Abstract
S-111-S-WB, a mixture of perfluoro fatty acid ammonium salts (C6-C13), was administered orally to Crl:CD (SD)IGS-BR rats. Higher hepatic β-oxidation and liver weights with hepatocellular hypertrophy were present at the 0.125 and 0.6 mg/kg/d dosage. The 0.6 mg/kg/d males developed hepatocellular degeneration and necrosis. Lower serum protein and higher bilirubin and BUN were seen in the 0.6 mg/kg/d males and lower globulin and higher alkaline phosphatase in the 0.125 mg/kg/d males and 0.6 mg/kg/d animals. After 2 weeks, serum concentrations of pentadecafluorooctanoic acid (C8), heptadecafluorononanoic acid (C9), perfluoroundecanoic acid (C11), and perfluorotridecanoic acid (C13) were constant for at least 8 hours. After 90 days, only C9 in the 0.025 mg/kg/d females had reached steady state. Serum C8 and C9 concentrations in the males were 10-fold higher than in the females, whereas C11 and C13 were similar for both genders. The main elimination was via the urine for C8 (males) and C9 (females), and via the feces for C11 and C13. The no-observed-effect level (NOEL) was 0.025 mg/kg/d for the males and 0.125 mg/kg/d for the females.
Introduction
S-111-S-WB (CAS No 72968-38-8) is a mixture of perfluoro fatty acid ammonium salts of different carbon chain lengths (C6-C13) that is used as an industrial processing aid. Perfluoro fatty acids and their salts have received considerable attention following reports that serum samples from individuals in the United States and around the globe have been shown to contain the perfluoroalkyl acid, perfluorooctanoic acid (PFOA). Samples from all age groups examined in the United States have been shown to contain PFOA at a mean concentration of approximately 5 ppb. 1 –3
Extensive toxicology data are available for PFOA. Data ranging from acute to chronic studies have been summarized in literature reviews and in hazard and risk assessments by regulatory and other authoritative bodies. In rats, oral PFOA is moderately toxic showing body weight loss, liver weight increase, liver effects reflected by higher serum transaminase levels, and diffuse hepatocellular hypertrophy at higher doses of acidophilic degeneration, and/or necrosis of the liver. Male rats are more sensitive than female rats as a result of a more efficient excretion which is primarily urinary (for review see Kennedy et al 4 ).
Toxicology data for perfluoroalkyl acids with longer carbon chain length than PFOA, such as perfluorononanoate (PFNA) the major component of S-111-S-WB, are much more limited. Perfluorononanoate (PFNA) and perfluorodecanoate (PFDA), like PFOA, have been shown to be peroxisome proliferators. 5 The acute oral lethal dose of PFDA is 4.6-fold lower than the estimated LD50 for PFOA, 6 whereas the approximate lethal dose for S-111-S-WB is 2.9-fold lower (unpublished data), suggesting that carbon chain length may be an important factor in determining the toxicity of perfluoroalkyl acids and salts. As part of a product stewardship program, this 90-day toxicology study in Sprague Dawley rats was conducted in accordance with United States Environmental Protection Agency (US EPA) and Organisation for Economic Co-operation and Development (OECD) guidelines to provide additional toxicology data on longer carbon chain length perfluoroalkyl acids as present in S-111-S-WB. A 2-generation reproductive toxicity study of S-111-S-WB in rats was conducted as well and was published in a separate manuscript. 7
Methods
Test Substance
The test substance, S-111-S-WB (CAS No 72968-38-8), is a mixture of perfluoro fatty acid ammonium salts of different carbon chain lengths (C6-C13). The major component is the ammonium salt of perfluorononanoic acid (PFNA, C9). S-111-S-WB was received from Solvay Solexis, Thorofare, New Jersey. It is a clear and colorless liquid and was formulated in deionized water. The test substance was stored at room temperature and was considered stable under this condition. Dosing formulations, to provide dosages of 0.025, 0.125, and 0.6 mg/kg/d (mg of active test substance per kg body weight per day), were stable following 1 day and 8 days refrigerated storage conditions. Formulations at the same concentrations were homogenous and contained the amounts of S-111-S-WB specified, confirmed by weekly concentration analyses for the first 4 weeks and monthly analyses thereafter.
Animals, Housing, and Randomization
Male and female Crl:CD (SD)IGS BR rats, approximately 45 days old, were received from Charles River Laboratories, Inc (Raleigh, North Carolina) and acclimated for 13 days prior to use.
On arrival, all animals were housed individually in clean, stainless steel, wire-mesh cages suspended above cage board. Animals for the toxicokinetic assessments were moved into clean Nalgene metabolism cages prior to urine and feces collections. All animals were kept under controlled environmental conditions (average daily temperatures ranged from 21.4°C to 22.1°C, average daily relative humidity ranged from 36.4% to 56.1%, 12/12-h light/dark cycle, and a minimum of 10 fresh air changes per hour). Animals had ad libitum access to reverse osmosis-purified water either from the automatic watering system or from water bottles when housed in metabolism cages. PMI Nutrition International, LLC Certified Rodent LabDiet was available ad libitum except for periods of fasting prior to clinical pathology blood collections. Animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals (National Research Council, 1996). The animal facilities at WIL Research Laboratories, LLC are accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC International).
Animals were observed twice daily for changes in appearance or behavior, body weights were recorded, feed consumption was determined, and general health was monitored. All animals that did not appear healthy were eliminated from a computerized randomization procedure. Individual group assignments were generated based on body weight stratification in a block design and individual body weights at randomization were within ±20% of the mean for each sex.
Study Design
S-111-S-WB was administered orally by gavage once daily to 3 toxicology groups (groups 2-4; main study animals) for a minimum of 90 consecutive days (starting on day 0, week 0), 3 peroxisomal proliferation satellite groups (groups 2A-4A) for 10 consecutive days, 3 single-dose toxicokinetic groups (groups 2B-4B), and 3 repeated dose toxicokinetic groups (groups 2C-4C) for a minimum of 90 consecutive days. The animals in the satellite groups were used only for the indicated purposes (Table 1).
Study Group Overview
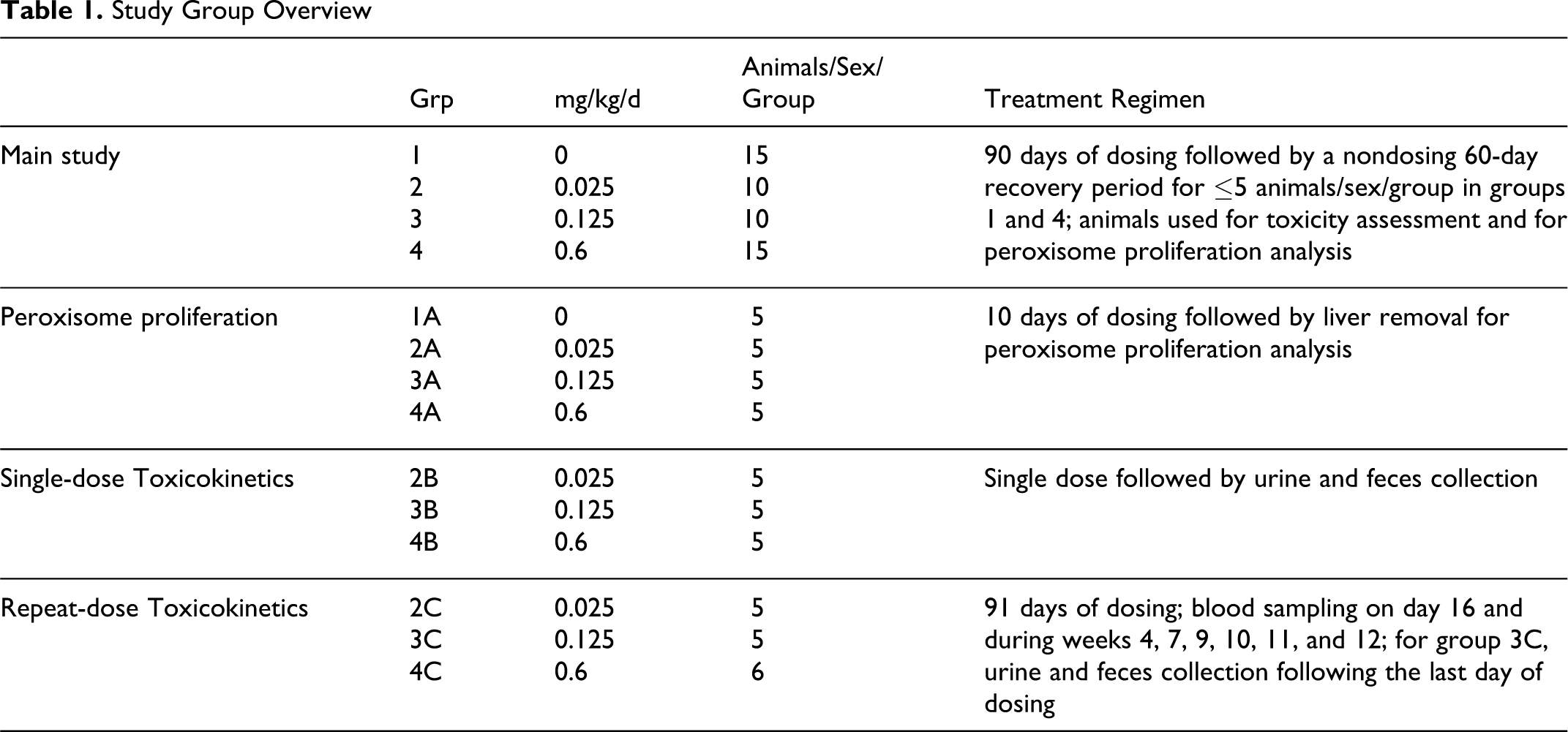
Dosage levels, selected based on results of an OECD 422 reproductive/developmental toxicity screening study, 8 were 0.025, 0.125, and 0.6 mg/kg/d and were delivered using a dosage volume of 2 mL/kg. Concurrent control groups (groups 1 and 1A) received deionized water on a comparable regimen. Groups 1 and 4 each consisted of 15 animals/sex; groups 2 and 3 each consisted of 10 animals/sex; groups 1A-4A, 2B-4B, and 2C-3C each consisted of 5 animals/sex; and group 4C consisted of 6 animals/sex. Following 90 days of dose administration, 10 rats/sex/group (toxicology groups) were euthanized; the remaining ≤5 rats/sex in the control and high-dose groups were euthanized following a 60-day nondosing (recovery) period.
