Abstract
Capsaicin, a pungent compound found in hot chili peppers, has been reported to have antitumor activities in many human cancer cell lines, but the induction of precise apoptosis signaling pathway in human nasopharyngeal carcinoma (NPC) cells is unclear. Here, we investigated the molecular mechanisms of capsaicin-induced apoptosis in human NPC, NPC-TW 039, cells. Effects of capsaicin involved endoplasmic reticulum (ER) stress, caspase-3 activation and mitochondrial depolarization. Capsaicin-induced cytotoxic effects (cell death) through G0/G1 phase arrest and induction of apoptosis of NPC-TW 039 cells in a dose-dependent manner. Capsaicin treatment triggered ER stress by promoting the production of reactive oxygen species (ROS), increasing levels of inositol-requiring 1 enzyme (IRE1), growth arrest and DNA-damage-inducible 153 (GADD153) and glucose-regulated protein 78 (GRP78). Other effects included an increase in cytosolic Ca2+, loss of the mitochondrial transmembrane potential (ΔΨ m ), releases of cytochrome c and apoptosis-inducing factor (AIF), and activation of caspase-9 and -3. Furthermore, capsaicin induced increases in the ratio of Bax/Bcl-2 and abundance of apoptosis-related protein levels. These results suggest that ER stress- and mitochondria-mediated cell death is involved in capsaicin-induced apoptosis in NPC-TW 039 cells.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is an endemic disease in southern China and Southeast Asia, with incidence rates of 15–50 per 100,000. 1 Overall, NPC exists with high frequency in Asian populations, especially among Chinese people.2,3 The NPC can spread to lymph nodes and metastasize to other tissue, which causes a poor prognosis. 4 Treatment of NPC has been problematic. Eye or facial paralysis and dietary problems have been demonstrated following chemotherapy and radiation therapy exposure; hence, the need for new therapeutic agents.5,6
Capsaicin (8-methyl-N-vanillyl-6-nonenamide) has been shown to act as a chemopreventive and chemotherapeutic agent in different human cancer models.7–12 In leukemia cells, capsaicin triggered apoptosis via a p53-dependent mechanism. 8 In human esophagus epidermoid carcinoma cells, capsaicin induced apoptosis through the elevation of intracellular reactive oxygen species (ROS) and Ca2+ productions and caspase-3 activation. 9 Capsaicin-affected prostate cancer cells were associated with apoptosis through a p53-independent pathway. 7 In colorectal cancer cells, capsaicin-induced apoptosis is elevated by cotreatment with the adenosine monophosphate-activated protein kinase activator. 13 Moreover, capsaicin treatment suppressed azoxymethane-induced aberrant cryptic foci formation in rats in vivo. 14
Numerous studies have shown that capsaicin has anticarcinogen, 15 antimutagenic 9 and anticancer activities. 16 However, there is no information regarding the anticancer activity of capsiacin in NPC cells and its molecular mechanisms of apoptosis induction are not understood. Therefore, the aim of this study was to investigate the apoptotic action of capsaicin on human NPC, NPC-TW 039, cells in vitro.
Materials and methods
Chemicals and reagents
Capsaicin, dimethyl sulfoxide (DMSO), propidium iodide (PI), RNase and trypan blue were obtained from Sigma-Aldrich Corp (St. Louis, MO, USA). RPMI-1640 medium, fetal bovine serum (FBS), penicillin–streptomycin, trypsin-EDTA, L-glutamine, 2,7-dichlorodihydrofluorescein diacetate (DCFH-DA), 1-[2-Amino-5-(6-carboxy indol-2-yl)phenol]-2-(2′-amino-5′methylphenoxy)ethane-N,N,N′,N′-tetra acetic acid pentaacetoxymethyl ester (Indo 1/AM), 3,3′-dihexyloxacarbocyanine iodide (DiOC6) were purchased from Invitrogen Life Technologies (Carlsbad, CA, USA). Caspase-3, -8 and -9 activity assay kits were obtained from OncoImmunin, Inc. (Gaithersburg, MD, USA).
