Abstract
Bee venom (BV) has been used as treatment against a wide variety of ailments, including inflammatory diseases. Various studies have demonstrated anti-inflammatory and anticancer effects of BV. Transforming growth factor (TGF)–β1 induces hepatocyte apoptosis via the mitochondrial permeability transition. However, there is no evidence or information regarding the antiapoptotic effect of BV on hepatocytes. The authors investigated the antiapoptotic effect of BV on TGF-β1-treated hepatocytes. The results showed significant protection from DNA damage by BV treatment compared to corresponding TGF-β1-treated hepatocytes without BV. BV suppressed TGF-β1-induced activation of the bcl-2 family and caspase family of proteins, which resulted in inhibition of poly ADP-ribose polymerase (PARP) cleavage. Furthermore, BV is not cytotoxic in the low concentrations used in this study. Low concentrations of BV potently suppress the apoptotic response in TGF-β1-treated hepatocytes; therefore, BV may have therapeutic potential for the treatment of liver diseases.
Transforming growth factor (TGF)–β1 is a multifunctional cytokine that mediates hepatocellular differentiation, growth, and apoptosis. 1 In the liver, a major function of TGF-β1, which is normally produced by the nonparenchymal stellate cells, is to limit regenerative growth of hepatocytes in response to injury by inhibiting DNA synthesis and inducing apoptosis. 2 In vitro and in vivo studies have also shown that TGF-β1 controls hepatocyte growth directly by inducing apoptosis. 3 TGF-β1 induces apoptosis through reactive oxygen species (ROS) generation, mitochondrial permeability transition, and caspase activation. Moreover, Smad-3-dependent cleavage of the mitochondrial proapoptotic protein, BAD, results in TGF-β1-induced apoptosis in FaO hepatoma cells and suggests a distinct role for Smad-3. These findings suggest that the TGF-β1 apoptotic pathway includes Smad-3 activation, mitochondrial involvement, and caspase cleavage. 4
Apoptosis is a continuous physiologic process of noninflammatory programmed cell death and is one of today’s most active fields of biomedical research. 5 Apoptosis is characterized by profound morphological alterations of the cell, specifically the nucleus. Typical features of apoptosis are the sequential occurrence of cell shrinkage, loss of cell-cell contact, membrane blebbing, and chromatin condensation. The nuclear DNA of apoptotic cells is often fragmented into oligonucleosomal-sized units. 6 The apoptotic signaling pathways mediated by death receptors and other proapoptotic stimuli have been well characterized. 7 Control of apoptosis plays a crucial role in liver regeneration. Among the regulators of apoptosis, the bcl-2 families of proteins determine the life or death of a cell by controlling the releases of mitochondrial apoptogenic factors, cytochrome c, and apoptosis-inducing factors (AIF), which activate the downstream execution phases of apoptosis, including the activation of caspases. The bcl-2 family of genes, which consists of both pro- and antiapoptotic genes, exerts their functions by forming dimers. 8 All apoptotic pathways appear to terminate in the activation of the caspase family of proteases. Caspases are synthesized as inactive precursors that must be cleaved autocatalytically or by other caspases for activation. Triggering of apoptosis results in a cascade of caspase activation, in which the last activated caspases digest cellular substrates, resulting in morphological changes and the death of the cell. 9
Bee venom (BV) consists of several biologically active peptides, including melittin, apamin, adolapin, and mast cell degranulating peptide. 10 BV has been used as a treatment against a wide variety of ailments, including inflammatory diseases, for centuries in Eastern medicine. 11 Several recent papers have reported the antiarthritic and/or anti-inflammatory mechanisms of BV. 12 Recent reports have indicated that acupuncture and administration of BV can significantly impart an antiarthritic response mediated by inhibition of inflammation mediators similar to nonsteroidal anti-inflammatory drugs. 13 BV exerts positive effects on melanocyte proliferation, melanogenesis, dendricity, and migration through complex signaling pathways in vitro. 14 BV also has been shown to have a radioprotective effect against basal and oxidative DNA damage. 15 However, the mechanisms underlying these events are poorly understood.
The purpose of this investigation is to test the hypothesis that BV protects against TGF-β1-induced hepatocyte cell death via inhibition of apoptosis signaling. We focused our analysis on the mechanism associated with TGF-β1-induced apoptosis in hepatocytes and the effect of BV on this mechanism. Our results indicate the existence of multiple survival pathways in cells and suggest that at least one transcription factor involved in modulating the apoptotic signaling cascade is modulated by BV.
Materials and Methods
Cell Culture
AML12 murine hepatocytes (America Tissue Culture Collection, ATCC CRL-2254) were cultured in a 1:1 mixture of Dulbecco’s modified Eagle’s medium/Ham’s F-12 medium (Gibco BRL, Gaithersburg, Maryland) containing 5 μg/mL insulin, 5 μg/mL transferrin, and 5 μg/mL selenium (ITS Premix, Sigma, St. Louis, Missouri), 40 ng/mL dexamethasone (Sigma), and 10% fetal bovine serum (Gibco BRL). Cell cultures were maintained at 37°C in a humidified atmosphere of 5% CO2.
