Abstract
Neuropathy target esterase has been shown to be a lysophospholipase in mouse. The authors investigate the effect of neuropathy target esterase inhibition in mouse nervous tissues in vitro on the homeostasis of phosphatidylcholine and lysophosphatidylcholine by treating the homogenates with tri-ortho-cresyl phosphate, paraoxon, paraoxon plus mipafox, and phenylmethylsulfonyl fluoride. The activity of neuropathy target esterase is significantly inhibited by phenylmethylsulfonyl fluoride and paraoxon plus mipafox but not by paraoxon alone. Tri-ortho-cresyl phosphate slightly but significantly inhibits neuropathy target esterase activity in brain. The levels of phosphatidylcholine and lysophosphatidylcholine in all 3 nervous tissues are not obviously altered after treatment with tri-ortho-cresyl phosphate, paraoxon, or paraoxon plus mipafox. However, phosphatidylcholine and lysophosphatidylcholine levels are clearly enhanced by phenylmethylsulfonyl fluoride. It is concluded that inhibition of neuropathy target esterase in mouse nervous tissues is not enough to disrupt the homeostasis of phosphatidylcholine and lysophosphatidylcholine and that the upregulation by phenylmethylsulfonyl fluoride may be the consequence of combined inhibition of neuropathy target esterase and other phospholipases.
Some organophosphorus compounds (OPs) are able to induce a delayed neurodegenerative condition known as OP-induced delayed neurotoxicity (OPIDN), which affects both the central and the peripheral nerves of birds and mammals. 1,2 Inhibition of neuropathy target esterase (NTE) has been proposed as the initial effect of OPs that induce OPIDN. 3-5 NTE is a nerve protein intimately associated with the cytoplasmic face of the endoplasmic reticulum. 6,7 The molecular basis of this disorder remains largely unresolved. 8 Nevertheless, significant progress has been achieved recently on the biochemical function of NTE that provides new clues to possible mechanisms of OPIDN. NTE may act as a kind of lysophospholipase (LysoPLA) with lysophosphatidylcholine (LPC) as its physiological substrate in mouse brain. 9 In mammalian cell lines and yeast, NTE has phospholipase B (PLB) activity, and its biochemical function is concerned with membrane phosphatidylcholine (PC) homeostasis. 10,11 These discoveries suggest that inhibition of NTE may affect the homeostasis of PC and LPC.
PC is the major phospholipid of eukaryotic cells and represents about 50% of the membrane phospholipids in animal cells. 12 Regulation of the PC biosynthesis, degradation, and relative distribution of different membranous structures is critical for cellular function. 13 PC homeostasis is regulated by a balance between the opposing actions of hydrolysis and synthesis. 14 PC is mainly synthesized from the cytidine-5′-diphospho-choline (CDP-CL) pathway via a phosphorylcholine intermediate for condensation with diacylglycerol to produce PC. 15-17 PC can be hydrolyzed by phospholipase A (PLA), PLB, and phospholipase D (PLD). 18 NTE is a devoted PLB against only CDP-CL pathway-derived PC, whereas PC molecules formed through the methylation pathway are not degraded. 10,19 LPC is another important phospholipid molecule in mammalian tissues. An elevated LPC level can induce neuronal sheath demyelination, together with a variable degree of axonal degeneration, 20,21 which is similar to the pathological changes of OPIDN. LPC can be deacylated by PLA and NTE or other LysoPLAs to form free fatty acids and glycerophosphocholine (GPC) (see Figure 1 for the pathway of NTE participating in PC metabolism).
