Abstract
Trastuzumab is indicated for the treatment of patients with breast cancer overexpressing human epidermal growth factor 2 (HER2). Women with HER2-positive tumors have an increased risk of brain metastases. The blood-brain barrier and blood-cerebrospinal fluid (CSF) barrier may prevent trastuzumab from reaching appropriate concentrations in the brain and CSF following standard intravenous administration. To evaluate the potential of effects on the central nervous system, a 4-week toxicology study with weekly intrathecal administration of trastuzumab was performed in cynomolgus monkeys at doses of 0, 3, or 15 mg. No trastuzumab-related effects on body weight, clinical signs, neurological function, clinical pathology, or anatomic pathology were noted. The applied doses and CSF concentrations achieved in the current study exceeded those reported in patients after intrathecal administration. The results support future studies for further evaluation of intrathecal application of trastuzumab in patients with brain metastases in HER2-positive breast cancer.
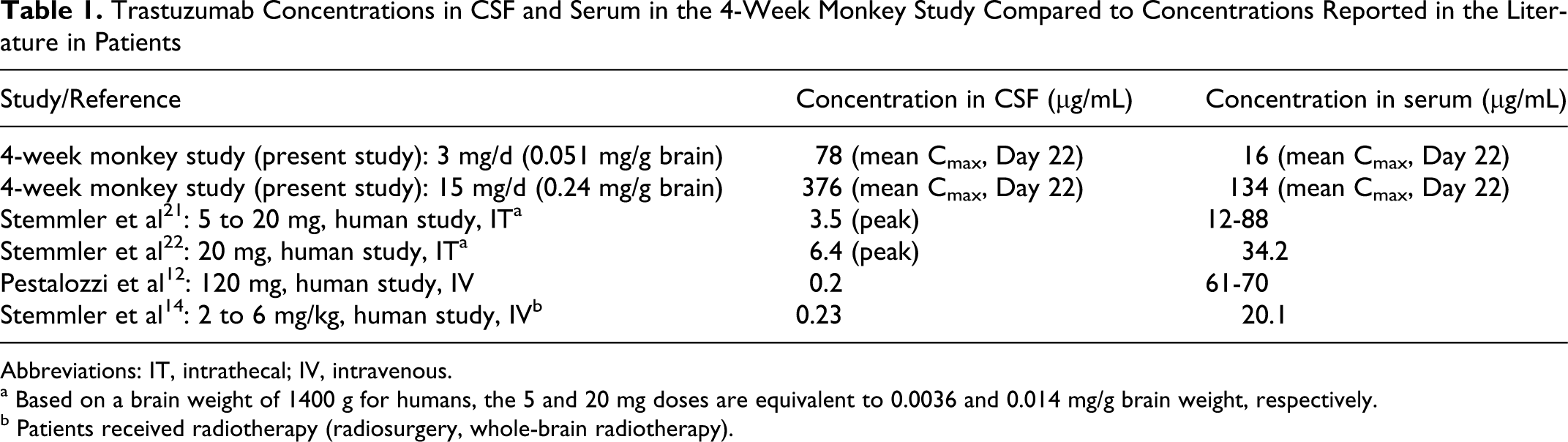
Trastuzumab (Herceptin;Genentech, South San Francisco, CA) is a humanized monoclonal antibody that selectively targets the extracellular domain of the human epidermal growth factor receptor 2 protein (HER2) and is known to extend survival across all stages of HER2-positive breast cancer. An increased rate of brain metastases (mCNS disease) has been observed in HER2-positive breast cancer patients. 1,2 Several studies have also documented a relatively high incidence of mCNS disease in patients with HER2-positive metastatic breast cancer (MBC) following treatment with trastuzumab. 3 –8 Evidence suggests that this apparent increase in the prevalence of mCNS disease is a consequence of trastuzumab prolonging survival. 9 The blood-brain barrier (BBB) and blood- cerebrospinal fluid (CSF) barrier (BCSFB), however, may prevent trastuzumab from reaching adequate concentrations in the brain and CSF following standard intravenous administration. 10 Indeed, the concentration of trastuzumab in CSF after intravenous administration in humans was low (~0.2 µg/mL) (Table 1 ). Increasing the permeability of the BBB and BCSFB by certain disease conditions such as meningitis or by having patients undergo whole-brain radiotherapy (WBRT) for the treatment of malignant brain tumors has shown an increased passage of trastuzumab across the BBB. However, these concentrations of trastuzumab compared to serum levels achieved after intravenous administration were still low. 11–14 As such, local delivery has been reported in which trastuzumab was intrathecally administered, directly or via an intrathecal delivery system such as an Ommaya reservoir, for the treatment of metastatic brain cancer in humans. 15–22
Trastuzumab Concentrations in CSF and Serum in the 4-Week Monkey Study Compared to Concentrations Reported in the Literature in Patients
Abbreviations: IT, intrathecal; IV, intravenous.
a Based on a brain weight of 1400 g for humans, the 5 and 20 mg doses are equivalent to 0.0036 and 0.014 mg/g brain weight, respectively.
b Patients received radiotherapy (radiosurgery, whole-brain radiotherapy).
