Abstract
Rosmarinic acid (RA) is a naturally occurring polyphenolic compound found in various plant families. We previously reported that RA exerted protective effects against 6-hydroxydopamine (6-OHDA)-induced neurotoxicity through antioxidative properties. In this study, we investigated whether RA could prevent effects of 1-methyl-4-phenylpyridinium (MPP+)-induced insult in MES23.5 dopamineric cells. 1-Methyl-4-phenylpyridinium treatment decreased cell viability and dopamine content, as well as caused apoptotic morphological changes. 1-Methyl-4-phenylpyridinium-induced mitochondrial dysfunction, indicated by inhibition of activity associated with mitochondrial respiratory chain complex I, suggested mitochondrial transmembrane potential collapse and generation of reactive oxygen species. Decreased Bcl-2/Bax ratio and caspase 3 activation were also observed. Rosmarinic acid pretreatment restored the complex I activity of the mitochondrial respiratory chain and partially reversed the other damaging effects of MPP+. Our results indicate that RA plays a neuroprotective role by ameliorating mitochondrial dysfunction against MPP+-induced cell apoptosis and suggest that RA has the potential to be considered an aid for prevention of Parkinson’s disease.
Introduction
Parkinson’s disease (PD) is a chronic neurodegenerative disorder characterized by the loss of dopaminergic neurons of substantia nigra (SN) pars compacta, leading to dopamine (DA) depletion that is responsible for the clinical features of PD, including bradykinesia, resting tremor, and rigidity. 1 A number of biochemical processes and molecular mechanisms have been identified as mediators of neuronal cell death in PD, including oxidative stress, increased iron content, and mitochondrial dysfunction. 2 –5 These interrelated events form a complex cascade and finally lead to neuronal death by apoptosis. 2,3,6 –8 Since apoptosis has been implicated as one of the important mechanisms leading to the death of dopaminergic neurons in PD, the classic neurotoxin 1-methyl-4-phenylpyridinium (MPP+) is used to produce a PD cell model in vitro in this study. 1-Methyl-4-phenylpyridinium, a metabolite of 1-methyl-4-phenyl- 1,2,3,6-tetrahydropyridine (MPTP), is selectively transported into dopaminergic neurons through the DA transporter and concentrated into mitochondria, 9 inducing pathological changes resembling PD.
Rosmarinic acid (RA), a polyphenolic compound distributed in many medicinal plants, has a number of interesting biological activities, for example antivirus, antibacteria, anti-inflammation, and antioxidation. 10 It exhibits strong free radical scavenging activities 11 and potent antioxidant effects on low-density lipoprotein (LDL) oxidation. 12 Several studies demonstrated the protective effects of RA in cardiac muscle cells, H2O2-treated astrocytes, and amyloid-β peptide-treated PC12 cells. 13 -16 We previously reported that RA could exert its protective effects against 6-hydroxydopamine (6-OHDA)-induced neurotoxicity through its antioxidation properties. 17 However, whether RA could exert neuroprotective effects against MPP+-induced apoptosis remains unclear. So the aim of current study was to explore the protective effects of RA against MPP+-induced neurotoxicity in MES23.5 cells as well as the underlying mechanisms. MES23.5 cells were used in this study because they have at least 3 neuronal features, namely, tyrosine hydroxylase, the DA synthesis system, and omega-conotoxin receptor expression. 18 The cells exhibit several properties similar to primary neurons originating from the SNs.
Materials and Methods
Materials
Unless otherwise stated, all chemicals were purchased from Sigma Chemical Co (St Louis, Missouri); Dulbecco’s modified Eagle’s medium Nutrient Mixture-F12 (Ham; DMEM/F12) was from Gibco (Gibco, Grand Island, New York); Hoechst 33258 was from Beyotime (Jiangsu, China); lactate dehydrogenase (LDH) assay kit was from Jiancheng Technology (Jian-Cheng Biochemical Engineering Co, Nanjing, China); mitochondria isolation kit and mitochondrial respiratory chain complexes I activity colorimetric assay kit were from Genmed Scientifics (Genmed Scientifics Inc, Arlington, Massachusetts); USA Carboxy-H2DCFDA was from Molecular Probes (Molecular Probes Inc, Carlsbad, California); RA was provided by the Department of Biology in Qingdao University; and MES23.5 cell line was kindly provided by Dr Weidong Le (Departments of Neurology and Molecular Physiology and Biophysics, Baylor College of Medicine, Houston, Texas). All other chemicals and reagents were of the highest grade available from local commercial sources.
Cell Culture
MES23.5 cells is a dopaminergic cell line hybridized from murine neuroblastoma−glioma N18TG2 cells with rat mesencephalic neurons exhibiting several properties similar to the primary neurons originated in the SN.
18
They were cultured in DMEM/F12 growth medium supplemented with 5% fetal bovine serum (FBS), 2% Sato’s components, 1%
Cell Viability Assay
MES23.5 cells were seeded in a 96-well plate at a density of 1 × 104 cells/well. After attachment, cells were pretreated with RA (final concentration 10−9 mol/L) for 30 minutes, and MPP+ (final concentration 200 μmol/L) was added to DMEM/F12 without serum supplement for the subsequent 24 hours. After incubation in methyl thiazolyl tetrazolium (MTT) (5 mg/mL) for 4 hours at 37°C, cell viability was measured at 570 nm by colorimetric assay (Rayto RT-2100C, Shenzhen, China).
To confirm the results obtained by MTT assay, cell viability was also assessed by measuring the release of the cytosolic enzyme LDH to the culture medium using a colorimetric LDH assay kit in an automatic colorimetric assay (Rayto RT-2100C), according to the manufacturer’s instructions. Briefly, at the end of cell treatment, 20 μL of cell medium was added into basic solution to measure extracellular LDH activity, which could catalyze the conversion of lactate to pyruvate and then reacted with 2,4-dinitrophenylhydrazine to give the brownish red color. The absorbance was measured at wavelength 440 nm, and LDH leakage was expressed as the percentage versus control cells designated as 100%.