Cell culture
The human NPC cell line (NPC-TW 039) was provided by Dr Chiou-Ying Yang (Institute of Molecular Biology, National Chung Hsing University, Taichung, Taiwan) and maintained in Dulbecco’s modified Eagle’s medium (DMEM; Invitrogen Life Technologies) supplemented with 10% FBS, 2 mM L-glutamine, 100 Units/ml penicillin and 100 μg/ml streptomycin in 75 cm2 tissue culture flasks at 37°C under a humidified 5% CO2 and 95% air atmosphere, as we have previously reported.17,18
Determinations of morphology, viability and cell cycle distribution of NPC-TW cells
The NPC-TW 039 cells were plated into 12-well plates at a density of 2 × 105 cells/well for 24 h. Capsaicin (0, 200, 250, 300 and 400 μM) was added to cell regimens and grown for 12, 24, 36 and 48 h. The DMSO (solvent) served as the vehicle control. For morphological examinations, cells in each well were examined and photographed under a phase contrast microscope. Cell viability was determined by a flow cytometric assay, as we have previously reported.19,20 For cell cycle and sub-G1 phase determination, cells were washed with PBS and were fixed in 70% ethanol at –20°C overnight then resuspended in PBS containing 40 μg/ml PI and 0.1 mg/ml RNase and 0.1% Triton X-100 in a dark room for 30 min at 37°C, and analysed with a BD FACSCalibur (Becton-Dickinson, San Jose, CA, USA) and BD CellQuest Pro software (Macintosh) as previously described.20,21
Determinations of apoptosis, DNA damage and fragmentation
The NPC-TW 039 cells (1 × 105 cells/ml) were placed in 12-well plates and treated with 0, 200, 250 and 300 μM of capsaicin for 24 h. Cells in each well were stained with 4,6-diamidino-2-phenylindole dihydrochloride (DAPI) and photographed using a fluorescence microscope as previously described.22,23 The NPC-TW 039 cells (1 × 106 cells/well) were treated with 0, 200, 250, 300 and 400 μM of capsaicin for 24 h to determine DNA fragmentation. DNA was isolated from each sample for gel electrophoresis, visualized with ethidium bromide (EtBr) staining and then photographed under fluorescence light as previously described.24,25
Determination of intracellular ROS, Ca2+ levels and mitochondrial membrane potential (ΔΨm)
The NPC-TW 039 cells (2 × 105 cells/ml) were placed in 12-well plates and treated with or without 300 μM of capsaicin for 1, 2 and 3 h. The cells were harvested, washed with PBS twice, and resuspended in 500 μl of DCFH-DA (10 μM for ROS), Indo 1/AM (3 μg/ml) for Ca2+ and DiOC6 (1 μmol/l) for mitochondrial transmembrane potential in dark room for 30 min. Cells were then analysed by flow cytometry as previously described.24,26
Assays for caspase-3, -8 and -9 activity
The NPC-TW 039 cells (2 × 105 cells/ml) were placed in 12-well plates and treated with 0 and 300 μM capsaicin for 24 and 48 h. Caspase-3, -8 and -9 activities were assessed using PhiPhiLux™-G1D2, CaspaLux™8-L1D2 and CaspaLux™9-M1D2 kits, respectively. Cells were incubated with cell permeable substrates for 1 h at 37°C according to the manufacturer’s instructions and measured using flow cytometric analysis.22,23
Western blotting analysis of protein levels associated with cell cycle and apoptosis
