Abstract
Cannabinoid neuroprotection is usually greater in vivo than in neuronal cell culture systems. To the authors' knowledge, a good in vitro culture model for the neuroprotective effects of cannabinoids does not exist. Therefore, a 3-dimensional (3D) culture system was developed to investigate the neuroprotective effects of the cannabinoid receptor agonist WIN-55,212-2 on apoptosis of differentiated PC12 cells, caused by the organophosphorus compounds paraoxon and diazinon. Cells pretreated with WIN-55,212-2 were exposed to a proapoptotic concentration of paraoxon and diazinon. TUNEL was used to detect apoptosis, and neurite length was assessed by morphometry. Both paraoxon and diazinon induced apoptosis, although the latter was more potent. WIN-55,212-2 also protected cells from neurite retraction and DNA fragmentation induced by the OPs. The results suggest that WIN-55,212-2 protects PC12 cells cultured under 3D conditions from organophosphorus-induced apoptosis. This 3D culture system may prove to be a useful tool for investigating the neuroprotective effects of cannabinoids.
Despite a history of more than 4000 years of medical use, 1 cannabinoids have only gradually been introduced into modern medicine. Recent studies have explored the utility of cannabinoids for treating muscle pain and weakness, nausea and emesis, appetite disorders, cancers, and neurodegenerative disorders. 2-5 The neuroprotective action of cannabinoids is attributed to different mechanisms, including inhibition of voltage-sensitive Ca2+ channels to reduce Ca2+ influx, glutamate release, and excitotoxicity; antioxidant capacity; and modulation of glial cell function. 6-8 .
Organophosphorus agents (OPs) are a well-known neurotoxin. In addition to being used as pesticides, some OP toxins have also been used as chemical warfare agents and continue to be potential weapons of chemical terrorism. 9 Exposure of neuronal cells to OPs can affect cell maintenance and ultimately cause cell death via apoptosis and necrosis. Previous studies of cell death induced by OPs demonstrated that long-term in vitro exposure of hippocampal neurons and neuronal cell lines to OPs resulted in necrosis 10 and short-term exposure–induced apoptosis. 11-13 Apoptotic cell death is accompanied by rounding of the cell and pseudopod retraction, reduction in cell volume, chromatin condensation, nuclear fragmentation, and plasma membrane blebbing. 14,15
The PC12 cell line was first characterized in 1976 and has been used as a neuronal model in many studies. 16 A notable characteristic of PC12 cells is that they can be induced readily to differentiate into neurons in culture with the neurotrophic factor nerve growth factor (NGF). 17 Although culturing cells in 2 dimensions is a convenient method for preparing, observing, and studying cells and their interactions with pharmaceuticals, biological factors, and biomaterials in vitro, it does not accurately mimic cell growth in vivo. In living organisms, cells generally exist under 3-dimensional (3D) conditions. PC12 cell differentiation in 3D cultures mimics cell growth in tissues and thus can be used as an in vitro model for investigating development, metabolism, regeneration, pathogenesis, and toxicity in neurons. 18,19
In this study, we used proapoptotic concentrations of 2 common OP toxicants (paraoxon and diazinon) to induce apoptosis in differentiated PC12 cells. We then investigated the neuroprotective effect of the cannabinoid receptor agonist WIN-55,212-2. Our objective was to explore the link between the neuroprotective effects of cannabinoids and OP neurotoxicity under 3D culture conditions.
Methods
Cell Culture
PC12 cell line was purchased from Pasteur Institute (Tehran, Iran) and maintained in RPMI 1640 (Gibco, Eggnestein, Germany) culture medium supplemented with 10% fetal bovine serum (Gibco), 2 mM L-glutamine (Sigma-Aldrich, St. Louis, MO), 100 µ/mL penicillin, and 100 mg/mL streptomycin. Cultures were incubated at 37°C in 5% CO2 and air. The culture medium was changed every 3 days until cells were confluent.
Cellular Differentiation Under 3D Culture Conditions
To induce differentiation of PC12 cells under 3D cell culture conditions (fibrin gel), we dissolved 3 mg/mL fibrinogen (Sigma) in culture medium for use as a pregel solution. PC12 cells resuspended by trypsinization were carefully mixed with the pregel solution; the mixture was then plated onto a 24-well culture dish at a density of 10 4 cells/well. Ten microliters of thrombin solution (Sigma) was added to 200 µL of the mixture. The dish was then placed at 37°C for approximately 1 hour to allow gelling to occur. The culture medium (RPMI supplemented with 50 ng/mL NGF) was changed every 2 days.
Experimental Protocol
There were 2 sets of experiments: (1) Different concentrations of paraoxon (Sigma) and diazinon (Sigma) were used to determine the concentration that would induce apoptosis after 18 hours of exposure. (2) The protective effect of WIN-55,212-2 (Tocris, Bristol, UK) against apoptosis induction and neurite retraction induced by paraoxon and diazinon was investigated.