Induction of Hepatocyte Apoptosis and Treatment With BV
TGF-β1 treatment previously has been demonstrated to induce cell death, specifically apoptosis. 16 Therefore, we examined hepatocytes treated with only TGF-β1 at a concentration of 10 ng/mL for 9 hours and hepatocytes treated with TGF-β1 for 30 minutes with additional administration of BV (Sigma) at a concentration of 1, 10, and 100 ng/mL for 9 hours.
Cytotoxicity Assay
To determine the effects of BV in hepatocyte apoptosis, we evaluated cell viability by the 3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyltetrazolium bromide (MTT) reduction assay. In brief, cells (1 × 105 cells/mL) were seeded in 96-well plates and treated with TGF-β1 and BV. The control was added to saline of equal volume. Cells were incubated for 24 hours, and the media were removed and the cells incubated with 0.5 mg/mL MTT solution. After being incubated for 4 hours at 37°C and 5% CO2, the supernatant was removed, and DMSO with ethanol was added. Cells were incubated at 37°C for 30 minutes and measured by a microplate reader at 540 nm. The percentage of cell survival was calculated by taking the optical density of cells given a particular treatment, dividing that number by the optical density for the untreated control cells, and then multiplying by 100.
DNA Fragmentation Assay
Apoptotic cells were detected as described by Lindenboim et al 17 with minor modifications. The cells were rinsed with phosphate-buffered saline (PBS) twice and lysed on ice for 30 minutes in 10 mM Tris-Cl (pH 8), 25 mM EDTA (pH 8), and 0.25% Triton X-100. After centrifugation at 8000 g for 15 minutes, the supernatant was incubated with RNase at 37°C for 1 hour and then with proteinase K at 56°C overnight. The contents were extracted sequentially with phenol, phenol/chloroform (1:1), and chloroform. The DNA in aqueous phase was precipitated and analyzed by 2% agarose gel electrophoresis. Gel was visualized and photographed under transmitted ultraviolet (UV) light.
Assessment of Apoptosis
Morphological evaluation was performed as described. 16 Briefly, medium was gently removed after treatment to prevent detachment of cells. Cells were harvested, washed in ice-cold PBS, fixed with 3.7% paraformaldehyde, and then permeabilized with 0.2% saponin in PBS. Fixed cells were washed with PBS, and the nuclei were stained with a Hoechst 33324 (2 μg/mL) solution. The blue fluorescence was visualized by using a fluorescence microscopy (excitation: 330-380 nm, emission: 460 nm). Apoptosis was evaluated by morphological criteria (i.e., condensed chromatin and fragmented nuclei), and the number of cells with apoptotic nuclei was determined within a field of view at a magnification of ×400. A total of 10 randomly prechosen fields were counted per well, and the number of apoptotic cells was averaged to obtain an apoptotic index. The percentage of apoptotic cells was calculated by taking the optical density of cells given a particular treatment, dividing that number by the optical density for the treated control cells, and then multiplying by 100.
Preparation of Protein Fractions and Immunoblotting
To prepare whole-cell lysates, we placed the cells on ice, aspirated the medium, and rinsed the cells once with ice-cold PBS. The cells were then scraped in 1 mL of lysis buffer containing 20 mM HEPES, 10 mM KCl (pH 7.5), 1.9 mM MgCl2, 1 mM EGTA, 1 mM EDTA, and protease inhibitor mixture (Complete Protease Inhibitor Mixture; Roche Diagnostics, Mannheim, Germany); transferred to microcentrifuge tubes; and incubated for 30 minutes at 4°C. Whole-cell lysates were centrifuged at 8000 g for 20 minutes at 4°C to remove insoluble material. For subcellular fractions, cultured cells were collected by scraping in homogenization buffer (70 mM sucrose, 220 mM mannitol, 3 mM EDTA, 5 mM MOPS, pH 7.4) and transferred to an ice-cold homogenizer. The crude homogenate was centrifuged at 8000 g for 20 minutes at 4°C to remove debris. The postnuclear supernatant was centrifuged at 7000 g for 10 minutes at 4°C to pellet mitochondria from cytosol. The supernatant from this step is the cytosolic fraction, and the pellet is the mitochondrial fraction. The mitochondrial pellet was rinsed in wash buffer (5 mM MOPS, 100 mM KCl, pH 7.4), collected by centrifugation, and resuspended in lysis buffer (30 mM Tris-HCl [pH 7.5], 150 mM NaCl, 10% glycerol, 1 mM phenylmethylsulfonyl fluoride, 1% Triton X-100, 1% Nonidet P-40, and a Complete Protease Inhibitor Tablet [Roche Diagnostics]). After incubating for 30 minutes at 4°C with gentle shaking, the mitochondrial lysate was centrifuged at 13 000 g for 10 minutes at 4°C to remove insoluble protein. Protein concentration was determined by the Bio-Rad Bradford kit (Bio-Rad Laboratories, Hercules, California). The mitochondrial, cytosolic, and whole-cell lysates were subjected to immunoblot analysis as described below. Proteins were electrophoresed in sodium dodecyl sulfate (SDS)–polyacrylamide gel and transferred onto nitrocellulose membranes (Millipore, Billerica, Massachusetts). Each protein was used to confirm equal sample loading. The membranes were blocked with 5% skim milk in Tris-buffered saline (TBS) containing 0.1% Tween-20 (TBS-T) and subsequently incubated with primary antibodies. The primary antibodies used in this study were the following: anti–caspase-3 and -9, anti-bax, anti–cytochrome c, and anti–poly ADP-ribose polymerase (PARP) antibodies were purchased from Cell Signaling Technology (Beverly, Massachusetts), and anti-bcl-2 and anti-β-actin antibodies were from Santa Cruz Biotechnology (Santa Cruz, California). Antibody binding was detected using horseradish peroxidase (HRPO)–conjugated secondary antibody and the enhanced chemiluminescence (ECL) detection system (Amersham, Piscataway, New Jersey). As loading control for mitochondrial, cytosolic, and whole-cell lysates, β-actin was used to confirm equal sample loading. 18-20 Membranes were then exposed to Kodak X-OMAT film. Signal intensity was quantified by image analyzer (Las 3000, Fuji, Tokyo, Japan).
