Abstract
The neurobehavioral effects of inhaled cyclohexane in rats and humans are investigated to define relationships between internal doses and acute central nervous system effects. Rats are exposed for 3 consecutive days at target concentrations of 0, 1.4, 8, and 28 g/m3, 8 h/d. Measurements include standardized observational measures, spontaneous motor activity assessments, and learned visual discrimination performance. Cyclohexane concentrations in blood and brain are measured to assess internal exposure. Human volunteers are exposed for 4 hours to 86 or 860 mg/m3 in 2 test sessions. Neurobehavioral effects are measured using a computerized neurobehavioral test battery. In rats, there are slight reductions in psychomotor speed in the high-exposure group but minimal central nervous system effects. In humans, there are no significant treatment-related effects at the levels tested.
The central nervous system (CNS) is a primary target for the acute effects of organic solvents, which are characterized, both in animals and in humans, by reversible signs of CNS depression that include slowing in behavioral performance, dizziness, drowsiness to anesthesia, and even death (eg, Ridgway et al 1 ). Occupational exposure limits (OELs) for organic solvents are designed to avoid signs of acute symptoms, and recommendations for solvent exposure levels in the workplace often cite CNS depressant effects as one basis for limiting exposure. 2,3 At levels that are well above current occupational exposure recommendations, exposures produce acute CNS effects including nausea, lack of coordination, and other signs of intoxication. At lower levels of exposure, subtle changes in behavioral functioning can occur, particularly those related to information processing and psychomotor function. 4-7 OELs that are based on acute CNS depression usually provide an adequate measure of protection for other, more profound manifestations of CNS effects as well as other nervous system effects. 8
Over the years, there has been significant progress in the development of methods for evaluating the effects of hydrocarbon solvents on the animal and human nervous system. 6,9-15 Automated tests designed for use with exposed workers and parallel tests for evaluating similar functions in exposed animals are now available. The studies reported here were part of a larger effort (see McKee et al 16 for more description) to determine whether acute CNS effects in animals could be used as a basis for occupational exposure recommendations.
Typically, the determination of a safe exposure level in humans is based on dose-response data from animal experiments conducted at levels much higher than human exposures. Joint neurobehavioral/toxicokinetic studies involving inhalation exposure of rats and humans are often needed to evaluate the neurobehavioral effects of hydrocarbon solvents and to establish physiologically based toxicokinetic (PBTK) modeling approaches for extrapolating animal test data to humans. 16
The specific objectives of the present studies were to assess the acute CNS effects of cyclohexane in animals and humans and, to the extent possible, determine internal doses at which adverse effects might occur. A battery of tests, including standardized observational measures, spontaneous motor activity assessment, and learned visual discrimination performance, were used in rats to examine the CNS effects of short-term exposure to cyclohexane. Blood and brain concentrations of cyclohexane were measured at various times to assess internal doses. In a volunteer study, 12 subjects were exposed to cyclohexane at 860 mg/m3, a level approximating occupational exposure standards in the Netherlands (250 parts per million [ppm]), and neurobehavioral effects were measured using a computerized neurobehavioral test battery. Blood samples were taken from the volunteers at the end of the treatment period. This design allows for comparisons of the animal and human data on an internal exposure basis. Assuming similar blood concentration–effect relationships for acute CNS effects in rats and humans, it should be possible to use toxicokinetic modeling to predict effect levels in humans from rodent data.
Materials and Methods
Test Substance
Cyclohexane (CAS no. 110-82-7) was purchased from 2 commercial sources (T. Baker, Deventer, The Netherlands, and E. Merck Nederland, Amsterdam, The Netherlands). In both cases the purity was given as greater than 99%. Other chemical characteristics included density = 0.78 kg/L; boiling point = 80°C at 105 Pa; ignition temperature = 260°C; explosive limits = 1.2%:8.3% (vol/vol); saturating concentration = 357 g/m3. The relationship between mg/m3 and ppm was 1 mg/m3 = 0.29 ppm.
Neurobehavioral tests in animals and humans
Attempts were made to study similar neurobehavioral effects as much as possible in both rats and humans. Test batteries used in animal and human studies contained many items that evaluated analogous nervous system functions (Table 1). In addition, a number of tests (excitation and sensorimotor responses in rats; mood and affect and perceptual coding in humans) were conducted that lack clear equivalents in the studies with the other species, although some overlap with other functions may be assumed.
Tests Performed in Animal and Human Volunteer Studies, With Dependent Variables and Assessed Nervous System Function
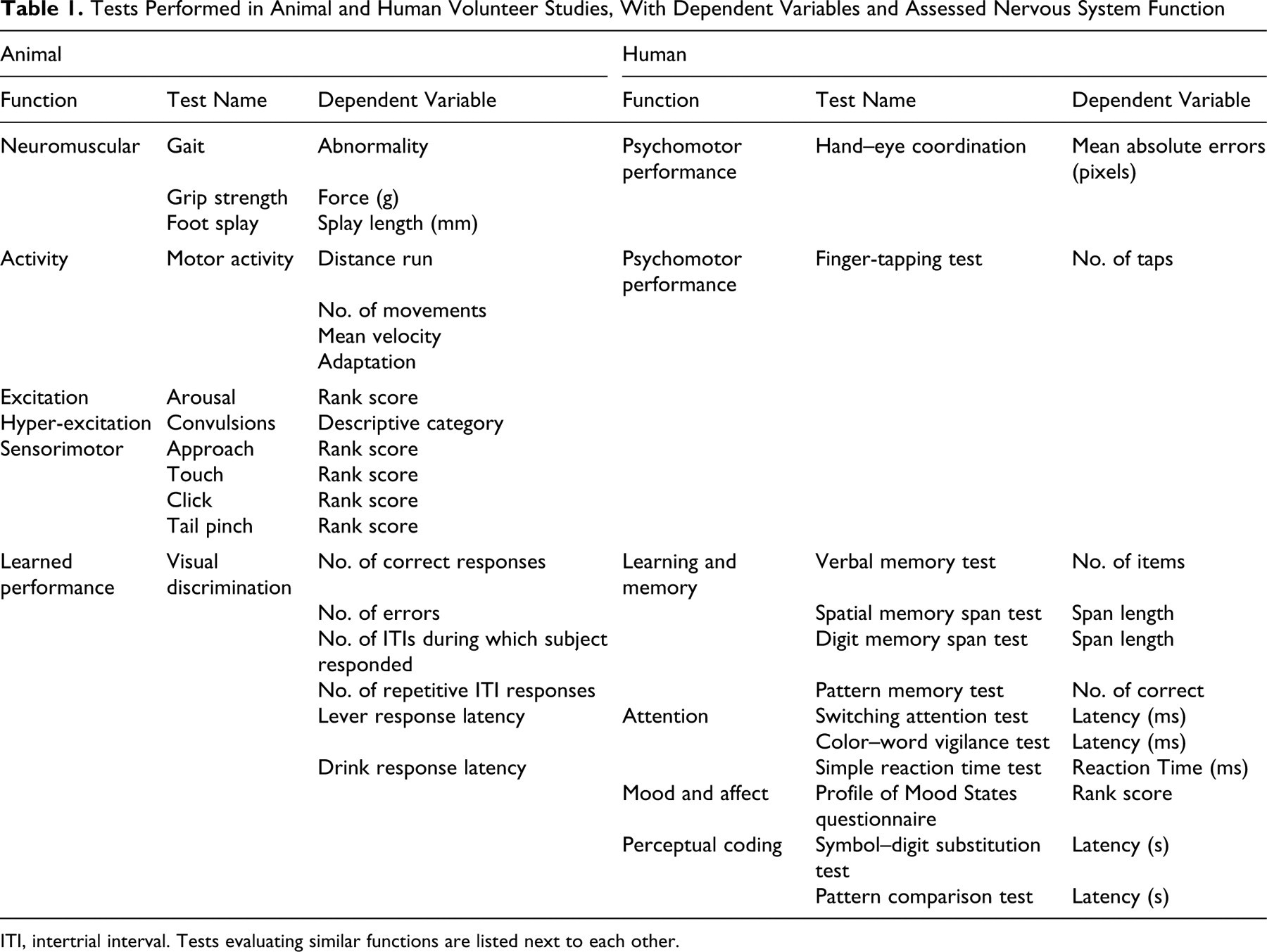
ITI, intertrial interval. Tests evaluating similar functions are listed next to each other.
