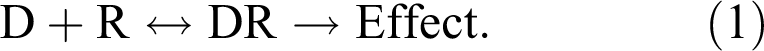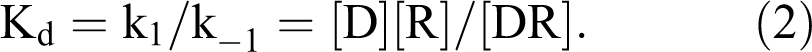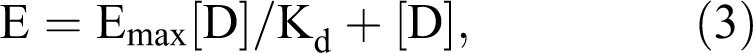Abstract
Hormesis describes dose-response relationships characterized by a reversal of response between low and high doses of chemicals, biological molecules, physical stressors, or other initiators of a response. Acceptance of hormesis as a viable dose-response theory has been limited until recently, in part, because of poor conceptual understanding, ad hoc and inappropriate use, and lack of a defined mechanism. By examining the history of this dose-response theory, it is clear that both pharmacological and toxicological studies provide evidence for hormetic dose responses, but retrospective examination of studies can be problematic at best. Limited scientific evidence and lack of a common lexicon with which to describe these responses have left hormesis open to inappropriate application to unrelated dose-response relationships. Future studies should examine low-dose effects using unbiased, descriptive criteria to further the scientific understanding of this dose response. A clear, concise definition is required to further the limited scientific evidence for hormetic dose responses.
Keywords
The term hormesis describes a dose-response relationship characterized by opposing effects at low and high doses. A diverse range of chemical (eg, Cd2+, Cu2+, phenol) and physical (eg, irradiation) stressors have been observed to elicit a hormetic-like response. Opposing effects of low and high doses have been observed across multiple experimental systems since the late 19th century. Lack of a conventional terminology and misconception has resulted in the use of numerous terms (eg, Arndt-Schulz law and Hueppe’s rule) and descriptors (eg, U-shaped, inverted U-shaped, J-shape, β-curve, biphasic, bidirectional, atypical, opposite effects, dual effects, and paradoxical effects) to describe the observed nonmonotonic nature of such responses. The term hormesis was coined in 1943 by Southam and Ehrlich 1 to describe the growth stimulation of wood-decaying fungi induced by low doses of natural antimicrobial agents in cedar wood.
Initial reports of hormetic-like responses described studies investigating the effects of heavy metals and other known toxic agents on endpoints primarily related to growth and survival. The key feature of those observations was a dichotomy between low and high doses of a toxic substance producing responses deemed to be “beneficial” and “harmful,” respectively. The general observation of opposite effects between low and high doses was subsequently reported in increasingly diverse model systems. Initially, hormesis was considered a concept describing beneficial effects of low-dose exposure to toxic substances. However, the idea of beneficial effects of toxins was intuitively paradoxical and consequently difficult to accept as a valid scientific treatise. As a result, hormesis was quickly marginalized and has only recently begun to regain scientific acceptance.
A contemporary view of the hormetic dose response has gained renewed interest as championed by a small group of investigators, most notably E. J. Calabrese. The increased number of publications referencing hormesis attests to the success that proponents of the concept have had in increasing awareness within the scientific community, particularly in the field of toxicology. In fact, the number of references to hormesis in a search of PubMed has gone from 35 in the 1980s to 79 in the 1990s and surpassed 520 in the 2000s. Despite this increase in acknowledgment, the term hormesis remains poorly defined in those publications. In addition, attempts to address the fundamental features of hormetic responses continue to be associated with pseudo-mechanistic features, such as adaptive response, resource allocation, and requirement of temporal components that are ill-defined and prove conceptually problematic. Furthermore, despite the lack of clear fundamental understanding and the lack of a mathematical model to define the hormetic response, some proponents have called for a paradigm shift that would place hormesis as “the fundamental” dose-response theory. 2 They suggest that the wide range of biological models in which hormetic responses were observed and the diversity in the types of substances tested and responses observed from those substances are evidence for their claim. 3 Unfortunately, the polarized and dogmatic position that threshold theory must be rejected and replaced by hormesis 4 has increased the resistance of accepting the existence of low-dose effects 5,6 (ie, effects below the threshold or no observable adverse effect level or NOAEL).
A Historical Review of Hormesis
Hormesis is not a new concept; biological scientists have theorized and provided experimental evidence for hormetic-like responses since the late 19th century. From inception, supporters of hormesis have argued that hormesis is the fundamental dose-response relationship because it appeared universal with respect to biological models, endpoints measured, and chemical or physical agents studied. Hormesis is also an attractive concept because it can account for effects at doses below reported threshold levels, thus extending the dose-response continuum.
Historical and Social Influences on the Nature of Concentration Response Relationships—“The Middle Way”
The concept of the dose response has been recognized, if not understood, since ancient times. Many ancient aphorisms expounded the importance of the right measure for healthy living. Ancient Greek adages such as meden agan (nothing too much), which was inscribed on the Apollo Temple at Delphi (~600
Influence of Paracelsus on Toxicology
In the 16th century, the Swiss-German physician Thiophrastus of Hohenheim (Paracelsus) extended the maxims of moderation to the context of drugs and poisons. Defending his practice of using poisons to treat the sick, Paracelsus proclaimed, “Alle Dinge sind Gift und nichts ist ohne Gift, allein die Dosis macht es, dass ein Ding kein Gift ist.” (All things are poison and nothing is without poison, only the dose permits something not to be poison. 8 ) Paracelsus had recognized that the toxicity of poisons is due to specific chemicals, and the body’s response to the chemicals is dependent on the dose. Paracelsus reasoned that therapeutic agents can be harmful at high doses, and conversely substances considered toxic can be harmless or even beneficial at lower doses. His understanding of dose and effects has been paraphrased by modern toxicologists in the statement “The dose makes the poison.”
Scientific Contributions of Hugo Schultz to Hormesis
One of the first scientific studies demonstrating low-dose stimulatory effects of toxic substances was performed by Hugo Schulz. 9 Schulz believed biological systems were distinct from chemical or physical systems in that the former produced bidirectional responses depending on dose, whereas chemical or physical systems functioned according to thermodynamic rules in a unidirectional manner regardless of reagent concentration. 10 Schulz believed reversal of response to be a universal biological phenomenon. Although never using the term hormesis to describe his understanding of the dose-response relationship, Schulz is commonly considered the founder of hormesis theory as demonstrated by the use of the name Arndt-Schulz law to describe hormetic-like dose responses in early literature. 11
Even though supporters of hormesis would defend the observations that were described by the Arndt-Schulz law, most would also agree that Schulz overextended his interpretation of his data. In addition, Schulz’s interpretations were limited by experimental design and analysis that would not meet modern scientific standards. 12 Holtz and colleagues 13 recognized that there was greater complexity to Saccharomyces cervisiae metabolism, the model system used in Schulz’s experiments, and that different dose ranges could induce different cellular responses. The observed hormetic-like dose response resulted from combining a low-dose stimulation of glycogenolysis and a high-dose inhibition of fermentation. Schulz’s lack of mechanistic understanding did not, however, invalidate the general observation of a hormetic-like effect. The validity of observing hormetic effects is dependent on the scientific quality of the work and reproducibility of the results, not upon the conceptual development or mechanistic understanding of hormesis. To paraphrase Hayes 14 in Toxicology of Pesticides, the existence of the hormetic dose response does not depend on understanding it.
