Abstract
Bisphenol A is an endocrine disrupting compound widely used in the production of polycarbonate plastics and epoxy resins. It is ubiquitously present in the environment, mostly in aquatic environments, with consequent risks to the health of aquatic organisms and humans. In the present study, we analysed the cytogenetic effects of bisphenol A on human lymphocytes using in vitro chromosomal aberrations and micronuclei assays. Lymphocyte cultures were exposed to five different concentrations of BP-A (0.20, 0.10, 0.05, 0.02 and 0.01 μg/mL) for 24 h (for chromosomal aberrations test) and 48 h (for micronuclei test). The concentration of 0.05 µg/mL represents the reference dose established by United States Environmental Protection Agency (US EPA); 0.02 μg/mL represents the higher concentration of unconjugated BP-A found in human serum and 0.01 μg/mL represents the tolerable daily intake established by European Union. Data obtained from both assays showed significant genotoxic effects of the bisphenol A at concentrations of 0.20, 0.10 and 0.05 μg/mL, whereas at the concentration of 0.02 μg/mL, we observed only a significant increase in the micronuclei frequency. Finally, at the concentration of 0.01 μg/mL, no cytogenetic effects were observed, indicating this latter as a more tolerable concentration for human health with respect to 0.05 μg/mL, the reference dose established by US EPA.
Introduction
In the last decades, global production of plastic has continued to rise. Recovery and recycling, however, remain insufficient and millions of tons of plastic compounds continue to accumulate in the environment. Most of these compounds have adverse effects on the biome and pose huge risks for the human health. Some of them are reported to be endocrine disrupting compounds (EDCs), mimicking or blocking natural hormone action and altering the normal functioning of the endocrine system of invertebrate and mammals species. 1
Among EDCs, bisphenol A (BP-A: 4,4′-(propane-2,2-diyl)diphenol) has received much attention due to its high production volume and widespread human exposure. 2 BP-A is an important synthetic compound used in the production of polycarbonate plastics and as a precursor of epoxy resins. A wide range of consumer products contain BP-A, including water and baby bottles, food packaging materials, internal coatings of cans and drums, toys, water pipes, sports equipment, medical and dental devices and household electronics. 3
Due to the daily use, BP-A was found in all environmental matrices such as air, water, soil, sediment and sewage sludge. It is released into the environment either through sewage treatment effluent, landfill leachate, untreated urban and industrial waste water or natural degradation of polycarbonate plastics. 4 For these reasons, BP-A was found at relatively high concentrations (up to hundreds of micrograms per litre) in several rivers and lakes worldwide. 5
Being BP-A used for food packaging, bottles and coatings for tins, human exposure to this compound is extremely variable depending on lifestyles, principally dietary consumption habits.
In general, humans are chronically exposed to low doses of the compound, mainly through food and drinking water. 6 However, the release of BP-A from polycarbonates is accelerated by UV light, ageing, heating or following pH changes due to the contact with acidic or basic compounds. 7 It has been shown that upon long-term heating, BP-A can leach from plastic baby bottles into an aqueous solution. 8 For these reasons, first in Canada, Denmark and next in all European countries, BP-A has been banned in infant formula bottles. 9
Nevertheless, many plastic food and drinking containers still contain BP-A and its presence has been reported in human serum and in 95% of the urine samples obtained from a reference population in the United States, 10 as well as in maternal and foetal plasma, 11 placental tissue 12 and in the milk of lactating mothers. 13 Several studies, using different analytical techniques, have also measured the unconjugated BP-A concentrations in human serum, reporting values ranging from 0.2 to 20 ng/mL. 2 Finally, the estimated exposure of the general population to BP-A is supposed to reach a body burden of up to 9 μg/kg/day. 14
In humans, BP-A is rapidly absorbed from the gastrointestinal tract and conjugated with glucuronic acid in the liver. The formed glucuronide is then cleared from blood by elimination with urine within 24 h after oral administration. This rapid excretion results in a low body burden of the BP-A in humans following oral absorption of low doses. 15 However, despite its rapid rate of excretion, as consequence of its ubiquitous and widespread distribution, BP-A has arisen worldwide concerns about its possible associations with human diseases such as obesity 6 and cancer. 16 At reproductive system level, exposure to ECDs seems to be associated with decreased fertility and increased risk of testicular or prostate cancer in men, 17 whereas in women exposure to ECDs seems to increase the risk of endometriosis, reproductive or other endocrine-related cancers. 18
