Abstract
Urotoxicity is a troublesome complication associated with cyclophosphamide (CP) and L-buthionine-SR-sulfoximine (BSO) treatment in chemotherapy. With this concern in mind, the present study investigated the potential effects of a hydroxytyrosol extract from olive mill waste (OMW) on urotoxicity induced by acute CP and BSO doses using a Swiss albino mouse model. Toxicity modulation was evaluated by measuring lipid peroxidation (LPO) and antioxidants in urinary bladder. The findings revealed that the hydroxytyrosol extract exerted a protective effect not only on LPO but also on enzymatic antioxidants. When compared to the controls, the CP-treated animals underwent significant decreases in the glutathione S-transferase (GST), glutathione reductase (GR), glutathione peroxidase (GP), and catalase (CAT) activities. The level of glutathione (GSH) was also reduced with increased doses of LPO in the CP-treated animals. L-Buthionine-SR-sulfoximine treatment exerted an additive toxic effect on the CP-treated animals. Interestingly, pretreatment with the hydroxytyrosol extract restored the activities of all enzymes back to normal levels and exhibited an overall protective effect on the CP- and BSO-induced toxicities in urinary bladder. The restoration of GSH through the treatment with the hydroxytyrosol extract can play an important role in reversing CP-induced apoptosis and free radical-mediated LPO.
Introduction
Cyclophosphamide (CP) is an alkylating antineoplastic agent with activity against a variety of human tumors. It is commonly used as an antineoplastic agent for the treatment of various forms of cancer and as an immunosuppressive drug for certain non-neoplastic conditions before organ transplantation. It is also widely employed in the treatment of lupus nephritis. 1 ,2 Several previous studies on murine models have also showed that the administration of CP retards the progression of kidney disease. 3 This agent has similarly been used extensively for the treatment of diffuse proliferative glomerulonephritis in patients with renal lupus. 3
L-Buthionine-sulfoximine (BSO) is a sulfoximine used in chemotherapy, which irreversibly inhibits g-glutamylcysteine synthetase, thereby depleting cells of glutathione (GSH), a metabolite that plays a critical role in protecting cells against oxidative stress and resulting in free radical-induced apoptosis. Elevated GSH levels are associated with tumor cell resistance to alkylating agents and platinum compounds. By depleting cells of GSH, this agent may enhance the in vitro and in vivo cytotoxicities of various chemotherapeutic agents in drug-resistant tumors. L-Buthionine-sulfoximine may also exhibit antiangiogenesis activity. 4
Despite being effective in the control of various malignant conditions, CP and BSO treatment has been occasionally reported to be associated with serious side effects. Although, CP was previously reported to be beneficial for the management of murine lupus, several concerns have been expressed about its side effects and the degree of immunosuppression. 3 ,5 In fact, CP intake is often reported to be associated with nausea, vomiting, hemorrhagic cystitis, hematopoietic depression, cardiac damage, alopecia, infertility, and leukemic CP-induced carcinogenicity. 6 ,7 Several reports have also linked cases of lung toxicity to this medication. 8
Similarly, several studies have demonstrated that bladder inflammation induced by CP in rats and mice increased transcript and protein expression 9 in the urinary bladder of several cytokines, including interleukin 6 (IL-6). Cyclophosphamide is bioactivated to its reactive electrophilic metabolites through the cellular cytochrome P450 enzyme system. This results in their ongoing production and slow accumulation in the plasma, causing severe cytotoxic effects on nontarget tissues. 10
Other studies demonstrated that CP treatment also results in the production of reactive oxygen species (ROS) that cause peroxidative damage to urinary bladder and other vital organs. 11 ,12 In fact, free radical generation is one of the mechanisms by which CP and its derivatives exert their toxic effects on different tissues. An overall decrease in the GSH content has often been reported in various tissues as a result of CP treatment. 8 ,13 The role of tissue antioxidants becomes important in the prevention of such peroxidative damage induced by CP treatment. A number of natural products and synthetic compounds have been shown to reduce CP toxicity mainly due to their antioxidant action. 13 ,14
The reduction in GSH (reduced GSH) levels as a result of CP treatment has also been reported in urinary bladder. 14 In fact, the reactive metabolites of CP are responsible for its various toxic as well as therapeutic actions. 6 ,15 Among these metabolites, phosphoramide mustard is specifically associated with the immunosuppressive action of CP. 15 Cyclophosphamide-induced immunosuppression is reported to prompt various types of infection. 16 Some of the infectious agents have GSH-depleting effects. 17 Cyclophosphamide treatment may, therefore, decrease the GSH content itself but the associated secondary infections are likely to cause an additional decrease in the GSH level. For that reason, a patient undergoing CP chemotherapy needs excessive supply of GSH-restoring antioxidants or compounds that induce GSH production.
A number of GSH-inducing compounds have been found to be effective in reducing CP toxicity in animals. 18 Several studies reported that olive mill waste (OMW) is a valuable compound particularly because it offers a rich source of a diverse range of biophenols with a wide array of biological activities. 10 ,19 Of particular interest, hydroxytyrosol (3,4-dihydroxyphenylethanol [3,4-DHPEA]; [HT]), an ortho-diphenol derived from the hydrolysis of oleuropein, is among the biophenol substances abundantly present in OMW that has recently received special attention because it may inhibit both the initiation and promotion steps of carcinogenesis in vitro.
In fact, 3,4-DHPEA possesses a clear scavenging activity toward different free radicals and is able to avoid several injuries caused by ROS, such as hydrogen peroxide-induced DNA strand breaks in Jurkat and prostate cancer cells, DNA base modification, and tyrosine nitration induced by peroxynitrite. 20 ,21 A complex of both a methanolic extract of the virgin olive oil containing several phenolic compounds and a purified 3,4-DHPEA has also been reported for its ability to induce apoptosis in different tumor cell lines. 22 Similarly, 3,4-DHPEA was shown to inhibit the proliferation of HL60 human promyelocytic leukemia cells and to alter cell cycle progression, inducing an accumulation of cells in the G0/G1 phase. 23 It should, however, be noted that the subcellular events contributing to the 3,4-DHPEA antiproliferative and cell cycle effects on tumor cells are still unknown.
Considering the serious concerns widely voiced about the undesirable side effects associated with CP treatment, the present study was undertaken to explore the potential effects that HT might offer to alleviate this disturbing problem. The study aimed to investigate, for the first time, the effects of a purified HT extract from OMW on the restoration of antioxidants and reduction of lipid peroxidation (LPO) in the urinary bladder of CP-treated animals that are predisposed or concomitantly exposed to GSH-reducing agents through either infection or the use of antibiotics.
Materials and Methods
Materials
The OMW used in the present study was supplied by a cooperative olive oil producer owning a discontinuous 3-phase olive processing mill (GREMDA, Sfax, Tunisia). The material was collected at the end of the olive harvest season (April 2010) and stored for 3 months at ambient temperature. Tyrosol, caffeic acid, para-coumaric acid, protocatechuic acid, and para-hydroxyphenylacetic acid (PHPA) were all purchased from Sigma-Aldrich, St. Louis, MO. The HT used as a standard was purified from OMW in the Center of Biotechnology at Sfax, as previously described. 24 All solvents were of analytical grade and used without further purification.
