Abstract
The objective of this study was to identify potential target organs for toxicity of recombinant human follicle stimulating hormone (r-hFSH) in female rhesus monkeys and to establish a no observed adverse effect level (NOAEL). In all, 24 female rhesus monkeys (Chinese origin, weighing 3.4-5.2 kg, approximately 5 years of age) received repeated subcutaneous (sc) r-hFSH at doses of 10, 60, and 300 IU/kg per d or vehicle once daily for 30 days followed by a 15-day recovery period. Endometrial hyperplasia and dermal edema in the external genitals were found in some animals at 300 IU/kg per d. Pharmacologic-related multiple cystic follicles were found in all r-hFSH-treated groups. A weak, anti-FSH antibody response was detected at the end of treatment in animals administered 60 and 300 IU/kg per d. These results indicate that the primary effects of r-hFSH in female rhesus monkeys were related to its pharmacological activity on the reproductive system. The NOAEL was considered to be 60 IU/kg per d.
Introduction
Follicle-stimulating hormone (FSH) is a heterodimeric glycoprotein hormone secreted by the anterior pituitary gland and is composed of 2 noncovalently linked protein subunits, the α-subunit (92 amino acids) which is common to other glycoprotein hormones, and the hormone-specific β-subunit (111 amino acids). 1,2 Human FSH (hFSH) is now produced by recombinant DNA technology (recombinant hFSH [r-hFSH]) and has been introduced to the market. Follicle-stimulating hormone is widely used in the treatment of anovulatory infertile patients and in stimulating multiple follicle development and maturation in patients undergoing assisted reproductive treatment (ART). 3,4 Recombinant hFSH is produced by Chinese hamster ovary (CHO) cell lines transfected with the genes encoding for the 2 subunits of FSH. 5,6 Recombinant hFSH has been shown to be similar to urinary FSH (u-hFSH) in amino acid sequence, glycosylation sites, receptor-binding activity, and in vitro biological activity. Urinary-derived FSH preparations will likely be replaced by recombinant products for stimulating ovarian follicular development. 7 –11 Use of recombinant FSH offers advantages over urine-derived preparations, because r-hFSH has been shown to be clinically more consistent, better defined, safer, more user-friendly, and is available in monotherapeutic preparations. Compared with u-hFSH, r-hFSH has the following characteristics: r-hFSH is independent of the urine production process and avoids any shortage of materials for clinical use due to lack of raw materials; the r-hFSH preparation has a high purity of about 99% and a high-specific activity, with more than 10 000 IU/mg protein. This high-purity and -specific activity allows for safe administration by subcutaneous (sc) injection, with good local tolerance at the injection site and low immunogenicity. 12 –14 Good batch-to-batch consistency can be achieved with r-hFSH because the manufacturing process of a recombinant DNA technology product can be better controlled; additionally, the r-hFSH preparation is free of luteinizing hormone (LH) activity, thereby providing a monotherapeutic agent for clinical use.
The clinical implications of differentiating isoform profiles of FSH products are unclear and there are no convincing data to suggest marked superiority of either human-derived or animal-derived FSH formulations for ovarian stimulation in terms of efficacy or safety. 15 –22 The purpose of the present study was to evaluate the safety of r-hFSH in female rhesus monkeys over a 30-day treatment period, to identify potential target organs of toxicity as well as reversibility (after a 15-day treatment-free period) and to identify a no observed adverse effect level (NOAEL), if possible.
Materials and Methods
Test and Control Drugs
Recombinant hFSH (Lot: 20061201) produced by genetically engineered CHO cells, with a purity of 99.31% (size-exclusion high-performance liquid chromatography [SEC-HPLC]) and specific activity of 12910 IU/mg protein, was obtained from GeneScience Pharmaceuticals Co, Ltd, China. The r-hFSH strengths for injection in animals were 50 IU/mL, 300 IU/mL, and 1500 IU/mL, respectively, which were stored at 4°C. Controls were administered a vehicle free of r-hFSH.
Instrumentation
Instruments used included a Vet ABC fully automatic blood analyzer manufactured by ABX Inc, France, a Cobas Integra 400 Plus automatic biochemical analyzer manufactured by Roche Inc, USA, a STAGO-4 Coagulation Analyzer manufactured by STAGO Inc, France, a Model 550 Micro-Plate reader manufactured by Bio-Rad Inc, USA, and a Columbus Microplate Washer manufactured by TECAN Inc, Austria.
Experimental Animals
In all, 24 adult female rhesus monkeys were obtained from the Suzhou Animal Experiment Research Center (Certificate No.: SCXK (Su) 2002-0007, Suzhou, China). On commencement of the study, the animals weighed 3.4 to 5.2 kg, and the mean age was about 5 years. The monkeys were housed in single stainless steel cages equipped with an automatic drinking water system and food hopper. Animals were fed fresh solid food, twice daily (150 g each time), with 100 g of fruit each day. The animals were monitored for body weight, hematology, clinical chemistry, urinalysis, and electrocardiography during a 4-week pretrial acclimatization period. The animal rooms had natural light with automatic control of temperature (20°C ± 2°C) and humidity (60% ± 10%).
