Abstract
In order to elucidate the mechanism of cytotoxicity photoinduced by 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B (EAHB), a derivative of hypocrellin B (HB), cellular uptake, subcellular localization as well as photodynamic therapy (PDT) efficiency of EAHB, and cell apoptosis photoinduced by EAHB were investigated in HeLa cells by laser confocal fluorescence microscopy, 3-(4,5-Dimethylthiazol-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide assay, flow cytometry, DNA fragmentation on agarose gel, and Western blot. The results showed EAHB was distributed throughout the cytoplasm of the cell, with no detectable penetration into the nucleus. The proportion of dead cells increased with increases in both the dosage of light and the concentration of EAHB. Its phototoxicity to HeLa cells proceeded via apoptosis. The EAHB-PDT treatment induced a cytochrome c release from the mitochondria into the cytosol followed by the activation of both caspase 3 and caspase 9 in HeLa cells. The results suggested that EAHB-PDT treatment induced apoptosis in HeLa cells, and the cellular apoptosis involved a mitochondria-/caspase-dependent mechanism.
Keywords
Photodynamic therapy (PDT) is a medical treatment that employs a combination of long-wavelength red light (600-800 nm) with a photosensitizing agent to produce reactive oxygen species and consequently leads to cell death and tissue devastation. 1 The use of PDT as a cancer therapy is particularly attractive owing to its fundamental specificity and selectivity. 2,3
Generally, the properties for an ideal photosensitizer include chemical purity, high quantum yield of singlet oxygen production, significant absorption in the long wavelength region (600-800 nm), minimal dark toxicity, and delayed phototoxicity. To date, a large number of photosensitizers have been tested in vivo and in vitro in PDT experiments. 4 According to previous studies, hypocrellins, owing to their easy preparation, purification, facility for site-directed chemical modification, high quantum yields of singlet oxygen, and fast metabolism in vivo, have been selected as potential photosensitizers for PDT. 5 –8 In particular, many derivatives of the parent hypocrellin B (HB) with strong absorption in the phototherapeutic window (600-800 nm), and with low dark toxicity, have been synthesized and studied. 9 –11 Some of them have shown promising anticancer properties. 12,13 Previously, we have screened another derivative of HB and proved that 2-ethanolamino-2-demethoxy-17-ethanolimino-HB (EAHB) exhibits a strong absorption shoulder peak at 632 nm with a high photopotentiation factor for MGC 803 cells. 14 However, mechanisms of cell death following photosensitization are poorly defined. Here we will further report the phototoxicity of EAHB to HeLa cells and the mechanism of the action.
The PDT is able to induce diverse cellular responses, including apoptosis, in which cells undergo characteristic biochemical and morphologic changes. 15 One pathway to trigger apoptosis is through the release of cytochrome c, a mitochondrial component, due to alterations in mitochondrial membrane potential. 16 The release of cytochrome c further activates caspases (a class of cysteine proteases), which then execute the cellular apoptosis. 17 Therefore, in the present study, we investigated the apoptotic induction of EAHB-mediated PDT and the relationship between the death mechanism and the mitochondria-/caspase-dependent apoptosis in HeLa cells.
Materials and Methods
Chemicals and Materials
The EAHB (Figure 1) was previously synthesized and purified as reported, 14 and the purities of photosensitizers assessed by high-performance liquid chromatography were higher than 95%. For the in vitro cellular experiments, the photosensitizers were dissolved in dimethyl sulfoxide (DMSO) at a concentration of 5.0 mmol/L and stored at −20°C in the dark. Immediately before use, they were diluted with culture medium to the desired concentrations. The concentration of DMSO in contact with the cells, both as a vehicle for dyes and as a control, did not exceed 1% (v/v), a concentration that has been demonstrated to have no effect on cell viability. 18 The photosensitizer and cells incubated with photosensitizer were protected from light at all times except for the time of planned exposure.
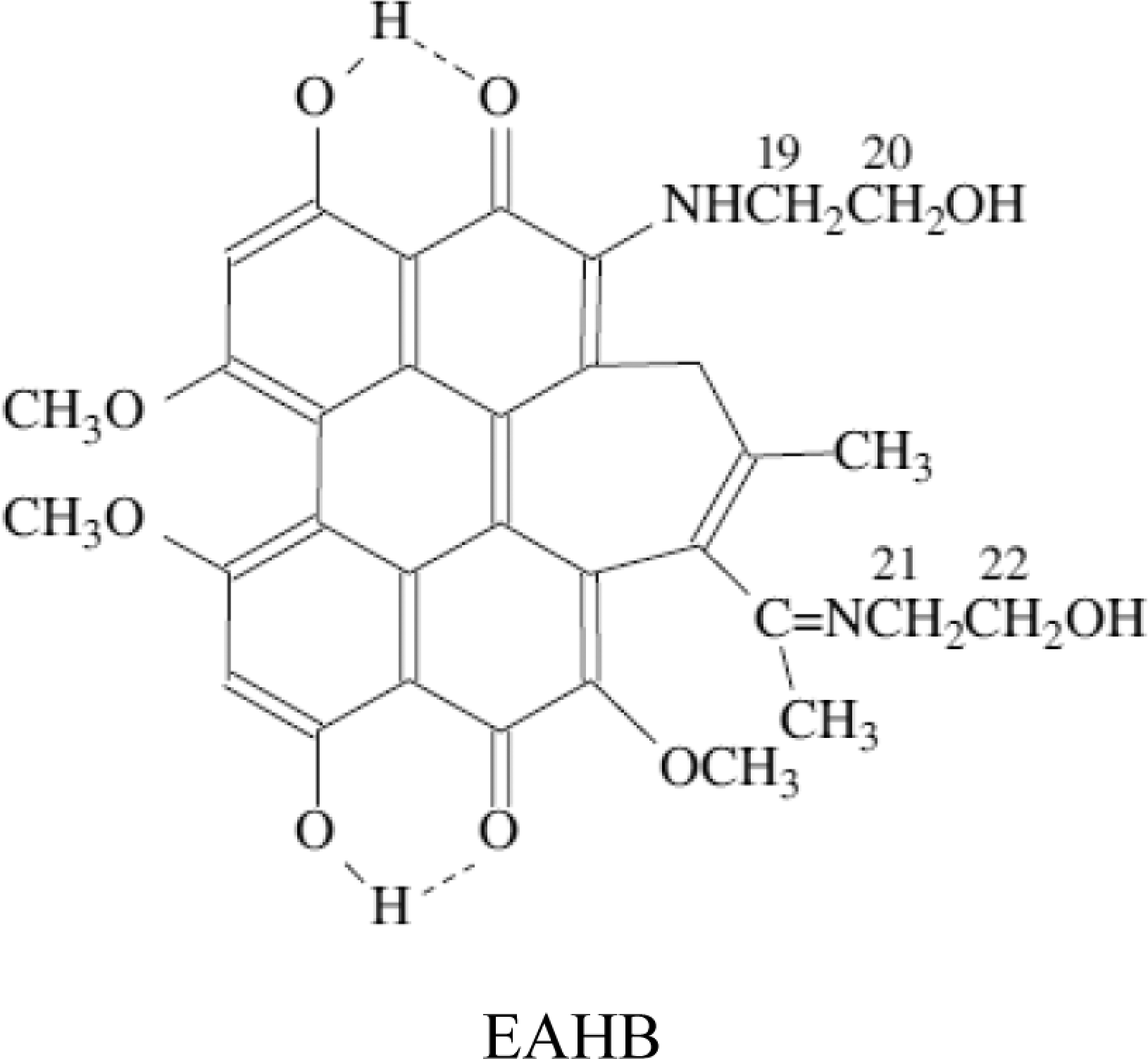
The structure of 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B (EAHB).