Chemicals
Cyclophosphamide monohydrate (2-[bis-(2-chloroethyl) amino] tetrahydro-2H-1,3,2-oxazaphosphorine 2-oxide monohydrate), CAS 6055-19-2, BSO, and CAS 5072-26-4 were purchased from Sigma-Aldrich. All other chemical products were also purchased from Sigma-Aldrich.
Extraction of Phenolic Compounds From OMW
Liquid-liquid extraction with ethyl acetate was carried out on OMW (2 L) in a separatory funnel. The mixture (solvent-OMW) was vigorously shaken for 10 minutes to achieve equilibrium and then allowed to settle for 30 minutes. The phases were separated and the extraction was repeated successively 4 times. All the runs were performed at ambient temperature (20°C-25°C). The ethyl acetate combined fractions were dried over anhydrous sodium sulfate and the ethyl acetate was evaporated in a rotary evaporator at 40°C to yield 21.35 g.
Hydroxytyrosol Purification and Identification
Hydroxytyrosol was purified as previously described by Hamden et al. 10 In brief, an aliquot (4 g) of the dry OMW extract was chromatographed on a silica gel column (4 × 80 cm) eluted with a solvent gradient: hexane-ethyl acetate-methanol. The chromatographic behavior was investigated by thin layer chromatography (TLC) analysis using silica gel plates (Merck, Quebec, Canada, 60 F-254). Nine groups of homogeneous fractions were collected. The third group was purified by preparative TLC that was eluted with a mixture of chloroform:methanol (8:2, v/v), which afforded 1 pure compound with Rf = 0.6. This compound was identified as HT by means of high-performance liquid chromatography-ultraviolet (HPLC-UV), gas chromatography–mass spectrometry (GC-MS) analyses, and proton nuclear magnetic resonance ( 1 H NMR) spectroscopy (VersaPakTM, C18 Cartridge 40 × 75 mm, MD). The 1 H NMR spectrum of the recovered HT was recorded at 303 K on a Bruker Avance 500 spectrometer operating at 500 MHz. Chemical shifts were reported in parts per million (ppm) downfield from the tetramethylsilane (TMS) signal reference (0.00 ppm).
High-Performance Liquid Chromatography-Ultraviolet Separation and Identification of Phenolic Compounds
The presence and amount of phenolic compounds in the OMW extract were studied by a reverse-phase HPLC-UV analysis with binary gradient elution using a Waters autopurification system equipped with a binary pump (Waters 2525), a UV-Vis diode array detector (190-600 nm, Waters 2996, Germany), and a PL-ELS 1000 ELS detector Polymer Laboratory. The chromatographic separation was achieved on a Kromasil C18 column (250 × 4.6 mm, ID, 5 µm; Thermo Göteborg, Sweden) with temperature being maintained at 40°C. The mobile phase consisted of 0.1% formic acid in water 24 versus 0.1% formic acid in acetonitrile 19 for a total running time of 60 minutes. The elution conditions were 0 to 30 minutes, 20% to 50% B; 30 to 35 minutes, 50% B; 35 to 45 minutes, 50% to 100% B; 45 to 55 minutes, 100% B; and 55 to 60 minutes, 100% to 20% B. The column was then subjected to washing and reconditioning steps for 10 minutes with 20% B. The flow rate was 0.6 mL/min and the injection volume was 50 mL. The retention times and UV spectra of the main phenolic compounds of the OMW extract were identified by comparison to those of pure standards.
Gas Chromatography–Mass Spectrometry Analysis
The identity of each peak was confirmed by GC-MS analysis performed on an HP Model 5872A equipped with a capillary HP5MS column (30-m length; 0.32-mm id.; 0.32-µm film thickness; France). The carrier gas was He with a flow rate of 1.7 mL/min. The oven temperature program was as follows: 1 minute at 100°C, from 100°C to 260°C at 4°C/min, and 10 minutes at 260°C. Trimethylsilyltrifluoroacetamide (BSTFA 100 µL) was added to 100 µL of the OMW ethyl acetate extract. The solution obtained was incubated within 20 minutes at 80°C. Ethyl acetate and BSTFA were evaporated under N2 flux. The residue was redissolved in ethyl acetate (1 mL) and analyzed by GC-MS. Identification of compounds by GC-MS analysis was carried out by comparing the retention times and mass spectra of the unknown peaks to those of the standards.
Animals
The experiments were performed on male Swiss albino mice (25 ± 2 g) provided by the animal service at the Pasteur Institute of Tunis, Tunisia. The study was approved by the Institutional Animal Ethics Committee (IAEC). The animals were bred and maintained under standard laboratory conditions (temperature 25°C ± 2°C; photoperiod of 12 hours). Commercial pellet diet and filtered water were given ad libitum.
Dosage and Experimental Groups
l-Buthionine-SR-sulfoximine, CP, and HT were suspended in normal saline. The animals were divided into 7 groups, namely groups I-VII, with each group consisting of 6 animals. Group I (control) referred to control mice that were administered normal saline orally (po) for 10 days and a single intraperitoneal (ip) injection on the 10th day. Group II (BSO) referred to the mice that received BSO (500 mg/kg body wt) and to which a single ip injection was administered on the 10th day. Group III (CP) designated the mice that received CP (50 mg/kg body wt) and a single ip dose on the 10th day of treatment. Group IV (BSO + CP) referred to animals that were administered BSO ip 5 hours before CP administration. Group V (CP + HT) referred to animals that were administered HT po for 10 days and a single ip injection of CP on the 10th day. Group VI (BSO + HT) designated animals that were given HT treatment (100 mg/kg body wt) po for 10 days and a single ip injection of BSO on the 10th day. Group VII (BSO + CP + HT) referred to the animals that were administered HT for 10 days and CP and BSO on the 10th day. Dosing was performed in such a way that all of the animals could be sacrificed on the same day, that is, day 11. The selection of BSO and CP doses was based on pilot experiments that involved the assay of a wide range of doses and on data provided from previously published reports. 13 ,25,26
Biochemical Investigations
Upon the completion of the treatment, the animals were sacrificed under mild anesthesia (methyl isopropyl ether) and their bladders were removed. The bladder tissue was homogenized in chilled phosphate buffer (0.1 mol/L, pH 7.4) using a Potter homogenizer. The homogenate was centrifuged at 10.500 rpm for 30 minutes at 4°C to obtain the post-mitochondrial supernatant (PMS), which was used for the biochemical measurements as described below.