The care and maintenance of animals were as per the approved guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals. Requirements of environment and housing facilities of laboratory animal complied with national standard, PR China (GB 14925-2001). All animal procedures were approved by the Institutional Animal Care and Use Committee (IACUC, China) and the experimental animal protocols were approved by the Institutional Animal Ethical Committee of Tianjin Institute of Pharmaceutical Research, Tianjin, China.
This safety study was performed according to Good Laboratory Practice (GLP) Guidelines for the Non-clinical Toxicology Testing promulgated by the State Food and Drug Administration (SFDA) of China.
Design of Safety Evaluation
After preliminary dose-ranging studies, a 30-day sc injection toxicity study was performed followed by a 15-day recovery period. The monkeys were randomly assigned to 4 groups with 6 animals per group. Female rhesus monkeys were given an alternating sc injection at the lateral side of both thighs of either vehicle or r-hFSH at doses of 10, 60, and 300 IU/kg per d (approximately equivalent to 2.5, 15, and 75 times the recommended human therapeutic dose), once per day for up to 30 days (from December 26 to January 24). The volume of injection was 0.2 mL/kg for all groups. At the end of the treatment period, 4 animals in each group were euthanized and necropsied. The remaining 2 animals per group were observed for an additional 15 days before necropsy. Clinical signs, food consumption, and body weight were evaluated throughout the study. Rectal temperature and respiratory rates were measured prior to dosing and 1 hour postdose on days 1 to 5, 8, 15, 22, and 30 during the dosing period, and on days 8 and 15 of recovery. Ocular examinations were conducted using direct ophthalmoscope after the animals were anesthetized with ketamine prior to the first dosing, terminal necropsy, and recovery necropsy. Heart rate, P-R duration, QRS duration, QT duration, ST interval, and T wave were averaged via 5 cycles of II-lead electrocardiogram during acclimatization, 1 hour postdose on days 15, 30, and on day 15 during recovery period. Clinical pathology, including standard hematology, serum chemistry, and urinalysis measurements was done during acclimation, on days 8, 15, and 30 of dosing and recovery day 15. Anatomic pathology assessments, which include macroscopic and microscopic examinations of tissues and organs, were conducted at the end of the treatment period (d31) and at termination of the recovery period (rd16). In addition, sternum marrow cell morphology was assessed at each necropsy interval.
Serum Sex Hormone Level Assay
The monkeys were given an sc injection of vehicle or r-hFSH at a dose of 10, 60, and 300 IU/kg per d. Serum samples for sex hormone determination were collected on days 0 (predose), 8, 15, and 30 (postdose), as well as on day 15 of the recovery period. The changes in serum sex hormone level, including FSH, estradiol (E2), LH, and progesterone (P), were determined by radioimmunoassay (RIA) using commercial RIA kits (China Institute of Atomic Energy, China).
Detection of Anti-r-hFSH Antibodies
Indirect enzyme-linked immunosorbent assay (ELISA) was used to detect the titer of anti-r-hFSH antibodies in the serum on days 0, 15, and 30 of dosing, and on day 15 of recovery. Briefly, 96-well microplates were coated with 10 μg/mL r-hFSH in sodium carbonate–bicarbonate buffer (Na2CO3-NaHCO3, pH 9.6), 100 μL/well overnight at 4°C. After washing with phosphate buffered saline (PBS) containing 0.05% tween 20, the wells were blocked with 2% bovine serum albumin (BSA), 200 μL/well, for 2 hours at 37°C. Plates were washed in PBS and then 100 μL/well serum samples of series of dilutions at 1:10, 1:100, and 1:1000 were added to alternate rows of the plate. Following incubation for 1 hour at 37°C, the plates were washed to dislodge any unbound substances, and a 1:30000-diluted goat-anti-monkey immunoglobulin G (IgG) polyclonal antibody labeled with horseradish peroxidase (HRP) was used to detect the monkey anti-r-hFSH antibodies. Microplates were washed to remove unbound antibodies, followed by incubation with tetramethylbenzidine (TMB) as a substrate in the dark. The color development was stopped and the intensity of the color was measured. The absorbance of each well was determined within 30 minutes using a microplate reader set at 450 nm. Each plate contained positive and negative controls. Each serum sample was measured in duplicate and the mean value was used for the anti-r-hFSH antibodies formation analysis.
The 2.1 times of the mean optical density (OD) of the vehicle group was defined as the threshold which produced anti-r-hFSH antibodies. Compared to the vehicle group at the same period, the serum sample was judged to be positive when the OD of the treatment group was greater than or equal to the threshold. The intensity of antibody was denoted with the logarithm of antibody titer. 23
Neutralization Tests
The serum samples (diluted at 1:10, 1:20, and 1:40 with PBS containing 1% BSA) were incubated with the quantified r-hFSH (50 mIU/mL) for 1 hour at 37°C, and then the serum FSH levels were determined using ELISA. When the concentration of FSH detected was lower than the total amount of the original serum FSH and exogenous FSH administration, neutralizing antibodies were considered to have been formed, otherwise, there was no neutralizing potency.