Factors Contributing to the Limited Acceptance of Hormesis
An Unfortunate Association With Homeopathy
Many factors have contributed to the limited acceptance of hormesis in the past. Most of these factors stem from poor conceptual understanding and inappropriate application of hormetic effects to unrelated beliefs. As such, undue skepticism and stigma has been leveled toward any data associable with hormetic-like responses. The perception that hormesis is associated with homeopathic beliefs illustrates this point well.
Homeopathy is an alternative field of medicine developed by the German physician Christian Friedrich Samuel Hahnemann during the beginning of the 19th century. 15 Practitioners of homeopathy attempt to treat the sick with exceedingly dilute agents that at higher concentrations produce similar symptoms in healthy individuals. For most preparations, Hahnemann advocated using dilution factors of 1060, making it a statistical improbability for any molecule of the original substance to be present in the diluted preparation. 15 Homeopathic practices are inconsistent with the laws of chemistry and physics and are without scientific or clinical merit.
Upon its conception, hormesis was conveniently co-opted by the homeopathic community as the scientific principle behind high-potency dilution. Hormesis, however, is only superficially similar to homeopathic therapeutic dilution practices. Homeopathy recognizes that adverse effects can be reduced by decreasing the concentration of a toxic substance but predicts that therapeutic effects can be achieved with dilution factors much greater than Avogadro’s number. Hormesis, like traditional dose-response theory, acknowledges the same correlation between adverse effects and dose; however, hormesis further describes a low-dose response at concentrations below the toxic threshold for some chemicals. Hormesis differs from homeopathy in that it predicts low-dose effects just below threshold. Thus, identified hormetic responses will be defined by a low-dose response range that is still physically relevant.
Acceptance of hormesis in the early 20th century was significantly hindered by criticisms presented by eminent scientists, such as the pharmacologist Alfred J. Clark. In his textbooks, Clark associated Schulz and his theory of hormetic dose response to homeopathy. 16 However, Clark failed to recognize the subtle differences between hormetic and pharmacological dose responses that could resolve experimental and mechanistic discrepancies between the two. Clark 17 saw a lack of evidence for hormetic responses by pharmacological drugs as proof that hormesis was an invalid scientific thesis.
When considering pharmacological concentration response relationships, it is important to realize that pharmacological drugs are either designed or selected for specific and maximal therapeutic responses with minimal adverse effects within their therapeutic window. In contrast, chemicals with hormetic actions are neither designed nor selected for their modest subinhibitory responses. Both pharmacological agents and hormetic chemicals may induce responses below inhibitory thresholds, but the response windows can appear unrelated because of differences in the amplitude of the response, the nature of the response being measured, and the actual dosage range analyzed. Clark 17 simply failed to recognize that the therapeutic window and hormetic stimulatory window are conceptually similar, although quantitatively different and dependent on the endpoints being measured.
Hormesis did gain support from a few prominent scientists, most notably from Ferdinand Hueppe, a distinguished bacteriologist and former student of the Nobel laureate Robert Koch. In his influential textbook Principles of Bacteriology, Hueppe claimed independent discovery of low-dose stimulatory effects in his bacteriological studies. However, neither experimental data nor references to experimental data were provided.
18
Hueppe gave credit of first discovery to Schulz, yet because of his strong scientific reputation, low-dose stimulatory effects of poisons became known as Hueppe’s rule.
11
Hueppe defended the scientific value of hormesis and accurately outlined the inappropriate association of hormesis with homeopathy. Low-dose stimulation stood on its own as a solid scientific foundation and should not be rejected because some homeopathic practitioners had adopted its premise to support their practices.
18
Concerning the General Nature of Hormesis
The fundamental understanding of hormesis remains limited because of a poor definition of a hormetic response. A specific criterion for classifying a response as hormetic does not exist. For example, some have insisted that low-dose responses must have “beneficial” effects, 19 whereas others have attempted to decouple beneficial effects from the definition of hormesis. 20 In addition, the lack of specificity in defining hormesis has resulted in indiscriminate and incorrect usage of the term. In the literature, hormetic responses are frequently not distinguished from other low-dose stimulatory responses or from different types of nonmonotonic responses. In part because of this ad hoc usage, 21 the term hormesis has been applied to responses as divergent as stimulation of bacterial growth by low concentrations of cations 22 and physiological preconditioning effects resulting from exercise and aging. 23
A common misinterpretation also relates to the proposed generalizability of hormesis, a point often used to support the argument that hormesis represents the most fundamental understanding of dose and effect. 3 The suggested general nature of hormetic responses, however, is no different from that of traditional threshold theory. Both models describe dose-response relationships for a variety of agents and endpoints across numerous biological systems. The generalizability of the agents refers to the broad classes of chemical or physical stressors that can induce a particular response. However, for a specific endpoint and a specific model system, not all substances are expected to induce a dose-dependent response (eg, the system is insensitive). This recognition holds true for both models. Thus, the hormetic dose response is a generalizable phenomenon much in the same way as the traditional threshold model.
Furthermore, the suggested generalizability of hormesis must not be confused with a suggestion of universality of the phenomenon. Early references to low-dose stimulation as Arndt-Schulz law or Hueppe’s rule conveyed an initial belief that these observations represented a universal biological phenomenon. Hormesis, as a biological law, would dictate that all chemical agents produce nonadverse responses at low doses. This, however, is not the case. For each biological model, only a subset of chemical agents that may span across broad chemical classes have the potential to induce modest responses at subinhibitory concentrations. To misinterpret hormesis as a universal phenomenon would justify the contention that the concept lacks sound scientific basis and would be at odds with the fact that it is often not observed even in appropriately designed studies. 3
Lack of Mechanistic Understanding for Hormetic Responses
Many reported hormetic responses are also criticized for lacking mechanistic understanding. The hormetic dose-response relationship is not adequately described by any model comparable to the receptor occupancy theory developed in pharmacology. In addition, little is known of the molecular and biochemical mechanisms associated with hormesis because they are likely the combination of multiple different responses as was the case in the effects defined by Schulz.
Stebbing 24 might be considered the first to have proposed a general mechanism for hormetic responses. He suggested that hormesis is a normal function of adaptive control mechanisms responsible for maintaining homeostasis in response to perturbation by chemical or physical stressors. Using different marine and aquatic systems to examine biomass accumulation under various chemical and physical challenges, the authors measured biomass accumulation and calculated growth rates at a range of doses. From the derivation of their data, Stebbing reported oscillations in rate of biomass accumulation in response to these various challenges over time; furthermore, he reported concentration-dependent changes in amplitude and duration of oscillations as well as overall rate of growth, suggesting a delay in regulatory response to perturbation. 24 The author interpreted the reported oscillations in growth rate as delayed hyperresponsiveness of adaptive control mechanisms responsible for maintaining homeostasis, or homeohoresis. The low-dose hormetic response was then interpreted as the result of this adaptive hyperresponsiveness that resulted in an apparent stimulation of colony growth.