In preclinical rat efficacy models, HER2-targeted monoclonal antibodies have been administered effectively by intraventricular or intracerebral administration by continuous microinfusion, resulting in tumor regression. 23,24 In addition, local delivery of the monoclonal antibody to the brain was well tolerated in these rat efficacy studies. However, the rat is not a trastuzumab pharmacologically relevant (cross-reactive) species. In addition, a slow infusion rate was used in these studies, which resulted in lower CSF concentrations when compared to patients described in the literature. In the rat studies, Grossi et al 24 administered a total dose of 4 mg over a 7-day period, and Bergman et al 23 used a 60-μg daily dose during a 4-week period. To date, no extensive preclinical safety evaluations have been performed via the intrathecal route after repeat dosing in a trastuzumab cross-reactive animal species using doses equivalent to or higher than those reported for patients (up to 100 mg per dose 18 ). In multiple-dose studies of up to 6 months' duration using weekly intravenous administration in monkeys, there was no evidence of trastuzumab-related toxicity. 25
The intrathecal route (a method to ensure cerebrospinal drug delivery) has been described in a preclinical safety evaluation in cynomolgus monkeys with rituximab, a monoclonal antibody to treat non-Hodgkin’s lymphoma, chronic lymphocytic leukemia, and rheumatoid arthritis. 26 In addition, the intrathecal route has been explored in standard toxicology studies using nonhuman primates in support of this clinical route. 27,28 It appeared that a relatively high dose and injection volume could be used in cynomolgus monkeys.
The present 4-week toxicology study in cynomolgus monkeys was initiated to elucidate the potential for CNS effects in a trastuzumab cross-reactive species and included measurements of trastuzumab in the CSF and serum. The highest dose used in the 4-week monkey study with trastuzumab (15 mg, or 0.242 mg/g brain weight) was approximately 3.4-fold higher than what has recently been used in the clinic. The low dose used in the 4-week monkey study with trastuzumab (3 mg) was equivalent to 0.051 mg/g brain weight. In the literature, doses up to 100 mg of trastuzumab have been reported in humans, equivalent to 0.071 mg/g brain weight based on a brain weight of 1400 g. 18,29 As recommended in the literature for clinical use by the intrathecal route, 30 the preservative-free marketed formulation (ie, a formulation without benzyl alcohol) was used in the present preclinical study.
Materials and Methods
Animals
Male and female cynomolgus monkeys (Chinese origin) were obtained from Primus Bio-Resources (Laval, Québec, Canada). At the start of dosing, the animals were approximately 2 to 3.5 years of age and ranged in weight from 2.1 to 3.6 kg. Monkeys were acclimated for 7 weeks before dosing. During the pretreatment and dosing periods, the monkeys were single-housed in stainless steel, mesh floor cages equipped with an automatic watering valve. During the 4-week recovery period, animals were socially housed with an individual of the same group and sex. The animal room was environmentally controlled to maintain a temperature of 24°C ± 3°C, a humidity of 50% ± 20%, and a 12-hour light cycle. Certified primate diet (2050C, Harlan Teklad) was provided twice daily, along with a variety of certified treats and/or fresh fruit. In addition, Prima-Foraging Crumbles, as part of the environmental enrichment program, was offered at least weekly. Water was provided ad libitum.
Experimental Design
Doses for the 4-week study were selected based on the results of an escalating-dose study (non-Good Laboratory Practice [GLP]). In this escalating-dose study, 3 male monkeys received weekly 30-minute intrathecal infusions at ascending doses of 1.5, 5, 10, and 15 mg at an infusion rate of 1.5 mL/hour. The high dose of 15 mg was selected, as it was considered the maximum feasible dose while still using an isotonic dosing formulation. All doses were well tolerated.
The 4-week study was conducted in compliance with the United States Food and Drug Administration GLP Regulations (21 CFR 58). In addition, this study was conducted in compliance with the guidelines and principles listed in the American College of Toxicology Policy Statement on the Use of Animals in Toxicology. Trastuzumab (marketed formulation) was provided by Roche Canada. Trastuzumab lyophilized powder was reconstituted with sterile water for injection USP and then further diluted with 0.9% sodium chloride for injection to the appropriate concentration of the day of dosing. Therefore, it did not contain any preservatives. The purity was 100%. Five male and 5 female monkeys were evaluated per dose group in this study, of which 2 monkeys/sex/group were evaluated after a 4-week recovery period. Animals were assigned to 3 groups that received 4 weekly 30-minute intrathecal infusions at dose levels of 0, 3, or 15 mg/dose at an infusion rate of 1.5 mL/hour, via an in-line 0.2 µm polyvinylidene fluoride filter on study days 1, 8, 15, and 22. At least 10 days were allowed between the surgical implantation of the catheter and treatment initiation to allow for adequate recovery of the surgery. Animals in the control group were dosed with 0.9% sodium chloride for 30 minutes at an infusion rate of 1.5 mL/hour.
Method of Dose Administration and CSF Sampling
At least 10 days before treatment initiation, surgery was performed to implant a silastic catheter at L5 into the intrathecal space, and the tip of the catheter was between L2 and L4 for continuous infusion. Another silastic catheter was placed in the cisterna magna and connected to a subcutaneous access port to permit CSF sampling. Catheter placements in the intrathecal space were confirmed by fluoroscopy during the surgical procedure and prior to treatment initiation. In the latter case, a contrasting agent (0.5-1 mL of Omnipaque 240) was injected into the catheter, and lateral and dorsal views of the catheter were taken.