Dulbecco-modified minimum essential medium (DMEM) and fetal bovine serum (FBS) were obtained from Gibco-BRL (Grand Island, New York). Trypsin-EDTA was from Gibco Invitrogen Corporation (Carlsbad, California). The 3-(4,5-Dimethylthiazol-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT), DMSO, and propidium iodide (PI) were all purchased from Sigma-Aldrich, Inc (St Louis, Missouri). Primary antibodies against cytochrome c (SC-81753), activated caspase 3 (SC-22171), activated caspase 9 (SC-22182),and β-actin (SC-47778) were purchased from Santa Cruz (California). Anti-mouse immunoglobulin G (IgG; H&L; donkey) antibody IRDye800-conjugated and anti-goat IgG (H&L; donkey) antibody IRDye800CW-conjugated secondary antibodies were from Rockland Immunochemicals, Inc (Pennsylvania). Other reagents were of analytical grade.
Cell Cultures
HeLa cells were maintained as a monolayer culture in DMEM containing 10% FBS and antibiotics PS (80 U/mL penicillin and 100 μg/mL streptomycin) at 37°C in 5% CO2 in a humidified incubator.
Cell Uptake and Intracellular Localization
Cells were cultured in sterile dishes on coverslips for 12 hours, then the medium was removed and the cells were exposed to EAHB (5 and 10 μmol/L) for various time intervals (0.5, 1, 2, and 4 hours) in serum-free medium at 37°C in the dark. Afterward, the cells were washed twice with phosphate-buffered saline (PBS) gently and the real-time fluorescence image of intracellular EAHB was registered by laser confocal scanning microscopy at an excitation wavelength of 543 nm. The intracellular fluorescence intensity was analyzed using image analysis software (Leica TCS SP2, Ver. 1.6.582) from Leica Confocal Microscope Systems.
Incubation and Irradiation
Stock EAHB solutions were diluted with FBS-free DMEM medium. Exponentially growing HeLa cells in 75 cm2 flasks (Costar, Cambridge, Massachusetts) were incubated with or without EAHB at various concentrations in DMEM medium (FBS-free) for 1 hour at 37°C. The cells were detached, centrifuge washed to remove unbound photosensitizer, and then transferred to 35-mm dishes (Costar), with 1 mL FBS-free and phenol red-free DMEM medium containing 3 × 105 cells. They were then irradiated with different doses of light. The light source was a Red Light Treatment Instrument (Institute of Electronics, Academia Sinica, China). Its total power output was more than 90% at 600 to 700 nm as measured using a SPR-4001 Spectroradiometer (Luzchem Research Inc, Ottawa Ontario, Canada). The irradiance was 47 mW/cm2 at the surface of the sample. An irradiation time of 1 minute led to a total fluence of 2.8 J/cm2. Immediately after irradiation, 2 mL DMEM medium containing 15% FBS was added to each dish. Then a portion of the cells was used for the assay of viability and the remaining cells were incubated in the medium prior to the use for DNA fragmentation assay, nuclear staining, flow cytometry analysis, mitochondrial transmembrane potential assay, mitochondrial cytochrome c release, and caspase activity measurement.
Cell Survival Studies
Treated and nontreated control cells (104) were transferred into flat-bottomed 96-well plates (Costar) and kept in the incubator for 24 hours. Cell viability was determined by adding 10 μL MTT (10 mg/mL in stock) to each well and incubated for a further 4 hours at 37°C. Culture medium was then replaced with DMSO to dissolve the formazan crystals. The plates were shaken at room temperature for 10 minutes and read immediately at 595 nm on a Bio-Rad model 550 microplate reader (Richmond, California). Samples were measured in 12 replicates and each experiment was repeated 3 times. Survival of PDT-treated cells was normalized against cells incubated with photosensitizers alone.
Flow Cytometric Analysis of DNA Fragmentation
A PI staining technique was used to assess the status of cellular DNA by flow cytometry with a Coulter Elite ESP Cytometer (Beckman Coulter, Brea, California). Histograms were analyzed with the Coulter Elite 4.5 MultiCycle instrument. The PDT treated and control cells were collected and washed twice with ice-cold PBS, and then fixed in 70% ethanol at 4°C for 24 hours. Cells were washed twice with PBS before resuspension in 1 mL of PBS containing 50 μg/mL PI and 100 μg/mL RNase A, and incubated for 1 hour at 37°C in the dark. Results are expressed as the percentage of cells exhibiting subdiploid (<2 N) amounts of DNA relative to the total number of cells analyzed.
DNA Gel Electrophoresis
DNA from treated and nontreated control cells was extracted using a DNA ladder kit (Applygen Technologies Inc, Beijing, China), according to the manufacturer’s instructions and the DNA ladder was visualized by electrophoresis on 1.5% agarose gel with ethidium bromide (0.5 μg/mL) staining.
Identification of Apoptotic Nuclei
Treated and control cells growing in 6-well plate were incubated in DMEM with 10% FBS for further 24 hours at 37°C in the dark, then Hoechst 33258 at final concentrations of 0.5 μg/mL, was added to the culture medium and incubated for another 30 minutes. After that, the cells were gently washed 3 times with PBS. The fluorescence image of nuclear morphology in a single cell was viewed under a Zaiss Axio Observer.A1 microscope. The excitation wavelength was 350 to 370 nm with emission monitored at 462 nm. Nuclei of both live and dead cells exhibit fluorescence; apoptotic cells can be identified by the appearance of bright fluorescent regions of nuclear condensation or fragmentation. 19
Mitochondrial Transmembrane Potential Assay
Treated and control cells were incubated in DMEM with 10% FBS for 12 hours at 37°C in the dark and stained with MitoCapture (Biovision, Mountain View, California), according to the supplier’s instructions. Briefly, cells were incubated with the MitoCapture reagent for 15 minutes at 37°C and visualized with laser confocal fluorescence microscopy. The fluorescent signal was detected by fluorescence microscopy using a band-pass filter (detects fluorescein isothiocyanate and rhodamine). MitoCapture that has aggregated in the mitochondria of healthy cells fluoresces red. In apoptotic cells, MitoCapture cannot accumulate in mitochondria, it remains as monomers in the cytoplasm and fluoresces green. 20
Preparation of the Cytoplasmic Fraction
Cytoplasmic protein was collected using a mitochondria/cytosol fractionation kit (BioVision), in accordance with the manufacturer’s instructions. Briefly, treated and control cells (5 × 106) were harvested and washed twice with ice-cold PBS, then incubated in 1.0 mL of cytosol extraction buffer mix containing dithiothreitol and protease inhibitors. After incubation with ice for 10 minutes, the cells were homogenized in an ice-cold dounce tissue grinder (BioVision). The homogenate was centrifuged at 800g for 10 minutes at 4°C and the supernatant was centrifuged at 10 000g for 30 minutes at 4°C. Then, the supernatant was collected as the cytosolic fraction. The protein concentration of the cytoplasm was measured by the bicinchoninic acid method using BSA as a standard.