Statistical Analysis
The test data are expressed as mean ± SD. Data statistical analysis was performed with the Statistical Package for Social Science program (SPSS) for Windows version 10.0. Statistical evaluation of data was performed with analysis of variance (ANOVA) for the repeated measured data, followed by Dunnett’s method to compare each test group with the control group and Ridit analysis for the ranked data. P < .05 is considered to be of significant difference and P < .01 highly significant difference.
Results
Safety and Tolerability of r-hFSH in Rhesus Monkeys
No animals died during the study, and observations of spirit, behavior, activity, feces, and urine did not show any abnormality related to r-hFSH treatment. No local irritation and/or inflammation (erythema, edema, lumps, and muscle atrophy) at the sites of injection were noted throughout the study. Following the administration of various dose levels of r-hFSH, all laboratory parameters including body weight, rectal temperature, food consumption, respiration, ophthalmic signs, and electrocardiography were unaffected by r-hFSH treatment. Clinical pathology measurements, including hematology, morphology of bone marrow cells, coagulation parameters, clinical chemistry, and urinalysis showed no changes related to r-hFSH treatment. In the external genitalia assessments, 2 animals in the high-dose group developed apparent swelling of the external genitalia associated with swelling of the area around the anus, which was most obvious at day 15 of the treatment period. All the above changes, except swollen external genitalia, were reduced at day 30. On day 15 of recovery period, that is, swelling of vulva and anus basically returned to the normal level, but swelling of external genitalia/edema was still noted.
In anatomic pathological assessments at necropsy, the ovaries were significantly increased in size and were translucent with the unaided eyes at the end of the treatment period in all r-hFSH-treated groups, compared to control. Moreover, a large number of mature follicles were observed on the ovary surface. Mean ovarian weights in the r-hFSH treated groups were significantly different (P < .05) from those in the control group, about 10 to 14 times greater than those of the control, and no apparent dose−effect relationship was observed. At the end of recovery, the ovary weights had declined but were still increased 4- to 5-fold over controls at all dose levels. In comparison to the control group, mean thymus weights were lower in both the mid-dose and high-dose groups (approximately 60%-70% of controls) but returned to control levels after the 15-day post dosing period (data not shown). Organ weight data are presented in Table 1 .
Organ Weights and Organ Indices in Subcutaneous Injection r-hFSH to Rhesus Monkeys at the End of Dosing (day 30; n = 4)
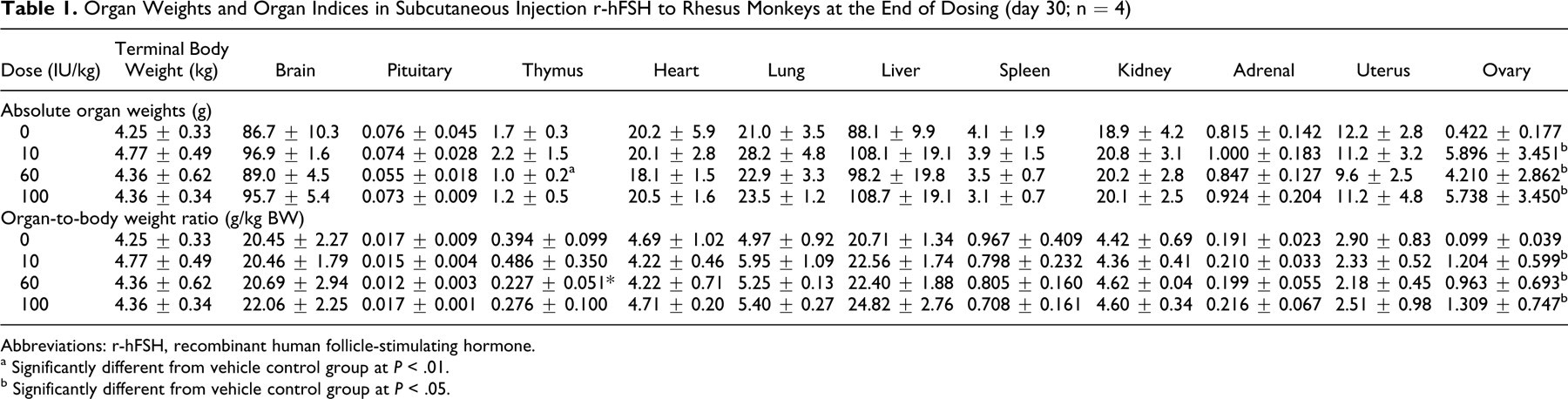
Abbreviations: r-hFSH, recombinant human follicle-stimulating hormone.
a Significantly different from vehicle control group at P < .01.
b Significantly different from vehicle control group at P < .05.
