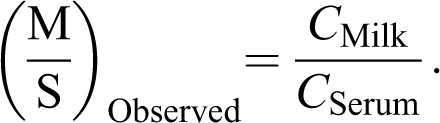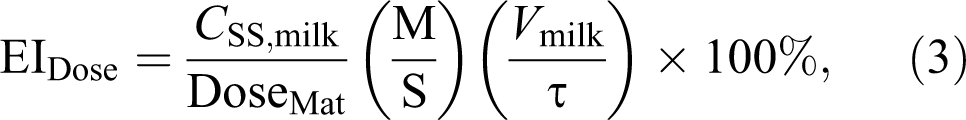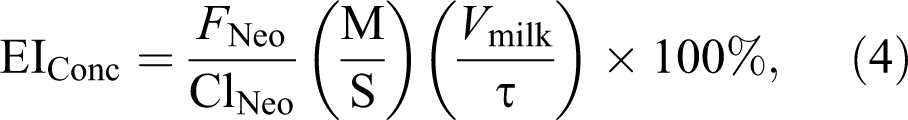Abstract
The purpose of our study was to assess the influence of lactation stage–dependent differences in milk-to-serum (M/S) ratio for an actively transported drug (cefepime) on the calculation of 2 exposure indices, EIDose and EIConc. Age-dependent differences in cefepime pharmacokinetics at postnatal days 4 and 10 as well as cefepime M/S values at lactation days 4 and 10 in rats were determined. Significantly higher elimination rate constant and systemic clearance and lower half-life were found in day 10 compared to day 4 pups with no differences in oral bioavailability. The EIConc was quantitatively higher than EIDose at both lactation stages. The ~7-fold decrease in cefepime M/S values at lactation day 10 resulted in ~7-fold reduction in the EIDose and ~13-fold reduction in EIConc. Our study confirms the need to evaluate M/S at different lactation stages for actively transported drugs to avoid over- or underestimation of neonatal exposure risk.
Introduction
Many chemicals have the potential to cross the lactating mammary epithelial barrier into milk.
1
–3
Any chemical (ie, drugs, environmental toxicants) transferred into breast milk may pose a health risk to a nursing neonate. To assess this risk, the literature posits several exposure indices that variably consider the key determinants affecting neonatal exposure to chemicals in breast milk.
2,4,5
Both maternal factors (ie, the bioavailable (F) maternal dose, maternal elimination capacity or systemic clearance (ClS), and the milk-to-serum [M/S] concentration ratio) and neonatal factors (ie, the volume of milk consumed during nursing [V
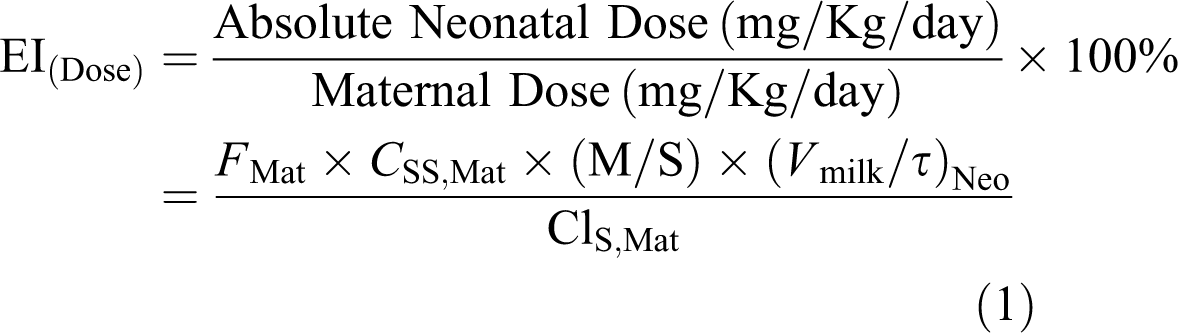
milk/τ], the bioavailable neonatal dose, and neonatal systemic clearance) determine neonatal exposure risks during breastfeeding. The most commonly referred to exposure index, EIDose, bases risk assessment on the dose a neonate receives from the mother during nursing. To provide some relative measure of risk, the EIDose expresses neonatal dose as a percentage of the maternal dose as shown in equation 1
4
:
A more comprehensive exposure index is the EIConc, 7 where risk is based on the systemic exposure of the neonate to the chemical. To provide a relative measure of risk, the EIConc expresses neonatal steady-state serum concentration as some percentage of the maternal steady-state serum concentration as shown in Equation 2 4 :
McNamara and Abassi 4 recently reviewed the theoretical use of these exposure indices and how the different determinants of exposure risk influence the values of EIDose and EIConc. This review highlighted the importance of including neonatal pharmacokinetics to more accurately reflect the risk as the EIConc bases the risk assessment on systemic concentrations and not simply on the ingested dose. 4 What was of further interest was the importance of the M/S on the value of these exposure indices (see Equations 1 and 2) where the higher the M/S value (ie, greater the compound distribution into milk), the higher the exposure. The M/S has been evaluated for a number of drugs. However, these are often determined without consideration of stage of lactation. Although the literature acknowledges the M/S variability between colostrum and mature milk, 4,9 –11 foremilk and hindmilk, 12 –14 and within a dosing interval, 14 limited discussion exists about the stage of lactation (ie, early, mid, and late) and its impact on the M/S value. 15
