Abstract
Hypocrellin B (HB) was encapsulated into a phosphatidylcholine liposome. Encapsulation of HB into liposomes not only improved the delivery of this photosensitizer but also increased its photodynamic efficacy compared to free HB molecules. Liposomal HB showed a higher cellular uptake than free HB as measured by confocal microscopy and was internalized into cultured HeLa cells by caveolar endocytosis, which was lipid-raft-dependent. Cell viability measurements demonstrated that liposomal HB was more phototoxic to HeLa cells than free HB as a result of the higher concentration of intracellular HB delivered by the liposomal formulation. The encapsulation of HB influenced the cell death pathway by an increased rate of necrotic cells after irradiation versus free HB, and a Type II (singlet oxygen) mechanism was responsible for the photocytotoxicity.
Photodynamic therapy (PDT) is an innovative modality for the treatment of small and superficial tumors. 1 Photodynamic therapy, as a multimodality treatment procedure, requires both a selective photosensitizer and a light source that matches the absorption spectrum of the photosensitizer. Hypocrellin B (HB), a photosensitizer isolated from natural fungus sacs of Hypocrella bambusae in China, has been employed in pioneering PDT applications owing to its high quantum yield of singlet oxygen generation. 2–6 It also exhibits several advantages over the presently used hematoporphyrin derivatives (HPD) in PDT, that is easy preparation and purification as well as red light absorbance and significantly reduced normal tissue photosensitivity because of the fast metabolism in vivo. 7,8 Under physiological conditions, HB, a lipophilic compound, is essentially insoluble and easily aggregates in water, which not only complicates the normal administration and biodistribution of this drug in vivo but also dramatically reduces its photodynamic activity. This may be a major limiting factor for its potential clinical application.
To solve this problem, some water-soluble derivatives of hypocrellin B were prepared by chemical modifications on the molecular structures. 9 –13 However, the water-soluble derivatives commonly show very poor photodynamic activity in vivo because of the low cellular uptake.
Liposomal delivery of photosensitizers may overcome or reduce these problems. 14,15 It reported that HB might become water-soluble by forming nanometer-sized liposome, 5 and the formation of active oxygen species (1O2, O2 ·−, and .OH) and semiquinone anion radical (HB·−) from liposomal HB upon photoexcitation has been measured in detail. 6 However, the phototoxicity of liposomal HB to the tumor cells has never been reported.
In this present work, encapsulation of HB into liposomes was studied against free HB with regard to the improvement of delivery of the HB into the cells, and the concomitant phototoxicity of liposomal and free HB to HeLa cells was assessed in vitro.
Materials and Methods
Chemicals and Materials
Hypocrellin B was obtained by dehydrogenation of HA. 16 Phosphatidylcholine (PC), cholesterol, dimethyl sulfoxide (DMSO), 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyl-2H-tetrazolium bromide (MTT), paraformaldehyde (PFA), Trypan blue (TB), and propidium iodide (PI) were all purchased from Sigma-Aldrich, Inc (St Louis, Missouri). Dulbecco's modified minimum essential medium (DMEM) and fetal bovine serum (FBS) were obtained from Gibco-BRL (Grand Island, New York). Trypsin-EDTA was from Gibco Invitrogen Corporation (Carlsbad, California). Methyl-β-cyclodextrin (MBCD) was purchased from Sigma-Aldrich.
Stock HB-DMSO solution for biological study of free HB was prepared by dissolving the dye in DMSO to 1 mmol/L. The stock solution was stored at 4°C in dark. Known volumes of stock were diluted in PBS or DMEM cell culture medium to achieve the desired experimental dose. The concentration of DMSO in contact with the cells, both as a vehicle for dyes and as a control, did not exceed 1% (v/v), a concentration that has been demonstrated to have no effect on cell viability. 17
Other regents were all of analytical grade. The water used was purified by triple distillation. The working solutions were prepared immediately before use.