Microscopic examination of both skin and muscle of the injection sites were unremarkable. Dermal and sc edema of the external genitalia were found histologically during both the experimental phase and recovery phase, primarily at the high dose. It was unclear whether these changes were related to animals entering estrus normally, linked to r-hFSH inducing estrus, or a combination of both.
The abnormal histopathology indices in rhesus monkeys that received repeat-dose r-hFSH in the 30-day toxicity study followed by a recovery period of 15 days are listed in Table 2 .
The Number of Abnormal Indices of Histopathology in Subcutaneous Injection r-hFSH to Rhesus Monkeys for 30-Day Toxicity Studies Following 15-Day Recovery Period (d30[rd15]) a

Abbreviations: r-hFSH, recombinant human follicle-stimulating hormone.
a At the end of the treatment period: multiple cystic follicles were observed in all treated animals (4 of 4); endometrial glandular hyperplasia and external genital derma edema happened in 3 of 4 animals and 1 of 4 animals, respectively, at the dose of 300 IU/kg per d; sex skin edema occurred in treatment groups and control group, but the high-dose group had a high incidence; thymic atrophy happened in mid-dose and high-dose groups as well as control group.
Multiple cystic follicles were observed in all r-hFSH-treated animals at the end of dosing. In the high-dose group (300 IU/kg per d), 3 of 4 animals also had endometrial glandular hyperplasia at the end of the treatment period. All the above changes, however, were reversible following treatment-free recovery. Thymic atrophy was also observed in 4 animals of the mid-dose group and 3 in the high-dose group at end of treatment, and none in recovery, while 2 cases of thymic atrophy in treatment phase and 1 case in recovery were also found in the controls. This pathology finding could be a result of physiological involution during the progress of sexual maturity with increase of endocrine hormone. 24 In addition, thymus is sensitive to stress, which leads to glucocorticoid-induced thymocyte apoptosis and subsequent thymic involution. 25 No other treatment-related pathological changes were observed during the entire experimental period (Figure 1 ).
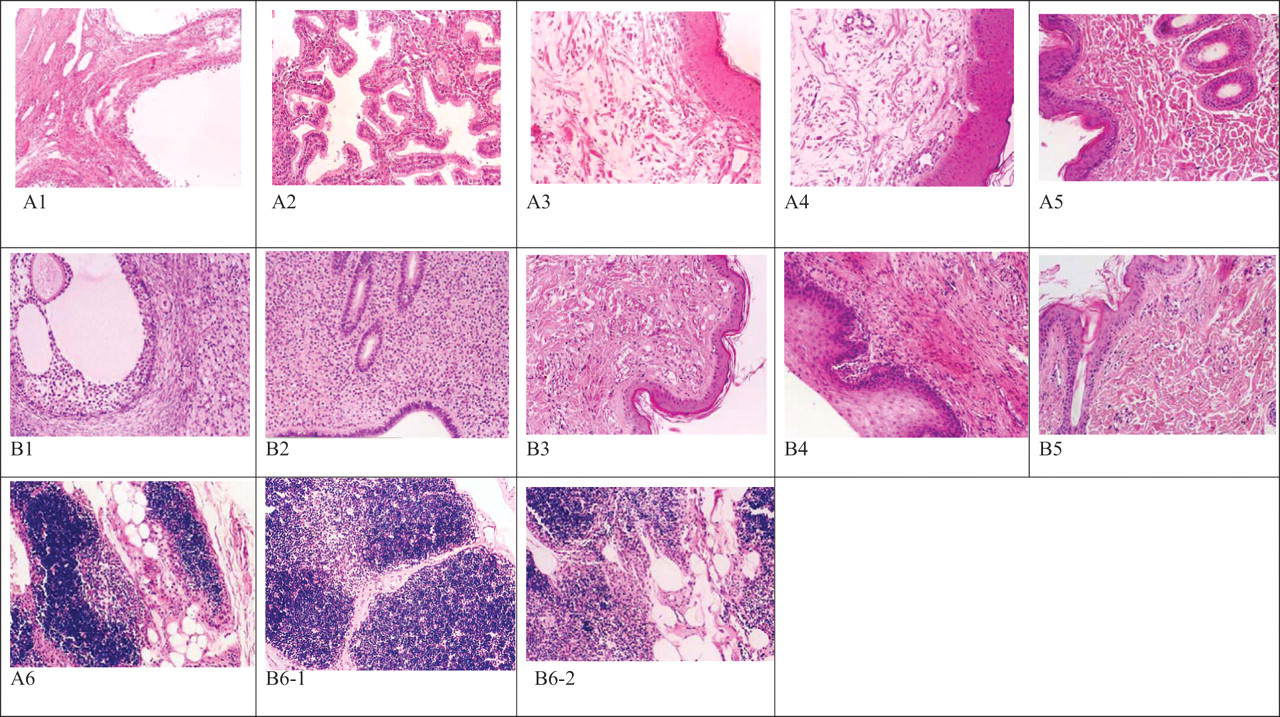
Histological changes by microscopic examination in monkeys that received recombinant human follicle-stimulating hormone (r-hFSH) at a dose of 300 IU/kg per d or control for 30-day toxicity study followed by a 15-day recovery period. The images (A) are pathological tissues in high-dose group, and images (B) are that of control group. Hematoxylin and eosin (H&E) staining, 10 × 10 magnification. (A1) Ovarian cystic follicles in high dose during treatment (×10, H&E); (A2) Endometrial glandular hyperplasia in high dose during treatment (×10, H&E); (A3) Sex skin dermal and hypodermis edema in high dose during treatment (×10, H&E); (A4) Hypodermis edema of the vulva in high dose during treatment (×10, H&E); (A5) Normal injection site in high dose during treatment (×10, H&E); (A6) Thymic atrophy in high dose during treatment (×10, H&E). (B1) Normal ovarian in control during treatment (×10, H&E); (B2) Normal uterus in control during treatment (×10, H&E); (B3) Normal sex skin in control during treatment (×10, H&E); (B4) Normal skin of the vulva in control during treatment (×10, H&E); (B5) Normal injection site in control during treatment (×10, H&E); (B6-1) Normal thymus in control during treatment (×10, H&E); (B6-2) Thymic atrophy in control during treatment (×10, H&E).