The NPC-TW 039 cells (1 × 106 cells/ml) were placed in 6-well plates and treated with 300 μM capsaicin for 0, 12, 24, 36 and 48 h. The cells were harvested and lysed in the PRO-PREP™ protein extraction solution (iNtRON Biotechnology, Seongnam, Gyeonggi-Do, Korea). For protein determination of each sample, the cell lysates (40 μg of each) were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) on a polyacrylamide gel followed by electrotransfer onto a sequi-blot polyvinylidene difluoride membrane (Bio-Rad, Richmond, CA, USA). Specific polyclonal antibodies (anti-p53, p21, p16, cyclin D, cyclin E, CDK2, CDK6, apoptosis-inducing factor [AIF], cytochrome c, pro-caspase-9, pro-caspase-3, PARP, Bax, Bcl-2, ATF6-α, inositol-requiring 1 enzyme-α [IRE1-α], PERK, glucose-regulated protein 78 [GRP78], pro-caspase-12 and growth arrest and DNA-damage-inducible 153 [GADD153]) were purchased from Santa Cruz Biotechnology, Inc.(Santa Cruz, CA, USA). Horseradish peroxidase-conjugated goat antirabbit or antimouse immunoglobulin G (IgG; Jackson Immuno-Research Laboratories, Inc., West Grove, PA, USA) was used as secondary antibodies for enhanced chemiluminescence (NEN Life Science Products, Inc., Boston, MA, USA) as previously described.24,27
RNA preparation and reverse transcriptase–polymerase chain reaction (RT-PCR)
The NPC-TW 039 cells (1 × 106 cells/ml) placed in 6-well plates were exposed to 300 μM of capsaicin for 6, 12, 24 and 36 h. Total RNA was extracted from the cells using a Qiagen RNeasy Mini kit (Qiagen, Inc., Valencia, CA, USA) as previously described.22,28 A high-capacity complementary DNA (cDNA) reverse transcription kit (Applied Biosystems, Foster City, CA, USA) was used for reverse transcription at 42°C for 30 min. The primer sets for the associated gene of transient receptor potential vanilloid type 1 (TRPV1) as F-CATGGCCAGTGAGAACACCATGG and R-AGCCTTTTGTTCTTGGCTTCTCCT. 29 The thermocycler parameters were 5 min at 95°C; denaturation for 1 min at 95°C; annealing for 1 min at 55°C; 40 cycles of 15 s at 94°C, 30 s at 55°C and 1 min at 72°C; final extension of 10 min at 72°C as described previously. 29 The reaction products were analysed by electrophoresis on 1% agarose gels as described previously.30,31
Statistical analysis
Differences between the capsaicin-treated and control groups were analysed by Student’s t test and a p value < 0.05 was considered significant.
Results
Effects of capsaicin on NPC-TW cell morphology, viability, cell cycle and apoptosis
Figure 1, panels A–C, shows that effects of different doses of capsaicin on morphological changes, viability and cell cycle distribution. Increasing the dose of capsaicin and/or time of incubation led to an increase in morphological changes (Figure 1(A)) and reduced the percentage of viable cells (Figure 1(B)). Capsaicin at 400 μM significantly decreased the viability by almost 65%, after 48-h treatment. Data in Figure 1(C) indicate that the percentage of cells in G0/G1 also was enhanced with increasing concentrations of capsaicin. Figure 1(C) reveals that capsaicin caused a dose-dependent increase in apoptotic cells. Further evidence in support of capsaicin-induced apoptosis can be seen in Figure 2 , panels A and B, which shows that capsaicin induced DNA condensation (Figure 2(A)) and stimulated DNA fragmentation (Figure 2(B)). These effects were concentration dependent.