For toxicology assessment, all main study animals (groups 1-4) were observed twice daily for mortality and moribundity prior to and during treatment. On initiation of dose administration, clinical examinations were performed daily prior to and approximately 1 hour following dose administration and detailed physical examinations were performed weekly. Individual body weights were collected weekly on all animals and feed consumption was determined weekly for the main study animals only.
Functional Observational Battery
Functional observational battery (FOB) assessments were performed for all main study animals without knowledge of the animals' group assignments during study weeks 12 and 21 (recovery animals). The FOB assessments were performed in a sound-attenuated room equipped with a white-noise generator set to operate at 70 ± 10 dB. The assessments included home cage, handling, open field, sensory, neuromuscular, and physiological observations and were based on previously developed protocols. 9 –14
Locomotor Activity
Locomotor activity was assessed for all main study animals during study weeks 12 and 21 (recovery animals) using the SDI Photobeam Activity System (San Diego Instruments, Inc, San Diego, California). Total and ambulatory movements were measured using a test session duration of 60 minutes (12 × 5 minutes intervals; 4 × 15 minutes epochs).
Ophthalmic Examinations
Ocular examinations were conducted on all main study animals prior to the start of treatment, near the end of treatment, and near the end of recovery by a board-certified veterinary ophthalmologist using an indirect ophthalmoscope and slit lamp biomicroscope preceded by pupillary dilation with an appropriate mydriatic agent.
Clinical Pathology: Hematology, Clinical Chemistry, and Urinalysis
Animals were fasted overnight prior to blood collection while in metabolism cages for urine collection. Blood was collected from the vena cava at the time of necropsy (study weeks 13 and 21) into tubes containing EDTA (hematology), sodium citrate (clotting determination), or no anticoagulant (serum chemistry).
Hematological parameters (groups 1-4) determined included total leukocyte and erythrocyte count, hemoglobin, hematocrit, mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), platelet count, prothrombin time, activated partial thromboplastin time (APTT), reticulocyte count, differential leukocyte count, platelet estimate, and red cell morphology. Hematological parameters were assayed using a Bayer Advia 120 analyzer and platelet estimate and differential leukocyte count were verified manually when necessary; prothrombin time and APTT were measured using a MLA Electra 1400 C automated coagulation analyzer. Clinical chemistry parameters determined included blood levels of albumin, total protein, globulin (calculated), albumin/globulin ratio (A/G ratio), total bilirubin, urea nitrogen, creatinine, alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, gamma glutamyltransferase, glucose, total cholesterol, calcium, chloride, phosphorus, potassium, and sodium using a Hitachi 912 analyzer. Urinalysis included macroscopic (total volume, color, appearance, and specific gravity) and microscopic (sediment) examination and determination of pH, urobilinogen, protein, glucose, ketones, bilirubin, occult blood, leukocytes, and nitrites using Ames CLINITEK 200+ reagent strips.
Macroscopic Examination and Organ Weights
A complete necropsy was conducted on all animals in the toxicology groups (groups 1-4) after euthanasia by isoflurane anesthesia followed by exsanguination. Necropsies included, but were not limited to, examination of the external surface, all orifices, and the cranial, thoracic, abdominal, and pelvic cavities, including viscera. Organ weights (adrenal glands, brain, heart, kidneys, liver, spleen, thymus, testes, epididymides, ovaries with oviducts, and uterus) were recorded and appropriate organ weight ratios were calculated (relative to body and brain weights). In addition, liver-to-body weight ratios were determined for peroxisomal proliferation satellite animals.
Microscopic Examination
A full complement of organs and tissues for groups 1 to 4 was prepared and microscopically examined by a veterinary pathologist. Microscopic examination was performed on all tissues in the control and 0.6 mg/kg/d groups at the primary necropsy and the liver, duodenum, and stomach (glandular and nonglandular) were examined from all males in the 0.025 and 0.125 mg/kg/d groups at the primary necropsy and from all males at the recovery necropsy.
Peroxisomal Proliferation Analysis
Livers were weighed and collected from all peroxisomal proliferation satellite group animals (groups 1A-4A) following the 10th day of dosing and from all animals/group in the toxicology groups (groups 1-4) at the scheduled necropsies. Animals (groups 1A-4A) were euthanized by isoflurane anesthesia followed by exsanguination. Livers were removed from the animal, rinsed in chilled saline, blotted dry, and weighed. As soon as possible after weighing, a liver sample was wrapped in aluminum foil and snap frozen in liquid nitrogen. Samples were stored frozen between −65°C and −85°C until shipment on dry ice to DuPont Haskell Laboratory for Health & Environmental Sciences for peroxisome proliferation analysis. Livers were homogenized (1 g tissue/4 mL buffer) in 50 mmol/L Tris-HCl, 50 mmol/L Trizma-base, 0.25 mol/L sucrose, and 5.4 mmol/L EDTA, pH 7.4 buffer. Hepatic peroxisomes were prepared using differential centrifugation. Resulting peroxisomal pellets were resuspended in the buffer and aliquots were stored between −65°C and −85°C until analyzed for peroxisomal β-oxidation activity. The peroxisomal suspensions were diluted to a protein concentration of approximately 0.5 mg/mL. Peroxisomal β-oxidation activity was determined using [14C] palmitoyl CoA as the substrate. 15 Protein content of the peroxisomes was determined before and after analysis using a method by Bradford. 16
Toxicokinetic Determinations (Single Dose)
For determination of urinary and fecal elimination after a single dose, 3 groups of 5 animals/sex received a single dose of S-111-S-WB at dosage levels of 0.025, 0.125, or 0.6 mg/kg/d. Following dosing, animals were housed in plastic metabolism cages and urine and feces were collected separately at 0 to 6, 6 to 12, and 12 to 24 hours after dosing and then daily through 7 days after dosing. Animals were euthanized following the last urine and fecal collection. Urine and fecal samples were analyzed for concentration of pentadecafluorooctanoic acid (C8), heptadecafluorononanoic acid (C9), perfluoroundecanoic acid (C11), and perfluorotridecanoic acid (C13) using validated high-performance liquid chromatography (HPLC) tandem mass spectrometry (HPLC/MS/MS) methods (Hewlett Packard 1100 LC with attached Quattro Ultima Mass Spectrometer). The total amount of each analyte excreted was calculated based on the mass of excreta.
Toxicokinetic Determinations (Repeat Dose)
For determination of serum concentrations and urinary and fecal elimination after repeated dosing, 2 groups of 5 animals/sex received daily oral doses of S-111-S-WB at dosage levels of 0.025 and 0.125 mg/kg/d and a group of 6 animals/sex received 0.6 mg/kg/d for 92 consecutive days. Blood samples (approximately 1 mL) were collected from the first 3 animals/sex in the 0.6 mg/kg/d group at 1 and 4 hours after dosing and the next 3 animals/sex in the 0.6 mg/kg/d group at 2 and 8 hours after dosing on study day 16 to determine sampling times for subsequent blood collections. During weeks 4, 7, 9, 10, 11, and 12, blood samples were collected from 5 animals/sex/group at approximately 2 hours after dosing. Samples were collected from the retro-orbital sinus while the animals were under isoflurane anesthesia. Serum was obtained by centrifugation. Animals in the 0.025 and 0.6 mg/kg/d groups were euthanized following the final blood collection. After dosing on day 91, animals in the 0.125 mg/kg/d group were housed in plastic metabolism cages and urine and feces were collected separately at 0 to 6, 6 to 12, and 12 to 24 hours after dosing and then daily through 7 days after dosing. These animals were euthanized following the final urine and fecal collection on day 98. All samples were stored at approximately −20°C and concentrations of C8, C9, C11, and C13 were measured using validated HPLC/MS/MS methods.
Statistics
All statistical tests were performed using appropriate computing devices or programs. Analyses were conducted using 2-tailed tests for minimum significance levels of 1% and 5%, comparing each test article-treated group to the control group by sex. Body weight, body weight change, food consumption, continuous FOB, locomotor activity, clinical pathology, and organ weight data were subjected to a parametric 1-way analysis of variance (ANOVA) 17 to determine the intergroup differences. If the ANOVA revealed statistically significant (P < .05) intergroup variance, Dunnett test 18 was used to compare the test article-treated groups to the control group. Functional observational battery parameters that yielded scalar or descriptive data were analyzed using Fisher exact test. 19 Clinical pathology values for white blood cell types that occur at a low incidence (ie, monocytes, eosinophils, and basophils) were not subjected to statistical analysis.
Results
Clinical Observations and Survival
All animals survived to the scheduled necropsies. S-111-S-WB-related clinical findings were generally limited to 2 males in the 0.6 mg/kg/d group. Beginning on days 72 and 79, these animals were noted with dermal atonia, thin bodies, decreased defecation, and smaller than normal feces. These observations correlated with lower body weight and food consumption. One of these males was also noted with hypoactivity (study days 80 and 81), pale extremities (study days 80, 81, 84, and 88), and partial closure of eyes (study day 81). No other S-111-S-WB-related clinical observations were noted during dosing or recovery periods.
Body Weights and Food Consumption
S-111-S-WB-related effects on body weight were observed in the 0.6 mg/kg/d group males with partial recovery noted by the end of the nondosing period (Figure 1). Statistically significantly lower mean body weight gains and/or mean body weight losses were observed in the 0.6 mg/kg/d group beginning on study weeks 2 to 3. By the end of the treatment period, mean body weight in this group was 24% lower compared with the control group. During the recovery period, higher mean body weight gains (generally statistically significant) were noted in these males when compared with the control group. As a result, the mean body weight was only 12.5% lower than control by the end of the recovery period. No effects on body weights were noted among the females. A slightly lower mean food consumption (predominately due to 2 animals) was noted in the 0.6 mg/kg/d group males during the last 4 weeks of the dosing period. No other S-111-S-WB-related effects on food consumption were noted throughout the study.