Western Blot Analysis
Cytoplasmic fractions containing equal amounts of proteins (30 μg) were boiled in sodium dodecyl sulfate (SDS) sample buffer for 5 minutes, then electrophoresed on a 12% SDS polyacrylamide gel and transferred onto a polyvinylidene difluoride membrane (Amersham Biosciences, Sweden). After being blocked in blocking buffer containing 5% no-fat milk for 2 hours at room temperature, the membranes were incubated with primary antibodies against cytochrome c, activated caspase 9, activated caspase 3, or β actin overnight at 4°C. The membranes were then rinsed with PBS 3 times (10 minutes each) and incubated for 45 minutes at room temperature with IRDye800-conjugated donkey anti-goat IgG (1:10 000 dilution) or donkey anti-mouse IgG (1:10 000 dilution). After 3 rinses (10 minutes each), the blots were visualized with Odyssey infrared imaging system (LI-COR Biosciences, Lincoln, Nebraska).
Statistical Analysis
All data were expressed as mean ± standard deviation (SD). For comparisons of the means, we used Student t test (P < .05).
Results
Cellular Uptake of EAHB
The cellular uptake kinetics of EAHB versus incubation time, studied through fluorescence intensity, indicated that cellular uptake of EAHB increased and reached a plateau after 1 hour of incubation (Figure 2A). The fluorescence imaging indicated that EAHB was distributed with well-defined spots throughout the cytoplasm of the cell, with no detectable penetration into the nucleus (Figure 2B). Due to the wide wavelength of EAHB, we were unable to isolate the localization of this compound to a specific organelle. No major differences were observed in the intracellular distribution of the sensitizer at different incubation times (data not shown).
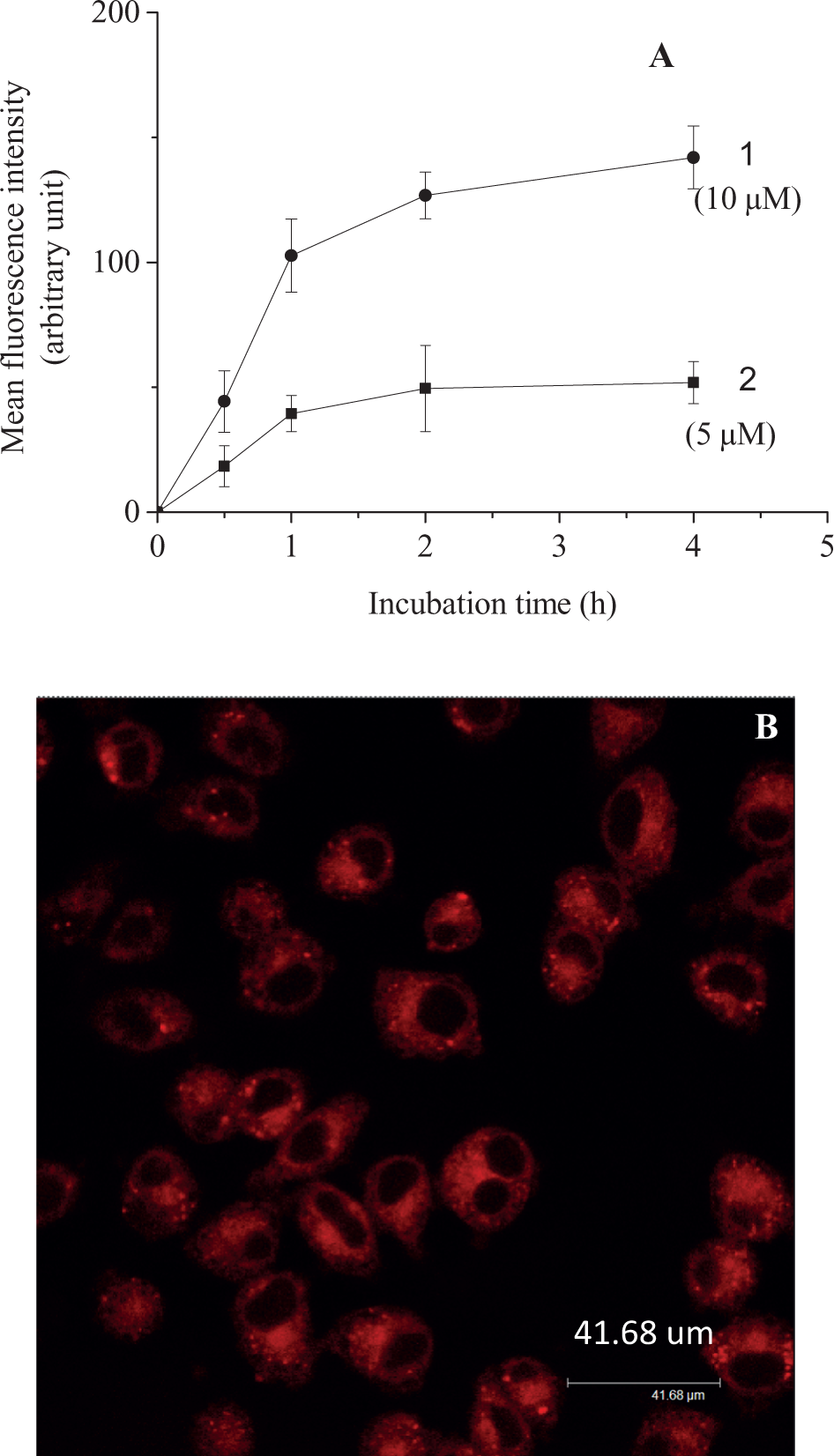
Uptakes and localization of 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B (EAHB) in HeLa cells. (A) Uptake of EAHB in HeLa cells as a function of incubation time at different concentrations of the photosensitizer as determined by fluorescence imaging (experiments were carried out in triplicate with 20 to 40 cells examined in each trial). (1) 5 μmol/L; (2) 10 μmol/L. Intracellular localization of EDAHB. (B) Cells were incubated with 10 µmol/L of EAHB for 1 hour in fetal bovine serum (FBS)-free Dulbecco-modified minimum essential medium (DMEM) and observed under a laser confocal fluorescence microscope. Magnification, ×60 oil, zoom factor: 1.