Many drugs distribute into milk via passive processes. In such cases, the physicochemical properties of the compound, fat partitioning, and plasma protein binding determines the milk levels of a compound 15 and often M/S values do not exceed unity. However, some drugs are substrates for transporters expressed in the lactating mammary epithelium. 16 –18 Actively transported drugs can achieve M/S values well exceeding 2. 19,20 Recently, our laboratory has shown clear evidence of changes in M/S values of an actively transported drug substrate with lactation stage due to lactation stage–dependent changes in the expression of transporters in the lactating mammary epithelium. 21 Reports of transporter expression in the lactating mammary gland also clearly indicate significant changes in transporter expression levels with lactation stage. 22 –24 Herein we provide experimental support for the need to consider lactation stage in the M/S determination of actively transported compounds and in the assessment of neonatal exposure risk with EIDose and EIConc using cefepime, a β-lactam antibiotic substrate of organic cation/carnitine (Octn) transporters, and the lactating rat dam-pup dyad.
Methods
Animals and Chemicals
Female Sprague-Dawley rats (250-300 g) ordered at gestation day 17 (for pups at postnatal day 4; n = 12) and lactation day 3 (for pups at postnatal day 10; n = 12) were obtained from Charles River Canada (St Constant, PQ, Canada) and were housed singly in a temperature- and humidity-controlled facility (22°C ± 2°C) on a 12-hour light:dark cycle (07:00-19:00 hours). All litters were reduced or increased (via cross-fostering) to 10 pups as necessary immediately following parturition or were requested from Charles River to have 10 pups per litter. All rats had free access to food (Prolab RMH 3000, Purina, Inc, Richmond, Indiana) and water throughout the study and were allowed a 7-day acclimatization period. All procedures were conducted in accordance with the Canadian Council of Animal Care guidelines for the care and use of laboratory animals and were approved by the Animal Research Ethics Board of the University of Saskatchewan. Cefepime was purchased as a sterile crystalline powder equivalent to 2 g of cefepime per vial from the Royal University Hospital at the University of Saskatchewan (Saskatoon, SK). High performance liquid chromatography (HPLC) solvents were HPLC grade and all other chemicals were highest analytical grade possible. A MilliQ Synthesis (Millipore, Bedford, Massachusetts) water purification system provided purified deionized water.
Cefepime Pharmacokinetics in Rat Pups
Rat pups at postnatal age days 4 and 10 were removed from their dams at least 2 hours prior to cefepime administration. These dams were used to assess steady-state milk levels of cefepime at lactation days 4 and day 10 as reported by Ling and Alcorn.