High-Performance Liquid Chromatography−Electrochemical Detection
Cells were seeded in 25 cm2 flask, pretreated with 10−9 mol/L RA for 30 minutes, followed by 200 µmol/L MPP+ treatment for 24 hours and then the samples were prepared as previously described, in our laboratory. 19 Separation was achieved on a PE C18 reversed-phase column. The mobile phase consisted of 20 mmol/L citromalic acid, 50 mmol/L sodium caproate, 0.134 mmol/L EDTA-2Na, 3.75 mmol/L sodium octane sulphonic acid, 1 mmol/L di-sec-butylamine, and contained 5% (v/v) methanol; the flow rate was 1 mL/min. Dopamine was determined by high-performance liquid chromatography (HPLC) equipped with a 2465 electrochemical detector (ECD) in direct current mode (Waters, USA). Results were presented as ng/106 cells.
Hoechst 33258 staining
Nuclear morphology was detected using the method previously described in our laboratory. 20,21 MES23.5 cells were seeded on sterile cover glasses in 24-well plates and treated as described above. Then the cells were fixed in 4% paraformaldehyde for 30 minutes, washed in phosphate-buffered saline, and stained with Hoechst 33258 dye for 30 minutes at room temperature. After washing 3 times to remove the excessive dye, the cells were examined and photographed under a fluorescence microscope (Olympus, Japan) with an excitation wavelength of 330 to 380 nm. Apoptotic cells were defined on the basis of nuclear morphological changes, such as chromatin condensation and fragmentation. The total number of condensed cells was counted manually by researchers blinded to the treatment schedule using unbiased stereology. For each well, we delineated a 400 µm2 frame and counted all condensed and normal nuclei in at least 10 different fields. Average sum of condensed and normal nuclei was calculated per well. The data were presented as the percentage of condensed nuclear number to the total number.
Mitochondrial Respiratory Chain Complexes I (NADH Dehydrogenase) Activity Assay
Mitochondria from MES23.5 cells treated as described above were isolated using a mitochondria isolation kit according to the manufacturer’s instructions. Nicotinamide adenine dinucleotide (NADH) dehydrogenase activity was determined at 340 nm by following the decrease in NADH absorbance that occurs when ubiquinone is reduced to ubiquinol (NADH is converted to oxidized NAD+). The reaction was started by adding the sample to the reaction mixture according to the manufacturer’s instructions. NADH dehydrogenase activity was expressed as changes in NADH level in micromoles per minute per milligram of protein.
Detection of Mitochondrial Transmembrane Potential (▵ΨM) and Intracellular Reactive Oxygen Species Production
Changes in the ▵ΨM and levels of intracellular reactive oxygen species (ROS) with various treatment in MES23.5 cells were measured by rhodamine123 or carboxy H2DCFDA using flow cytometry (Becton Dickinson, Franklin Lake, New Jersey, USA) as described before. 22,23 The uptake of rhodamine123 into mitochondria is an indicator of the ΔΨm. The fluorescence coming from carboxy H2DCFDA reflects the intracellular ROS generation. Cells treated as described above were incubated in 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES)-buffered saline (HBS) containing rhodamine123 or carboxy H2DCFDA in a final concentration of 5 μmol/L for 30 minutes at 37°C and followed by washing twice with HBS. For analysis, fluorescent intensity was recorded at 488 nm excitation and 525 nm emission wavelengths (fluorescence 1 [FL1]). Results were demonstrated as fluorescence 1-Histogram (FL1-H), setting of the gated region M1 and M2 as a marker to observe the changing levels of fluorescence intensity using Cellquest Software.
Total RNA Extraction and Reverse Transcriptase−Polymerase Chain Reaction
Total RNA was isolated using Trizol reagent (Invitrogen, California, USA) from MES23.5 cells treated as described above according to the manufacturer’s instructions. Then 5 µg of total RNA was reverse transcribed in a 20 µL reaction using the AMV reverse transcription system (Promega Corporation, Madison, Wisconsin). We amplified Bcl-2 complementary DNA (cDNA) fragment (amplified products were 409 bp length) with the primers (forward: 5'-GTC CCG CCT CTT CAC CTT-3'; reverse: 5'-CCC ACT CGT AGC CCC TCT-3'), Bax cDNA fragment (amplified products were 307 bp length) with the primers (forward: 5'-GGC GAA TTG GAG ATG AAC-3'; reverse: 5'-CCG AAG TAG GAG AGG AGG-3') and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) cDNA fragment (amplified products were 236 bp length) with the primers (forward: 5'-TTC ACC ACC ATG GAG AAG GC-3'; reverse 5'-GGC ATG GAC TGT GGT CAT GA-3'). The expression of housekeeping gene, GAPDH messenger RNA (mRNA), was used as an internal standard. Polymerase chain reactions (PCRs) were run for 36 cycles in an Eppendorf Mastercycler. Denaturing, annealing, and extension reactions were performed at 94°C for 30 seconds, 51°C for 30 seconds, and 72°C for 45 seconds. The PCR products were electrophoresed on 1% agarose gel, stained with ethidium bromide, and detected by UV irradiation. The levels of Bcl-2 and Bax mRNA were expressed as their respective ratios to GAPDH mRNA.