From the genetic point of view, the genotoxicity of BP-A has been evaluated in several in vitro and in vivo studies, although with controversial results. BP-A was found to be able to induce increased chromosome aberrations (CAs) and DNA adducts in Syrian hamster embryo cells, 19 micronuclei (MNs) in human MCL-5 cells 20 and in fish erythrocytes, 21 MNs and CAs formation in chines hamster ovary (CHO) cells. 22 A significant genotoxic effect by BP-A was also seen on bovine peripheral lymphocytes in vitro, but only at the highest concentration of 1 × 10−4 M. 23 Moreover, Tiwari et al. 24 observed a significant increase of MNs in polychromatic erythrocytes, structural CAs in bone marrow cells and DNA damage in lymphocytes of rats exposed to BP-A. Interestingly, in HEK293 cells, BP-A was found to alter the expression of some genes involved in important biological processes including ion transport, cysteine and glycogen metabolic processes, apoptosis, DNA damage repair and so on. 25
On the other hand, BP-A failed to induce gene mutations in Syrian hamster cells, 19 polyploidy or MNs in male mice 26 and sister chromatid exchanges in mouse lymphoma cells. 27
Nevertheless, based on published data, the United States Environmental Protection Agency (US EPA) has set a reference dose (RD) for BP-A oral exposure of 0.05 mg/kg body weight (BW)/day. 28 More recently, the European Union (EU) 14 established a temporary tolerable daily intake (TDI) for BP-A of 10 μg/kg BW/day, based on a consideration of much toxicological data, including the effects of BP-A on reproductive organs and functions and hormonal effects in animals.
No data are reported in literature about in vitro cytogenetic effects of BP-A on human lymphocytes.
For this reason, in the present study, we assessed the possible in vitro clastogenic and aneugenic effects of BP-A exposure on human lymphocyte cells by CAs and MNs assays.
It has been shown that a high frequency of the chromosomal damage is a predictive value of health risk. 29 In this scenario, CAs and MNs assays are important tools in the measurement of the genotoxic potential of many chemicals, including BP-A. The CAs assay allows the detection of cells carrying unstable aberrations (chromosome and chromatid breaks, deletions, fragments, rings, dicentrics and chromatid exchanges) that will lead to cell death during proliferation. 30 Previous studies have provided strong evidence in support of the hypothesis that high CAs frequency in peripheral blood lymphocytes is a powerful predictor of cancer risk. Indeed, it is known that cancer incidence among healthy individuals of a population increases with increased levels of CAs in their circulating lymphocytes. 29 On the other hand, MNs are used as a fast and reliable assay for detecting both potential clastogenic (chromosome breakage) or aneugenic (chromosome lagging due to dysfunction of mitotic apparatus) effects of different xenobiotics. In particular, MNs represent acentric chromosomal fragments or whole chromosomes left behind during mitotic cell division and appear in the cytoplasm of interphase cells as small additional nuclei. Bonassi et al. 31 provided evidences that also increased MNs frequencies in peripheral blood lymphocytes are positively associated with early events in carcinogenesis.
In the present study, we tested concentrations of 0.20, 0.10, 0.05, 0.02 and 0.01 μg/mL, where 0.05 µg/mL represents the RD established by US EPA; 28 0.20 and 0.10 μg/mL are multiples of this RD in order to establish a possible dose-dependent increase in the genomic damage; 0.020 μg/mL represents the higher concentration of unconjugated BP-A found in literature in human serum 2 and 0.01 μg/mL is the TDI established by EU. 14
Methods
Chemicals and media
The IUPAC name of bisphenol A (BP-A, CAS n. 80-05-7) is 4,4′-(propane-2,2-diyl)diphenol. The BP-A was dissolved in dimethyl sulfoxide (DMSO, CAS no. 67-68-5). Gibco RPMI 1640 cell culture media supplemented with
Subjects
Peripheral venous blood was collected from five healthy female subjects (mean age ± SD, 27.60 ± 5.22, range 22–30 years), non-smoking, non-alcoholic, not under drug therapy, and with no recent history of exposure to mutagens. Informed consent was obtained from all blood donors. The study was approved by the University of Turin ethics committee and was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki.