21
One day prior to the study, the right jugular vein was surgically cannulated (silastic tubing, 0.64 mm inner diameter [ID] × 1.19 mm outer diameter [OD], Dow Corning Corporation, Midland, Michigan) under isoflurane anesthesia (4% for induction of anesthesia, 2% for maintenance of a surgical plane of anesthesia). Cefepime (250 mg/h) was administered to rat dams at lactation days 4 or 10 by continuous intravenous (IV) infusion (4 hours) via the jugular catheter following an IV loading dose (50 mg). Cefepime was reconstituted with sterile saline just prior to administration. Blood (via saphenous venipuncture) and milk (via manual expression of the mammary glands) was collected under light isoflurane anesthesia just prior to the start of the infusion (0 hour) and just before the termination of the infusion (4 hours). All samples were stored frozen (−20°C) until analysis. The observed M/S of cefepime was determined from the ratio of the serum and milk concentrations (C) using the equation
Freshly prepared cefepime was diluted according to pup body weight. For IV injection, 20 mg/kg body weight in 50 μL was administrated via intracardiac puncture under isoflurane anesthesia using a 27-gauge needle. For oral administration, 40 mg/kg body weight in 100 μL was administrated via a 24-gauge 1 inch gavage needle. Blood samples were collected from pup heart via cardiac puncture (under isoflurane anesthesia) at 0, 10, 20, 30, 45, 60, 120, 240, 360, and 480 minutes after administration and the pups were immediately euthanized. Each rat pup in a litter was assigned to a single time point following drug administration (ie, destructive sampling scheme). Each litter was considered as n = 1 and a total of 6 litters were used for each route of administration (consequently we used 6 dams/route of administration/postnatal age group to provide n = 6 for each blood collection time point for each route of administration). Blood samples were allowed to clot for 30 minutes at room temperature and serum was collected following centrifugation at 1000g for 10 minutes. Serum was stored in Eppendorf microcentrifuge tubes at −20°C until analysis.
Cefepime Analysis
Milk and serum cefepime concentrations were measured by a validated high performance liquid chromatography with ultraviolet detection (HPLC-UV) method as described by Ling and Alcorn. 21 Briefly, in screw-top glass culture tubes, 20 μL of rat plasma or milk was added with 180 μL acetonitrile:methanol (9:1), vortex-mixed for 5 minutes, and centrifuged at 10 000g for 20 minutes. The supernatant was transferred to HPLC vials and 10 μL was injected onto a CN (cyano) column (HyperClone 5 μm, 250 × 4.6 mm, Phenomenex, Torrance, California) with methanol:double distilled water (1:1) as mobile phase delivered at 1 mL/min. UV detection wavelength was set at 260 nm. The standards were prepared in male rat serum as above. The standard curve was linear (r 2 > .99) in the range of 3.1 to 100 mg/L. Intra- and interassay accuracy and precision ranged from 5% to 14%. Quality control samples at 3 different concentrations performed in duplicate were assessed as acceptance criteria for individual HPLC analyses.
Pharmacokinetic Analysis
The pharmacokinetic parameters of cefepime were characterized using GraphPad Prism 4.0 (GraphPad Software, San Diego, California). Plots of average serum cefepime concentration following oral or IV administration to Sprague-Dawley rat pups at postnatal day 4 and postnatal day 10 are shown in Figure 1. The C max values were obtained directly from the serum concentration versus time data. The area under the serum concentration–time curve (AUC) for both oral and IV administration was calculated by the linear trapezoidal method with extrapolation to infinity determined using the ratio of the last serum concentration and elimination rate constant (ie, C last/k). The elimination rate constant (k) was determined by linear regression analysis of the postdistributive log-linear terminal serum concentration–time determinations. The elimination half-life (t 1/2) was determined by the ratio 0.693/k. The systemic clearance (ClS) was calculated as DoseIV/AUC. Cefepime absolute bioavailability (F) after oral administration was determined as (AUCOral × DoseIV)/(AUCIV × DoseOral). The apparent volume of distribution (V d) was determined as DoseIV/(k × AUC).