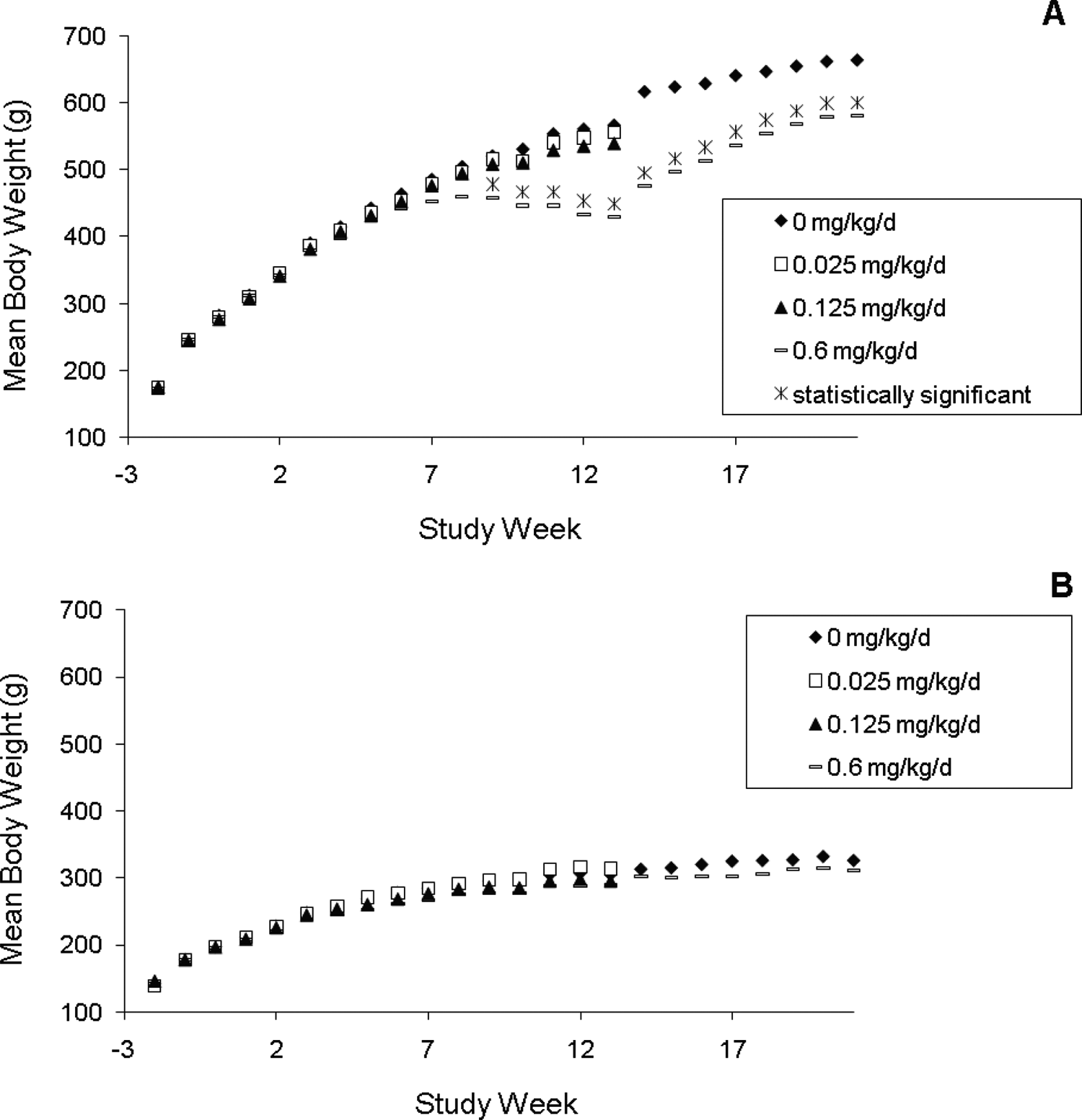
Mean body weights of male (A) and female (B) Sprague Dawley rats orally dosed with S-111-S-WB for 90 days followed by a 60-day nondosing recovery period. Asterisk indicates a statistically significant difference from control value, P < .05 or .01.
Functional Observational Battery and Locomotor Activity (Data Not Presented)
Home cage, handling, open field, sensory, and neuromuscular observations were unaffected by test article administration. There were no test article-related effects on locomotor activity patterns (total and ambulatory activity counts).
Ophthalmic Examinations (Data Not Presented)
No ophthalmic lesions indicative of toxicity were observed in any of the test article-treated groups. All findings observed were typical in prevalence and appearance for laboratory rats of this age and strain.
Clinical Pathology: Hematology, Clinical Chemistry, and Urinalysis
S-111-S-WB-related hematology findings consisted of statistically significantly higher mean prothrombin time and lower mean reticulocyte counts in the 0.6 mg/kg/d group males during study week 13 compared with the control group (Table 2). Similar differences were not observed in the females. Following the recovery period, at study week 21, mean prothrombin time and mean reticulocyte counts in the 0.6 mg/kg/d group were similar to the control group. At this interval, statistically significantly higher lymphocyte counts and lower mean red blood cell count, hemoglobin, and hematocrit levels were noted in the 0.6 mg/kg/d group males (Table 2). The values at study week 21 were similar to the control group values at study week 13, suggesting that the differences at study week 21 could be a reflection of the delayed growth of the 0.6 mg/kg/d group males. These differences at study week 21 were therefore a secondary effect and not a direct test article-related effect.
Summary of Hematology Values in Male and Female Sprague Dawley Rats After 90 Days of Oral Dosing of S-111-S-WB and Following 60-Day Recovery Period a
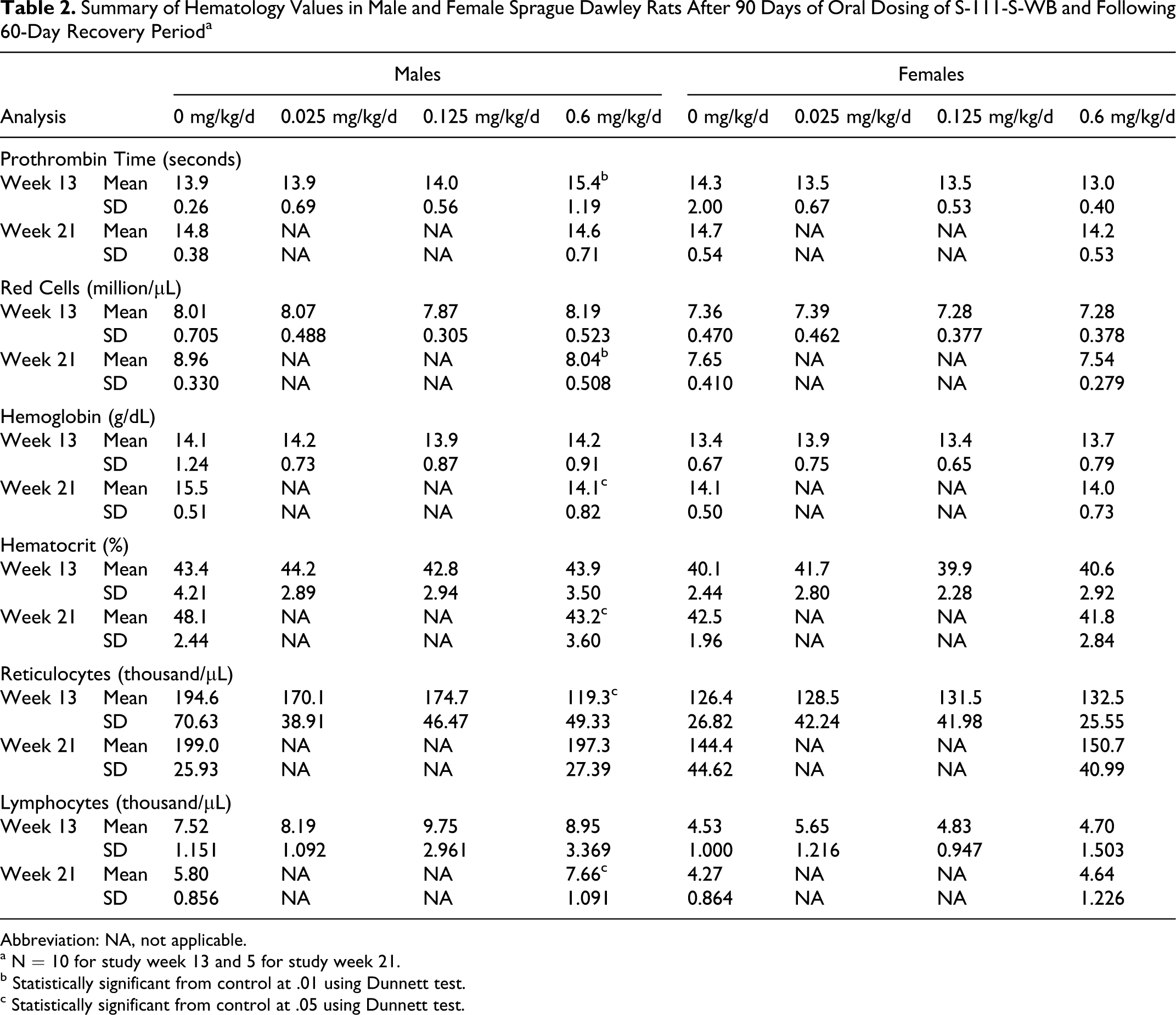
Abbreviation: NA, not applicable.
a N = 10 for study week 13 and 5 for study week 21.
b Statistically significant from control at .01 using Dunnett test.
c Statistically significant from control at .05 using Dunnett test.