Serum Sex Hormone Levels
Endogenous FSH levels prior to r-hFSH administration were very low, about 0.7 to 1.6 mIU/mL, and the level of E2, LH, and P were 133 to 173 pg/mL, 0 to 0.15 mIU/mL, and 0.2 to 0.3 ng/mL, respectively. Serum FSH concentration was markedly increased 7 days after treatment initiation, as expected. The ratio of FSH concentration in the treatment groups to control (FSHdose groups/FSHcontrol) at dosages of 10, 60, and 300 IU/kg per d showed a clear dose-dependent increase, for example, the ratios for low-dose, mid-dose, and high-dose were 15, 81, and 311, respectively, after 2 weeks. The serum E2 level also increased markedly for all dose levels. Serum FSH and E2 reverted to the pretreatment level at the end of the recovery period. At doses from 10 to 300 IU/kg per d, serum LH level increased dose dependently during the experimental period, while serum P level showed no obvious change. The results of the sex hormone levels were summarized in Table 3 .
Changes in Serum Hormone Levels in Subcutaneous Injection r-hFSH to Monkeys for All Dose Levels at Various Time Points (n = 6)
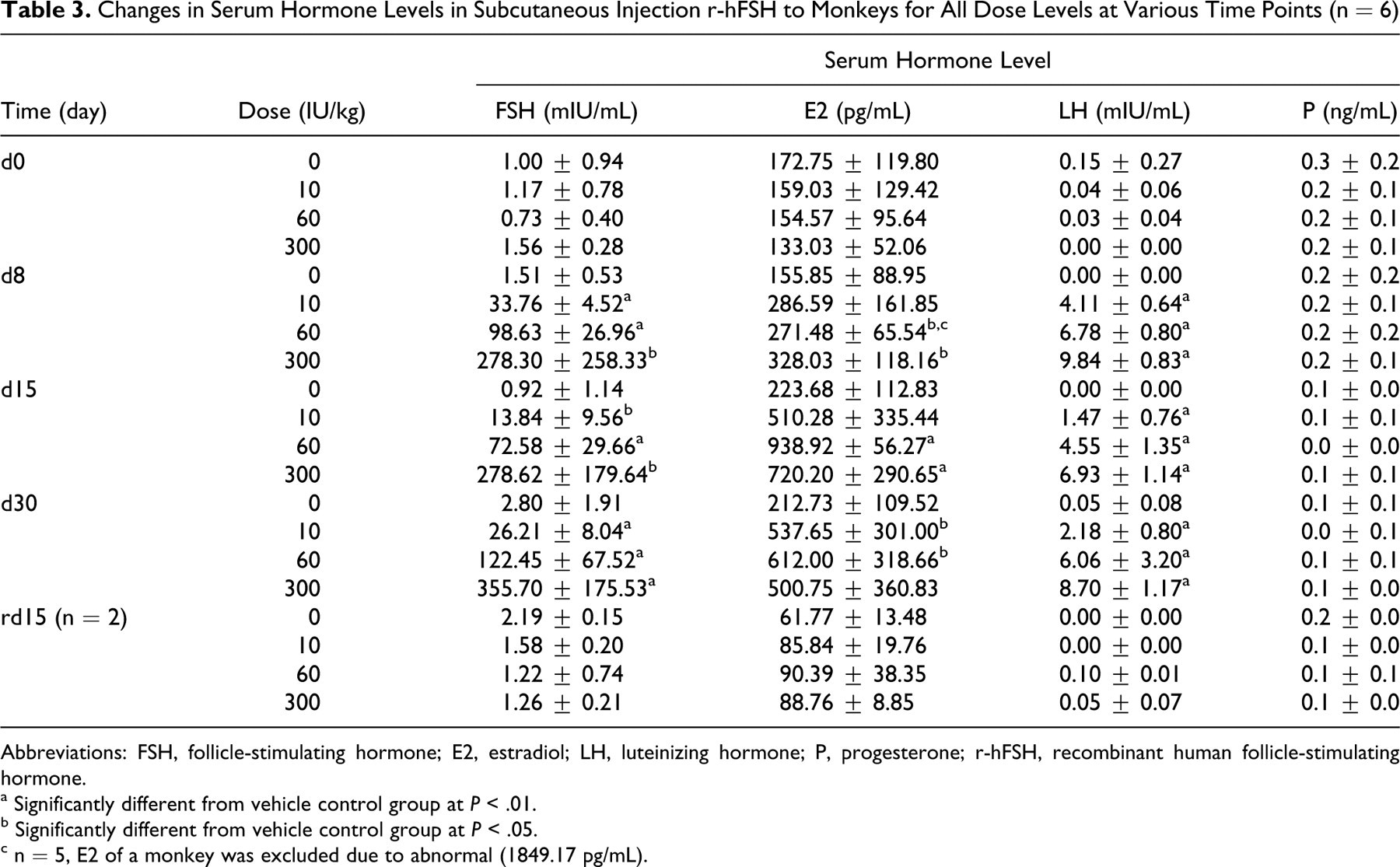
Abbreviations: FSH, follicle-stimulating hormone; E2, estradiol; LH, luteinizing hormone; P, progesterone; r-hFSH, recombinant human follicle-stimulating hormone.
a Significantly different from vehicle control group at P < .01.
b Significantly different from vehicle control group at P < .05.
c n = 5, E2 of a monkey was excluded due to abnormal (1849.17 pg/mL).
Anti-r-hFSH Antibodies
No anti-r-hFSH antibodies were observed in the low-dose group during the experiment of all groups in recovery phase. As shown in Table 4 , antibodies to r-hFSH were detected in half of all the monkeys (3 of 6) in the mid-dose and high-dose groups, respectively, compared with controls at the end of dosing (day 30). However, antibody titer was lower (1:10-1:100) and there was no apparent dose−response relationship in either the incidence of antibody positivity or titer of antibody development. Following the recovery period, antibody titers decreased and returned to preinjection values (data not shown).
