Abstract
This report describes a physiologically based pharmacokinetic model for cyclohexane and its use in comparing internal doses in rats and volunteers following inhalation exposures. Parameters describing saturable metabolism of cyclohexane are measured in rats and used along with experimentally determined partition coefficients. The model is evaluated by comparing predicted blood and brain concentrations to data from studies in rats and then allometrically scaling the results to humans. Levels of cyclohexane in blood and exhaled air are measured in human volunteers and compared with model values. The model predicts that exposure of volunteers to cyclohexane at levels of 4100 mg/m3 (∼1200 ppm) will result in brain levels similar to those in rats exposed to 8000 mg/m3 (the no-effect level for acute central nervous system effects). There are no acute central nervous system effects in humans exposed to 860 mg/m3, consistent with model predictions that current occupational exposure levels for cyclohexane protect against acute central nervous system effects.
Keywords
Physiologically based pharmacokinetic (PBPK) models have been useful in risk assessments for a number of compounds. 1-3 These models are particularly useful for extrapolations beyond the observed dose range, from one dosing route or scenario to another, or from high to low dose levels. In addition, extrapolation from laboratory animals to humans is possible, as is evaluation of interindividual variability. In each case, the PBPK model provides a framework for determining the relationship between the external (environmental) exposure and the internal (biologically effective) target tissue concentration. 4,5
The central nervous system (CNS) is a target for the acute effects of organic solvents, characterized by reversible signs of CNS depression both in animals and in humans. Occupational exposure limits (OELs) for organic solvents are often designed to avoid signs of acute intoxication, and recommended OELs often cite CNS depression as an effect of concern. 6 At levels that are high by comparison to occupational exposure recommendations, acute, nonlethal CNS effects typically include nausea, loss of coordination, and signs of intoxication. At lower levels of exposure, subtle changes in behavioral function, particularly those related to information processing and psychomotor function, can arise. 7,8 Eye or respiratory irritation and/or target organ toxicities of various types may also contribute to the setting of occupational exposure levels for individual solvents. However, the onset of signs of acute CNS effects often occurs at lower levels than these other effects. Thus, OELs that are based on acute CNS effects usually provide an adequate measure of protection for chronic toxicity and other effects.
As part of a project designed to help set occupational exposure limits, 9-11 PBPK modeling has been proposed as a tool for predicting the levels of exposure to complex hydrocarbon solvents that can be expected to produce acute behavioral effects in humans based on the results of effects testing in animals. No appropriate cyclohexane (CyHx) data were available to relate internal dose to CNS effects in relevant species (eg, human, rat, monkey, or dog). Therefore, in separate studies, rats were exposed by inhalation to CyHx at 0 g/m3, 1.4 g/m3 (∼400 ppm), 8 g/m3 (∼2000 ppm), and 28 g/m3 (∼8000 ppm) 8 h/d for 3 consecutive days and then tested for effects on motor activity, functional observational measures, and learned performance. 12 CyHx produced a dose-responsive psychomotor slowing in learned performance with small but significant effects of CyHx at 28 g/m3 (ie, the lowest observed effect level [LOEL]) with a no observed effect level (NOEL) of 8 g/m3. In addition, dosimetry studies provided measures of concentrations of CyHx in blood and brain. Having information regarding the exposure levels at which effects occur and the concentration of CyHx in the brain of rats at these levels, however, one is still confronted with the question of how these results relate to human populations. In the risk assessment process, this is typically handled by the use of “uncertainty” (or “safety”) factors 13 that are used to relate predicted exposure levels in humans to effect levels measured in animal studies and/or to account for a possible range of sensitivity in human populations. There is, however, another approach to address the question of cross-species extrapolation: the use of PBPK modeling to integrate the effects and dosimetry data.
In the present program, studies were undertaken to develop a PBPK model for CyHx that could be used to integrate human and animal test data. Experiments were conducted to obtain the Michaelis-Menten constants, and a PBPK model was developed for the rat. The model was tested by comparison to time course data on CyHx uptake and elimination in rat blood and brain. The rat model was then allometrically scaled by comparing the values predicted by the model with blood concentration data from the human volunteers.
CyHx is a widely used industrial solvent with a total volume in commerce in Europe estimated as approximately 900 000 tons/y. 14 As with other volatile organic solvents, the CNS is a primary target for acute effects. The rationale for selection of CyHx as one of the model compounds for this study was the availability of a substantial database on the neurobehavioral effects of CyHx in rodents. 15,16 Human modeling studies have also been published. 17 A unique feature of the present study is that PBPK modeling has been used to link animal and human data to predict the levels of exposure at which CNS effects might be expected to occur in humans.
Methods
Chemicals
Cyclohexane was purchased from commercial suppliers at a purity of greater than 99%. For the rat uptake study and the kinetic study in volunteers, CyHx was obtained from E. Merck Nederland B.V. (Amsterdam, The Netherlands). For determination of blood and brain concentrations in rats, CyHx was obtained from J. T. Baker (Deventer, The Netherlands). Other chemical characteristics included density = 0.78 kg/L, boiling point = 80°C at 105 Pa, ignition temperature = 260°C, explosive limits = 1.2%:8.3% vol/vol, and saturating concentration = 357 g/m3. The relationship between mg/m3 and ppm was 1 mg/m3 = 0.29 ppm. All other chemicals used for analyses were obtained from Merck (Darmstadt, Germany) and were of analytical grade.
Animals and Housing
Adult male Wistar-derived WAG/RijCR/BR rats were purchased from Charles River Wiga (Sulzfeld, Germany). Animals were 9 to 14 weeks of age at the time of exposure with body weights of approximately 200 to 250 g. Animals were housed in suspended wire mesh steel cages under normal housing conditions in temperature-controlled (20°C-25°C) animal rooms with a relative humidity between 40% and 70%. Animals were maintained on a 12-hour light/dark cycle. For animals in the cyclohexane uptake study, food and water were available ad libitum except during the exposure period. For animals used in the kinetic study, food was available during exposure and was changed after each exposure period.
Cyclohexane Uptake Study in Rats
Exposure conditions
To determine the Michaelis-Menten parameters Vmax and Km for CyHx, a closed exposure system was used to collect data on the uptake of CyHx for a range of initial concentrations using techniques similar to those described by Gargas et al. 18 Rats were exposed in a desiccator that was part of a closed-loop system. The volume of the system was 11.2 L, and the system consisted of a desiccator, an inert pump to circulate the test atmosphere, an oxygen-measuring device, a mass flow control unit to supplement oxygen, a column with lime to remove carbon dioxide, and an impinger filled with 1 N cooled sulfuric acid to remove ammonia. To generate the test atmosphere, CyHx was injected into the system through a septum and quickly evaporated by local heating.
Groups of 3 rats were exposed to CyHx for 6 hours at target concentrations of 0.1, 0.6, 4, or 24 g/m3. In addition, to evaluate the adsorption of CyHx to rat fur, 3 rats were killed by CO2 overdose prior to exposure. The bodies were then exposed at a target concentration of 6 g/m3, and the concentration inside the exposure chamber was measured for 6 hours.
Analysis of the test atmosphere
To monitor CyHx concentrations, 250-μL samples of air were automatically drawn from the exposure chamber at approximately 5-minute intervals using a sample loop. CyHx concentrations were measured online using a Mega 8000 gas chromatograph (Fisons Interscience, Breda, The Netherlands) equipped with a flame ionization detector. A CP-Sil-5-CB column (30 m × 0.53 mm inner diameter, film thickness 1.5 μm) was used. Column temperature was 120°C (isotherm), and column flow was 3.65 mL/min. The gas chromatograph was calibrated using Tedlar bags (Chrompack, Bergen op Zoom, The Netherlands) filled with measured amounts of air into which metered amounts of liquid CyHx were allowed to evaporate.
Rat Kinetic Study
Exposure conditions and sample collection
Groups of rats were exposed to different concentrations of the test atmosphere using 4 modified H 1000 inhalation chambers (Hazleton Systems, Aberdeen, Md). Each chamber had a pyramidal top and bottom and was constructed of stainless steel with glass doors on 2 sides. The test atmosphere was generated by pumping liquid CyHx into stainless steel tubing that led through an oven at 400°C. The vapor was transported with an air stream from a compressed air source and added to the main airflow system. During exposure, samples were automatically drawn from each exposure chamber at approximately 12-minute intervals and analyzed using gas chromatography. Gas chromatograph readings were calibrated using Tedlar bags filled with air and 1.04, 8.00, or 31.20 g/m3 CyHx.