Lipid peroxidation
Lipid peroxidation was measured using the procedure of Mihara et al. 27 The assay mixture consisted of 0.67% thiobarbituric acid (TBA; Central Pharmacy, Tunis, Tunisia), 10 mmol/L butylated hydroxy toluene ([BHT] Amresco Inc, Solon, Ohio), 1% ortho-phosphoric acid (Central Pharmacy), and tissue homogenate in a total volume of 3 mL. The rate of LPO was expressed as nmol of TBA reactive substances (TBARS) formed/h/g of tissue using molecular extinction coefficient epsilon (є) of 1.56 × 105/mol/L cm−1.
Measurement of GSH
GSH content was measured in the PMS of urinary bladder using the method of Haque et al 13 . Post-mitochondrial supernatant (1 mL) was precipitated with 1 mL of 4% sulfosalicylic acid (Amresco). The samples were incubated at 4°C for 1 hour and then centrifuged for 15 minutes at 1200 rpm and 4°C. The assay mixture consisted of 0.2 mL of filtered aliquot, 2.6 mL of sodium phosphate buffer (0.1 mol/L, pH 7.4), and 0.2 mL 100 mmol/L dithio-bis-2-nitrobenzoic acid ([DTNB] Sigma–Aldrich) in a total volume of 3 mL. The absorbance of the reaction product was measured at 412 nm and the results expressed as nmol GSH/g tissue.
Antioxidant enzyme measurements
Glutathione S-transferase (GST) activity was assayed using the method of Haque et al. 13 The reaction mixture consisted of 1.675 mL sodium phosphate buffer, 0.2 mL of 1 mmol/L GSH (Sigma-Aldrich), 0.025 mL of 1 mmol/L 1-chloro-2,4-dinitrobenzene ([CDNB] Amresco), and 0.1 mL of PMS in a total volume of 2 mL. The change in absorbance was recorded at 340 nm and the enzyme activity calculated as nmol CDNB conjugates formed/min/mg protein using epsilon (є) of 9.6 × 103/mol/L cm−1. Glutathione reductase (GR) activity was assayed by the method of Sharma et al. 28 The assay mixture consisted of 1.6 mL sodium phosphate buffer, 0.1 mL of 1 mmol/L ethylenediamine tetra acetic acid disodium salt ([EDTA] Amresco), 0.1 mL nicotinamide adenine dinucleotide phosphate reduced ([NADPH] Sigma-Aldrich), and 0.1 mL oxidized GSH (Sigma-Aldrich) and PMS (0.1 mL) in total volume of 2 mL. The enzyme activity measured at 340 nm was calculated as nmol NADPH oxidized/min/mg of protein, using epsilon (є) of 6.22 × 103/mol/L cm−1. Glutathione peroxidase (GP) activity was assayed using the method of Sharma et al. 28 The assay mixture consisted of 1.49 mL sodium phosphate buffer, 0.1 mL EDTA (1 mmol/L), 0.1 mL sodium azide (1 mmol/L; Central Pharmacy), 0.1 mL of 1 mmol/L GSH (Sigma-Aldrich), 0.1 mL NADPH (0.02 mmol/L), 0.01 mL of 0.25 mmol/L hydrogen peroxide (H2O2; CDH Chemicals, Mumbai, India), and 0.1 mL PMS in a total volume of 2 mL. The oxidation of NADPH was recorded spectrophotometrically at 340 nm. The enzyme activity was calculated as nmol NADPH oxidized/min/mg of protein using epsilon (∊) of 6.22 × 103/mol/L cm−1. Catalase (CAT) activity was assayed using the method of Haque et al. 13 The assay mixture consisted of 1.95 mL phosphate buffer, 1 mL H2O2 (0.09 mol/L), and 0.05 mL of PMS in a final volume of 3 mL. The change in absorbance was recorded kinetically at 240 nm. Catalase activity was calculated in terms of nmol H2O2 consumed/min/mg protein.
Protein measurement
Protein was measured by the method of Lowry et al. 29
Statistical Analysis
Single factor 1-way analysis of variance (ANOVA) was performed to determine significant differences in results of various groups. The statistical significance level was set at P values <.05. A Student–Newman–Keuls test was then carried out to analyze and compare the significance of the treatment groups. The values were expressed as mean ± standard error (SE).
Results
Identification, Quantification, and Isolation of Biophenols Extracted From Stored OMW
The chromatogram that was generated revealed several simple peaks corresponding to different phenolic monomers, among which 7 compounds were identified, namely HT, 3,4-dihydroxyphenyl acetic acid, tyrosol, cafeic acid, protocatechuic acid, p-coumaric acid, and p-hydroxyphenyl acetic acid. Minor unidentified components were also present in the ethyl acetate extract. The identification of these phenolic compounds was confirmed by GC-MS analysis (Table 1). The calculated concentrations are illustrated in Table 2. As expected, HT was the most abundant phenolic monomer in the OMW sample under investigation (2.652 mg/L in stored crude OMW). Tyrosol was also present at a significant concentration (1.286 mg/L in stored crude OMW). In fact, the HT concentration in fresh OMW (1.453 mg/L) was lower than that in stored OMW (2.652 mg/L). The chromatogram reported in Figure 1 contains 1 single peak at 16.59 minutes. Its UV spectrum shows 2 main bands at 230 nm and 277 nm that were identical to those observed in the UV spectrum of the standard sample used (Figure 2).
