Abstract
L-(+)-ergothioneine has antioxidant and anti-inflammatory properties in vitro and in vivo and has uses as a dietary supplement and as an ingredient in foods, cosmetics, and as a pharmaceutical additive. The clastogenic potential and mutagenic of ergothioneine were assessed in vitro and in vivo. Ergothioneine concentrations up to 5000 μg/mL, with and without metabolic activation, was tested in the chromosome aberration assay with CHL cells and found not to induce structural chromosome aberrations. In the in vivo mammalian erythrocyte micronucleus test, ergothioneine was administered orally to male mice at doses up to 1500 mg/kg for potential genotoxic activity. No increase in the frequency of micronucleated polychromatic erythrocytes was observed. Overall, ergothioneine was not genotoxic in these studies and provides additional experimental evidence supporting the safety of its use as a potential dietary supplement.
Keywords
Introduction
L-(+)-Ergothioneine (Figure 1) is a hydrophilic sulfur-containing amino acid thiourea derivative of betaine of histidine antioxidant that can be derived from the diet that is highly sequestered in mammalian tissues. 1 –4
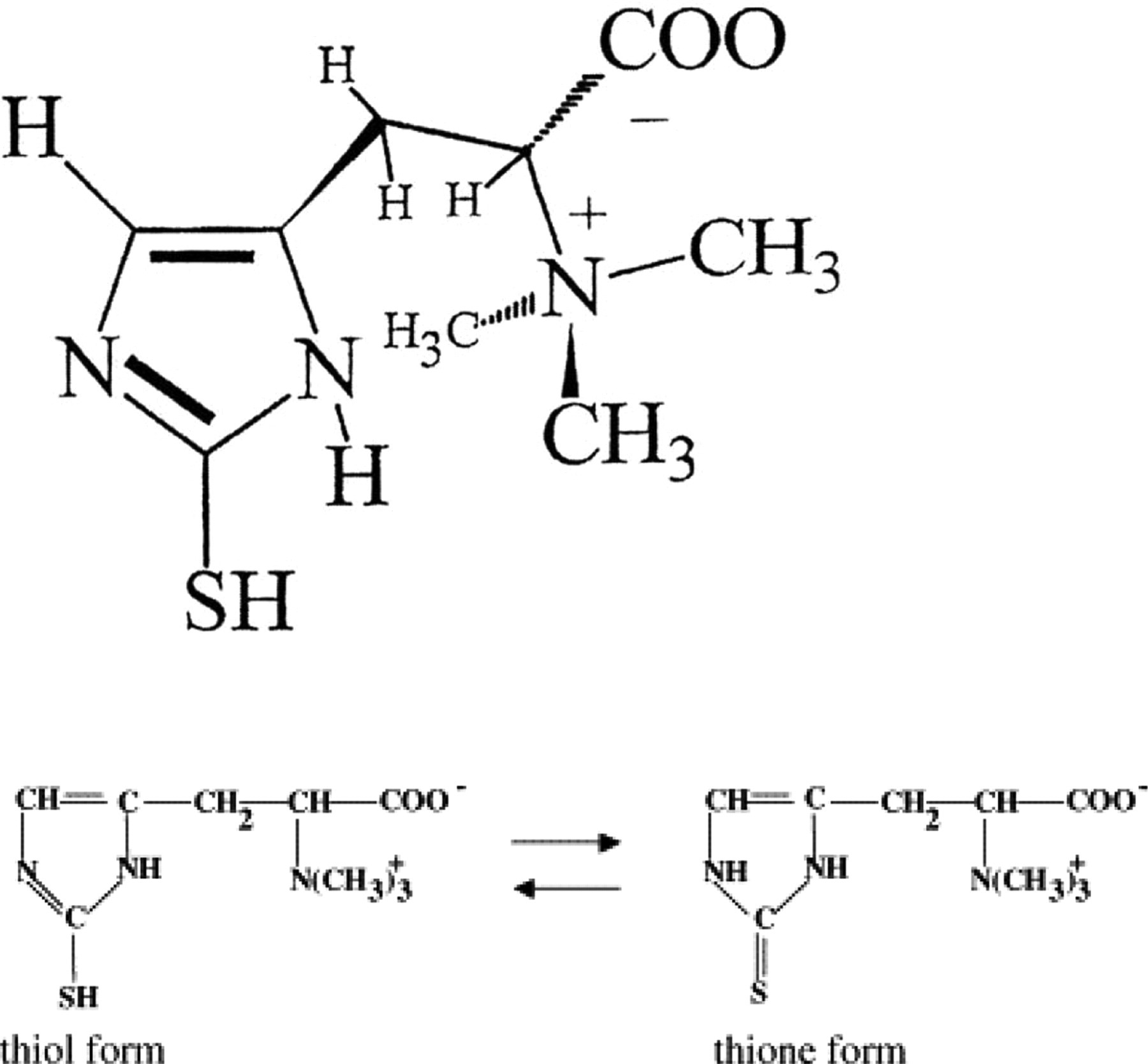
Chemical structure of L-(+)-ergothioneine
The highest levels of ergothioneine per kilogram of a particular food are found in 2 commonly consumed mushroom species: Boletus edulis (528 mg/kg) and the oyster mushroom (Plerotus ostreatus; 119 mg/kg). Other foods with appreciable amounts per kilogram include black beans (13.9 mg/kg), kidney beans (4.52 mg/kg), oat bran (4.41 mg/kg), and garlic (3.11 mg/kg), 5 as well as, Lentinula edodes and Grifola frondosa. 6 Park and colleagues have reported the accumulation of ergothioneine in the rhizomes of medicinal plant Gastrodia elata and suggested that this may account for the beneficial health benefits of this achlorophyllous orchard plant in human diseases. 7