To determine the blood and brain concentrations of CyHx during uptake of the solvent, groups of 3 or 4 rats were exposed to CyHx at target concentrations of 0 g/m3 (control group), 1.4 g/m3 (∼400 ppm), 8 g/m3 (∼2300 ppm), and 28 g/m3 (∼8000 ppm) for 2, 4, or 8 hours and sacrificed immediately thereafter. For evaluation of elimination, groups of 3 or 4 rats were exposed for 8 hours and sacrificed 0.5, 1, 2, 4, or 8 hours after exposure. To determine whether repeated exposures would produce differing CyHx concentrations in blood and brain, groups of rats were exposed to CyHx 8 h/d for 3 consecutive days at the concentrations listed above. These rats were sacrificed immediately following the termination of exposure on the third exposure day. Rats were killed by decapitation, and trunk blood was collected in heparinized glass tubes and kept on ice. Immediately after decapitation, brains were removed from the skulls. After weighing, the brains were homogenized in chilled saline (dilution 1:4 wt/vol) and kept on ice.
Analysis of rat blood and brain concentrations
CyHx concentrations in rat blood and rat brain homogenate were determined with gas chromatography using headspace analysis with a flame ionization detector. A 0.2-mL aliquot of blood or brain homogenate was added to a headspace vial (9 mL) containing 1.8 mL of physiological saline solution (9 g NaCl/L). Samples were stored for at least 1 night at –20°C before analysis. Preliminary validation studies of the analytical techniques indicated that blood and brain samples were stable for 20 days when stored at this temperature. After thawing, the samples were placed in a water bath at a temperature of 60°C for at least 1 hour to establish equilibrium.
For analysis, a Carlo Erba 4160 gas chromatograph (Carlo Erba, Milan, Italy) equipped with a split injector and flame ionization detector was used. One milliliter of vapor phase was injected with an autosampler HS 250 using split injection. A DB-1 capillary column (30 m × 0.536 mm [inner diameter], film thickness 1.5 μm) was used. The column was operated isothermally at 45°C. The temperature of the injection syringe was 80°C. The injection port temperature was 200°C, and the detector base temperature was 300°C. The carrier gas was helium with a column inlet pressure of 29 kg/cm2. Data acquisition was performed using a Spectra Physics Chromnet/Labnet (Spectra Physics, Eindhoven, The Netherlands) station. Calibration curves were made in physiological saline and were linear from 50 to 100 000 ng per vial. The limit of quantitation for CyHx in blood and brain samples was 30 ng/mL.
Kinetic Study in Volunteers
Subjects
Three healthy male subjects were recruited from a pool of individuals living near the laboratory who had previously indicated their willingness to serve as volunteer subjects in human studies at TNO Quality of Life (Zeist, The Netherlands). Volunteer 1 was 27 years old, weighed 80.2 kg, was 189.9 cm tall, had a body fat measurement of 18% (calculated from skin caliper data), drank approximately 7 alcoholic beverages per week, and smoked 10 to 15 cigarettes per day. Volunteer 2 was a 22-year-old nonsmoker, 174.6 cm tall, 64.0 kg body weight, 12.1% body fat, and drank approximately 6 alcoholic beverages per week. Volunteer 3 was a 23-year-old nonsmoker, 190.2 cm tall, 93.2 kg body weight, 18.1% body fat, and drank approximately 12 alcoholic beverages per week.
The principles of informed consent in the current revision of the Declaration of Helsinki 19 were implemented, and the studies were conducted in accordance with the EC Principles of Good Clinical Practice. Prior to the start of the study, a check of each subject’s general health, including a medical history, physical examination, clinical laboratory tests, and a drug screen, was conducted. For inclusion into the study, volunteers had to meet the following requirements: male, between 20 and 50 years of age, body weight between 60 and 90 kg, fat volume percentage less than 30% (measured with a skin caliper), in good medical health as determined by medical and laboratory examination, Dutch as native language, and willing to sign the informed consent form. Exclusion criteria included a history of a serious medical condition or disease, a history of alcohol or drug abuse, present use of psychopharmacological medications, excessive use of alcohol (>28 drinks per week), a positive drug screen, liver or kidney dysfunction, participation in a clinical study within 3 months of the start of the present study, a history of occupational exposure to organic solvents, a blood donation within 1 month prior to the start of the study, claustrophobia, need to wear contact lenses, or TNO employee or first- or second-removed relative of a TNO employee.
Exposure
Subjects were exposed to CyHx at a targeted concentration of 875 mg/m3 (∼250 ppm), the maximum allowable concentration value for CyHx in The Netherlands at the time the study was conducted.
20
The 3 volunteers were exposed simultaneously in a specially constructed temperature and relative humidity–controlled exposure room. The room was equipped with a transparent viewing window, an intercom system, 3 chairs and tables; the room size was 2.9 m × 2.3 m × 2.1 m (length × width × height). The air ventilation system serving the exposure room allowed for the partial recirculation of the test atmosphere. Subjects were provided with breakfast at 8
The test atmosphere was generated by total evaporation at an elevated temperature of a constant flow of liquid CyHx in a constant flow of fresh air. During the experiment, the concentration of CyHx vapor was continuously monitored by an infrared absorption spectrophotometer at 3.49 μm using a path length of 3.75 m (Miran 1A, Foxboro, Norwalk, Conn). The measured concentration of CyHx in the exposure room was recorded at approximately 15-minute intervals and averaged to provide the mean exposure concentration. Examination of the chart recording made during exposure indicated that concentrations inside the exposure chamber were within 10% of the target concentration of 875 mg/m3.
Sampling
To examine blood levels of CyHx during and shortly after exposure, each subject was fitted with an indwelling cannula inserted into the median cubital vein on the ventral side of the forearm prior to the start of exposure. On the day of exposure, venous blood samples were drawn at predetermined times prior to, during, and approximately 9 hours following the termination of exposure. On the 3 consecutive days following exposure, blood samples were obtained from the median cubital vein by venipuncture. An aliquot of 1.0 mL of blood was pipetted into a headspace vial filled with 1.0 mL of physiological saline solution (9 g NaCl/L). These samples were stored frozen at –20°C for at least 1 night prior to analysis.
Breath samples were collected at predetermined time points before and following the termination of exposure. Samples of end-exhaled alveolar air were collected in glass sampling tubes (approximately 70 mL) after 30 seconds of breath holding. Breath holding was required to achieve equilibrium between mixed venous blood and exhaled alveolar air. The sampling tubes were closed on both sides with screw caps containing rubber septa and Teflon linings.
Analysis in human blood and exhaled alveolar air
CyHx concentrations in human blood were determined with a Carlo Erba 4160 gas chromatograph using the same techniques as those described above for the analysis of rat brain and blood. For the human study, CyHx calibration samples prepared using blood from unexposed individuals were linear from 10 to 5000 ng/mL of blood. The detection limit and the limit of quantitation for CyHx in human blood were 8 ng/mL and 15 ng/mL, respectively.
For analysis of the exhaled alveolar air, a Carlo Erba 5300 gas chromatograph equipped with a split injector and flame ionization detector was used. A 1-mL gas sample was injected into a DBB-1 capillary column (30 m × 0.548 mm [inner diameter], film thickness 1.5 μm). Further GC conditions were similar to those used for the analysis of blood samples. Calibration samples were made using glass sampling tubes filled with nitrogen gas. The CyHx calibration curves were linear from 12.5 to 500 μg/L of air. The limit of quantitation for CyHx in human exhaled alveolar air was 1 ng/mL.
Rat and Human PBPK Modeling Studies
The model used in this study was a 5-compartment model that is similar to the 4-compartment model described by Ramsey and Andersen 21 for evaluating the kinetics of inhaled styrene and by Gargas et al 18 for determining the kinetic constants from gas uptake data of different inhaled solvents.
The 5 compartments defined in the present model included (1) the liver, serving as the metabolizing organ; (2) fat; (3) a lumped compartment representing slowly perfused tissue consisting mainly of muscle; (4) a lumped compartment representing richly perfused tissue; and (5) the brain. The brain was added as a separate tissue compartment because the CNS is considered to be the primary target for the expression of the acute effects of hydrocarbon solvents. In addition, a lung compartment was included for gas exchange. To take account of possible binding of CyHx to the fur coat of the rats, 22 a fur compartment was incorporated into the rat PBPK model with parameters based on the measurements obtained in the fur control study. The model is represented schematically in Figure 1.
