Abstract
Multiple changes in metabolic levels could be useful for understanding physiological toxicity. To explore further risk factors for the convulsions induced by the interaction of nonsteroidal anti-inflammatory and new quinolone antimicrobial drugs, the effect of sparfloxacin, enoxacin, and felbinac on fatty acid metabolism and glucose concentrations in the liver, brain, and blood of rats was investigated. The levels of long-chain acyl-CoAs (C18:1 and C20:4) in the liver and brain were decreased at the onset of convulsions induced by the coadministration of enoxacin with felbinac. Then, glucose concentrations in the liver and blood were decreased, whereas they were increased in a dose-dependant manner in the brain. However, the formation of acyl-CoAs and glucose levels in the liver, brain, and blood was not significantly influenced by enoxacin, felbinac, and sparfloxacin alone, respectively. The disturbance of both fatty acid metabolism and glucose levels might be associated with the increased susceptibility to convulsions, which may contribute to further understanding of the toxic effects associated with these drugs.
Introduction
The interaction of nonsteroidal anti-inflammatory (NSAIDs) and new quinolone antimicrobial drugs (NQs) is well known to induce adverse reactions such as convulsions. 1 –7 New quinolone antimicrobial drugs have potent convulsant activity, and the side effects are often enhanced by concomitant administration with NSAIDs. A widely accepted mechanism for the onset of convulsions involves the blockage of γ-aminobutyratergic (GABA) neurotransmission in the central nervous system. However, the details of the interactions between NQs and NSAIDs have not been fully understood.
Bough and Rho focused particular attention on the role that limited glucose, fatty acids, and ketone bodies may play in anticonvulsant activity of the ketogenic diet (KD). 8 Metabolic signatures could be useful for predicting physiological toxicity and elucidating the mechanism of adverse reactions. Multiple changes in metabolic levels may contribute to the increased susceptibility to convulsions induced by the interaction of NSAIDs and NQs.
We have previously found that a medium-chain acyl-CoA synthetase purified from mitochondria of mouse liver and kidney and bovine liver was inhibited by NSAIDs and NQs. 9 –11 Diflunisal, nalidixic acid, and salicylic acid inhibited competitively the purified medium-chain acyl-CoA synthetase activity for hexanoic acid, with Ki values of 0.8, 12.4, and 19.6 μmol/L, respectively. In addition, enoxacin and ofloxacine inhibited this medium-chain acyl-CoA synthetase in a mixed-type manner with Ki values of 23.7 and 38.2 μmol/L, respectively. The administration of salicylic acid decreased the levels of both the medium-chain and the long-chain acyl-CoAs in mouse liver and only long-chain acyl-CoAs in mouse brain. The control of acyl-CoA synthetases, which metabolize not only xenobiotic carboxylic acids but also fatty acids, may be disturbed by drugs. It would therefore be of interest to study the inhibition of the medium-chain acyl-CoA synthetases by NSAIDs and NQs.
Much information is available about the adverse effects of NQs, such as alterations in blood glucose, QTc interval prolongation, seizures, phototoxicity, and tendinopathy, etc. 12,13 The mechanism of hypoglycemia is suggested to be that NQs commonly cause insulin secretion through direct inhibition of pancreatic β-cell potassium channel activity. 14 On the other hand, gatifloxacin and levofloxacin cause hypoglycemia and hyperglycemia, especially in patients with diabetes. 15,16 The rate of hyperglycemia is less encountered. Although the mechanism of hyperglycemia is not known, it may be related to the vacuolation of pancreatic β-cells, leading to a decrease in insulin secretion. 17
The metabolism of fatty acids and glucose is closely related. Convulsions induced by NSAIDs and NQs may be connected in some way with the disturbance of both fatty acid metabolism and glucose levels. We have found a report in which the reduction of glucose levels and/or increases in fat might result in an elevated seizure threshold. 18 The KD, which is very high in fats and low in carbohydrates, has been employed as an effective treatment for medically refractory epilepsy. The KD is designed to create the metabolic conditions of fasting and makes the body burn fat for energy instead of glucose. The anticonvulsant activity of the KD is widely accepted. 8,19 –24 Despite nearly a century of use, however, the anticonvulsant mechanisms of the KD have proven elusive. Ketone bodies (metabolites of fatty acids), fatty acid, and glucose restriction might each lead directly or indirectly to seizure control. 8,22 –26 On the other hand, it is interesting that the KD is generally most effective against seizures evoked by a GABAergic antagonist, pentylenetetrazole. 8,27,28
To understand the mechanisms of action involved in convulsions, it is important to discover their metabolic signature. To explore further risk factors that may be associated with the development of convulsions induced by NSAIDs and NQs, we investigated the effect of sparfloxacin, enoxacin, and felbinac on the intracellular situation of acyl-CoA esters and glucose levels. Acyl-CoAs are key metabolites in fatty acid metabolism and were analyzed by Liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS).
Materials and Methods
Materials
Male Wistar rats (250-300 g) were purchased from SLC Co Ltd (Shizuoka, Japan). Hexanoyl-CoA, oleoyl-CoA, and arachidonoyl-CoA were obtained from Sigma Chemical Co (St Louis, Missouri). Enoxacin, sparfloxacin, felbinac, glucose, and hexokinase were purchased from Nacalai Tesque (Kyoto, Japan), CoA and ATP from Kohjin Co Ltd (Tokyo, Japan). Glucose 6-phosphate dehydrogenase (G-6-PDH) and nicotinamide adenine dinucleotide phosphate oxidized form (NADP+) were purchased from Wako Pure Co Ltd (Kyoto, Japan). Other chemicals used were of analytical grade and were used as received.
All the experiments were approved by the Ethical Committee for Experimental Work of Kobegakuin University and were performed in accordance with the guidelines for the Care and Use of Laboratory Animals published by our institute.
Administration of Felbinac, Enoxacin, and Sparfloxacin
Wistar rats were housed 1 per cage in plastic cages and fed a commercial chow. The animal laboratory was maintained at 20°C to 23°C with 12-hour light and dark cycles. Wistar rats were fasted for 24 hours with free access to water. Experiments were performed in rats fasted for 24 hours. Concomitant administration of enoxacin and felbinac was considered to be the most risky combination. 7,29 Sparfloxacin has very weak convulsion-inducing activity. 13 To compare multiple changes in metabolic levels at the onset of convulsion versus nonconvulsion, enoxacin, felbinac, and sparfloxacin were selected. Enoxacin (10, 50, and 150 mg/kg) was coadministered intraperitoneally (ip) with felbinac (10, 50, and 150 mg/kg) to rats at an equal dose of each according to a modification of the method described by Kita et al, 29 respectively. On the other hand, felbinac, enoxacin, and sparfloxacin alone were administered ip to rats at 50 mg/kg, respectively. Combination of each equal dose of enoxacin (50 and 150 mg/kg) with felbinac (50 and 150 mg/kg) always induced convulsions in all of the 6 rats within 20 to 30 minutes. Therefore, at 30 minutes (in the case of nonconvulsion) or the onset of convulsion after each dose, blood samples were drawn from the heart under anesthesia with diethylether, and the brain and liver were removed. Each tissue was immediately stored frozen at −40°C. In all, 6 rats were used at each dose.