Preparation and Characterization of Liposome Suspensions
Liposomes were prepared following the procedures reported before. 18 In a typical experiment for preparing 5 mL dispersion of liposomes, 22 mg of PC and 3.8 mg of cholesterol (molar ratio PC: CHOL 3:1) were dissolved in chloroform/methanol (6:1, v/v) for the formation of lipid films, while for the preparation of the liposomes containing HB, 1.4 mg (2.5 μmol) of HB was added to the above lipid mix solution. After evaporation of the solvent, the thin lipid film was hydrated with 5 mL of PBS buffer (pH 7.4). The resulting suspension was then sonicated for 60 minutes at above the phase transition temperature. The liposome suspension was then passed through 2 stacked polycarbonate membrane filters with 0.22 μm pores, and a homogeneous liposome suspension was thus obtained. Removal of nonencapsulated HB was achieved on a Sephadex G25 column (Pharmacia, Peapack, New Jersey). The final encapsulation rate of HB in the suspension was determined by HPLC (Agilent 1200). The particle sizes of the liposomes were measured with a Microtrac Nanotrac 150 particle size analyzer (Microtrac Inc., Florida, America). The liposomal HB particles were also characterized by fluorescence scanning confocal microscopy using a Leica TCS-SP2 microscope with a 488 nm excitation laser. Stock suspensions of liposomal HB prepared as described were transparent, with no visible signs of sedimentation, even after 4 weeks of storage at 4°C. The elution was performed with FBS-free DMEM solution, with no effect on the stability and the particle size of the suspension.
Cell Cultures
HeLa cells were maintained as monolayer culture in DMEM containing 10% FBS 80 U/mL penicillin and 100 μg/mL streptomycin at 37°C in a humidified 5% CO2 incubator. Cells were seeded in new medium every 2 days using 0.25% trypsin–EDTA.
Cell Uptake of HB
HeLa cells were cultured in sterile dishes with coverslips for 12 hours, then medium was removed and cells were exposed to liposomal or free HB (50 μmol/L) for 0 minute, 20 minutes, 40 minutes, and 60 minutes in serum-free medium at 37°C. Afterward, cells were washed twice with PBS gently and the fluorescence image of intracellular HB was registered by laser confocal scanning microscopy at 488 nm excitation wavelength.
To study the uptake kinetics, the intracellular HB fluorescence intensity was analyzed using image analysis software (Leica TCS SP2, Ver. 1.6.582) of Leica Confocal Microscope Systems.
To investigate the cell uptake processing, cells were incubated with or without MBCD (5 mmol/L) at 37°C for 30 minutes, then exposed to liposomal or free HB in serum-free medium at 37°C for 60 minutes. The fluorescence intensity of intracellular HB was measured by laser confocal scanning microscopy.
Cell Survival Studies
For in vitro PDT tests, cells were cultured in 96-well plate for 12 hours, and then were exposed in the dark for 1 hour at 37°C to certain concentration of liposomal or free HB in DMEM (FBS free). After incubation, the medium was replaced, and the cells were then irradiated with a Red Light Treatment Instrument (Institute of Electronics, Academia Sinica, China), which total power output, more than 90% at 500 to 700 nm, was 47 mW cm-2 at the position of the samples as measured with a SPR-4001 Spectroradiometer (Luzchem Research Inc., Ottawa Ontario, Canada). An irradiation time of 1 minute led to a total fluence of 2.8 J/cm2. After exposure, FBS was added to the DMEM medium with a final concentration of 10%. Cell viability was determined by adding 10 μL MTT (10 mg/mL in stock) to each well and the mixture was incubated for 4 hours at 37°C. Culture medium was then replaced with dimethylsulfoxide to dissolve the formazan crystals. The plates were shaken at room temperature for 10 minutes and read immediately at 620 nm on a Bio-Rad model 550 microplate reader (Richmond, California). Samples were measured in 12 replicates and each experiment was repeated twice.
