in the brain regions of rats after BDE-99 treatment. 5 BDE-99 exposure in rats showed similar response of decreased antioxidant enzymes at 0.6 mg/kg bw. 2 In another report on BDE-99, rat liver and kidney showed an upsurge of lipid peroxidation and unstable antioxidant system, 19 rendering support to our observations.
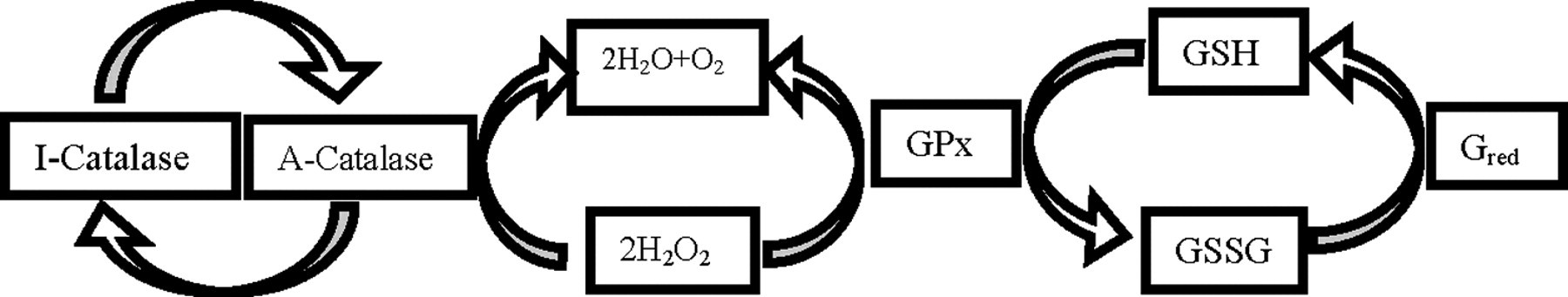
H2O2 is disposed by catalase and GPx. Glutathione acts as an electron donor and is recycled by glutathionc redutase (Gred). Auto-inactivation of catalase (I-catalase) by H2O2 is prevented by GPx, which maintains catalase in active form (A-catalase). 18 GPx indicates glutathione peroxidase.
In summary, BDE-85 has a grave potential to elicit a severe oxidative stress in mice. When compared to the work of other researchers on the toxic effects of BDE-47 and BDE-99, the present findings on the profile of BDE-85 stand consistent and appear to resonate the same theme that these fire retardants subject organisms to oxidative stress. A striking difference in BDE-85 congener is that in the liver tissue the percentage variation is more pronounced and is reflected not only in lowered antioxidant enzymes but also in elevated lipid hydroperoxides, whereas the same extent of upheaval is not turned on in the brain tissue.
Compound Action Potential and Nerve Conduction Studies on Rats
The results show that BDE-85 at 5 or 20 µg/mL dosage can alter sciatic nerve function (Figure 4A, B). Compound action potential amplitudes (Figure 4A) depend on the stimulus strength in a nonlinear way. A cubic polynomial fit was found to result in the least residual error. A significant attenuation in the CAP amplitudes was noted at both the dosages indicating that BDE-85 is quite toxic at concentration as low as 5 μg/mL. The area under the CAP curve is an index of membrane depolarization and electrical summation of action potentials. A decrease in the area under the curve (Table 1) points to an impairment of cell membrane function, active transport of ions, alteration in the pre- and postsynaptic thresholds, and consequent degradation in the overall axon. It could also mean some degree of phase dispersion in the spatial and temporal summation of individual action potentials. This also could explain why PBDE-exposed animals are predisposed to neurological dysfunctions. 12
Another observation of this study is a pronounced reduction in conduction velocity at both the dosages (Figure 4B). The reduction in NCV was dose dependent. A precursor to delay in conduction speed is often damage to the myelin sheath; as such, BDE-85 according to the present study might deteriorate similarly. This comes as no surprise as myelin sheath is predominantly lipid and BDE-85 is a lipophilic toxicant. A reduction in conduction velocity was also observed during heavy metal lead exposure. 23 Although there are no direct reports on the involvement of PBDEs in human peripheral neuropathies, their structural counterparts, PCBs, are identified to cause peripheral neuropathy, 24 implicating PBDEs, potential to cause it. Overall, our observations of this study show a serious disruption in the sciatic nerve function in terms of reduced conduction property and low CAP amplitudes. Due to paucity in the supporting literature on the neurotoxicity of BDE-85, more studies are needed to confirm the myelin sheath integrity following exposure and to render more elaborate reasons for reduced CAP amplitudes.
Footnotes
Notes
Acknowledgments
Mary Vagula thanks Gannon University for the award of Faculty Research Grant. The authors also thank Dr Michael Rutter, Penn State Behrend, for his help in data analysis.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The first author (MCV) acknowledged receipt of the faculty research grant from the Gannon University, Erie, PA, USA.