Extraction of Medium-Chain and Long-Chain acyl-CoA Esters, Enoxacin, Felbinac, and Sparfloxacin
The medium-chain and long-chain acyl-CoA esters, enoxacin, felbinac, and sparfloxacin were extracted from each tissue as described previously. 30,31
Briefly, the weight of each frozen tissue was determined and 1 mL of 10 mmol/L ammonium acetate buffer (pH 5.3) was added to per 0.5 g of wet tissue. Tissues were homogenized and then 3 mL of ice-cold chloroform/methanol (2:1, v/v) was added to the tissue homogenate. The mixture was rehomogenized and centrifuged at 1300 g for 30 minutes. The upper phase (the buffer phase) was removed and the tissue pellet was also rehomogenized in 3 mL of chloroform/methanol (2:1) and centrifuged as described above. The supernatant was added to the first supernatant. The upper phase was used for the analysis of acyl-CoAs and the lower phase was for enoxacin, felbinac, and sparfloxacin.
For the analysis of acyl-CoA esters, the 2 combined upper phases were washed twice with 2 mL of chloroform/10 mmol/L ammonium acetate buffer (pH 5.3; 1:1, v/v). The upper phases were combined and evaporated below 40°C. The acyl-CoA esters were stable below 40°C under evaporation. The dry residue was dissolved in 0.1 mL of 10 mmol/L ammonium acetate buffer (pH 5.3) for LC-ESI-MS/MS.
For the analysis of enoxacin, felbinac, and sparfloxacin, the 2 chloroform phases (the lower phases) were combined and evaporated below 40°C. The dry residue was dissolved in 0.5 mL of 50% methanol solution for LC-ESI-MS.
Calibration curves of the medium-chain and long-chain acyl-CoA esters were obtained by adding known amounts of authentic medium-chain and long-chain acyl-CoA esters to the tissues, and extracting them. The amount of each acyl-CoA ester was determined by linear regression of peak area representing each acyl-CoA ester versus concentration. As the tissues already contain various acyl-CoA esters, net peak areas for the known concentrations of the individual acyl-CoAs were calculated by subtracting peak areas of acyl-CoAs in tissues from those of samples added with standards.
Calibration curves of enoxacin, sparfloxacin, and felbinac were obtained by adding known amounts of authentic enoxacin, sparfloxacin, and felbinac, and extracting them. The amounts of enoxacin, sparfloxacin, and felbinac were determined by linear regressions of peak areas representing enoxacin, sparfloxacin, and felbinac versus concentrations, respectively.
The recoveries of 1 nmol of hexanoyl-CoA, oleoyl-CoA, and arachidonoyl-CoA added to the liver were 48.1%, 60.2%, and 61.5%, respectively. The recoveries of 5 nmol of enoxacin, sparfloxacin, and felbinac added to the liver were 83.2%, 85.3%, and 75.5%, respectively.
LC-ESI-MS/MS Analysis of the Medium-Chain and Long-Chain Acyl-CoAs
The medium-chain and long-chain acyl-CoA esters were determined as described previously. 31
High-performance liquid chromatography (HPLC) separation was performed with a Waters 2690 instrument (Micromass, Manchester, UK) having a Capcellpak C18, UG120 column (2.0 mm id × 150 mm). The mobile phases were A, 10 mmol/L acetate ammonium buffer (pH 5.3) and B, acetonitrile. The eluting gradient was as follows: the column was equilibrated with, 84% A for 5 minutes, 84% A to 16% A in 20 minutes, and 16% A to 84% A in 7 minutes. The flow rate was 0.2 mL/min. Quantitation was carried out on a Quattro Ultima (Micromass, Manchester, UK). The mass spectrometer was operated in the positive-ion mode with the source temperature set to 110°C, a corn voltage of 45 V, and a capillary voltage of 3.5 kV. Collision energy was individually tuned at 30 eV. All positive-ion mass spectral data were obtained by scanning the mass range from m/z 100 to 1200.
The medium-chain and long-chain acyl-CoAs in tissues were quantified by selective multireaction monitoring (MRM) with a positive ionization mode. The medium-chain acyl-CoA of C6 and long-chain acyl-CoAs of C18:1 and C20:4 were selected. The peak of each acyl-CoA was monitored by the product ion obtained from [M+H]+ ion of each medium-chain and long-chain acyl-CoA ester (ie, m/z 866 > 359 for hexanoyl-CoA, m/z 1032 >525 for oleoyl-CoA, and m/z 1054 > 547 for arachidonoyl-CoA).
LC-ESI-MS Analysis of Enoxacin, Felbinac, and Sparfloxacin
High-performance liquid chromatography separation was performed with a Waters 2690 instrument (Micromass, Manchester, UK) having a 5-μm Cosmosil C8-MS column (4.6 mm id × 150 mm). Enoxacin, felbinac, and sparfloxacin were eluted with acetonitrile:10 mmol/L acetate ammonium buffer (pH 3.8; 20:80, v/v) at a flow rate of 0.2 mL/min. Quantitation was carried out on a Quattro Ultima (Micromass, Manchester, UK). The mass spectrometer was operated in both the positive- and the negative-ion modes with the source temperature set to 100°C, a corn voltage of 30 V, and a capillary voltage of 3.5 kV. All positive- and negative-ion mass spectral data were obtained by scanning the mass range from m/z 100 to 1000.
The positive-ion mass spectra of enoxacin and sparfloxacin were dominated by [M+H]+ ions at m/z 321 and 393, respectively. The negative-ion mass spectrum of felbinac gave a dominant [M-COOH]− ion at m/z 167. Enoxacin, sparfloxacin, and felbinac in the tissues were quantified by selective ion monitoring (SIM) with the positive and negative ionization modes. The peaks of enoxacin and sparfloxacin were monitored by m/z 321 and 393, and the peak of felbinac was by m/z 167, respectively. The retention times of enoxacin, sparfloxacin, and felbinac were 5.9, 14.2, and 16.6 minutes, respectively.
Analysis of Glucose in the Liver, Brain, and Blood
Glucose was measured with a UV-1700 spectrophotometer (Shimadzu Co Ltd, Japan), using a method similar to de Vries et al. 32 Briefly, plasma samples (0.3 mL) were mixed with 1.0 mL reaction mixture (1.7 μmol MgCl2, 3 μmol ATP, 3 μmol NADP+, 4.0 units hexokinase and 0.4 units G-6-PHD in 100 mmol/L Tris-HCl buffer [pH 8.0]) and incubated at 37°C for 2 minutes. Absorbance of the reaction mixture was measured at 340 nm.
Glucose in the liver and brain was extracted as described by Karcher et al. 33 Briefly, the weight of each frozen tissue was determined and 2 mL of 100 mmol/L Tris-HCl buffer (pH 8.0) was added to per 1.0 g of wet tissue. The tissues were homogenized and then 2.5 mL of 0.3 mol/L HClO4 was added to the tissue homogenate. The mixture was rehomogenized and centrifuged at 12 000g for 30 minutes. In all, 1 mL of 1.0 mol/L NaOH was added to the upper phase removed and centrifuged as described above. The supernatant was filtrated with Ultrafree-MC (0.45 μm) and aliquots of the filtrate were subjected to glucose analysis as described above.
Glucose levels in the liver, brain, and blood were determined by linear regressions of absorbance representing glucose versus concentrations, respectively.