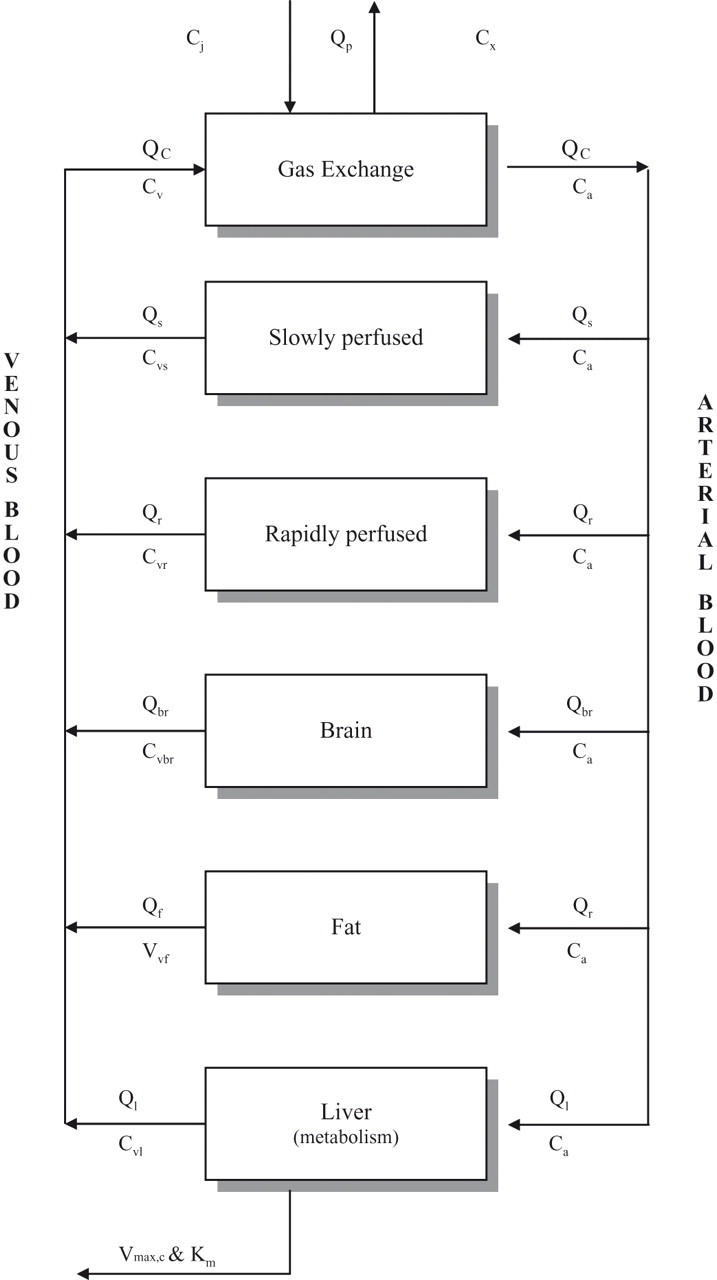
Schematic presentation of the PBPK model for cyclohexane. Ca, concentration of solvent in arterial blood; Ci, concentration of solvent in ambient air; Cv, concentration of solvent in venous blood, leaving specific tissue compartments, that is, Cvs (slowly perfused tissue), Cvr (richly perfused tissue), Cvbr (brain), Cvf (fat), Cvl (liver); Cx, concentration of solvent in expired air; Km, Michaelis-Menten constant (the concentration of solvent that results when the metabolic rate is equal to half the maximum metabolic rate); Qbr, rate of blood flow through brain; Qc, cardiac output; Qf, rate of blood flow through fat; Ql, rate of blood flow through liver; Qp, pulmonary ventilation rate; Qr, rate of blood flow through richly perfused tissue; Qs, rate of blood flow through slowly perfused tissue; Vmax,c, maximum metabolic rate (at high substrate concentrations).
Values for the physiological parameters were obtained from the literature 23 and are given in Table 1. In the PBPK simulations for the rat, the value for body weight was the average of the 3 rats used in each experiment.
Physiological (Flows and Volumes) and Biochemical (Vmax and Km) Parameters and Partition Coefficients Used in the PBPK Model for Cyclohexane for Rat and Human
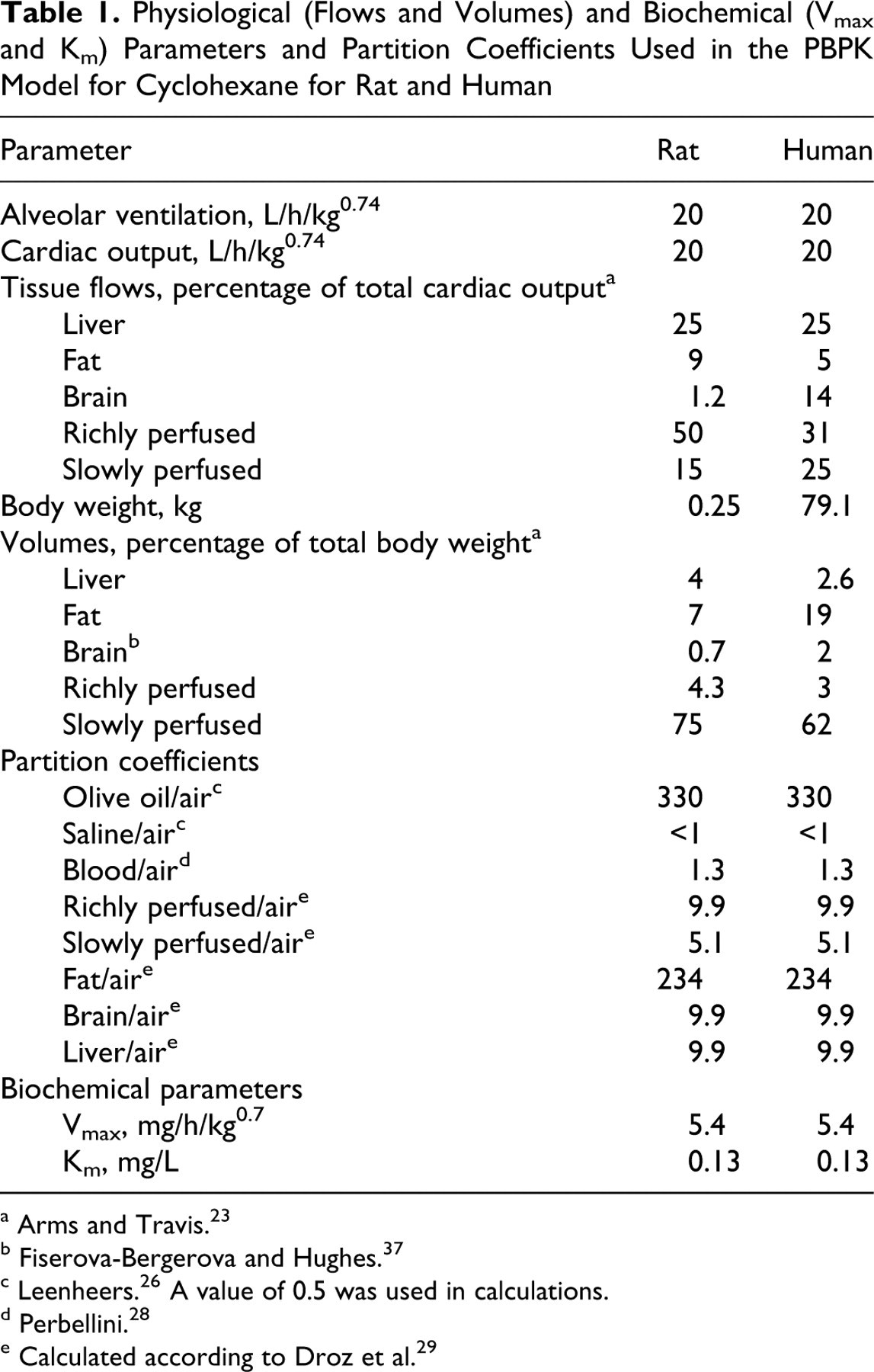
a Arms and Travis. 23
b Fiserova-Bergerova and Hughes. 37
c Leenheers. 26 A value of 0.5 was used in calculations.
d Perbellini. 28
e Calculated according to Droz et al. 29
Pulmonary ventilation rate (QP) was taken from Davies and Morris 24 for a 0.25-kg rat and was then scaled to body weight (BW), expressed in kilograms according to the equation QP = 20 L/h × (BW)0.74. Cardiac output (QC) was then described by the same equation.