Our treatment groups comprised the following: (1) Differentiated PC12 cells treated with proapoptotic concentrations of paraoxon (200 µM) or diazinon (50 µM) for 18 hours (POX and DZ groups, respectively). (2) Differentiated PC12 cells pretreated with 0.1 µM WIN-55,212-2 for 18 hours and then exposed to 200 µM paraoxon or 50 µM diazinon (WP and WD groups, respectively). (3) Differentiated PC12 cells pretreated with 1 µM cannabinoid receptor antagonist AM-251 (Tocris) and 0.1 µM WIN-55,212-2 (Tocris) for 18 hours, followed by exposure to 200 µM paraoxon or 50 µM diazinon (AWP and AWD groups, respectively).
Apoptosis Detection
An in situ cell death detection kit (Roche, Mannheim, Germany) was used to identify the apoptotic cells by TUNEL (terminal uridine deoxynucleotidyl transferase dUTP nick end labeling) staining, following the manufacturer’s protocol. Briefly, all cells were fixed with 4% wt/vol paraformaldehyde for 10 minutes, permeabilized, blocked, and incubated with a mixture of fluorescent labeled nucleotides on terminal deoxynucleotidyl transferase catalyzed the polymerization of labeled nucleotides to 3/0H terminals of DNA fragments. The cells were then counterstained with 10 µg/mL propidium iodide (red) at room temperature for 15 minutes and washed with phosphate-buffered saline. A positive apoptosis control, cells induced into apoptosis by 5% ethanol treatment, was included in each assay. TUNEL-positive cells were counted in 8 randomly selected fields from each culture under a fluorescent microscope (Olympus AX-70; Tokyo, Japan), and apoptotic index was calculated by dividing the number of apoptotic cells by the total cells.
Morphological Estimation
Differentiated PC12 cells have an irregular shape, characterized by several branched neurites of uniform caliber and varying length. For differentiated PC12 morphology, total neurite length was estimated by the recommended method of Ronn et al. 20 Six microscopy fields in each group were randomly selected for morphological estimation.
Statistical Analysis
Data are shown as mean ± standard error of the mean and were analyzed using 1-way analysis of variance (ANOVA) followed by Tukey’s test and t test. Statistical analysis was performed using SPSS 16 for Windows, and P values less than .05 were considered statistically significant.
Results
Upon exposure to NGF in the 3D fibrin gel, PC12 cells stopped dividing, attached more strongly to the substratum, and extended long neurite process (Figure 1). After 3 days, cells that had long neurite outgrowths (approximately 2-fold longer than the cell nucleus) and resembled neurons were considered to be differentiated PC12 cells and were used in further experiments.
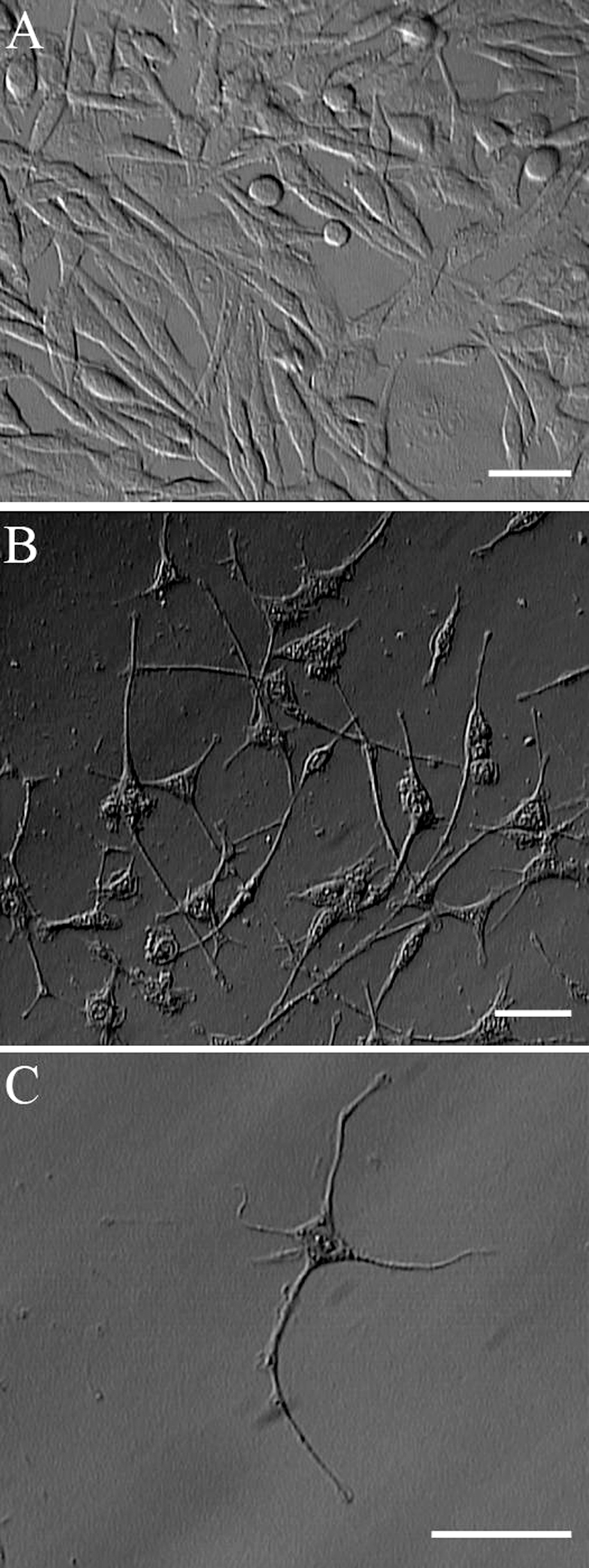
PC12 differentiation under 3D culture conditions. After 3 days, mitotic and undifferentiated PC12 cells (A) plated in 3D fibrin gel ceased proliferation and acquired many features of neurons, including outgrowth of long-branching neurites (B and C). Scale bar = 30 µm. 185 × 493 mm (72 × 72 DPI).
