Abstract
The acute toxicity of zinc threoninate chelate was assessed. The oral lethal dose 50% (LD50) was 2710 mg/kg in female rats and 3160 mg/kg in male rats. Genotoxicity was assessed by Ames test in
Zinc (Zn) is one of the important essential trace elements found in biological systems and also in many metalloproteins and metalloenzymes that exist in living organisms. As a component and cofactor of enzymes, it can regulate and control the structure and function of nearly 300 enzymes in vivo and thus affect various metabolic activities of the body. Zinc also has many pharmacological and nutritional roles, such as promoting growth performance, 1 –3 enhancing immunity, 4 and affecting reproductive ability. 5 Another interesting pharmacological role of zinc is that zinc can act as an insulinomimic 6 and improve noninsulin-dependent diabetes mellitus. 7,8
Inorganic zinc salts (such as zinc sulfate, zinc chloride, zinc acetate, zinc carbonate, and zinc oxide) had been used in animal feed to promote animal growth in the past. However, it was found that zinc ion in inorganic zinc salts could react with phytic acid, oxalic acid, and carbonic acid in intestine to form insoluble salts and result in a low bioavailability of zinc. 9 Increasing zinc absorption by increasing the zinc content of a diet causes problems because it may lead to the interaction of zinc with other minerals, such as iron, copper, and selenium, whose physical and chemical properties are similar to zinc, and reduce the absorption of the minerals. 10 –13
It is known that zinc amino acid chelates (such as zinc-methionine, zinc-lysine) are much better than inorganic zinc salts in bioavailability because they can prevent Zn from binding to nonabsorbable compounds in the diet. Alternatively, the ligands may directly facilitate Zn absorption as shown in a Zn absorption model proposed by Cousins. 14 Because Zn exerted its biological activity almost entirely as part of complex molecules, 15,16 one speculated that the complexed or chelated organic Zn sources were more effective than inorganic Zn sources in restoring Zn concentrations by increasing Zn absorption and (or) enhancing Zn retention. It was found that when dogs were administered zinc through the diet at an equivalent dose of 50 mg Zn/kg for 2 weeks, zinc from the zinc amino acid chelate led to dogs growing more hair than when they were fed zinc from zinc oxide. This indicated that the zinc from the amino acid chelate may be more bioavailable to the dog. 11 It was also found that higher bone Zn concentrations were observed in chicks fed by diets supplemented with zinc-methionine than in those supplemented with zinc sulfate or zinc oxide. 17 In young pigs, chicks, and lambs, Zn was considerably more available in zinc-methionine than in zinc sulfate. 1,18 So some zinc amino acid chelates are prepared to replace the inorganic zinc salts now.
Zinc threoninate chelate is a new zinc fortifier and can supply zinc and threonine (Thr), which are both essential for human beings and animals. As the compound is chelated by zinc ion and Thr and can supply more available Zn than the inorganic zinc salts, it has plenty of advantages like other zinc amino acid chelates,
12,17,18
including good stability, high absorption and utilization rate, high biological value, and so on. It had first been prepared by adding zinc sulfate to an aqueous solution of the lithium salt of
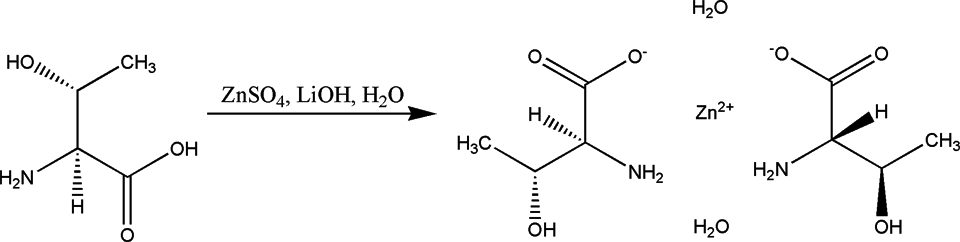
The synthetic route of zinc threoninate chelate.
The effect of zinc threoninate chelate in lowering blood glucose had been studied. The high blood glucose level of KK-Ay mice with type 2 diabetes mellitus could be lowered by daily intraperitoneal injections of a zinc(II) complex, cis-[Zn(
It was envisioned that zinc threoninate chelate may be used as a kind of nutritional fortification substances and antidiabetic drugs. To prepare for its clinical use, safety assessment of zinc threoninate chelate was measured by acute, subacute toxicity, genotoxicity, and teratogenicity trials in mice and rats in this study.
Materials and Methods
Source of Zinc Threoninate Chelate
Zinc threoninate chelate used in this study was provided by State Key Laboratory of Food Science and Technology in Nanchang University in China. The purity of zinc threoninate chelate was more than 99%.