S-111-S-WB-related effects on serum chemistry parameters consisted of lower protein concentrations and higher total bilirubin, urea nitrogen, and chloride concentrations in the 0.6 mg/kg/d males (Table 3; data not shown for chloride concentrations) and of lower globulin values, correspondingly higher A/G ratios and higher alkaline phosphatase concentrations in the 0.125 mg/kg/d males and 0.6 mg/kg/d males and females; the differences were often statistically significant and were more pronounced in the males.
Summary of Serum Chemistry Values in Male and Female Sprague Dawley Rats After 90 Days of Oral Dosing of S-111-S-WB and Following 60-Day Recovery Period a
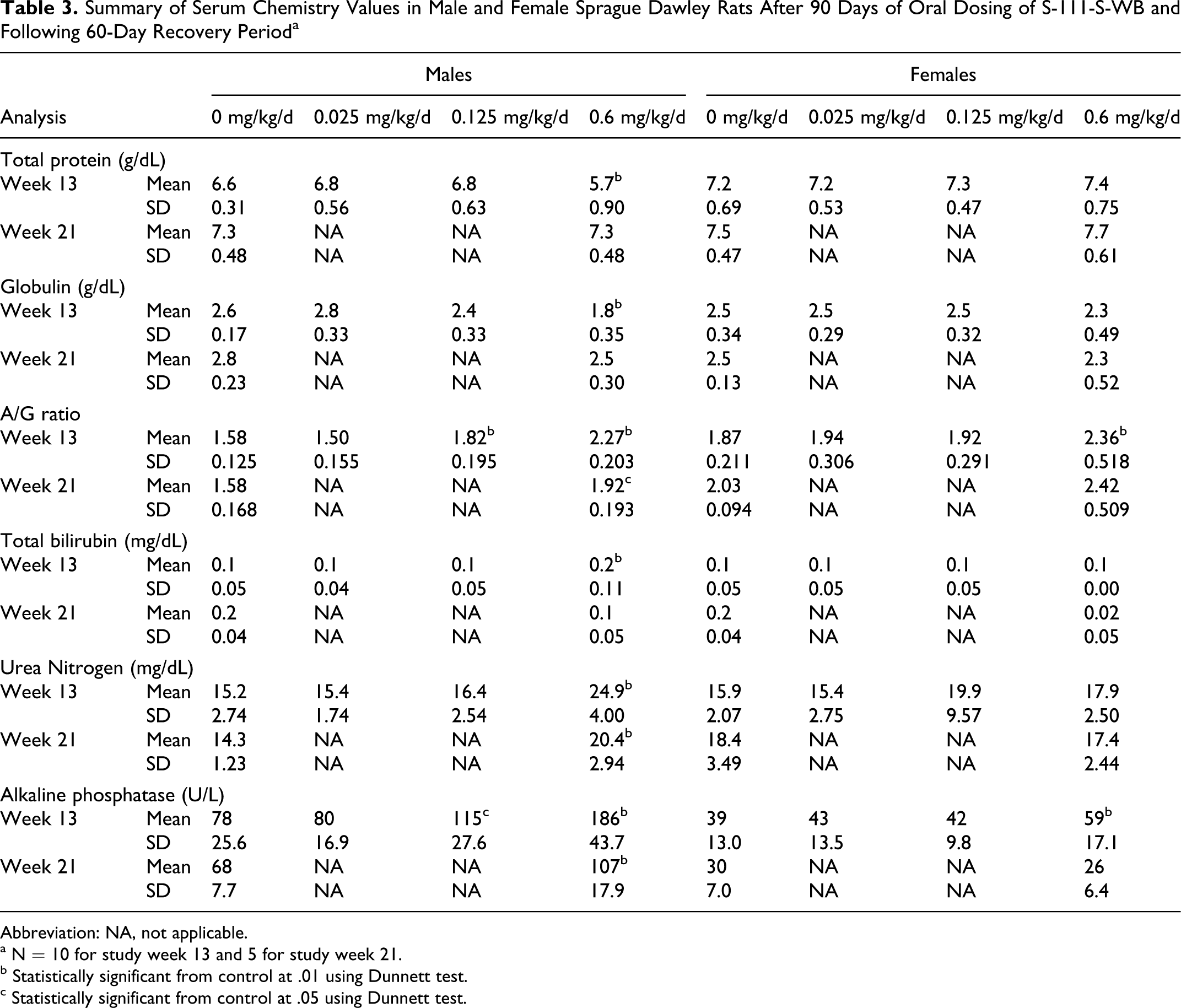
Abbreviation: NA, not applicable.
a N = 10 for study week 13 and 5 for study week 21.
b Statistically significant from control at .01 using Dunnett test.
c Statistically significant from control at .05 using Dunnett test.
Partial to complete recovery of these serum chemistry effects was shown at the end of the recovery period. Total protein values that were lower in the 0.6 mg/kg/d group males at study week 13 had returned to normal values at study week 21. Globulin values were lower in the 0.6 mg/kg/d group males and only slightly lower in the 0.125 mg/kg/d group males and 0.6 mg/kg/d females at study week 13, but following the recovery period, only slightly lower globulin values were present in the 0.6 mg/kg/d group males and females. Consequently, higher A/G ratios in the 0.125 mg/kg/d group males and the 0.6 mg/kg/d group males and females were observed at study week 13. The differences from the control group values decreased over time from approximately 44% to 22% and 26% to 19% in the males and in the females, respectively, from study weeks 13 to 21. Urea nitrogen values were higher in the 0.6 mg/kg/d group males at study week 13 and decreased by study week 21 to a value slightly higher than the control group mean. In the absence of a higher creatinine concentration and any histopathological evidence of nephrotoxicity, the urea nitrogen differences were most likely a reflection of higher protein catabolism which would be consistent with the lower total protein values and lower body weights present in these animals. Mean total bilirubin concentration was higher (predominantly due to 1 animal) in the 0.6 mg/kg/d group males at study week 13. Alkaline phosphatase in the 0.125 mg/kg/d group males and 0.6 mg/kg/d group males and females were higher than control group values at study week 13. Following the recovery period, only the 0.6 mg/kg/d group males still had a slightly higher mean alkaline phosphatase concentration. Higher alkaline phosphatase levels were consistent with microscopic liver observations and higher hepatic β-oxidation measured in these animals. The slight statistically significant higher mean chloride concentration in the 0.6 mg/kg/d group males at study week 13 (107 mEq/L compared to the control value of 104 mEq/L) was not considered adverse because values in the high-dosage group were comparable to the control values following the recovery period. There were no other test article-related differences in serum chemistry parameters. Urinalysis parameters were unaffected by test article administration.
Macroscopic Examination and Organ Weights
No S-111-S-WB-related macroscopic findings were noted at the scheduled necropsies. S-111-S-WB-related higher liver weights were present at study week 13 in the 0.125 mg/kg/d males and 0.6 mg/kg/d males and females (Table 4). In the males, the liver weight effects were reflected not only in the relative to final body weight values but also in the absolute and relative to brain weight values. In the females, the liver effect was less pronounced and was reflected only in the relative to final body weight values. These liver weight effects persisted in the 0.6 mg/kg/d males to study week 21. No other S-111-S-WB-related changes were observed.
Liver Weights in SD Rats After 90 Days of Oral Dosing of S-111-S-WB and Following 60-Day Recovery Period
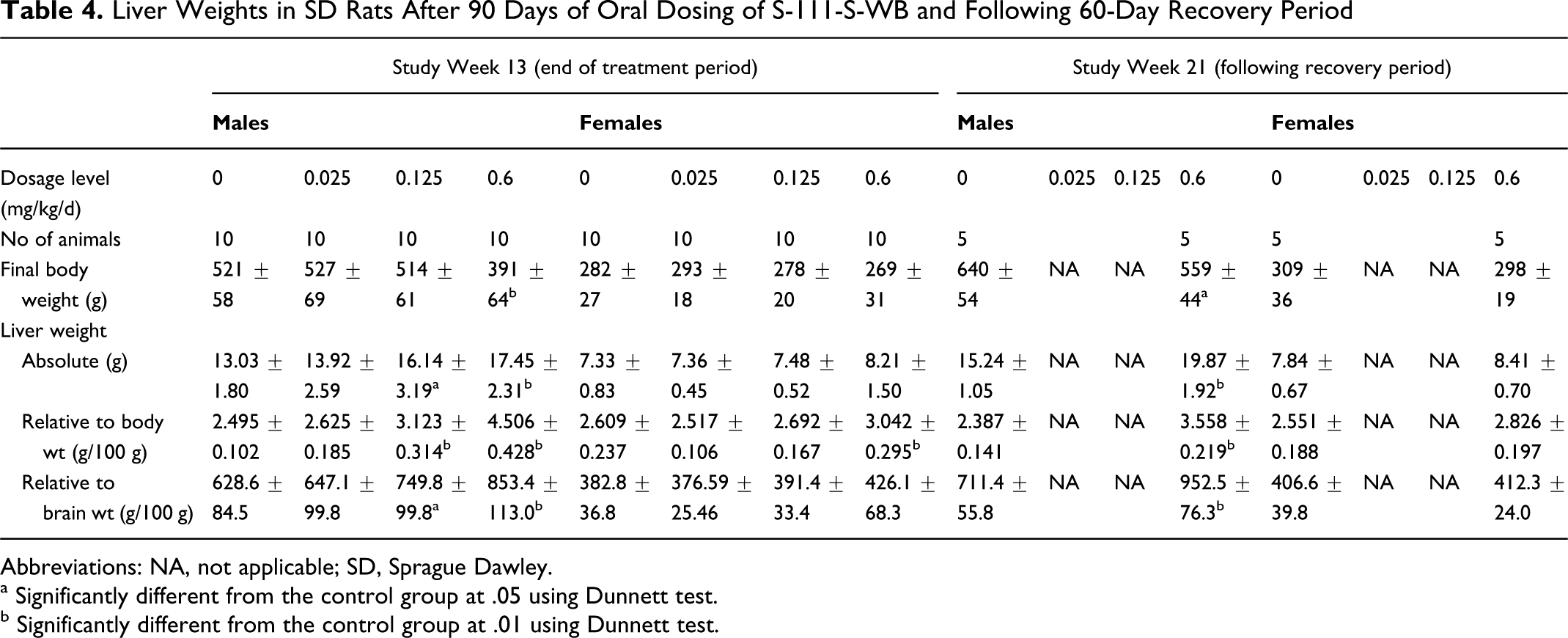
Abbreviations: NA, not applicable; SD, Sprague Dawley.
a Significantly different from the control group at .05 using Dunnett test.
b Significantly different from the control group at .01 using Dunnett test.