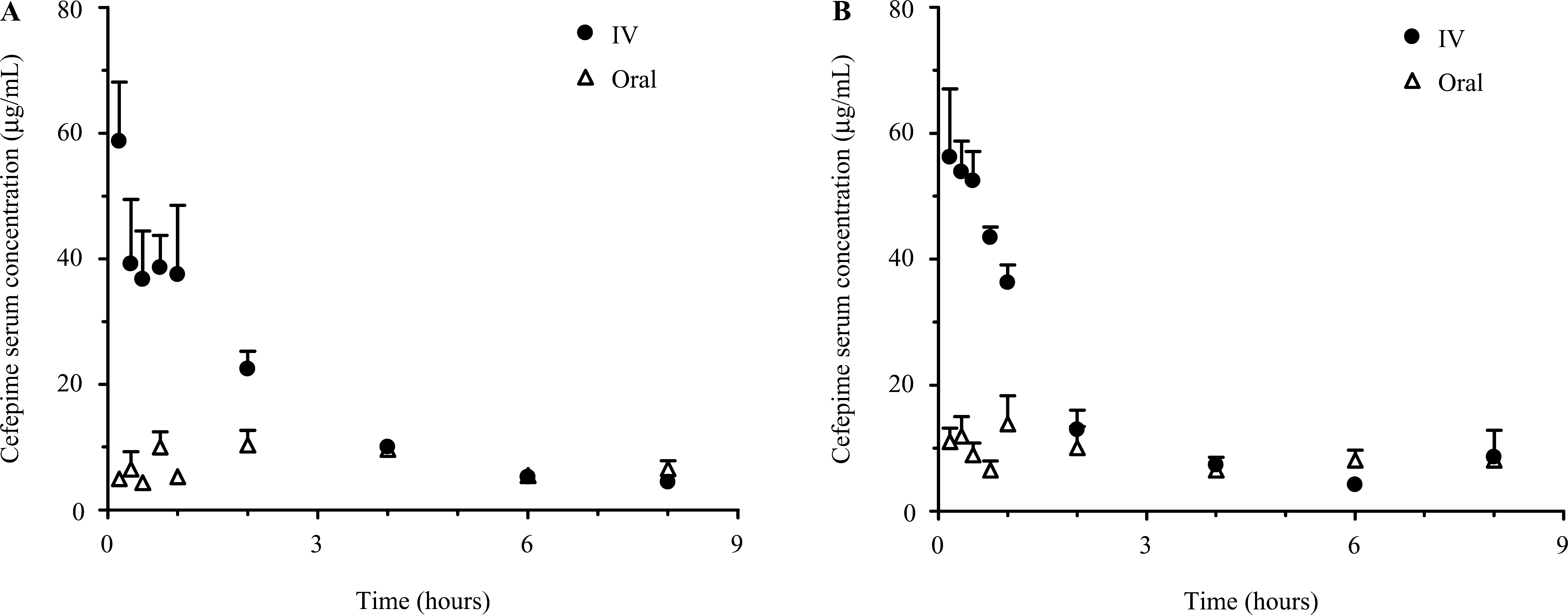
Mean ± SEM serum concentration versus time curves of cefepime after 20 mg/kg intravenous (IV) dose or 40 mg/kg oral dose in Sprague-Dawley rat pups (n = 6) at postnatal day 4 (A) and postnatal day 10 (B).
Calculation of Exposure Indices
The exposure index related to maternal dose (EIDose) was calculated as
Statistical Analysis
Data are presented as mean ± SEM. Means between different age groups within the same route of administration were compared using the unpaired t test. Means for F were compared using the unpaired t test. The significance level was set at .05.
Results
Table 1 presents the mean pharmacokinetic parameter estimates for cefepime following oral and IV administration to rat pups 4 and 10 days of age. Following IV administration, elimination rate constant, half-life, and systemic clearance were significantly higher in 10-day-old rat pups relative to postnatal day 4 pups. Although not statistically significant, oral bioavailability was also increased in postnatal day 10 pups. The volume of distribution (V d) of cefepime was similar between the 2 rat pup age groups.