Zinc threoninate chelate used in current study was synthesized in aqueous solution with zinc oxide and
Animals
All female and male imprinting control region (ICR) mice and Sprague–Dawley (SD) rats for acute, subacute toxicity, genotoxic tests were supplied by Shanghai Slack Laboratory Animal Co, Ltd, China. Sixty healthy mature virgin female SD rats (228-298 g) and 50 healthy mature male SD rats (300-350 g), which were all 3 months old, for teratogenic tests were supplied by Zhejiang Laboratory Animal Center, China. All animals were bred in the animal facility and housed in a temperature- (20°C-25°C) and humidity- (40%-70%) controlled environment with free access to water and food.
The study was approved by Laboratory Center for Health Analysis of Nanjing Medical University, China, and conducted in compliance with the Code of Federal Regulations (CFR), Title 21, Part 58 (Good Laboratory Practice for Nonclinical Laboratory Studies).
Acute Toxicity Test in Rats
Acute toxicity test was performed to determine the lethal dose 50% (LD50) using Horn’s method. A total of 40 clean healthy SD rats (20 per sex) with body weight 185 to 200 g were used after medical inspection for 3 days. Rats were fasted for 16 hours with free drinking before the test. Zinc threoninate chelate was diluted with corn oil. Female and male rats were randomly divided into 4 dosage groups at doses of 1000, 2150, 4640, and 10 000 mg/kg and received intragastric administration at a dosing volume of 20 mL/kg for each administration. They were consecutively observed for 14 days for toxic signs and mortality.
Genetic Toxicity Studies
Ames test
According to reference 20, Ames test was performed as plate incorporation test with
Triplicate plates were evaluated, in the presence or absence of S9 mix, with the sample volume of 0.1 mL/plate. Then 0.1 mL bacterial culture, 0.1 mL test sample solution, and 0.5 mL S9 mix (when metabolic activation was required) were added into the top agar (2 mL) and they were poured onto the basal medium after blending. The strains were incubated at 37°C for 48 hours. The revertant colony number of each plate was evaluated.
If the mean revertant colony number of each cheated group was 2-fold or more than that of the negative control group and a dose-reaction relation existed, the result was considered positive. The whole test was repeated once under the same conditions. 21 –23
Bone marrow micronucleus test
To design the administration dose in mice, the LD50 of zinc threoninate chelate for mice was tested. A total of 40 ICR healthy mice with body weight 19.0 to 22.0 g were used after they had adapted to the environment for 3 days. All mice were randomly divided into 4 groups with 10 mice each (5 per sex). Mice in 4 treated groups were fed at doses of 215, 464, 1000, and 2150 mg/kg to perform the acute oral toxicity test. According to Horn’s table, LD50 of zinc threoninate chelate for female and male mice were 794 mg/kg and 501 mg/kg, respectively.
A total of 50 ICR healthy mice with body weight 27.0 to 29.8 g were used in the micronucleus test after they had adapted to the environment for 3 days. All mice were randomly divided into 5 groups with 10 mice each (5 per sex). Samples were diluted with corn oil. Female mice in the zinc threoninate chelate groups were fed once at doses of 100, 200, and 400 mg/kg while male mice in the zinc threoninate chelate groups were fed once at doses of 62.5, 125, and 250 mg/kg. Mice in negative and positive control groups were given once corn oil and 40 mg/kg cyclophosphamide, respectively.
According to the 30-hour method, the mice received intragastric administration twice at the dosing volume of 10 mL/kg over a 24-hour interval. Six hours after the second administration, the mice were sacrificed and harvested for bone marrow of mediastinum. Their breast bones were conventionally prepared to slice and observed under microscope. A total of 1000 polychromatic erythrocytes (PCEs) per mouse were evaluated for the occurrence of micronuclei by permillage.
The results were statistically analyzed by using Poisson distribution
Mice sperm abnormality test
A total of 25 ICR healthy mature male mice with body weight 29.5 to 33.1 g were used in the sperm abnormality test after they had adapted to the environment for 3 days. All mice were divided into 5 groups with 5 mice each by body weight. Zinc threoninate chelate was diluted with corn oil and the dosing volume was 10 mL/kg. Mice in treated groups received intragastric administration once daily at doses of 6.25, 12.5, and 25.0 mg/kg, while mice in the negative and positive control group, respectively, received intragastric administration once daily with corn oil and 40 mg/kg cyclophosphamide for 5 consecutive days.
The mice were sacrificed 35 days after the first administration, 2 sides of the epididymal sperm filtrate was conventionally prepared to slice and observed under microscope. A total of 1000 sperm cells of integrity structure per mouse were assessed for the occurrence of abnormality by percentage. 24 The results were statistically analyzed by using rank sum test.
Thirty-Day Subacute Toxicity Study in Rats
A total of 80 healthy SD rats were used after medical examinations for 3 days. The rats were randomly divided into 4 groups with 20 rats each (10 per sex) by body weight. Zinc threoninate chelate was mixed into basic feed and the rats were orally administered daily at doses of 42, 169, and 675 mg/kg. The target dosage levels were 1/64, 1/16, and 1/4 of LD50 of zinc threoninate chelate in female rats, respectively, according to the acute toxicity test. The negative control group was given basic feed for 30 consecutive days.