Blood sample collection and lymphocyte cultures
Heparinized blood samples were obtained by venipuncture and collected in heparinized tubes for genotoxicity testing. All blood samples were coded, cooled (4°C) and processed within 2 h after collection. Heparinized venous blood (0.3 mL) was cultured in 25 cm2 flasks in 6 mL of RPMI-1640 medium supplemented with 20% FCS, 2% of the mitogenic agent PHA,
Three control cultures were assessed: (1) positive control, by adding only MMC (final concentration 0.1 µg/mL culture); (2) solvent control, by adding only 0.1% of DMSO; (3) negative control, culture without both BP-A and DMSO. Only for MNs assay, after 44 h of incubation, cytochalasin-B was added to the cultures at a concentration of 6 µg/mL to block cytokinesis.
After 48 h (for CAs assay) and 72 h (for MNs assay) of incubation at 37°, the cells were collected by centrifugation and treated for 10 min with a pre-warmed mild hypotonic solution (75 mM KCl). After centrifugation and removal of the supernatant, the cells were fixed with a fresh mixture of methanol/acetic acid (3:1 v/v). The treatment with the fixative was repeated three times. Finally, the supernatant was discarded and the pellet, dissolved in a minimal volume of fixative, was seeded on the slides to detect CAs and MNs by conventional staining with 5% Giemsa (pH 6.8) prepared in Sörensen buffer.
Cytokinesis-block micronucleus assay
Microscope analysis was performed at 40× magnification on a light microscope (Dialux 20, Leica, Germany). MNs were scored in 1000 binucleated lymphocytes with well-preserved cytoplasm per subject (total 5000 binucleated cells per concentration), following the established criteria for MNs evaluation. 32 A total of 1000 lymphocytes per donor per concentration were scored to evaluate the percentage of cells with one to four nuclei. The cytokinesis-block proliferation index (CBPI) was calculated, according to the following formula: [1 × N1] + [2 × N2] + [3 × (N3 + N4)]/N, where N1–N4 represents the number of cells with one to four nuclei, respectively, and N is the total number of cells scored.
Chromosomal aberrations assay
Microscope analysis was performed at 1000× magnification on a light microscope (Dialux 20, Leica, Germany). Although the Organization for Economic Co-operation and Development guidelines call for 300 metaphases to be scored for each dose, 33 in order to obtain results more significant and suitable for statistical analysis, it is the normal practice in our laboratory to score 200 metaphases per subject per dose. Thus, in the present work, for each subject and BP-A concentration, 200 well-spread first-division complete metaphases (for a total of 1000 metaphases for each dose) containing 46 ± 1 chromosomes were analysed for the following categories of CAs: gaps, chromatid breaks (B′), chromosome breaks (B′′, dicentrics (Dic), rings (R), tri- or tetra-radials (TR), acentric fragments (AF), rearrangements (Re) and numerical aberrations. Cells containing one of more types of CAs were scored as ‘aberrant cell’ (Ab.C).
With regard to the opportunity to include gaps in the statistical analyses, the discussion is open.
Although some authors considered gaps as the appropriate indicator of genotoxic potential of chemicals, 34 the molecular mechanism of BP-A to induce achromatic lesion/gaps is yet to be revealed. Thus, in our statistical analyses, we decided to exclude gaps.
Statistical analysis
Comparison of mean values of the percentage of cells with MNs, CBPI and CAs between exposition levels and their controls was assessed by the non-parametric Wilcoxon Mann–Whitney U test. Statistical calculations were carried out using the SPSS software package program (version 23.0; SPSS, Inc., Chicago, Illinois, USA). All p values were two tailed, and p values of 5% or less were considered statistically significant for all tests carried out.
Results
Effect of BP-A on CAs formation
Table 1 shows values of CAs found in the human peripheral lymphocytes cultured in the presence of different concentrations of BP-A. BP-A was found to induce seven types of structural CAs (gaps, chromatid and chromosome breaks, dicentric chromosomes, rings, tri- or tetraradials, acentric fragments and rearrangements). No numerical aberrations were found.