Measurement of Caspase 3 Activation
Caspase 3 activity was measured by flow cytometry using a PE-conjugated monoclonal active caspase 3 antibody apoptosis kit according to the manufacturer’s protocol (BD Biosciences, Franklin Lake, New Jersey, USA). Briefly, cells were seeded on 6-well plates and treated as described above. After washing twice with cold phosphate buffered saline (PBS), cells were resuspended in Cytofix solution at a concentration of 1 × 106 cells/0.5 mL and incubated for 20 minutes. The cells were then washed with Perm/Wash buffer twice and incubated in Perm/Wash buffer with antibody (1:5) for 30 minutes. After washing once with Perm/Wash buffer, cells were resuspended with 0.5 mL Perm/Wash buffer and analyzed by flow cytometry. The extent of apoptosis was determined by counting the number of active caspase 3 immunoreactive cells as a percentage of total MES23.5 cells using Cellguest Software.
Statistical Analysis
Data were analyzed using the SPSS12.0 software and expressed as mean ± standard error of the mean (SEM). One-way analysis of variance (ANOVA) followed by Student-Newman-Keuls test was used to compare the differences between means. A probability value of P < .05 was considered statistically significant.
Results
Rosmarinic Acid Protected MES23.5 Cells Against MPP+-Induced Cytotoxicity
Initial studies were performed to examine the potential cytotoxicity of RA in MES23.5 cells. Rosmarinic acid, 10−9 mol/L, did not show any cytotoxicity to MES23.5 cells (data not shown). To investigate whether RA could antagonize MPP+-induced cytotoxicity in MES23.5 cells, cells were pretreated with 10−9 mol/L RA for 30 minutes, followed by 200 µmol/L MPP+ treatment for 24 hours. As shown in Figure 1A, the cell viability in 200 µmol/L MPP+-treated group was decreased to 69.01% compared with control, however, it was significantly increased to 83.81% with 10−9 mol/L RA pretreatment. It implied that 10−9 mol/L RA could protect MES23.5 cells against MPP+-induced cytotoxicity. Lactate dehydrogenase assay further confirmed that 10−9 mol/L RA pretreatment for 30 minutes could significantly inhibit cytotoxicity induced by MPP+ as evident from less LDH leakage outside the cells (Figure1B). Accordingly, DA content was 2.45 ± 0.15, 1.38 ± 0.07, and 1.71 ± 0.05 ng/106 cells in the control, MPP+-treated, and RA-pretreated groups, respectively (Figure1C).
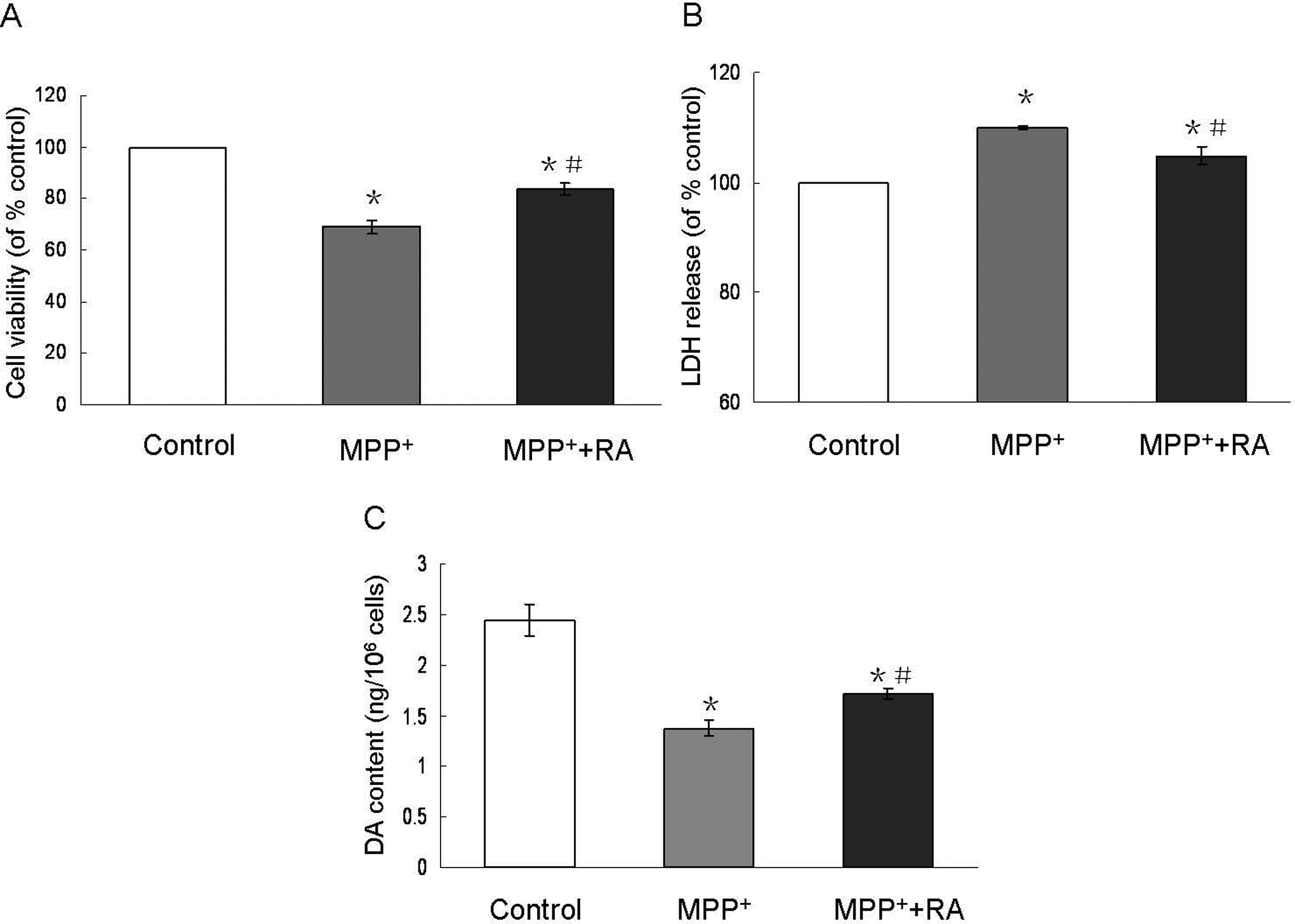
Changes in cell viability and DA content in MPP+-treated MES23.5 cells with RA pretreatment. Cell viability was determined by MTT assay (A) and LDH assay (B). The cell viability in 200 mol/L MPP+-treated group was reduced compared with the control. A significant increase in cell viability was observed when cells were pretreated with 10−9 mol/L RA for 30 minutes. Lactate dehydrogenase assay confirmed that 10−9 mol/L RA significantly inhibited LDH leakage outside the cells induced by 200 μmol/L MPP+ C, Dopamine (DA) content in 200 μmol/L MPP+-treated group was decreased, however, 10−9 mol/L RA pretreatment significantly attenuated MPP+-induced decreased DA content. Data were presented as means ± SEM of 6 independent experiments. *P < .05 compared with control; #P < .05 compared with MPP+-treated group (y axis should be DA content [ng/106 cells]). MPP+ indicates 1-methyl-4-phenylpyridinium; RA, rosmarinic acid; SEM, standard error of the mean.