Although ergothioneine has been shown to be as potent as glutathione in combating the cytotoxic effects of pyrogallol, a superoxide-generating trioxybenzene, 8 the mechanism with which both exert their antioxidant effects differs. Deiana and colleagues have shown that glutathione can be conserved by ergothioneine in conditions of oxidative stress in vivo. 4 Sang and colleagues reported that ergothioneine protects against neuronal injury by the chemotherapeutic drug cisplatin. 9 The neuroprotective potentials of ergothioneine have been substantiated. 10,11
Ergothioneine has been previously reported to be nonmutagenic in a bacterial reverse mutation assay. 12 The present study extends previous findings to include a mammalian chromosomal aberration assay and an in vivo mammalian erythrocyte micronucleus test.
Materials and Methods
Chemicals
L-(+)-Ergothioneine was obtained from Oxis International (Beverly Hills, California) and determined to be 99% pure by Cambridge Major Laboratories (Germantown, Wisconsin) using high-performance liquid chromatography (HPLC), chiral-HPLC, infrared spectroscopy, and Liquid chromatography/mass spectrometry (LC/MS).
Chromosomal Aberration Assay
All chemicals were provided by Sigma-Aldrich Chemie (Budapest, Hungary), unless otherwise indicated.
The solution used in the chromosomal aberration assay was prepared in Dulbecco Modified Eagle (DME) medium prior to the treatment of cells. The positive control, without S9, was ethylmethane sulphonate (EMS). Ethylmethane sulphonate was dissolved in DME medium and used as a positive control for the nonactivation phase of the study at a concentration of 0.4 and 1.0 µL/mL. N-nitrosodimethylamine was used as the positive control for the activation study (1.0 µL/mL).
The V79 cell line for culture was purchased from the European Collection of Cells Cultures. During the 3- and 20-hour treatments with ergothioneine, negative solvent, and positive controls, the serum content was reduced to 5%. The cells were incubated in the presence or absence of S9 obtained from Trinova Biochem (Giessen, Germany).
Mammalian Erythrocyte Micronucleus Test
Ergothioneine was dissolved in Aqua purificata (Hungaropharma, Budapest, Hungary) at concentrations of 37.5, 75, and 150 mg/mL. The positive control, cyclophosphamide, was dissolved in 0.9% NaCl solution (Salso, Budapest, Hungary).