Statistical Analysis
The data are presented as means ± SD of 3 to 5 independent experiments. Data were analyzed using Student t tests (SAS Institute, Cary, North Carolina). P values less than .05 were considered statistically significantly different.
Results
Effect of BV in Hepatocytes and DNA Fragmentation Induced by TGF-β1
The MTT assay was carried out to determine the optimal treatment time of TGF-β1. At 9 hours after treatment, the viability of cells treated with TGF-β1 decreased by about 30% as compared to normal control cells. There were no significant changes in cell viability when the hepatocytes were incubated with BV alone (Figure 1A). Adding 10 ng/mL of BV resulted in a significant increase in the viability of TGF-β1-induced hepatocytes compared to those without addition of BV. To determine whether cells were killed via apoptotic processes, we examined for internucleosomal DNA fragmentation after TGF-β1 treatment. DNA fragmentation was observed in TGF-β1-induced hepatocytes but was decreased in the TGF-β1-induced hepatocytes with addition of 10 ng/mL of BV (Figure 1B). From this result, we confirmed that BV protected TGF-β1-induced apoptosis.
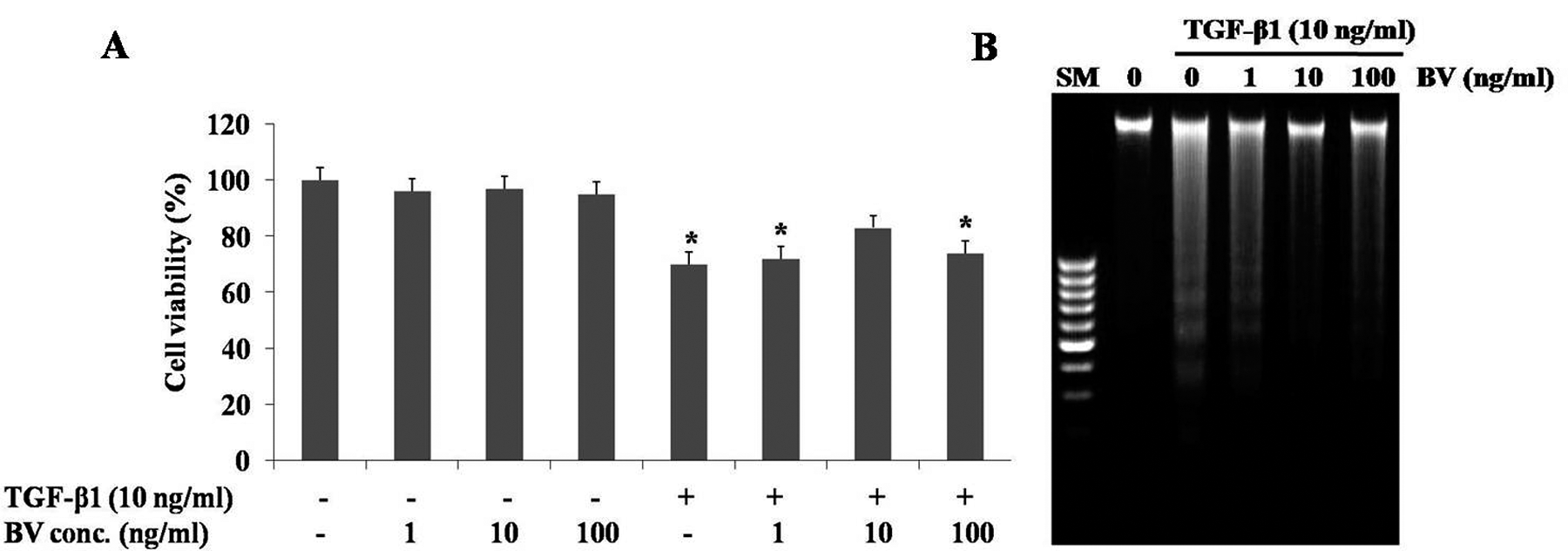
Effects of different concentrations of BV on hepatocytes. (A) The 3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyltetrazolium bromide (MTT) reduction was used to indicate cell viability. (B) Electrophoretic examination of genomic DNA in TGF-β1-induced hepatocytes. Hepatocytes were treated with 1, 10, and 100 ng/mL BV after TGF-β1 treatment. SM, standard marker; TGF-β1, transforming growth factor–β1; BV, bee venom. Results are expressed as mean ± SD of 3 to 5 independent determinations. *P < .05 compared to normal control.