Trypan Blue and Propidium Iodide Staining
After incubated in DMEM with 10% FBS for 24 hours at 37°C, both treated and control cells were stained with trypan blue (0.4%), and then fixed with 4% paraformaldehyde (PFA) for 1 hour at room temperature. The fixed cells were washed twice in PBS, and permeabilized at 4°C for 5 minutes with 0.1% Triton X-100, then stained for 10 minutes at room temperature with PI (5 μg/mL). Cells were washed again and observed with the fluorescence microscope (Olympus Inverted Microscope IX71) equipped with software (IMAGE-PRO MAC 5.1). Pictures of 4 random fields were taken. Cells with bright condensed chromatin and fragmented nuclei were counted as apoptotic cells. 19,20 Cells with normal-shaped nuclei and stained by trypan blue were counted as necrotic. At least 500 cells were scored for each sample. The results shown above are the mean of 3 independent experiments.
Statistical Analysis
All data were expressed as mean ± SD (standard deviation); for comparisons of the means, we used Student t-test. P values less than .05 were considered statistically significant.
Results
Characteristics of Liposomal HB
The particle sizes of the liposome entrapping HB in FBS-free DMEM solution were mainly 170 nm, which were measured by Microtrac NanotracTM150 particle size analyzer. The encapsulation rate of HB in liposomes was about 48.2% ± 8.6% measured by HPLC. Therefore, the final concentration of HB in stocking liposomal formulation is 48.2 ± 8.6 μmol/L based on the encapsulation rate and the original mol of HB added for the preparation of liposomal HB.
Laser confocal scanning microscopy was used to investigate the actual distribution of HB in liposomes. As shown in Figure 1 , the fluorescence was only detected from hydrophobic domains of the liposome, defined strictly by the phospholipid boundary. It indicated that HB was located in the liposome phospholipid bilayer. The fluorescence images were obtained by focusing on the bigger particles for a better visualization.