Microscopic Examinations
S-111-S-WB-related microscopic findings consisted of hepatocellular hypertrophy and eosinophilic foci in the 0.125 mg/kg/d and 0.6 mg/kg/d males and of hepatocellular degeneration and necrosis; ulceration, epithelial hyperplasia, and acute inflammation in the duodenum; and ulceration, erosion, squamous hyperplasia, and chronic active inflammation in the stomach in the 0.6 mg/kg/d males. Many of these lesions were of lessened severity or not observed following the recovery period (Tables 5 and 6).
Incidence of S-111-S-WB-Related Microscopic Observations in Male SD Rats After 90 Days of Oral Dosing a
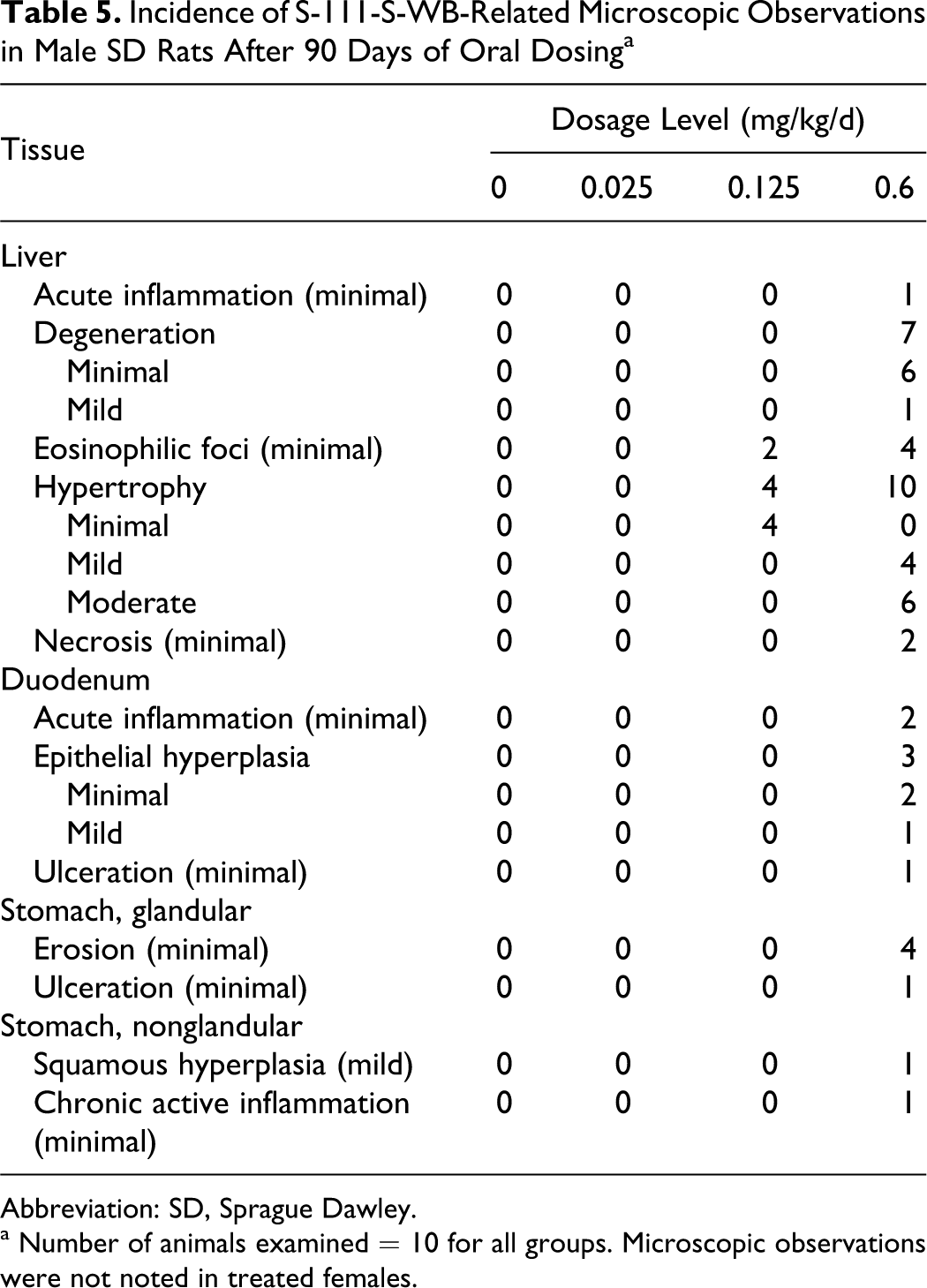
Abbreviation: SD, Sprague Dawley.
a Number of animals examined = 10 for all groups. Microscopic observations were not noted in treated females.
Incidence of S-111-S-WB-Related Microscopic Observations in Male SD Rats Following a 60-Day Recovery Period a
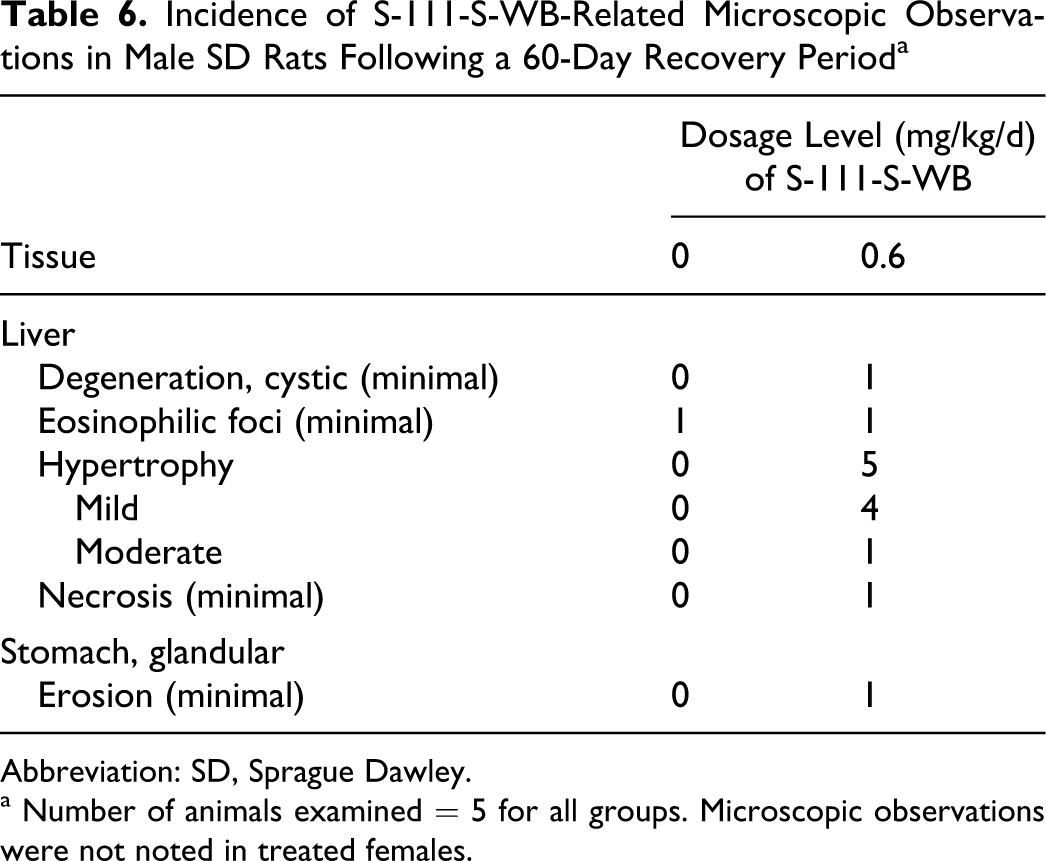
Abbreviation: SD, Sprague Dawley.
a Number of animals examined = 5 for all groups. Microscopic observations were not noted in treated females.
At study week 13, liver hypertrophy was observed in 4 of 10 and 10 of 10 males in the 0.125 and 0.6 mg/kg/d groups, respectively. This histologic finding corresponded to the higher liver weights. Hepatocellular hypertrophy was characterized by cells with abundant granular eosinophilic cytoplasm consistent with proliferation of peroxisomes. In the 0.125 mg/kg/d group males, the distribution of the hypertrophy was centrilobular (minimal severity). In the 0.6 mg/kg/d group males, this finding was observed diffusely within the lobule (mild severity) and in the most extensive cases, there was accentuation of the cytoplasmic change in the centrilobular areas (moderate severity). Eosinophilic foci of cellular alteration, consistent with “ground glass foci,” 20 were observed in the livers of 2 of 10 and 4 of 10 males in the 0.125 and 0.6 mg/kg/d groups, respectively.
Hepatocellular degeneration was characterized by individual cells with increased cytoplasmic eosinophilia and condensation of nuclei and was noted in 7 of 10 of the 0.6 mg/kg/d group males at study week 13. Two males in the 0.6 mg/kg/d group at study week 13 had minimal hepatocellular necrosis, with or without acute inflammation. Similar changes were not observed in the test article-treated females.
A period of 60 days without treatment allowed for significant recovery of these hepatic changes. Eosinophilic foci of alterations were present only at background level (a single incidence in each of the control and 0.6 mg/kg/d groups) and hepatocellular degeneration was absent. Mild to moderate hepatocellular hypertrophy persisted in all (5 of 5) 0.6 mg/kg/d group males, and minimal hepatocellular necrosis was present in 1 of 5 males in this group. No other S-111-S-WB-related microscopic findings were observed in the liver.