CyHx was assumed to be eliminated through saturable biotransformation in the liver.
25
Calculation of the metabolic clearance of the parent compound by the liver compartment was based on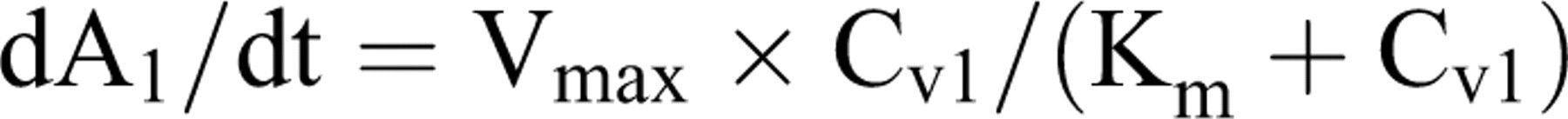
Saline/air and olive oil/air partition coefficients for CyHx were obtained in a separate study 26 according to the method described by Sato and Nakajima 27 and are presented in Table 1. The rat blood/air partition coefficient for CyHx was obtained from the literature. 28 These data were used to calculate the tissue/blood partition coefficients for the different tissue compartments using the equations of Droz et al. 29 For these calculations, a partition coefficient of 0.5 was used for saline/air. The liver/blood and the richly perfused tissue/blood partition coefficients were taken as equal to the brain/blood partition coefficient. 30 Tissue/air partition coefficients were calculated as the predicted tissue/blood partition coefficient multiplied by the measured blood/air partition coefficient.
The metabolic parameters Vmax and Km were derived by optimization of the fit of the PBPK model simulations to the experimental data obtained in the closed chamber study at 4 different concentrations corrected for adsorption to the fur (described in Results). Because metabolic rate is dependent on body weight, Vmax was allometrically scaled in the following way: 18 Vmax = Vmax,c × (BW)0.70.
The differential equations used to describe the model have been reported previously. 21 Optimizations were conducted using acslXtreme v2.4.2.1 (Aegis Technologies, Huntsville, Ala) software. The optimization criterion was the minimization of the log-likelihood function using the Nelder-Mead algorithm, assuming a relative error model (heteroscedasticity = 2). All simulations were conducted using the Gear algorithm.
Sensitivity analyses were conducted by changing a single parameter’s value by 0.001, calculating the changes in the response of interest, and normalizing the resulting change to the baseline values of the input and output. That is, the normalized sensitivity coefficient (NSC) = (% change in output)/(% change in input).
Further Model Validation
The rat model was further tested against CyHx exhalation data from the literature. Breath and excreta were collected from 3 male Fisher 344 rats injected (intravenously) with 10 mg/kg 14C-CyHx. 25
Results
Cyclohexane Uptake Study in Rats
The measured CyHx concentrations in the exposure chamber for the 0.1, 0.6, 4, and 24 g/m3 exposure groups at the 5-minute point were 0.105 g/m3, 0.401 g/m3, 3.841 g/m3, and 22.401 g/m3, respectively. The CyHx concentrations in the exposure chamber decreased at different rates for the different exposure groups, indicating metabolic clearance ( Figure 2). Reductions of approximately 50% in the concentration of CyHx in the exposure chamber were seen after approximately 68, 110, 125, and 180 minutes for exposures with initial chamber concentrations of 0.1, 0.6, 4, and 24 g/m3, respectively. In the fur control experiment, a very slight reduction in the CyHx concentration in the chamber at the very beginning of the measurement period was seen, indicating some uptake of CyHx by the fur. Because concentrations were stable after this initial decrease, a simple description as a constant chamber “loss” rate would not accurately describe the time course of the chamber decline (initially steeper, then flat). The model simulations indicate approximately 8% of the cyclohexane was adsorbed to fur in the control experiment.
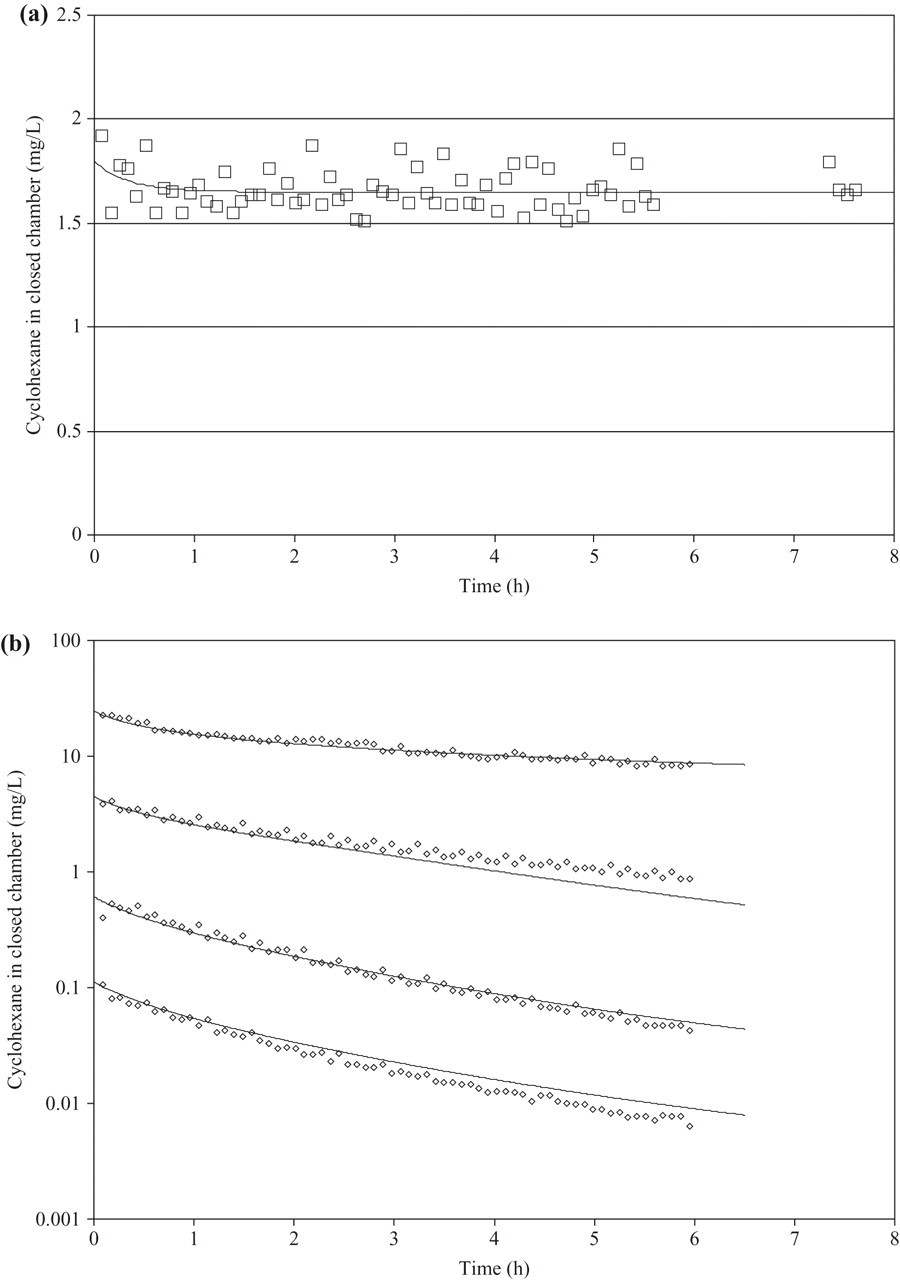
Measured (symbols) and predicted concentrations of cyclohexane during closed chamber exposures. (a) Control exposure of 3 dead rats at a starting concentration of 1.7 g/m3. (b) Exposure of 3 live rats at 4 different starting exposure concentrations (0.1, 0.6, 4, and 24 g/m3).
To take into account the loss of CyHx due to adsorption to the fur in estimating kinetic parameters, the data obtained from the fur control condition across the exposure session were curve-fitted using simulation techniques. A simulated fur compartment characterized by a reversible flow of 2.6 L/h, a fur/air partition coefficient of 0.97, and a virtual volume of 1.0 L best fit the measured data ( Figure 2a). These values were subsequently incorporated into the simulations of CyHx uptake in living rats. The results of simultaneously fitting the 4 measured concentration time courses in the closed chamber for the 4 exposure levels using the PBPK model corrected for fur adsorption is illustrated in Figure 2b. Best visual agreement between the experimental data and the results of the PBPK modeling procedure for CyHx was obtained for a value of 5 mg/h for Vmax,c and 0.1 mg/L for Km. These values were used in the PBPK model to predict blood and brain kinetics of CyHx in rats.