Photocytotoxicity and Lethal Dosage of EAHB to HeLa Cells
No cytotoxicity was observed upon exposure of HeLa cells to red light alone (Figure 3A) and the corresponding MTT optical density was taken as 100% cell survival. However, as shown in Figure 3A and C, with the light, EAHB caused a remarkable phototoxicity to HeLa cells, and the cell survival rate decreased with dependents of light dose, concentration of EAHB, and time post-PDT. At the light dose of 8.4 J/cm2, EAHB caused 50% cytotoxicity at a concentration of 1.2 μmol/L (median lethal dose [LD50] light; Figure 3A).
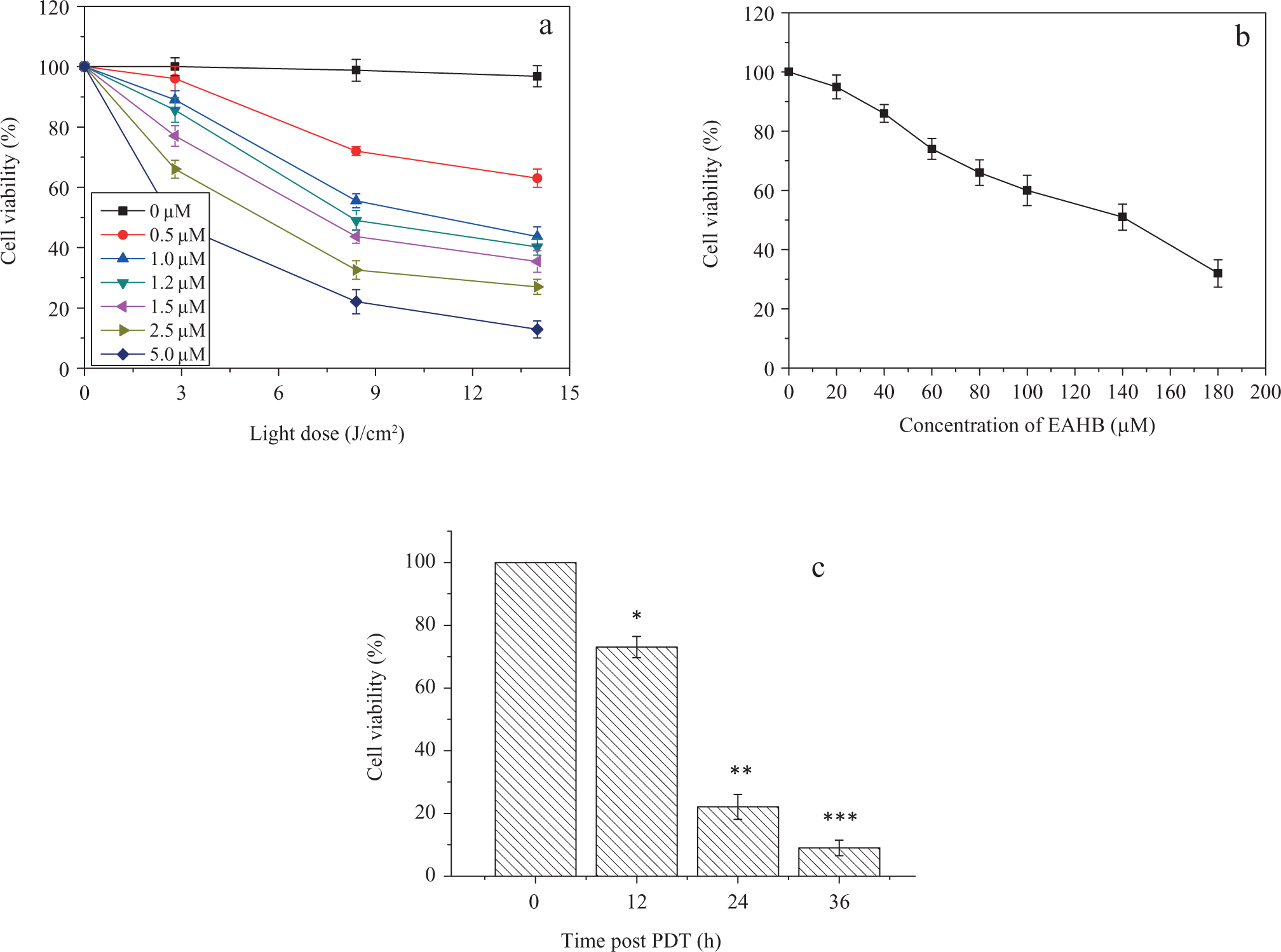
Phototoxicity (A) and cytotoxicity (B) of 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B (EAHB) to the HeLa cells using 3-(4,5-dimethylthiazol-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay, and (C) time dependent cell survival post-PDT (5.0 μmol/L EAHB plus 8.4 J/cm2 light). Representative results are shown as the mean ± standard deviation (SD) from 2 independent experiments. *, **, and *** P < .05 versus group of 0 hour post-PDT.
Photopotentiation is defined as the molar concentration ratio of the amount of PS required to achieve a 50% cell death (LD50) in vitro in the dark to the amount required to achieve an LD50 with irradiation of red light, as determined by the MTT assay. 12 The LD50 of EAHB in the dark (LD50 dark) was 140 μmol/L (Figure 3B). Therefore, EAHB was characterized by 120-fold photopotentiation factor toward HeLa cells at the light dose of 8.4 J/cm2.
The EAHB Photoinduces Apoptosis in HeLa Cells
The photocytotoxicity of EAHB to cells was further analyzed by flow cytometry. The results showed the appearance of hypoploid cell populations in photosensitized cells after 12 hours of PDT treatment (Figure 4). Percentages of apoptotic HeLa cells increased along with incubation time post-PDT treatment.