The interpretation of hormesis as an adaptive response has more recently been adopted by Calabrese and Baldwin. 20 The concept was interlaced into their definition of hormesis by saying that it is “an adaptive response characterized by biphasic dose-responses of generally similar quantitative features with respect to amplitude and range of the stimulatory response that are either directly induced, or the result of compensatory biological processes following an initial disruption in homeostasis.” 20 Calabrese and Baldwin argued, as suggested by Stebbing, that the hormetic stimulatory effect is a consequence of overcompensation from the reestablishment of homeostasis. They considered the “modest overcompensation” to be a key feature of hormetic responses because it “functionally links hormetic responses to homeostasis, a universal biological concept, providing the theoretical foundation for the broad generalizability of hormetic phenomenon.” Thus, hormesis was rationalized to represent a common evolutionary-based strategy for the reestablishment and maintenance of homeostasis following a toxicological insult. 20
It is apparent that the results observed by Stebbing, much like those of Schulz, involve complex inter- and intracellular processes not definable by observation of a gross endpoint such as biomass accumulation. After further analyzing published data, it appears that there are multiple oscillations in colony growth rates per population doubling, an effect not explained by delayed hyperresponsiveness in individual organisms as suggested by Stebbing 25 and Calabrese and Baldwin. 20 As in the case of Schulz, there appears to be a clear hormetic effect in the chosen endpoint. However, the mechanisms driving that observed effect are not clear and thus speculative. In short, the adaptive mechanism has been used as a convenient, though unjustified and unnecessary, explanation for specific features of hormetic dose responses, such as the modest amplitude of the response and the perceived temporal delay.
Including such justifications for the existence of the phenomenon does not contribute to the conceptual understanding of hormesis. Furthermore, the adaptive mechanistic component is irrelevant to the definition of hormesis; the principle function of the definition is to describe the common features of an observable biological phenomenon (ie, a reversal of response from low to high doses), thus allowing for accurate identification of said responses. Therefore, compounds that elicit hormetic responses can be identified, and subsequently the specific molecular and physiological mechanisms responsible for generating a hormetic response can be investigated and understood with the same set of experimental approaches used to investigate other growth-related responses. In addition, the diversity of agents observed across chemical classes that can induce hormetic-like responses would suggest that the responses are likely due to many different mechanisms that cannot be characterized adequately by a single general mechanism.
Hormesis Defined
Here, we propose that a single, unambiguous, and streamlined definition of hormesis be used during all discussion and investigation of the concept. Hormesis is a dose-response relationship for a single endpoint that is characterized by reversal of response between low and high doses of chemicals, biological molecules, physical stressors, or any other initiators of a response.
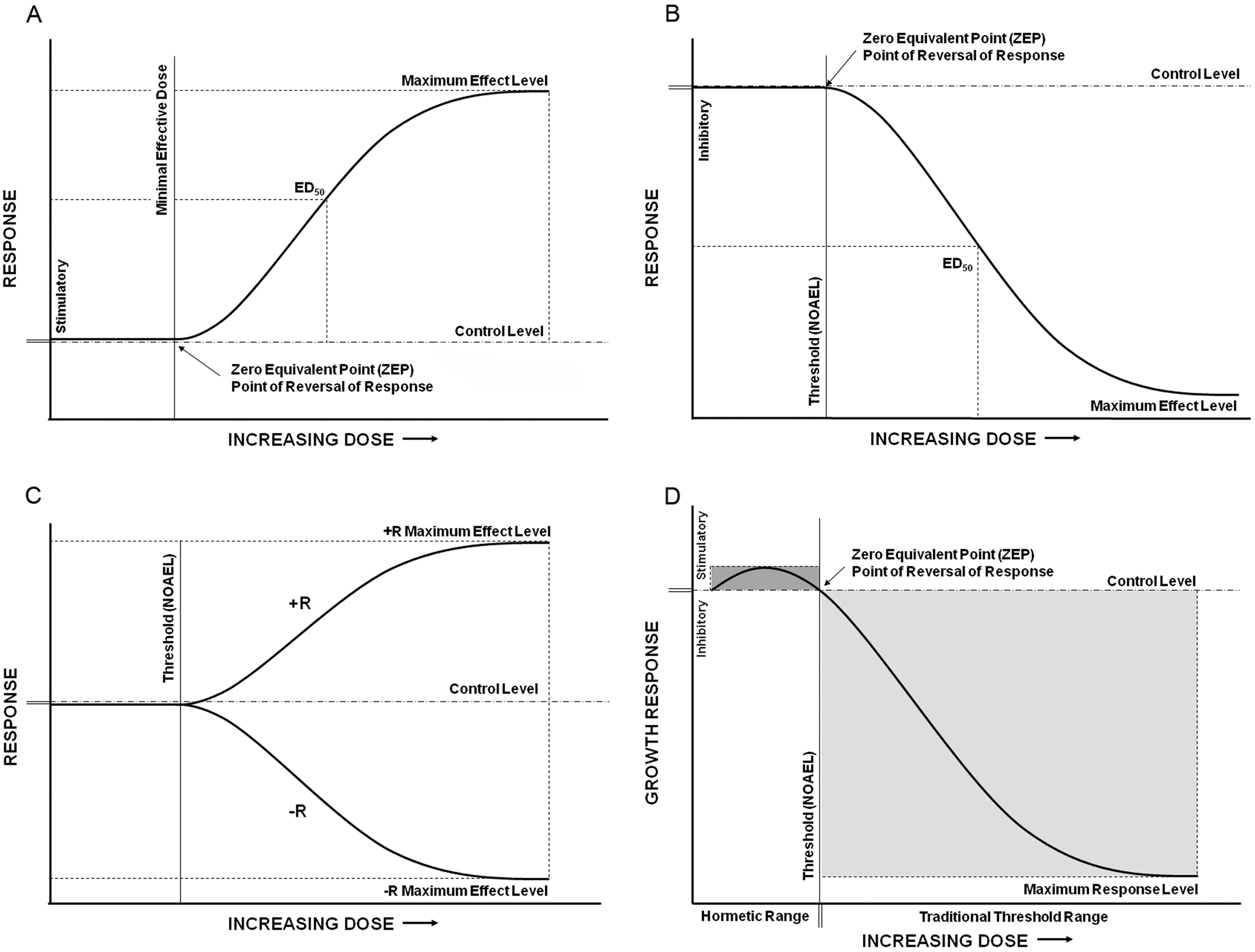
(A) A classic pharmacological dose-response curve where the response is plotted on the y-axis with the log of the dose plotted on the x-axis. The ED50 value represents a dose that produces a response that is 50% of the maximum response. (B) The classic toxicology endpoint of lethality also produces a sigmoid-shape response curve, which is typically inverted to reflect the adverse nature of the response. (C) The α-curve is indistinguishable from control at low doses but becomes progressively inhibitory at doses above the threshold. The direction of the response is indicated by + R and – R for stimulatory and inhibitory/adverse effect, respectively. (D) The β-curve response type is characterized by a single stimulatory peak (outlined by a dark gray box) at doses immediately below the inhibitory threshold and an inhibitory response (as shown within the light gray box), similar to the α-curve, at doses above the threshold.