Morphological Changes After Treatment With BV in TGF-β1-Induced Hepatocytes
To observe the effect of BV on cell morphology, we also observed cells using fluorescence microscopy following treatment with Hoechst 33342, which specifically stained the nuclei. BV (10 ng/mL) significantly decreased apoptosis-induced hepatocytes by treatment with TGF-β1 for 9 hours (Figure 2). These differences in morphological characteristics suggest that BV protects against apoptotic cell death induced by TGF-β1.
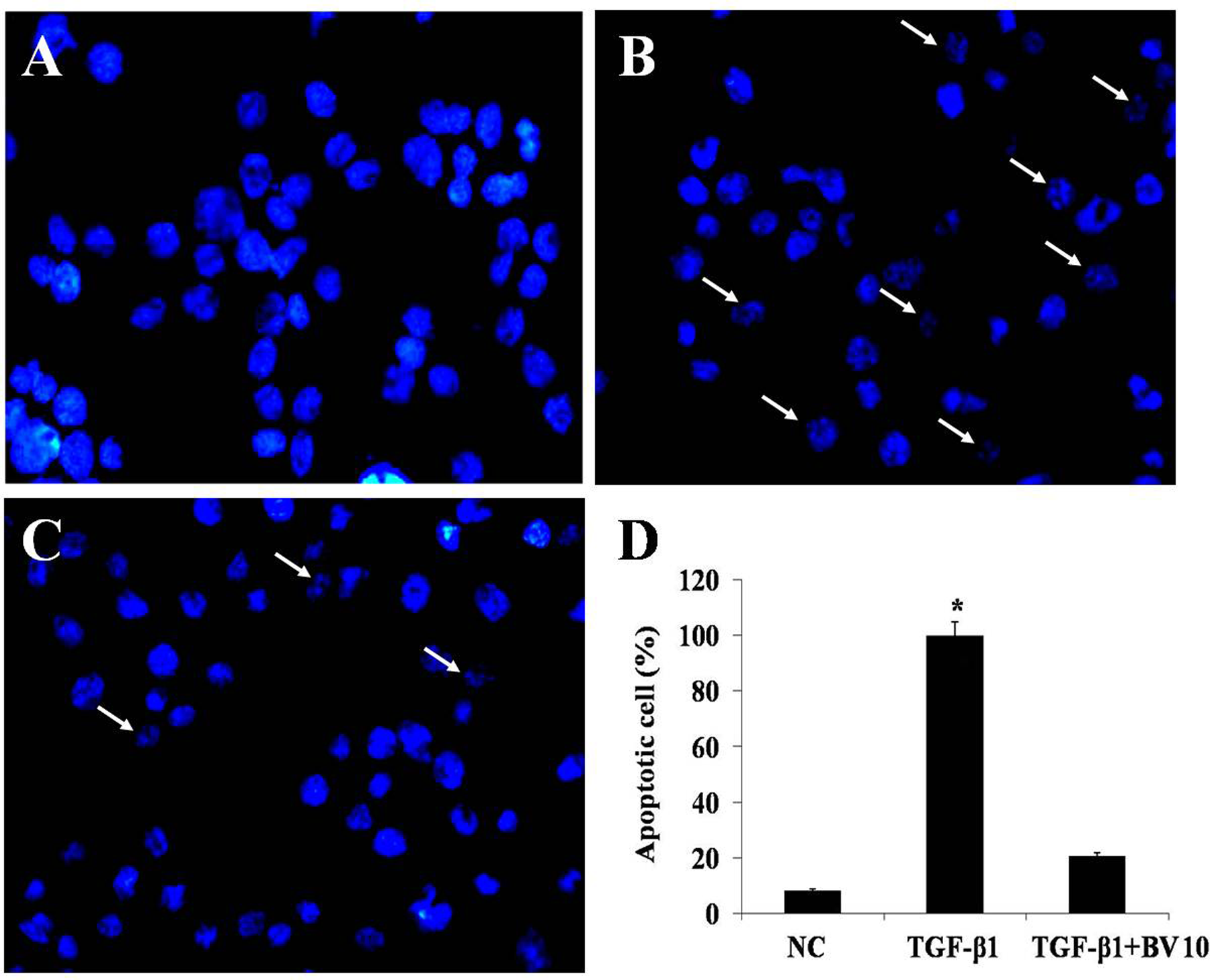
Morphology of cells incubated with or without BV in TGF-β1-treated hepatocytes. (A) Fluorescent microscopy showing nuclei by Hoechst 33342 stain blue color. NC observed had typical round nuclei and stained with blue color. (B) Hepatocytes treated with TGF-β1 showed fragmented typical nuclei. (C) TGF-β1-treated hepatocytes with addition of BV 10 ng/mL had decreased fragmented typical nuclei compared with TGF-β1-treated hepatocytes. Arrows indicate cells with condensed chromatin. (D) The data are expressed as the percentage of apoptotic cells. BV, bee venom; NC, normal control; TGF-β1, transforming growth factor–β1-treated hepatocytes; TGF-β1 + BV, TGF-β1-treated hepatocytes with addition of BV 10 ng/mL. *P < .05 compared to NC; n = 3.