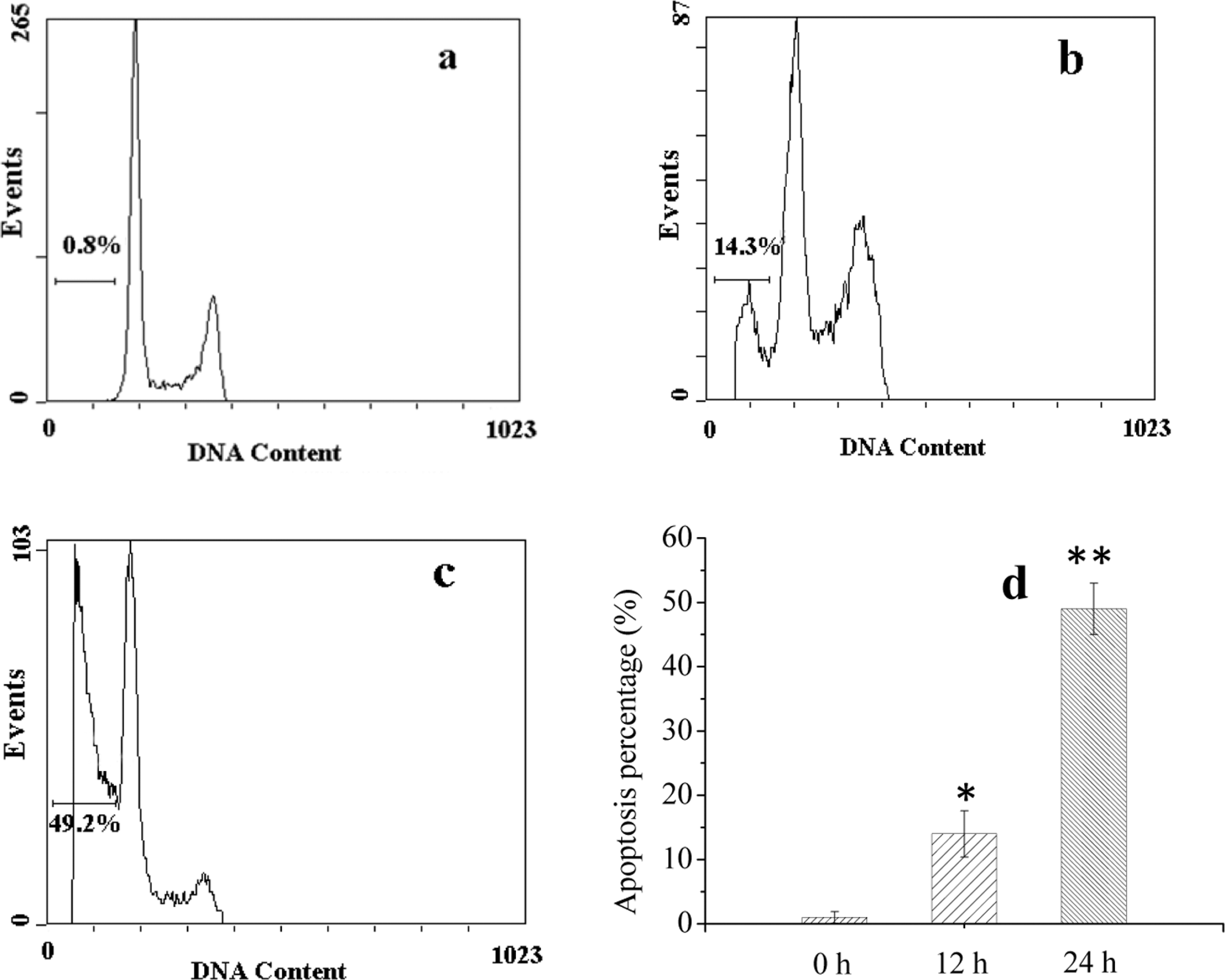
The DNA histogram of HeLa cells treated with 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B (EAHB)-mediated photodynamic therapy (PDT) at 5.0 μmol/L EAHB plus 8.4 J/cm2 light. (A) 0 hour after EAHB photosensitization; (B) 12 hours after EAHB photosensitization; (C) 24 hours after EAHB photosensitization; and (d) the percentages of apoptotic HeLa cells at different incubation time postirradiation. The results are representative of 3 independent experiments. Columns were shown as the mean ± standard deviation (SD) from 3 independent experiments.* and ** P < .05 versus group of 0 hour post-photodynamic therapy (PDT).
Degradation of DNA into a specific fragmentation pattern with multimers of approximately 200 base pairs is a characteristic feature of apoptosis. Agarose gel electrophoresis was used to examine the production of apoptotic DNA fragments in HeLa cells after EAHB-mediated PDT. As shown in Figure 5 the characteristic DNA ladders in HeLa cells 24 hours post-EAHB-mediated PDT (5 μmol/L EAHB plus 8.4 J/cm2 light) were observed (Figure 5, lane 4). No DNA ladders were observed when the cells were exposed to PS or red light alone (Figure 5, lanes 2 and 3).

DNA fragmentation in HeLa cells 24 hours postirradiation. Lane 1, DNA marker; lane 2, cells were incubated with EAHB alone (5.0 μmol/L, 1 hour); lanes 3 and 4, cells were incubated with 0 μmol/L (lane 3) and 5.0 μmol/L (lane 4) of 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B (EAHB) for 1 hour and followed by 8.4 J/cm2 of irradiation, respectively. The results are representative of 3 independent experiments.
The morphology of dying cells was investigated using Hochest 33258 nuclear staining. In the absence of light irradiation, the nuclei of EAHB-treated HeLa cells remained intact (Figure 6A), similar to the situation for cells exposed to red light alone (Figure 6B). However, characteristic apoptotic nuclei were detected in EAHB-photosensitized cells 24 hours postirradiation (Figure 6C).