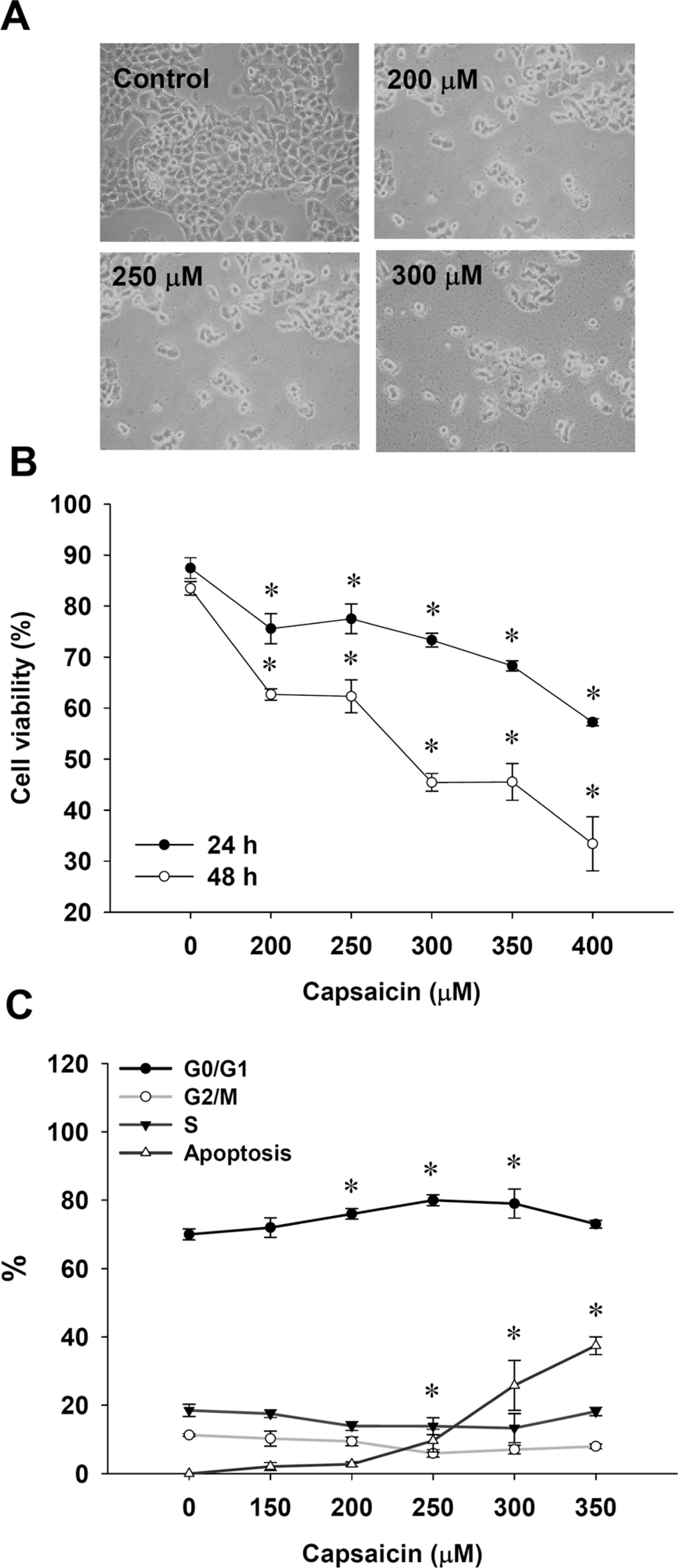
Capsaicin affected the human nasopharyngeal carcinoma cells’ viability and cell cycle distribution of NPC-TW 039 cells. Cells were incubated with or without 0, 200, 250, 300 and 400 μM capsaicin for 24 h and 48 h then were harvested for examining the morphological changes (A), the percentage of viable cells (B) and the distribution of cell cycle (C) by flow cytometry as described in the section on Materials and methods. *p < 0.05.

Capsaicin induced DNA damage and apoptosis in NPC-TW 039. Cells were treated with 0, 200, 250, 300 or 350 μM capsaicin for 24 h or treated with 300 μM capsaicin for 0, 12, 24 and 48 h, and then were harvested for 4,6-diamidino-2-phenylindole dihydrochloride (DAPI) staining (A) and for DNA gel electrophoresis (B) as described in the section on Materials and methods.