Animal Study
Detailed descriptions of testing conditions, generation of test atmosphere, behavioral testing, and determination of blood and brain concentrations are provided in previous articles. 16,17
Animals and housing
One hundred fifty-nine adult male Wistar-derived WAG/RijCR/BR rats were obtained from Charles River Wiga (Sulzfeld, Germany). Thirty-two (n = 8 rats per exposure concentration) were used for testing of visual discrimination performance, 32 (n = 8 rats per exposure concentration) were used for functional observations and motor activity assessments, and 95 (n = 3 or 4 rats per exposure concentration per time point) were used for determination of blood and brain levels of cyclohexane. Animals were approximately 14 weeks of age at the time of exposure and testing. Body weights ranged from 222.2 to 267.8 g (functional observations), 180.6 to 391.7 g (visual discrimination, water deprived), and 215.7 to 274.7 g (blood and brain concentrations). Note that the majority of the blood and brain data were for a pharmacokinetic study that is being reported separately. 18
Rats were housed in suspended stainless steel cages under standard laboratory conditions. A 12-hour light/dark cycle was maintained with the light phase commencing at 7:30
Viability checks, clinical signs, and body weights
All rats were checked at least once daily for health and viability. Clinical signs were taken and body weights were recorded prior to randomization and on days of testing and treatment in the functional observation study; at least once weekly during operant training, prior to randomization, and after testing was completed on each exposure day in the visual discrimination test; and prior to randomization and prior to sacrifice in the blood and brain level determination study.
Exposure conditions
Groups of rats were exposed to cyclohexane at target concentrations of 0 g/m3 (control group), 1.4 g/m3 (~400 ppm), 8 g/m3 (~2300 ppm), and 28 g/m3 (~8000 ppm) in modified H1000 inhalation chambers (Hazleton Systems, Aberdeen, MD). Animals were exposed to the test atmosphere 8 h/d for 3 consecutive days. To create the test atmosphere, cyclohexane was pumped through an oven at 400°C via stainless steel tubing. The vapor was carried by an airstream from a compressed air source and added to the main airflow system. During exposures, samples were automatically drawn from each exposure chamber hourly and analyzed by gas chromatography. Gas chromatograph readings were calibrated by measuring the contents of Tedlar bags filled with air or 1.04, 4.00, 8.00, 16.85, or 31.20 g/m3 cyclohexane.
Functional observational battery
Neurobehavioral functioning was evaluated using selected measures from a standardized functional observational battery (FOB) and a motor activity assessment protocol similar to that used in the WHO/IPCS Collaborative Study on Neurotoxicity Assessment. 19,20 Body weight and rectal temperature were also measured. Functional observations required about 5 minutes per animal to complete and were carried out 6 days prior to the start of exposure and immediately after the first and third exposure periods.
Automated locomotor activity assessment
Changes in spontaneous motor activity were assessed after FOB testing of each animal using an automated quantitative microprocessor-based video image analysis system (Ethovision, Noldus Information Technology, Wageningen, The Netherlands). Motor activity testing started immediately after each round of FOB testing was completed, so animals were placed in the motor activity testing device 25 to 40 minutes after removal from the exposure chambers.
Visual discrimination performance
Effects of cyclohexane on cognitive performance were evaluated by a discrete-trial 2-choice visual discrimination task using a water reward as the reinforcement. The apparatus consisted of 16 operant chambers (32 × 30 × 28 cm [l × w × h]) and programming and recording equipment programmed with the MedState notation system (Med Associates, St Albans, Vt).
Test sessions consisted of 100 trials or 60 minutes, whichever came first, and were conducted at approximately the same time each day. Well-trained rats were tested on the day prior to exposure and on each day of exposure immediately after the exposure period. A postexposure test was performed the day after the last exposure period to evaluate the persistence of effects.
The FOB measurements were initiated immediately after the first and third exposure periods. This took approximately 5 minutes per rat or approximately 20 minutes in total. Motor activity measurements were then initiated (approximately 25-40 minutes after exposure) and required 30 minutes per rat. The visual discrimination tasks were initiated approximately 55 to 70 minutes after exposure and required 60 minutes per rat.
Determination of blood and brain concentrations
To determine the blood and brain concentrations of cyclohexane at which behavioral effects occurred, groups of rats were killed immediately after a single 8-hour exposure period and after 8-hour exposure periods on 3 consecutive days. Trunk blood was collected in heparinized glass tubes and stored at –20°C until analysis. Immediately after euthanasia, brains were removed from the skulls. After weighing, the brains were homogenized in chilled isotonic saline (dilution 1:4 wt/vol) and stored at –20°C until analysis. Cyclohexane concentrations in rat blood and brain homogenates were determined by gas chromatography (Carlo Erba 4160 gas chromatograph, Carlo Erba, Milan, Italy) using headspace analysis and flame ionization detection. Calibration samples were prepared in physiological saline. The curves were linear from 10 to 20 000 ng per vial (50-100 000 ng/mL matrix). The lower quantification limit for the blood and brain samples was 30 ng/mL. At these concentrations recoveries ranged from approximately 86% to 90%.