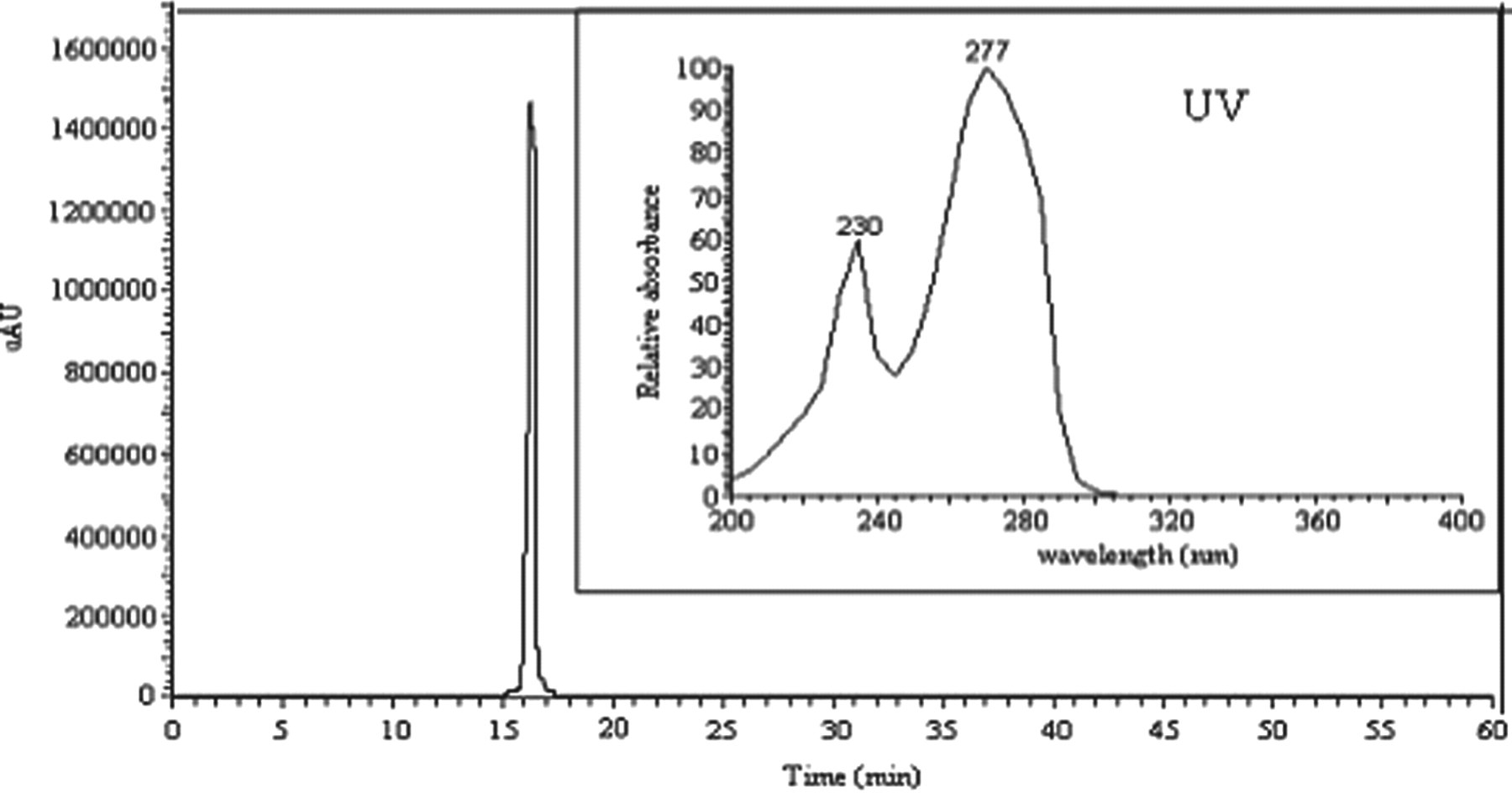
Analysis of the purified hydroxytyrosol from the stored OMW using HPLC-UV. HPLC-UV indicates high-performance liquid chromatography-ultraviolet; OMW, olive mill waste.
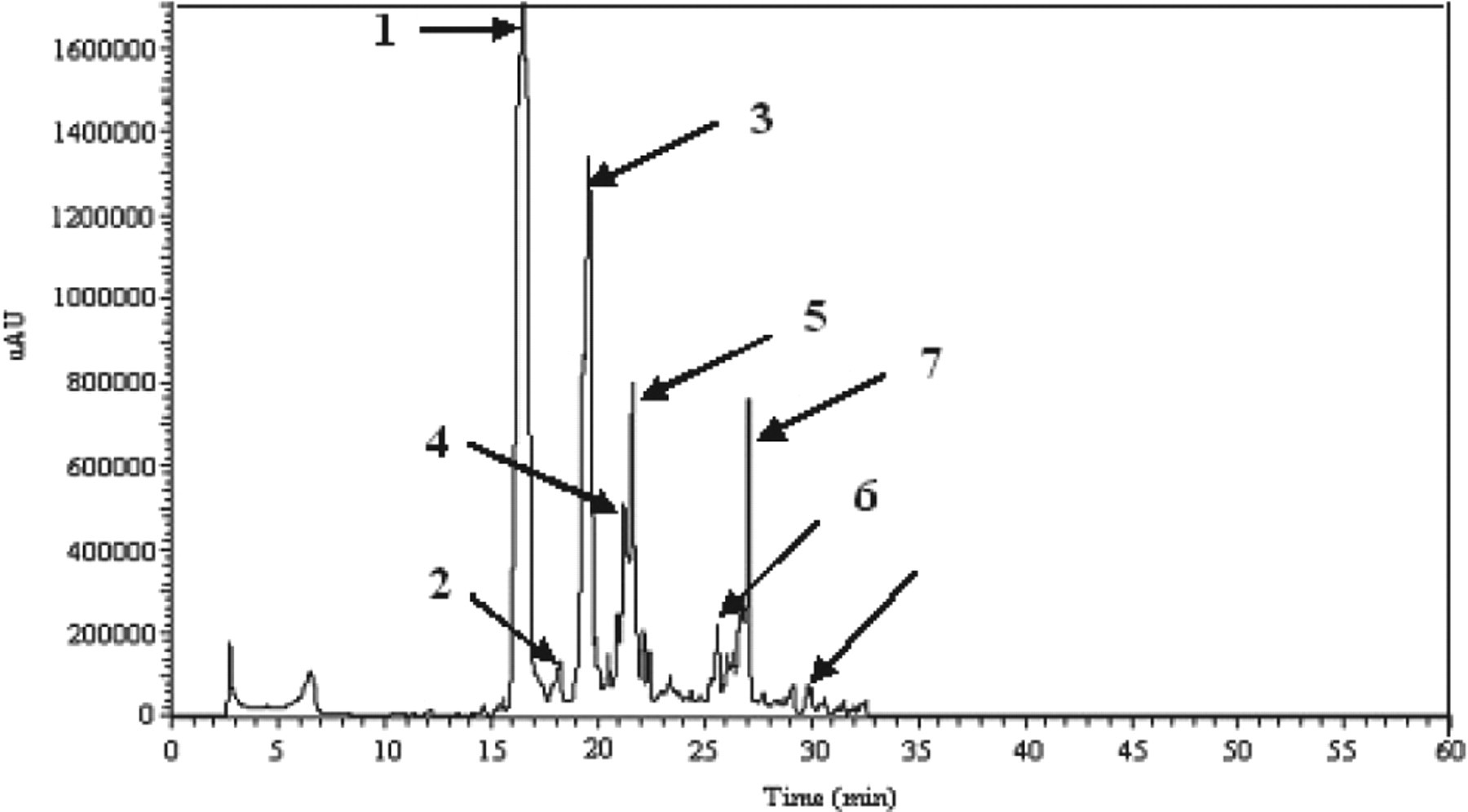
High-performance liquid chromatography analysis of ethyl acetate extract from stored olive mill waste (OMW): (1) hydroxytyrosol, (2) 3,4-dihydroxyphenylacetic acid, (3) tyrosol, (4) unknown, (5) cafeic acid, (6) protocatechuic acid, (7) p-coumaric acid, and (8) p-hydroxyphenylacetic acid.