Low-Dose Stimulation and High-Dose Inhibition
Recent discussions on hormesis have typically described the concept as a dose-response phenomenon displaying low-dose stimulatory and high-dose inhibitory effects, with greater emphasis placed on the low-dose effects. These discussions are consistent with our proposed definition of hormesis. However, the requirement of hormetic responses to display both a low-dose effect and an opposing high-dose effect is rarely stated explicitly. Observations of low-dose stimulation alone do not constitute evidence for a hormetic response. Yet such observations were often mislabeled as hormetic and often result from experiments biased to detect low-dose effects while ignoring the opposing high-dose effects. Studies may also have been analyzed in ways that limit or eliminate detection of high-dose effects. Similarly, identifying hormetic responses on the basis of only low-dose responses perpetuates misunderstanding of the concept.
Despite attempts to dissociate defining hormetic responses with the context-dependent assessment of beneficial, the characterization of responses as stimulatory for most measured endpoints is still frequently associated with beneficial effects. The default bias is that stimulatory responses add to physiological functions, whereas inhibitory responses detract from normal function. By specifying the requirement of response-reversal in the definition of hormesis, those limitations are circumvented. Critically, reversal of response can only be observed when there is a pair of complementary (opposing) responses and is absent of potential subjective interpretation of the data. In discussions that follow, the terms positive/negative and stimulatory/inhibitory will be used to provide specific directional descriptions to the responses. These terms are used with the understanding that a pair of opposing responses must be observed and recognition of the caveats associated with using subjective terminology to discuss hormetic dose responses.
The Dose-Response Relationships of Pharmacology and Toxicology
The emergence of the field of toxicology has been heavily influenced by established principles of pharmacology. In fact, many of the founding members of the Society of Toxicology (SOT) in 1961 were initially trained as pharmacologists. 26 Thus, it is not surprising that toxicology would adopt a threshold model to explain the observed relationships between toxic agents and adverse effects. For most cases, the dose-response relationships observed in toxicological studies are qualitatively similar to those observed in pharmacological studies.
Classic responses investigated in pharmacology often produce sigmoidal dose-response curves when the responses are plotted on the y-axis and the log value of the doses is plotted on the x-axis (Figure 1A). Such responses asymptotically approach 0% and 100% at the extreme low and high concentrations tested, respectively. Those observations led to the development of the traditional threshold dose-response model, which defined the response as a function of receptor occupancy. That model predicts that no response can be observed below a threshold concentration and the response is sustained at maximum level upon receptor saturation. If responses below threshold are observed, they are considered to have arisen due to random variation. This understanding of the dose-response relationship represents the most fundamental principle of pharmacology.
The Threshold Model in Pharmacology
As discussed above, the development of the threshold model was based on observations from pharmacological studies of dose-response relationships with receptor-mediated effects. Building on the theories of Paul Ehrlich and John Langley that stated drugs initiate responses by binding to receptors, Alfred J. Clark developed a mathematical model that defines the dose-response relationship in terms of drug and receptor interaction that became known as receptor occupancy theory. Clark suggested that the interaction between drug and receptor followed the law of mass action and the concentration of the drug-receptor complex (DR) is proportional to the amplitude of response:
The interaction between drug (D) and receptor (R) is reversible, and the rate of association (k1) and dissociation (k–1) was used to calculate the dissociation constant (Kd). The dissociation constant defines the concentration of the drug that will result in half of the receptors being occupied by the drug. The dissociation constant is also considered a measure of the affinity of a drug for a receptor; the lower the value of Kd, the higher the affinity.
Clark, like others at the time, had observed the similarities between the hyperbolic nature of the drug dose-response relationship and the enzyme-substrate relationship described by the Michaelis-Menton equation. The dissociation constant (Kd) is conceptually similar to the Michaelis constant (Km), an indicator of the concentration of a substrate at which the rate of reaction is half its maximum, and is also a measure of the affinity of the substrate for its enzyme. Clark assumed that the effect of a drug is proportional to the fraction of the receptors bound by the drug, whereby maximal response is reached when all receptors are bound by the drug. Clark then described the drug dose-response relationship as
where E is the measured response for the concentration of drug [D], Emax is the maximal response produced when all receptors are bound by the drug, and Kd is the dissociation constant of the drug for the receptor system being studied. In this situation, Kd is related to EC50, which is the drug concentration that will induce a half-maximal response, and is also a measure of the potency of a drug.
Equation (3) defines a rectangular hyperbola; plotting responses on the y-axis and the log of the drug concentration on the x-axis produces a sigmoid-shaped curve. The observable dose-response range is defined by the threshold level and the maximal effect level (Figure 1A). Drug concentrations below the threshold level do not induce effects that are different from control. Increasing drug concentrations above the maximal effect level cannot induce further increases in response because the receptors are already fully occupied.
It is important to recall that the dose-response relationship defined in equation (3) refers to responses mediated by a single receptor system. Clark made some important assumptions on the nature of the drug-receptor interaction in deriving this equation. Assumptions included the following: (1) drug and receptor binds with a one-to-one relationship, (2) the drug concentration is much greater than the receptor concentration and thus is not the limiting factor, (3) the drug-receptor complex initiates an all-or-nothing response, (4) the receptor numbers are constant for the duration of the investigation, and (5) maximal response correlates with receptor saturation. 27 These assumptions are known to not be valid for all situations. The threshold model, however, remains a reasonably accurate and functional description of many dose-response relationships investigated in pharmacology.
Toxicity, Lethality, and Hormesis as a 2-Threshold Phenomenon
The dose-response curves for the classic toxicological endpoint of lethality are sigmoidal and thus reminiscent of the dose-response curves seen in pharmacological studies. The curve, however, is typically inverted to reflect an inhibitory nature of the endpoint (Figure 1B). By definition, a maximal effect concentration of a substance is lethal to 100% of a test population. Obviously, this quantal response becomes saturated with increasing dose. Lethality in response to a substance, however, is reduced with reduction of dose. The dose that induces 50% lethality in the population is the LD50 of a substance; the LD50 value of a substance is the toxicological counterpart to the ED50 value in pharmacology (ie, the dose that elicits 50% maximum response) and is an indicator of a substance’s potency. Below the threshold dose, no adverse effect can be observed—this effect threshold is labeled as NOAEL. From repeated observation of this type of concentration-response relationship, it was assumed that the dose-response relationships observed in toxicological and pharmacological studies were fundamentally the same. The assumption of the fundamental similarity between therapeutic and toxic effects implicitly accepted the threshold model as accurate and universally applicable.
Although traditional threshold theory, used to investigate inhibitory responses in toxicological studies, assumes no response below the threshold dose, the more accurate description for inhibitory dose-response relationships is that there is no inhibitory response below the inhibitory threshold that is different from the baseline of the control. Thus, it is theoretically possible that the stimulatory phase of the hormetic concentration-response curve is also sigmoidal, but effects are not recognized because of a lack in experimental sensitivity or statistical power. Limited sensitivity or power would make it impossible to detect differences even if the subthreshold dosage range is characterized. The existence of subthreshold effects is frequently considered in concentration-response studies by the observation of the lowest observable effect level (LOEL). As the name suggests, a LOEL is the lowest concentration at which a given response is observed.