Rat Kinetic Study and PBPK Modeling for CyHx in the Rat
The analytically determined mean concentrations over the exposure sessions ranged from 1.51 to 1.59 g/m3 in the low-exposure group, 7.92 to 8.22 g/m3 in the mid-exposure group, and 28.48 to 28.79 g/m3 in the high-exposure group. Concentration–time curves of CyHx in rat blood and brain during uptake and elimination of CyHx for acute exposure are shown as individual data points in Figures 3 and 4. At the end of the third 8-hour day of exposure with CyHx, the blood and brain levels were no different than the 8-hour acute exposure measurement, and thus there was no evidence of induced metabolism (data not shown). As expected, concentrations of CyHx during the uptake phase were approximately 10-fold higher in the lipophilic brain compartment than the corresponding blood concentrations.
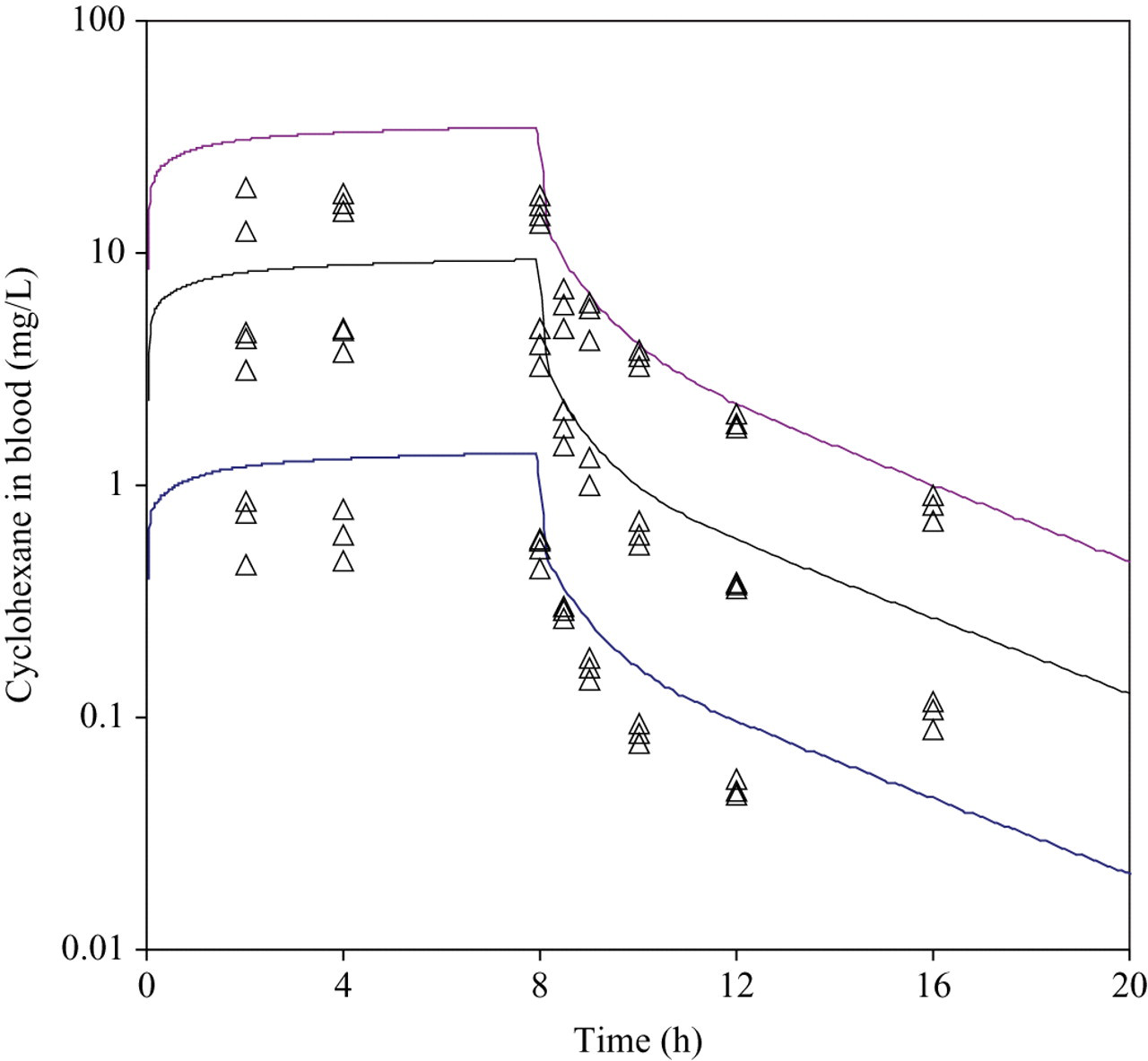
Measured (symbols) and predicted concentrations of cyclohexane in rat blood at 3 different exposure concentrations (1.4, 8, and 28 g/m3).
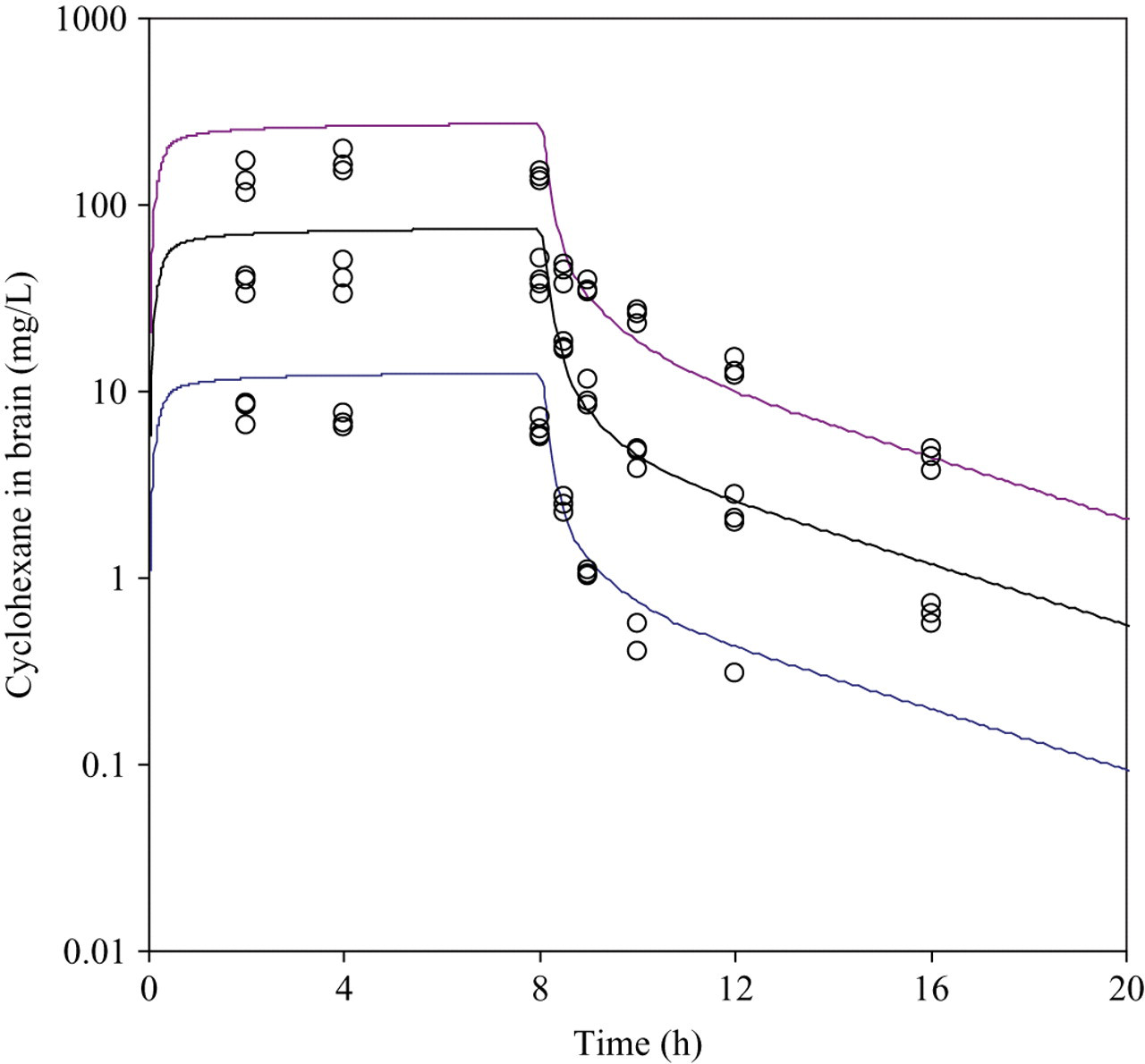
Measured (symbols) and predicted concentrations of cyclohexane in rat brain at 3 different exposure concentrations (1.4, 8, and 28 g/m3).