Activation of bcl-2 Family of Proteins During TGF-β1-Induced Hepatocytes
To characterize the mechanism of TGF-β1-mediated apoptosis, we compared the steady-state levels of several bcl-2 family proteins by immunoblot analysis. We detected no change in the expression of bcl-2 and bax with BV treatment alone (data not shown). The expression of antiapoptotic proteins bcl-2 was decreased in the mitochondrial fraction, and the expression of proapoptotic protein bax was increased in the total lysates of TGF-β1-treated hepatocytes for 6 hours (Figure 3). The expression of bcl-2 in the TGF-β1-treated hepatocytes with addition of 10 ng/mL of BV was significantly increased, whereas the expression of bax was decreased. Cells undergoing apoptosis release their mitochondrial cytochrome c into the cytosol. The cytosol from untreated control cells contained negligible amounts of cytochrome c. However, cytochrome c accumulated in the cytosol of cells exposed to TGF-β1 (Figure 4). This TGF-β1-induced release of cytochrome c was significantly inhibited by treatment with BV (10 ng/mL).
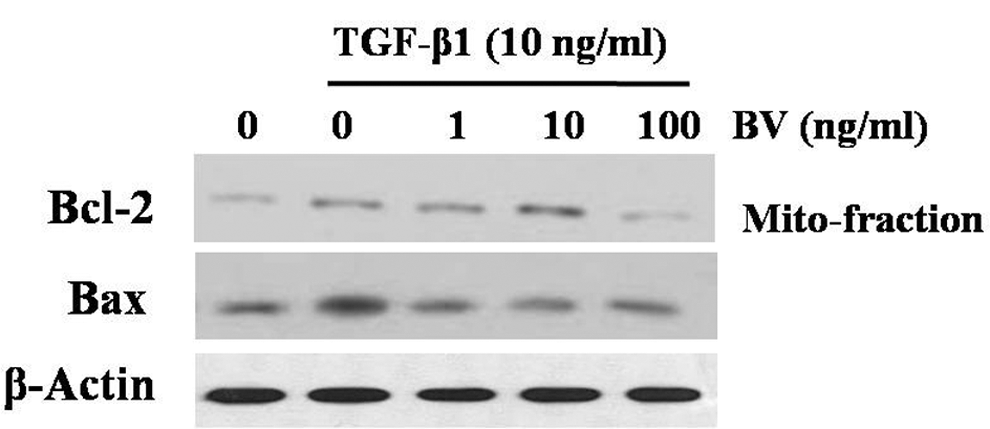
Effect of BV on the expression of bcl-2 and bax in TGF-β1-induced hepatocytes. Western blot of mitochondria fractions of bcl-2 and total lysates of bax were examined. The expression of bcl-2 was decreased in the TGF-β1-induced cells, whereas that of bax was increased. BV protected the decrease in bcl-2 as well as the increase of bax in the TGF-β1-induced hepatocytes. As loading control, β-actin was used to confirm equal sample loading. BV, bee venom; TGF-β1, transforming growth factor–β1.