Statistical Analysis
The data were expressed as mean ± SEM (standard error of mean) and the results were analyzed by one-way analysis of variance (ANOVA) followed by Dunnett’s “t” multiple comparison test. A difference was considered significant when P < .05.
Results
LC-ESI-MS/MS Analysis of Acyl-CoA Esters
The medium-chain and long-chain acyl-CoA esters were extracted from tissues as described previously. 30,31 The liver and brain contained the relatively high amounts of acyl-CoAs of C6:0, C18:1, and C20:4 (data not shown). The acyl-CoA of C18:1 is the major composition of triacylglycerol. Polyunsaturated fatty acids, as in arachidonic acid, might be involved in the anticonvulsive effect of KD. 8,24 To examine the effect of enoxacin, felbinac, and sparfloxacin on fatty acid metabolism, a medium-chain acyl-CoA ester of C6:0 and 2 long-chain acyl-CoAs of C18:1 and C20:4 were selected. The typical MRM chromatograms of 3 acyl-CoA esters of C6:0, C18:1, and C20:4 of the extract obtained from rat brain are shown in Figure 1 . Excellent separation of medium-chain and long-chain acyl-CoAs was achieved without interference of biological materials.
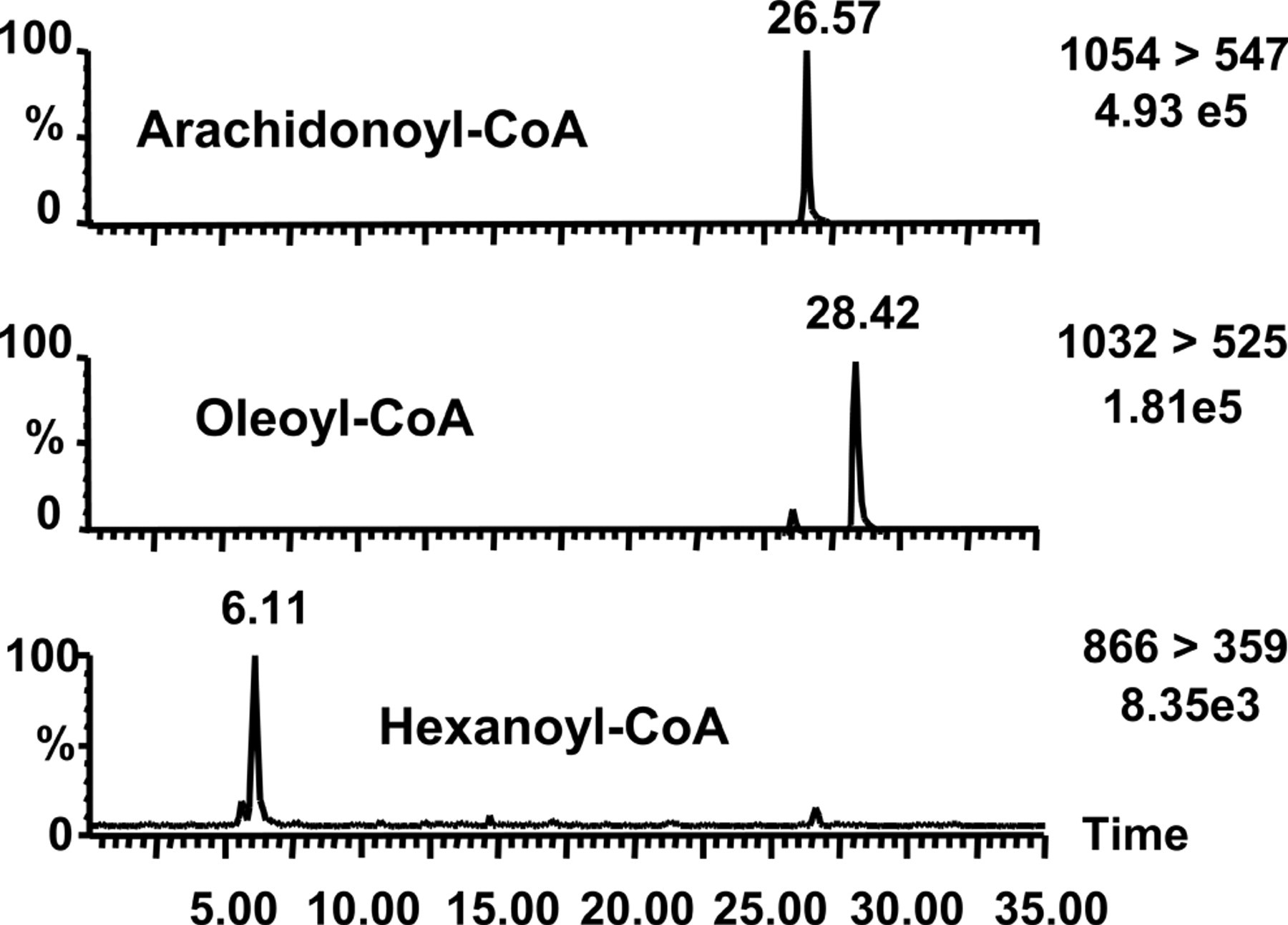
The typical multireaction monitoring (MRM) chromatograms of 3 acyl-CoA esters of C6:0, C18:1, and C20:4 of the extract obtained from rat brain. The targeted ions of acyl-CoAs are m/z 866 > 359 (C6:0), m/z 1032 > 525 (C18:1), and m/z 1054 > 547 (C20:4).
Concentrations of Enoxacin, Felbinac, and Sparfloxacin in Rat Liver and Brain
Felbinac is known to enhance the convulsant activity of enoxacin strongly. Therefore, enoxacin (10, 50, and 150 mg/kg) was coadministered ip with felbinac (10, 50, and 150 mg/kg) to rats at an equal dose of each, respectively. Sparfloxacin has weak convulsion-inducing activity. Felbinac, enoxacin, and sparfloxacin alone were administered ip to rats at 50 mg/kg, respectively. Blood samples were drawn and each tissue was removed 30 minutes (in the case of nonconvulsion) or at the onset of convulsions (always induced within 20 to 30 minutes) after each dose. To obtain insights into multiple changes in metabolic levels at the onset of convulsions compared with nonconvulsion, it is necessary to obtain information concerning the concentrations of each drug in tissues.
The combination of each equal dose of enoxacin (50 and 150 mg/kg) with felbinac (50 and 150 mg/kg) induced convulsions in all of the 6 rats within 20 to 30 minutes, respectively. However, convulsions were not induced at all after the ip administration of felbinac (50 mg/kg), enoxacin (50 mg/kg), and sparfloxacin (50 mg/kg) alone, and of enoxacin (10 mg/kg) in combination with felbinac (10 mg/kg) to rats, respectively.
Hepatic concentrations of enoxacin and felbinac after the coadministration of enoxacin (50 mg/kg) with felbinac (50 mg/kg) were 500.3 nmol/g and 211.0 nmol/g wet tissue, respectively (Table 1 ). In the brain, 3.0 nmol/g enoxacin and 96.8 nmol/g felbinac were observed.