The body weights of rats were evaluated once per week and their intake of feed was evaluated twice per week. Weekly and total food utilization of rats was also calculated and the rats were observed everyday to see whether there were toxic signs and deaths. The following parameters were measured during the study: Body weight and food utilization rate. Hematological parameters: Blood in the rats was analyzed with CELL-DYN 3700 blood cell analyzer for white blood cell count, RBC count, hemoglobin level, and the rate of various white blood cells. Blood biochemical parameters: Serum in the rats was analyzed with Japan Hitachi 7020 biochemical automatic analyzer for determination of serum aspartate aminotransferase (AST), alanine aminotransferase (ALT), total protein, albumin, urea nitrogen, creatinine, total cholesterol, triglyceride, and glucose. Organ weight: The rats were killed and their liver, kidneys, spleen, and gonads were carefully separated and washed with normal saline. They were weighed after being hydrated with filter paper. The absolute and relative weights (organ/body weight) were calculated. Pathological examination: The liver, spleen, kidneys, stomach, duodenum, and gonads of rats in the control group and the high-dose group were fixed with 10% formalin, embedded with paraffin, prepared to slice, and hematoxylin and eosin (H&E) stained, and then were examined histopathologically.
25
Data were tested for homogeneity of variance by Statistical Package for the Social Sciences (SPSS) 10.0 software. Single factor variance analysis was used if their variances were homogeneous and variable transformation was used if there were abnormal distributions or their variances were heterogeneous. Rank test was used for abnormal distribution.
Teratogenic Toxicity Study in Rats
Same number of female and male SD rats was arranged in same cage. The vaginal plug was checked daily to determine the pregnancy. The day of checked pregnancy was set as day 0. If the female rats were not pregnant, the male rats were changed. The pregnant rats were randomly divided into 4 groups by body weight with 15 rats each. Zinc threoninate chelate was diluted with corn oil and the dosing volume was 10 mL/kg. The rats in treated groups were treated once daily at doses of 42, 169, and 675 mg/kg while the negative control group was treated once daily with corn oil by gavage on day 7 to 16 of pregnancy. Body weights were taken on the days 0, 7, 12, 16, and 20. According to the body weight, the dose volume of the test article was individually calculated.
During the study, the animals were checked for general health condition daily. Food and water consumption were recorded. On day 20, pregnant rats were sacrificed by CO2 anesthesia and the uterus was collected and weighed. The number of corpora lutea, implantations, absorbed fetuses, early dead fetuses, late dead fetuses, and live fetuses were examined and recorded. Sex, body weight, body length, and external malformations for live fetus were recorded as well.
Half of the live fetuses in each litter were fixed in Bouin’s fluid for 2 weeks, followed by organs examination by the razor-section technique. 26 The remaining fetuses were decorticated, eviscerated, degreased, and then stained with alizarin red solution until skeleton became red. 27 Fetal rats were put into transparent liquid A for 1 to 2 days and then into transparent liquid B for 2 to 3 days. After bones had become incarnadine, and purple in soft tissues had basically faded, they were put into glycerine to observe for skeletal malformations. 28 –30
The SPSS 10.0 software was used to perform statistics. All rates were analyzed by χ2 test. Weight gains of pregnant rats were analyzed by analysis of variance (ANOVA) or nonparametric statistics. Fetus body length and body weight, average live fetus number of each litter, uterus and fetus weight were compared by
Results
Acute Toxicity
Mortality was observed in both male and female rats only on days 1 and 2 following dosing. The oral LD50 of zinc threoninate chelate was 2710 mg/kg in female rats and 3160 mg/kg in male rats.