Statistical Analysis
Functional observational battery and motor activity assessment
Effects of treatment on continuous variables were analyzed by repeated-measures analysis of variance (ANOVA). If a significant effect of treatment or a significant treatment-by-time point interaction was indicated, ANOVA was conducted at each time point. Group comparisons were made using Dunnett’s multiple comparison tests. Motor activity variables (distance run, number of movements, mean velocity) and habituation of activity were analyzed by 3-way repeated-measures ANOVA, using treatment as a between-subjects factor and test time point and time blocks within each session as within-subjects factors. Each session consisted of 5 time blocks of 6 minutes each. Rank data (gait score, arousal, approach response, touch response, click response, tail-pinch response) were analyzed by Kruskal-Wallis 1-way ANOVA on each test day followed by multiple comparisons when differences were significant.
Visual discrimination performance
For all variables, effects of treatment were analyzed by univariate repeated-measures ANOVA of the data recorded during the 3-day exposure period, using treatment as a between-subjects factor and test time point as a within-subjects factor. Huynh-Feldt adjustment of P values of the repeated-measures factor was applied in case the assumption of sphericity of observations was violated. When a significant treatment-related effect or treatment–by–time point interaction was identified, pair-wise group comparisons were carried out to determine which treated group differed significantly from the control group. Persistence of effects was evaluated by univariate ANOVA of postexposure data.
All analyses in animal studies were conducted using the BMDP statistical software package 21 with a P < .05 significance level.
Human Volunteer Study
Subjects
Testing was conducted in 12 healthy male subjects (20-40 years, 66-95 kg) with no history of occupational exposure to organic solvents, using a within-subjects study design. Subjects were exposed for 4 hours to either 86 mg/m3 (25 ppm) or 860 mg/m3 (250 ppm) cyclohexane, in 2 test sessions spaced 7 days apart. All subjects were exposed to both concentrations and therefore served as their own controls. The principles of informed consent in the current revision of the Declaration of Helsinki 22 were implemented in this study. The study protocol was reviewed and approved by TNO’s Institutional Review Board. Further details of the exposure conditions used in this study may be found in Hissink et al. 18
Exposure conditions
An exposure level of 860 mg/m3, the 8-hour threshold limit value/time-weighted average in The Netherlands 23 at the time of testing, was selected. The threshold limit value of 860 mg/m3 was intended to minimize the potential for either narcotic effects or irritation. A concentration of 86 mg/m3 cyclohexane, rather than clean air, was chosen as a reference in an effort to control for the odor associated with cyclohexane. Awareness of odor could provide the subjects with a cue as to whether they were being exposed and thereby potentially confound the double-blinded nature of the study design. Assignment to treatment sequence was by random selection, and the sequence of test conditions was counterbalanced.
The test atmosphere was generated by total evaporation at an elevated temperature of a constant flow of liquid cyclohexane in a constant flow of fresh air. During the experiments, the concentration of cyclohexane vapor was measured by infrared absorption of 3.49 μm using a path length of 3.75 m (Miran 1A, Foxboro, Mass). The Miran was calibrated at the beginning of each exposure week. Tedlar bags were filled with 44.1 L of dry nitrogen gas into which 50.0 μL of liquid cyclohexane was allowed to evaporate, producing an 860-mg/m3 concentration. Similarly, two 86-mg/m3 bags, two 690-mg/m3 bags, and one 1035-mg/m3 bag were prepared and used to construct a calibration curve. The values recorded at 15-minute intervals were converted to concentrations and then averaged for each session.
Testing
Tests included assessments of learning and memory, attention, mood and affect, and perceptual coding. Performance tests were also conducted. On each experimental day, behavioral testing was conducted 90 minutes before exposure; 45 minutes and 165 minutes after the start of exposure; and 90 minutes after the 4-hour exposure period. These testing time points are referred to as the pretesting, 1-hour and 3-hour, and posttesting time points. In addition, the simple reaction time test was administered as a stand-alone test beginning 15 and 135 minutes after the start of the exposure. These are subsequently referred to as the 0.25-hour and 2.25-hour testing time points. The tests are summarized below. More detailed descriptions of the testing procedures are provided by Lammers et al. 17
Vocabulary test
The vocabulary test was administered at a pre-experimental training session in order to obtain an estimate of general verbal ability. Vocabulary test scores are regarded as relatively stable estimates of premorbid intelligence. The vocabulary test was adapted from the vocabulary subtest of the Groningen Intelligence Scale. 24 The vocabulary score was used as a demographic parameter and not as an outcome parameter.
Computerized behavioral testing
Cognitive functioning was assessed using selected tests from the Neurobehavioral Evaluation System, originally described by Baker et al
25
and translated for use in Dutch populations.
26,27
Additional tests were used to examine cognitive function in more detail. Based on earlier studies within the testing laboratory, the test–retest reliability was on the order of 0.80.
26
An overview of computerized tests used in this study is given in Table 1. The tests include the following: Mood States Questionnaire: This is a modification of the Profile of Mood States,
28,29
which contains 32 adjectives that describe different mood states. Finger-tapping test: The test consisted of 3 subtests in which the subject was asked to press (tap) a button on a 2-choice response box as many times as possible within an interval of 30 seconds. Hand–eye coordination test: Subjects were asked to use a joystick to trace over a sine wave/sawtooth pattern on the video display. A cursor moved horizontally at a constant velocity while the subject controlled the vertical motion of the cursor with the joystick. Simple reaction time test: The subject was asked to press a button as quickly as possible after a red square (2.5 × 2.5 cm) appeared on the screen. Switching attention test: This test is meant to test the ability of the subject to switch rapidly between responses to simple 2-choice visual discriminations based on changing verbal cues. Color word vigilance test: Colored stimuli (words) were presented for 1.5 seconds with 2.2 seconds between stimuli. The Dutch words for green, yellow, blue, and red were presented in each of these 4 colors. Subjects had to press the button as quickly as possible when there was congruence between word and color. Symbol–digit substitution test: In this test of perceptual coding and attention, 9 symbols and 9 digits were paired at the top of the screen. The subject had to press the digit keys corresponding to a test set of the 9 symbols scrambled. Pattern comparison test: This test is similar to a computer-administered test given by Acker.