Confocal microscopy fluorescence images of phosphatidylcholine (PC) liposomal Hypocrellin B (HB), using excitation laser of λex = 488 nm.
Cellular Uptake of HB
After incubation with liposomal or free HB for 1 hour and upon excitation at 488 nm, the HeLa cells in both cases showed intracellular fluorescence indicating substantial uptake of HB (Figure. 2 ). The cells incubated with liposomal HB obtained higher intracellular fluorescence intensity than the cells incubated with free HB (Figures 2 and 3 ). The fluorescence of HB in both cases was seen mainly in cytoplasm, and in no case fluorescence was nucleus-localized.

Confocal microscopy fluorescence images of uptake of Hypocrellin B (HB) in HeLa cells. A, Incubation with free HB (50 μmol/L) for 1 hour. B, Incubation with liposomal HB (stocking suspension) for 1 hour.
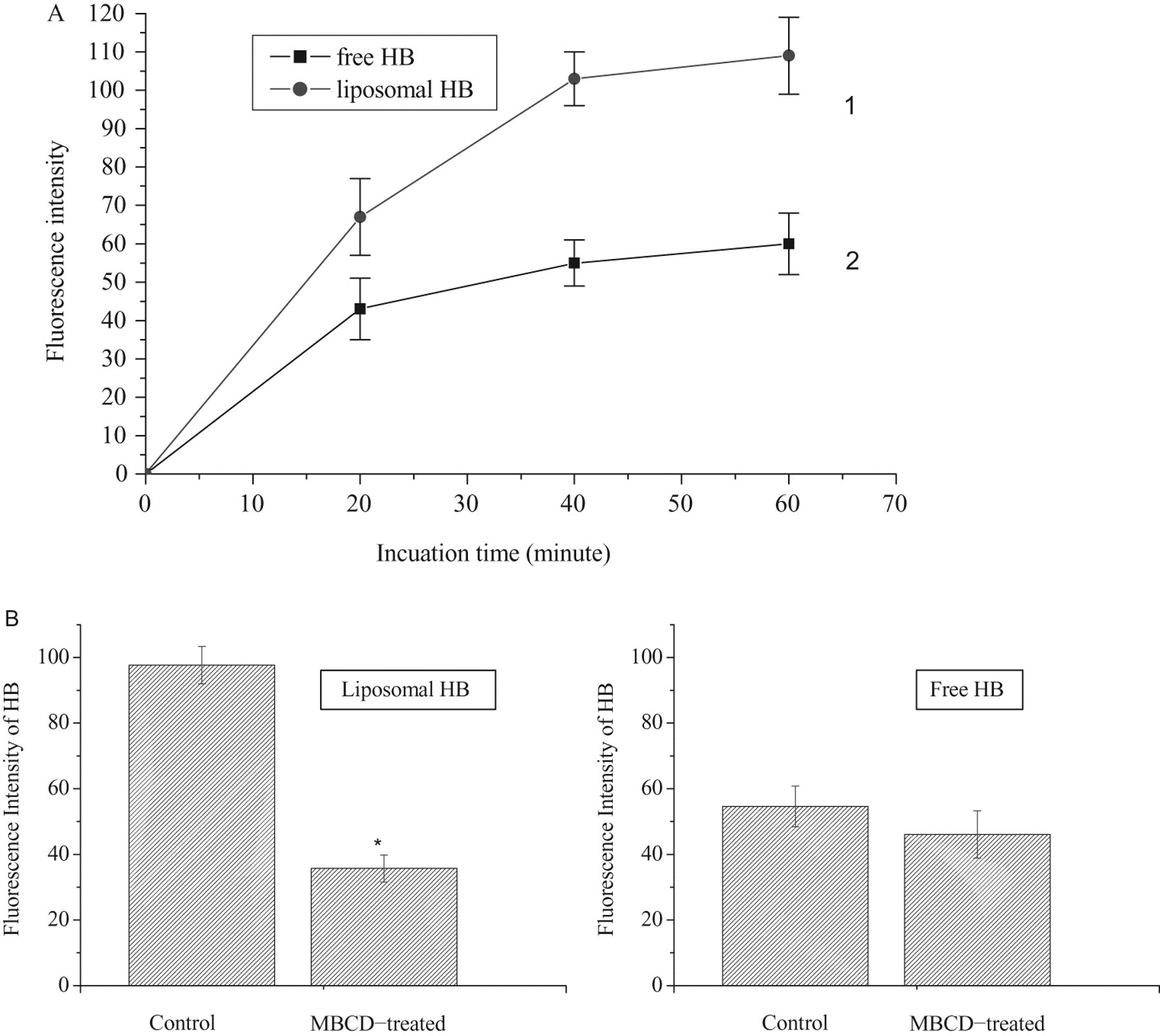
The fluorescence intensity of intracellular Hypocrellin B (HB). A, Uptake kinetics of liposomal HB (stocking suspension) (1) and free (2) HB (50 μmol/L) by HeLa cells. B, Effect of methyl-β-cyclodextrin (MBCD) treatment on the cellular uptake of liposomal HB (stocking suspension) and free HB (50 μmol/L). At least 200 cells were scored for each sample. Bars represent the standard deviation of 3 experiments. *P < .05 versus control (no MDCB treated).
The cellular uptake kinetics of liposomal or free HB versus incubation time, studied through fluorescence intensity, is presented in Figure 3A. Cellular uptake of HB increased and reached a plateau after 40 minutes and 20 minutes of incubation with liposomal and free HB, respectively. However, greater rate of uptake was measured for liposomal HB group, which reached the higher intracellular concentrations at all incubation times than those for free HB group. Thus, it was noticed that the cellular uptake of the HB in HeLa cells was strictly dependent on the means of photosensitizer delivery.