Levels of Enoxacin, Felbinac, and Sparfloxacin in the Tissues After the Intraperitoneal Administration of Enoxacin, Felbinac, and Sparfloxacin alone, and Enoxacin in Combination With Felbinac a
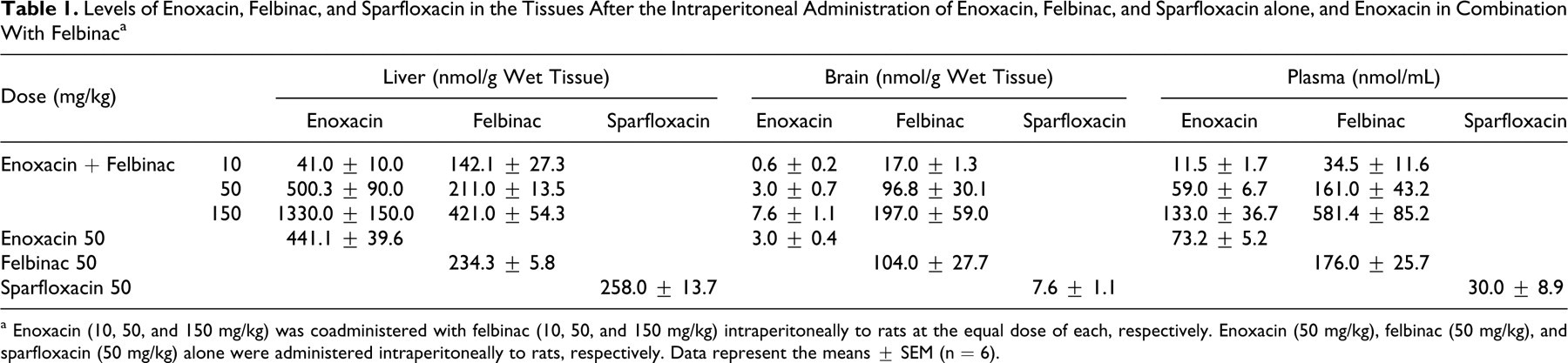
a Enoxacin (10, 50, and 150 mg/kg) was coadministered with felbinac (10, 50, and 150 mg/kg) intraperitoneally to rats at the equal dose of each, respectively. Enoxacin (50 mg/kg), felbinac (50 mg/kg), and sparfloxacin (50 mg/kg) alone were administered intraperitoneally to rats, respectively. Data represent the means ± SEM (n = 6).
After ip administration of enoxacin (150 mg/kg) in combination with felbinac (150 mg/kg), the liver contained a high 1330.0 nmol/g concentration of enoxacin, whereas the plasma concentration of enoxacin was low at 133.0 nmol/mL compared with felbinac (581.4 nmol/mL). The levels of enoxacin (7.6 nmol/g) and felbinac (197.0 nmol/g) in the brain were 0.6% and 46% of those in the liver, respectively.
In spite of the different doses (10, 50, and 150 mg/kg) of enoxacin in combination with felbinac, the transport rates into the brain (brain/plasma concentration) did not change in a dose-dependent manner. The transport rate of enoxacin into the brain (brain/plasma concentration) was low, 0.051, whereas that of felbinac was high, 0.6. Less enoxacin was transported into the brain than felbinac.
Similar levels of enoxacin and felbinac in the liver, brain, and plasma were observed in the case of enoxacin (50 mg/kg) and felbinac (50 mg/kg) alone, and enoxacin (50 mg/kg) in combination with felbinac (50 mg/kg), respectively (Table 1). The interaction between enoxacin and felbinac had no effect on the distribution of each drug.
The levels of sparfloxacin were 30.0 nmol/mL in plasma, 7.6 nmol/g in the brain, and 258.0 nmol/g in the liver after ip administration of sparfloxacin (50 mg/kg) alone (Table 1). The transport rate of sparfloxacin into the brain (brain/plasma concentration) was low, 0.25.
Effect of Enoxacin, Sparfloxacin, and Felbinac on the In Vivo Formation of Acyl-CoA Esters
We examined the effect of enoxacin, sparfloxacin, and felbinac on 1 medium-chain acyl-CoA ester of C6:0 and 2 long-chain acyl-CoAs of C18:1 and C20:4. The tendency on decreases of acyl-CoAs of C6, C18:1, and C20:4 in the liver and brain was observed with the dose of 50 mg/kg of enoxacin and felbinac alone, respectively, but was not significant (Table 2 ). Sparfloxacin did not exert an influence over levels of acyl-CoAs (C6, C18:1, and C20:4) in the liver and brain.
Levels of Acyl-CoAs in the Tissues After the Intraperitoneal Administration of Enoxacin, Felbinac, and Sparfloxacin Alone a
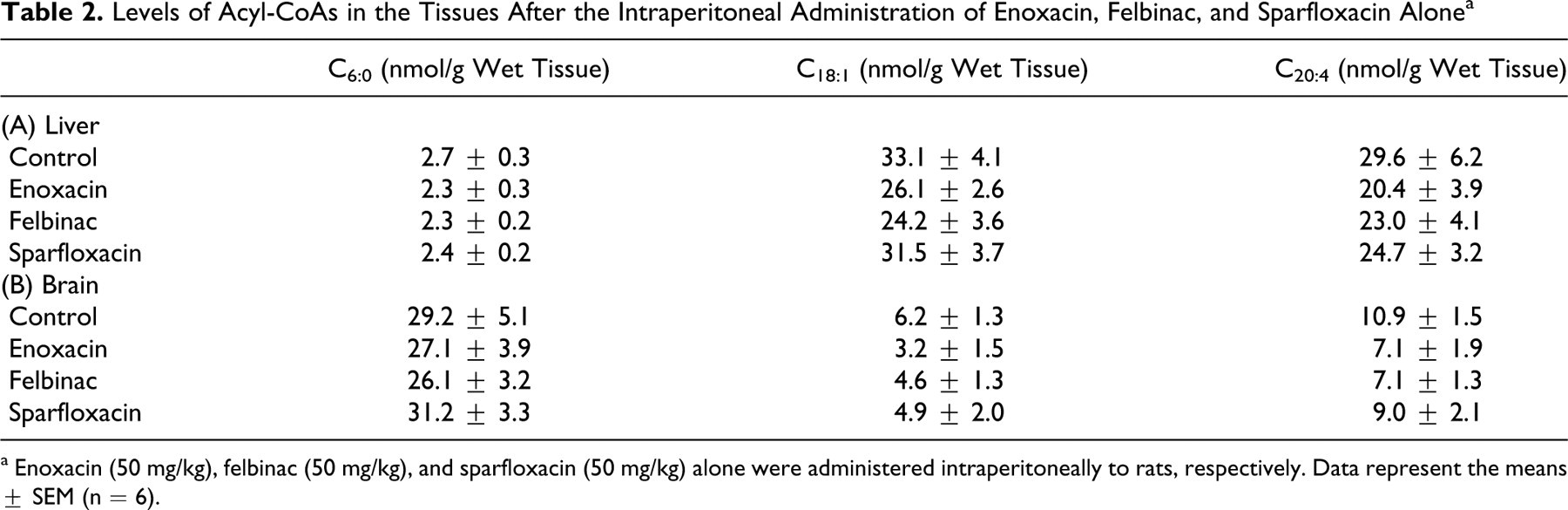
a Enoxacin (50 mg/kg), felbinac (50 mg/kg), and sparfloxacin (50 mg/kg) alone were administered intraperitoneally to rats, respectively. Data represent the means ± SEM (n = 6).