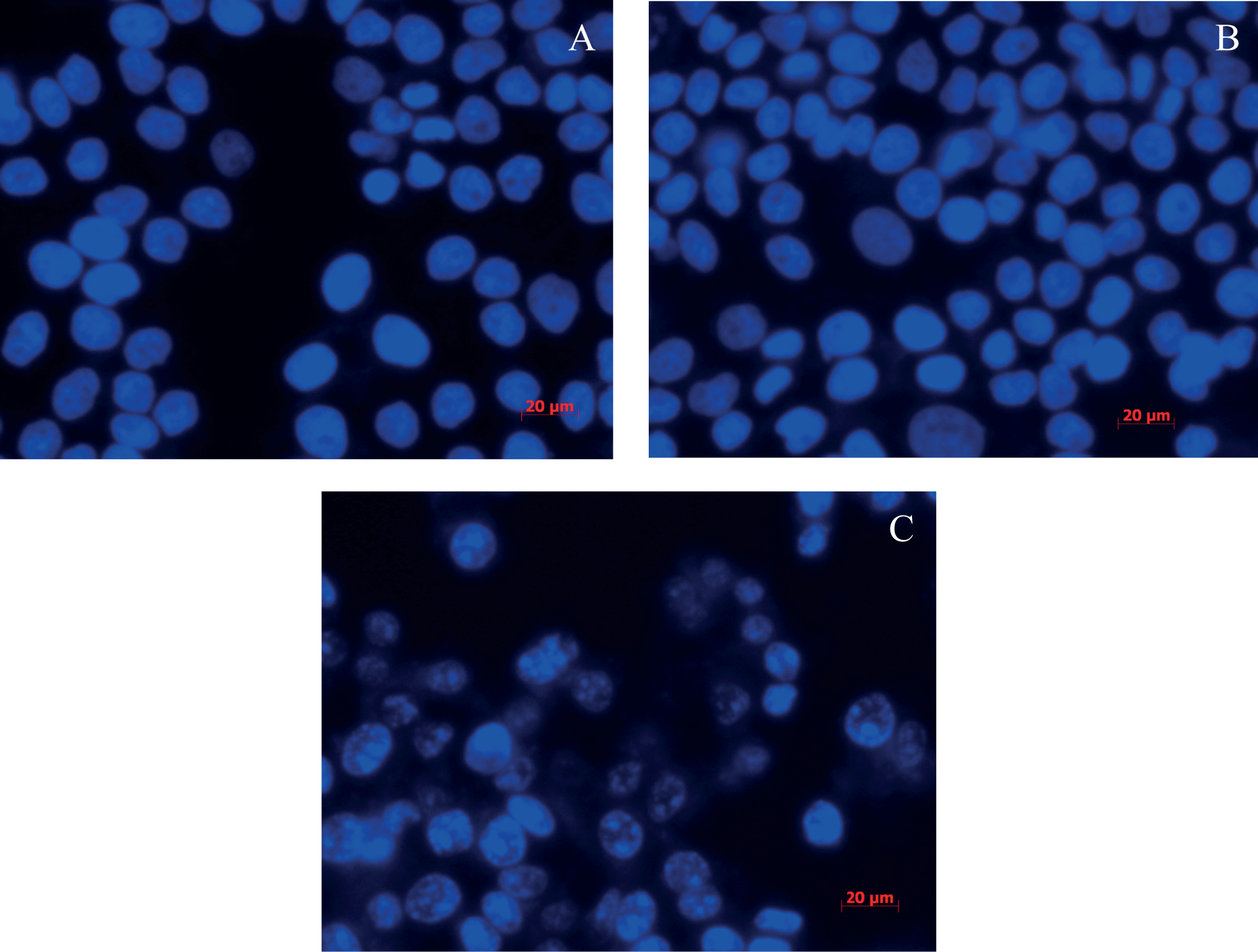
Fluorescence images of Hoechst 33258-stained nuclei of HeLa cells 24 hours postirradiation. (A) Cells were incubated with 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B (EAHB) alone (5.0 μmol/L, 1 hour); (B) and (C), cells were incubated with 0 μmol/L (B) and 5.0 μmol/L (C) of EAHB for 1 hour and followed by 8.4 J/cm2 of irradiation, respectively. Magnification, ×40. The results are representative of 3 independent experiments.
The EAHB Photoinduces a Mitochondria-/Caspase-Dependent Apoptosis Pathway
To examine whether EAHB photoinduced mitochondrial membrane dysfunction, cells were incubated with the MitoCapture reagent to detect intact or collapsed mitochondrial membrane potentials after PDT treatment. The results showed that EAHB-mediated PDT caused green diffuse cytoplasmic fluorescence (Figure 7C), while exposure to EAHB or red light alone produced red punctuate fluorescence (Figure 7A and B).
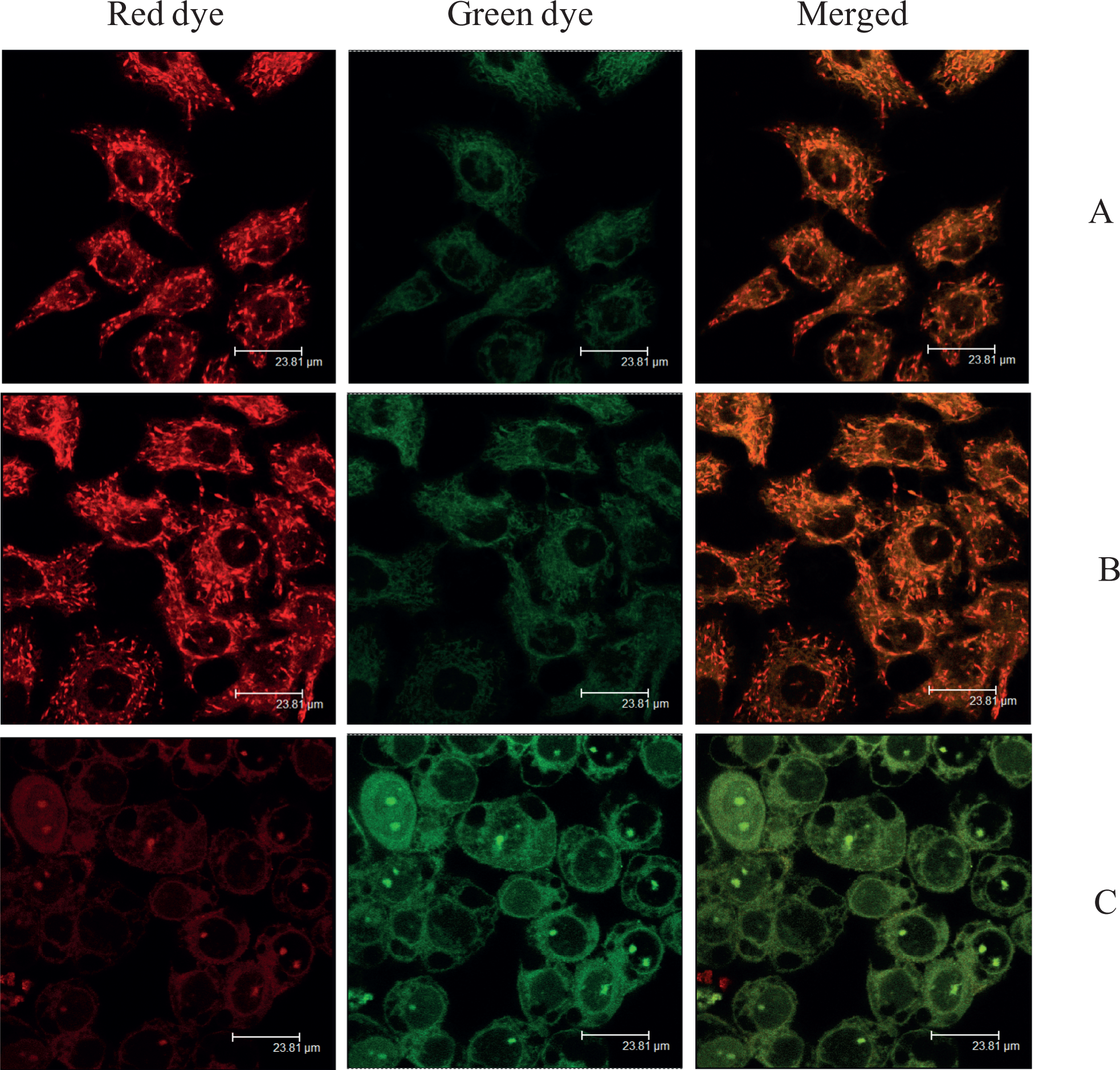
Detection of intact or collapsed mitochondrial membrane potentials with MitoCapture reagent 12 hours postirradiation. (A) Cells were incubated with 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B (EAHB) alone (5.0 μmol/L, 1 hour); (B) and (C), cells were incubated with 0 μmol/L (B) and 5.0 μmol/L (C) of EAHB for 1 hour and followed by 8.4 J/cm2 of irradiation, respectively. Punctate red fluorescence (intact mitochondria); diffuse red/green fluorescence (permeabilized mitochondria). Magnification, ×60 oil, zoom factor: 2. The results are representative of 3 independent experiments.