Examination of the figures shows that the exposure levels used in this study were probably sufficient to saturate CyHx metabolism. Concentrations of CyHx in blood measured during a single 8-hour exposure period were relatively high in the 28 g CyHx/m3 group, and the concentration/exposure ratio was higher in this group than in the other groups.
The predicted blood and brain concentrations were simultaneously calculated using the PBPK model for all 3 exposure levels. The Vmax and Km obtained from the gas uptake study were used to describe liver metabolism. In Figures 3 and 4, simulations are shown using the model parameters that are presented in Table 1. As is demonstrated, generally good fits were obtained for both the blood and brain concentrations. However, predicted concentrations during the 8-hour uptake were roughly double the experimentally determined values for both blood and brain.
The rat model was further validated against CyHx exhalation data identified from the literature. CyHx was eliminated primarily via exhalation of parent compound when 3 male Fisher 344 rats were administered 10 mg/kg 14 C-CyHx by intravenous injection. 25 The model predictions were consistent with the total CyHx exhalation, with the predicted exhalation occurring more rapidly than observed experimentally ( Figure 5).
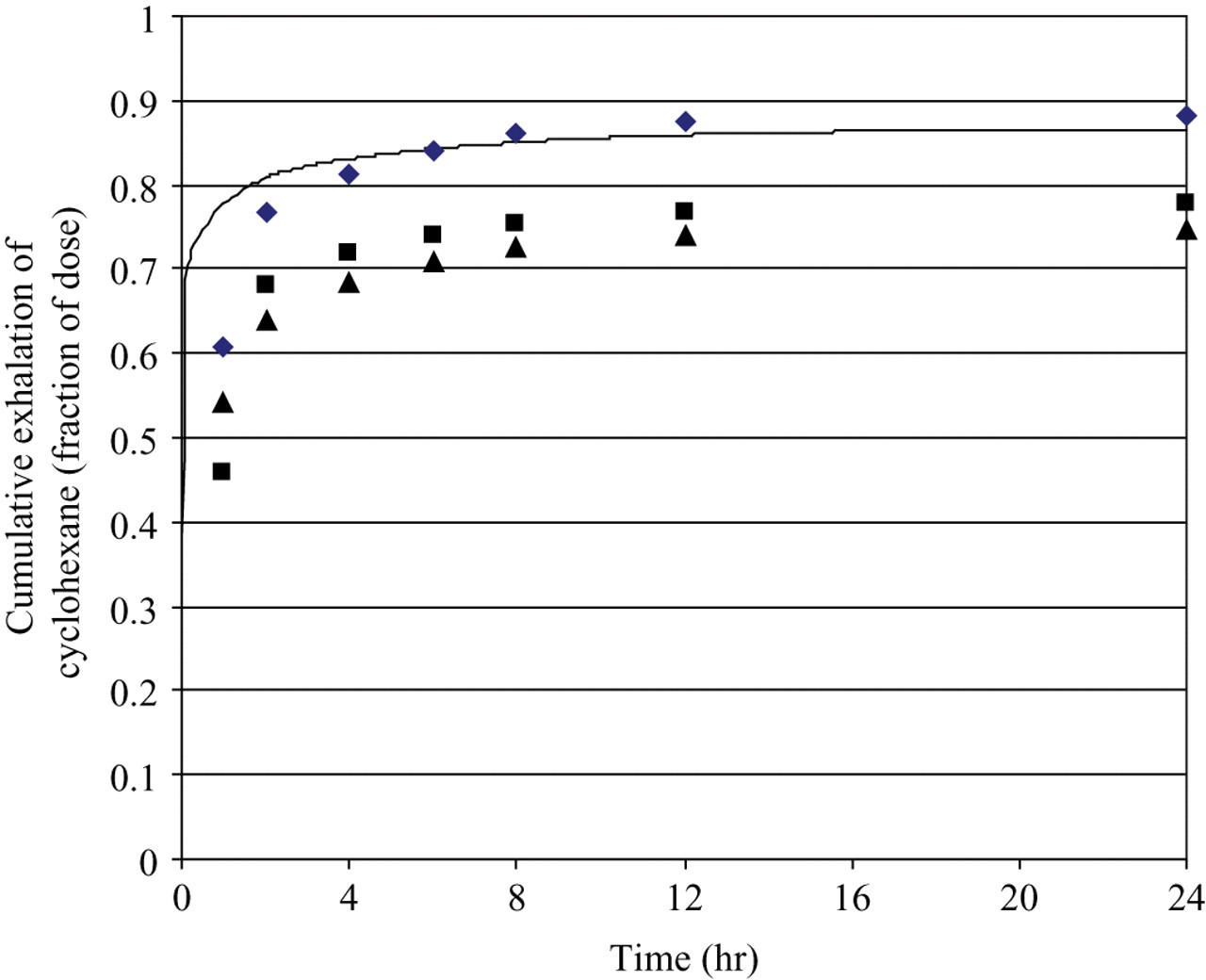
Cyclohexane exhalation by Fisher 344 rats (average body weight = 243 g) after intravenous injection of 10 mg/kg cyclohexane. Symbols represent experimental data. 25 The line represents the model prediction.
Sensitivity analyses were conducted to identify those model parameters with the greatest influence on the predicted concentrations of CyHx in blood (Cv) and brain (Cbr) of rats exposed to 8 g/m3 CyHx for 8 hrs. This concentration was selected because it corresponds to the NOAEL (No Observed Adverse Effect Level) for the rat behavioral toxicity study, and the dose metrics were selected because they were measured (both Cv and Cbr) and had relevance to the likely mode of action (Cbr). Sensitivity analyses were conducted at time points corresponding to the sampling times. NSCs were reported (Table 2) only if the absolute values of the NSC exceeded 0.05 for at least 1 time point. NSCs were generally similar at all sampling points during exposure, so only the 8-hour values were included for simplicity. At the end of exposure, predicted blood and brain concentrations were determined almost entirely by the chemical’s concentration and solubility.