The levels of acyl-CoA esters of C6:0, C18:1, and C20:4 in the liver and brain after the coadministration of enoxacin with felbinac are shown in Figures 2 and 3 , respectively. At the onset of convulsions, the levels of acyl-CoAs of C18:1 and C20:4 in the liver and brain were significantly decreased, respectively. The interaction between enoxacin and felbinac had no effect on the distribution of each drug. These findings suggest that the combination of individual decrease rate of acyl-CoA of C18:1 and C20:4 induced by enoxacin with that by felbinac resulted in the significant decrease of each acyl-CoA. The levels of hexanoyl-CoA in the liver and brain were not significantly decreased, respectively.
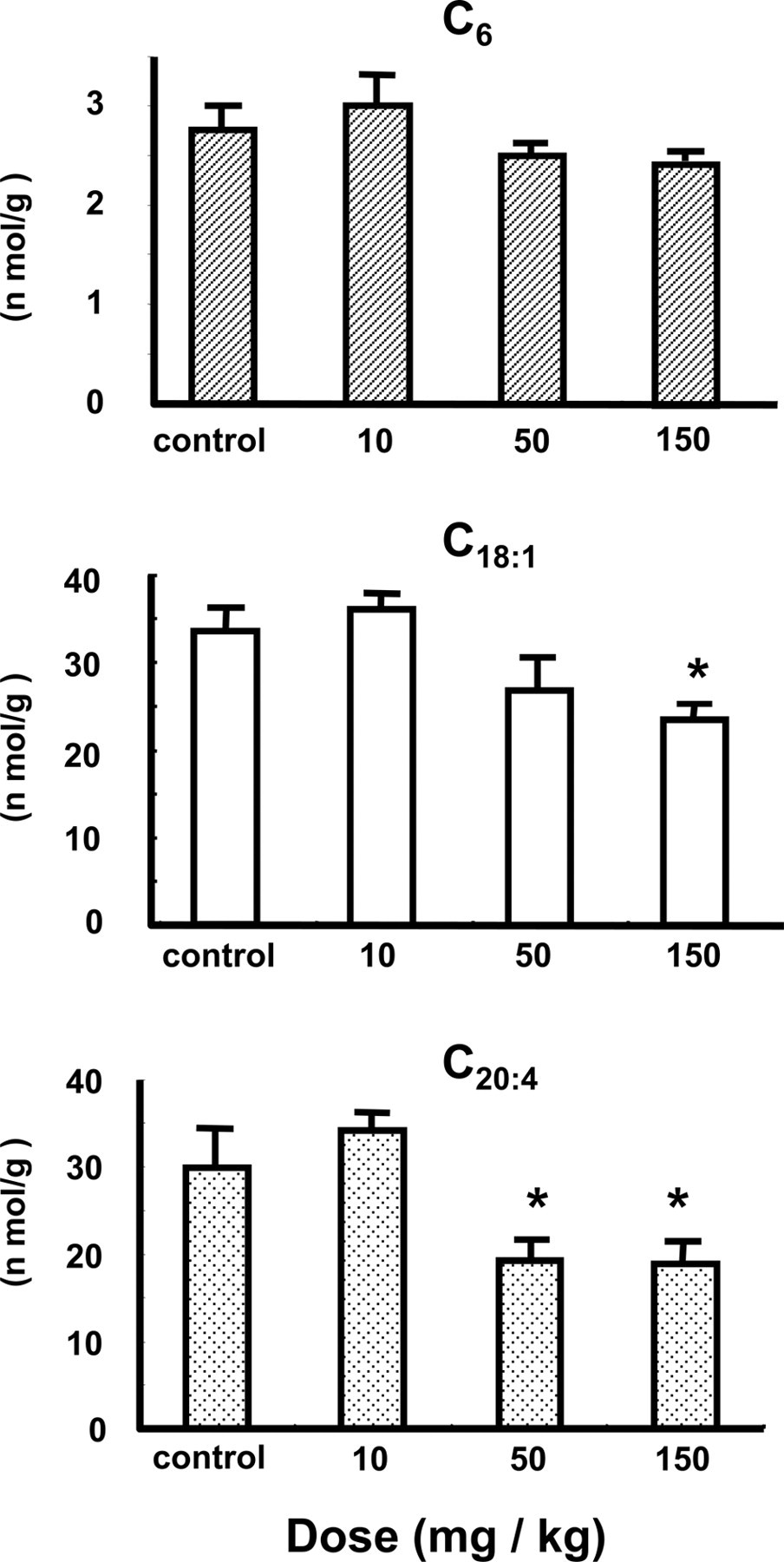
Effect of the combination of enoxacin with felbinac on acyl-CoA formation in rat liver. Enoxacin (10, 50, and 150 mg/kg) was coadministered with felbinac (10, 50, and 150 mg/kg) intraperitoneally to rats at an equal dose of each, respectively. Data represent the means ± SEM (n = 6). *Significantly different from control rats (P < .05).
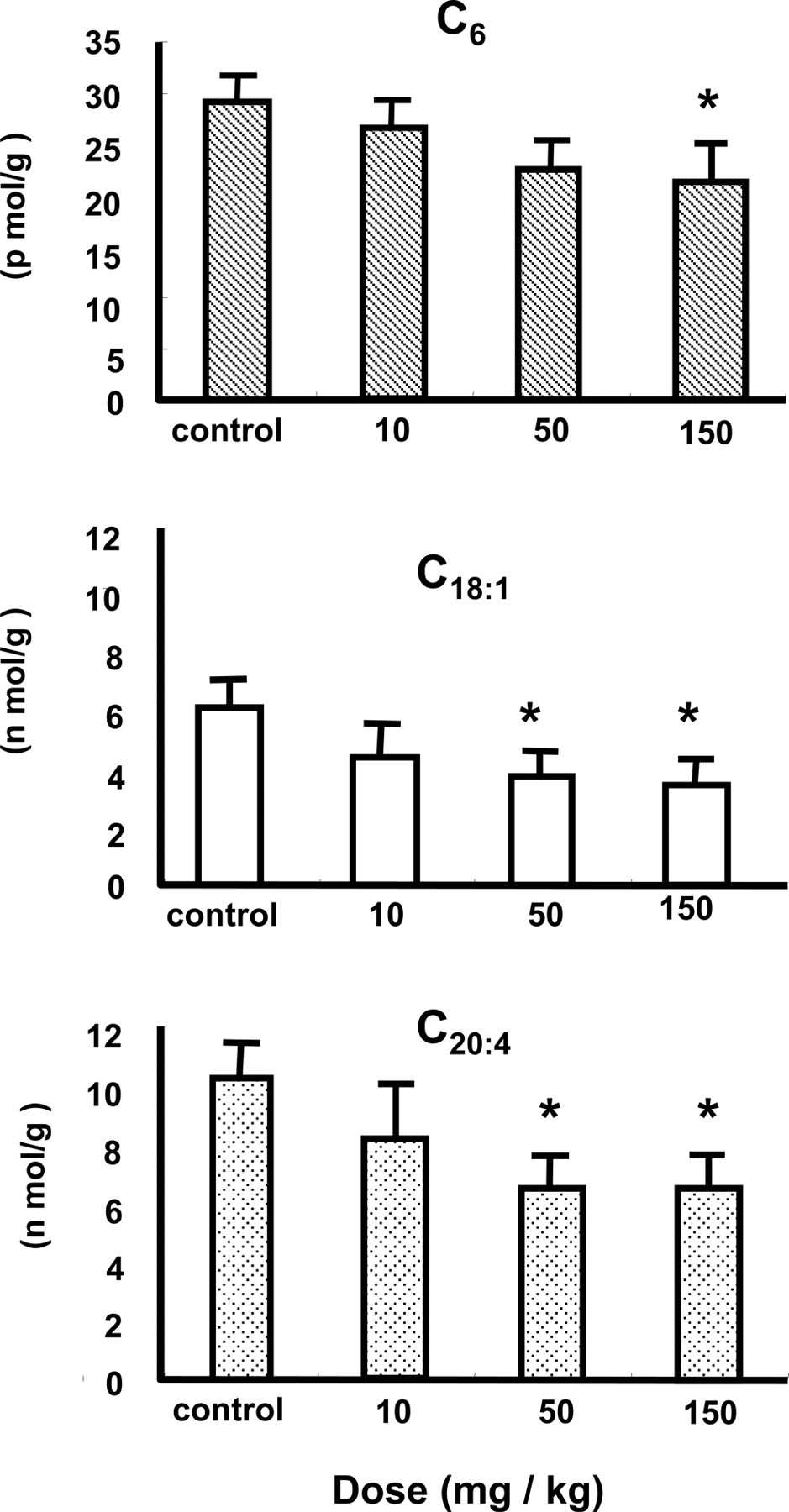
Effect of the combination of enoxacin with felbinac on acyl-CoA formation in rat brain. Enoxacin (10, 50, and 150 mg/kg) was coadministered with felbinac (10, 50, and 150 mg/kg) intraperitoneally to rats at an equal dose of each, respectively. Data represent the means ± SEM (n = 6). *Significantly different from control rats (P < .05).