Genotoxicity
Ames test
The revertant colony number of each treated group did not exceed 2-fold of that in the negative control group and had no dose-reaction relation (Table 1). This result indicated zinc threoninate chelate had nonmutagenic effect on
The Revertant Colony Number (
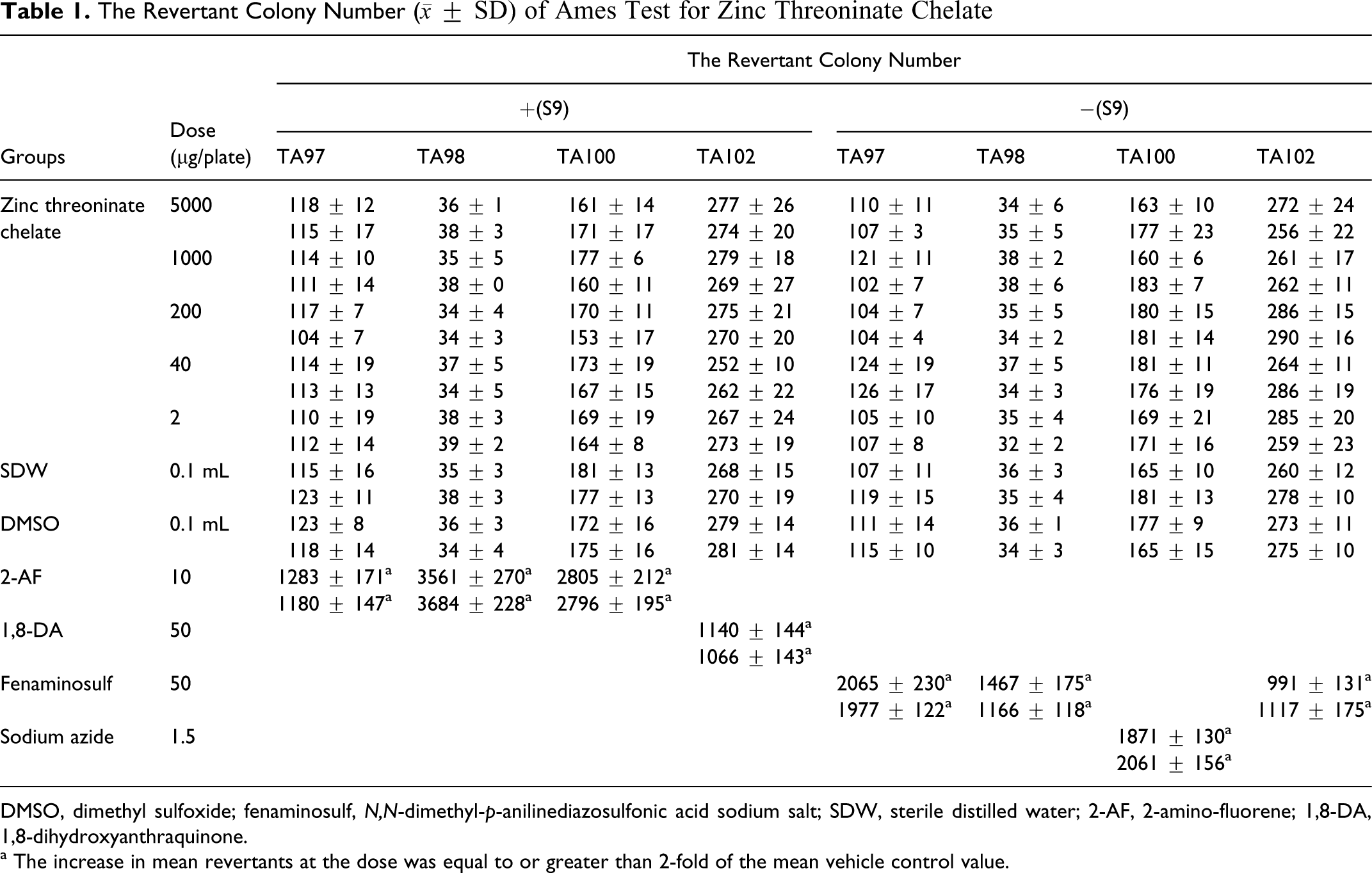
DMSO, dimethyl sulfoxide; fenaminosulf,
a The increase in mean revertants at the dose was equal to or greater than 2-fold of the mean vehicle control value.
Bone marrow micronucleus test
The difference of the PCE micronuclei rate was not significant between each treated group and the negative control group (
The Effect of Zinc Threoninate Chelate on the Mice Bone Marrow Micronucleus Rate and the PCE/RBC