S-111-S-WB-related microscopic findings in the gastrointestinal tract included ulceration, erosion, inflammation, and hyperplasia (regenerative) and were observed only in the 0.6 mg/kg/d group males. Acute inflammation, epithelial hyperplasia (regenerative), or ulceration (minimal to mild severity) in the duodenum was observed in 5 of 10 males at study week 13. Minimal erosion or ulceration primarily in the pyloric region of the glandular stomach was observed in 5 of 10 males, with one of these rats also having squamous hyperplasia and chronic active inflammation of the nonglandular stomach. Similar microscopic findings were not observed in the S-111-S-WB-treated females. Significant recovery occurred by study week 21, as only 1 of 5 males in the 0.6 mg/kg/d group had minimal erosion in the pyloric region of the stomach following the recovery period. No other S-111-S-WB-related microscopic findings were observed.
Peroxisomal Proliferation Analysis
Higher hepatic β-oxidation was measured in 0.125 mg/kg/d group males and in 0.6 mg/kg/d males and females, indicating that S-111-S-WB-related is a mild peroxisome proliferator (Table 7). At the 10-day interval, hepatic β-oxidation was 1.7-fold higher in the 0.6 mg/kg/d males than in control rats. At the primary toxicology necropsy, hepatic β-oxidation was 2-fold and 3.1-fold higher in the 0.125 and 0.6 mg/kg/d group males, respectively, and 1.5-fold higher in the 0.6 mg/kg/d group females than in control rats. Analysis at the end of the recovery period showed partial recovery in the males and complete recovery in the females.
Summary of Peroxisomal β-Oxidation Activity in Male Sprague Dawley Rats
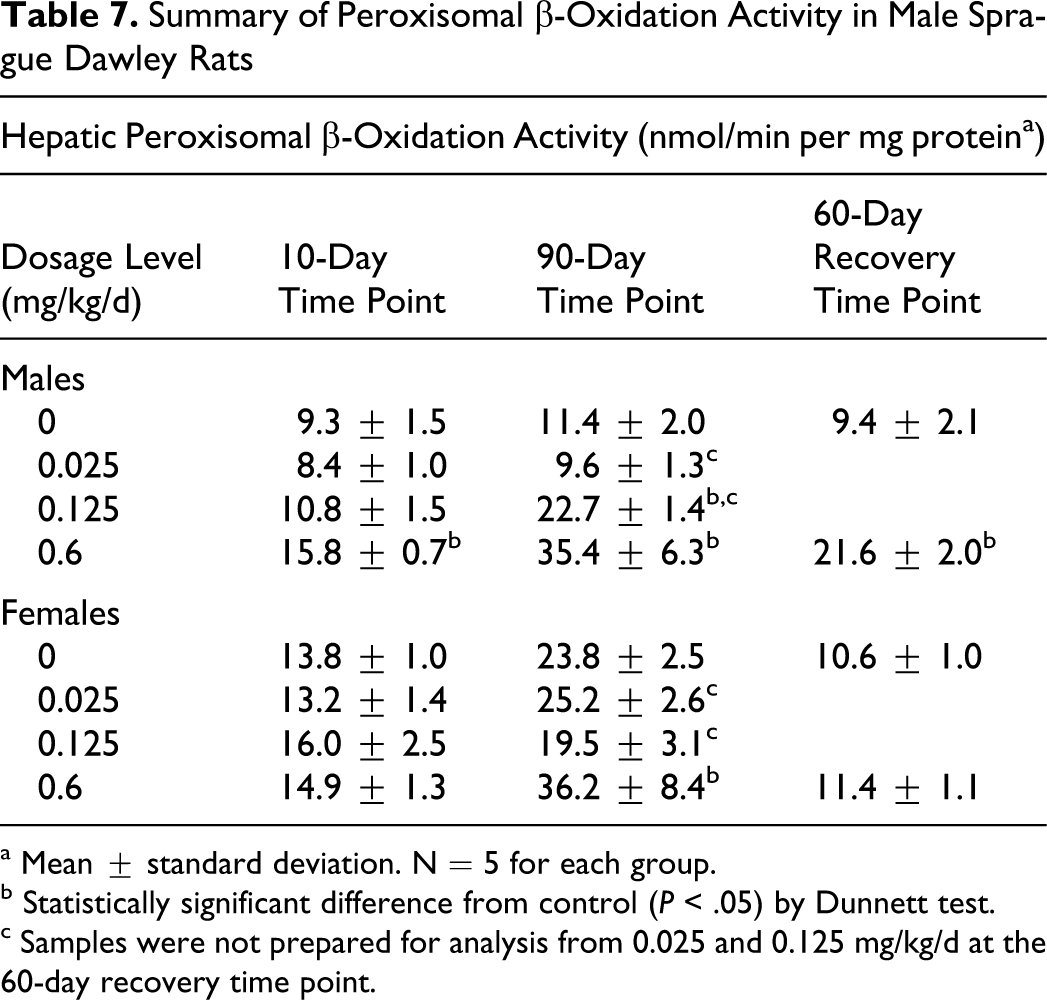
a Mean ± standard deviation. N = 5 for each group.
b Statistically significant difference from control (P < .05) by Dunnett test.
c Samples were not prepared for analysis from 0.025 and 0.125 mg/kg/d at the 60-day recovery time point.
Toxicokinetic Determinations
After 2 weeks of oral administration of S-111-S-WB at 0.6 mg/kg/d, the mean serum concentrations of C8, C9, C11, and C13 were relatively constant for at least 8 hours after dosing. Therefore, there was no unequivocal tmax to serve as the sampling time for continued evaluation of serum concentrations of the test article components; 2 hours after dosing was selected as the sampling time for subsequent blood collections. After 2 weeks of dosing (Figure 2), serum concentrations of C8 and C9 were considerably higher in the 0.6 mg/kg/d group males than the females (approximately 30× and 5×, respectively). The serum concentrations of C11 and C13 were similar between genders.
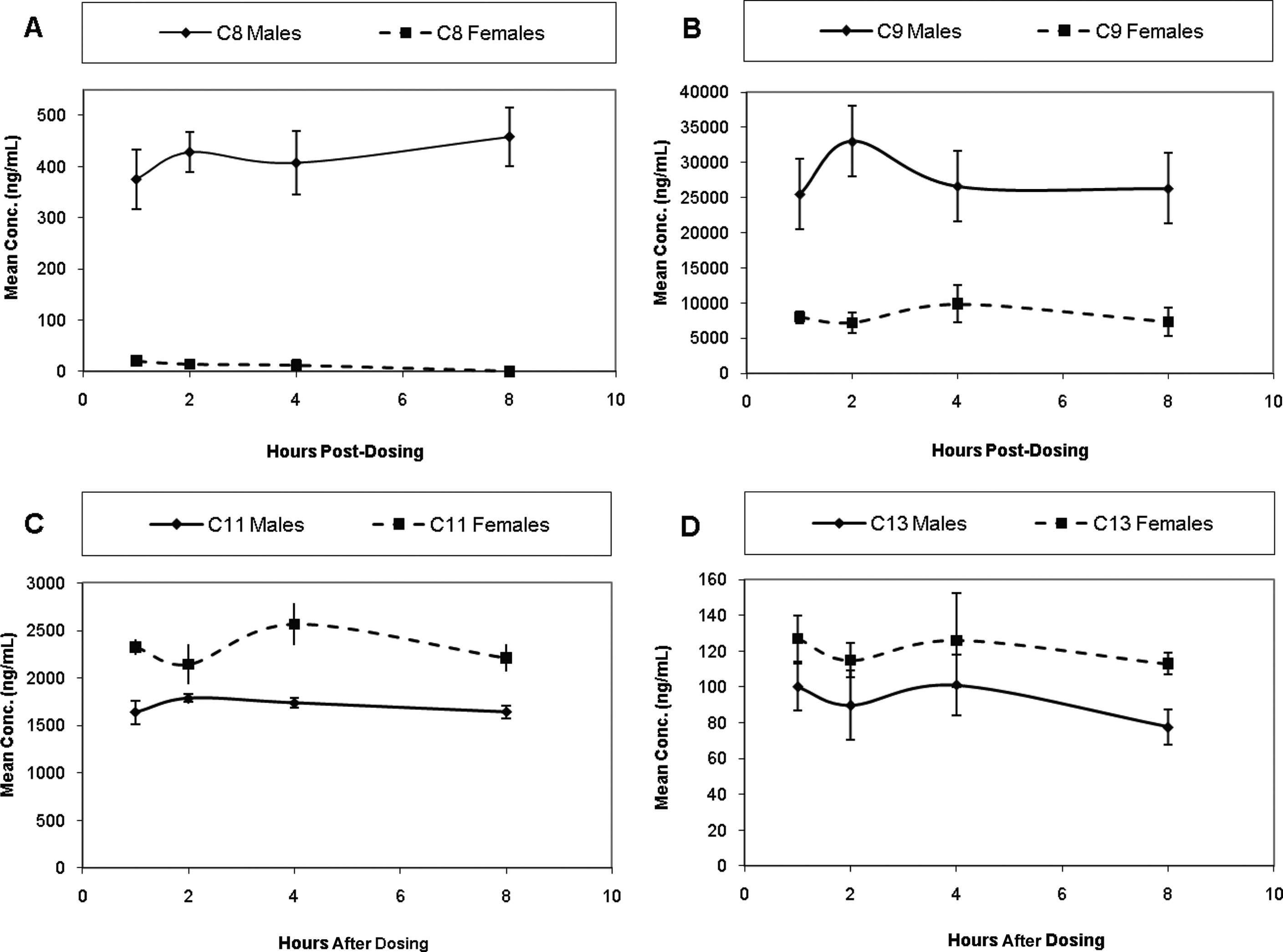
Mean (± SD) concentrations of C8, C9, C11, and C13 in serum of male and female Sprague-Dawley rats following 2 weeks of oral administration of S-111-1-WB at 0.6 mg/kg/d.