30
The stimuli used in this test were similar to those used in the pattern memory test (see below). Three 10 × 10-block matrices of blocks were presented side by side on the screen. Two of the matrices were identical, and the third was similar but with a number of the array elements changed. The subject was asked to identify the one that was different. Verbal memory test: This is a computer-administered learning and memory test adapted from the California Verbal Learning Test.
31
Spatial memory span test: This test is based on a traditional test developed by Albert and Moss
32
for measuring the capacity of short-term memory store for visuospatial information and uses an up–down procedure. Subjects were required to remember the location of a number of red-colored disks, distributed semi-randomly across 30 locations on the video screen. Digit memory span test backwards: In this up–down procedure for estimating the 50% threshold for short-term digit memory span, subjects were instructed to memorize a list of digits that were visually presented one by one on a video screen at the rate of 1 per second. Starting list length was 3 digits, and the subject’s task was to repeat the sequence in reverse order by pressing the relevant numbers on the keyboard. Pattern memory test: This nonverbal memory test is an elaboration of a manually administered test used by Warrington and James
33
to test patients with localized cerebral lesions. The Neurobehavioral Evaluation System 2 version is similar to a computer-administered test given by Acker.30 A single stimulus array consisting of a 10 × 10-block matrix (similar to those used for the pattern comparison test) was presented for 4 seconds, and then the screen was blanked. After a 3-second retention interval, three 10 × 10-block matrices were presented together, side by side, on the screen. One of these was the original array, and the other two had 3 array blocks changed. For each of 25 trials the subject had to choose which of the 3 arrays was identical to the original array.
Determination of blood concentrations
To quantify the internal dose of cyclohexane, a venous blood sample was drawn approximately 10 minutes before the end of the 4-hour exposure session. Additional blood and breath samples were taken for the development of a pharmacokinetic model as described elsewhere. 18 Samples were collected in heparinized tubes and stored at –20 °C until analysis. Blood samples were analyzed by the same methods as those used for analysis of rat blood samples. Calibration samples, prepared in blood from unexposed individuals, were linear from 10 to 5000 ng/mL. The lower quantification limit of cyclohexane in human blood was 15 ng/mL.
Statistical analysis
To determine possible differences between the low (86 mg/m3) and the high (860 mg/m3) treatment conditions, the outcome parameters were compared by univariate and repeated-measures ANOVA. In addition, the relationship between performance and blood levels of cyclohexane was examined for the 3-hour and postexposure testing time points by Pearson correlation coefficients. Analyses were conducted using the BMDP statistical software package 21 with P < .05 as the level of significance.
Results
Animal Study
Exposure concentrations
Target exposure concentrations were 1.4, 8, and 28 g/m3, respectively. The analytically determined mean concentrations (±SE) over the exposure sessions ranged from 1.32 ± 0.11 to 1.59 ± 0.12 g/m3 in the low-exposure group, 7.92 ± 0.25 to 8.22 ± 0.29 g/m3 in the intermediate-exposure group, and 28.13 ± 3.93 to 29.56 ± 1.33 g/m3 in the high-exposure group.
Clinical signs and physiological parameters
No treatment-related clinical signs were observed, and there were no significant differences in body weight during the study (data not shown). Analysis of body temperature data showed a significant difference between groups during the exposure period (repeated-measures ANOVA, F 3,28 = 2.98, P < .05) as well as a significant treatment–by–time point interaction (repeated-measures ANOVA, F 6,56 = 6.71, P < .001). Body temperature was significantly reduced by approximately 1°C in the high-exposure group (P < .001) after the first 8-hour exposure period (ANOVA, F 3,28 = 9.99, P < .001) (see Table 2). Body temperature was also reduced after the third 8-hour exposure, but the difference was much smaller and not significantly different from control.
Results of Functional Observations and Physiological Parameters in Rats Following Exposure to Cyclohexane
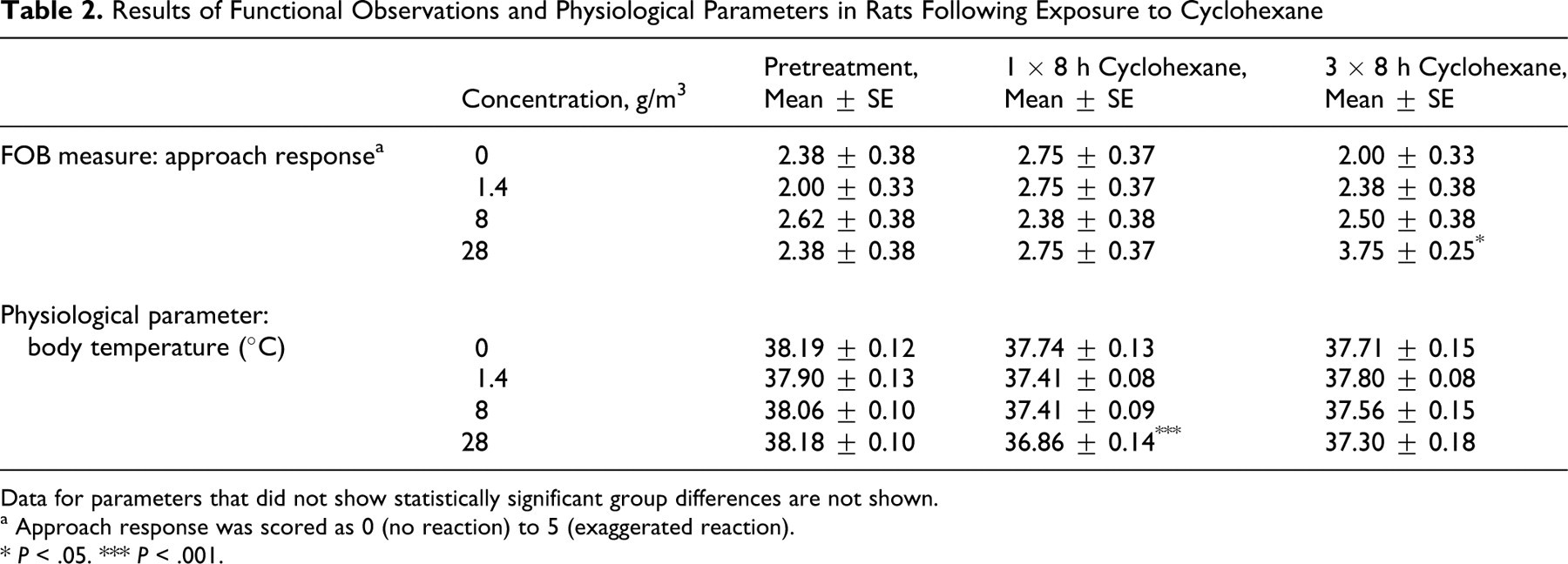
Data for parameters that did not show statistically significant group differences are not shown.
a Approach response was scored as 0 (no reaction) to 5 (exaggerated reaction).