Induction of chromosomal aberrations by bisphenol A in human lymphocytes in vitro.
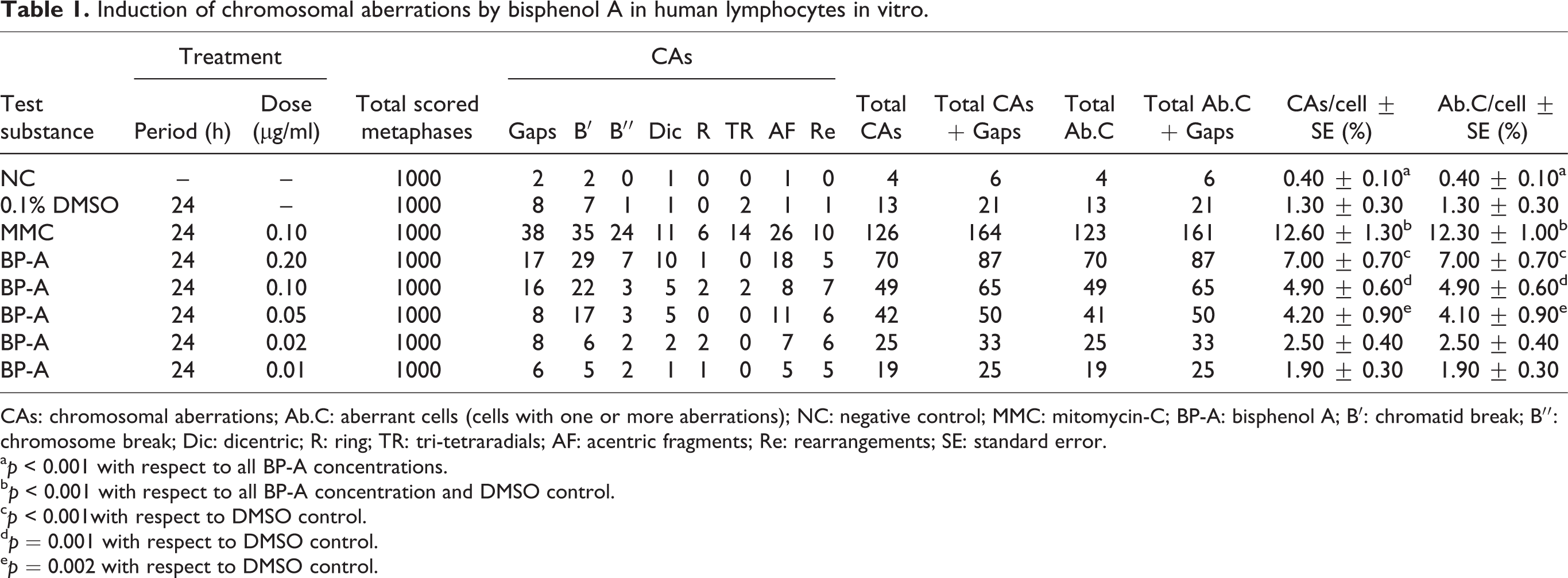
CAs: chromosomal aberrations; Ab.C: aberrant cells (cells with one or more aberrations); NC: negative control; MMC: mitomycin-C; BP-A: bisphenol A; B′: chromatid break; B′′: chromosome break; Dic: dicentric; R: ring; TR: tri-tetraradials; AF: acentric fragments; Re: rearrangements; SE: standard error.
a p < 0.001 with respect to all BP-A concentrations.
b p < 0.001 with respect to all BP-A concentration and DMSO control.
c p < 0.001with respect to DMSO control.
d p = 0.001 with respect to DMSO control.
e p = 0.002 with respect to DMSO control.
Data obtained indicated that human lymphocytes treated in vitro with BP-A at concentrations of 0.20, 0.10 and 0.05 μg/mL significantly increased the CAs frequency compared with the solvent control, whereas at the lower concentrations of 0.02 and 0.01 μg/mL, this effect was not observed.