Effect of Enoxacin, Felbinac, and Sparfloxacin on Glucose Concentrations in the Liver, Brain, and Blood
Glucose levels in the liver, brain, and blood of rats were determined after the ip coadministration of enoxacin with felbinac (Figure 4 ). At the onset of convulsions, blood glucose was decreased by 25% compared with the control after the administration of enoxacin (50 mg/kg) in combination with felbinac (50 mg/kg), whereas it was decreased by 10% after the 150 mg/kg dose. Hepatic glucose concentrations were significantly reduced by 32% and 32% with the combination of equal doses of enoxacin and felbinac at 50 and 150 mg/kg, respectively, whereas glucose levels in the brain were increased by 21% and 55% compared with the control, respectively. Alterations in glucose levels were observed in the liver, brain, and blood. As blood glucose was increased after the 150 mg/kg dose, a similar tendency (decrease of hepatic glucose and increase of brain glucose) for glucose levels in the liver and brain was shown in the case of 50 and 150 mg/kg doses.
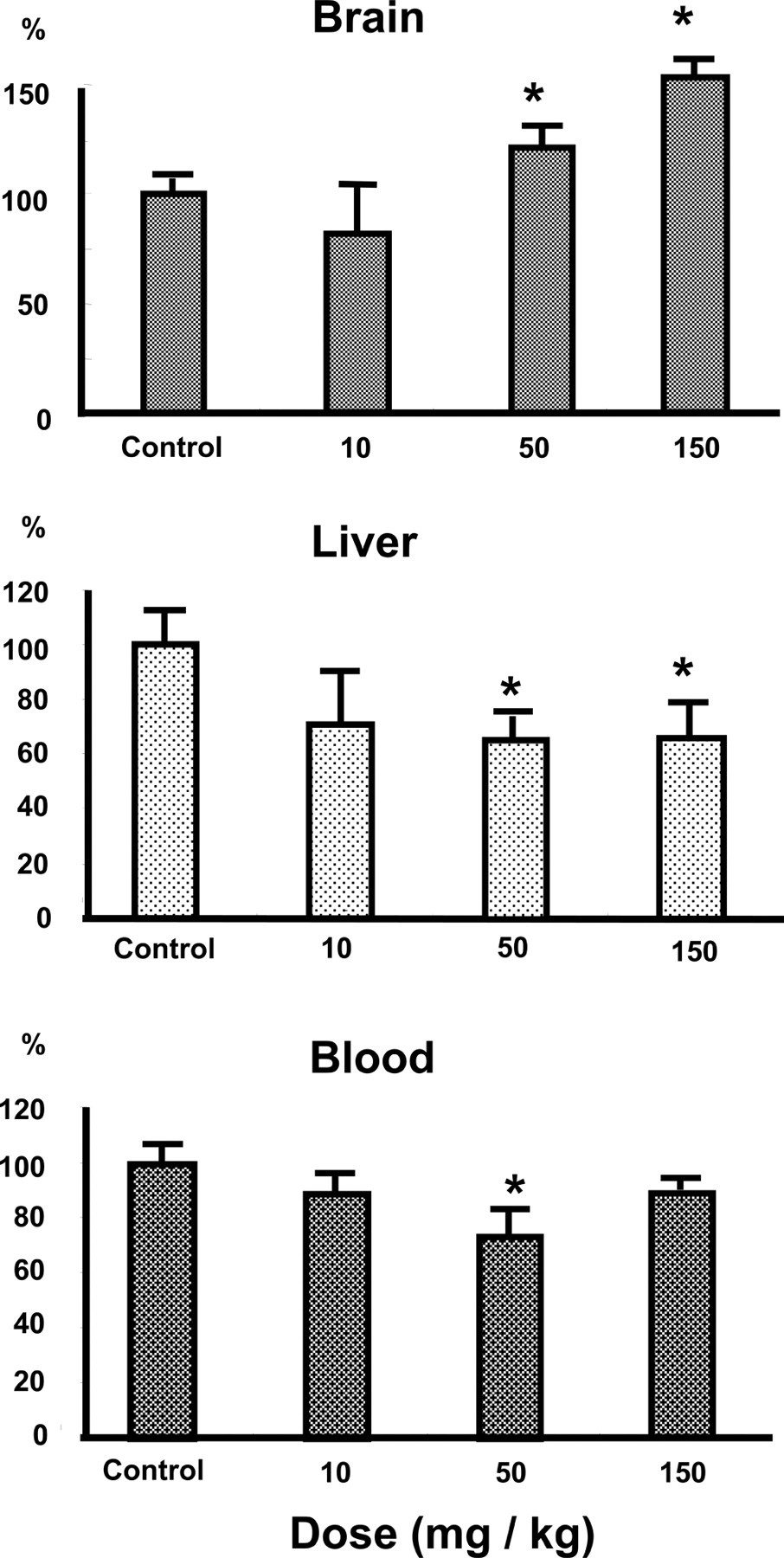
Effect of the combination of enoxacin with felbinac on glucose concentrations in the liver, brain, and blood of rats. Enoxacin (10, 50, and 150 mg/kg) was coadministered with felbinac (10, 50, and 150 mg/kg) intraperitoneally to rats at an equal dose of each, respectively. Data represent the means ± SEM (n = 6). Glucose levels in the brain, liver, and blood of rats were 72.3 μg/g wet tissue, 3.2 mg/g wet tissue, and 121.8 mg/dL as the control values, respectively. *Significantly different from control rats (P < .05).