* P < .05.
*** P < .001.
Functional observations
No statistically significant differences between groups were observed for any of the FOB parameters when tested prior to exposure.
Following exposure to cyclohexane, there were very few effects in any of the FOB measures. During the observations of gait after the first exposure period, 1 rat from the high-exposure group exhibited a slight ataxic movement. This received the lowest severity rating and was not observed after 3 consecutive 8-hour exposures. However, the observation of altered gait was exposure responsive in the sense that it was found only in the high-exposure group, and so a relationship to treatment is possible. During open field observations, 1 rat from the high-exposure group showed a slight tremor after the first 8-hour exposure. As above, this was a single observation that was not observed after 3 consecutive 8-hour exposures, but because it was exposure responsive, a relationship to treatment is possible. Forelimb grip strength, hind limb grip strength, and mean foot splay were not significantly affected by treatment. There was an effect on approach response that increased with exposures, and the high-exposure group was significantly different from controls (P < .05) (see Table 2). However, this was observed only after 3 consecutive 8-hour exposures (Kruskal-Wallis statistic = 10.57, df = 3, P < .05), and there were no apparent differences after the first 8-hour exposure period. Touch response, click response, tail-pinch response, and arousal were not affected by treatment.
To summarize this section, the FOB observations indicated minimal effects of treatment on gait, tremor, and approach response in the highest exposure group. The differences were small and not consistently observed, and the effects on gait and tremor were not statistically significant.
Motor activity
No statistically significant differences between groups were observed for any of the motor activity parameters when tested prior to treatment or after exposure.
Visual Discrimination
General measures of responding were not affected by treatment (see Table 3). All animals completed 100 trials on every exposure day, and the mean percentage of reinforcements was greater than 99% in all groups. Furthermore, mean discrimination ratio was 0.86 or more for all groups. Measures of stimulus control and disinhibition were unaffected by treatment. There was a significant treatment–by–time point interaction in the number of repetitive errors (repeated-measures ANOVA, F 6,56 = 2.51, P < .05); however, this was not exposure responsive; that is, the significant increase was in the 8-g/m3 group (group comparisons, F 1,14 = 4.78, P < .05), whereas the performance of the 28-g/m3 group was not different from controls.
Mean Group Data for Visual Discrimination Performance Tests in Rats Exposed to Cyclohexane
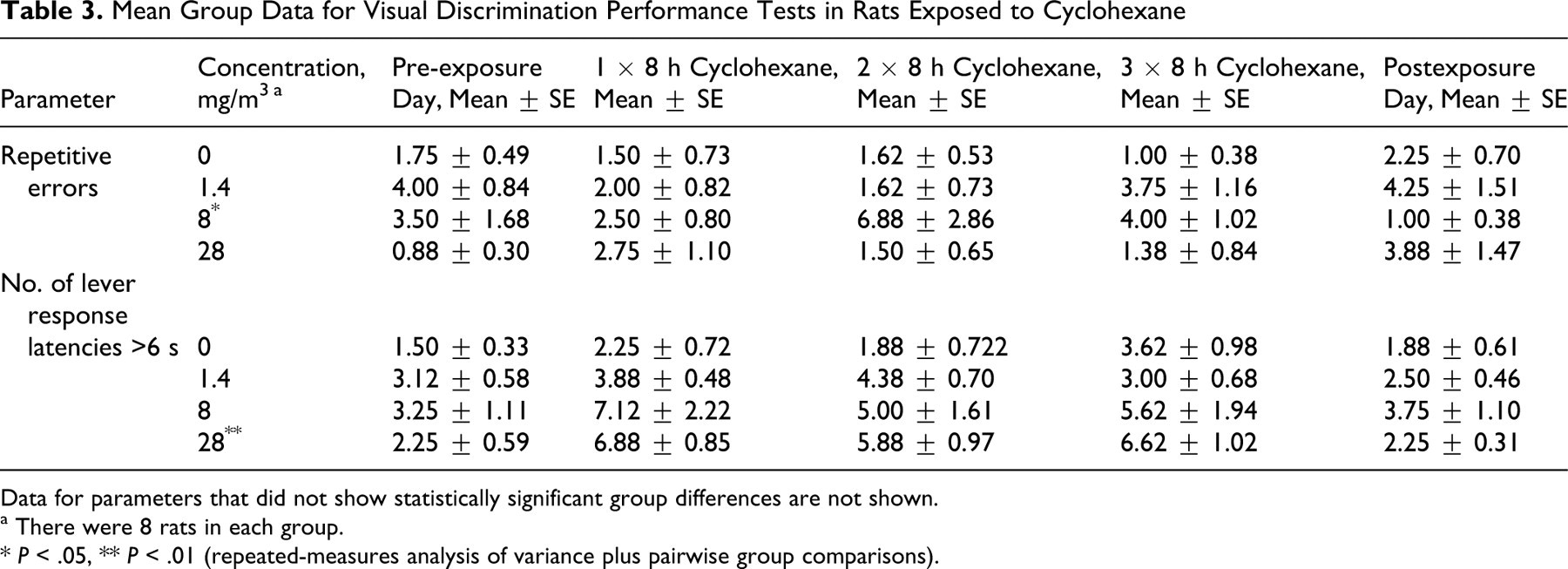
Data for parameters that did not show statistically significant group differences are not shown.
a There were 8 rats in each group.
* P < .05
** P < .01 (repeated-measures analysis of variance plus pairwise group comparisons).
In the domain of psychomotor speed, there were some changes associated with exposure, but these were small and not always statistically significant. Lever response latency appeared to increase with increasing exposure, and there also appeared to be a reduction in the number of fast (ie, latency <1 seconds or <2 seconds) responses. However, the differences between exposed and control groups were not statistically significant. There was a time point–dependent increase in the number of long latencies (ie, >6 seconds) (repeated-measures ANOVA, F 6,56 = 2.66, P < .05), and in this case the high-exposure group had significantly (group comparisons, F 1,14 = 12.42, P < .01) more long latencies than the control group. No significant differences were observed when the rats were tested the day following the final exposure, indicating that these effects were rapidly reversible once exposure stopped.