For maintenance of continuous infusion into the intrathecal space, a jacket was placed on the animals to hold the tether system. The lumbar port catheter was continuously infused with 0.9% sodium chloride injection, USP, via an in-line 0.2 µm polyvinylidene fluoride filter at a rate of 0.1 mL/hour until initiation of treatment and between dosing occasions. Following the end of the last infusion, the catheters were tied off, cut, and allowed to slip subcutaneously.
Toxicokinetics
Blood samples (approximately 1 mL) were collected by venipuncture from all animals at 0 (predose), 6, 24, 96, and 168 hours post-dose (prior to next dose) on days 1 and 22. Blood samples were allowed to clot at room temperature for at least 20 minutes. The serum was obtained by centrifugation at approximately 2700 rpm for 10 minutes in a refrigerated centrifuge (approximately 4°C) and transferred to a freezer at approximately –80°C. In addition, on days 1 and 22, CSF samples (300-500 µL) were collected from the cervical port placed in the animals at 6, 24, 96, and 168 hours post-dose. All samples were stored at approximately –80°C. Serum and CSF samples were analyzed using a validated sandwich ELISA method with a sensitivity of 20 ng/mL and a range of 20-2000 ng/mL. Briefly, plates were coated with recombinant p185 hER2, followed by blocking, washing, and then incubating with 1/25 diluted calibrators, quality controls, and samples. A horseradish peroxidase (HRP)-conjugated goat anti-human IgG was used to detect the immobilized trastuzumab. After appropriate incubation and plate wash, bound HRP conjugate was detected with a tetramethyl benzidine substrate, which was read colorimetrically on a plate reader. Noncompartmental toxicokinetic analysis was performed on the serum and CSF concentration data. Tmax (time to achieve maximal serum or CSF concentration), Cmax (maximal/peak serum or CSF concentration), AUC (area under the serum or CSF concentration–time curve), CSF clearance (CL), and volume of distribution at steady-state (Vss) were determined to characterize drug exposure. Cmax and Tmax were taken directly from the serum or CSF concentration–time profiles. The reported pharmacokinetic parameters were calculated by using WinNonlin, version 5.2.1 (2008; Pharsight Corporation, Mountain View, CA, USA).
Clinical and In-Life Observations
Clinical signs and food evaluation were monitored daily, and body weights were measured weekly.
Neurological Examinations
Neurological examinations were performed twice prior to initiation of treatment (before and following surgery), toward the end of weeks 1 and 4 of dosing, and toward the end of the recovery period. The neurological examinations included assessment of general attitude and behavior, postural reactions (assessments of proprioceptive positioning, and visual and tactile placing reactions), cranial nerve function (assessments of head movement/symmetry, head muscle tone, eye reactions, eye symmetry, vestibular nystagmus, eye position, corneal reflex, pupillary light reflex, and nasal septum), spinal nerve function (assessments of muscle tone, flexor reflex, and perineal reflex), and gait.
Clinical Pathology
Clinical pathology evaluations were conducted once prior to treatment, during week 4 of treatment, and toward the end of the recovery period. Food was removed overnight prior to blood sampling for clinical pathology evaluations. Clinical pathology evaluations included a full battery of hematology (blood cell morphology, erythrocyte indices, hematocrit, hemoglobin, mean platelet volume, platelet count, red blood cell count, reticulocytes, and white blood cell count [including differential]), coagulation (activated partial thromboplastin time and prothrombin time), clinical biochemistry (albumin/globulin ratio, alanine aminotransferase, albumin, alkaline phosphatase, aspartate aminotransferase, blood urea nitrogen, calcium, chloride, cholesterol, creatinine, globulin, glucose, inorganic phosphorus, potassium, sodium, bilirubin [total, direct, and indirect], total protein, and triglycerides), and urinalysis (bilirubin, blood, color, appearance, glucose, ketones, microscopy of centrifuged deposit, nitrite, pH, protein, specific gravity, urobilinogen, and volume). Blood samples were collected from a femoral vein. No clinical pathology evaluations were performed on the CSF. Urine was collected overnight at room temperature.
Anatomic Pathology
Animals were euthanized on completion of the treatment (7 days after the last dose) and recovery periods and underwent exsanguination by incision of the axilliary or femoral arteries following anesthesia by intravenous injection of sodium pentobarbital. All animals were fasted overnight before necropsy. The terminal body weight was recorded prior to necropsy. The necropsy consisted of an external examination, including identification of all clinically recorded lesions, as well as a detailed internal examination and confirmation of the integrity of the lumbar catheter system by injection of ink through the catheter in order to visualize the catheter track. The brain weight was recorded. On completion of the necropsy of each animal, the brain including meninges (with dura mater, arachnoid, and pia mater), eyes, infusion site including meninges, optic nerves, sciatic nerve, and the spinal cord including meninges (cervical, thoracic, and lumbar, with emphasis on L2-L6 [lumbar catheter position]) were retained for histopathological processing. The brain, with inclusion of meninges, was evaluated at 5 levels (caudate putamen, frontal cerebral cortex, piriform cortex, thalamus and hypothalamus, hippocampus, midbrain, cerebellum, and medulla oblongata), and these sections were processed for histopathological examination and stained with hematoxylin and eosin, and with Kluver–Barrera stain for myelin. The infusion site was evaluated at 4 levels (1 section at entry of the catheter, 1 section at the catheter tip, 1 section 1 cm cranial to the catheter tip, and 1 section 1 cm caudal to the catheter tip), and these sections were processed for histopathological examination and were also stained for myelin with Kluver–Barrera stain and hematoxylin and eosin. Spinal cord tissues were also stained for myelin with Kluver–Barrera stain and hematoxylin and eosin. The eyes and optic nerves were fixed in Davidson’s fluid. Histopathological examination was performed on the brain, infusion sites, and spinal cord from all dosed animals. A peer review of pathology findings was performed.