When convulsions were not induced, glucose levels in the liver, brain, and blood were not significantly affected (Table 3 ).
Levels of Glucose in the Tissues After the Intraperitoneal Administration of Enoxacine, Felbinac, and Sparfloxacin Alone a
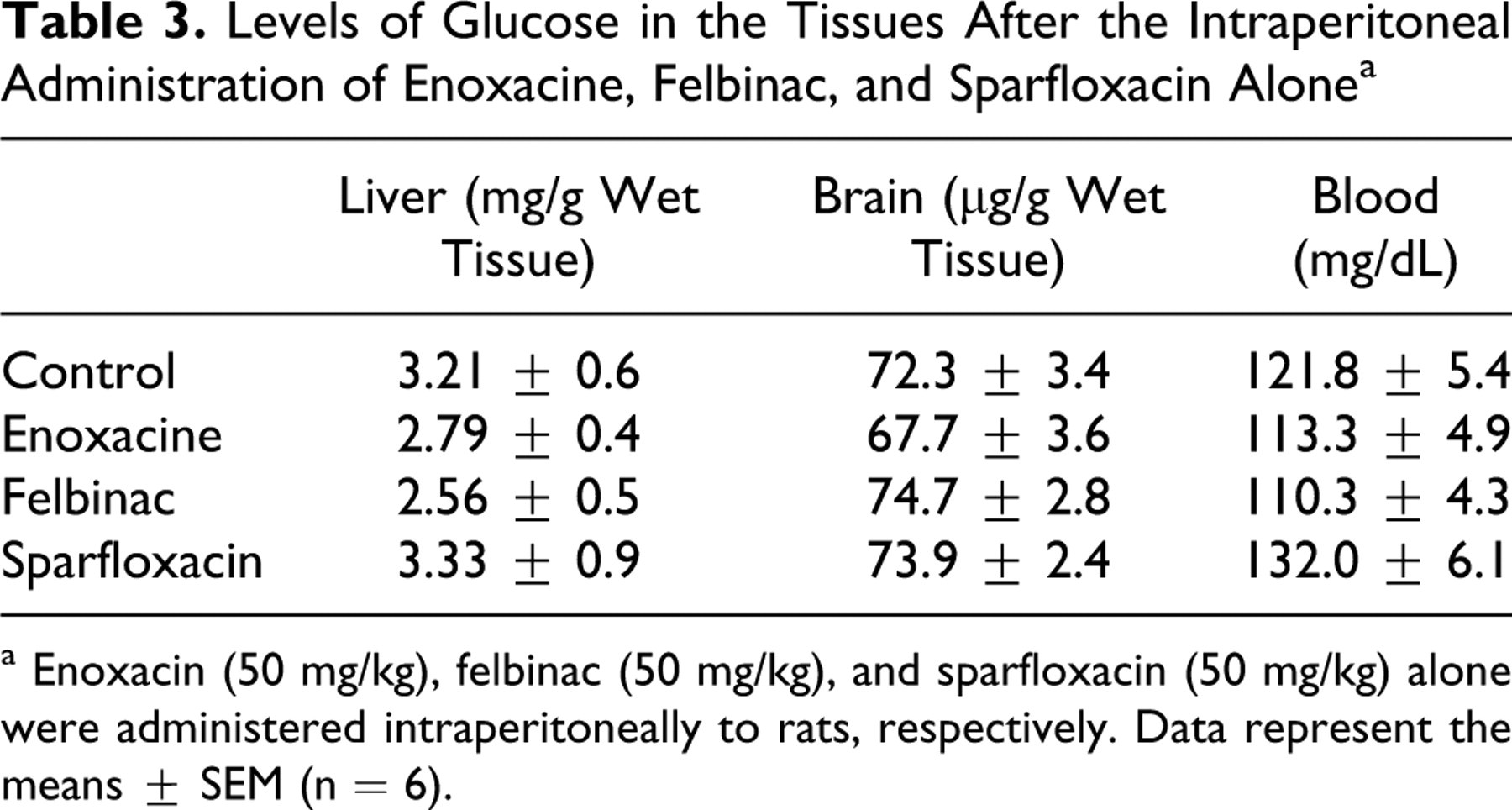
a Enoxacin (50 mg/kg), felbinac (50 mg/kg), and sparfloxacin (50 mg/kg) alone were administered intraperitoneally to rats, respectively. Data represent the means ± SEM (n = 6).
Discussion
The combined application of NQs and NSAIDs has been shown to attenuate ligand-binding affinity at the GABAA receptor. However, the mechanisms of action involved in convulsions are not clearly understood. Multiple changes in metabolic levels may be associated with the drug-induced convulsions. Metabolic signature at the onset of convulsion compared with nonconvulsion may be useful for elucidating physiological toxicity. Then, we examined the effect of enoxacin, felbinac, and sparfloxacin on fatty acid metabolism and glucose levels.
The coadministration of enoxacin with felbinac is known to be the most risky combination, whereas sparfloxacin has very weak convulsion-inducing activity. The levels of acyl-CoAs (C18:1 and C20:4) in the liver and brain at the onset of convulsion induced by the coadministration of enoxacin with felbinac were significantly decreased. The levels of acyl-CoAs in the liver and brain were not significantly reduced with the administration of enoxacin (50 mg/kg) and felbinac (50 mg/kg) alone, respectively. Glucose levels in the liver and blood at the onset of convulsions were significantly decreased, whereas they were significantly increased in the brain. The interaction between enoxacin and felbinac had an effect on both the levels of acyl-CoAs (C18:1 and C20:4) and glucose in the liver and brain at the onset of convulsions. However, sparfloxacin alone, which has weak convulsion-inducing activity, did not exert an influence over the metabolism of fatty acids and glucose.
We have reported that the medium-chain acyl-CoA synthetase purified from mitochondria of mouse liver and kidney, and bovine liver was inhibited by NSAIDs and NQs. 9 –11 Diflunisal, salicylic acid, nalidixic acid, enoxacin, and ofloxacin inhibited the medium-chain acyl-CoA synthetase activity toward hexanoic acid. The inhibitory carboxylic acids did not act as the substrate toward the purified medium-chain acyl-CoA synthetase. The hydroxyl or ketone (oxo) groups on the carbon adjacent to the carboxylic acid group of benzene ring were required for the inhibitory activity of the medium-chain acyl-CoA synthetase, suggesting that there is an interaction (ie, intermolecular hydrogen bond formation) between the ketone group of NQs and the thiol group of this enzyme. 9 –11 Salicylic acid in combination with enoxacin had very weak convulsion-inducing activity. 3 Salicylic acid had strong inhibitory activity (IC50 = 0.1 mmol/L) for the mitochondrial medium-chain acyl-CoA synthetases, whereas it showed very weak inhibition for long-chain acyl-CoA synthetases (IC50 = 25.5 mmol/L). 31 The disturbances of metabolism of long-chain fatty acids may play an important role in the development of convulsions. This study showed that the combination of enoxacin with felbinac also inhibits the enzymes involved in metabolism of long-chain fatty acids. Convulsions may be in some way associated with the production of energy. The amounts of acyl-CoAs of C16:0, C18:1, and C20:4 were more than those of C6:0, C8:0, and C10:0 in the liver and brain of mice and rats. Therefore, salicylic acid may play a role to less extent in the development of convulsions. Yoshino et al have reported that celecoxib did not induce convulsions in combination with enoxacin, lomefloxacin, and ciprofloxacin. 34 Celecoxib is one of the anti-inflammatory agents but is structurally distinct from conventional ones. Celecoxib is classified as a diaryl-substituted pyrazole, whereas the conventional anti-inflammatory agents are as arylacetic acid-derived or propionic acid-derived ones. Long-chain acyl-CoA synthetases activate fatty acids and xenobiotics such as arylacetic acid derivatives to acyl-CoA derivatives. Then, there is the difference in the structure between the celecoxib and the conventional anti-inflammatory agents, suggesting that celecoxib may not exert an influence over fatty acid metabolism. These findings indicate that the combination of enoxacin with felbinac inhibited fatty acid metabolism in the liver and brain at the onset of convulsions.