Rosmarinic Acid Antagonized MPP+-Induced Cell Apoptosis Indicated by Hoechst 33258
Morphological changes of the cells in the above groups were further observed by Hoechst 33258 staining. MPP+ treatment, 200 μmol/L, caused nuclear condensation, 10−9 mol/L RA pretreatment significantly attenuated this effect (Figure 2A). The percentage of apoptotic cells in 200 µmol/L MPP+-treated group was 26.9% ± 2.04%, however, with 10−9 mol/L RA pretreatment this number decreased to 11.7% ± 1.05% (Figure 2B). Meanwhile, only a small part of apoptotic nuclei changes could be observed in the control group (5.63% ± 0.96%).
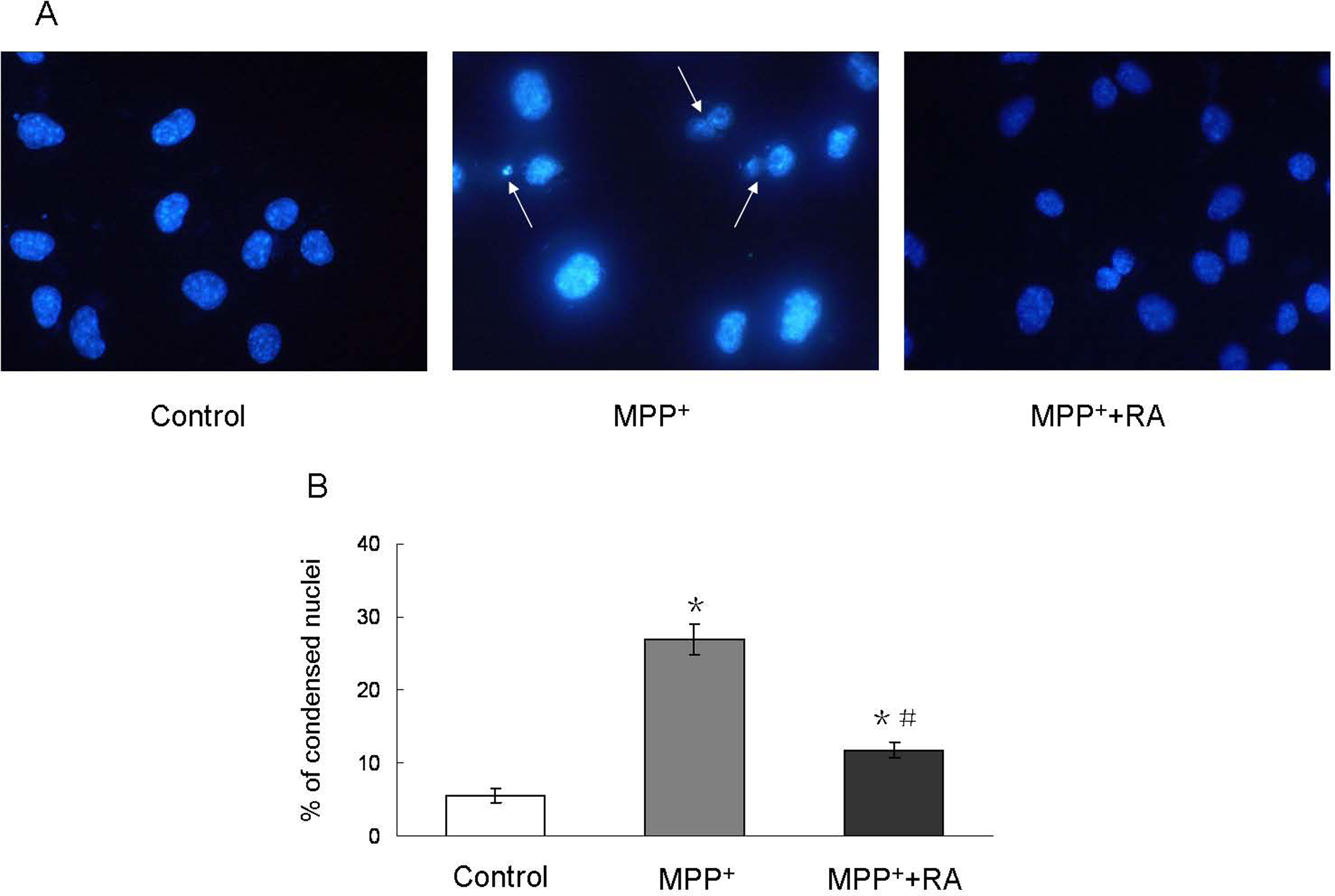
Morphological changes in MES23.5 cells with different treatments. A, Representative photographs of Hoechst 33258 staining from control, MPP+- treated and RA-pretreated groups. MPP+ treatment resulted in nuclear condensation, however, 10−9 mol/L RA pretreatment significantly attenuated MPP+-induced nuclear condensation. Magnification ×400. B, Statistical analysis. Data were presented as means ± SEM of 6 independent experiments. *P < .05 compared with control; #P < .05 compared with MPP+-treated group. MPP+ indicates 1-methyl-4-phenylpyridinium; RA, rosmarinic acid; SEM, standard error of the mean.