The full hormetic dose-response continuum comprises 2 sigmoid-shaped curves, one curve describing a stimulatory response and the other describing an inhibitory dose-response relationship. The point at which the 2 curves intersect represents both the maximum stimulatory response and the minimum inhibitory response (Figure 2). Hormesis is, therefore, characterized by 2 thresholds, a stimulatory threshold that defines the lower dose limit for stimulatory responses and an inhibitory threshold that marks the upper limit. The upper limit coincides with the threshold of adverse effects (ie, NOAEL; Figure 1D). Each type of response is not observed below its respective threshold, although activation of specific molecular mechanisms resulting in those physiological responses may occur over a full range of doses.
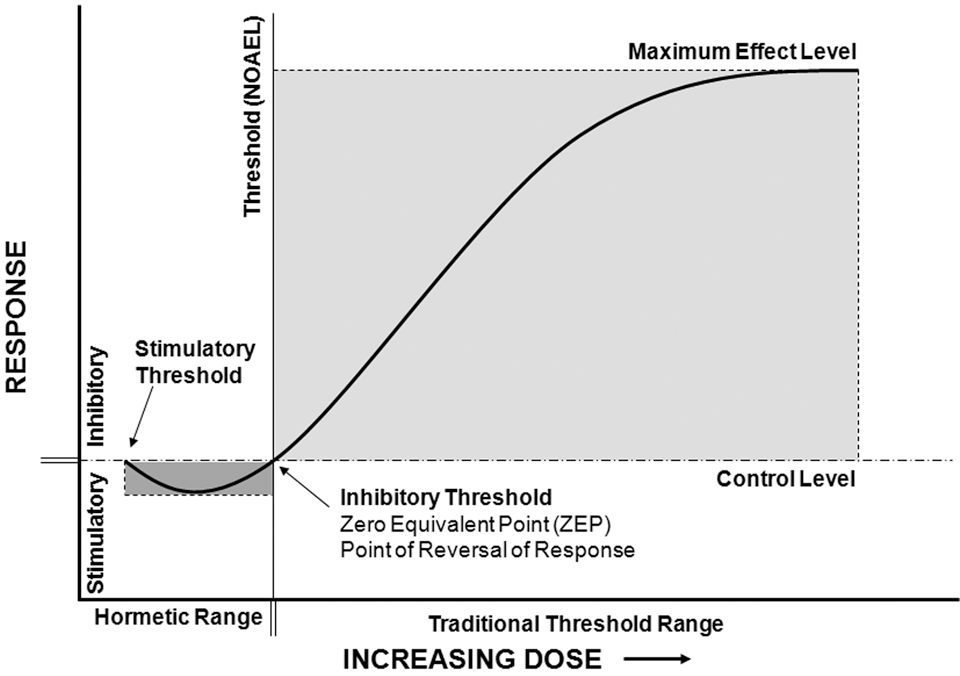
From the quantitative features of hormetic responses (modest amplitude and narrow range of response), the hormetic zone (dark gray) is predicted to be small in comparison to the inhibitory, or traditional, zone (light gray). The stimulatory range is finite and is defined by a lower limit, or stimulatory threshold, and an upper or inhibitory threshold.
It is important to recognize that above the threshold concentration defined by the NOAEL, hormesis describes a dose-response relationship that is identical to that of the threshold model. Thus, hormesis clearly conforms to the principles of threshold response. The perception that there is a discrepancy between the two is the result of overlooking the dual-threshold response; the full response is the result of both the stimulatory threshold and the inhibitory threshold. From this perspective, it becomes clear that hormesis does not deviate from the fundamental principles of the dose-response relationship; rather, it describes a dose-response relationship that includes two different thresholds.
Hormesis as a Threshold Model
The traditional threshold dose-response model is regarded as the central pillar for the science of pharmacology and toxicology, burdening hormesis with a great deal of skepticism. The criticisms relating to the hormetic β-curve and subthreshold responses can be resolved by considering the development and the general understanding of the dose-response relationship as applied to the disciplines of pharmacology and toxicology as detailed above.
Hormesis describes dose-response relationships that are not sigmoidal and do not display saturation of response with increasing dose. Deviations from the sigmoid-shaped curve, however, do not necessarily violate the general understanding of concentration and effect. The biological literature contains many examples of nonmonotonic responses. For example, Belcher and coworkers had carefully characterized the dose-response relationship of the steroid hormone 17β-estradiol (estrogen)–mediated extracellular signal-regulated kinases (ERK1/2) activation in cerebellar granule cells and reported a nonmonotonic response relationship. 28,29 Although initially thought as hormetic, this low-dose, biphasic response cannot be considered a hormetic response because the requisite reversal-of-response feature of hormesis was not detected. Low-dose stimulation of ERK1/2 by estradiol was observed, but high-dose inhibition with respect to baseline control levels was not observed. These studies were neither designed nor analyzed in a way that could detect a high-dose inhibition below control levels; thus, they were limited by design and can only be interpreted as having identified a complex nonmonotonic dose-response relationship.
Similar nonmonotonic responses are also frequently observed in other hormone–mediated processes. These responses are real and reproducible, yet they do not conform to the sigmoid-shaped response curve as predicted by the threshold model. One simple explanation is that nonmonotonic responses involve multifactorial mechanisms that do not adhere to the assumptions used in the mathematical modeling of the threshold model. In the above example, the inverted U-shaped dose dependency of estrogen-induced ERK1/2 activation can result from differential activities and/or different ligand affinities of the 2 ligand interaction sites found in the estrogen receptor. 29 The contribution of 2 ligand binding sites violates the assumption of a one-to-one binding relationship between ligand and receptor. Another contributing factor identified in this specific example is that estradiol simultaneously activates protein phosphatase 2A (PP2A) in cerebellar granule cells via a different signaling pathway. 30 Protein phosphastases inactivate kinases by removing specific phosphate groups. This suggests that in cerebellar granule cells, the modulation of ERK-pathway signaling and the resulting nonmonotonic dose–response relationship for estradiol involves concurrent activation of both phosphorylation and de-phosphorylation mechanisms. As discussed in more detail, examples such as this are not a violation of any aspect of threshold theory but are simply more complex responses that cannot be described with such a simplistic model.
Similarly, hormesis and traditional threshold model only appear different because each describes fundamentally different endpoints. Endpoints that are consistent with the threshold model such as those typically observed in pharmacology are initiated at receptors on the cell surface, and the amplitude of response is directly related to receptor occupancy. Below the threshold concentration, receptor activation is not sufficient to illicit a measurable response. With increasing dose, the response becomes saturated as ligand binding at the receptor is saturated. The mathematical relationship between receptor activation and amplitude of response is straightforward and predicts a sigmoid-shaped response curve. 27 In contrast, hormetic stimulatory responses do not follow the relationship defined by the threshold model. Hormesis differs from the threshold model in that for a given endpoint, it displays two types of responses, stimulatory and inhibitory. This is a unique feature of hormesis that stemmed from the nonsaturable nature of the hormetic stimulatory responses. The reversal of response feature also distinguishes hormesis from other nonmonotonic responses where the response may be reduced from peak stimulatory levels but does not become inhibitory as compared to control.