Summary of Mean ± SEM Pharmacokinetic Parameters for Cefepime After Intravenous (20 mg/kg by Intracardiac Injection Under Isoflurane Anesthesia) and Oral (40 mg/kg by gastric gavage) Administration in Rat Pups (Postnatal Days 4 and 10; n = 6/Time Point Blood Collection) a
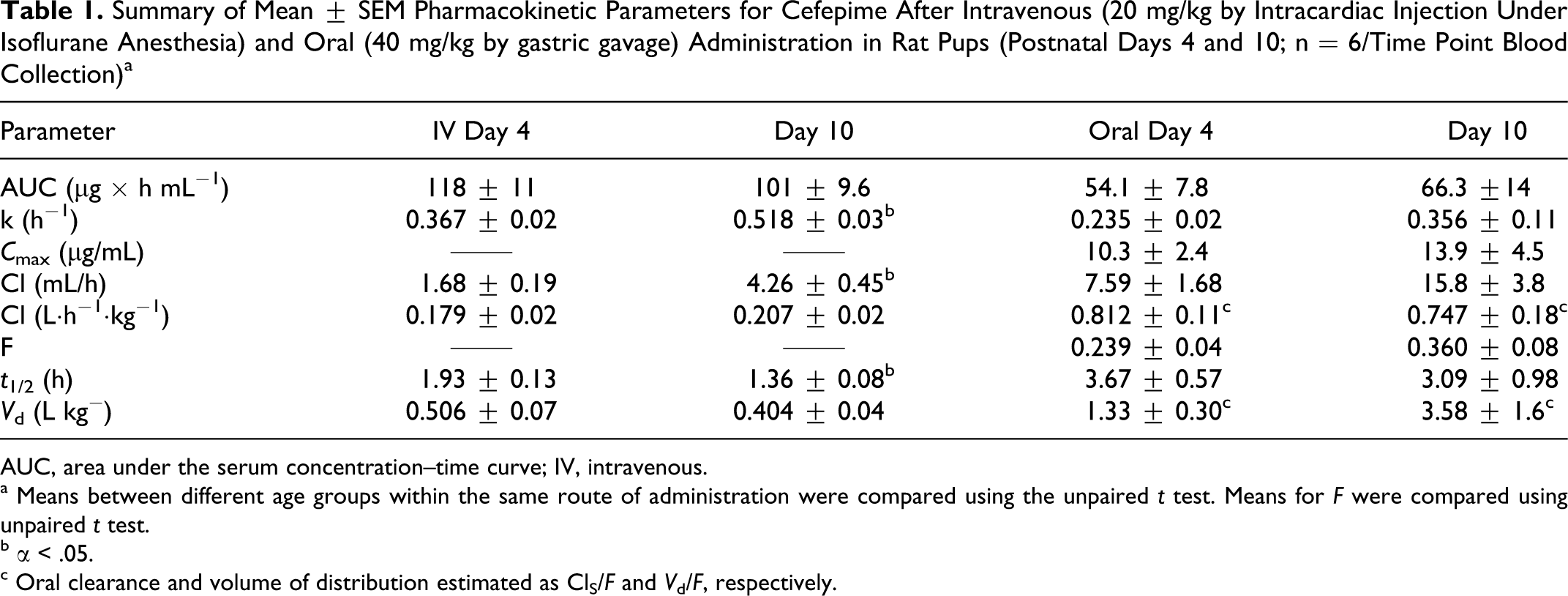
AUC, area under the serum concentration–time curve; IV, intravenous.
a Means between different age groups within the same route of administration were compared using the unpaired t test. Means for F were compared using unpaired t test.
b α < .05.
c Oral clearance and volume of distribution estimated as ClS/F and V d/F, respectively.
Table 2 presents additional parameters required for the calculation of exposure indices. Note that the cefepime M/S was higher at early lactation stage (day 4) relative to day 10 of lactation. 21 At postnatal day 4, the EIDose and EIConc were similar and represented about 3% of maternal exposures. The ~7-fold decrease in M/S values of cefepime at day 10 of lactation resulted in ~7-fold reduction in the calculated EIDose (considering the age-dependent changes in body weight and the volume of milk consumed per day) and ~13-fold reduction in the EIConc (considering the age-dependent increases in systemic clearance and the volume of milk consumed per day).