Identification and Concentrations of the Principal Phenolic Compounds in the Stored OMW Extract
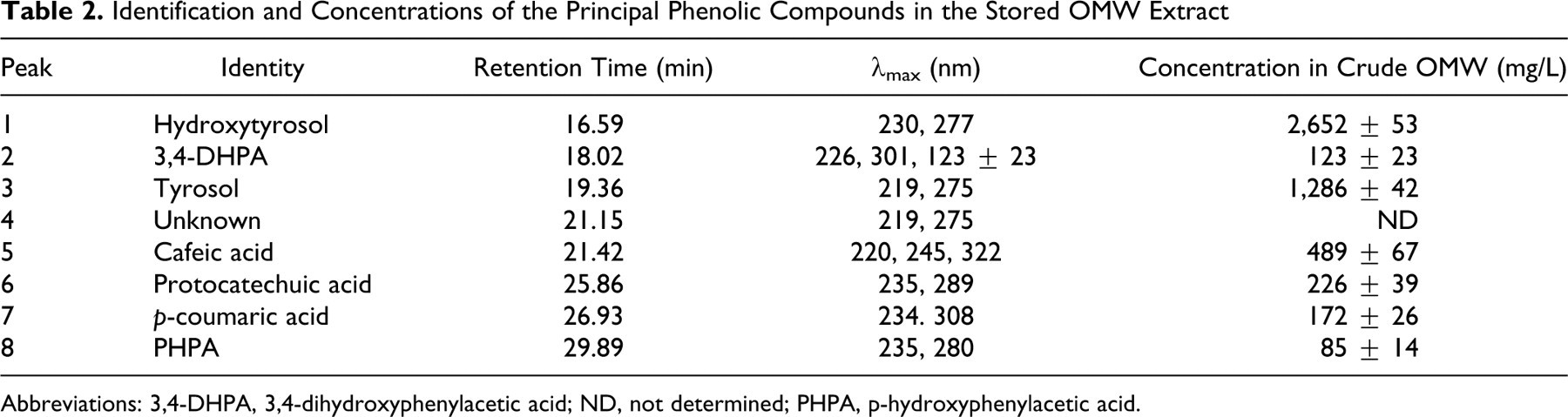
Abbreviations: 3,4-DHPA, 3,4-dihydroxyphenylacetic acid; ND, not determined; PHPA, p-hydroxyphenylacetic acid.
Abbreviated Mass Spectra of the Major Phenolic Monomers Identified in the Ethyl Acetate Extract From Stored OMW
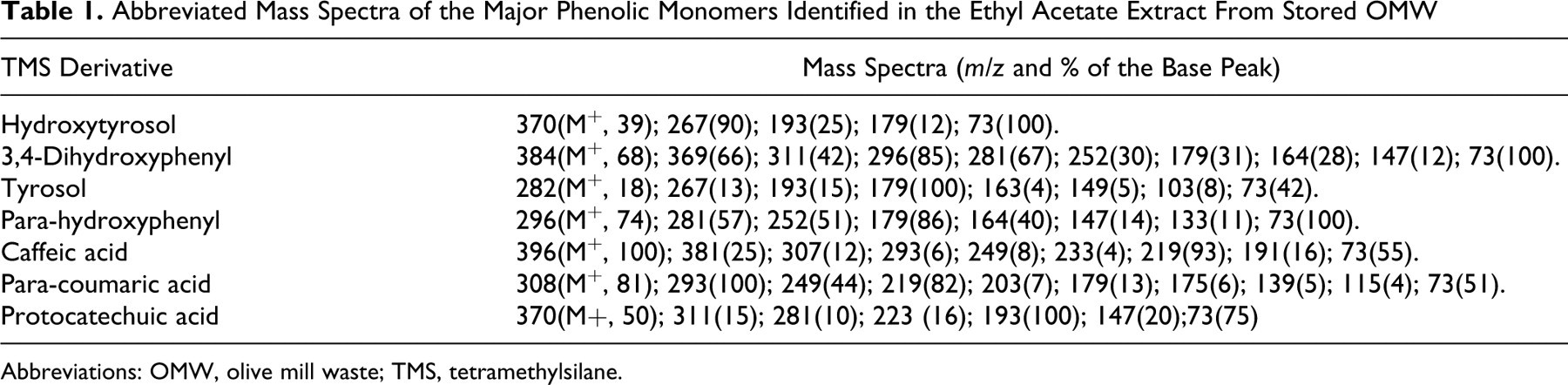
Abbreviations: OMW, olive mill waste; TMS, tetramethylsilane.
Lipid Peroxidation
The findings revealed that BSO treatment brought about a significant increase (P < .01) in the LPO levels in the bladder over the control values (Figure 3). The administration of CP also induced LPO in a significant way. Likewise, the cumulative effect of BSO + CP resulted in a significant increase in LPO as compared to group I (controls). The BSO + CP group (group IV) also underwent a significant (P < .01) increase in terms of LPO when compared to the CP group (group III) alone (Figure 3). The animals pretreated with HT and subsequently exposed to BSO (BSO + HT), on the other hand, were noted to undergo a significant (P < .01) reduction in terms of LPO levels in the bladder. When the values obtained for the CP + HT group (group V) were compared to those of the CP group (group II), HT treatment was also observed to significantly (P < .05) reduce the levels of LPO in the bladder. As illustrated in Figure 3, the comparison between the LPO values obtained for the BSO + CP group (group IV) and the BSO + CP + HT treatment group clearly show a significant decrease (P < .01) in the case of the latter (group VII).