Effect on Apoptosis Induction of Different Concentrations of Paraoxon and Diazinon
Both paraoxon and diazinon induced apoptosis in differentiated PC12 cells in the 30- 50-µM concentration range, although the effect of diazinon was more pronounced. As shown in Figure 2, apoptosis induced by 50 µM diazinon was similar to that induced by 200 µM paraoxon (Figure 2). Given these findings, we used 50 µM diazinon and 200 µM paraoxon as equivalent proapoptotic concentrations.
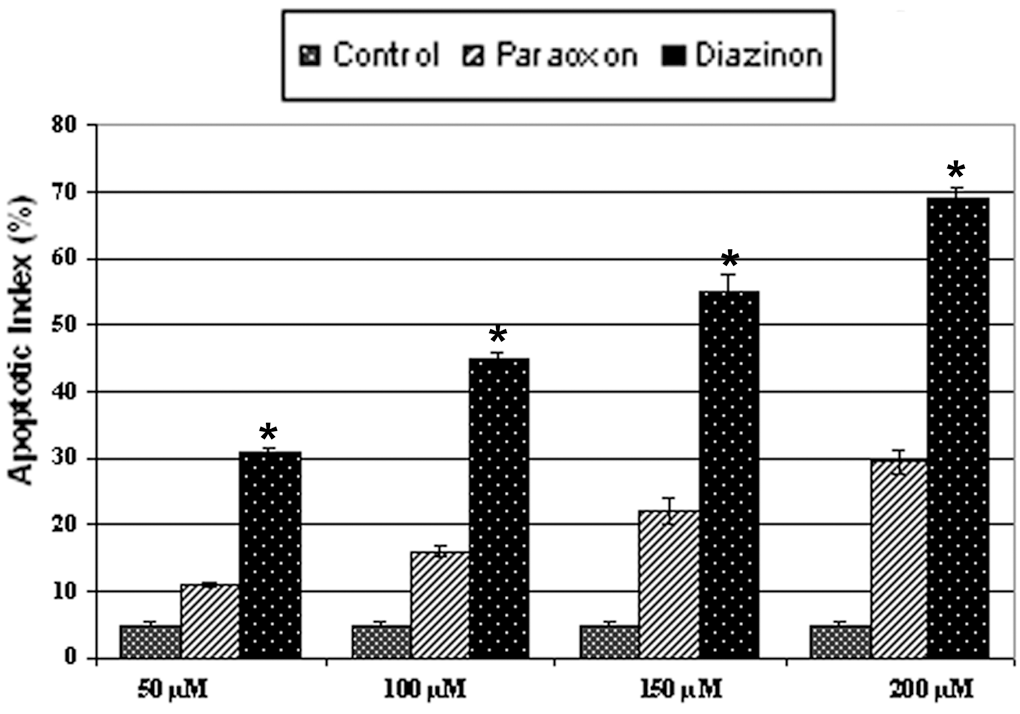
Paraoxon and diazinon induced apoptosis in a dose-dependent manner (18 hours of exposure). *Statistically significant difference in apoptotic index between paraoxon and diazinon groups (n = 4, P < .05). 165 × 115 mm (120 × 120 DPI).
WIN-55,212-2 Protects Differentiated PC12 Cells From Paraoxon- and Diazinon-Induced Apoptosis
Differentiated PC12 cells displayed the characteristic features of apoptosis when visually inspected by fluorescent microscopy; apoptotic cells exhibited bright labeling by TUNEL staining of fragmented nuclear DNA (Figure 3). The apoptosis index in cells pretreated with 0.1 µM WIN-55,212-2 for 18 hours was about 5- to 6-fold lower than in the AWP, AWD (cells pretreated with 1 µM cannabinoid receptor antagonist AM-251 for 20 hours and 0.1 µM WIN-55,212-2 for 18 hours, followed by exposure to 200 µM paraoxon or 50 µM diazinon), POX, and DZ groups (cells treated with proapoptotic concentrations of paraoxon [200 µM] or diazinon [50 µM] for 18 hours) (Figure 4 ; P < .05, ANOVA). Thus, these data demonstrate that WIN-55,212-2 protects differentiated PC12 cells from apoptosis induced by OPs.