Additional Parameters Used for the Calculation of EIDose and EIConc Values for Postnatal Days 4 and 10 Sprague-Dawley Rat Pups
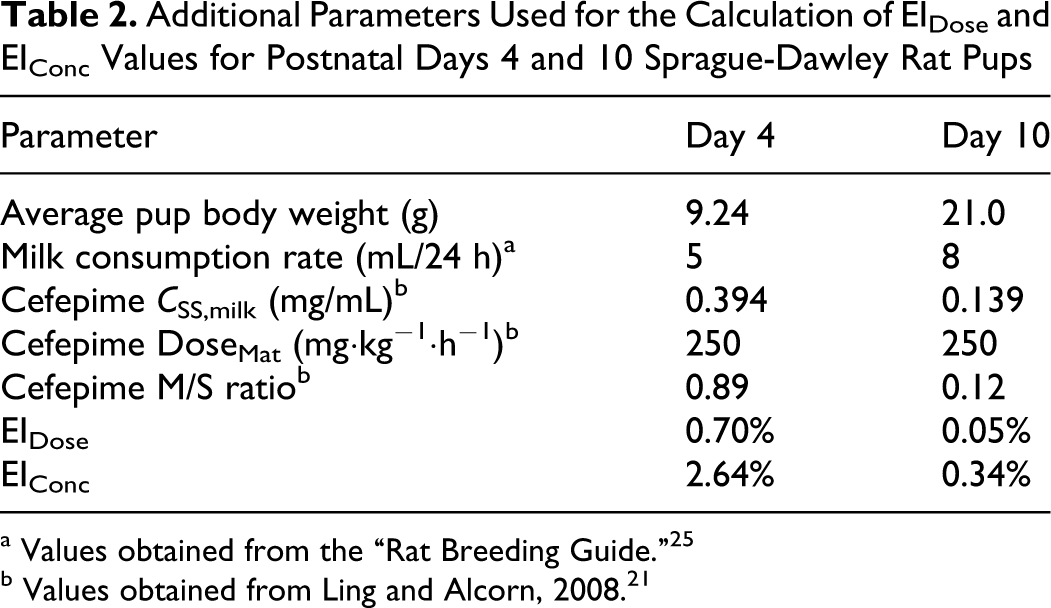
a Values obtained from the “Rat Breeding Guide.” 25
b Values obtained from Ling and Alcorn, 2008. 21
Discussion
Following maternal exposure to chemicals, exposure indices that relate risk to the nursing neonate as a percentage of maternal dose (EIDose) or of maternal steady-state serum concentration (EIConc) incorporate M/S values of the chemical as a significant determinant of the exposure risk. 4 For most compounds, the literature reports single M/S values and the reported value is assumed to reflect the distributional characteristics of the drug throughout most of the lactation period. 26 Our study clearly demonstrates through experimentation that the M/S value of an actively transported chemical changes considerably with stage of lactation, which, in turn, significantly affects the magnitude of the exposure risk calculated by the EIDose or EIConc.
The magnitude of the risk posed to the neonate following exposure to chemicals via the breast milk depends on the amount of drug in the milk and the pharmacokinetic and pharmacodynamic properties of the drug in the neonate. 27,28 Given the proportional relationship between M/S and the EIDose or EIConc (see equations 1 and 2), M/S estimates have a significant impact on the value of the EI. M/S estimates are available for some drugs and environmental toxicants, but most, if not all, are determined without regard to lactation stage. The literature does acknowledge differences in M/S between colostrum and mature milk 10,29 and hindmilk and foremilk. 13,30 However, the breast milk levels of many solutes (ie, critical nutrients, drugs) also depend on the activity of solute carrier (SLC) and ATP-binding cassette (ABC) transporters located on the apical and/or basolateral membranes of the polarized lactating mammary epithelium. 31,32 Although the role of most transporters in the mammary gland is largely unknown, recent reports on transporter expression profiles during advancing lactation clearly identify lactation stage–dependent changes in the messenger RNA (mRNA) and functional expression levels of various transporters. 17,21,33,34 Changes in expression can result in alterations in the distributional characteristics and extent of accumulation of drugs and environmental toxicants that are substrates for transporters expressed at the lactating mammary gland. The known ontogeny of transporters coupled with other elimination mechanisms in the developing neonate 35,36 demonstrates that the assessment of risk exposure in the nursing neonate can be significantly affected by postnatal age of the neonate and stage of lactation.