Because initial observations of rates of uptake as well as intracellular distribution suggested comparable behavior for liposomal and free HB (Figures 2 and 3A), we investigated the liposomal and free HB for detailed analysis of uptake mechanisms. HeLa cells pretreated with MBCD, an inhibitor of endocytosis, showed significantly reduced uptake of liposomal HB, but no effect on the uptake of free HB (Figure 3B). This suggested liposomal HB was internalized into cultured HeLa cells by endocytosis, but free HB might be through other processes, such as adsorption and plasma membrane fusion. 7
Survival Rate of HeLa Cells Induced by PDT
The photodynamic activity of liposomal and free HB toward HeLa cells was measured using the MTT assay. Figure 4A and B shows the characteristics of free and liposomal HB phototoxicity to the cells after exposure to 4 doses of light irradiation, respectively. Cell viability was measured 24 hours postirradiation in HeLa cells. When the cells were exposed to light in the absence of the photosensitizer, no toxicity was recorded (Figure 4A1 and B1). Only negligible proliferative change was observed when the treatment with photosensitizer was conducted in the absence of light (data not presented). This suggested that light, liposomes or photosensitiser alone was negligibly toxic in this treatment. When incubating cells with a certain concentration of the studied agents and irradiating them with various energy doses cells, cell survival was dependent on the dose of energy used and significantly decreased when the light dose was raised in both groups (Figure 4A and B). We have calculated the LD50, that is, the light dose, required for killing 50% of the cells at the given agent concentration. 