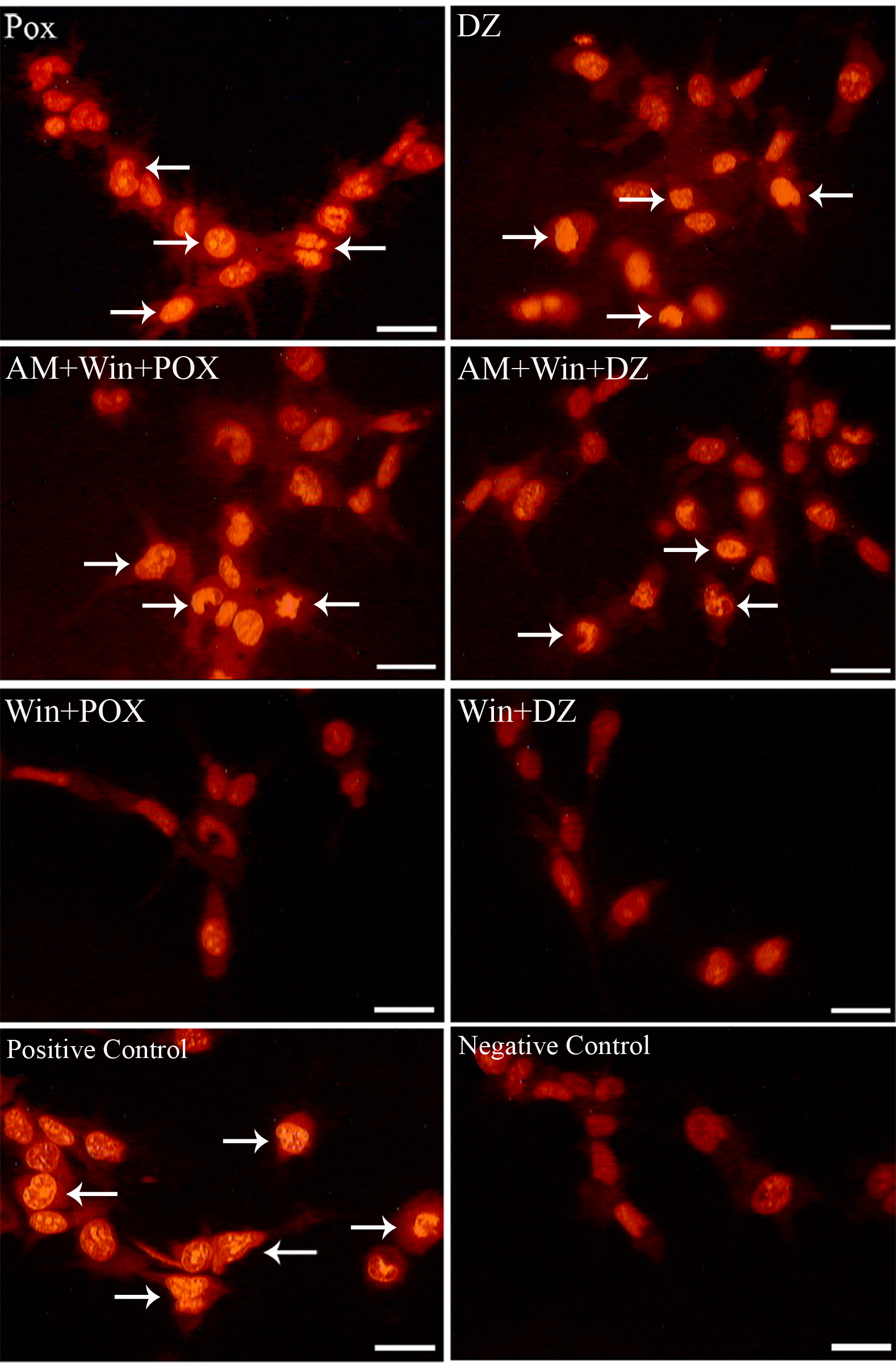
WIN-55,212-2 protected differentiated PC12 cells from apoptosis induced by paraoxon and diazinon. TUNEL staining labeled apoptotic cells (arrows) and revealed DNA fragmentation and chromatin condensation. Scale bar = 20 µm. 327 × 498 mm (72 × 72 DPI).