The Distribution of Incidence of Anti-r-hFSH Antibodies After Subcutaneous Injection r-hFSH to Monkeys for 30-Day Toxicity Studies at Different Dilution Serum a
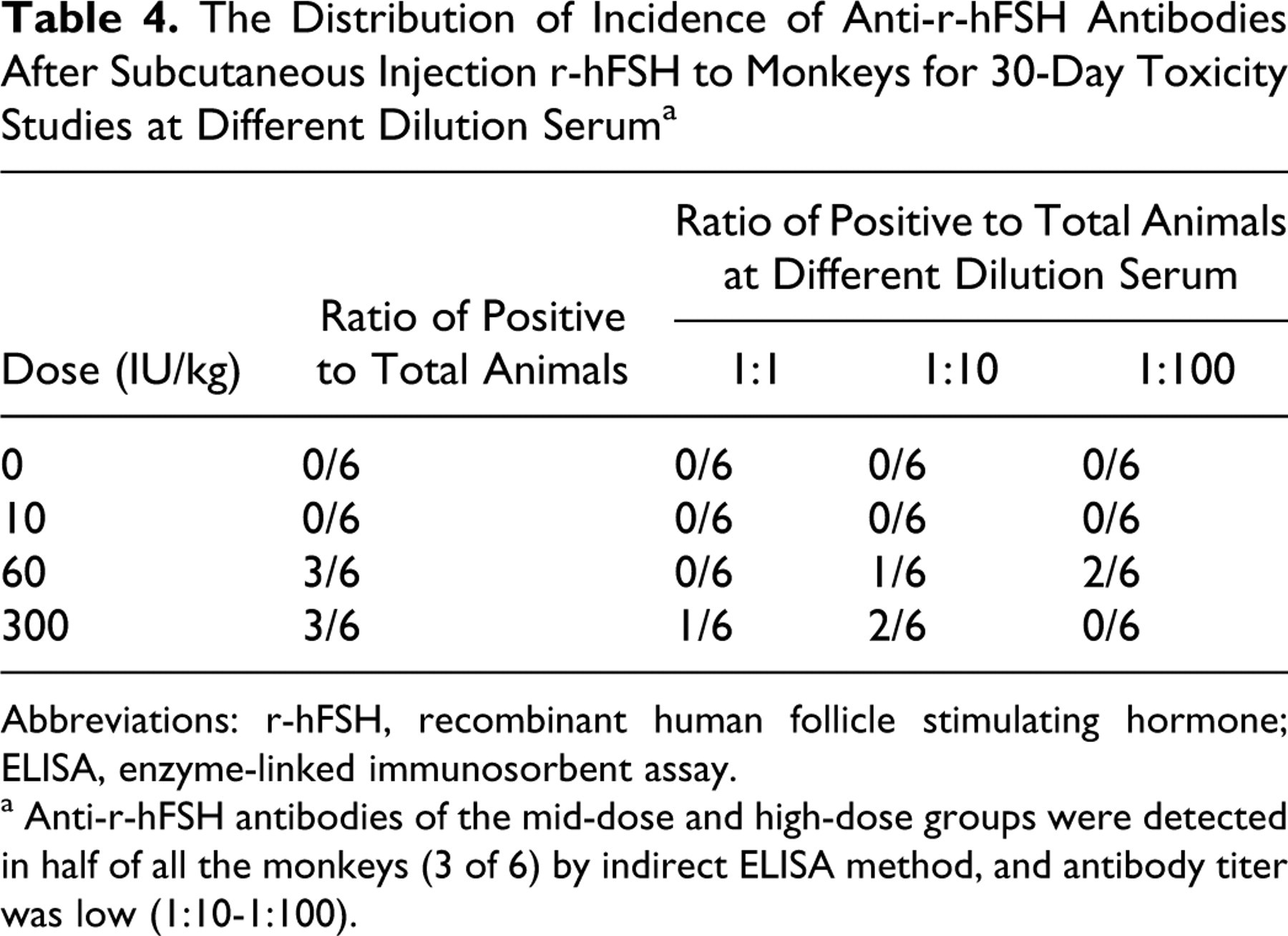
Abbreviations: r-hFSH, recombinant human follicle stimulating hormone; ELISA, enzyme-linked immunosorbent assay.
a Anti-r-hFSH antibodies of the mid-dose and high-dose groups were detected in half of all the monkeys (3 of 6) by indirect ELISA method, and antibody titer was low (1:10-1:100).
In order to further determine neutralizing antibody activity in serum samples which developed binding antibodies, r-hFSH (50 mIU/mL) was quantitatively added to the samples with positive antibodies. The serum r-hFSH levels were measured by ELISA. The results showed that the amount of FSH detected was almost equal to the r-hFSH added, which suggested that the binding antibodies were nonneutralizing antibodies.
Analysis of Safety and Target Organ Toxicity
No mortality or moribundity was found in the present study. For all groups, no changes were observed in food consumption, general activity, clinical observations, respiratory rate, or body weight. There were no local reactions, and no changes were found in hematology, marrow morphology, serum chemistry, coagulation, urinalysis, or electrocardiography (ECG) evaluations.
For the low-dose group (10 IU/kg per d), no r-hFSH effects were noted except ovarian cystic follicles (all treated animals). Ovarian cystic follicles were pharmacologic-related and recovered by 15 days postdose. Anti-r-hFSH antibodies were not detected in any of the low-dose samples.