21 Given a 5 μmol/L of culture photosensitizer, the LD50 of liposomal HB treatment was about 3 J/cm2 (Figure 4B2), while that of free HB treatment was more than 5 J/cm2 (Figure 4A2). Apparently, liposome encapsulation increases the concentration of HB in the cells, hence more cells are killed upon a particular light dose.
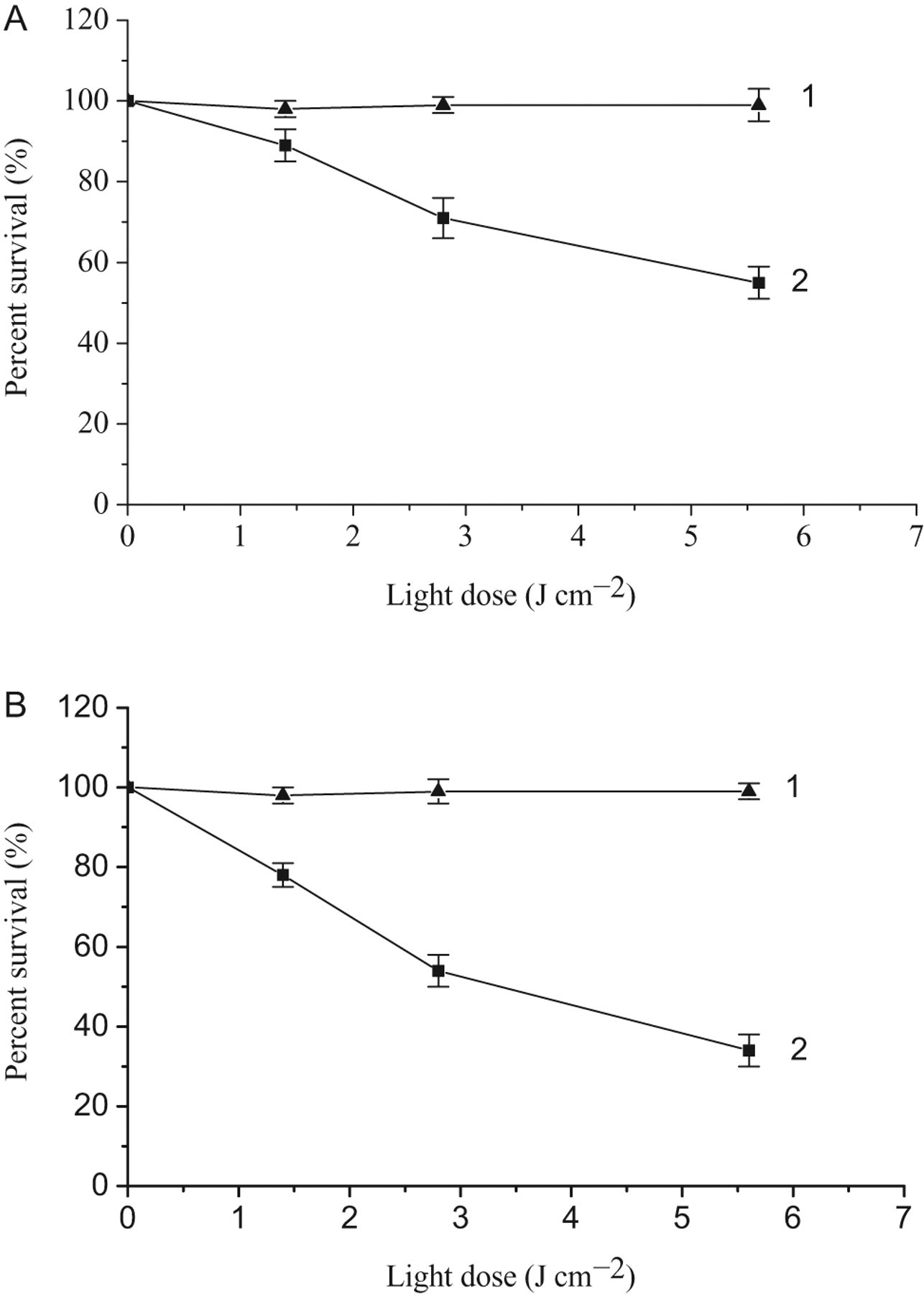
Phototoxicity of free and liposomal HB to the HeLa cells. A: (1) incubation with 0 μmol/L HB for 1 h; (2) incubation with 5 μmol/L Hypocrellin B (HB) for 1 hour; B, (1) incubation with liposomes containing 0 μmol/L HB for 1 h; 2, incubation with liposomal HB (1:10 dilution of stocking suspension) for 1 hour. Representative results were shown as the mean ± SD from 2 independent experiments.
Cell Death Determination
The phototoxic effect of the studied HB to HeLa cells was further determined assessing cellular death mechanisms, apoptosis, and necrosis. Figure 5 shows the evaluation of necrotic and apoptotic percentages after sensitization with liposomal or free HB followed by irradiation. Both the percentage of total dead cells and the ratio of necrosis versus apoptosis photoinduced by the liposomal HB were much higher than those of free HB (P < .05; Figure 5 A and C). Both the increased cytotoxicity and the increased fraction of necrosis seen in cells treated with the liposomal formulation are likely related to the increased concentration of HB in the cells. It reports that singlet oxygen (1O2) is a highly reactive oxygen species and believed to play a key role in the efficacy of PDT. 4 To confirm the generation of 1O2, we used the singlet oxygen quencher NaN3 to pretreat the cells, and the results showed a significant protection against the liposomal and free HB-induced cell killing on light exposure (Figure 5 B and D, P < .05).