Statistical Analysis
Numerical data obtained during the conduct of the study were subjected to calculation of group mean values and standard deviations for each sex. For each parameter of interest, group variances were compared using Levene’s test at the .05 significance level. When differences between group variances were not found to be significant, a parametric 1–way analysis of variance (ANOVA) was performed. For each pairwise group comparison of interest, significance was reported at the .05, .01, and .001 levels. Due to the small group size, numerical data collected during the recovery period were subjected to calculation of group means. No statistical analyses were performed on the toxicokinetic analyses.
Results
Clinical and In-Life Observations
There were no unscheduled deaths during this study. During the course of the study, there were no clinical signs related to test article administration (data not shown). Clinical signs on or around the access port used for CSF collection included red skin, swelling in the thoracic/cervical region, and scabs in all study groups. However, it was considered that these clinical signs were related to the experimental procedures and not to the test article. There were no test article-related changes in body weight or body weight gain during the dosing and recovery periods. In addition, no test article-related changes in appetite were noted during the dosing and recovery periods.
Neurological Examinations
There were no test article–related changes in neurological examinations during the dosing and recovery periods (data not shown).
Toxicokinetics
Because of occasional technical difficulties encountered in collecting CSF samples through access ports, some samples were not collected at all the prescribed time points. In general, CSF samples were successfully collected from 3 animals/sex/group at all time points, except for the females at a dose level of 3 mg on study day 22. On this day, CSF samples could be collected only from 1 female in this group. Following intrathecal infusion, trastuzumab passed across the BBB into the blood. A sharp reduction in CSF concentration of trastuzumab