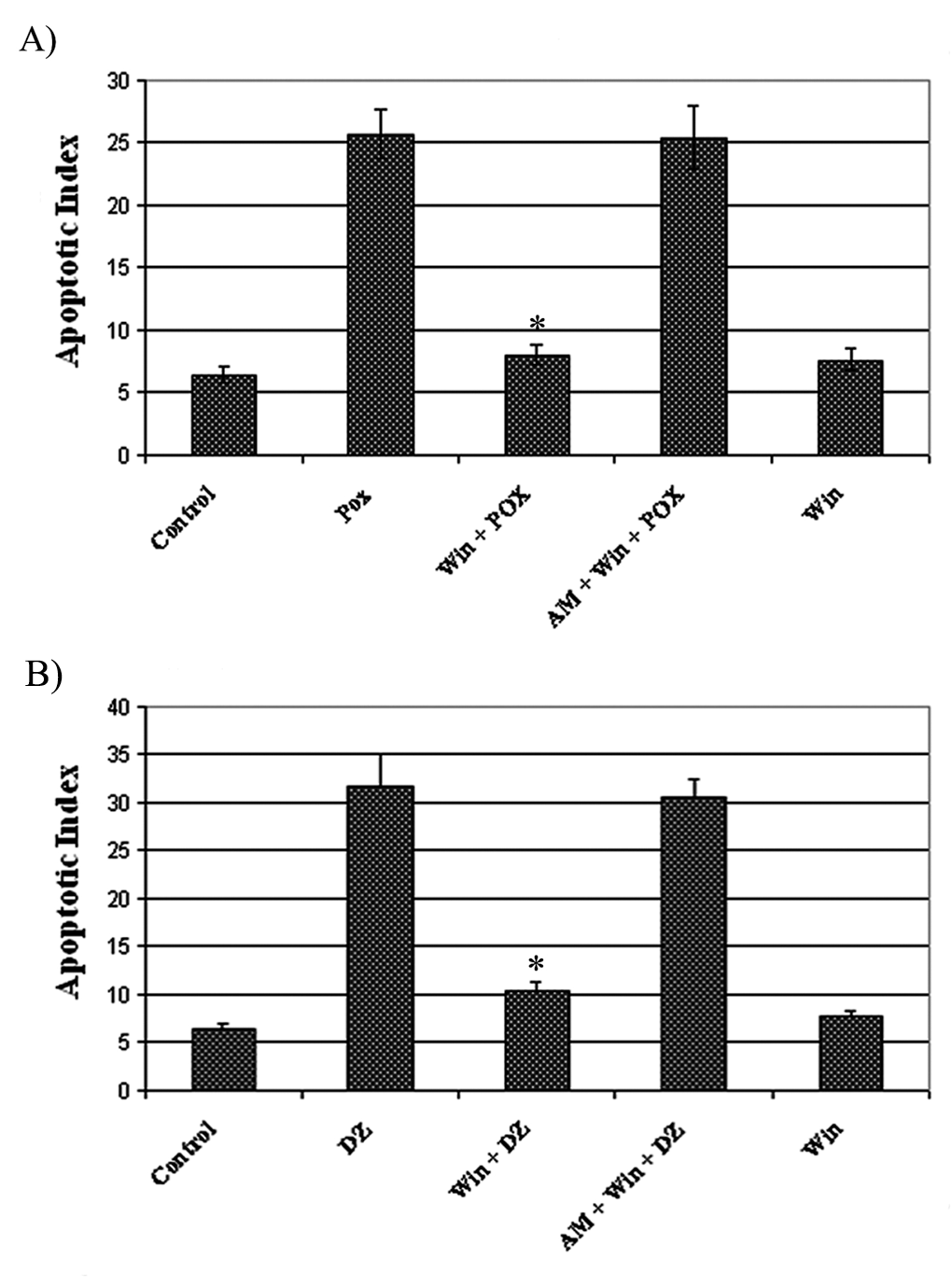
Effect of WIN-55,212-2 on apoptosis induced by paraoxon and diazinon. Quantitative analysis of apoptotic index revealed statistically significant differences (asterisk) between cells pretreated with WIN-55,212-2 and the other groups (n = 7, P <.05). 185 × 250 mm (200 × 200 DPI).
Because several studies have reported cannabinoids induced apoptosis in some neuronal cell lines, we only examined effect of WIN-55,212-2 on differentiated PC12 cells. Results showed that WIN-55,212-2 did not induce apoptosis in differentiated PC12 cells in comparison with the control group (P < .05, t test).
Protective Effect of WIN-55,212-2 on Morphological Damages Induced by Paraoxon and Diazinon
Cells that were treated with paraoxon and diazinon displayed retraction of neurites and a decrease in the total neurite length ( Figure 5). Our morphometric findings revealed that the total neurite length in the WP and WD groups (cells pretreated with 0.1 µM WIN-55,212-2 for 18 hours and then exposed to 200 µM paraoxon or 50 µM diazinon, respectively) was approximately 2- to 4-fold greater than those in the AWP, AWD (cells pretreated with 1 µM cannabinoid receptor antagonist AM-251 for 20 hours and 0.1 µM WIN-55,212-2 for 18 hours, followed by exposure to 200 µM paraoxon or 50 µM diazinon, respectively), POX, and DZ groups (cells treated with proapoptotic concentrations of paraoxon [200 µM] or diazinon [50 µM] for 18 hours) (Figure 6, P < .05, ANOVA). Thus, PC12 cells pretreated with WIN-55,212-2 did not undergo the morphological changes typically induced by OPs.