Animals
Healthy (SPF) CRL:NMRI BR mice were used in the study and randomized according to body weight. Two males and 2 females were used in the dose-ranging study (2 animals/sex per group), and 42 males were used in the main study: 5 male animals per dose group were used for sampling on each occasion; 5 males in the low- and the middle-dose group; 10 males in the highest and control groups. Animals were kept in polypropylene/polycarbonate cages and maintained in a 12-hour light at 22°C ± 3°C, and 30% to 70% relative humidity. Rodents received water and a pellet diet of SSNIFFSM R/M-Z+H (Experimental Animal Diets, Soest, Germany) ad libitum.
Experimental Procedures
Chromosomal aberration assay
The chromosomal aberration assay was performed in compliance with Organisation for Economic Co-operation and Development (OECD) 473, 13 US Environmental Protection Agency (EPA) 870.5373, 14 and European Commission 15 guidelines. The studies were conducted according to Good Laboratory Practices certified by the Hungarian Ministry of Health and Ministry of Agriculture and Regional Development. The assay was conducted as 2 independent experiments in the presence and in the absence of the S9 mix. The test item concentrations 1250, 2500, 3750, and 5000 μg/mL of the main test were selected on the basis of cytotoxicity investigations in a preliminary study with and without metabolic activation using S9, in experiment A consisted of 3-/20-hour treatment/sampling time and experiment B consisted of 20- and 3-/28-hour treatment/sampling time (both run in duplicate). Parameters evaluated for statistical analysis were the number of cells with aberrations, with and without gaps.
Mammalian erythrocyte micronucleus test
The in vivo mammalian erythrocyte micronucleus test followed OECD 474, 16 Guidance S2A, 17 and European Commission 440/2008 18 guidelines and was approved by the Animal Care and Use Committee of TOXI-COOP (Budapest, Hungary).
A preliminary toxicity test was performed to identify the appropriate maximum dose level for the main test. Groups of 2 male and female mice were treated orally once at dose levels of 2000 mg/kg (20 mL/kg). Animals were examined for adverse reactions and mortality. On the basis of the results of this test, the male and female doses for the mouse micronucleus test were 375, 750, and 1500 mg/kg (n = 5/dose). Two additional male mice were also dosed in the highest treated group to replace any animals that might die. Clinical signs were recorded.
The test/negative items were orally administered once. In the untreated control, low- and mid-dose groups, the bone marrow was collected at 24 hours after treatment and at 24 and 48 hours after treatment in the high-dose and negative (solvent) control groups. Cyclophosphamide (positive control; 60 mg/kg) was administered intraperitoneally (10 mL/kg) and mice (n = 5) were sacrificed 24 hours after treatment. Bone marrow was obtained from 2 exposed femurs of the mice from every time point immediately after sacrificing (by cervical dislocation). The bone marrow was flushed with fetal bovine serum (5 mL) and followed by vortex mixing; the cell suspension was concentrated by centrifugation and the supernatant was discarded. Smears of the cell pellet were fixed for 5 minutes in methanol, allowed to air-dry, stained with Giemsa (10%) solution, and coated with EZ mounting (Thermo Fisher Scientific, Geel, Belgium). A total of 2000 polychromatic erythrocytes (PCEs) were scored per animal by a blinded reader. The frequency of micronucleated cells was expressed as a percentage of micronucleated cells based on the first 2000 PCEs counted in the optic field. The proportion of immature cells among the total (immature + mature) erythrocytes was determined for each animal by counting a total of at least 200 immature erythrocytes. To identify micronucleated erythrocytes, the standard criteria were applied.
The frequencies of micronucleated PCEs (MPCEs) in animals in the test and positive control groups were compared to the values found in the corresponding negative control group using Kruskal-Wallis nonparametric analysis of variance (ANOVA) test (SPSS, PC+, Budapest, Hungary). Ergothioneine was considered to show genotoxic activity if there was a statistically significant increase in the frequency of micronucleated PCEs in treated animals compared to corresponding controls, the increases were dose related and the increases exceeded the historical control range for the laboratory. Otherwise, the test item is considered to have given a negative response as no reproducible, statistically significant increases were observed.