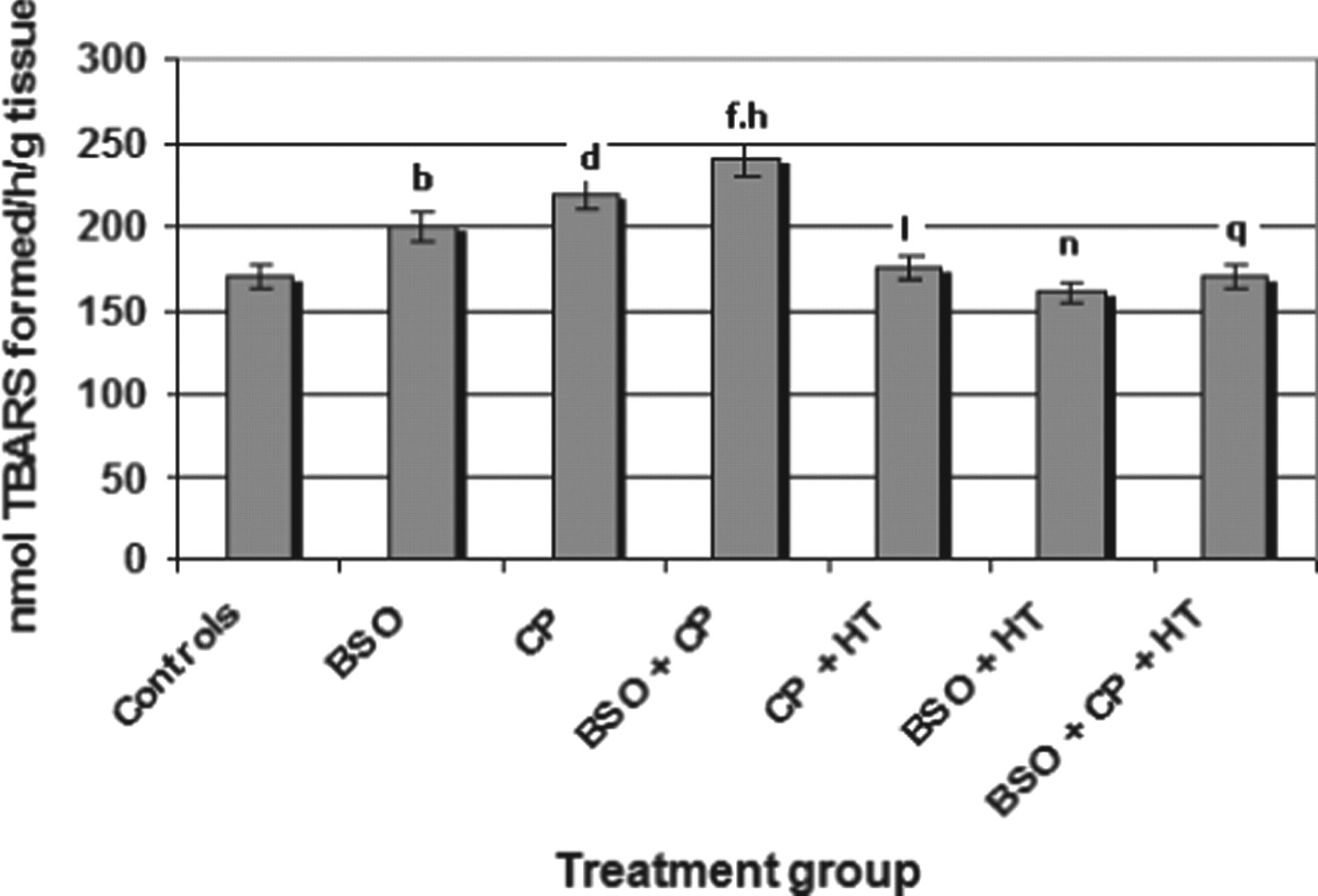
Effects of HT, BSO, and CP on the lipid peroxidation in the urinary bladder of mice. Significant differences are indicated by b P < .01 and d P < .01 in group II (BSO) and group III (CP) of treated animals, respectively; and f P < .01 in group IV (BSO + CP), when compared with control animals (group I). h P < .01 indicates significant levels for the BSO + CP group when compared to the CP group. I P < .01 and n P < .05 indicate significant difference of data related to group V (CP + HT) and group VI (BSO + HT) when compared with group III and group II, respectively. q P < .01 when data related to group IV were compared with those of group VII. Values are means ± SE (n = 6). HT indicates hydroxytyrosol; BSO, L-buthionine-SR-sulfoximine; CP, cyclophosphamide; SE, standard error.
Reduced GSH
The findings revealed that, when compared to the control values (2.20 nmol GSH/g tissue), the BSO-, CP-, and BSO + CP-treated groups underwent significant (P < .01) decreases of 0.95, 1.45, and 0.45 nmol GSH/g tissue in GSH, respectively (data generated for the cellular GSH of urinary bladder are shown in Figure 4). Likewise, when compared to the CP group (group III), the BSO + CP group (group IV) showed a significant (P < .01) decrease in terms of GSH levels. As shown in Figure 4, when the BSO and BSO + HT groups were compared, the GSH content of the latter was noted to undergo a significant (P < .01) increase. Similarly, the GSH content in the bladder of the CP + HT group (group V) showed a significant increase (P < .01) when compared to the group given only CP (group III). When the GSH values obtained for the BSO + CP group (group IV) were compared to those of the BSO + CP + HT group (group VII), a significant (P < .01) restoration in GSH was recorded (Figure 4).
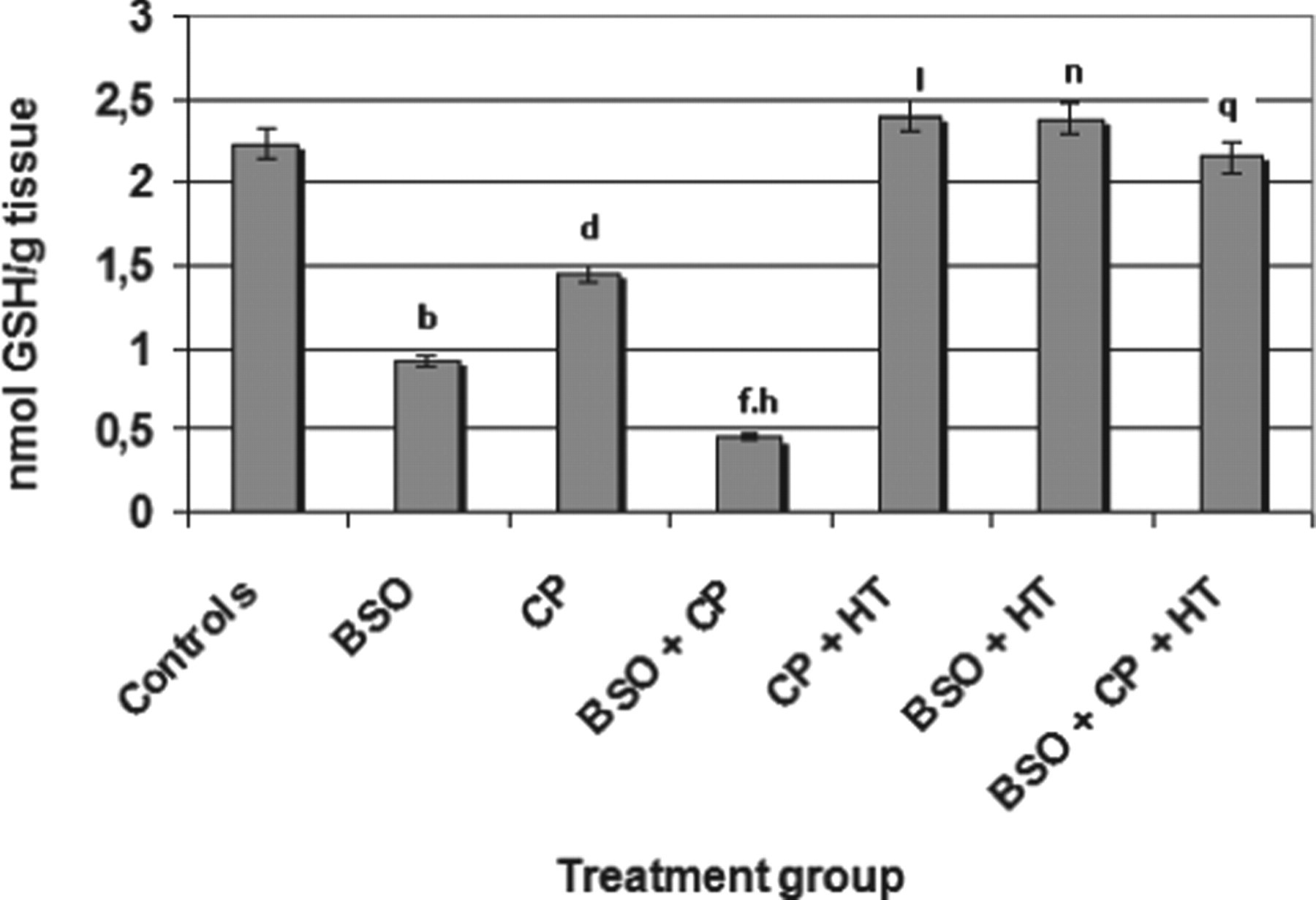
Effects of HT, BSO, and CP on GSH content in the urinary bladder of mice. Significant differences are indicated by b P < .01 and d P < .01 in group II (BSO) and group III (CP) of treated animals, respectively; and f P < .01 in group IV (BSO + CP) when compared with control animals (group I). h P < .01 indicates significant change in the BSO + CP group when compared with the CP group. l P < .01 and n P < .01 indicate significant differences in observations in group IV (CP + HT) and group VI (BSO + HT) when compared with data related to the GSH of groups III and II, respectively. q P < .01 indicates significant differences when data related to the GSH of Group IV were compared with Group VII. Values are means ± SE (n = 6). HT indicates hydroxytyrosol; BSO, l-buthionine-SR-sulfoximine; CP, cyclophosphamide; GSH, glutathione; SE, standard error.