Rosmarinic Acid Restored MPP+-Induced Inhibition of Mitochondrial Respiratory Chain Complexes I Activity
Since MPP+ exerts its neurotoxicity mainly by inhibiting the activity of mitochondrial respiratory chain complexes I, 9,24 we then analyzed the activity of mitochondrial respiratory chain complexes I to evaluate the specific neuroprotection of RA against MPP+ insult. As shown in Figure 3 , a significant inhibition of mitochondrial respiratory chain complexes I activity as indicated by changes of NADH level was observed in 200 µmol/L MPP+-treated group, and this effect could be completely blocked by 10−9 mol/L RA pretreatment.
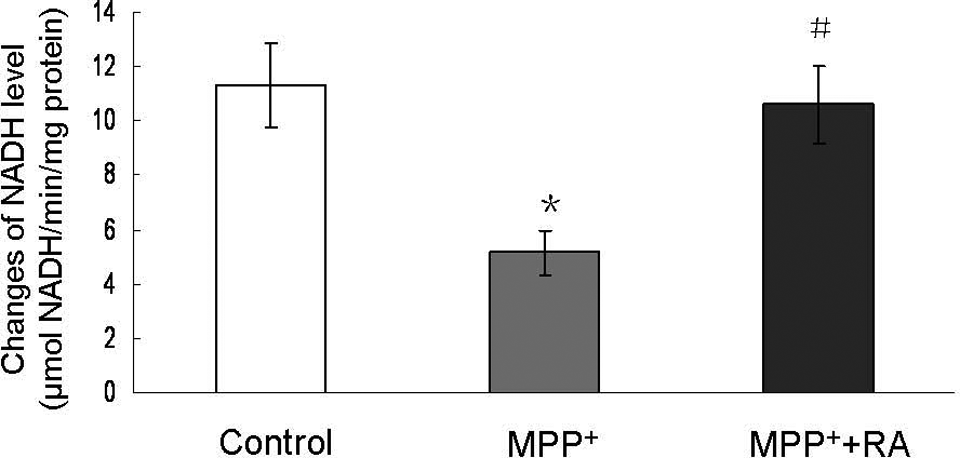
Changes of NADH level in MPP+-treated MES23.5 cells with RA pretreatment. Rosmarinic acid pretreatment completely blocked MPP+-induced inhibition of mitochondrial respiratory chain complexes I activity. NADH dehydrogenase activity was expressed in micromoles per minute per milligram of protein (μmol NADH/min per mg protein). Data were presented as means ± SEM of 4 independent experiments. *P < .05 compared with control; #P < .05 compared with MPP+-treated group. MPP+ indicates 1-methyl-4-phenylpyridinium; RA, rosmarinic acid; NADH, nicotinamide adenine dinucleotide; SEM, standard error of the mean.
Rosmarinic Acid Prevented MPP+-Induced Mitochondrial Transmembrane Potential (ΔΨm) Decrease and ROS Generation
Mitochondrial transmembrane potential is the marker of mitochondria function, which is involved in a variety of key events in oxidative stress and apoptosis. 25 We measured the mitochondrial transmembrane potential in MPP+-treated cells and RA-pretreated cells. As shown in Figure 4A and C, 200 µmol/L MPP+ significantly reduced ΔΨm, whereas pretreatment with 10−9 mol/L RA could partially block this effect. Since ROS played an important role in cell apoptosis and changes in mitochondrial transmembrane potential were considered involved in ROS production, we also measured ROS production using fluorescent dye H2DCFDA. As shown in Figure 4B and D, the levels of intracellular ROS markedly increased with MPP+ treatment. Rosmarinic acid, 10−9 mol/L, could significantly suppress MPP+-induced ROS production. These suggested that 10−9 mol/L RA could protect cells from MPP+-induced neurotoxicity by restoring the mitochondria function.