Effects of capsaicin on ROS, Ca2+ and mitochondrial membrane potential (ΔΨm) in NPC-TW cells
The ROS production contributes to DNA damage and release of ROS into the cytoplasm. 32 Capsaicin treatment increased ROS and Ca2+ as shown in Figure 3, panels B and C. In contrast, there was a decrease in the mitochondrial transmembrane potential (Figure 3(A)), suggesting that the mitochondria were depolarized in NPC-TW 039 cells.

Capsaicin affected the productions of reactive oxygen species (ROS) and Ca2+, the levels of mitochondria membrane potential (ΔΨ m ) in NPC-TW 039 cells. Cells were treated with 300 μM of capsaicin for 0, 1, 2 and 3 h and then were collected and stained by 3,3’-dihexyloxacarbocyanine iodide (DiOC6) for the determination of mitochondrial transmembrane potential levels (A), stained with 2,7-dichlorodihydrofluorescein diacetate (DCFH-DA) for the determination of ROS levels (B) and stained by 1-[2-Amino-5-(6-carboxy indol-2-yl)phenol]-2-(2′-amino- 5′methylphenoxy)ethane-N,N,N′,N′-tetra acetic acid pentaacetoxymethyl ester (Indo 1/AM) for Ca2+ levels, determined (C) as described in the section on Materials and methods. *p < 0.05.
Effects of capsaicin on the activities of caspase-3 and -9 in NPC-TW cells
It can be seen in Figure 4, panels A and B, that 300 μM capsaicin promoted caspase-9 and -3 activities in a time-dependent manner. However, capsaicin at 300 μM did not stimulate caspase-8 activity (Figure 4(C)). Based on these results, we suggest that capsaicin-triggered apoptosis is mediated through mitochondria- and caspase-dependent pathways rather than extrinsic signaling pathway.

Capsaicin affected caspase-9 and -3 activities in NPC-TW 039 cells. Cells were treated with 300 μM of capsaicin for 24 and 48 h and were collected for determination activities of caspase-9 (A), -3 (B) and -8 (C) as described in the section on Materials and methods. Data represents mean ± SD of three experiments. *p < 0.05.
Levels of proteins associated with cell cycle and apoptosis
Effects of capsaicin on levels of G0/G1 phase, extrinsic, intrinsic and ER stress-related proteins are shown in Figure 5(A–D ). Protein levels of p53, p21 and p16 were increased (Figure 5(A)) but levels of cyclin D, cyclin E, CDK2 and CDK6 were reduced (Figure 5(A)). Capsaicin promoted the levels of Bax (Figure 5(B)), AIF, cytochrome c and PARP (Figure 5(C)), ATF6-α, IRE1-α, PERK, GRP78 and GADD153 (Figure 5(D)) but decreased the levels of Bcl-2 (Figure 5(B)), pro-caspase-9 and pro-caspase-3 (Figure 5(C)), and pro-caspase12 (Figure 5(D)).

The effects of capsaicin on the associated protein levels of apoptosis in NPC-TW 039 cells. Cells were incubated with or without 300 μM capsaicin for 0, 12, 24, 36 and 48 h, and total protein from cells were collected and analysed for Western blotting as described in the section on Materials and methods. A, p53, p21, p16, cyclin D, cyclin E, CDK2 and CDK6; B, Bax and Bcl-2; C, AIF, cytochrome c, pro-caspase-9, pro-caspase-3, PARP; D, ATF6-α, inositol-requiring 1 enzyme-α (IRE1-α), PERK, glucose-regulated protein 78 (GRP78), pro-caspase-12 and growth arrest and DNA-damage-inducible 153 (GADD153).
Effect of capsaicin on mRNA expression
After 300 μM capsaicin treatment in NPC-TW 039 cells for 0, 6, 12, 24 and 36 h, the gene expression of the mRNA levels of TRPV1 were observed. Compared with 0 h, TRPV1 elevation in NPC-TW 039 cells is time dependent (Figure 6).

Capsaicin promoted the expression of transient receptor potential vanilloid type 1 (TRPV1) in NPC-TW 039 cells. The cells were exposed to 300 μM of capsaicin for 0, 6, 12, 24 and 36 h as described in the section on Materials and methods.