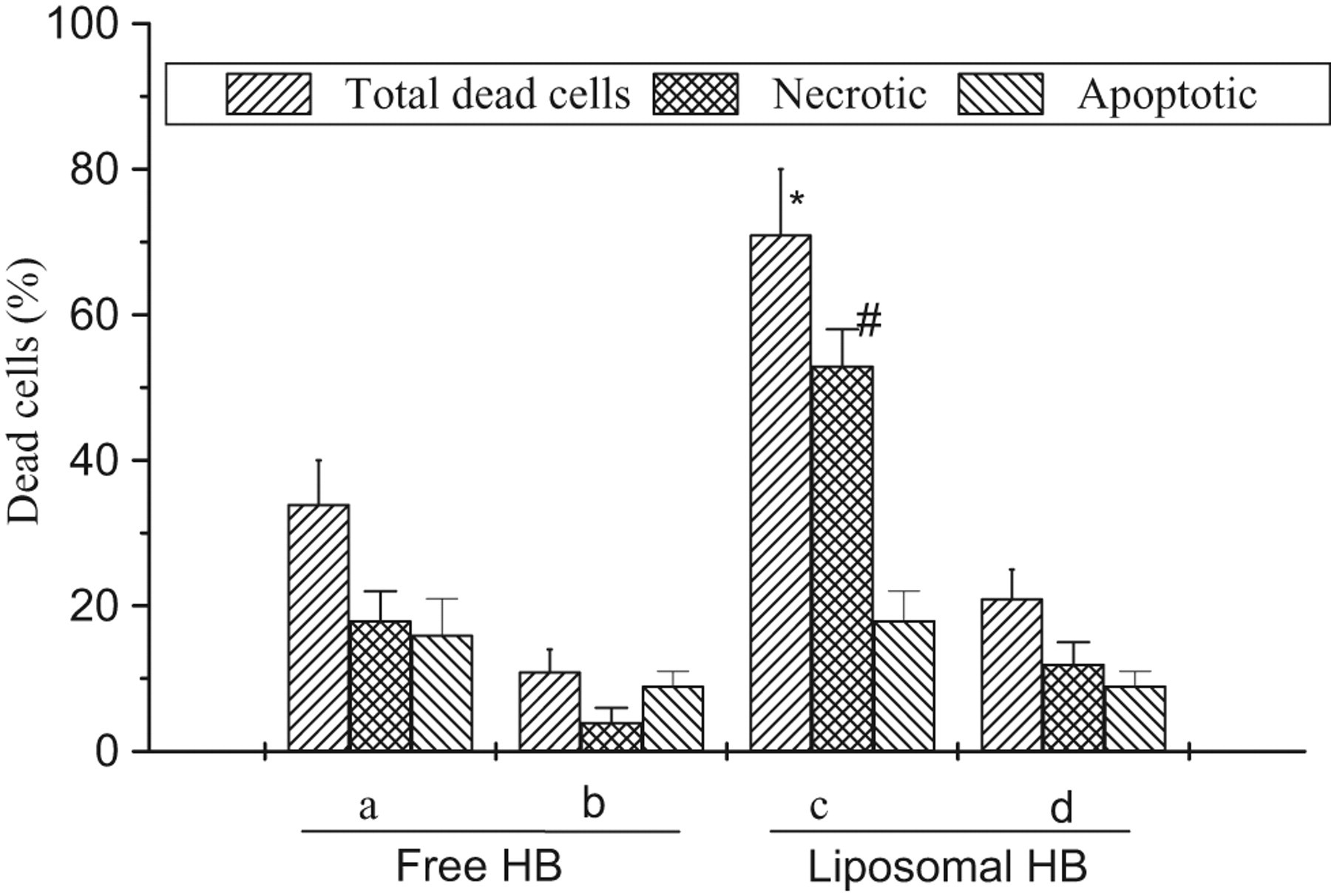
Free and liposomal Hypocrellin B (HB) induced apoptotic and necrotic death in HeLa cells after irradiation. Cells were incubated with free HB (5 μmol/L) (A) or liposomal HB (1:10 dilution of stocking suspension) (c) for 1 hour, followed by irradiation dose of 5.6 J/cm2, the cells were incubated overnight in cell culture medium, then stained with Trypan blue and propidium iodide (PI). The cell percentage was calculated through cell counting. At least 300 cells were scored for each sample. For NaN3 protection assay (b and d), cells were incubated with 20 mmol/L NaN3 during the incubation period as well as the irradiation period. Bars represent the standard deviation of 2 independent experiments. * and # P < .05 versus free HB group.
Discussion
It is reported that the hydrophobic HB might become water-soluble by encapsulation into liposome particles. 5 The aim of the present study was to investigate whether or not the incorporation of hydrophobic HB into liposomes enhanced the therapeutic efficacy of PDT.