Antioxidant Enzymes
l-Buthionine-SR-sulfoximine and CP treatments were observed to induce significant (P < .01) decreases in terms of GST, GR, GP, and CAT activities in the bladder when compared to the control group (Table 3). The BSO + CP group also showed an additive significant (P < .01) decrease in the activities of GST, GR, and GP when compared to the CP and the control groups. However, no significant difference was observed in terms of CAT activity between the animals of group III (CP) and group IV (BSO + CP). The activity of those antioxidant enzymes increased significantly (P < .01) in both the CP + HT (group V) and BSO + HT (group VI) groups when compared to their respective controls, the BSO (group II) and CP (group III) groups (Table 3). The animals treated with HT and subsequently exposed to BSO + CP (group VII, BSO + CP + HT treatment) showed a significant increase (P < .01) in the activities of all the antioxidant enzymes when compared to the BSO + CP group (group IV).
Activities of Antioxidant Enzymes in the Urinary Bladder of Mice in Different Treatment Groupsa
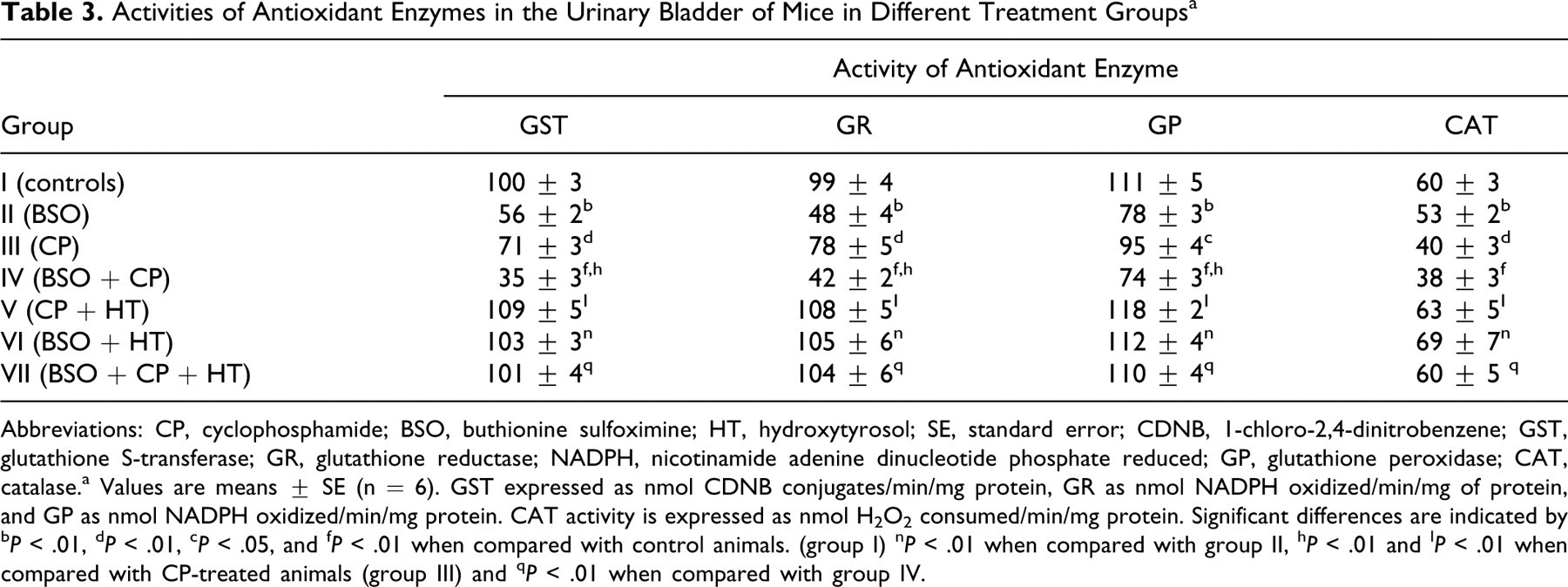
Abbreviations: CP, cyclophosphamide; BSO, buthionine sulfoximine; HT, hydroxytyrosol; SE, standard error; CDNB, 1-chloro-2,4-dinitrobenzene; GST, glutathione S-transferase; GR, glutathione reductase; NADPH, nicotinamide adenine dinucleotide phosphate reduced; GP, glutathione peroxidase; CAT, catalase.a Values are means ± SE (n = 6). GST expressed as nmol CDNB conjugates/min/mg protein, GR as nmol NADPH oxidized/min/mg of protein, and GP as nmol NADPH oxidized/min/mg protein. CAT activity is expressed as nmol H2O2 consumed/min/mg protein. Significant differences are indicated by b P < .01, d P < .01, c P < .05, and f P < .01 when compared with control animals. (group I) n P < .01 when compared with group II, h P < .01 and I P < .01 when compared with CP-treated animals (group III) and q P < .01 when compared with group IV.
Discussion
The present study was undertaken to explore the potential effects that HT might offer to alleviate or prevent the serious concerns widely voiced about the undesirable side effects associated with CP and BSO treatment.