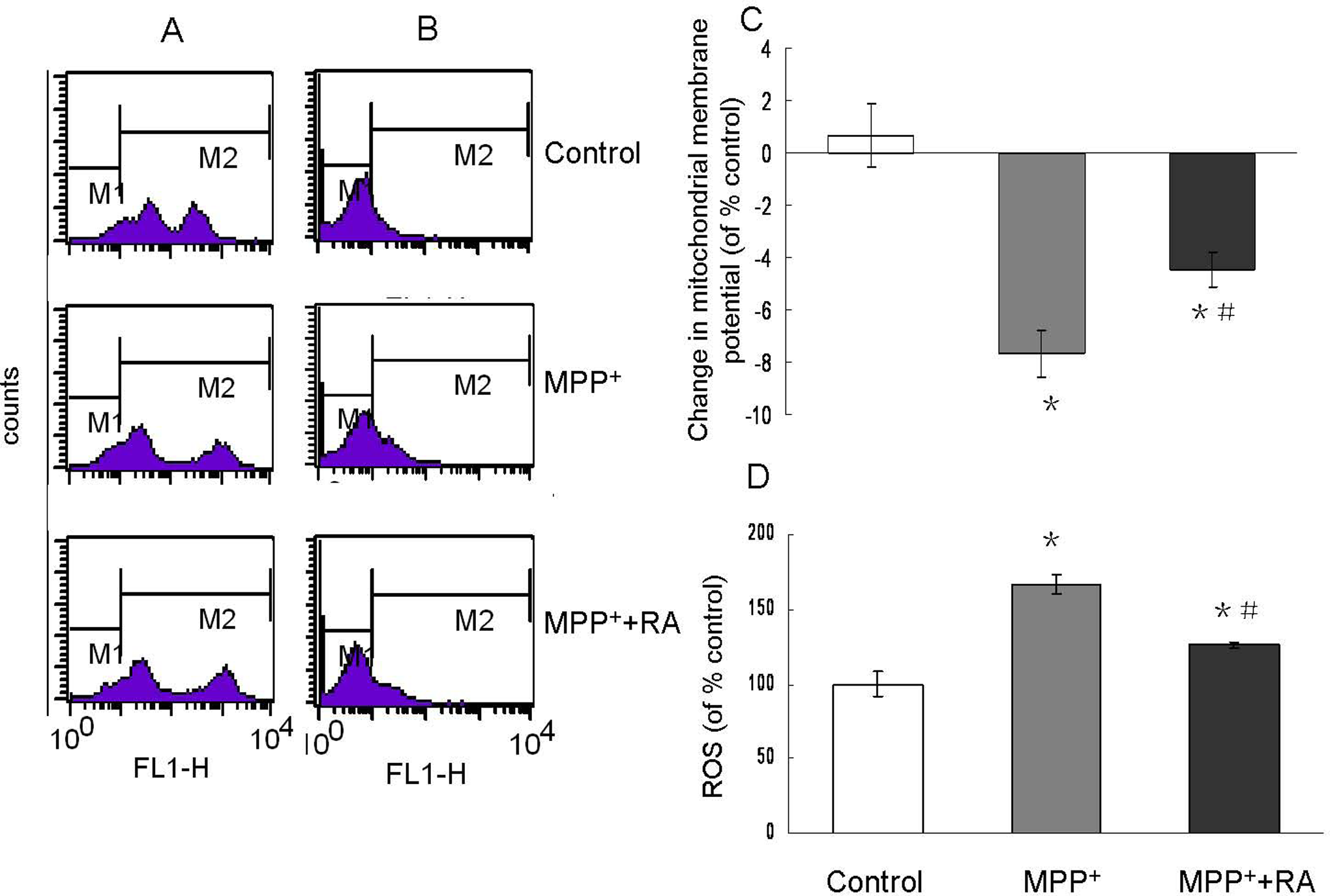
Mitochondrial transmembrane potential (▵Ψm) and ROS generation assessed by flow cytometry in MPP+-treated MES23.5 cells with RA pretreatment. A/B, Representatives of the fluorometric assay on ▵Ψm and ROS generation of different groups. 10−9 mol/L RA pretreatment significantly prevented the ▵Ψm reduction and ROS generation induced by MPP+. C/D, Statistical analysis. Data were presented as means ± SEM of 3 independent experiments. Fluorescence values of the control were set to 100%. *P < .05 compared with control; #P < .05 compared with MPP+-treated group. MPP+ indicates 1-methyl-4-phenylpyridinium; RA, rosmarinic acid; SEM, standard error of the mean; ROS, reactive oxygen species.
Rosmarinic Acid Antagonized MPP+-Induced Downregulation of Bcl-2/Bax Ratio
The Bcl-2 family of proteins is associated with mitochondrial function during apoptosis. Bcl-2, as an antiapoptotic member of the Bcl-2 family, can bind Bax to form Bcl-2:Bax heterodimers, thereby attenuating the proapoptotic effect of Bax. 26 MPP+ treatment, 200 µmol/L, caused a dramatic downregulation of Bcl-2 mRNA and upregulation of Bax mRNA (Figure 5A and B), resulting in the downregulation of Bcl-2/Bax ratio. Rosmarinic acid pretreatment could inhibit the decrease of Bcl-2 and the increase of Bax induced by MPP+, with the ultimate increased Bcl-2/Bax ratio (Figure 5C).
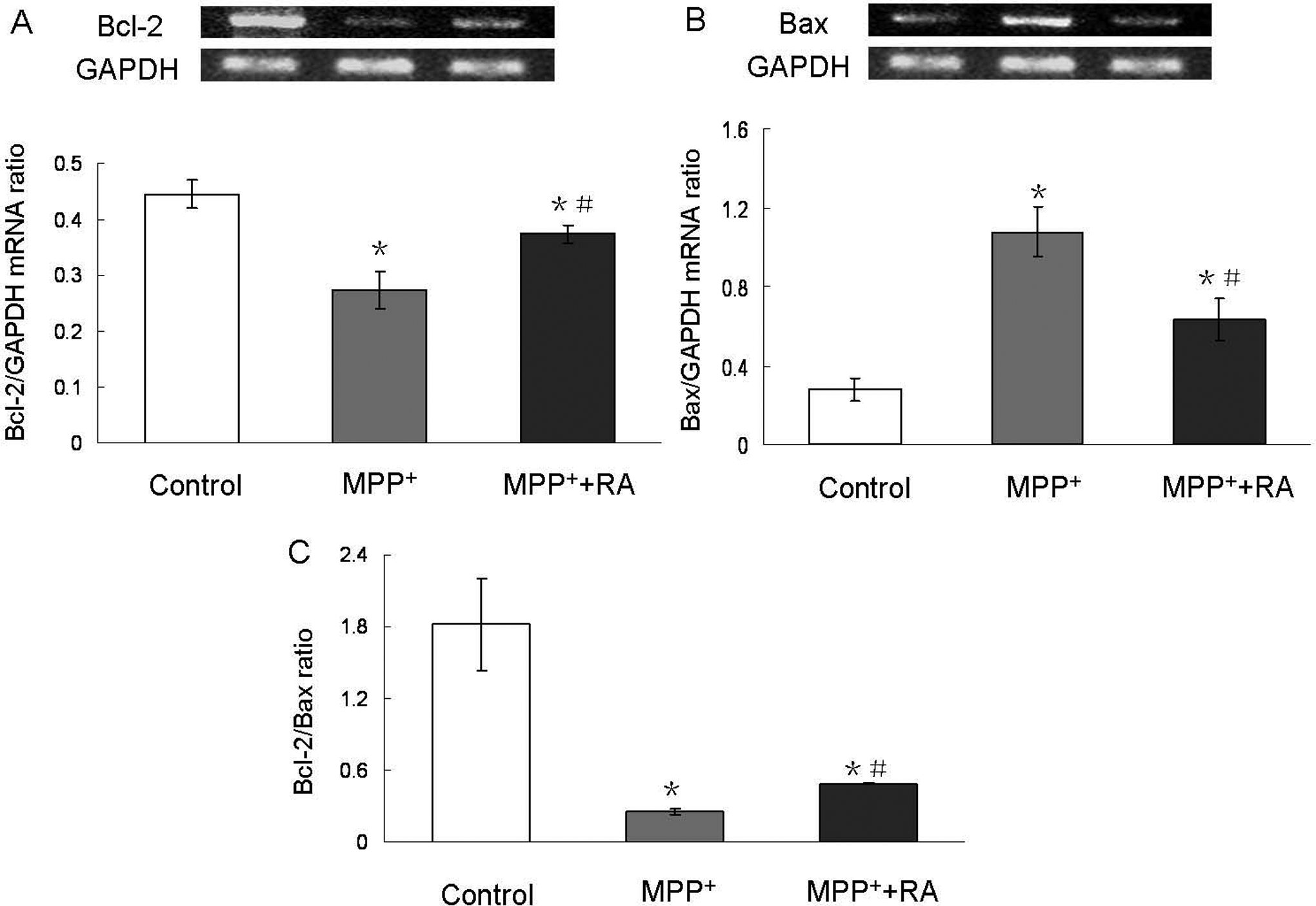