Sensitivity of Rat Blood and Brain Cyclohexane Concentrations in Rats Exposed to 8 g/m3 Cyclohexane for 8 Hours to Changes in Model Parameters
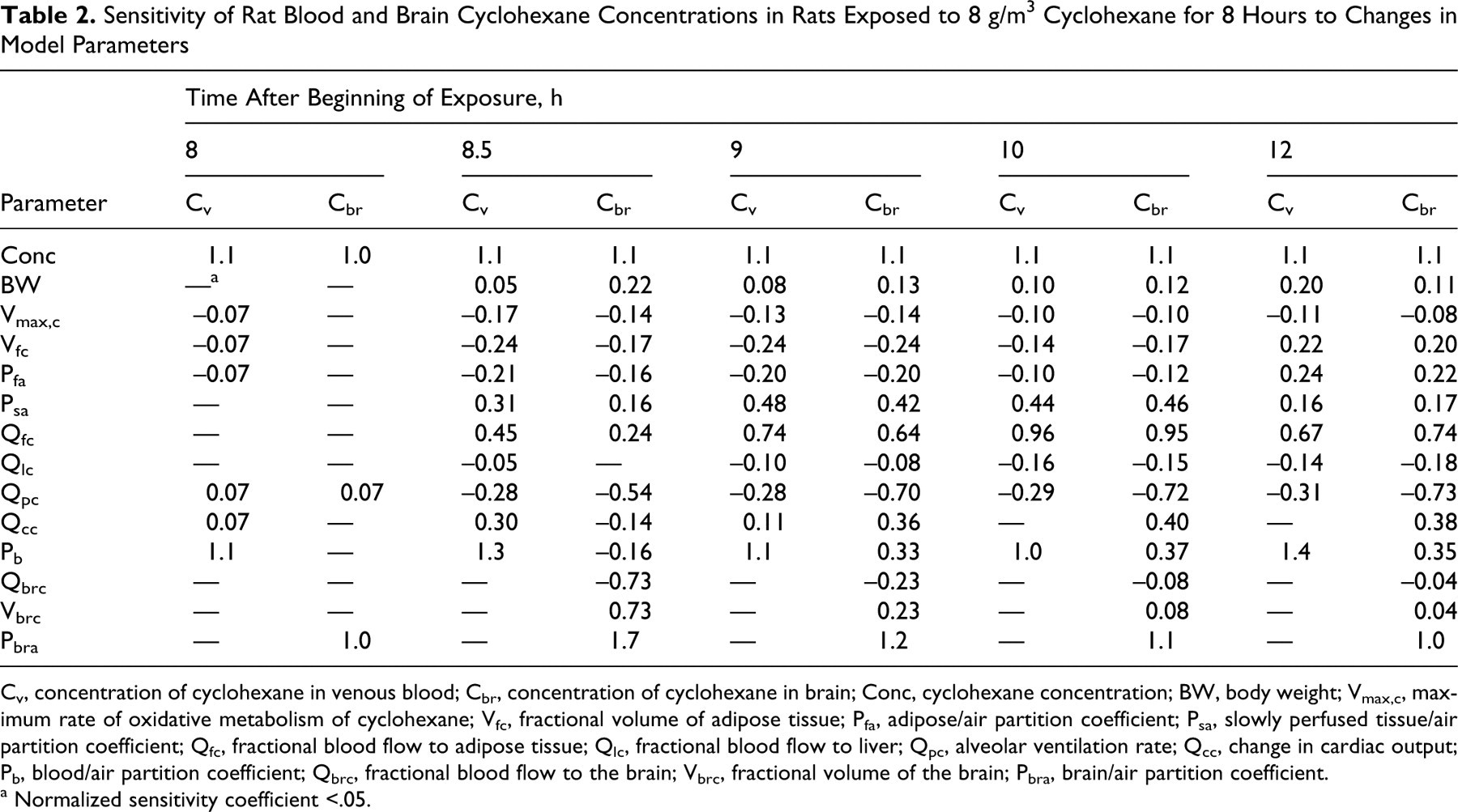
Cv, concentration of cyclohexane in venous blood; Cbr, concentration of cyclohexane in brain; Conc, cyclohexane concentration; BW, body weight; Vmax,c, maximum rate of oxidative metabolism of cyclohexane; Vfc, fractional volume of adipose tissue; Pfa, adipose/air partition coefficient; Psa, slowly perfused tissue/air partition coefficient; Qfc, fractional blood flow to adipose tissue; Qlc, fractional blood flow to liver; Qpc, alveolar ventilation rate; Qcc, change in cardiac output; Pb, blood/air partition coefficient; Qbrc, fractional blood flow to the brain; Vbrc, fractional volume of the brain; Pbra, brain/air partition coefficient.
a Normalized sensitivity coefficient <.05.
The parameters with |NSC| > 0.05 may be considered as 2 distinct groups. The first group of parameters (Conc through Qlc in Table 2) show similar NSC for blood and brain concentrations at a given time. For the most part, these parameters influence the total body burden of CyHx (storage in fat and muscle) rather than its distribution between blood and tissues. The remaining parameters differ in impact on blood versus brain concentrations. In the early part of the postexposure period, the changes in predicted brain concentration were determined by the tissue-specific perfusion and partitioning characteristics (brain blood flow, tissue volume, and tissue/air partition coefficient), which became less important at later times, as equilibrium among blood and tissues was reestablished. Cardiac output, breathing rate, and the blood/air partition coefficient were influential parameters during the postexposure period because of their importance to clearance via drawing CyHx from storage tissues into the blood, from which it can be exhaled or delivered to the liver for metabolism. In general, the metabolic rate was not a key determinant of blood or tissue CyHx concentrations for this rat exposure.
Human Kinetic Study and PBPK Modeling for CyHx in Humans
The measured cyclohexane concentration during exposure of human volunteers was 880.9 mg/m3 (range, 865.0-905.0 mg/m3).
Individual concentrations of CyHx in venous blood of the 3 volunteers are shown in Figure 6. Inhaled CyHx was rapidly taken up by all subjects, and interindividual variation was relatively small. A rapid first elimination phase followed by a somewhat slower phase was observed. The mean blood concentration for the 3 subjects after 2 hours of exposure was 617 ng/mL and was approximately 90% of the concentration measured at the end of the 4-hour exposure period (mean = 696 ng/mL). The concentration profile of CyHx in blood remained above the detection limit for approximately 7 hours after termination of exposure in 2 subjects. For the third subject, only 3 blood samples (t = 360, 480, and 765 minutes) were collected after exposure. The mean CyHx blood concentrations decreased rapidly and were 23.5% (151.5 ng/mL) and 10% (68 ng/mL) of the maximum measured concentrations 1 and 2 hours after termination of exposure, respectively. Thereafter, blood levels were near to or below the level of quantitation of 15 ng/mL.
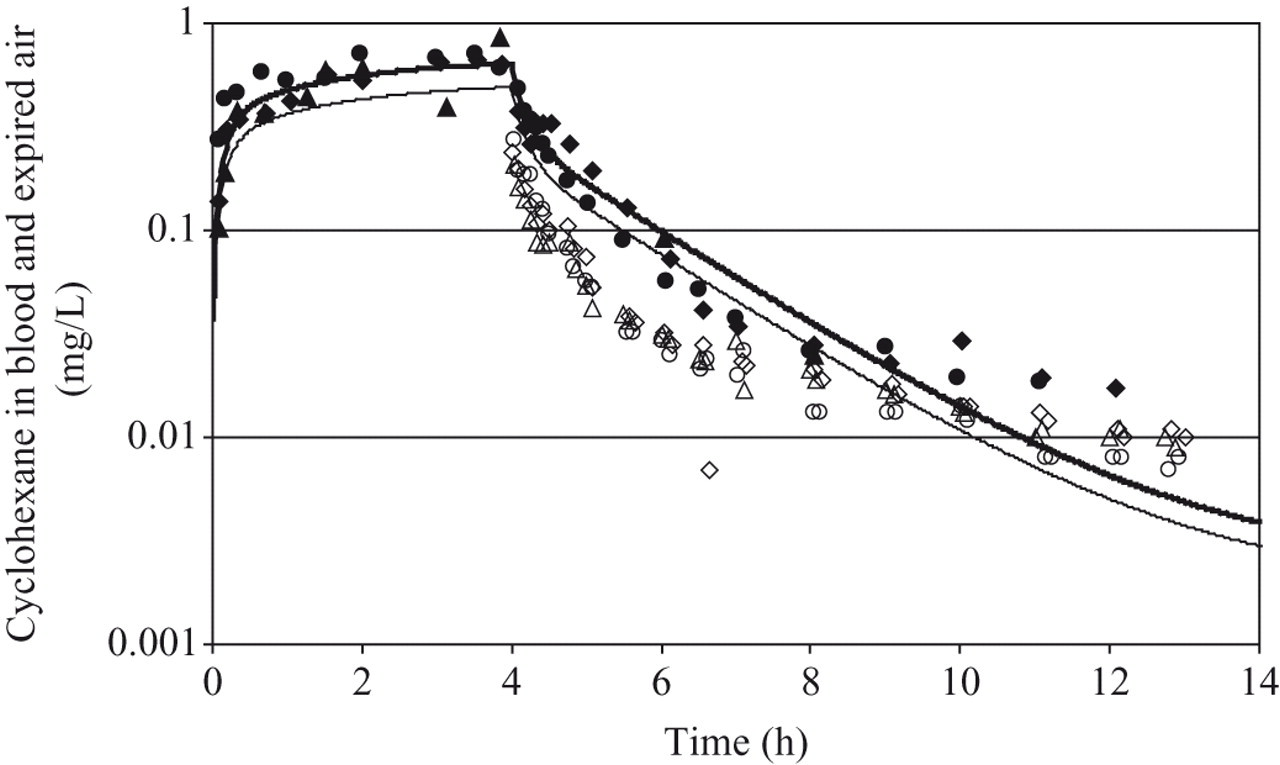
Measured (open symbols, exhaled air; filled symbols, blood) and predicted concentrations of cyclohexane in human blood (solid line) and end-exhaled air (dashed line) of 3 volunteers at an exposure concentration of 875 mg/m3 (∼250 ppm). Filled and open symbols of the same shape (ie, triangle, circle, or diamond) correspond to the same individual: triangle, volunteer 1; circle, volunteer 2; diamond, volunteer 3.