Identification and comparisons of the phenolic compounds of OMW using HPLC-UV showed several simple peaks corresponding to different phenolic monomers, among which 7 compounds were identified: HT, 3,4-dihydroxyphenyl acetic acid, tyrosol, cafeic acid, protocatechuic acid, p-coumaric acid, and p-hydroxyphenyl acetic acid. Minor unidentified components were also present in the ethyl acetate extract. The identification of these phenolic compounds was confirmed by GC-MS analysis (Table 1) where the GC-MS spectra of the isolated compounds were compared to those reported in the GC-MS apparatus library. The results obtained for the mass fragments were in agreement with those previously described. 24
The biophenols identified in the stored OMW extract were quantified using standard curves in the concentration range of 50 to 2000 ppm, in accordance with the method reported by Akasbi et al. 30 The concentrations that were calculated are reported in Table 2. As expected, HT was the most abundant phenolic monomer in the OMW sample under investigation (2.652 mg/L in stored crude OMW). In fact, the HT concentration in the fresh OMW (1.453 mg/L) was lower than that in the stored OMW (2.652 mg/L). This increase can be attributed to a number of hydrolysis reactions of oligomeric polyphenols (HT derivatives) which contain HT units attached to other compounds via ester and/or glucosidic linkages.
In order to evaluate the inhibitory action of HT on bladder toxicity in the animals, this sample was first purified from the OMW phenolic extract using silica gel column chromatography. The purity of this biophenol was then evaluated using HPLC-UV. The chemical structure and purity of the isolated HT was confirmed by 1 H NMR spectroscopy. The spectroscopic data obtained (Figure 2) are in full agreement with those previously described. 31
Hydroxytyrosol has been reported to have anticancer effects on human colon adenocarcinoma HT-29 cells and human promyelocytic leukemia HL-60 cells. 23 In this context, several studies have suggested that oxidative stress could play an important role in CP- and BSO-induced toxicity. Since the bladder is the site where urine is stored, the concentration of CP toxic metabolites is higher in the bladder than in other organs, which is likely to increase the toxic load of CP in the bladder. 32 As far as the present study is concerned, the findings indicated that CP caused a significant reduction in the levels of all the antioxidants in urinary bladder. It also increased the LPO in the bladder. The oxidative products of the CP responsible for the induction of LPO and the generation of ROS resulted in inflammation, thus disturbing the overall redox cycling of the bladder. 33 It is worth noting in this context that BSO, a known depletor of GSH, inhibits c-glutamylcysteine synthetase (c-GCS). 34
When the data pertaining to GSH reduction were compared, BSO was found to exert a more significant effect than CP. In fact, a difference of 42% was observed between the 2 items involved, confirming that BSO is a more potent depletor of GSH than CP, a result which is in good agreement with the findings previously reported. 35 The CP-induced depletion of GSH is primarily mediated by the interaction of its reactive metabolite, acrolein, with GSH. Acrolein interacts not only with GSH but also with cysteine, which is one of the constituent amino acids of GSH. It is for this reason that a number of sulfhydryl (–SH) compounds, and cysteine itself, have been observed to protect the animals from toxic effects of CP. 35 ,36
Furthermore, the ip administration of CP was observed to induce LPO in bladder in a significant way. In fact, CP-induced LPO has been described in different tissues of exposed animals. 11 ,13 The role of acrolein in the CP induction of LPO also has been implied. 37 It has been suggested that by binding to nucleophilic amino acids, acrolein could directly affect transcription and modulate this process through its ability to deplete GSH. 36
Moreover, BSO treatment was noted to result in the depletion of GSH and the increase of LPO in urinary bladder. The depletion of GSH was, in fact, previously reported to increase the susceptibility of cells to apoptosis. 38 When BSO and CP were administered together, an additive effect was observed in the cases of GSH, LPO, and other parameters. The purpose of using BSO together with CP was to study the likely scenario where a host is exposed to a combination of GSH-depleting agents, including pathogens, and to assess whether an herbal extract treatment of HT can have any modulatory impacts on their commutative/additive effects. Not only did HT pretreatment show protective effects on CP urotoxicity but it was also effective in protecting the animals treated with the CP + BSO combination (group VII). The HT pretreatment was observed to restore the depleted GSH and other antioxidants and to simultaneously reduce LPO in the bladder. Cyclophosphamide-induced immunosuppression is likely to increase the occurrence of infections, which may deplete GSH since several infectious agents have been reported to do so. 17 Immunomodulatory herbal extracts, such as the HT that has valuable GSH-restoring effects presented in the current work, hold great promise in reducing the adverse effects of CP in patients with cancer.
The results of the present study, in agreement with previously reported findings, 35 ,36 demonstrate that treatment with CP and BSO significantly enhances ROS formation in the bladder as evidenced by the increased LPO and decreased GSH. Evidence for the involvement of oxidative stress, resulting in increased LPO from the onset of several diseases is accumulating. Epidemiological studies indicate negative correlations between the dietary intake of antioxidants and the incidence of such diseases. Furthermore, the production of ROS, following CP administration, also occurs in other tissues, as demonstrated by directly measuring ROS formation in the aorta and renal artery. 39 Among the several antioxidants available, the naturally occurring phenol HT was selected and investigated in terms of potential effects for reducing CP toxicity in vivo.
The findings of this work revealed that HT completely counteracts CP-induced oxidative stress, which is evidenced by the increased GSH and the decreased LPO levels in urinary bladder. These results are in agreement with other previous reports indicating that HT is able to completely prevent CP-induced ROS generation in rats. 21
As recently reviewed by Manna et al, 40 the biological activities of HT have been extensively investigated. The application of HT in vitro was found to prevent low density lipoprotein (LDL) oxidation as well as platelet aggregation and to inhibit 5- and 12-lipoxygenases. 41 –43 It was also reported to exert an inhibitory effect on peroxynitrite-dependent DNA base modifications and tyrosine nitration. 21 Several studies have reported that a diet rich in the materials tested had no effect on the levels of DNA damage in the human bladder, while others found that such links exist between these materials and genotoxicity. The efficacy of these vegetable products probably depends on human polymorphisms in DNA repair genes, which may be associated with differences in the repair efficiency of DNA damage. 44,45
The pre-incubation of intestinal Caco-2 cells with HT was also described to prevent damage caused by oxidative stress, such as LPO and alterations of cell permeability and viability. 46 Moreover, HT was found to exert a protective effect against the H2O2-induced oxidative hemolysis and malondialdehyde formation in red blood cells. 40 Hydroxytyrosol was also shown to have an antiproliferative effect, inducing apoptosis in HL-60 cells and in resting and activated peripheral blood lymphocytes. 22
In short, the present study investigated, for the first time, the potential antitoxic activities of HT focusing on the reduced LPO effect and increased antioxidant activities. The findings demonstrate that HT is endowed with promising properties and attributes that make it a potential attractive candidate for application as an antitoxic and therapeutic agent in the medical industry. For this reason further studies, some of which are currently under way, are needed to determine other pharmacological activities pertaining to this natural compound.
Footnotes
Acknowledgments
This research was supported by the Tunisian Ministry of Higher Education and Scientific Research and Technology and the Tunisian Ministry of public health. The authors wish to express their sincere gratitude to Prof Anouar Smaoui from the English Unit at the Sfax Faculty of Science for accepting to proofread and polish the language of the current paper.
The authors Kamel Rouissi and Bechr Hamrita have contributed equally in this work.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