Changes of Bcl-2 and Bax mRNA level in MPP+-treated MES23.5 cells with RA pretreatment. Rosmarinic acid pretreatment blocked MPP+-induced Bcl-2 mRNA level downregulation (A), Bax mRNA level upregulation (B), and downregulation of Bcl-2/Bax ratio (C). Bcl-2 and Bax band intensities were normalized with GAPDH band intensity. Each value represented the means ± SEM. *P < .05 compared with control; #P < .05 compared with MPP+-treated group. MPP+ indicates 1-methyl-4-phenylpyridinium; RA, rosmarinic acid; SEM, standard error of the mean; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Rosmarinic Acid Attenuated MPP+-Induced Caspase 3 Activation
Caspase 3 is a key protein in the process of apoptosis. Consistent with the above observation, inactivation of the effector caspases was also observed with RA pretreatment. As shown in Figure 6 , cells incubated with 200 µmol/L MPP+ resulted in a 2-fold increase in caspase 3 activity, and this effect could be significantly attenuated by 10−9 mol/L RA pretreatment.
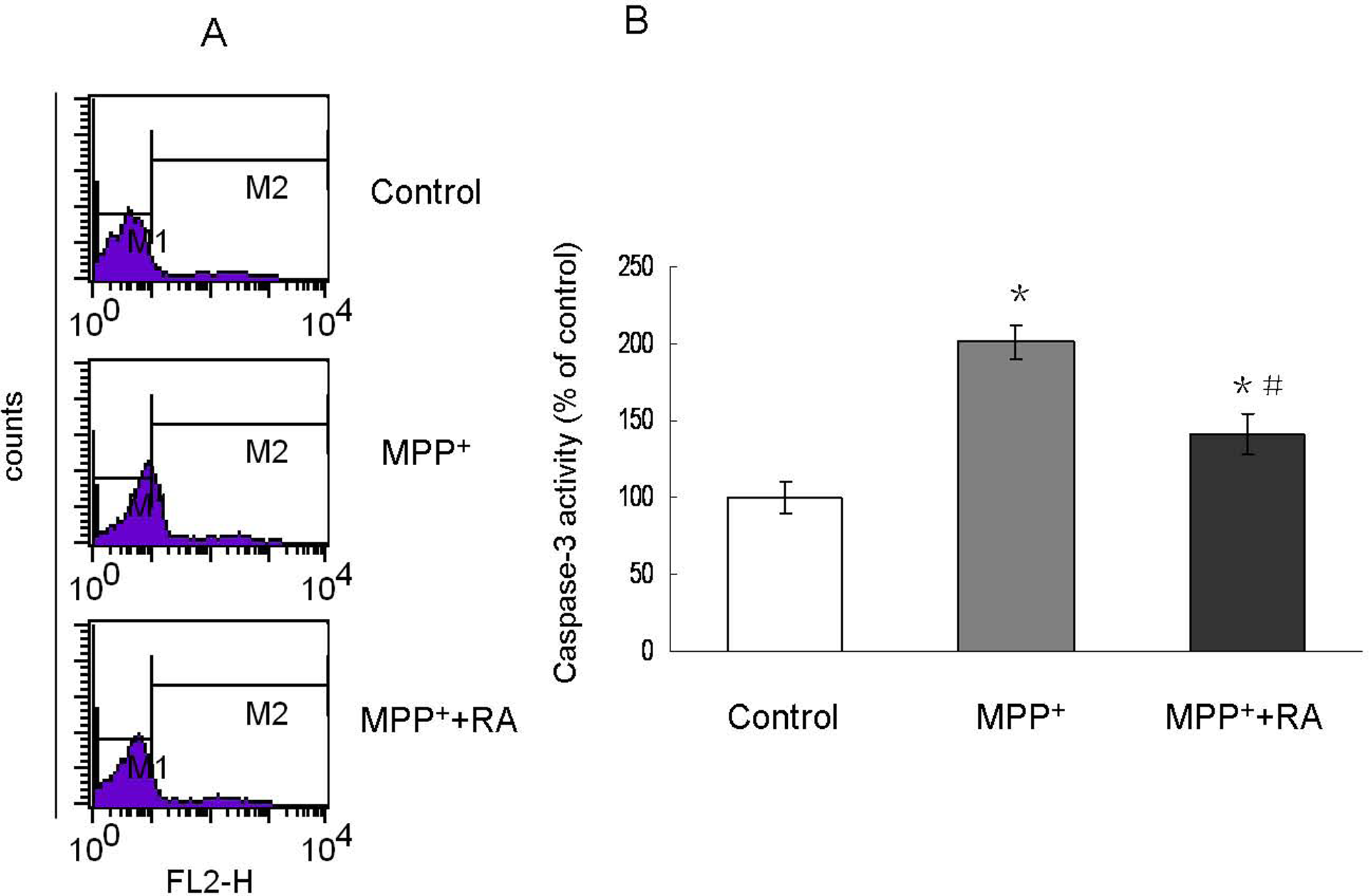
Caspase 3 activation assessed by flow cytometry in MPP+-treated MES23.5 cells with RA pretreatment. A, Representatives of the fluorometric assay on active caspase 3 of different groups. 10−9 mol/L RA pretreatment significantly attenuated caspase 3 activity increase induced by MPP+. B, Statistical analysis. Data were shown as percentage of control and presented as mean ± SEM of 3 independent experiments. *P < .05 compared with control; #P < .05 compared with MPP+-treated group. MPP+ indicates 1-methyl-4-phenylpyridinium; RA, rosmarinic acid; SEM, standard error of the mean.