Individual concentration data of CyHx in exhaled alveolar air of the 3 subjects were also collected ( Figure 6). As with the blood data, interindividual variation was quite small. The concentrations of CyHx in exhaled alveolar air, measured in samples taken after the end of the exposure period, showed an initial rapid elimination phase followed by a slower second phase, as was seen with the blood concentration data. The mean exhaled alveolar air concentration, measured at the termination of exposure, was 240 μg/L. Exhaled alveolar air concentrations decreased by approximately 50% during the first 20 minutes following exposure. At 8 hours and 45 minutes after the end of exposure, the mean concentration in exhaled alveolar air was 9.33 μg/L. On the third day of the observation period (approximately 71 hours after exposure termination), the concentration of CyHx measured in exhaled alveolar air was below the limit of quantitation of 1 μg/L.
The parameters used in the PBPK model to describe CyHx kinetics in man are presented in Table 1. To describe metabolism, the parameter Vmax that was optimized for the rat model (5 mg/kg/h) was allometrically scaled as described in the Methods section. A Km of 0.1 mg/L was used, as for the rat model. With this model, simulations were performed to predict blood kinetics in humans, as for the rat, and concentrations in alveolar air. These simulations are shown in Figure 6, along with the experimental data. Predictions agreed well with the measured blood concentrations, however, exhaled air concentrations were overestimated by approximately a factor of 2.
Discussion
The CyHx PBPK models presented here were developed to facilitate the comparison of no-effect levels for CNS end points evaluated in the rat to occupational exposure levels in humans. As such, the focus was on levels of parent compound in blood and tissues in environments with stable exposure levels. Under these circumstances, blood and tissue levels appear to be determined largely by partitioning (solubility), with a limited role for metabolism in influencing total CyHx body burden. Therefore, the current model can be confidently used for estimation of blood and tissue levels of CyHx in rats and humans during and after acute or repeated exposure to CyHx in the range of concentrations described here. Although the model would be expected to provide reasonable estimates of the total metabolism of CyHx for rats and humans, the model does not characterize the further disposition of the CyHx metabolite cyclohexanol. Indeed, differences between humans and other species (eg, rabbits and dogs) have been noted for the excretion of down-stream metabolites of cyclohexanol. 31
To obtain input parameters for the PBPK model for CyHx, the closed chamber gas exchange technique described by Gargas and colleagues 18,22 was used. For highly metabolized compounds, parameters describing metabolism are of major importance as input parameters in PBPK models. Metabolism of CyHx was assumed to occur by saturable biotransformation in the liver, which can be described by Michaelis-Menten kinetics. 25 The values of the metabolic rate constants for CyHx were estimated for the rat by optimization to obtain the best simultaneous visual fit to the set of gas uptake curves. Consistent with biotransformation of CyHx by a single saturable process, these simulations produced a value for the maximum metabolic rate, Vmax,c, of 5.4 mg/(hc·kg0.7). The value obtained for the Michaelis-Menten constant, Km, was 0.13 mg/L. The kinetic parameters determined for CyHx using this approach are of the same order of magnitude as those found for toluene and m-xylene 32 and for several other compounds studied by Gargas and colleagues. 18,22
The accuracy to which this PBPK model predicted concentrations of body burdens of CyHx in rats and humans was subsequently tested by comparing the concentration–time curves of the parent compound in rats and in humans with those predicted by the model. For the rat, the model tended to overestimate the experimental values during exposure. However, there was excellent agreement between predicted values for both blood and brain in the postexposure period. For the postexposure period, the model predictions were, on average, within a factor of 1.4 of the mean of the experimental data, and in only 1 instance did the rat model differ by a factor of 2 or more. The small difference between predicted values and experimental data collected during exposure to CyHx may be due to the short delay between the termination of exposure and sacrifice of the animals because there is a rapid decrease in tissue concentrations immediately following the termination of exposure. Comparison of the experimental data and model predictions for the experimental data at t = 8 hours suggests a collection delay of 8 minutes for blood and 11 minutes for brain. Because blood and brain kinetics were well predicted for the elimination phase, it was concluded that the kinetic parameters estimated from the gas uptake study could be used to predict concentrations of body burdens of CyHx in the rat.
With respect to the validation of the human PBPK model, results indicated good agreement between predicted and measured concentrations of CyHx in blood. However, predicted values of exhaled air concentrations were higher by a factor of about 2 compared with the measured concentrations. A number of different values have been reported for the blood/air partition coefficient of CyHx. 22,28,33,34 The difference between the accuracy in the predicted blood concentration and predicted alveolar air concentration may have resulted from the blood/air partition coefficient of 1.3 used in the model. 28 However, the experimentally determined values for the concentrations in expired air compared with those in blood point to somewhat greater partitioning. In contrast to the value of 1.3 (human) obtained by Perbellini 28 and the value of 1.4 (equal values for male F344 rats and humans) published by Gargas et al, 34 Imbriani et al 33 published a human blood/air partition coefficient of 2.4. When this higher value was used in the present model, predicted concentrations were indeed closer to the experimental values for the exhaled air concentrations obtained in the present experiment. With a higher blood/air partition coefficient, however, human blood concentrations, particularly during exposure, were greatly overestimated. Moreover, the value of 2.4 for the blood/air partition coefficient gave poor results for the rat model predictions. Hypothetically, the ratio of the blood and end-exhaled air values should be constant and equal to the blood air partition coefficient, but inspection of the data indicates that the ratio changes during the postexposure period. Thus, the differences between model predictions and measured values are likely to involve factors other than blood/air partitioning. Because PBPK models are typically validated on the basis of blood concentrations rather than exhaled air 35 and the model reported here adequately predicted human blood levels as well as rat brain and blood levels, it was concluded that the present model was adequate for its intended use in cross-species extrapolation.
A central issue in the use of PBPK modeling in the present project is its application in predicting exposure levels at which CNS effects would be expected. To achieve this, a similar brain concentration–effect relationship was assumed for rat and human. Results from previous effects testing studies in the rat demonstrated that exposure to 28 g/m3 CyHx (∼8000 ppm) for 8 hours resulted in small but statistically significant effects in measures of performance, whereas exposure to 8 g/m3 (2286 ppm) produced no effects. 12 This exposure level corresponded to an experimentally measured brain concentration of approximately 125 mg/L, as shown in Figure 4. At the 8 g/m3 exposure level, a brain concentration of 36.2 mg/L was measured. Using the PBPK model described in the present report, it was calculated that for humans exposed for 8 hours, the external concentrations of CyHx corresponding to these brain levels were 13.7 g/m3 and 4.1 g/m3, respectively. If it is then assumed that there are similar concentration–effect relationships in rats and humans, the predicted human-equivalent concentrations for the NOEL and LOEL values for CNS effects are approximately 4100 and 13 700 mg/m3. Accordingly, the margin of exposure between the current occupational exposure level (in The Netherlands) of 860 mg/m3 and the human equivalent concentration for acute CNS effects would be approximately 5-fold. In volunteer studies, no acute CNS effects were associated with exposure to 860 mg/m3. 12
Although a number of human experimental exposure studies have been conducted to examine the acute neurobehavioral effects of solvents, 7 the practical and sometimes ethical constraints of such studies mean that the results of animal studies with neurobehavioral end points will continue to be used in setting occupational exposure limits. In this respect, PBPK models may serve as an excellent tool in the risk assessment process because uncertainties in cross-species extrapolation can be minimized. 36
The present report demonstrates that a PBPK model for CyHx, validated in the rat, can be used as a tool to extrapolate animal test data to humans. An important feature of PBPK modeling is that target tissue doses that cannot be directly measured in humans may be calculated. Correlating these target tissue doses to tissue dose–effect relationships found in animal tests may provide a scientific basis for the establishment of occupational exposure levels of chemical compounds and may add significantly to the process of chemical risk assessment.
Footnotes
Acknowledgement
The authors gratefully acknowledge the assistance of L. H. Leenhers in the determination of partition coefficients.
Two of the authors (RHM, DEO) are employed by companies that manufacture cyclohexane.
This work was sponsored by the CEFIC Hydrocarbon Solvent Producers Association and a grant (to Dr. Beverly M. Kulig) from The Netherlands Ministry of Economic Affairs.