Among tested concentrations, significant differences were found between 0.02 and 0.01 μg/mL and the other three tested concentrations (with p values of 0.001, 0.028 and 0.023, respectively, for 0.02 μg/mL and <0.001, 0.006 and 0.006, respectively, for 0.01 μg/mL), as well as between the higher tested concentration (0.20 μg/mL) and 0.05 μg/mL (p = 0.010). As already observed in our previous work, the DMSO solvent-control cultures did not show any difference compared to the control cultures (p = 0.058) confirming that, at this low dose (0.1%), DMSO has no relevant biological effects. Finally, as expected, control cultures treated with the known mutagen MMC showed a significant increase of CAs with respect to all BP-A tested concentrations (p < 0.001), as well as to the negative and solvent control cultures (p < 0.001).
Effect of BP-A on MNs formation
To verify both the aneugenic and clastogenic effects of BP-A, the MN test was assessed in parallel with CAs test (Table 2). Our results indicated that BP-A significantly, and in a dose-dependent manner, increased the MNs formation compared with the negative and solvent controls, at all concentrations tested, including 0.02 μg/mL, that represents the higher concentration of unconjugated BP-A found in literature in human serum. 2 Similar to what we already observed with the CAs assay, at the concentration of 0.01 μg/mL, that represent the TDI established by EU, 14 no significant cytogenetic effects were observed.
Induction of micronuclei by bisphenol A in human lymphocytes in vitro.

BNCs: binucleated cells; MNs: micronuclei; Ab.C: aberrant cells (cells with one or more MNs); NC: negative control; MMC: mitomycin-C; BP-A: bisphenol A; SE: standard error; CBPI: cytokinesis-block proliferation index.
a p < 0.001 with respect to all BP-A concentrations and MMC.
b p = 0.042 with respect to DMSO control.
c p = 0.039 with respect to DMSO control.
d p = 0.043 with respect to DMSO control.
The highest tested concentration (0.20 μg/mL) caused a fourfold increase in MNs as compared with the solvent culture. Moreover, significant differences in MNs frequency were also found between 0.20 μg/mL and all other concentrations, as well as between 0.10 μg/mL and the last two lower concentrations (0.02 and 0.01 μg/mL). The solvent-control cultures did not show any difference with the negative controls (p = 0.059), further confirming that at this low concentration, DMSO has no cytogenetic effects. Similar to what we observed with CAs assay, cultures treated with the mutagen MMC showed a substantially increased MNs formation compared with the negative control, solvent controls and all tested concentrations of BP-A. Finally, after 48-h exposure, a significant reduction of the CBPI value in cultures treated with BP-A was not observed, indicating that BP-A does not seem to produce effects on the proliferation/mitotic index when its concentration is ≤0.20 μg/mL.
Discussion
The genotoxicity of BP-A has been evaluated in several in vitro and in vivo studies, although with controversial results. 22 –24,26 Here, we discuss data obtained from our in vitro study conducted in order to establish the clastogenic and aneugenic potential of BP-A on human cultured lymphocytes. The results obtained in the present in vitro study with CAs and MNs assays evidenced a possible clastogenic effect of the BP-A on human lymphocytes, also at the concentration of 0.05 μg/mL that represents the RD established for humans by the US EPA. 28 This result is consistent with that obtained by Tayama et al., 35 who demonstrated that BP-A, in cultured CHO-K1 cells, is able to induce significant structural CAs, such as gaps, breaks and exchanges. Similarly, Xin et al. 22 observed a significant increase in MNs frequency in CHO cells after 24-h exposure to doses of 80 and 120 μM of BP-A (corresponding to 0.018 and 0.027 μg/mL, respectively). Finally, Parry et al. 20 and Johnson and Parry 36 showed that BP-A causes MNs induction, also in these cases in a dose-dependent manner, on MCL-5 and AHH-1 human lymphoblastoid cell lines and Chinese hamster V79 cells.
The increased cytogenetic damage observed with both CAs and MNs assays at concentration of 0.05 μg/mL pushes towards the adoption of lower reference limits. Indeed, increased CAs and MNs frequencies in peripheral blood lymphocytes have been positively associated with increased cancer risk and early events in carcinogenesis, respectively. 29,31 Vice versa, at the concentration of 0.01 μg/mL, the TDI established by the EU (2002), 14 no cytogenetic effects were observed, indicating that this latter represents a more safe concentration for human health with respect to 0.05 μg/mL.