Tissue levels of cyclohexane
Concentrations of cyclohexane in brain were approximately 10-fold higher than in blood. There were no striking differences in tissue levels between rats exposed for 8 hours and those exposed for 8 hours on 3 consecutive days (see Table 4).
Comparison of Blood and Brain Levels of Cyclohexane in Rats After Exposure for 8 Hours Once or for 3 Consecutive Days
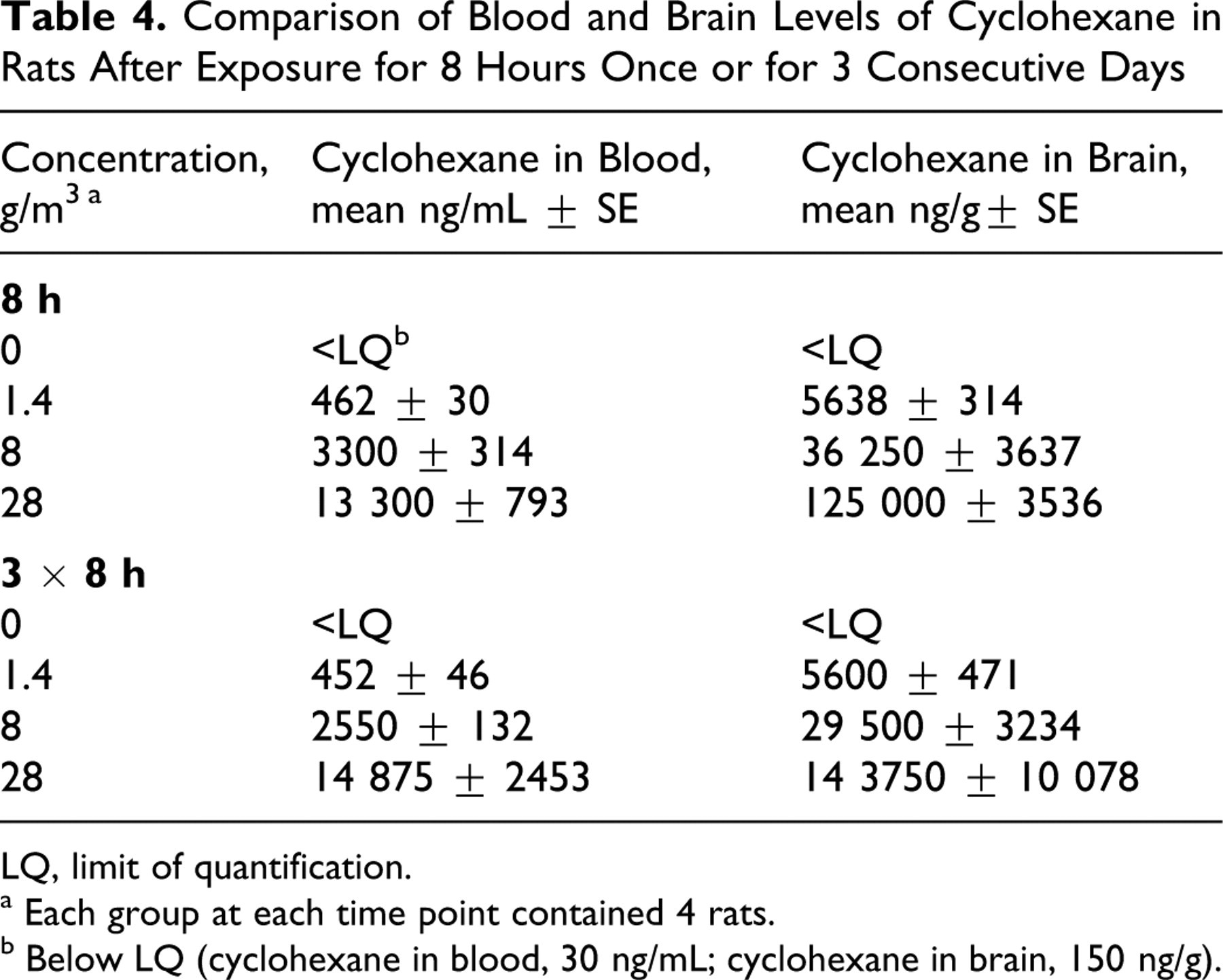
LQ, limit of quantification.
a Each group at each time point contained 4 rats.
b Below LQ (cyclohexane in blood, 30 ng/mL; cyclohexane in brain, 150 ng/g).
Human Volunteer Study
Exposure levels
The mean concentrations (± SD) for the 860-mg/m3 cyclohexane sessions were 859 ± 28, 873 ± 17, 869 ± 12, and 866 ± 8 mg/m3, respectively. The mean concentrations (± SD) for the 86 mg/m3 cyclohexane sessions were 89 ± 2.1, 88 ± 1.4, 87 ± 3.5, and 90 ± 5.2 mg/m3, respectively.
Subjective measures
On each test day, prior to and following the exposure periods, subjects were asked to complete short “well-being” questionnaires. The questionnaire consisted of items designed to elicit subjective reactions related to exposure, including reaction to odor, headache, nausea, throat dryness, or coughing and tearing. In addition, subjects were asked to indicate on a dichotomous scale whether they were in the high-or low-exposure group.
As shown in Table 5, more subjects in the high-exposure group reported headache and dry throat than did subjects exposed to the lower concentration. There were no consistent reports of eye or respiratory irritation. In the assessment of these observations as potential treatment-related findings, we noted that subjects exposed to 860 mg/m3 cyclohexane were more likely to have been aware that they were being exposed to the higher concentration. It should also be noted that although headache was not reported in the well-being questionnaires prior to the first exposure, it was reported by 3 individuals in forms completed prior to the second exposure, which took place 7 days later.