Due to the linkage between cytochrome c release and loss of mitochondrial membrane potentials, we next examined the release of cytochrome c from mitochondria as caused by EAHB-mediated PDT treatment. Cytosolic fraction was prepared from EAHB-photosensitized cells 12 hours postirradiation, and cytochrome c was detected by Western blot analysis. As shown in Figure 8, cytochrome c accumulated in the cytosol at 12 hours post-EAHB-mediated PDT.
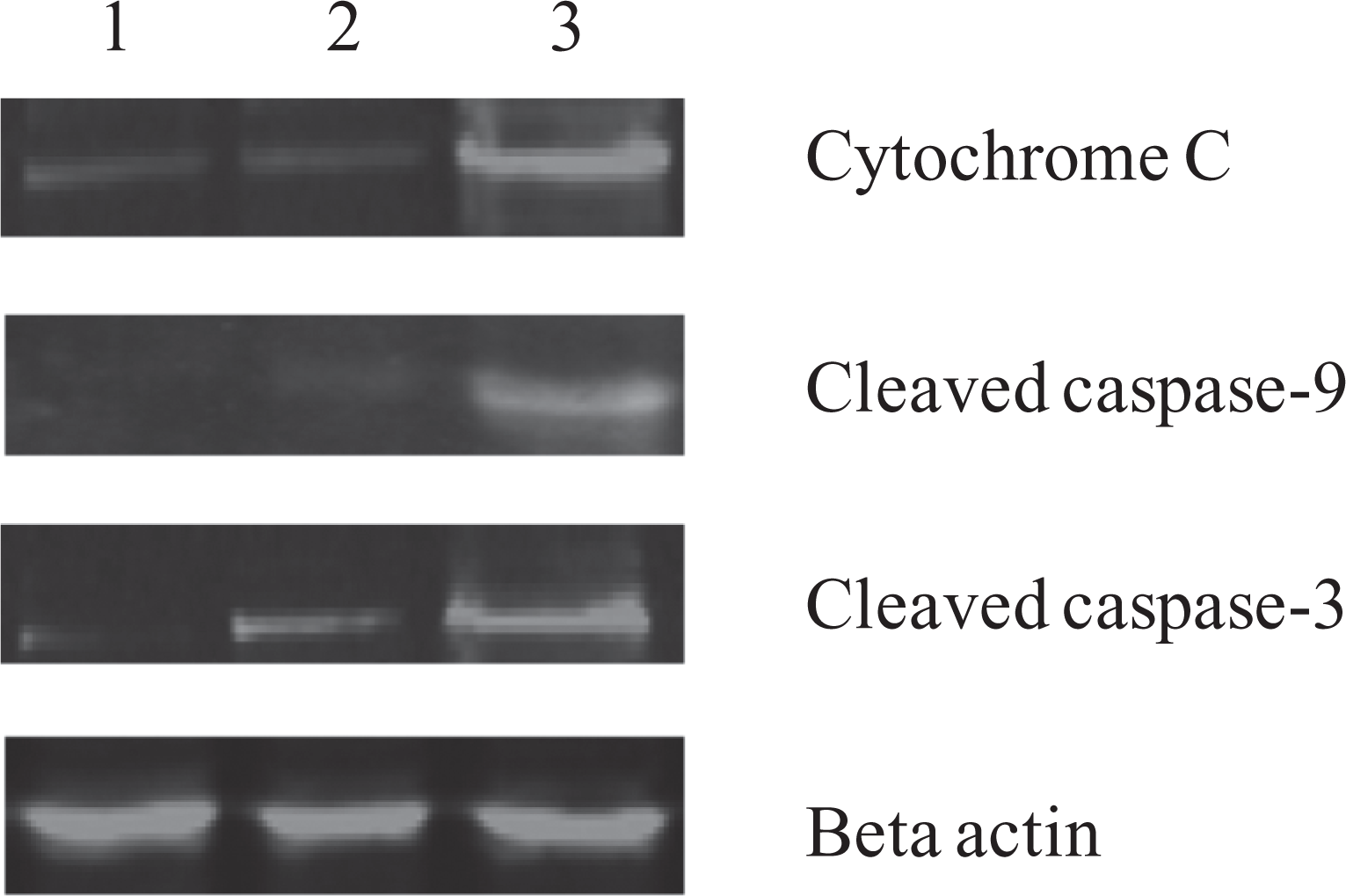
Western blot analysis of cytoplasmic cytochrome c, cleaved caspase 9 and cleaved caspase 3 in HeLa cells 12 hours postirradiation. (1) Cells were incubated with 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B (EAHB) alone (5.0 μmol/L, 1 hour); (2) and (3), cells were incubated with 0 μmol/L (B) and 5.0 μmol/L (C) of EAHB for 1 hour and followed by 8.4 J/cm2 of irradiation, respectively. The results are representative of 3 independent experiments.
Since the release of cytochrome c from mitochondria can activate the caspase cascade, we investigated the involvement of caspase 9 and caspase 3 in apoptosis induced by EAHB-mediated PDT treatment. As shown in Figure 8, the active form of caspase 9/3 was observed in the cytosolic faction of EAHB-photosensitized cells 12 hours post-EAHB-mediated PDT.
Discussion
The EAHB is a derivative of HB in which the ethanolamino group is substituted in position 2. The product has the properties of an ideal photosensitizer, including strong absorption of clinically used red light, low dark cytotoxicity, and excellent photopotentiation of cellular damage. 21 However, the mechanism of cytotoxicity caused by EAHB-mediated PDT has not been elucidated. The purpose of this study was to investigate the molecular pathway of EAHB-induced cell death after PDT.
First, we applied the photodynamic effect to the HeLa cell line. The effects of both the concentration and the light dose on the phototoxic potential of EAHB were evaluated by the MTT assay in Figure 3. The results from Figure 3B suggested that EAHB alone evoked 50% lethality at a concentration of 140 μmol/L. The results from Figure 2A indicated that EAHB exhibited 50% phototoxicity to HeLa cells at 1.2 μmol/L at 8.4 J/cm2 of irradiation. Therefore, EAHB demonstrated a 120-fold photopotentiation in vitro. This showed that EAHB exhibited a wide safety margin for drug administration, and this therefore made it one of the promising derivatives of HB for use as a PDT photosensitizer.