Our review of studies that contained evidence of hormesis 31 indicated that 62.2% of the measured endpoints were growth responses, with longevity, survival, and reproduction accounting for 16.6% of the endpoints. Metabolic and physiological responses accounted for 15.2% of the remaining endpoints analyzed. Identified responses included metallothionein synthesis, DNA synthesis, RNA synthesis, mitosis, oxygen consumption, altered hepatic foci, photosynthesis rate, tissue regeneration, immune response, stress protein synthesis, and seed germination. 32 Many of these metabolic responses are also involved in growth-related mechanisms. Thus, the purported generalizability of responses demonstrating hormesis is actually limited to endpoints relatable to growth.
Clearly, hormetic physiological responses are not the manifestation of a specific single signaling cascade but require the coordination of multiple cellular processes. As a result, there is no simple mathematical relationship connecting ligand concentration, receptor occupancy, and the observed response. Toxicological endpoints such as lethality often involve multiple mechanisms and may not directly correlate with chemical binding at a specific receptor system, yet toxicological studies frequently display the same sigmoidal dose-response relationship observed in pharmacological studies. In addition to the quantal nature of many toxicological dose-response studies, this sigmoidal dose-response relationship is likely due to fundamental differences between stimulatory and inhibitory responses. The inhibitory effects of chemicals investigated in toxicology can be saturated with increasing dose because at no point beyond the maximal-effect dose would the response be less inhibitory. The subthreshold effects of hormesis are stimulatory responses that do not display saturation with saturation of receptor occupancy. Responses such as growth or longevity do not sustain maximum stimulation with increasing dose. Metabolic endpoints are also frequently regulated by complex negative feedback or substrate inhibition mechanisms. Thus, for these endpoints, a β-curve dose-response relationship is not only acceptable but expected.
Hormesis as a Nonsaturable Stimulatory Response
Hormetic responses are similar to other stimulatory responses that do not display saturation of response. Chemicals that elicit a hormetic dose response, however, are typically defined by their adverse effects. This is evident in discussions of hormesis, which are largely limited to the toxicological literature, in which the low-dose stimulatory responses are minimized. Moreover, the proposed mechanism of hormesis as an adaptive response is based on the idea that hormetic chemicals are disruptors of normal cellular function. 20 Stebbing 24 suggested that hormetic stimulatory responses are indirect consequences of overcompensatory reactions that were activated by the initial disruption to homeostasis. This hypothesis implies that high-dose inhibition represents the primary effect of hormetic chemicals, whereas low-dose stimulation is the effect of an organism’s response to stress.
A number of therapeutic agents, particularly substances that were initially recognized as toxins, can be characterized as causing hormetic-like dose responses. Such agents are rarely considered in the context of hormesis, however, because of the continued focus on chemical exposures, which are primarily considered harmful. Cardiac glycosides serve as prime examples of chemicals used clinically that demonstrate hormetic properties—the cardiac glycosides are extracted from plants of the genus Digitalis, of which the common foxglove (Digitalis purpurea) is most well known. Before the discovery of their therapeutic values by William Withering in 1785, the deadly effects of digitalis extracts had long been recognized. 33 The purified cardiac glycoside, digoxin, is indicated for treatment of various heart conditions such as atrial fibrillation, atrial flutter, and congestive heart failure that is unresponsive to other medications. The dose-response relationship of digoxin is characterized by a narrow therapeutic window (plasma concentration of 1-2.6 nmol/L). Exposure to doses above that range result in numerous systemic adverse effects mediated via inhibition of the sodium potassium ATPase and altered intracellular calcium concentrations. The digoxin dose-response relationship is consistent with a hormetic response, that is, low-dose clinical efficacy and high-dose toxicity.
Some antibiotics provide another example of a class of agents that display dose-response relationships consistent with hormesis but are generally not associated with hormesis because these agents are only considered by their therapeutic effects. In nature, antibiotics are synthesized by bacteria, fungi, and molds to kill or prevent the growth of bacteria. The release of an antibiotic substance by a microorganism produces a concentric concentration gradient. The high concentration of the antibiotic closest to the microorganism has a bactericidal effect and protects that microorganism from surrounding (presumably competing) bacteria in the immediate proximity. As the antibiotic diffuses outward, the concentration decreases. At some distance, the antibiotic concentrations may fall into the growth stimulatory range. Thus, some antibiotics may have dual effects that promote the survival of the antibiotic-producing microorganism by acting to eliminate competing bacteria that are within close proximity and by encouraging survival of bacterial populations away from local resources. Antibiotic-induced stimulatory responses are reproducible and have been suggested to be an endogenous function of antibiotics. 34
Antibiotics represent a broad class of compounds that use multiple mechanisms to produce the shared feature of bactericidal or bacteriastatic actions. 35 With such diversity in the mechanisms of action, not all antibiotics are expected to demonstrate a low-dose stimulatory effect. However, such low-dose effects are well documented for penicillins. Following 16 hours of exposure, the dose-response relationship of penicillin’s actions on Staphylococcus growth exhibited the β-curve response shape, a classic feature of hormesis. 36 This observation tends to be underappreciated because it runs counter to clinical/therapeutic function of penicillins. The unfortunate clinical approach adopted in response to the limited supply of penicillin in England during World War II further demonstrates this interrelationship between bacterial growth and therapeutic effectiveness. Penicillin was prescribed below normal therapeutic doses as an attempt to meet the needs of the large number of patients. This strategy of dealing with the short supply inadvertently exacerbated Staphylococcus infections in many patients, an effect distinct from development of resistance. 36
In pathogenic bacteria, antibiotics (eg, penicillins) induce multiple nonlethal effects at subinhibitory concentrations that differ from the inhibitory effects observed at higher concentrations. Early observations of subinhibitory effects included morphological changes at the cell surface and induction of transcriptional and translational activities unrelated to DNA repair mechanisms. 34,35 Features of hormetic responses are also present in reports of altered transcriptional activity in response to exposure to some classes of antibiotics (ie, low-dose stimulation and high-dose inhibition) and growth stimulation and bactericidal effects at low and high concentrations of penicillins, respectively. The dosage that marks the point of response-reversal is related to the minimal inhibitory concentration (MIC; Figure 3 ), which is the lowest concentration of an antibiotic that will inhibit the bacterial growth. In fact, the stimulatory doses are often reported as fractions or percentages of the MIC values.