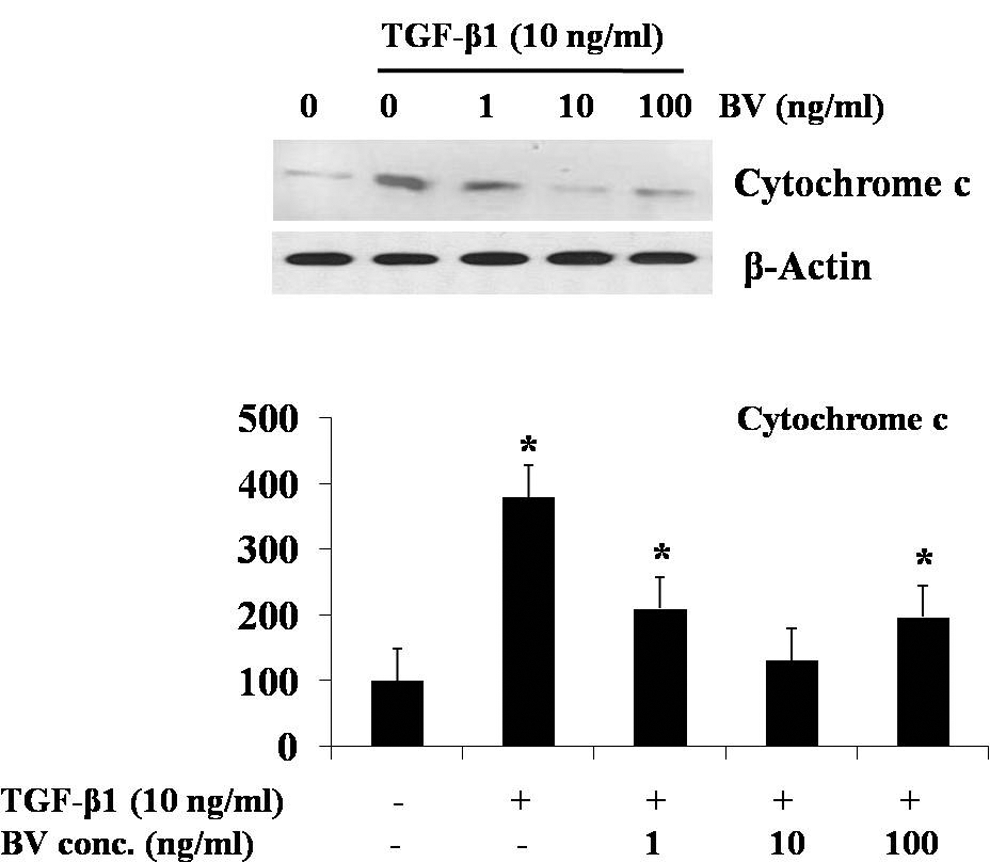
BV attenuates mitochondrial cytochrome c release. Western blot of cytosol fractions of cytochrome c was examined. Mitochondrial cytochrome c release increased in the TGF-β1-induced cells but was decreased with additional BV 10-ng/mL treatment. BV, bee venom; TGF-β1, transforming growth factor–β1. *P < .05 compared to normal control; n = 3.
BV Protects Cleavage of Caspase and PARP in TGF-β1-Induced Hepatocytes
Caspase activation during TGF-β1-induced apoptosis was assessed by immunoblot analysis of proteins from the cells. TGF-β1-treated hepatocytes showed increased cleavage of caspase-9 (37 kDa) and caspase-3 (17 kDa; Figure 5). The addition of BV (10 ng/mL) inhibited the proteolytic fragmentation of the same caspases in TGF-β1-treated cells. We detected no change in the expression of caspase-9 and -3 after BV treatment, and no cleaved products of these proteins were seen. PARP is known to be a target of caspase protease activity associated with apoptosis. During apoptosis, PARP is cleaved by activated caspase-3. The cleavage of PARP (89 kDa) was decreased because caspase-3 