Glucose levels in the tissues may be closely connected with the disturbance of fatty acid metabolism. In the present study, glucose concentrations in the liver and blood at the onset of convulsions were significantly decreased, whereas they were increased in the brain. Recent reports suggest that glucose alterations (hypoglycemia or hyperglycemia) may occur with fluoroquinolones. 12,13,15,16 The mechanism of hypoglycemia is suggested to be that fluoroquinolones cause the release of insulin through direct inhibition of pancreatic β-cell potassium channel activity. 14 Insulin secretion increased in a dose-dependent manner as the concentrations of fluoroquinolones increased. On the other hand, gatifloxacin and levofloxacin commonly cause hypoglycemia and hyperglycemia, especially in patients with diabetes. 15,16 The mechanism of hyperglycemia is not known, but may be related to the vacuolation of pancreatic β-cells, leading to a decrease in insulin secretion. 17 In the present study, the decrease of blood glucose at the onset of convulsions may be induced by the same mechanism as proposed for fluoroquinolone-induced hypoglycemia. However, brain glucose levels at the onset of convulsions were significantly increased.
The mechanism linking fatty acid metabolism, glucose levels, and convulsion remains unknown. The KD (a high-fat and low-carbohydrate diet) has been employed as an effective treatment for medically refractory epilepsy. Despite nearly a century of use, the anticonvulsant mechanisms of the KD have proven elusive. 8,19 –21,23,24 Although several major hypotheses have been advanced, none has been widely accepted. The collective data indicate that the critical condition necessary for achieving seizure control is a metabolic shift toward fatty acid oxidation from glycolysis, 8,22 –26 which might result in an elevated seizure threshold. 18 Yamada has also reported that inhibiting glycolysis or diverting glucose from the glycolytic pathway inhibits seizures, suggesting an interaction between the energy regulation and the anticonvulsant actions of the KD in experimental seizure models. 23 Ketone bodies (metabolites of fatty acids), fatty acid, and glucose restriction might each lead directly or indirectly to seizure control. 8,22 –26
Bough and Rho have summarized the hypothetical pathways leading to the anticonvulsant effects of the KD, and indicated a variable rise in blood/brain ketone levels and a concomitant reduction in blood/brain glucose as the critical condition necessary for achieving seizure control. 8 Several factors may likely contribute to prevent seizures. As one of the anticonvulsant effects, the reduction in glycolytic energy supply may activate selectively ATP-sensitive potassium (KATP) channels to increase the resistance to onset of ictal activity. 35,36 KATP channels are ligand-gated receptors broadly expressed throughout the central nervous system. 37 When glucose is limited, KATP channels might open to hyperpolarize the cell as the intracellular ATP concentration falls. Conversely, when glucose is present and ATP concentrations rise, KATP channels close. As such, KATP channels may provide a measure of protection against a variety of metabolic stressors, such as hypoxia, ischemia, and hypoglycemia, and are believed to regulate the seizure threshold. 8,38 KATP channels would be ideally positioned to metabolically regulate the onset of several different seizure types. 8 In the present study, the condition observed in the brain at the onset of convulsion was opposite that treated with the KD. Namely, the increase in brain glucose may result in closing KATP channels and would contribute to neuronal/glial cell membrane depolarization. These findings suggest that the glucose rise in the brain may be an increased risk of convulsion intensity. On the pathway leading to anticonvulsant effects, an increase in oxidative phosphorylation coupled with the induction of uncoupling proteins and mitochondrial biogenesis can diminish reactive oxygen species generation and increase energy reserves, both of which would be expected to prevent neuronal dysfunction, seizures, and even neurodegeneration. 8
On the pathways involved in fatty acid metabolism of the KD, elevated free fatty acids were metabolized through the pathway of β-oxidation, leading to chronic ketosis. Chronic ketosis is predicted to lead to increased levels of acetone, which might activate the 2-pore domain of potassium channels to hyperpolarize neurons and limit neuronal excitability. 8 In the present study, the formation of acyl-CoAs from fatty acids was inhibited, suggesting that the decreased levels of acetone might inactivate the 2-pore domain of potassium channels to increase neuronal excitability. Namely, the decrease of acyl-CoA levels in the liver may be an increased risk of convulsion intensity. It is improbable that 1 mechanistic target of the mediator will produce complete seizure protection associated with the KD. Rather, several factors will contribute to the anticonvulsant action. As a result, this study indicates that inhibition of fatty acid metabolism in the liver and brain and the increase in brain glucose may contribute to increase the susceptibility to convulsions induced by the interactions between NSAIDs and NQs.
Kushner et al have suggested that the clinical conditions of patients and concomitant treatment with another drug that may lower the seizure threshold are important risk factors for the development of seizures induced by NQs. 39 On the other hand, a defect in fatty acid metabolism and concomitant limited glucose, in a particular case, have been reported to be very important factors associated with unexplained sudden clinical deterioration. In any clinical situation, such as periods of fasting or metabolic stress due to concurrent illness or infection, fatty acid oxidation is critical in supplying energy. Patients with medium-chain acyl-CoA dehydrogenase deficiency will continue glucose consumption with reduced or absent formation of ketones. The result of this is severe hypoglycemia and hypoketonuria. Fatty acid oxidation defect can result in acute metabolic episodes or sudden death. 40 –42 These findings indicate that metabolic crisis, such as inhibition of fatty acid metabolism and alteration of glucose levels, may develop various clinical symptoms.
We propose the hypothesis that the disturbance of both fatty acid metabolism and glucose levels is caused by fasting or metabolic stress due to infection deteriorates and by the interaction of enoxacin and felbinac, resulting in an increased risk of convulsant activity. These findings may contribute to further understanding of the toxic effects associated with these drugs.
Conclusions
The combination of reduction of acyl-CoAs induced by enoxacin with that by felbinac resulted in the significant decrease of acyl-CoAs. Alterations in glucose levels were observed in the liver, brain, and blood. Inhibition of fatty acid metabolism in the liver and brain and increased brain glucose might be associated with the increased susceptibility to convulsions induced by the interactions of enoxacin and felbinac.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
This work was supported in part by the “Academic Frontier” Project 2006-2010 for Private Universities from the Japanese Ministry of Education.