Following 90 consecutive days of dosing, mean serum concentrations of C8 were much higher for males in the 0.125 and 0.6 mg/kg/d groups than for the female animals in the 0.6 mg/kg/d group (Figure 3A). For females, the concentrations of C8 were below the lower limit of quantitation (LLOQ) except at a dosage of 0.6 mg/kg/d. A steady-state concentration of C8 was not attained during the 12 weeks of repeated daily dosing at any of the dosage levels tested. Within the 0.6 mg/kg/d groups, the serum concentrations of C8 and C9 were approximately 10-fold higher for males than for females (Figure 3B). Of all the treated groups, only the 0.025 mg/kg/d female group reached a steady-state concentration of C9 (approximately 400 ng/mL) in the serum by week 10.
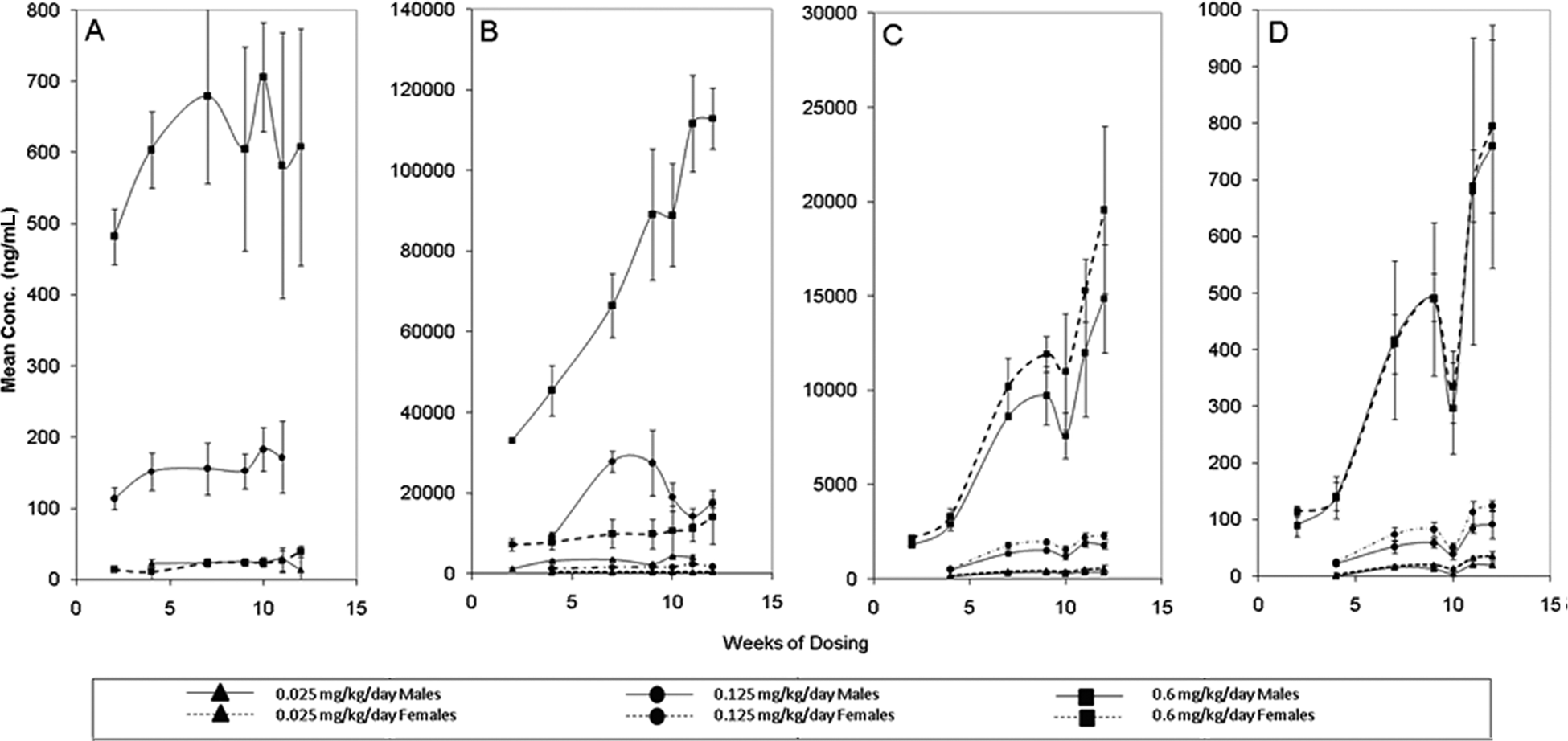
Mean (± SD) concentrations of C8 (A), C9 (B), C11 (C), and C13 (D) in serum of male and female Sprague-Dawley rats during 90 days of oral administration of S-111-S-WB at 0.025, 0.125, or 0.6 mg/kg/d. Note: C8 for females was below the low limit of quantitation except at a dosage of 0.6 mg/kg/d. N = 5 except for males on study week 4 at 0.025 mg/kg/d (N = 4) and for males and females on study week 2 at 0.6 mg/kg/d (N = 3).
Mean serum concentrations of C11 and C13 were similar between the genders (Figure 3C and D) and steady-state concentrations were not considered to have been reached during the 12 weeks of dosing at any of the dosage levels. Mean serum concentrations of total S-111-S-WB (sum of C8, C9, C11, and C13) for male and female rats were approximately 4- to 6-fold higher for males than for females (Figure 4).
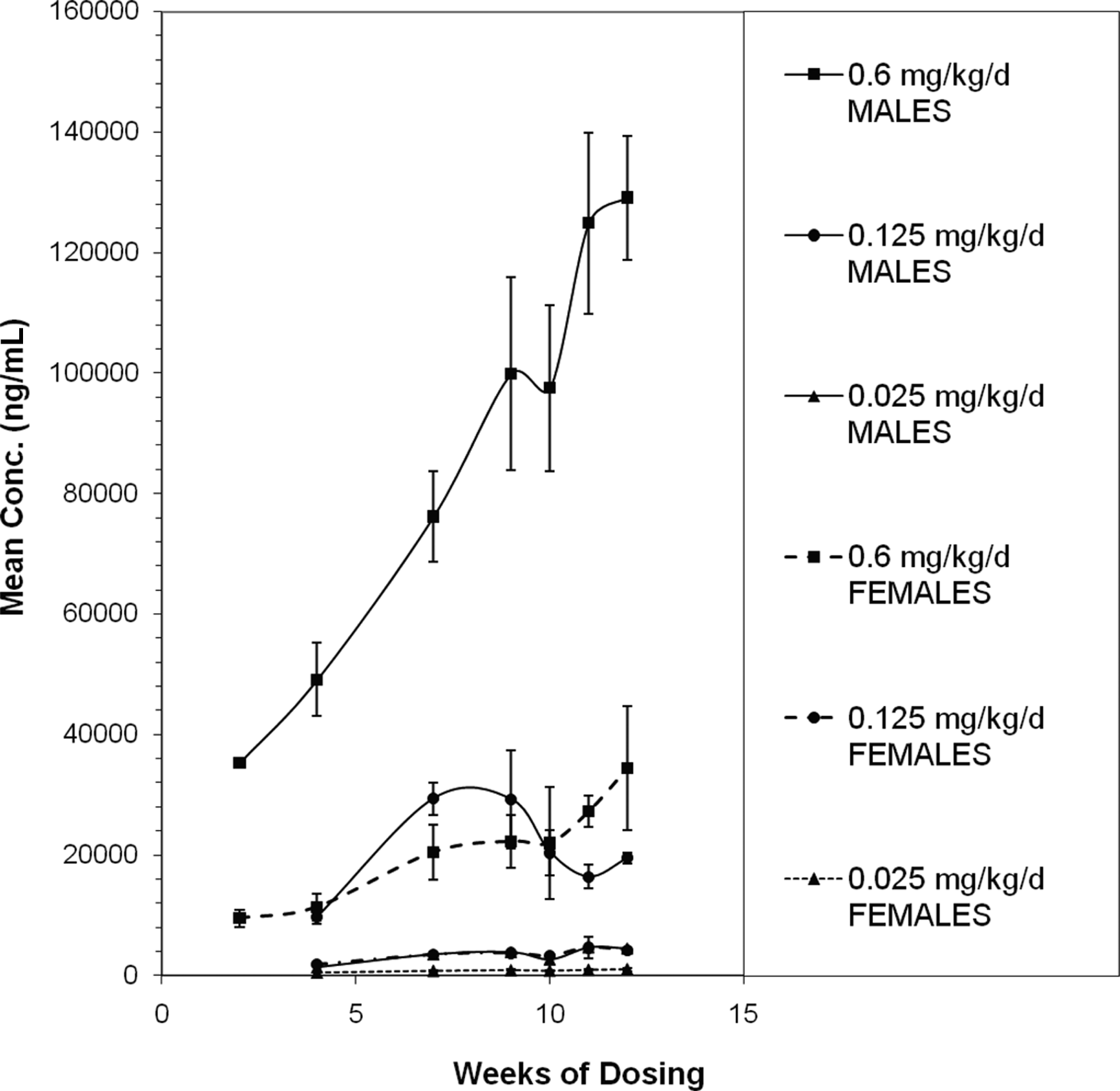
Mean (± SD) concentration of total S-111-S-WB in serum of male and female Sprague Dawley rats during 90 days of oral administration of S-111-S-WB at 0.025, 0.125 or 0.6 mg/kg/d.
Urinary and fecal elimination half-lives were conditionally defined as the time required for elimination of half the amount excreted over the 7-day post dosing period. For all analytes, the percentage of the daily dose eliminated after repeated dosing was higher than after a single dose, suggesting accumulation of each analyte had occurred. Elimination of C8 could only be evaluated following repeated dosing in the 0.125 mg/kg/d male rats because of the low levels excreted in the remaining male and female groups. The low level of C8 elimination (most below the LLOQ) following a single dose for male and female rats and following repeated dosing for female rats suggests accumulation of C8. Following repeated dosing in male rats, urinary elimination was the predominant route of elimination for C8, and correlated to higher serum values measured in the male animals. The amount eliminated in the male rats during the 168 hours following the final dose (>250% of the daily dose of C8) suggests that accumulation of C9 had occurred during 90 days of dosing (Table 8). For males, urinary and fecal elimination half-lives of C8 were 53 and 28 hours, respectively.