In this work, we formulated the stable liposomes containing the hydrophobic HB with a diameter of around 170 nm. The encapsulation of HB into liposomes caused a broadened and redshift of the optical spectrum compared to the free HB (data not shown). The phenomena indicated that the reduction of HB molecular movement and the enhancement of intramolecular proton transfer were owing to the association of liposomes, as observed before. 22,23 Hypocrellin B dye appeared uniformly distributed in hydrophobic domains of the liposome, defined strictly by the phospholipid boundary (Figure 1). The results suggest that liposomes represent suitable carriers of hydrophobic HB because lipid vesicles induce a uniform distribution of this photosensitizer.
The efficiency of PDT treatment is highly dependent on the photosensitizer cellular uptake and subcellular localization. 24,25 In the current studies, the cellular uptake of the HB encapsulated in liposomes was evaluated using a HeLa cell line. It was compared with results obtained for free HB. The encapsulation of HB into liposomes enhanced the fluorescence of HB in the cytoplasm, a faster kinetics of cellular uptake and almost a 2 times higher intensity plateau were found after 1 hour incubation of liposomal HB with respect to that of free HB (Figure 3). This suggests a more efficient uptake mechanism for liposomal HB than that for free HB. Endocytosis, especially caveolar endocytosis pathway, is important for efficiently modulating uptake and cellular sorting of extracellular particulates such as nanoparticle diagnostic probes or delivery devices, into the intracellular membrane system. 26 Methyl-β-cyclodextrin, an inhibitor of endocytosis, can deplete cholesterol from the cell membranes effectively inhibiting diverse cholesterol-dependent endocytic routes including caveolar endocytosis, 27 and in some cases, even clathrin-mediated endocytosis. 28 HeLa cells pretreated with MBCD showed little effect on the cellular uptake of free HB but significantly reduced uptake of liposomal HB (Figure 3B). This finding suggests liposomal HB uptake is mainly dependent on caveolar endocytosis, which leads to a higher intracellular concentration of photosensitizer than the adsorption or membrane fusion process of free HB.
Our work has focused on assessment of internalization of the liposomal and free HB, but an equally important question that remains to be addressed is the postendocytic processing including the intracellular trafficking of endosome, release of photosensitizer from intracellular lysosomal compartments, and relocalization of the photosensitizer.
The increased cellular uptake of encapsulated HB versus free HB is reflected in greater photoinduced toxicity of liposomal HB than free HB toward HeLa cells as measured by the MTT assay (cell viability; Figure 4). Photodynamic therapy is known to elicit both necrosis and apoptosis, depending on the light/drug dose, cell type, and the nature of the photosensitizer used. 25,29,30 Under the optimized experimental protocol conditions used to elicit cell death, both apoptosis and necrosis induced by PDT were identified in this experiment (Figure 5). However, liposomal HB-mediated PDT caused both higher percentage of total dead cells and larger ratio of necrosis to apoptosis than free HB (Figure 5A and C). The higher percentage of necrotic cells after higher PDT doses was reported by various authors and was found to be accompanied by inhibition of the apoptotic pathway. 31,32
It is generally accepted that the singlet oxygen mechanism (photochemical reaction of type II) predominates during PDT and that singlet oxygen is the most important cytotoxic species produced by HB. 2,4 The decrease of phototoxic damage in the presence of the singlet oxygen quencher NaN3 (Figure 5B and D) confirmed the involvement of singlet oxygen, and the results suggested an enhancement of necrotic cells to the detriment of apoptotic ones is correlated with a higher concentration of reactive oxygen species (singlet oxygen) photoinduced by higher intracellular concentration of photosensitizer in liposomal HB group than that in free HB group during PDT as shown in Figure 3.
Conclusion
In summary, in the present work, we have reported the preparation and photodynamic efficacy of liposomal HB toward HeLa cells. The results showed that the liposomes not only imparted solubility to the hydrophobic HB in aqueous solution with less aggregation but also transported HB into cells efficiently and improved its photocytotoxicity significantly.
Footnotes
Acknowledgement
This work was supported by the Beijing Talents Training Project (20081D0501500180) and by the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