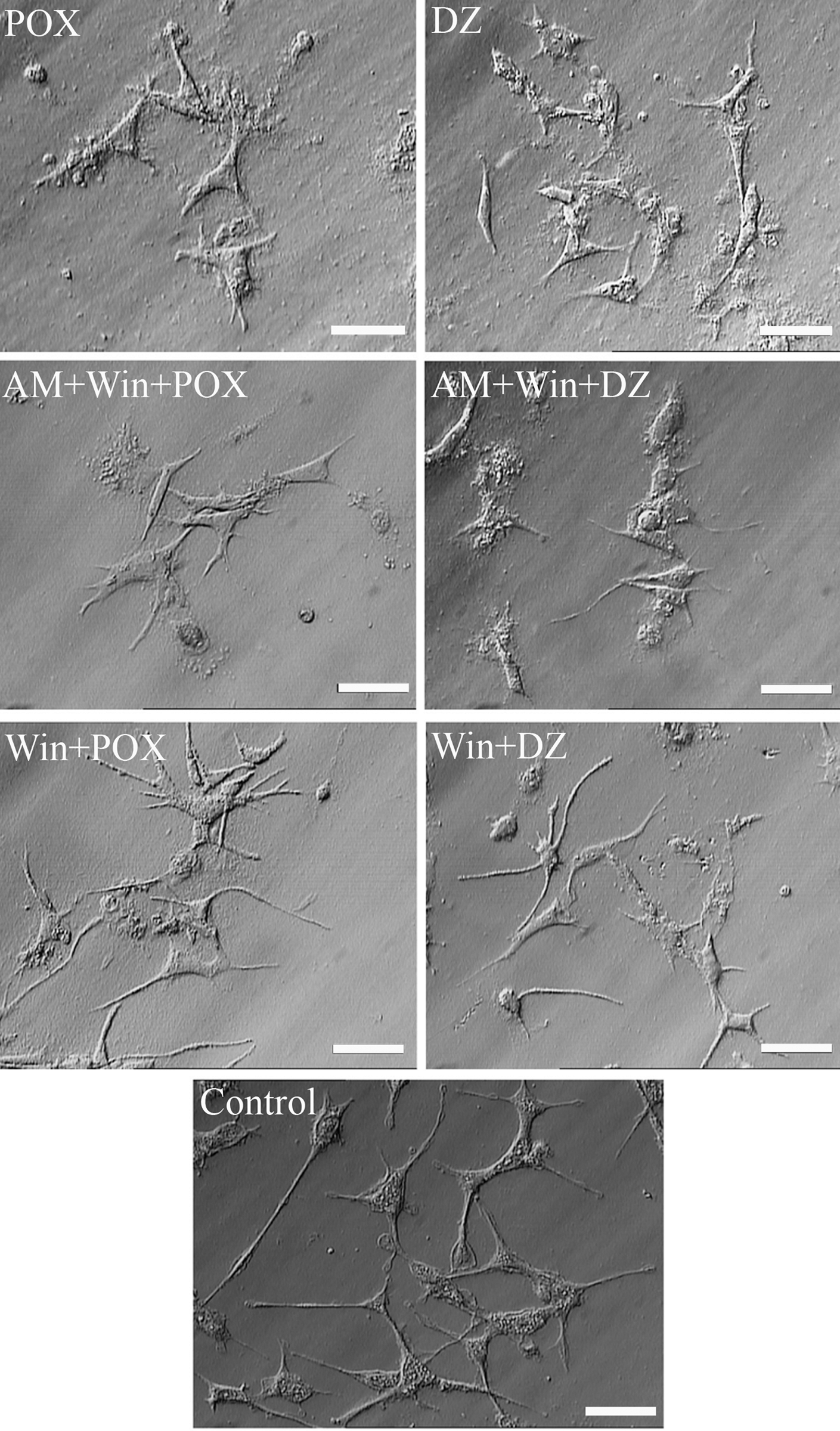
WIN-55,212-2 ameliorated morphological damage and neurite retraction induced by paraoxon and diazinon. Scale bar = 40 µm. 272 × 464 mm (72 × 72 DPI).