was observed, whereas a stable serum concentration was generally reached after 24 hours post-infusion (Figure 1 ). Table 2 summarizes the mean toxicokinetic data in the 4-week toxicology study. Intrathecal infusion of trastuzumab in cynomolgus monkeys resulted in high variability in the CSF exposure between animals and within an individual animal. This high variability might be the reason of the sex difference observed in the CSF exposure concentrations of trastuzumab, namely, that a higher Cmax and AUCt value was noted in females on study day 1 in the 15-mg dose group when compared to males. However, this sex difference might be a result of the high variability in CSF exposure on that day, since no sex difference was noted on study day 22 or at the lowest dose. Results for serum sample exposures were less variable. Accumulation of trastuzumab in the CSF was not observed, however, it was 1.8- to 3.4-fold higher in the serum in the 3- and 15-mg dose groups, respectively, after 4 intrathecal administrations as compared to the mean values of Cmax and AUCt on study day 1. Compared to the 3-mg dose group, the mean AUCt and Cmax values in the CSF increased less than dose proportionally at a dose level of 15 mg/dose on study day 1, but dose proportionally on study day 22. The mean AUCt and Cmax values in the serum increased proportionally on study day 1, but greater than proportionally on study day 22 at a dose level of 15 mg/dose.
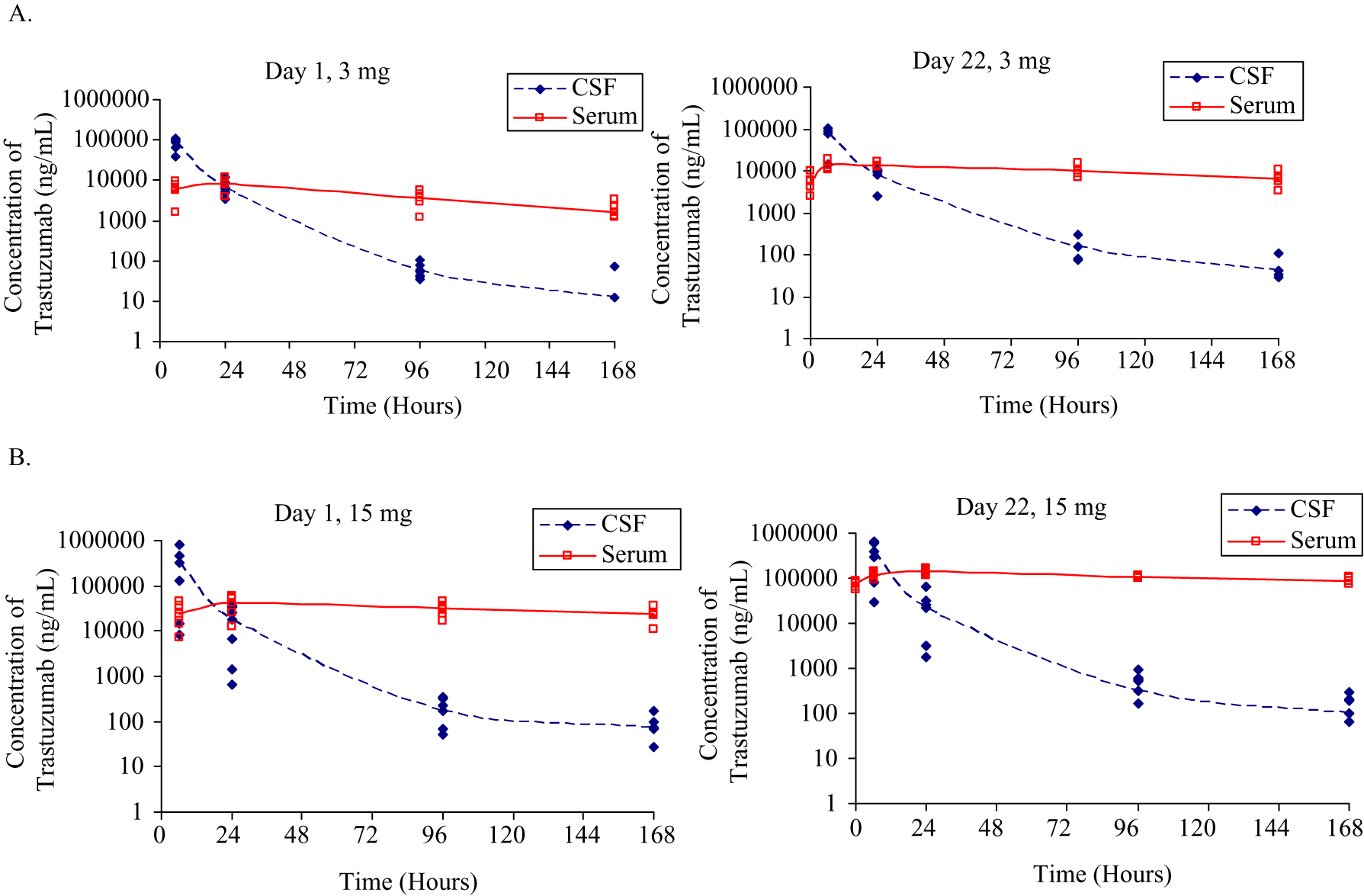
Individual and mean trastuzumab concentrations in the cerebrospinal fluid and serum in cynomolgus monkeys after intrathecal dosing at 3 mg per dose (A) or 15 mg per dose (B).
Mean Toxicokinetic Parameters in CSF and Serum Following Weekly Intrathecal Infusion of 3 or 15 mg Trastuzumab in Cynomologus Monkeys
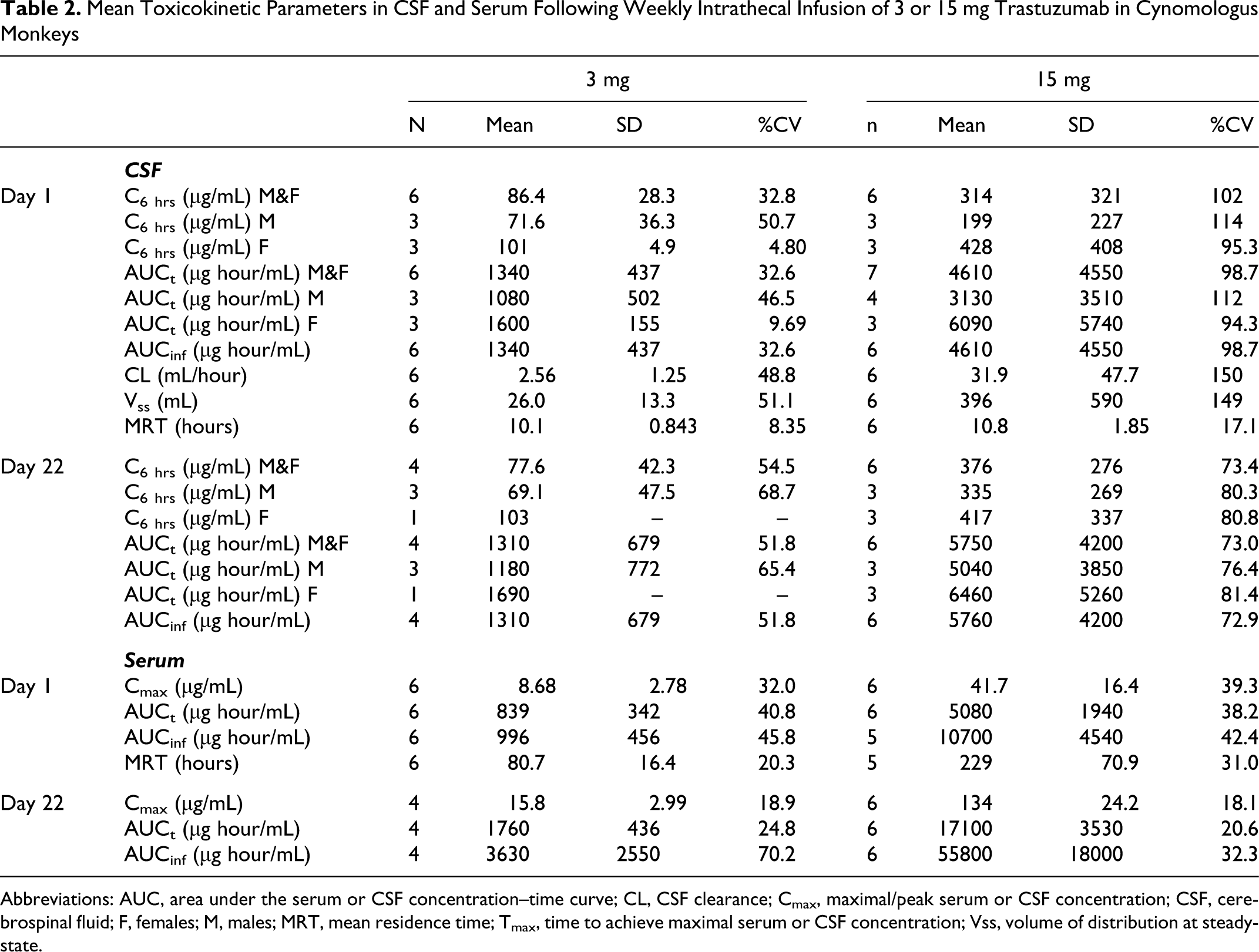
Abbreviations: AUC, area under the serum or CSF concentration–time curve; CL, CSF clearance; Cmax, maximal/peak serum or CSF concentration; CSF, cerebrospinal fluid; F, females; M, males; MRT, mean residence time; Tmax, time to achieve maximal serum or CSF concentration; Vss, volume of distribution at steady-state.