The clastogenic properties of the BP-A were attributed by Adamkis et al. 37 to its capacity to interact with the cell microtubules causing their depolymerization and consequent manifestation of chromosomal abnormalities. Moreover, it is evident from the literature that BP-A is able to generate reactive oxygen species (ROS) by decreasing the activities of antioxidant enzymes and increasing lipid peroxidation. A possible consequence of this action could be the increase in the cellular oxidative stress that is considered one of the possible mechanisms underlying DNA damage. 38 BP-A was also found to be able to up-regulate the expression of ERCC5 encoding a DNA endonuclease involved in nucleotide excision repair. This is an important issue because a reduced repair capacity has been reported to be associated with increased risk of genomic damage and cancer development. 39
However, we cannot ascribe the observed cytogenetic damage exclusively to the direct effect of BP-A on lymphocytes. Indeed, in vitro studies showed that BP-A is converted to DNA-binding metabolites. In particular, BP-A is oxidized to bisphenol-o-quinone whose chemical reaction with DNA produces adducts in vitro and in vivo. 40 The irreversible binding of BP-A-derived compounds to DNA could cause bases loss from the DNA strand and could be responsible for some of the BP-A-observed toxic effects. It is known that many chemicals, either directly or after conversion to reactive metabolites, covalently modify nucleosides of DNA leading to mutational changes and, thus, playing a role in cellular toxicity or tumorigenesis induction. 40
Moreover, it should also be emphasized that the link between observed toxicological effects and BP-A exposure, especially at low doses, is complicated by the fact that BP-A is prevalent in many consumer products, including common laboratory plastics, and has the potential to leach from these products. Consequently, these background concentrations of BP-A have the potential to affect the results of in vitro studies that detect low concentration effects. 41 Indeed, some in vitro studies showed that BP-A induces significant effects also to nominal concentrations. For example, a BP-A concentration of 0.23 pg/mL was found to promote proliferation of human seminoma cells (JKT-1). 42
Interestingly, BP-A increased MNs frequency also at the concentration of 0.02 μg/mL, where CAs failed. These apparently discordant results could be explained by the fact that the MNs assays reveal alterations in either chromosome number or chromosome structure, whereas the CAs analysis can detect alterations only in the chromosome structure. One of the mechanisms of action of aneugenic chemicals is the induction of alterations to centromeric DNA, with consequent failure of the damaged chromosomes to attach to the mitotic spindle. As also postulated by other authors for other xenobiotics, 43 it is possible that BP-A may also damage the chromosomes centromeres, determining MNs formation. Moreover, disruption of the mitotic machinery, that is, microtubules and microtubule organizing centres, has been shown to be the mechanism of action for BP-A-induced spindle-damaging effects. 44 In this scenario, it can be hypothesized that BP-A might induce aneuploidy through non-disjunction.
Conclusion
In conclusion, the results reported herein demonstrate cytogenetic effects of BP-A on cultured human lymphocytes by increasing MNs and CAs frequencies. A large number of studies showing the clastogenic and toxic properties of BP-A were performed using different cell models. For these reasons, although simple experimental models, like mammalian and bacterial cells, cannot accurately mimic the complex kinetics of xenobiotic compounds in vivo, our results suggest the adoption of more stringent measures in order to reduce the presence of this compound in the environment and to minimize the adverse health effects of BP-A exposure. Finally, being BP-A converted in vivo and in vitro in DNA binding metabolites, it would be useful and interesting to test in vitro also the genotoxicity of the BP-A metabolites or a combination of both BP-A and its metabolites. Indeed, BP-A was found to have a short half-life (47 h in H295R cells) 45 and therefore it is conceivable that in in vitro systems about 50% of BP-A could not be metabolized after 48-h treatment (typical of MNs assay) and more than 50% after 24-h treatment (typical of CAs assay). In this sense, a combined genotoxic action of both BP-A and its metabolites cannot be excluded.
Footnotes
Acknowledgement
The authors are grateful to all volunteers who participated in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grant from the Italian Ministry of University and Scientific Research (ex 60%).