Previously, we had reported of the acute interaction between the β-lactam antibiotic, cefepime, and
In addition to the bioavailable dose to the neonate via the breast milk, neonatal elimination capacity is an additional critical determinant of exposure risk. 4 In the current study, bioavailability was unchanged between the 2 age groups but rat neonates showed an enhanced capacity to eliminate cefepime with age resulting in a considerable decrease in the EIConc at postnatal day 10 relative to postnatal day 4 (Table 2). These data suggest that for a given dose via the breast milk, the internal dosimetry metrics (ie, blood concentrations, area under the blood concentration vs time curve) change with age of the neonate. The potential for drug accumulation and, hence, susceptibility for adverse outcomes may also change with neonatal age. These data confirm the current discourse on the importance of the developmental maturation of pharmacokinetic processes in the assessment of exposure risks during postnatal development.
Our study provides evidence of the importance of lactation stage in the determination of M/S ratios and brings into question the accuracy of M/S ratios of some chemicals reported in the literature. This becomes a critical consideration when exposure indices are used in the risk/benefit analysis concerning the safety of medication use during breastfeeding or the safety of maternal environmental exposures to a nursing neonate. The literature on drug use during breastfeeding 37 suggests health care professionals should pay particular attention when prescribing drugs to breastfeeding mothers. Most literature on drug use during breastfeeding focuses on drug transfer into breast milk and the amount of drug the neonate becomes exposed to during breastfeeding. 4,38 Our study suggests the need to consider lactation stage in the risk/benefit analysis for drugs that are known transporter substrates. What may be safe to use at one stage of lactation may not be considered safe to use at an earlier or later lactation period due to differences in M/S ratios of a drug. An additional consideration in the risk/benefit analysis is the recent publications that demonstrate the importance of SLC and ABC transporter expression at the lactating mammary in nutrient and drug transport into breast milk. 21,33,39 We recently showed a significant drug–nutrient transport interaction at the lactating mammary gland, 21 and our current data suggest that the outcomes of a drug–nutrient transport interaction may vary qualitatively and quantitatively at different lactation stages. Furthermore, this new evidence in general is disconcerting because drugs considered “safe” during breastfeeding are known to inhibit nutrient transport at other blood–epithelial barriers that have led to adverse outcomes, 40,41 possibly requiring nutritional interventions to alleviate a nutrient deficiency. 42,43 The outcome of drug–nutrient transport interactions at the lactating mammary gland and in the developing neonate following breast milk exposures to drug is relatively uninvestigated.
Chemicals transferred into milk may pose a significant health risk to the nursing neonate. Exposure indices that estimate neonatal drug exposure following maternal exposures to chemicals variably consider the key determinants of risk. Our study confirms the importance of considering the ability of neonate to eliminate an exposed dose due to age-dependent changes in elimination mechanisms as a critical determinant of exposure risk. More importantly, our study confirms the importance of M/S and the need to consider lactation stage for compounds that are substrates for transporters expressed at the lactating mammary epithelium. For such chemicals, an evaluation of M/S at different lactation stages is necessary to avoid an over- or underestimate of the risk when M/S values are assumed to remain constant throughout most of the lactation period. Investigations are needed concerning the role of transporters in the transfer of chemicals across the blood–mammary epithelial barrier, their role in defining M/S, and how the M/S may change with lactation stage as the functional expression of these transporters change with advancing lactation. Such considerations also prompt questions concerning the influence of drugs and other exogenous factors on nutrient transport at the lactating mammary gland and on M/S values.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was funded in part by Natural Sciences and Engineering Research Council of Canada (NSERC), Grant # 261219, and by a College of Pharmacy and Nutrition Research Trust Grant.