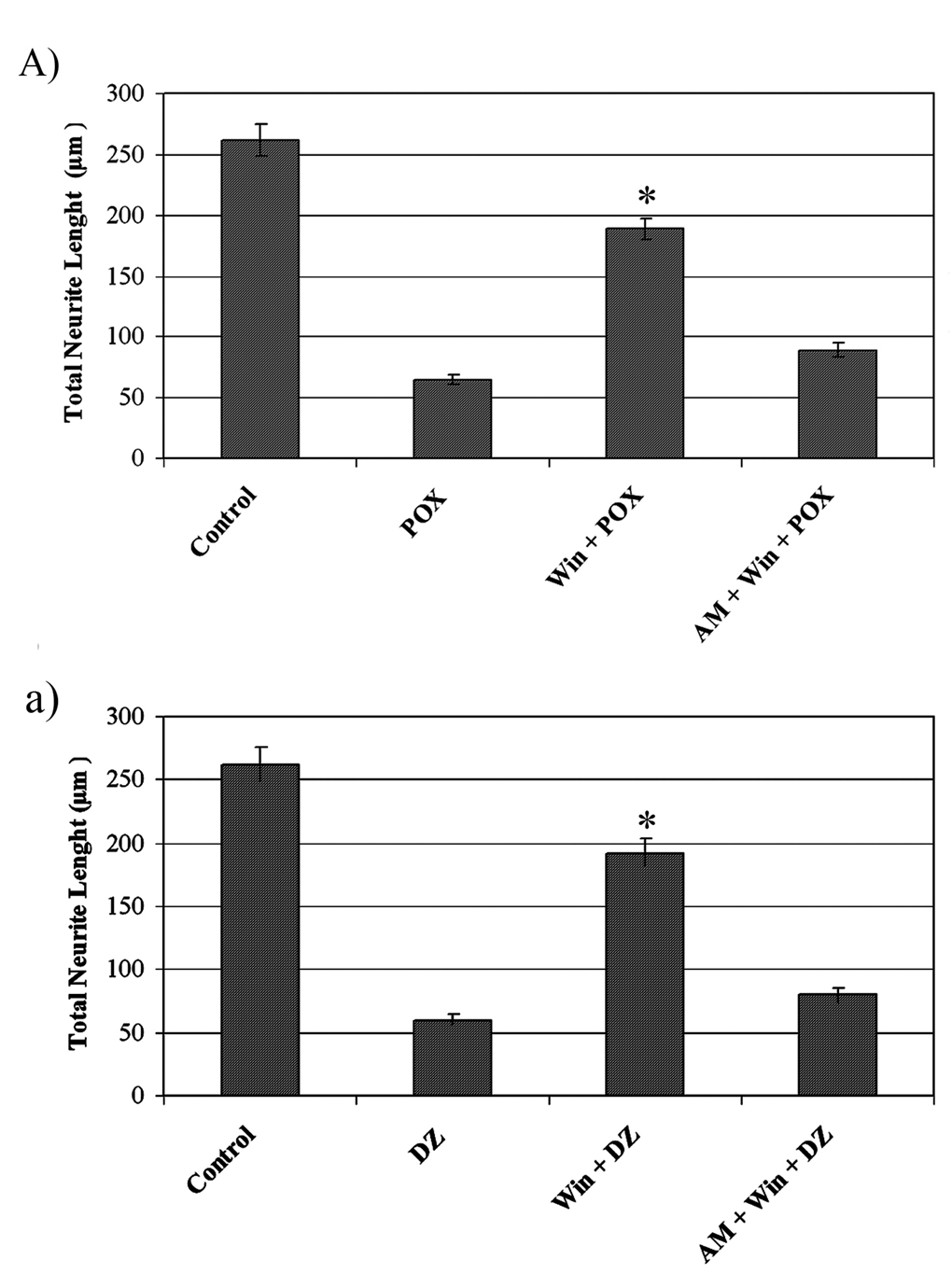
WIN-55,212-2 protected differentiated PC12 cells from morphological damage induced by paraoxon and diazinon. Quantitative analysis of morphometric findings revealed statistically significant differences (asterisk) between cells pretreated with WIN-55,212-2 and the other groups (n = 6, P < .05). 185 × 250 mm (200 × 200 DPI).
Discussion
Over the last few years, a number of studies have reported that cannabinoids protect neuronal cells from toxicological insults. 21,22 In line with these studies, our data suggest that the cannabinoid receptor agonist WIN-55,212-2 protects differentiated PC12 cells from OP-induced apoptosis.
In the present study, we used differentiated PC12 cells, a standard in vitro model for neuronal research; these cells have been used to characterize important features of OP-induced developmental neurotoxicity. 23 Differentiated PC12 cells have an advantage over cultured primary neurons, which do not maintain cell division. Furthermore, primary neurons do not provide a uniform population with regard to either cell types or differentiation states. PC12 cells in 3D culture systems possess characteristic cell functions such as extracellular matrix production and expression of differentiation markers. The resulting cellular structures can be used as models for investigating development, pathogenesis, and toxicity. Upon exposure to NGF in 3D fibrin matrices, PC12 cells gradually exit the mitotic cycle and begin to differentiate. They develop axonal projections, express a battery of neuronal genes, and, finally, acquire the characteristics of neurons. 24,25
Degradation (desulfuration) of parathion by sunlight or liver enzymes results in the formation of paraoxon, which, like other OP compound insecticides, has acute effects on both insects and mammals. 26 In our earlier work with OPs, we concluded that long-term (3 days to 2 weeks) exposure of hippocampal neurons to paraoxon induced concentration-dependent morphological and neuropathological changes. Discontinuity of the plasma membrane, the production of numerous lysosome and lipid vacuoles, mitochondrial swelling, the formation of whorled phospholipid masses, and rough endoplasmic reticulum breakdown, all seen via transmission electron microscopy, were evidence of paraoxon-induced necrosis in hippocampal neurons. 10 Similarly, Wu et al 13 demonstrated that paraoxon neurotoxicity was time and concentration dependent in cultured rat cerebellar granules. They noted that short-term (12-18 hours) paraoxon exposure induced apoptosis in cultured cerebellar granule cells. Moreover, Saleh et al 26 observed necrosis in EL4 T lymphocytes pretreated with high concentrations (0.1 mM) of paraoxon. The discrepancy in the dose effects between these studies may be related to the different cell lines that were investigated.