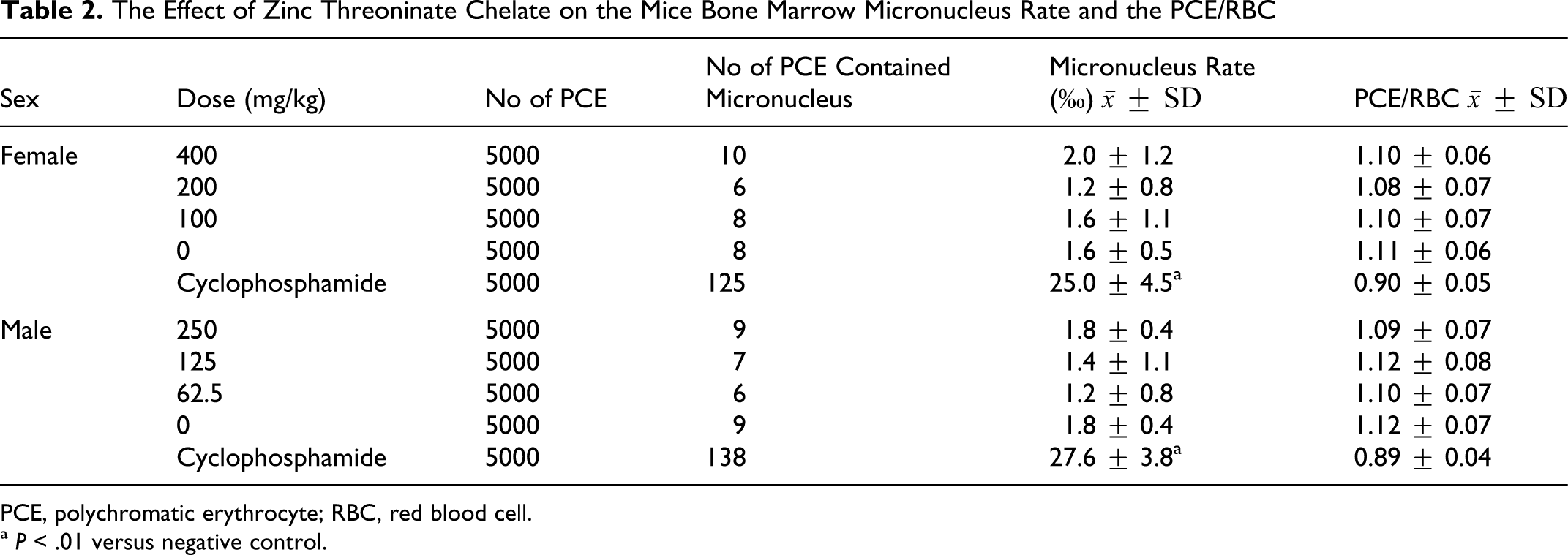
PCE, polychromatic erythrocyte; RBC, red blood cell.
a
Mice sperm abnormality test
The difference of the sperm abnormality rate was not significant between each treated group and the negative control group. However, the incidence of abnormally shaped sperm cells was significantly increased in the positive control group where mice were given orally 40 mg/kg cyclophosphamide as compared with the negative control group (
The Effect of Zinc Threoninate Chelate on Abnormality in Mice Sperms
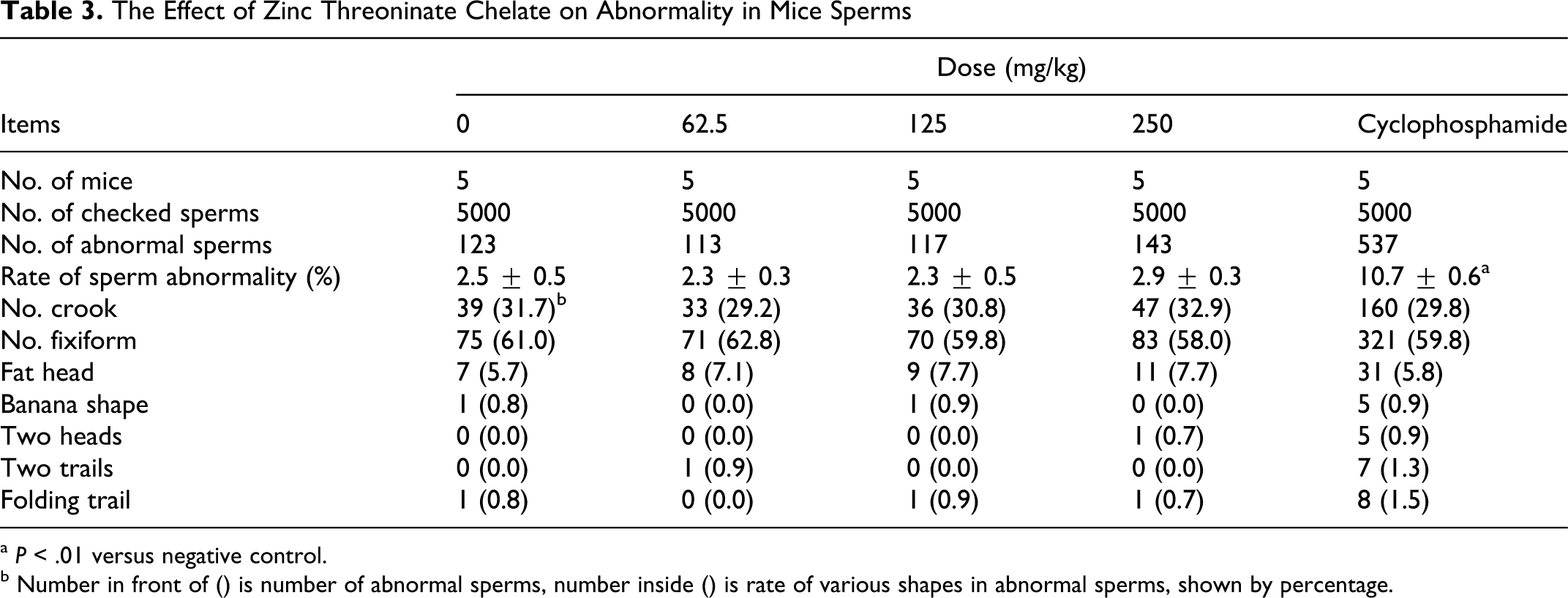
a
b Number in front of () is number of abnormal sperms, number inside () is rate of various shapes in abnormal sperms, shown by percentage.