Results of Well-Being Questionnaires Taken After Exposure Periods
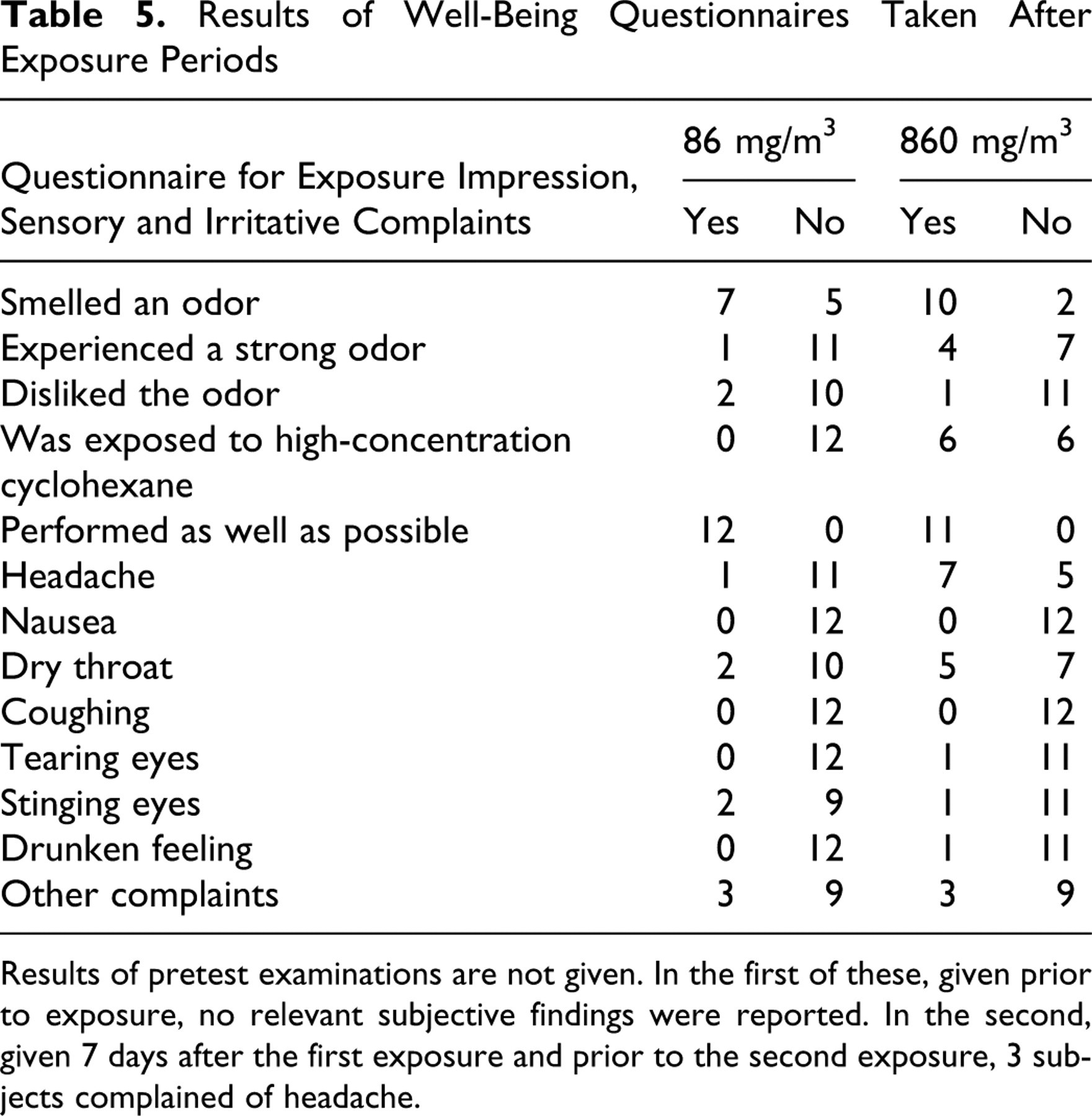
Results of pretest examinations are not given. In the first of these, given prior to exposure, no relevant subjective findings were reported. In the second, given 7 days after the first exposure and prior to the second exposure, 3 subjects complained of headache.
Cyclohexane levels in blood
Blood samples were taken 10 minutes before the end of the exposure sessions and reflect what were assumed to be the steady-state levels. Among volunteers exposed to 860 mg/m3 cyclohexane, the mean blood concentration (± SD) was 618 ± 68 ng/mL, and it was 55 ± 7 ng/mL among subjects exposed to 86 mg/m3.
Behavioral Assessments
The following behavioral assessments were used: Mood and affect: There were no treatment-related effects on measures of depression, anger, vigor, fatigue, or tension. Psychomotor skills: There were no apparent treatment-related differences between groups in either the hand–eye coordination tests or the finger-tapping tests. Attention: There were no treatment-related differences in either the simple reaction time test or the color–word vigilance test. There were longer latencies to respond in all 3 of the switching attention tests, but none of the differences was statistically significant. Perceptual coding: There were no treatment-related differences between the exposed groups in either the symbol–digit substitution test or the pattern comparison test. Learning and memory: There were no treatment-related differences between groups in any of the learning and memory tests (digit memory span test, spatial memory span test, pattern memory test). Among the verbal memory tests, most of the differences were not significant, although there was a small but statistically significant reduction in the semantic cluster ratio (ANOVA, F
1,11 = 6.04, P < .05) (see Table 6), but given there were no differences in learning or memory, it is unlikely that this was due to treatment. Relationship with specific blood levels: There were no significant relationships between blood levels of cyclohexane and any of the parameters measured.