activation was inhibited by the 10 ng/mL of BV, and BV did not have any effects on caspase activity alone. The appearance of PARP cleavage coincided with the timing of procaspase processing (Figure 6). These results demonstrate that BV protects TGF-β1-induced hepatocytes at the site upstream of caspase-3 activation.
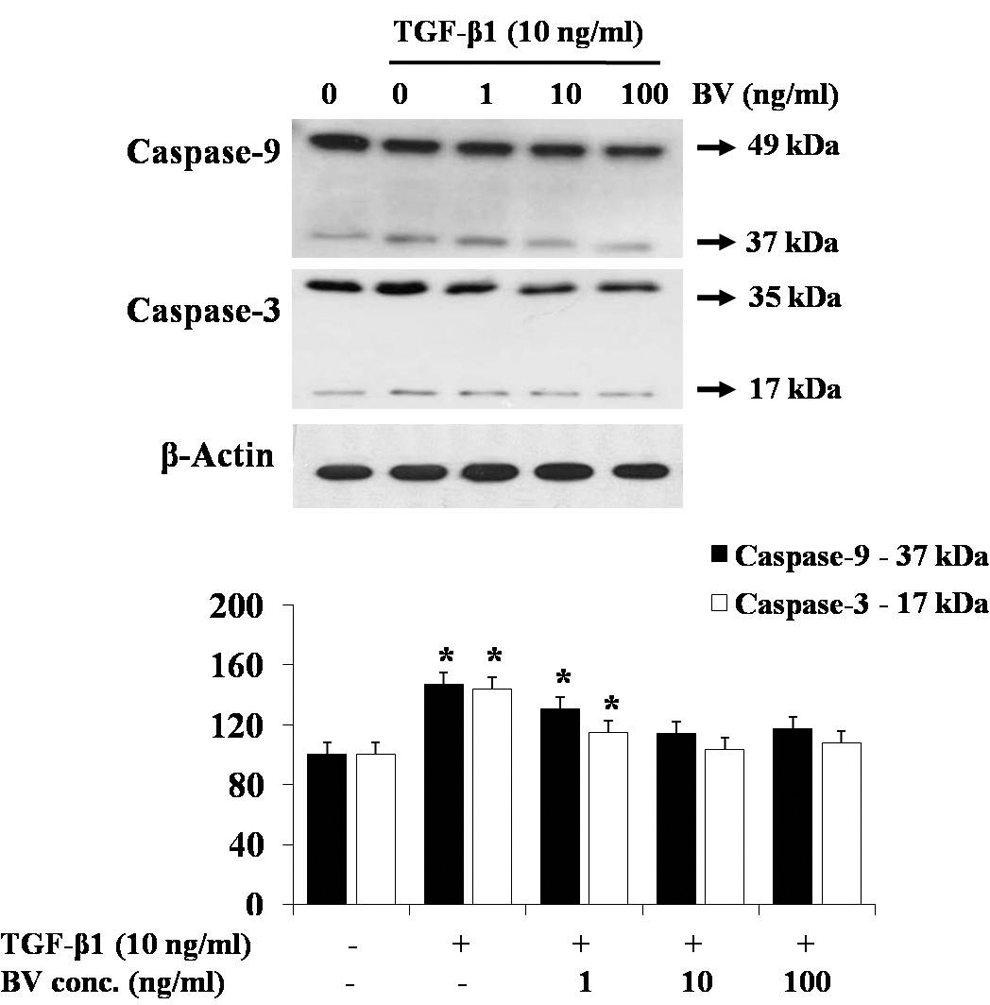
Effect of BV on TGF-β1-induced caspase in hepatocytes. Western blot of cytosol fractions of caspase-9 and -3 was examined; 10 ng/mL of BV treatment attenuated caspase-9 (37 kDa) and caspase-3 (17 kDa) cleavage in TGF-β1-induced hepatocytes. BV, bee venom; TGF-β1, transforming growth factor–β1. *P < .05 compared to normal control; n = 3.
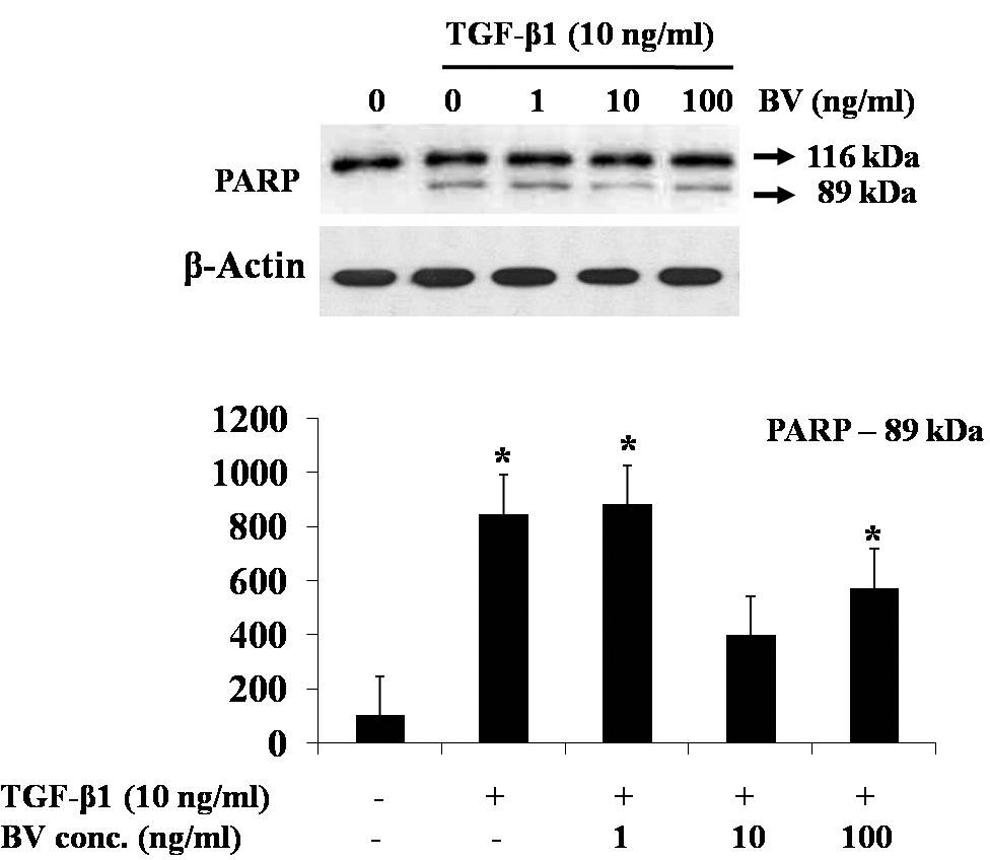
Effect of BV on TGF-β1-induced poly ADP-ribose polymerase (PARP) activation in hepatocytes. Total lysates from hepatocytes were examined by Western blot. The expression of cleavage PARP (89 kDa) was increased in the TGF-β1-induced hepatocytes but was decreased with additional BV 10-ng/mL treatment. BV, bee venom; TGF-β1, transforming growth factor–β1. *P < .05 compared to normal control; n = 3.