Thirty-Day Subacute Toxicity
All rats had a similar body weight gain throughout the experiment and there was no difference in food intake and food utilization rate between the treated group and the control group.
The data for hematology and blood biochemistry are shown in Tables 4 and 5. It could be seen that the eosinophil rate of female rats in 675 mg/kg group was significantly higher than that of the control group (
The Effect of Zinc Threoninate Chelate on Hematological Parameters of Rats (
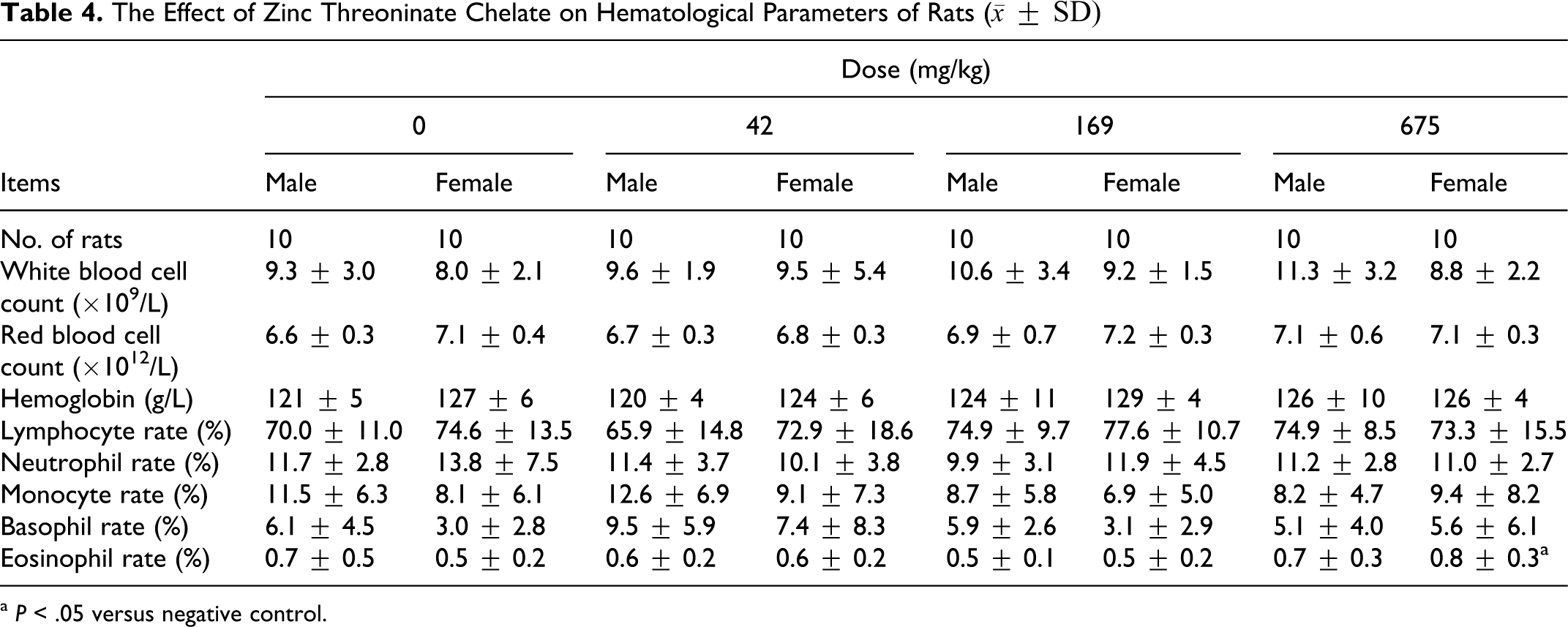
a
The Effect of Zinc Threoninate chelate on some blood biochemical parameters of rats (
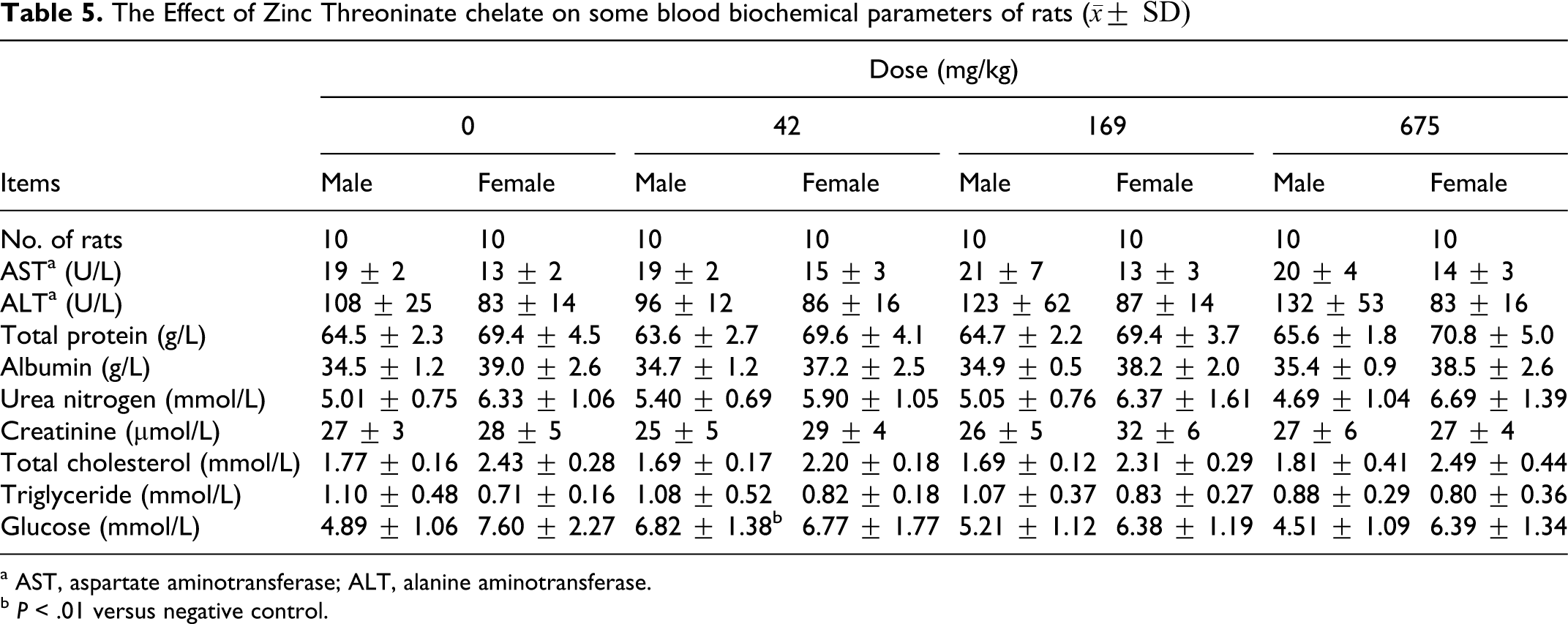
a AST, aspartate aminotransferase; ALT, alanine aminotransferase.
b
The absolute and relative weights of liver, kidneys, spleen, and gonads of zinc threoninate chelate–treated rats remained unchanged in comparison with the control animals.
Gross anatomy of rats in each group was conducted. The organs of female and male rats in the high dose and the control group (10 cases each) were observed under microscope. No significant changes were observed in liver, kidneys, spleen, stomach, duodenum, and gonads of rats in the control group as well as the female rats in the high dose group. There was 1 case of low-grade inflammatory cell infiltration at portal area and 2 cases of low-grade fatty degeneration in liver but no significant abnormality in other organs of the male rats in the high dose group.
Teratogenicity
No abnormality was detected for activities of pregnant rats in each dose group during the observation period. Moreover, no effects were observed on body weight gains and food consumption of pregnant rats between each treated group and the control group, but water consumption in 42 mg/kg and 169 mg/kg groups was more than that of the control group.
No significant differences between each treated group and the control group were observed in embryonic development in the fetuses (Table 6 ). Furthermore, external and splanchnic malformations were not observed in the fetuses throughout the teratogenic toxicity test.