There have been reports that the subcellular localization of photosensitizer is one of the key factors that determine the type of photodynamic effects. 13,22 These reports show that hypocrellins and their derivatives could be detected in mitochondria, lysosomes, Golgi, endoplasmic reticulum and, to a minor extent, in cellular membranes. 13,23 But the exact localization of each compound will originate in its physicochemical property characteristics, which may ultimately impact on the primary modality of phototoxicity. Because the absorption spectrum of EAHB is across a wide range of wavelength, 14 we were unable to isolate the localization of this compound to a specific organelle with fluorescence probes as suggested in literatures. 22,23 However, the intracellular localization of EAHB occurred in the form of punctuate granular structures inside the cytoplasm. This signified that the cytoplasmic distribution of EAHB was the major cause of its phototoxicity.
Second, characteristic apoptotic features included chromatin condensation and DNA fragmentation, finally ending with the engulfment by macrophages or neighboring cells; while during necrosis, the cellular contents are released uncontrolled into the cell’s environment which results in damage to surrounding cells and a strong inflammatory response in the corresponding tissue. Therefore, apoptosis is an important target mechanism in cancer therapies, including PDT. 24 Apoptotic responses have been observed in tumor cells treated with various sensitizers, such as hemoporfin, 25 silicon phthalocyanine (Pc 4), 26 hypocrellin A, and its derivatives. 12,13,27 In this study, we demonstrated that EAHB photoinduced apoptosis in HeLa cells as evidenced by flow cytometric detection of DNA fragments, agarose gel electrophoresis, and PI nuclear staining (Figures 4, 5, and 6). The percentages of apoptotic HeLa cells also increased with the incubation time postirradiation (Figures 3C and 4), indicating that DNA fragmentation was caused by an enzymatic process rather than by direct photochemical damage to DNA. 28
Third, the mitochondrion has been reported to play a key role in the regulation of apoptosis. 16,29 Mitochondrial dysfunction including the loss of mitochondrial membrane potential (ΔΨm), permeability transition, and release of cytochrome c from the mitochondrion into the cytosol, are all associated with apoptosis. 30 In this study, the loss of ΔΨm and release of cytochrome c were observed in EAHB-mediated PDT-treated HeLa cells (Figures 7 and 8), suggesting that the cellular apoptosis induced by EAHB-mediated PDT treatment involved a mitochondria-dependent pathway. Some results have already demonstrated that PDT could open the permeability transition pore, which then leads to the loss of integrity of the outer mitochondrial membrane; this would then release the intermembrane proteins—such as cytochrome c—into the cytosol. 31
Mitochondria-dependent apoptosis might imply that caspase activation is involved in the cell death. 17 When released from the mitochondria, cytochrome c combines with an inactive protease precursor, procaspase 9, to form the “apoptosome.” The apoptosome attacks procaspase 3 and cleaves it to form active caspase 3, which can lead to DNA breakage, nuclear chromatin condensation, and ultimately cause cell death (Figure 9). The present study showed that EAHB-mediated PDT treatment caused activation of caspase 9 and caspase 3 in HeLa cells, suggesting that cell death was through a caspase-dependent mechanism.
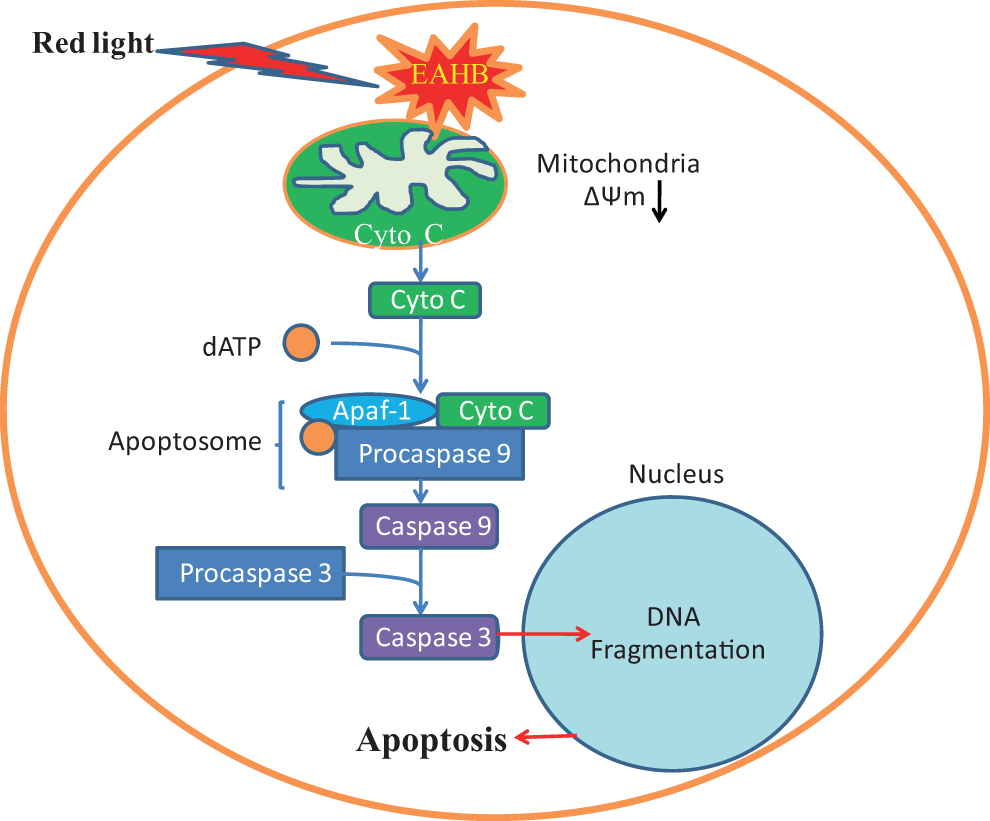
Cellular signaling pathways leading to apoptosis in cells after 2-ethanolamino-2-demethoxy-17-ethanolimino-hypocrellin B–photodynamic therapy (EAHB-PDT).
Conclusion
The PDT has emerged as a promising therapeutic procedure for the treatment of many tumors. Cell death in PDT may occur by apoptosis or by necrosis, depending on the sensitizer, PDT dose, and cell genotype. We have demonstrated that EAHB-mediated PDT treatment induced apoptosis in HeLa cells. The mechanism for the apoptosis in HeLa cells induced by EAHB-PDT appeared to be via a mitochondrially mediated pathway, accompanied by cytochrome c release from the mitochondria into the cytosol, which was then followed by the activation of caspase 9/3. These results suggest that EAHB is an apoptosis-inducing photodynamic agent and provide a fundamental basis for the clinical application of EAHB as a candidate in antitumor treatments.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Funding Project for Academic Human Resources Development in Institutions of Higher Learning Under the Jurisdiction of Beijing Municipality (PHR(IHLB)) and by the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry of China.