Discussion
TGF-β1 is the prototype of a widespread and evolutionarily conserved superfamily of cytokines that regulate a broad spectrum of cellular responses, including proliferation, differentiation, and apoptosis. 21 TGF-β1 has been shown to elicit apoptotic cell death in a variety of cell types, including hepatocytes. Moreover, TGF-β1-induced apoptosis plays important roles in the selective elimination of damaged or abnormal cells from various normal tissues. 22 TGF-β1 has also been implicated in controlling liver size, and an intravenous injection of TGF-β1 induces atrophy and apoptosis in the normal and regressing liver. 23 Thus, there is very convincing evidence that TGF-β1 is a potent hepatic apoptogen, although the mechanisms by which it induces cell death are still poorly understood. Some reports have suggested that some members of the bcl-2 family of proteins and caspases involved in the apoptotic effectors system are activated in hepatocytes undergoing TGF-β1-induced apoptosis. 24-26
BV contains several peptides, including melittin, apamin, adolapin, mast cell degranulating peptides, enzyme, biologically activity amines, and nonpeptide components. 27 Each BV component has been studied to elucidate their anti-inflammatory effects. 28 The medicinal use of substances derived from BV has been practiced since ancient times. 29 Over the past few years, a number of studies were published on the beneficial role of BV, including anti-inflammatory, 29 antimutagenic, 30 antinociceptive, 31 and anticancer activities. 32 In addition, recent studies reported several effects of BV such as radioprotection of lymphocytes, induction of apoptosis and necrosis, and effects on proliferation, cytotoxicity, and growth inhibition of different types of cancer cells. 15,33-38 BV was proven to exert its anti-inflammatory properties by inhibiting iNOS and COX-2 expression possibly through the suppression of NF-κB and MARK activities. 39 After 70% of the rat liver volume has been resected, the stimulatory effect of a single administration of BV on hepatocyte proliferation is revealed. 40 BV inhibits the production of tumor necrosis factor–α (TNF-α) in lipopolysaccharide (LPS)–stimulated microglia, and this inhibition is closely associated with the suppression of nitric oxide (NO). 38 BV contains a variety of proteins that could act as allergens causing anaphylactic shock as well as local inflammation. 41 However, it has also been demonstrated that long-term BV treatment induces T cell hyporesponsiveness to these allergens and also modulates cytokine secretion in both humans. 42 When human blood cells were treated with BV (more than 20 μg/mL), hemolysis occurred due to the enhanced permeability of erythrocytes caused by melittin. 43 Garaj-Vrhovac and Gajski 44 reported that a high concentration of BV (100 μg/mL) can lead to cellular instability in human lymphocytes. In contrast, low concentrations of BV are demonstrated to have a radioprotective effect against basal and oxidative DNA damage. In addition, low concentrations of BV are not genotoxic and do not produce oxidative damage. 15
Our results provide strong evidence suggesting that BV can affect a critical downstream component in the apoptotic pathway induced by TGF-β1 in hepatocytes. Treatment with BV resulted in a significant reduction in the morphological characteristics of apoptosis. BV (10 ng/mL) protected TGF-β1-treated hepatocytes from apoptosis. Assessment of cell viability using the MTT assay, morphological changes, DNA fragmentation, and immunoblot analysis revealed that BV produced a dose-dependent antiapoptotic effect on TGF-β1-treated hepatocytes, and this effect was apparent at a low concentration of BV (10 ng/mL). PARP is a posttranslational modification of proteins that, in eukaryotic cells, plays a crucial role in DNA repair and replication, transcription, and cell death and represents a cellular emergency reaction. 45 It has also been reported that PARP is cleaved before or concomitantly with the degradation of nuclear DNA into nucleosomal fragments, and the presence of PARP inhibitors delays or prevents apoptosis. PARP cleavage was also widely used as a characteristic phenotype of cell apoptosis and an indirect evidence for the activation of caspase-3 or -7. 46 Our results demonstrated that cleavage of PARP was decreased because caspase-3 and -9 cleavages were inhibited by 10 ng/mL BV.
In summary, we demonstrated that BV protects hepatocytes against TGF-β1-induced apoptosis. BV showed antiapoptotic effects that were associated with a decrease in the levels of proapoptotic proteins in the cytoplasm and activation of proteins related to apoptosis in the mitochondria. Our results demonstrated that 10 ng/mL BV protects TGF-β1-treated hepatocyte injury by inhibiting the activation of bcl-2 family of proteins, caspases, and PARP cleavage. This suggests that BV can serve to protect cells and organs against TGF-β1-mediated injury.
Footnotes
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors disclosed receipt of the following financial support for research and/or authorship of this article: This work was supported by a grant (Code#2007030103400101) from the BioGreen 21 Program, Rural Development Administration, Republic of Korea.