Results
Chromosomal Aberration Assay
A dose selection cytotoxicity assay was performed to establish the concentration range for the chromosome aberration assays, both in the absence and in the presence of rodent S9. Toxicity was determined by cell counting and results noted as cell survival in the treatment group (in %) in relation to the negative (solvent) control. The cytotoxicity of ergothionene at the dose ranges used in this study was not found to be cytotoxic. No polyploidy was observed. When ergothioneine was tested for cytotoxic concentrations of up to 5000 µg/mL, both with and without metabolic activation, it did not induce structural chromosome aberrations in Chinese hamster lung cells and therefore is not considered clastogenic in this system (see Tables 1 and 2).
a Experiment A with 3-/20-hour treatment/sampling time.
b Positive control (−S9) EMS: ethylmethane sulphonate (1.0 μL/mL). Positive control (+S9) NNDA: N-Nitrosodimethylamine (1.0 μL/mL). Negative control: DME medium.
c P < .01.
a Experiment B with 20- and 3-hour/28-hour treatment/sampling time.
b Positive control (−S9) EMS: ethylmethane sulphonate (0.4 μL/mL). Positive control (+S9) NNDA: N-Nitrosodimethylamine (1.0 μL/mL). Negative control: DME medium.
c P < .01.
Mammalian Erythrocyte Micronucleus Test
In the preliminary toxicity test, no mortality occurred among the 2 males or 3 females treated with 2000 mg/kg body weight (bw) of ergothioneine. Males exhibited a moderate decrease in activity, squatting position, and piloerection at 24 hours after treatment, which decreased in incidence at 48 hours. The female mice dosed 2000 mg/kg showed a slight decrease in activity, hunched position, and piloerection at 24 hours after treatment. No clinical signs of toxicity were observed in female mice between 24 and 48 hours after treatment. In the main study, 4 of 12 animals dosed at 1500 mg/kg bw showed a decrease in activity, hunched position, and piloerection, between 5 and 24 hours after the treatment. All animals treated at the 375 and 750 mg/kg bw dose levels did not exhibit any clinical signs of toxicity.
Table 3 shows the number of PCEs analyzed and the mean number of MPCEs and PCE/normochromatic erythrocytes (NCEs) in each male group.
Summary of Mouse Micronucleus Test at 24 and 48 hours a