Total Elimination as Percentage of Mean Daily Dose 168 hours Following 90 Days of Repeated Oral Dosing of S-111-S-WB at 0.125 mg/kg/d in SD Rats
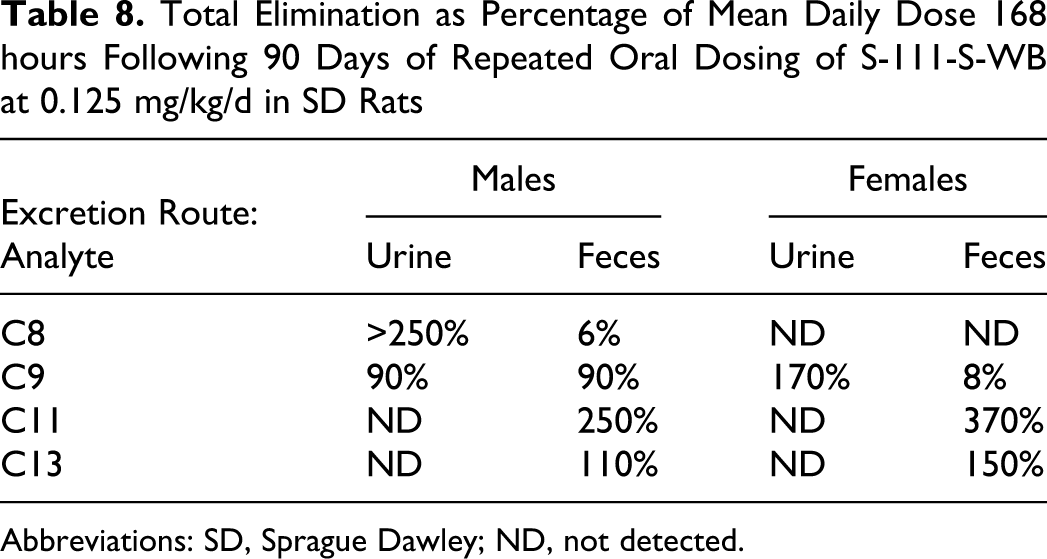
Abbreviations: SD, Sprague Dawley; ND, not detected.
The extent of urinary and fecal elimination of C9 were similar in male rats with only a few percentages of dose being eliminated by either route during the 168 hours following a single dose at any of the dosage levels. For female rats, urinary elimination was the predominant route of elimination (50% of the dose), with only a few percentages of the dose being eliminated in the feces. After repeated dosing, a similar trend was observed for males, approximately 180% of the daily dose was eliminated via both routes combined during the 168 hours following the final dose, suggesting that some accumulation of C9 had occurred during 90 days of dosing (Table 8). Elimination following repeated dosing is expressed in terms of the daily average dose and can exceed 100% because of the continued elimination of previous doses. For female rats, >170% of the daily dose was eliminated in the urine and about 8% in the feces during the 168 hours following the final dose in the 13-week study, again suggesting that some accumulation of C9 had occurred. Urinary and fecal half-lives of C9 were relatively similar within each sex but were generally longer for males (typically 50 hours) than for females (typically 35 hours; Table 8).
Elimination of C11 and C13 could only be evaluated in feces because these analytes could not be accurately determined in the urine because of low recovery in the analytical methodology. Fecal elimination of C11 and of C13 was generally similar for male and female rats (half-life approximately 40-50 hours), with typically about 15% and 5% to 10%, respectively, of the dose being eliminated during the 168 hours following a single dose. Approximately 250% to 370% of the daily dose of C11 and approximately 110% to 150% of the daily dose of C13 was eliminated during the 168 hours following the final dose in the 13-week study (Table 8). These results suggested that both C11 and C13 were absorbed and accumulation had occurred during 90 days of dosing.
Elimination of total S-111-S-WB (sum of C8, C9, C11, and C13) following oral administration could only be evaluated in feces because of low recoveries of C11 and of C13 in urine samples. Analysis of fecal elimination of total S-111-S-WB resulted in approximately 15% of the dose being eliminated during the 168 hours following a single oral dose. Approximately 70% to 115% of the daily dose was eliminated during the 168 hours following the 90th dose, again suggesting accumulation of S-111-S-WB had occurred during the 90 days of dosing (data not shown).
Discussion
The primary target organ for S-111-S-WB-mediated toxicity was the liver and males were more sensitive than females. Higher hepatic β-oxidation and liver weights indicative of mild peroxisome proliferation were present in the 0.125 mg/kg/d males and 0.6 mg/kg/d males and females. Lesions consisted of hepatocellular hypertrophy and eosinophilic foci in the 0.125 and 0.6 mg/kg/d males and hepatocellular degeneration and a limited incidence of necrosis in the 0.6 mg/kg/d males. Prothrombin time values were higher in the 0.6 mg/kg/d males. Serum chemistry parameters affected consisted of lower protein concentrations and higher total bilirubin, blood urea nitrogen, and chloride concentrations in the 0.6 mg/kg/d males and of lower globulin values, higher A/G ratios, and higher alkaline phosphatase concentrations in the 0.125 mg/kg/d males and 0.6 mg/kg/d males and females. The higher alkaline phosphatase concentrations were consistent with the microscopic liver findings and the higher hepatic β-oxidation in these animals. There were no elevated alanine aminotransferase and aspartate aminotransferase concentrations. Furthermore, there was no suggestion of a hypolipidemic effect as has been reported for PFOA where lower serum cholesterol and triglyceride levels were observed following treatment with PFOA. 21 In the current study, following administration with S-111-S-WB, there were no lower serum cholesterol levels, however, triglycerides were not included in the serum chemistry panel. In contrast, in a previous combined repeated-dose toxicity study with a reproduction/developmental toxicity test at dosage levels of 0.1, 0.5, and 2 mg/kg/d S-111-S-WB, lower serum cholesterol, phospholipids, and calcium levels were observed in the males in all S-111-S-WB groups following 14 days of treatment. 8 The reason for this difference is not clear. It is possible that the longer duration of dosing in this 90-day study could be a factor.
Males were more sensitive to the S-111-S-WB-mediated toxicity than females not only based on the liver-related effects described above but also based on the mean body weight differences. The 0.6 mg/kg/d males had a 24% lower mean body weight at the end of the dosing period compared with the control animals, whereas there were no body weight differences between the control and S-111-S-WB-treated females, The no-observed-effect level (NOEL) was 0.025 mg/kg/d for males and 0.125 mg/kg/d for females because of the serum chemistry differences and the higher hepatic β-oxidation and liver weights present in the 0.125 mg/kg/d males and 0.6 mg/kg/d females and the hepatocellular hypertrophy and eosinophilic foci present in the 0.125 mg/kg/d males. Thus, the low-observed-effect level (LOEL) in the current study was 0.125 mg/kg/d for males and 0.6 mg/kg/d for females. In contrast, PFOA was reported to have a NOEL of 0.05 mg/kg/d in a 90-day dietary rat study, indicating that S-111-S-WB is more potent than PFOA. 4,22 Similarly data from the companion 2-generation reproductive toxicity study with S-111-S-WB showed that S-111-S-WB was more toxic than PFOA in rats. 7
Perfluoroalkyl acid serum levels were higher in males than in females. The difference between males and females became smaller with increasing chain length. After 2 weeks of dosing, serum concentrations at the 2 hours postdose time point showed male/female ratios for C8 and C9 of approximately 30× and 5×, respectively, whereas the serum concentrations of C11 and C13 were essentially similar between genders. A similar trend was also present after 90 days of dosing. This comparison is complicated by the fact that C8, C9, C11, and C13 are not present in equal amounts in S-111-S-WB and neither concentration nor competing effects on the transporters involved with absorption and excretion were addressed in this study. Nevertheless, this suggested trend of decreasing gender difference with increasing chain length might be related to the potential decreasing relative importance of the excretion via the urine compared to the excretion via the feces.
Other studies have shown that urine was the primary route of elimination for C8 in both sexes. In males, urinary excretion accounted for approximately 43% to 62%, and fecal excretion 6% to 14% of the administered dose. In females, urinary elimination was approximately 76% to 84% and fecal excretion 2%-6% of the administered dose. 23 Table 8 summarizes the total elimination of each constituent as a percentage of the mean daily dose. In this study, there was a gender difference in analyte serum concentration for C9, which was excreted in the males equally via urine and feces, whereas in the females excretion occurred predominantly via the urine. Thus, these results suggest that there may be a shift from urinary excretion toward fecal excretion with increasing carbon chain length (Table 8). These results are consistent with those of Goecke et al and Goecke-Flora and Reo who showed that C7 and C8 perfluorinated acids were excreted primarily via the urine, C9 acid via both urine and bile, and C10 and C11 derivatives mainly via the bile. 24,25
There are several reports that suggest the involvement of organic anion transporters in the urinary excretion of PFOA (for review see Kennedy et al 4 ). Kato et al reported that females exhibited a higher urinary excretion of taurocholate, dibromosulfophtalein, and zenarestat, all substrates and/or inhibitors of organic anion transporting polypeptide 1, than male Sprague Dawley rats. 26 Thus, a decreased affinity of longer carbon chain perfluoracids for these transporters could explain the above-described differences in excretion and the resulting serum levels.
In conclusion, based on the results of this study the NOEL was 0.025 mg/kg/d for the males and 0.125 mg/kg/d for the females.
Footnotes
Acknowledgments
This work was paid for by the Society of Plastics Industry, Inc. The authors thank the staff of WIL Research Laboratories, LLC, for their technical expertise in the conduct of this study; Mr John C. O’Connor and the staff of DuPont Haskell Laboratory for Health & Environmental Sciences for their expertise in the performance of the peroxisome proliferation analysis; and Dr Christopher Chengelis from WIL Research Laboratories, LLC, for helpful discussions in the preparation of this manuscript.
The author(s) declared a potential conflict of interest (eg, a financial relationship with the commercial organizations or products discussed in this article) as follows: Four of the authors are or were employed by companies that manufactured or used S-111-S-WB. The remaining 5 authors are or were employed by the CRO that was contracted to perform the research.
The author(s) received no financial support for the research and/or authorship of this article.