Mean Test Scores and Standard Error of the Mean at Different Time Points During and After Exposure of Human Volunteers to 86 or 860 mg/m3 Cyclohexane
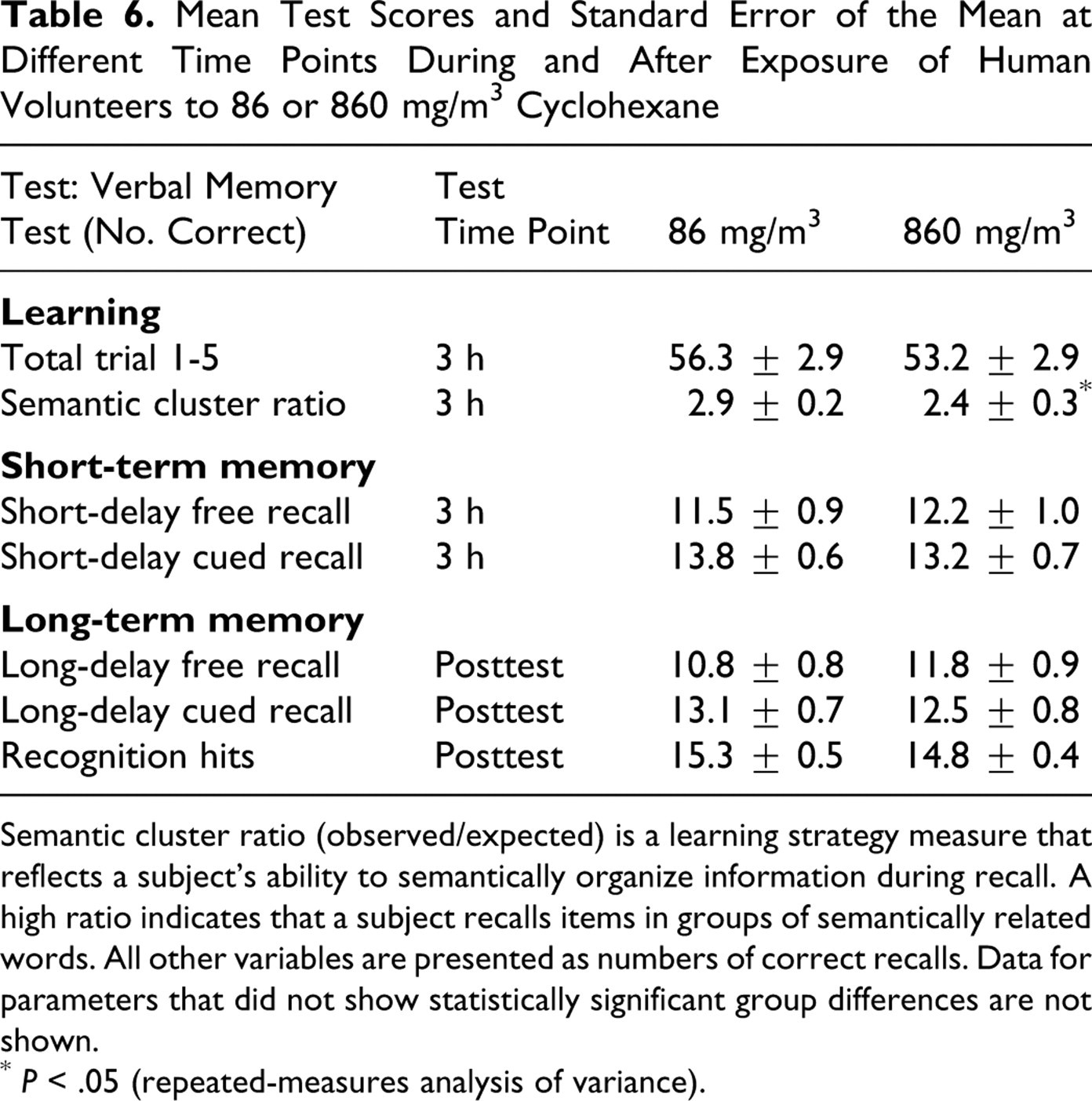
Semantic cluster ratio (observed/expected) is a learning strategy measure that reflects a subject’s ability to semantically organize information during recall. A high ratio indicates that a subject recalls items in groups of semantically related words. All other variables are presented as numbers of correct recalls. Data for parameters that did not show statistically significant group differences are not shown.
* P < .05 (repeated-measures analysis of variance).
Discussion
The objectives of this study were to characterize the acute CNS effects of cyclohexane in rats and humans and to compare these effects between species. The overall goal was to use these results, along with those of companion studies of ethanol 16 and white spirit, 17,34 to define and validate a set of procedures that could be used to predict levels of acute CNS effects in humans from neurobehavioral studies in rats.
The rat data from the present study indicated that cyclohexane produces limited effects on the CNS at exposure levels up to 28 g/m3. There were no effects on body weight and minor effects on body temperature at 28 g/m3, indicating minimal systemic effects at this level. The functional observation battery provided evidence of minimal CNS effects via small, not statistically significant effects on gait and tremor as well as a small but statistically significant effect on approach. Although these effects were only detected in the highest exposure group, satisfying the criteria for dose response, the lack of consistency between assessments (ie, differences between the first and third exposure days) reduced the likelihood that these differences were really due to treatment.
The visual discrimination tests suggested that cyclohexane may have affected psychomotor speed. But even in this case, the effects were small and, for the most part, not statistically significant. An overall conclusion from the rodent data would be that at 28 g/m3, cyclohexane may have affected gait, tremor, sensorimotor reactivity, and psychomotor speed, but all of the changes were small and their relationship to treatment was uncertain.
In other related studies, cyclohexane did not produce any acute CNS effects following acute exposures at levels up to 7000 ppm (~24 g/m3) for 6 hours. 35 In a subchronic neurotoxicity study, cyclohexane was reported to produce transient overt signs of mild sedation during and immediately after repeated exposure to 2000 (~6.9 g/m3) or 7000 ppm (~24 g/m3). However, there were no significant effects in functional observations or motor activity and no evidence of treatment-related pathologic changes in the nervous system. 36
The kinetic studies indicated that cyclohexane is taken up by the blood and brain. The blood/brain ratio was in the range of 10 to 11, somewhat higher than that reported by Zahlsen et al. 37 The similarity in brain levels after either 1 or 3 exposures indicated that cyclohexane did not induce its own metabolism. Zahlsen et al 38 observed some reduction in steady-state levels of 1,2,4-trimethylcyclohexane with repeated exposure and suggested that this might be attributable to metabolic induction. However, to our knowledge induction of metabolism has not been directly demonstrated for aliphatic or cycloaliphatic hydrocarbons.
The human studies provided no compelling evidence for treatment-related effects at 860 mg/m3, the recommended occupational exposure limit for cyclohexane in the Netherlands at the time the study was conducted. Based on self-reporting, some subjective measures including headache and dry throat were increased, but there was no apparent eye or respiratory tract irritation. There were no treatment-related effects on mood and affect, psychomotor skills, perceptual coding, attention, or learning and memory, except for 1 statistically significant but implausible reduction in semantic cluster ratio. The evidence of minimal effects in rats exposed to 28 g/m3 (approximately 8000 ppm) suggests that effects in humans exposed at much lower levels would be unlikely. A PBPK model predicted that the lowest effect level in humans would be greater than approximately 4100 mg/m3 but below approximately 13 700 mg/m3, assuming effects in humans and animals are associated with similar brain concentrations. 18
In summary, cyclohexane produced minimal acute effects on the CNS in rats and humans. In the rodent studies, even at levels of 28 g/m3, the only effects were small effects on gait and a statistically significant reduction in psychomotor speed in visual discrimination tests. In humans exposed to 860 mg/m3, there were no clear treatment-related effects on any of the objective measures of acute CNS effects, although there were some possible differences in some subjective measures.
Footnotes
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
This work was sponsored by the CEFIC Hydrocarbon Solvent Producers Association and a grant (to Dr Beverly M. Kulig) from the Netherlands Ministry of Economic Affairs. Two of the authors (R. H. McKee, D. E. Owen) are employed by companies that manufacture cyclohexane.