Discussion
Capsaicin treatment led to ROS production and induced ER stress in NPC-TW 039 cells. It was also observed that capsaicin-induced ER stress occurred based on the GRP78 and GADD153 increases and ER stress, which elicited a rise in intracellular Ca2+ and subsequent mitochondrial membrane depolarization, followed by mitochondrial release of cytochrome c and consequent activation of caspase-9 and -3 in NPC-TW 039 cells. Additionally, we also observed AIF release from mitochondria after capsaicin treatment in NPC-TW 039 cells. Under normal conditions, tumor suppressor p53 is maintained at a low level 33 ; however, after various cellular stresses, p53 is modified and activates downstream target proteins, including Bax or p21, which have been shown to be involved in apoptosis or cell cycle arrest. 34 Herein, we observed that capsaicin promoted the level of p53 and also its downstream target proteins p21 and p16 (Figure 5(A)). Our results are in agreement with other reports showing that capsaicin elevates intracellular Ca2+ levels,9,34,35 suggesting that Ca2+-mediated signaling is involved in capsaicin-induced apoptosis.
We found that capsaicin treatment induced the expression of ER stress-related proteins including IRE1, GADD153, and GRP78, and the activation of caspase-12. It was previously reported that caspase-12 is activated during constitutive ER stress and is involved in ER stress-mediated apoptosis. 36 The ER stress has earlier been reported to inhibit p53-dependent apoptosis. 37 In the present study, capsaicin treatment increased intracellular Ca2+ levels and induced the loss of the mitochondrial transmembrane potential, and these findings are in agreement with our earlier reports, showing that capsaicin causes apoptosis by increasing intracellular Ca2+ levels and loss of the mitochondrial transmembrane potential. 9
It was reported that the Bcl-2 family of proteins plays an important role in the regulation of outer mitochondrial membrane permeability. 38 An increase in the ratio of Bax/Bcl-2 can lead to an alternation in mitochondrial membrane permeability, which can cause the release of apoptotic mediators such as cytochrome c or AIF-producing caspase activation and DNA degradation.38,39 Our results showed that capsaicin-induced apoptosis was caspase-8 independent (capsaicin did not affect caspase-8 activity). Other reports showed that the mitochondrial outer membrane permeabilization by pro-apoptotic Bcl-2 family members is involved in the release of AIF from the mitochondria.40,41 It was reported that AIF translocates from the mitochondria to the nucleus, then induces caspase-independent peripheral chromatin condensation, and large-scale DNA fragmentation and apoptosis are induced.39,42 The TRPV1 is a nonselective, ligand-gated cationic channel that can be activated by capsaicin, noxious heat, and protons. 43 We found that capsaicin promoted the gene expression of TRPV1 in NPC-TW 039 cells (Figure 6). Capsaicin has been shown to bind to the TRPV1 channel specifically and mediate transient Ca2+ entry and increase cytosolic Ca2+ release. 44 However, the role of TRPV1 in capsaicin-induced apoptosis of NPC-TW 039 cells is still unclear. The NPC-TW 039 cells were pretreated with an inhibitor of TRPV1 then treated with capsaicin. Treatment with the TRPV1 inhibitor altered the effects of capsaicin on the percentage of viable cells, and further investigations are needed.
In conclusion, our results suggest that capsaicin induces ER stress-mediated apoptosis and mitochondria-dependent apoptosis as depicted in the model shown in Figure 7. Capsaicin-induced apoptosis may be regulated by multiple signaling pathways in NPC-TW 039 cells.

The proposed possible signal pathways of capsaicin-induced cell cycle arrest and apoptosis in human nasopharyngeal carcinoma NPC-TW 039 cells.
Footnotes
This study was supported by a grant DOH100-TD-C-111-005 from Taiwan Department of Health, China Medical University Hospital Cancer Research Center of Excellence.