Discussion
Recently, the involvement of oxidative stress in the etiology and progression of neurodegenerative diseases including PD, has led to interest in the use of naturally occurring antioxidants. They are perceived as safe and functional compounds for neurodegenerative diseases therapy. Among these, it was reported that RA is the most potent antioxidant in the hydroxylcinnamic group of polyphenols. 27 It was reported that RA displayed a strong antiviral and anti-inflammatory effect against Japanese encephalitis and diesel exhaust particles-induced lung injury. 28,29 In the current study, we showed that RA could exert its antiapoptotic effects against MPP+-induced neuronal cell apoptosis in MES23.5 dopaminergic cells by ameliorating mitochondrial dysfunction, mainly through restoring the mitochondrial respiratory chain complexes I activity. Accordingly, the intracellular DA content was significantly recovered after RA pretreatment.
1-Methyl-4-phenylpyridinium is widely used as a common neurotoxin for inducing PD models. The neurotoxicity of MPP+ was due to its uptake by dopaminergic neurons and accumulation by electrochemical gradient to intramitochondria. By this way, MPP+ in cytoplasma was first concentrated into mitochondria and then penetrated into hydrophobic binding site on NADH dehydrogenase (complex I) and thus inhibited the latter activity.
30
This eventually causes dopaminergic cell function loss due to the inhibition of electron transport chain. In the current study, we observed that RA could completely restore mitochondrial respiratory chain complexes I activity, which might be due to the interruption of these 2 steps: (1) RA blocked the concentration of MPP+ into mitochondria and (2) RA competitively combined to hydrophobic binding site on NADH dehydrogenase. With a low concentration reached in mitochondria or low binding to complex I or both, MPP+-induced mitochondrial respiratory chain complexes I activity inhibition was fully blocked by RA pretreatment, because complex I damage only occurs at a high concentration of MPP+ in mitochondria.
30
This suggested that the restoration of mitochondrial respiratory chain complexes I activity might contribute to the neuroprotective effects of RA, in accordance with a previous study that MPP+ induced an increase of intracellular net NADH content in SH-SY5Y cells and
Mitochondrial respiratory chain complexes I activity is closely related to mitochondrial functions. One of the events reflecting mitochondrial functions is the changes in mitochondrial transmembrane potential. The mitochondrial transmembrane potential collapse induces the formation of ROS, and further ROS formation damages the mitochondrial membrane, such mitochondrial dysfunctions are implicated as key events in the pathogenic cascades leading to apoptosis. 25,32,33 It is well known that the Bcl-2 family of proteins plays an important role in intracellular apoptotic signal transduction by regulating the permeability of the mitochondrial membrane. 34 They are also involved in the regulation of cleavage and activation of effector caspases, such as caspase 3, which is the main executioner caspase inducing cell apoptosis. 35 In this study, despite the complete restoration of mitochondrial respiratory chain complexes I activity, MPP+-induced decrease of the mitochondrial transmembrane potential and increased ROS generation, as well as proapoptotic Bax upregulation, antiapoptotic Bcl-2 downregulation and caspase 3 activation in MES23.5 were only partially attenuated with RA pretreatment. This indicated that mitochondrial dysfunctions and the subsequent classic apoptotic pathway activation were still present in RA-pretreated cells. We supposed that this might be due to superoxide and hydroxyl radicals generated during intracellular reduction and reoxidation of MPP+, which could not be fully blocked by RA. However, even a higher concentration of RA pretreatment did not show greater effects (data not shown), indicating that other factors undefined could also be involved in the limitation of protective processes regulated by RA. Since DA-dependent oxidative stress is also involved in MPP+ toxicity, 36 the possibility that RA could not fully block MPP+-induced DA oxidation indicated that another mitochondrial-independent mechanism might contribute to the partial protection against MPP+ conferred by RA.
In conclusion, our results demonstrated that RA could antagonize the MPP+-induced neurotoxicity in MES23.5 cells via ameliorating the mitochondrial dysfunction, with completely restoring the mitochondrial respiratory chain complexes I activity. Despite the positive results in vitro, since the metabolites might interfere the effects of RA, 37 application of RA in vivo also needs to be further explored. Our results suggest that RA may be a novel therapeutic strategy for the preventive and/or complementary therapies of PD.
Footnotes
Acknowledgments
We thank Dr Wei-dong Le to give us the MES23.5 cell line.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the Natural Science Foundation of Shandong Province (Z2008C01).