At the mid-dose of 60 IU/kg per d, ovarian cystic follicles and thymic atrophy were found in all animals (4 of 4). In the control group, 3 cases of thymic atrophy were also found; hence it was not considered related to r-hFSH. Ovarian cystic follicles were related to the primary pharmacodynamics of r-hFSH. Nonneutralizing antibodies were detected in 3 animals on day 30 and their titers were very low (with 1:10 in 1 case and 1:100 in other 2 cases).
At the high dose of 300 IU/kg per d, histopathological examination showed evidence of ovarian cystic follicles (4 of 4), thymic atrophy (3 of 4), endometrial glandular hyperplasia (3 of 4), and dermal edema at the external genitals (3 of 4) at the end of administration. Thus, the dose of 300 IU/kg per d could cause a mild toxicity to reproductive tissues. All these changes, however, were reversible during the 15-day recovery period. Thymic atrophy was not considered definitely related to the treatment. The titers of nonneutralization antibody detected on day 30 were 1:1 in 1 case and 1:10 in the other 2 animals, respectively. Thus, based on the endometrial hyperplasia and edema of the external genitalia, the NOAEL for r-hFSH in rhesus monkey was considered 60 IU/kg per d, approximately equivalent to 15 times the recommended human therapeutic dose.
Discussion
From the reports on Gonal-f, 26,27 studies on the toxicity of r-hFSH in experimental animals showed that in monkey studies, ovarian and endometrial changes were observed. However, these findings were considered related to the pharmacological action of the active substance. The serum FSH levels measured during dosing period confirmed extensive exposure despite the development of antibodies to the foreign protein. The general similar toxicological findings demonstrate that some hormonal activity of FSH was maintained.