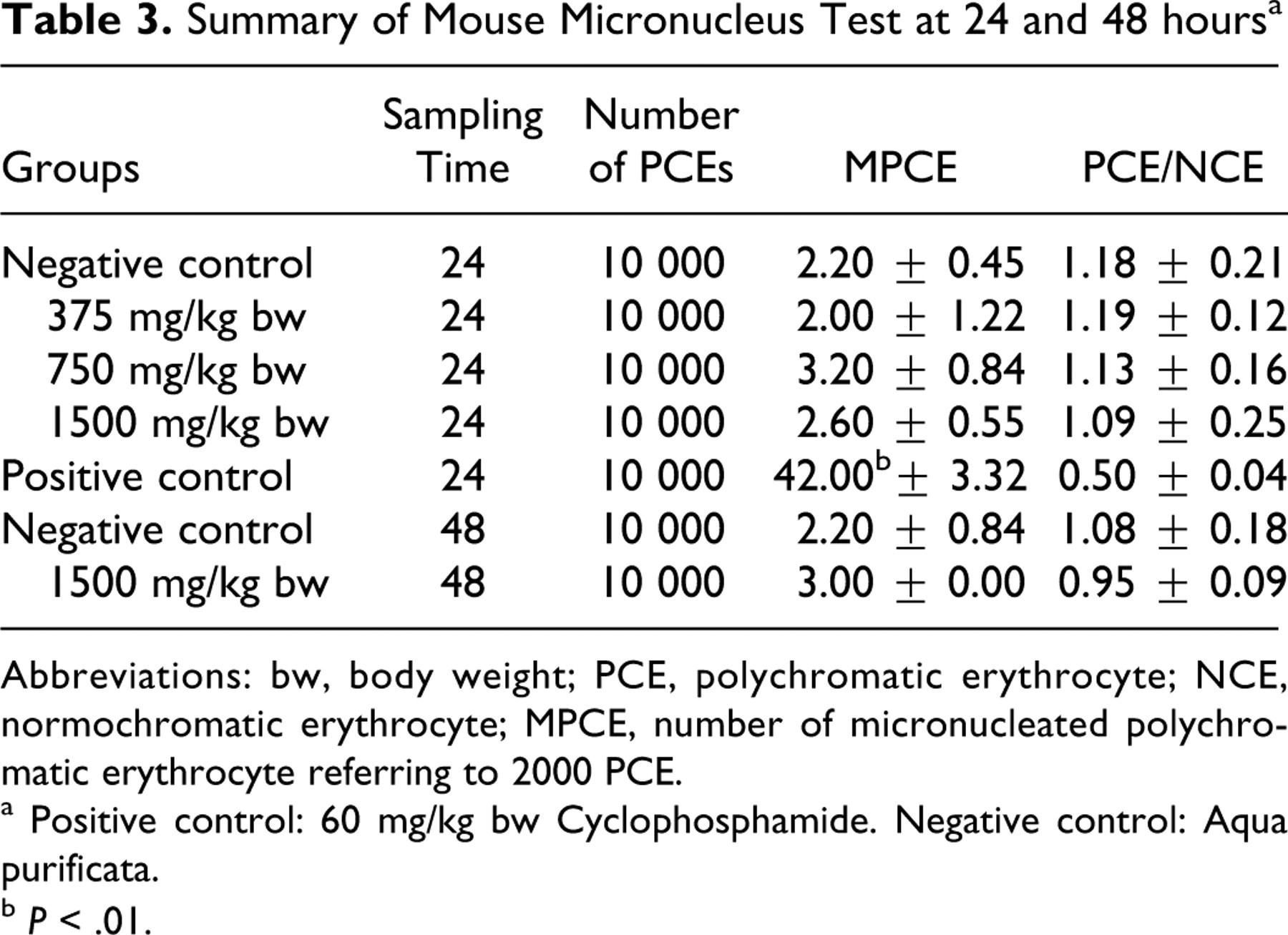
Abbreviations: bw, body weight; PCE, polychromatic erythrocyte; NCE, normochromatic erythrocyte; MPCE, number of micronucleated polychromatic erythrocyte referring to 2000 PCE.
a Positive control: 60 mg/kg bw Cyclophosphamide. Negative control: Aqua purificata.
b P < .01.
Discussion
Ergothioneine accumulates in cells and the plasma membrane when subjected to high levels of oxidative stress. Its protective function is restricted to cells that express the ergothionene-specific receptor/transporter, organic cation/carnitine transporter 1 (OCTN1). 18 Functionally expressed in nonparenchymal liver cells, the transporter serves to enhance small intestinal absorption of ergothioneine, as has been demonstrated in the OCTN1 gene knockout mouse. 19 The transporter is concentrated in mitochondria, where it plays a role in protecting mitochondrial components such as DNA from oxidative damage due to mitochondrial superoxide production. 8 Cells that do not express OCTN1 are susceptible to oxidative stress, resulting in increased mitochondrial DNA damage, lipid peroxidation, and protein oxidation. 20 Despite the evidence that ergothioneine may play a protective role as an anti-genotoxic agent, there is a dearth of genotoxicity data.
The very low frequencies of micronucleated PCEs were within acceptable ranges for the negative and positive control mice in the mammalian erythrocyte micronucleus test. With oral administration of ergothioneine at doses of 375, 750, and 1500 mg/kg, no biologically or statistically significant increases in the frequency of MPCEs was observed at either 24 or 48 hours compared to the concurrent control group.
The lack of clastogenic potential and mutagenic activity by ergothioneine observed in these studies adds to the results reported for the bacterial reverse mutation assay using Salmonella typhimurium strains TA98, TA100, TA1535, and TA1537, and Escherichia coli WP2 uvrA, which showed no increases in the number of revertant colony observed in any of the test strains at any concentration level. 12
Footnotes
Acknowledgments
The authors wish to thank Oxis International for their financial support to perform the studies.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.