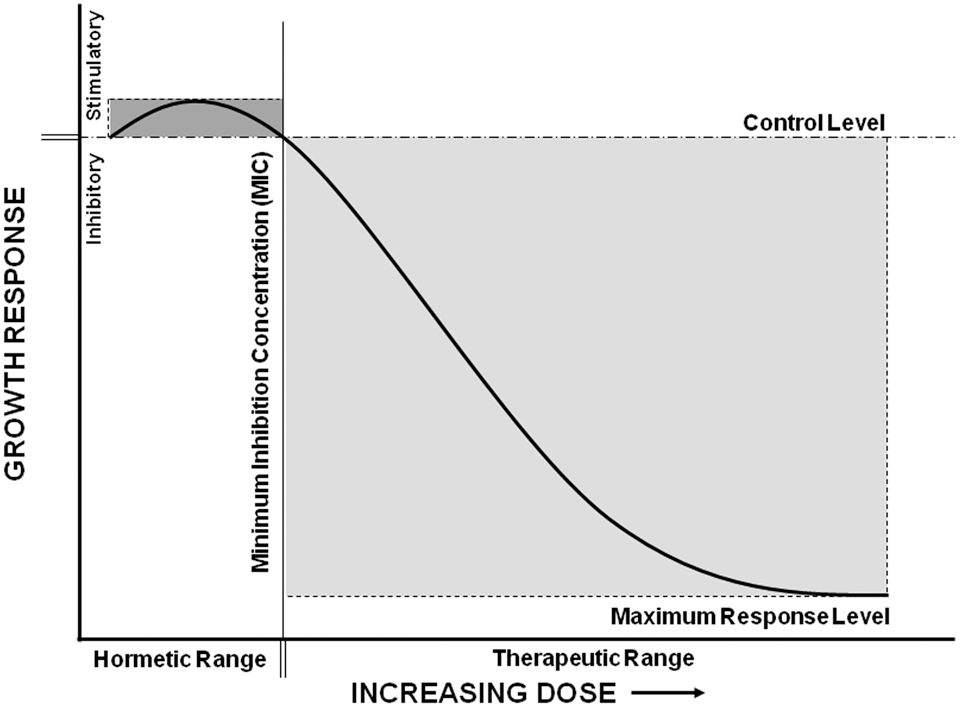
Some antibiotics may display a stimulatory growth response at low dose that is consistent with hormesis. There is a single stimulatory peak (dark gray) immediately below the inhibitory threshold (minimal inhibitory concentration, MIC) and an inhibitory zone (light gray) above the threshold. The therapeutic effectiveness of the antibiotic as observed for the host is inversely related to the bacterial growth response. Doses below the inhibitory threshold may reduce the therapeutic response below control level (ie, subinhibitory doses may enhance infection).
The dose-response relationships of antibiotics can be considered from the perspective of the host or that of the pathogen. From either perspective, examples of hormetic-like responses abound in peer-reviewed literature. The clinical interrelationship between host and pathogen predicts that the dose-response relationship measuring therapeutic effectiveness is inversely related to that of the bacteria growth response. Concentrations that inhibit bacterial growth would produce an increase in therapeutic effectiveness, and conversely, concentrations that stimulate bacterial growth would result in a reduction of therapeutic effectiveness (Figure 3). Below the inhibitory threshold is a nonsaturable zone where infections are exacerbated. This nonsaturable zone corresponds to the nonsaturable growth zone seen in hormesis. Above the inhibitory threshold, infection is suppressed, and this response can be saturated with increasing dose. Doses above a therapeutic window of antibiotics are not used clinically because of associated side effects, not because of ineffectiveness in the treatment of infections. Doses below a therapeutic window are not used because they are ineffective in reducing infections. Such subinhibitory doses, however, are not generally recognized for their potential to enhance infections even though the evidence is clear that low-dose stimulatory effects are a normal function of many antibiotics.
The Shapes of Dose-Response Curves
In evaluating different types of pharmacological and toxicological dose-response relationships, Townsend and Luckey 37 identified 4 types of curves. The 2 most common types were the α-curve and the β-curve (Figures 1C,D). The α-curve represents the classical understanding of a toxic concentration-response relationship and is consistent with the threshold model such that toxicity is reduced as the dose decreases with no response observed below the threshold dose.
By contrast, the β-curve exhibits a single stimulatory peak followed by a progressively inhibitory response as the dose increases beyond the inhibitory threshold dose (Figure 1D). The β-type curve is observed for many responses reported to demonstrate hormetic-like properties. Indeed, the β-curve represents the graphical presentation of various descriptive definitions of hormesis, such as reversal of response, biphasic effects, or opposing low-dose and high-dose effects. Significantly, the β–curve also differs from the α-curve in that it represents 2 thresholds, the inhibitory threshold seen in the α-curve and a stimulatory threshold that is absent from the α–curve (Figure 1D). The hormetic dose-response curve may also be described as inverted U-shaped or J-shaped depending on the endpoints measured. Inverted U-shaped curves depict low-dose stimulation and high-dose inhibition characteristic of growth and longevity, whereas J-shaped curves describe low-dose reduction and high-dose enhancement of adverse effects such as disease incidence (Figure 4).
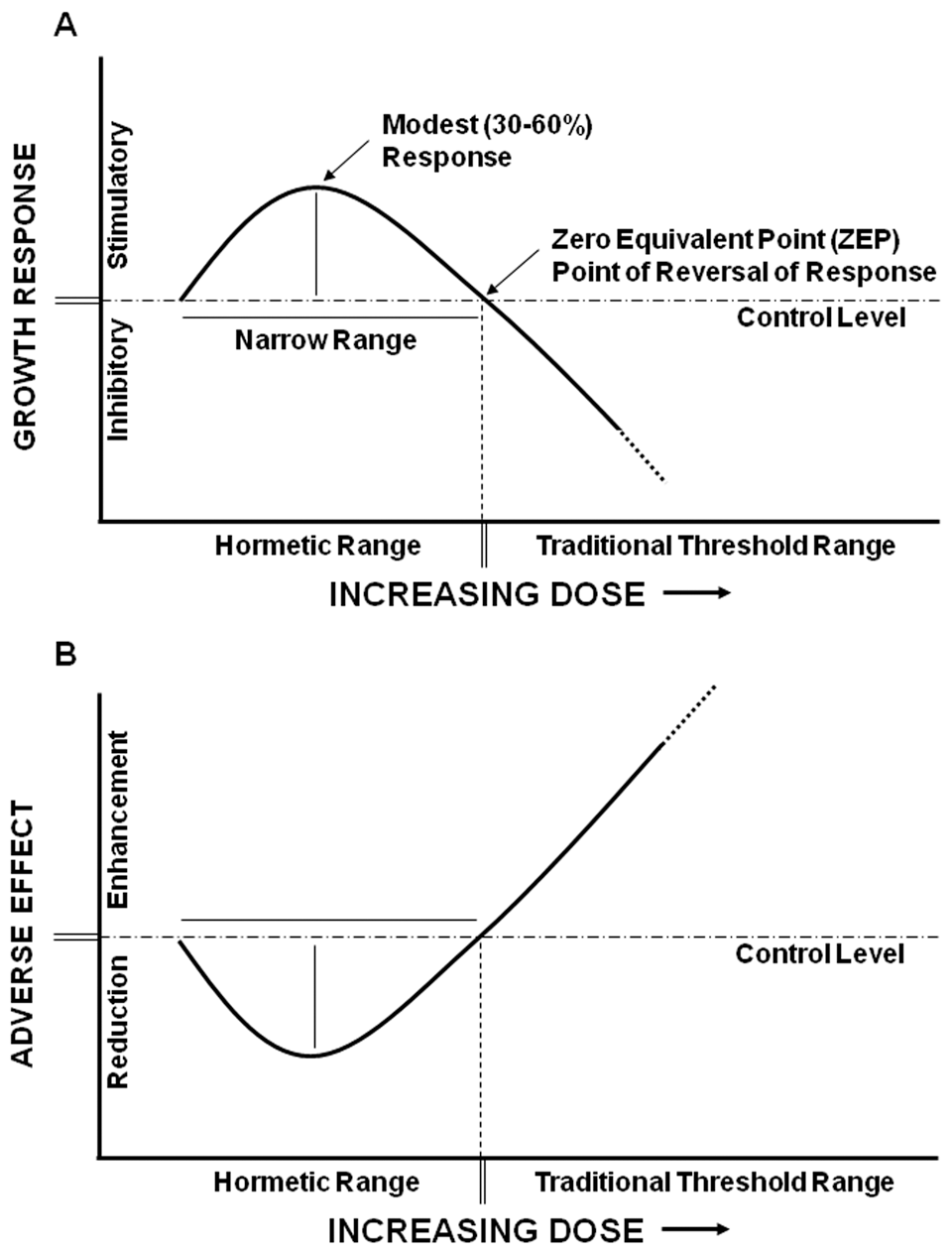
The hormetic dose-response curve is also commonly described as inverted U-shaped or J-shaped. (A) The U-shaped curve describes low-dose stimulatory and high-dose inhibitory responses like growth and longevity. Quantitative assessment of the hormetic response revealed that the stimulatory response is modest (30%-60% increase from control) and the stimulatory range is typically less than 10-fold below the inhibitory threshold. (B) The J-shaped curve represents low-dose reduction and high-dose enhancement of adverse effects such as disease incidence.