Changes in Serum Sex Hormone Level, Ovaries, and Endometrium
The levels of endogenous FSH in the animals were low (about 0.7-1.6 mIU/mL) before dosing. The serum FSH concentration was significantly increased in all r-hFSH-treated groups after 1 week, and FSH level peaked at day 30 to a mean of 26, 122, and 356 mIU/mL in the low-dose, mid-dose, and high-dose group, respectively. Ovarian weights were also markedly increased (about 10- to 14-fold higher compared with controls) at all dose levels associated with the rise in serum FSH level; however, no clear dose relationship was observed. At the same time, serum E2 level was also higher than that of the controls (about 2.4-2.9 times higher). The histopathological findings showed that multiple cystic follicles occurred in all groups receiving r-hFSH. In the high-dose group, endometrial glandular hyperplasia was observed in 3 of 4 animals at the end of dosing, while the uterine weights did not increase obviously. The serum LH level rose slightly during the treatment period but reverted to baseline levels in the treatment-free recovery period. Although unknown, it is possible that excessive secretion of ovarian estrogen, as noted in the high circulating E2 levels in this study, produced feedback inhibition on basal FSH secretion and stimulated LH secretion as evidenced by the mild increase in LH during the study period. No significant changes in serum P levels were observed during the experiment. The serum FSH level returned to pretreatment level at the end of the recovery period, and other indices also returned to the normal levels.
Changes in the Swelling of External Genitalia
At the dose of 300 IU/kg per d, swelling of the external genitalia was found in 2 females given r-hFSH during dosing. In general, swelling of the external genitalia in female monkeys is a special physiological phenomenon during estrus, which occurs in response to hormonal fluctuations. During this study (occurring from late December to early February), rhesus monkeys were in the nonmenolipsis period, which meant that the animals with swollen external genitalia might be in natural estrus. However, since r-hFSH administration may stimulate excessive secretion of ovarian estrogen, resulting in a more significant swelling of the external genitalia, this effect might be related to r-hFSH alone, or to a combination of factors. According to a literature report, the external genitalia of monkeys showed reversible swelling when the r-hFSH dose was equal to or more than 300 IU/kg per d (intramuscularly [im]). 26,27 These reported findings were consistent with that of the present study.
Neutralizing Antibody Analysis
To identify whether anti-FSH antibodies could neutralize the activity of FSH, the most direct method is to bioassay the activity of FSH using rat ovary weight gain as an end point, based on the Chinese Pharmacopoeia. In the present study, however, this method was difficult as it required additional serum samples, hence given the robust pharmacologic activity at all doses, as well as internal projection of the fraction of free FSH measured in our assays, we estimated that the antibodies were likely nonneutralizing.
As reported in the Product Monograph of GONAL-f Pen, a similar product marketed in Canada, intramuscular doses between 10 and 1000 IU/kg per d for 4 weeks in male and female cynomolgus monkeys resulted in ovarian weight increases in all animals, ovarian cystic follicles, proliferation in mammary glandular cells, and hypertrophy in pituitary acidophilic cells for all the groups. At doses ≥300 IU/kg additional findings included swollen genitalia (reversible) and thymic atropy. Antidrug antibodies were detected at all doses.
Our results were similar to those of GONAL-f, although we did not find proliferation of mammary glandular cells, hypertrophy of pituitary acidophilic cells, muscle degeneration, and/or inflammation at injection sites by examining the pathological sections.
Conclusions
In a 30-day repeated-dose toxicity study of r-hFSH in female rhesus monkeys at sc doses of 10, 60, and 300 IU/kg per d once daily (approximately equivalent to 2.5, 15, and 75 times the recommended human therapeutic dose), no mortalities were observed, and no distinct clinical signs of systemic toxicity or local inflammation at the injection sites were noted. Body weight and food consumption were unaffected by r-hFSH treatment. In addition, there were no effects on ophthalmology or electrocardiography. There were no significant changes related to treatment in the clinical pathologic analyses, including hematology and sternum marrow cell morphology, serum chemistry, and urinalysis parameters. Anatomically pathological assessments revealed ovarian enlargement and the formation of multiple cystic follicles at all dose levels, and endometrial hyperplasia as well as dermal edema in the external genitals at 300 IU/kg per d. All these changes were reversible. In addition, the serum FSH level was significantly increased during treatment. A weak, anti-FSH antibody response was detected at the end of treatment in the monkeys treated with 60 and 300 IU/kg per d.
In conclusion, our results indicate that the toxicity of r-hFSH in female rhesus monkeys was generally related to exaggerated pharmacology at the high dose (300 IU/kg) under the conditions of this study. The NOAEL in rhesus monkeys can be considered 60 IU/kg per d, based on mild toxicity to reproductive tissues at 300 IU/kg per d.
Footnotes
Acknowledgments
The authors thank GeneScience Pharmaceuticals Inc, Changchun, China for technical support. We are also grateful to Hairong Wang, Jun Zhang, and Zihui Song for technical assistance.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research of this article: the Key Science and Technology Program of China State Science and Technology Agency (No. 2007CB516807), and State Major Scientific Special Project (No: 2008ZX09305-005).