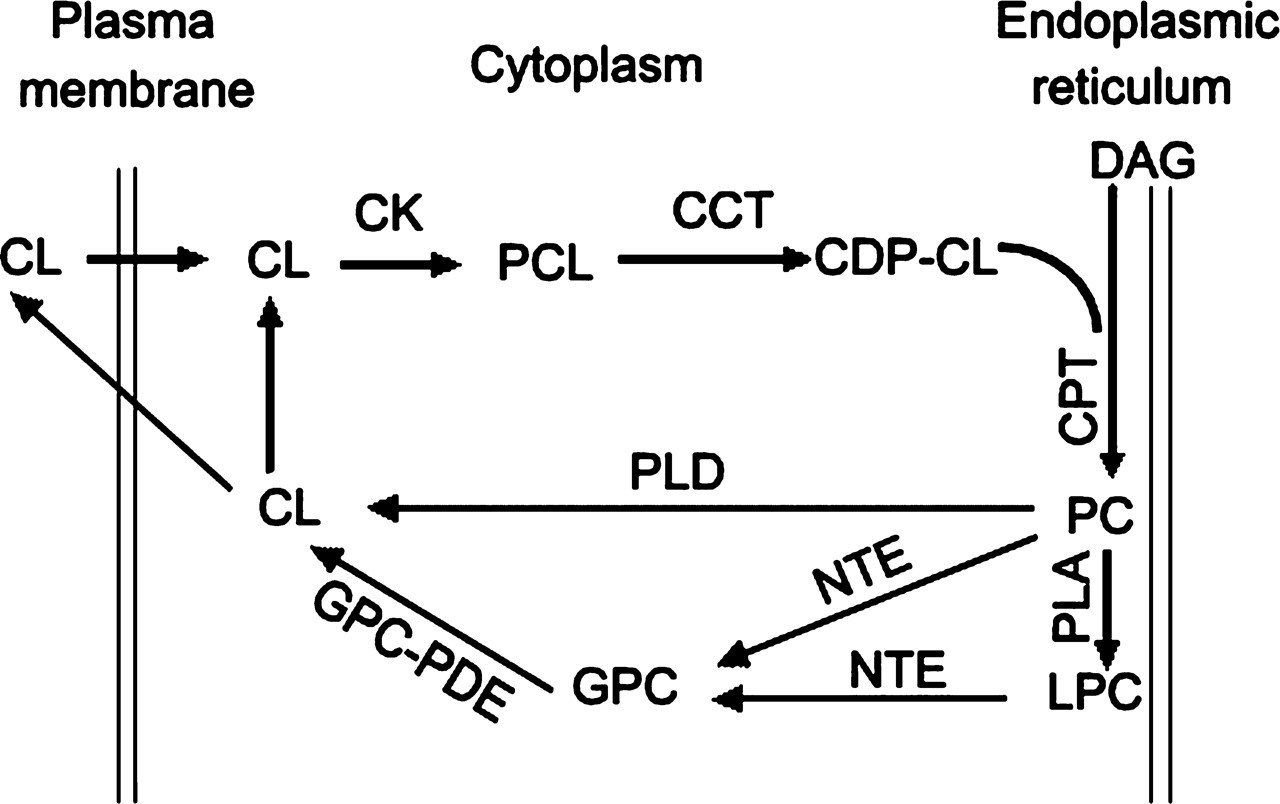
The pathway of neuropathy target esterase (NTE) participating in phosphatidylcholine (PC) metabolism. Choline (CL) is transported into cells and then phosphorylated by choline kinase (CK). Phosphocholine (PCL) reacts with cytidine triphosphate (CTP) in the pathway’s rate-limiting step catalyzed by CTP/phosphocholine cytidylyltransferase (CCT), forming cytidine-5′-diphospho-choline (CDP-CL). CDP-CL reacts with diacylglycerol (DAG) catalyzed by choline phosphotransferase (CPT) to form membrane-associated phosphatidylcholine (PC). PC can be hydrolyzed by phospholipase D, forming phosphatidic acid and CL. PC can also be deacylated by NTE at the cytoplasmic face of the endoplasmic reticulum to form soluble products: free fatty acids, and glycerophosphocholine (GPC). In addition, phospholipase A and NTE can sequentially deacylate PC to GPC. GPC is hydrolyzed by glycerophosphorylcholine phosphodiesterase (GPC-PDE), forming CL and glycerophosphate. CL can be phosphorylated or secreted from the cell. The above information is taken from references 9, 10, 11, and 37.
Tri-ortho-cresyl phosphate (TOCP) has been the prototype delayed neurotoxicity–inducing agent, because it can inhibit and age NTE. TOCP requires metabolism to saligenin cyclic-o-tolyl phosphate, the active neurotoxic metabolite of TOCP to elicit OPIDN. 22 Phenylmethylsulfonyl fluoride (PMSF) cannot initiate OPIDN because NTE inhibited by PMSF does not undergo aging. 3 However, PMSF can modify the clinical effects of both toxic and traumatic insults. 1 PMSF protects the animal from OPIDN when given before the neurotoxic OPs yet exacerbates the polyneuropathy when given after OP administration. 23,24 Mipafox (MPX) can inhibit NTE and cause OPIDN in susceptible species, whereas paraoxon (PXN) is a nonneuropathic OP that reacts more readily with acetylcholinesterase than NTE and causes acute cholinergic toxicity rather than delayed neuropathy. 25 MPX and PXN are used in biochemical assays to operationally define NTE activity as PXN-resistant, MPX-sensitive, phenyl valerate-hydrolyzing activity. 26,27
In the present study, homogenates of mouse brain, spinal cord, and sciatic nerve were treated with TOCP, PMSF, PXN, and PXN+MPX to investigate the effect of NTE inhibition on the homeostasis of PC and LPC.