Clinical Pathology
There were no changes in hematology, coagulation, clinical chemistry, or urinalysis parameters associated with the administration of trastuzumab at doses of 3 or 15 mg/dose during the treatment period or at the end of the recovery period (data not shown).
Anatomic Pathology
During the dosing and recovery periods, there were no treatment–related effects on the brain weight of monkeys given trastuzumab. Based on the mean brain weight at the end of the treatment period in the 3- and 15-mg dose groups of 58.98 (g) and 62.00 (g), respectively, the mg administered trastuzumab/g brain weight was equivalent to 0.051 and 0.24. There were no macroscopic findings considered to be related to the administration of trastuzumab during the dosing and recovery periods. The gross findings observed in control and treated monkeys were considered incidental or related to the experimental procedures (eg, thickening of surgical site) or to sexual immaturity (ie, small epididymides, prostate, seminal vesicles, and testes). Infusion site integrity (ie, ink dissipating in the spinal canal following injection at the time of necropsy) was considered adequate in all animals.
A summary of the microscopic findings is given in Tables 3a and 3b. There were no microscopic findings interpreted to be related to the administration of trastuzumab. Changes attributed to the experimental procedures (ie, lumbar port catheterization and cisterna magna catheterization) were observed at the infusion site in animals from all groups including controls. These changes included minimal to marked chronic inflammation of the meninges (due to the presence of the catheter in the lumbar intrathecal space), often surrounding the catheter site. Other changes at the infusion site (eg, meningeal granuloma or mononuclear cell infiltration, degeneration of nerve fiber), in the spinal cord (eg, meningeal mononuclear cell infiltration or granuloma), and in the brain (eg, meningeal mononuclear cell infiltration or granuloma, degeneration), reported in both control and treated animals, were most likely secondary to alterations (chronic inflammation of the meninges) observed at the intrathecal infusion site. Inflammation of the meninges seen in the medulla oblongata area of some control and treated animals was regarded as secondary to the cisterna magna catheterization.
Incidences of Histopathological Lesions in Monkeys After 4 Weekly Intrathecal Doses of Trastuzumab
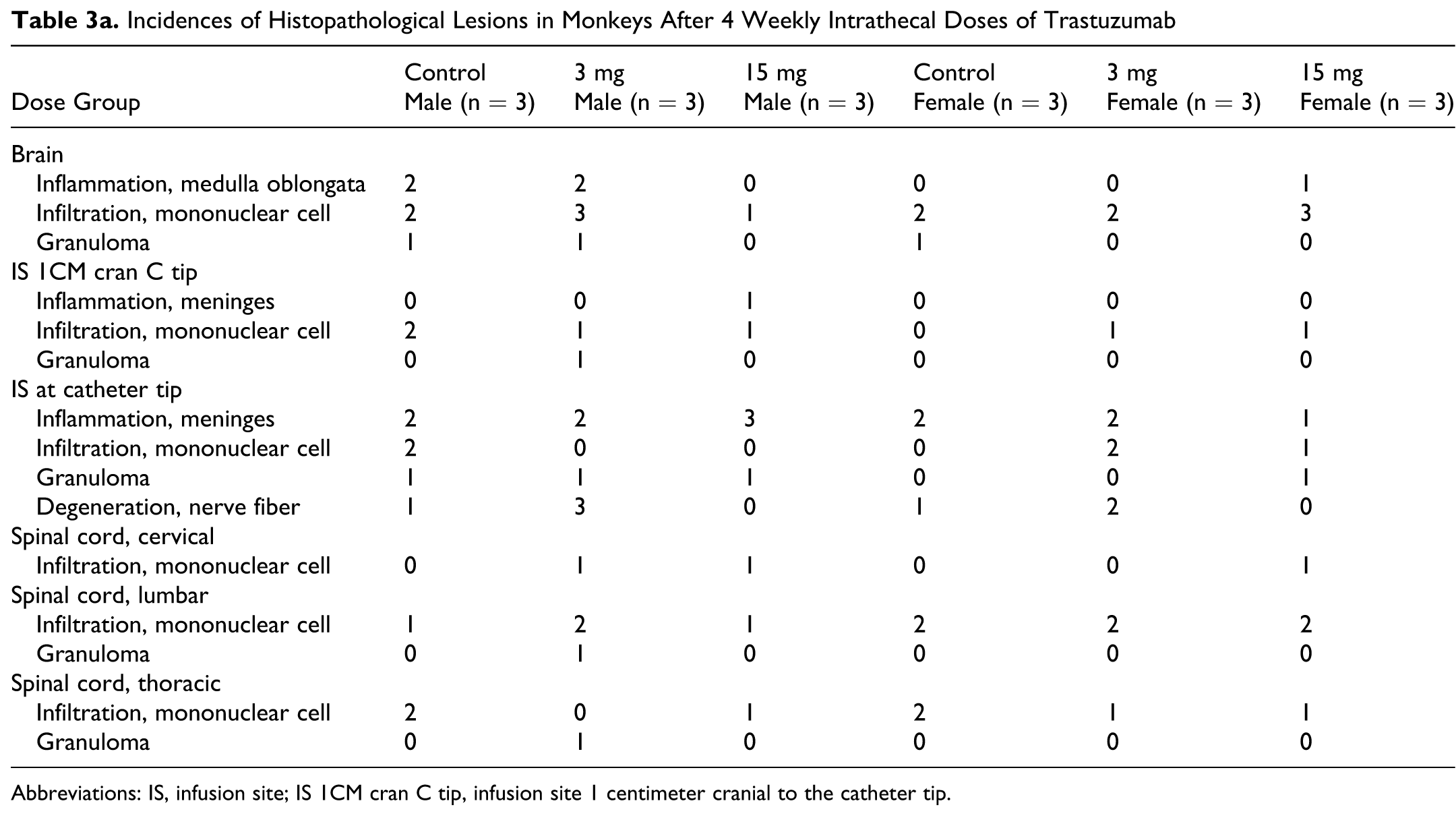
Abbreviations: IS, infusion site; IS 1CM cran C tip, infusion site 1 centimeter cranial to the catheter tip.
Incidences of Histopathological Lesions in Monkeys After a 4-Week Recovery Period Following 4 Weekly Intrathecal Doses of Trastuzumab
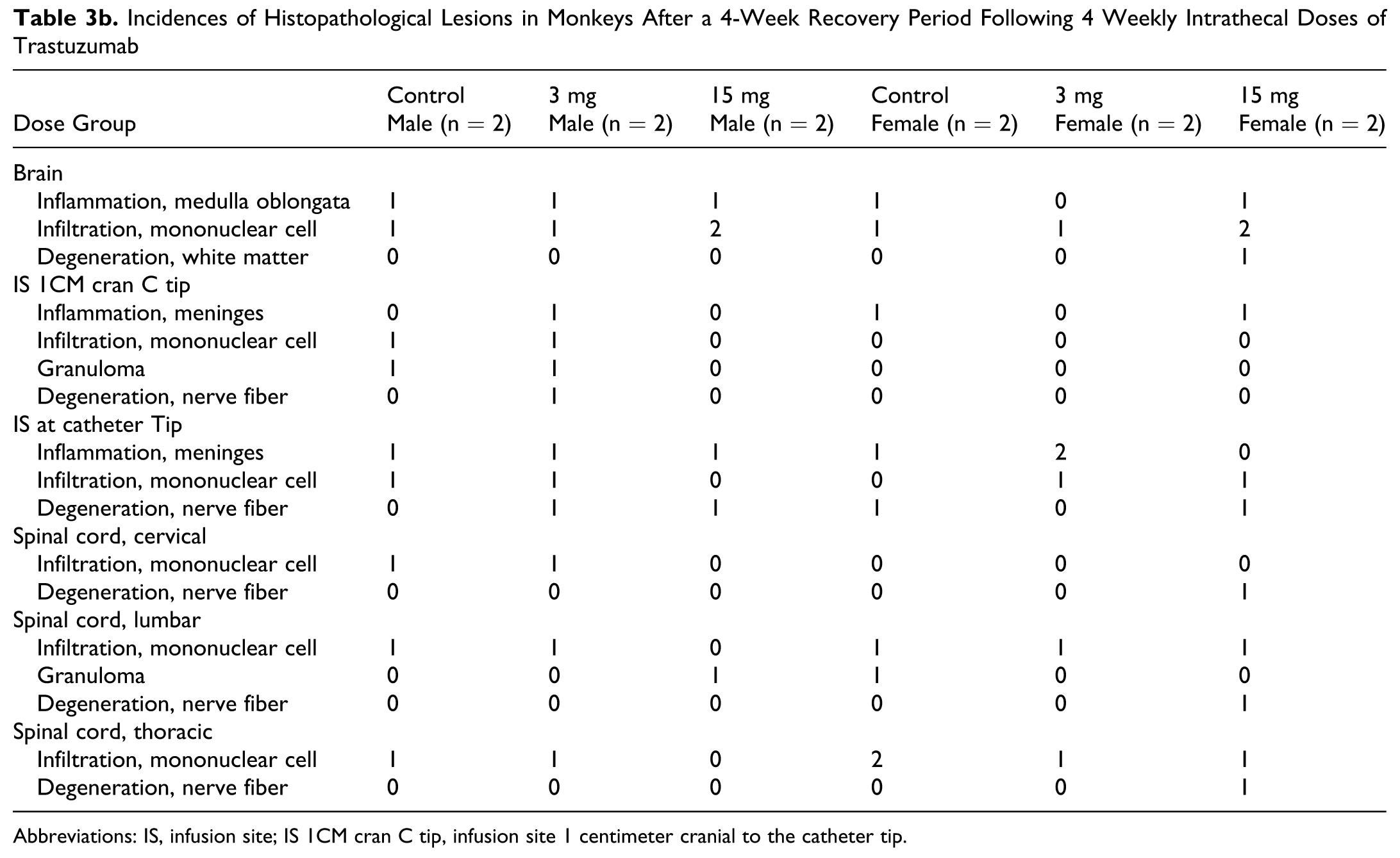
Abbreviations: IS, infusion site; IS 1CM cran C tip, infusion site 1 centimeter cranial to the catheter tip.