The Effect of Zinc Threoninate Chelate on Embryonic Development in the Fetuses
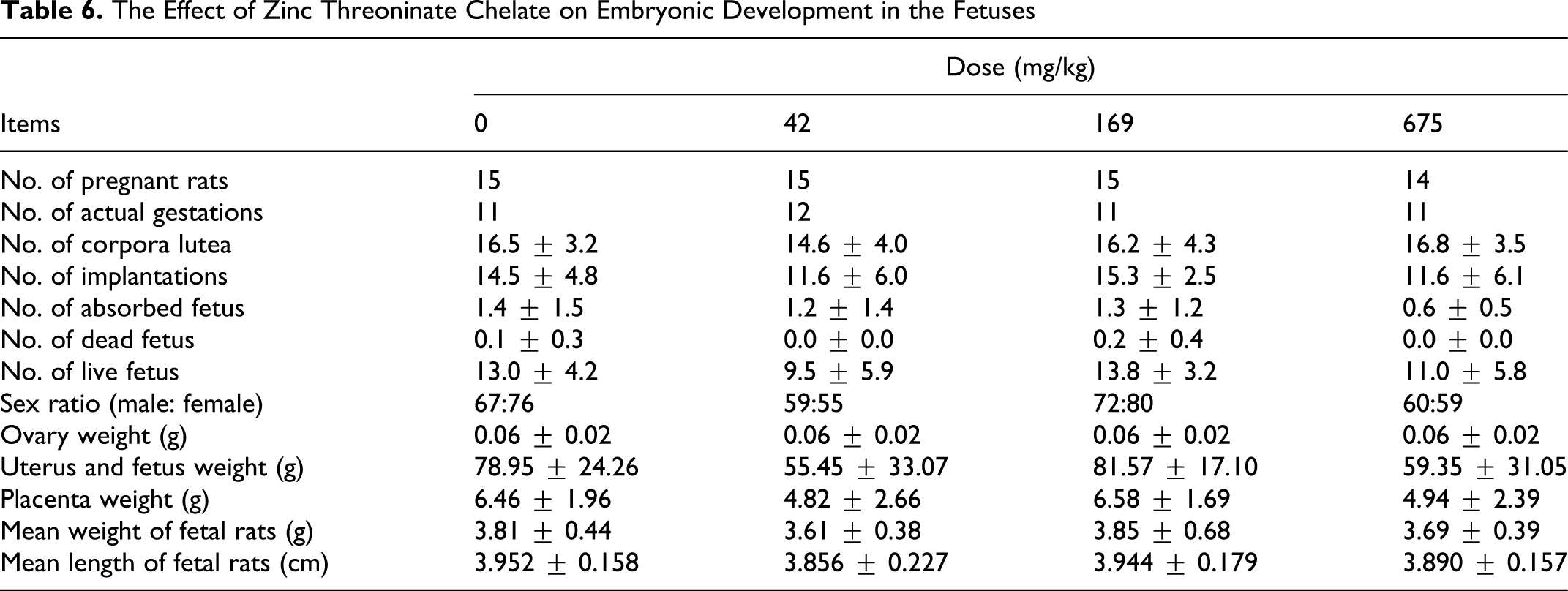
Table 7
showed that the rate of sternum malformations in the fetuses was significantly higher in 169 and 675 mg/kg groups than that in the control group (
The Effect of Zinc Threoninate Chelate on Skeleton Development in the Fetuses

a Number in front of () is number of malformed fetuses, number inside () is rate of abnormality, shown by percentage.
b
c
Discussion
In the current study, the acute, subacute, and genetic toxicity of zinc threoninate chelate was evaluated in mice and rats. The acute toxicity test showed the LD50 of zinc threoninate chelate was between 501 mg/kg and 5000 mg/kg, which was considered low toxicity according to Chinese Standard for Toxicological Assessment of Health Food. 31
The genotoxic effects were examined using Ames test, bone marrow micronucleus, and sperm abnormality assays in mice. No significant alteration on morphologically abnormal spermatozoa was observed, which suggested that there was no genetic damage on male reproductive organs by supplementation of zinc threoninate chelate. Moreover, no mutagenic effects and abnormal micronucleus formation were detected, which indicated that zinc threoninate chelate produced no genetic damage on mice.
The teratogenicity test revealed that the chelate had no significant teratogenic effect in rats at a dose of 42 mg/kg. When pregnant rats were administered zinc threoninate chelate at doses of 169 and 675 mg/kg, malformations in fetal rats were mainly incomplete development of the sternum, while other parameters were not significantly different from the control group. The observation may be due to the antagonistic effect of zinc on calcium in skeleton development of fetal rats, 32 yet the occurrence of which could be avoided by adding calcium. This result also further illustrated the role of zinc supplementation in zinc threoninate chelate.
In the repeated toxicity studies up to 675 mg/kg in rats for 30 days, no genotoxicity or adverse effects were observed on physical examination, hematology, blood biochemistry, and histopathology, indicating that zinc threoninate chelate produced no adverse effects. Zinc threoninate chelate had no deleterious effects on health status, growth, or development of the animals. Renal function was evaluated by means of urea nitrogen and creatinine serum levels. Urea and creatinine are compounds derived from proteins, which are eliminated by the kidney. The fact that rats treated with zinc threoninate chelate at doses of 42, 169, and 675 mg/kg showed unchanged urea nitrogen and creatinine levels suggested no alterations in kidney function. Liver enzymes, such as AST and ALT, were also not affected by zinc threoninate chelate administration, which indicated that there was no liver cell injury or hepatic function changes.
In summary, the investigation on the acute, subacute, genetic toxicity of zinc threoninate chelate in mice and rats was performed. No dose-dependent effect was observed in any of the parameters measured, which suggested the absence of a treatment-related adverse effect of zinc threoninate chelate. Below a daily dose of 42 mg/kg, zinc threoninate chelate had no teratogenic effect. The data in this article supported the safety of zinc threoninate chelate for human clinical trials and indicated that zinc threoninate chelate could be used as a zinc fortifier.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The support to this project by Laboratory Center for Health Analysis of Nanjing Medical University and the Program for Chang Jiang Scholars and Innovative Research Team in University (No IRT0540) is gratefully acknowledged.