Recent evidence indicates that exposure to diazinon also causes neurotoxic effects in brain cell development, neuronal cell signaling, and neurotransmitter systems. 27,28 Diazinon is a phosphorothionate, which is rapidly metabolized in vivo by liver microsomal enzymes, mostly to its oxon analog, diazoxon. 29 However, neuroblastoma cell lines and neuronal cells that were differentiated in in vitro systems are generally unable to convert phosphorothionate insecticides to their oxon forms. 30 Hence, diazinon has immediate and direct inhibitory actions on neural cell replication. 31 Sidiropoulou et al 32 found that although diazoxon and its parent compound share the ability to impair outgrowth of long neurites in N2a cells under identical conditions, the patterns of biochemical effects induced by these 2 substances were somewhat different.
Based on these reports, and because of the time- and concentration-dependent nature of OP-mediated cell death, we examined several concentrations of paraoxon and diazinon to determine the optimal concentration for inducing apoptosis in differentiated PC12 cells after an 18-hour exposure. We found that both paraoxon and diazinon began to induce apoptosis in the 30- to 50-µM concentration range. In addition, diazinon was more potent than paraoxon. For example, the apoptosis index of cells treated with 50 µM diazinon was approximately equivalent to that of cells treated with 200 µM paraoxon.
One of the most exciting areas in cannabinoid research is the ability of these compounds to control the cell survival/death decision. Cannabinoids have been shown to induce the apoptotic death of glioblastoma cells, 33 CHP100 neuroblastoma cells, 34 and undifferentiated PC12 cells. 35 In contrast, the results of Shen and Thayer 21 suggest that low concentrations of WIN-55,212-2 protect hippocampal neurons from presynaptically evoked glutamate excitotoxicity. In addition, Gomez del Pulgar et al 22 used cannabinoids to protect astrocytes from ceramide-induced apoptosis via activation of the PI3/PKB cytoprotection pathway. They claimed that protection by WIN-55,212-2 reached a maximum 18 hours after the addition of the cannabinoid. 22 These studies demonstrate that the dual effect of cannabinoids on cell survival and cell death depends upon the cell type and the cannabinoid concentration. In our study, we used differentiated PC12 cells to investigate the protective effects of WIN-55,212-2 against paraoxon- and diazinon-induced apoptosis. We found that apoptosis in cells pretreated with 0.1 µM WIN-55,212-2 was approximately 3-fold lower than in cells not pretreated with WIN-55,212-2, or those pretreated with 1 µM AM-251, a cannabinoid receptor antagonist. Thus, low concentrations of WIN-55,212-2 not only did not induce apoptosis in differentiated PC12 cells but actually protected them from paraoxon- and diazinon-induced apoptosis.
Cells undergoing apoptosis proceed through various stages with characteristic morphological and biochemical changes. To determine at what point in the process apoptosis is interrupted by WIN-55,212-2, we measured the total neurite length and performed TUNEL staining. Neurite extension is a critical process in development and nervous system function; it plays an important role in neuronal survival and maintenance. 36 OPs can disrupt neuronal processing through inhibition of growth factor receptors 37 and alterations in the amount of F-actin. 38 In addition, diazinon has been shown to impair the outgrowth of neurites (as shown by a decrease in the membrane/total protein ratio) in the forebrain and brainstem of neonatal rats. 39
Our morphometric findings indicated that cells not pretreated with WIN-55,212-2 or AM-251 showed a statistically significant decrease in total neurite length. These results suggest that WIN-55,212-2 protected differentiated PC12 cells from OP-induced morphological damage, which is indicated by neurite retraction.
Our 3D culture condition study yielded 3 findings: (1) Both paraoxon and diazinon induced apoptosis in differentiated PC12 cells, and diazinon appeared to have a greater effect. (2) WIN-55,212-2 protected differentiated PC12 cells from apoptosis and morphological changes induced by paraoxon and diazinon. (3) Because one of the most important signs in the early stages of neuronal apoptosis is neurite retraction, WIN-55,212-2 may protect differentiated PC12 cells during the early stages of apoptosis.