The Identifying Characteristics of Hormetic Responses
From an extensive database search assessment of the toxicological literature, Calabrese and coauthors 20,32,38 provided a quantitative characterization of hormetic dose responses. The authors characterized 2 features of the hormetic response curve: (1) the amplitude of stimulatory response and (2) the width of stimulatory dose range. Their analyses revealed that the maximal stimulation of the hormetic response was most typically a percentile increase, ranging 30% to 60% over control, with some exceptions producing greater than 2-fold stimulation. The range of stimulatory dose responses is within a 5- to 100-fold dose range for 90% of studies examined. However, the majority are 5- to 10-fold below the point where the response curve crosses the reference level of response, or the zero equivalent point (ZEP). The ZEP is also the point of response-reversal, the inhibitory threshold, and the NOAEL. Stimulatory dose ranges were only rarely reported to be greater than 1000-fold below the ZEP. From their analysis, it was clear that hormetic responses were modest, reproducible, and often statistically significant; however, specific experimental considerations are required to reliably discerned responses from random variation. 2 Critics have raised some valid objections concerning the methodologies used to characterize hormetic dose responses. Specific criticism was leveled at the inclusion criteria used for dose-response analysis and for determination of statistical significance of analyzed data. 6,21,39,40 Despite such criticisms, these studies provide a potentially useful framework of quantitative features associated with hormetic responses.
Considering the quantitative features of defined response amplitude (30%-60%) and concentration range (5- to 10-fold below ZEP) as guidelines, the definition of hormesis proposed here establishes consistent and quantitative experimental guidelines for identification of most hormetic dose responses. Furthermore, the application of these guidelines makes assessment of the toxicological impact of hormetic mechanisms economically feasible for general toxicological evaluation, as they identify a common low-dose range for targeting hormetic dose responses. Routine assessment of hormetic effects is possible by including sensitive low-dose measures within the typical hormetic range of many chemicals/inducers after a NOAEL has been established by conventional assessment approaches. These guidelines may prove effective for identification of many hormetic responses. However, it is important to stress that the above criteria should not be considered definitive limits for detection or definition of hormetic responses, nor should they be used to define all nonmonotonic responses.
As described above, the quantitative features of hormetic dose responses have clearly hindered detection. Study design is a major limiting factor in the detection of hormetic responses, as the likelihood of observing hormesis is directly related to the number of subthreshold doses used. 24,32 To further the detection and understanding of hormetic responses, investigators must acknowledge the potential existence of low-dose hormetic responses and design their studies accordingly. The detection of hormetic responses requires inclusion of a low-dose bias in study design to increase the probability of detecting these more modest responses, a practice not commonly used in the determination of response to a toxicant because of the associated increase in cost.
Concluding Remarks
The existence of hormesis is not dependant on understanding the molecular mechanism(s) responsible for the observed effects. As such, hormesis is defined here as a reversal of single response between low and high doses of chemicals, biological molecules, physical stressors, or other initiators of a response. Hormetic responses are conceptually similar to other nonsaturable stimulatory responses, the shape of which is graphically represented best by a β-curve with a stimulatory zone within a defined and finite range below the inhibitory threshold.
Hormesis is analogous to the concepts of physiological range or therapeutic window for an endogenous agent or a pharmacological drug, respectively. Several factors have contributed to the failure to recognize this similarity. First, agents that were reported to display hormesis are mostly chemicals investigated in regard to toxicity and are best known for their adverse effects. 32,38 The bias inherent in associating a substance with toxicological endpoints makes it appear counterintuitive to expect stimulatory (opposing) responses from such agents. The focus of toxicological studies on the inhibitory dose-response relationship is justified because the inhibitory response is the more predominant, if not the only, response for most chemicals displaying toxic effects. As a result, the potential for low-dose stimulatory responses is often left uninvestigated.
Second, chemicals that induce a hormetic dose response may not be recognized as such because of the inherent difficulties in detecting the modest stimulatory response. Hormetic stimulatory responses are limited both in amplitude and dose range. This makes it particularly difficult to detect these responses with normal experimental practices, which generally lack the power to detect such modest variations from control, particularly at subthreshold doses. For example, based on preliminary range-finding studies, the characterization of the inhibitory dose-response curve may stop at a dose where the response had been reduced to control level (ie, the inhibitory threshold), and the hormetic stimulatory response was not observed because doses below the inhibitory threshold were not investigated. A second scenario may be that the 2 lowest doses selected had spanned the stimulatory dose range, and as a result, the stimulatory response was inadvertently not observed. Furthermore, without a detailed characterization of the dose-response relationship at the low-dose range, a dose that produced a response that is the same as control cannot be distinguished as either an inhibitory threshold dose or a substimulatory threshold dose. Only if the stimulatory range is known could the distinction between the stimulatory and inhibitory thresholds be made.
Third, only a subset of chemicals can induce low-dose stimulatory responses consistent with hormesis. The ambiguous language in describing the generalizability of hormesis was often construed to mean hormesis is a universal phenomenon. This is not a reasonable assumption. For example, growth hormesis was reported for a broad range of chemicals with no apparent shared features but is not expected for all chemicals. The diversity of substances across multiple chemical classes suggests that growth hormesis is mediated by a variety of compound-specific cellular mechanisms. This flexibility is not an exception applicable only to hormesis but is a general observation for all growth responses. All biological systems contain multiple pathways that are specifically regulated to induce the common endpoint of growth. These multitudes of pathways allow biological systems to respond to a broader range of stimuli, which confers obvious evolutionary advantages.
Footnotes
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors disclosed receipt of the following financial support for research and/or authorship of this article: This work was supported by NIH/NIEHS grants RO1 ES015145, RO1 ES017263 and RC2 ES018765.