Discussion
This study was conducted in cynomolgus monkeys, a trastuzumab pharmacologically active species, to provide a robust preclinical safety evaluation for the further clinical use of intrathecal trastuzumab. Preservative-free trastuzumab was used in this preclinical safety study, as caution has been raised regarding the use of benzyl alcohol-containing solutions by the intrathecal route. 30,31 The 4 weekly intrathecal doses to cynomolgus monkeys were well tolerated. No test article-related in-life, neurological, clinical pathology, or anatomic pathology findings were noted.
The doses used of 3 and 15 mg per dose resulted in 0.051 and 0.24 mg/g brain doses in cynomolgus monkeys, respectively. Doses administered in 8 clinical case reports ranged from 5 to 100 mg per dose. 15–22 Based on a 1400-g human brain weight, these intrathecal doses would be equivalent to 0.0036 and 0.071 mg/g brain weight in humans. 29 Thus, the doses used in the 4-week monkey study were 3.4- to 67-fold higher than those reported in the 8 case reports. In addition, the CSF concentrations achieved in the 4-week monkey study (mean CSF concentration of 376 µg/mL) were also substantially higher than those reported in patients (6.4 µg/mL at an intrathecal dose of 20 mg) (Table 1). Furthermore, the CSF concentrations achieved in the 4-week monkey study were also higher than those achieved in the serum of patients by intravenous administration (Table 1). As such, the doses and local concentrations achieved in the 4-week monkey study were well in excess of those described in the case reports reported in the literature. Although not all CSF samples were collected in this study because of technical difficulties consisting of blocked ports (see also Perron et al 27 ), sufficient sample time points were available for a full toxicokinetic evaluation.
The toxicokinetic profile of trastuzumab in this monkey study was similar to that described in the literature with intrathecally administered rituximab, a monoclonal antibody (mAb), to cynomolgus monkeys. 26 Pharmacokinetically, the CSF is not considered a “well-stirred” compartment. Following an intraventricular or intrathecal injection, drugs unevenly distribute in the CSF, which is shown to be influenced by injection, sampling sites, and gesture position after administration and sampling. 32 As a result, CSF concentrations have been shown to vary within a large range. 32 In the current study, the mAb trastuzumab was intrathecally infused at the lumbar level in cynomolgus monkeys, and CSF samples were taken from a catheter placed in the cisterna magna at the cervical level. A large variation in the CSF concentration was observed between monkeys (Figure 1 and Table 2). In addition, a large variation in the CSF concentration was observed within an individual monkey between day 1 and day 22 (data not shown). The values of % CV were between 30% and 100% for mean C6 hrs and AUCs. The CSF concentration of the mAb appeared to vary more when compared to a small molecular drug after intrathecal injection. 33 As a result, the fitted PK parameters were significantly variable, with %CV values between 50% and 150% (Table 2). The value of mean residence time (MRT) in the CSF was 10.1 ± 0.843 hours and 10.8 ± 1.85 hours at the dose level of 3 and 15 mg, respectively, which was much less than those in the serum (Table 2), but higher than has been reported after intrathecal administration of the mAb rituximab in cynomolgus monkeys. 26 This observation indicated rapid transfer of trastuzumab from the CSF into the serum after an intrathecal injection of this mAb.
Combined with the results of clinical intrathecal case studies and the previous intravenous preclinical safety information, the results of this 4-week toxicology study in monkeys support further studies to evaluate intrathecal application of trastuzumab in patients with brain metastasis in HER2-positive breast cancer.
Footnotes
Acknowledgments
This study was sponsored by Hoffmann-La Roche, Inc. The authors are very grateful for the dedication and help of many people at Charles River Laboratories, with special thanks to Y. Trudel, S. Caron, H. Simard, and N. Grégoire for their support in the conduct of the studies. For the pathology peer review, the authors thank M. Odin (Hoffmann-La Roche). The expert help of Nadia Kulagina (Covance) for the bioanalyses of the serum and CSF samples is appreciated. Lastly, the authors are grateful to Drs. L. Benincosa and D. Brewster for reviewing this manuscript.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