Method
Reagents
TOCP was purchased from BDH Chemicals (Poole, UK). Coomassie brilliant blue G-250 and PMSF were purchased from Fluka Chemika (Buchs, Switzerland). MPX and phenyl valerate were synthesized in our laboratory as described previously. 28 PXN, PC, LPC, and GPC standards were purchased from Sigma (St. Louis, Mo). Silica gel 60 F254 plates (20 × 20 × 0.25 mm) were purchased from Merck (Darmstadt, Germany).
Animals and Nervous Tissue Samples Preparation
Adult Kunming mice (20-25 g) used in this study were obtained from the Animal Center, Institute of Genetics and Developmental Biology, Chinese Academy of Sciences. They were maintained under standard conditions with access to water and food ad libitum. The animals were acclimatized for 1 week prior to the start of the experiment. Mice were sacrificed by cervical decapitation, and then brain, spinal cord, and sciatic nerves were quickly dissected on ice, meninges were removed, and wet weight was recorded. All animal procedures were performed in accordance with current Chinese legislation and approved by the Institute of Zoology Animal and Medical Ethics Committee. Tissues were homogenized in 10% (wt/vol) TE buffer (50 mM Tris-HCl, 0.2 mM EDTA, pH 8.0) at 4°C.
Treatment of Mouse Nervous Tissues Homogenate With Toxicants In Vitro
The tissue homogenates prepared as above were preincubated with 0.1% dimethyl sulfoxide (DMSO) (as control), 1 mM TOCP, 1 mM PMSF, 40 µM PXN, and 40 µM PXN plus 50 µM MPX (all concentrations are final, and all toxicants were dissolved in DMSO) for 30 minutes at 37°C in 5 tubes under gentle agitation. Homogenates were then centrifuged at 100 g for 5 minutes to remove large debris. Aliquots of the resulting supernatant were collected for determination of protein concentrations and NTE activities; the remaining homogenates were used for lipids extraction.
NTE Activity Assay
NTE activity was assayed in mouse nervous tissues as the difference in phenol liberated from the PXN-resistant and MPX-sensitive hydrolysis of phenyl valerate according to Johnson 27 with modification for reduced volume microassay, as previously described in our laboratory, and expressed as nanomoles of phenol formed per minute per milligram of protein with phenol as the standard. 29 Protein concentrations in the homogenates were measured by the method of Bradford using bovine serum albumin as standard. 30
Lipids Extraction
Lipids were extracted from the tissue homogenates (prepared as described above) with a chloroform/methanol mixture (2:1, vol/vol) using the procedure of Folch et al 31 with some modifications. Briefly, tissue homogenate was centrifuged at 3000 rpm for 10 minutes. The supernatant (S1) was removed, and a 5-fold its volume of chloroform/methanol mixture (2:1, vol/vol) was added and stirred vigorously. After centrifugation (1000 rpm, 5 minutes), the lower organic phase (L1) was recovered. The sediment (P1) was homogenized again in 20-fold volume chloroform/methanol mixture (2:1, vol/vol) of the tissue sample weight at 4°C, and the supernatant (S2) was recovered after centrifugation (3000 rpm, 10 minutes). The sediment (P2) was re-extracted in 10-fold volume chloroform/methanol mixture (2:1, vol/vol) of the tissue sample weight, and supernatant (S3) was recovered after centrifugation (3000 rpm, 10 minutes). A 0.2-fold volume of 0.1 M KCl was added in combined S2 and S3 and stirred vigorously. The lower organic phase (L2) was recovered after centrifugation (1000 rpm, 5 minutes). Then L1 and L2 were combined. Organic extracts were evaporated to dryness under a nitrogen stream. The lipid extract was redissolved in a small volume of chloroform/methanol (1:1, vol/vol) for thin-layer chromatography (TLC) analysis.
Separation of Phospholipids by TLC
Analysis of phospholipids was achieved by 1-dimensional TLC on silica gel 60 plates using chloroform/methanol/acetic acid/acetone/water (40:25:7:4:2, v/v) as the solvent according to the method of Wang and Gustafson. 32 The individual phospholipids were visualized on TLC plates by staining with iodine vapor. The bands corresponding to PC and LPC (based on comigration with standards) were scraped into glass tubes for quantification.
Measurement of Inorganic Phosphorus
Inorganic phosphorus in each phospholipid fraction was measured using the method of Vaskovsky et al 33 with some modifications. Briefly, 0.2 mL of 72% HClO4 was added to the scrapings, the tubes were incubated at 190°C for 20 minutes in an electrically heated metal block, and then the temperature was elevated to 220°C to allow the HClO4 to evaporate completely. After cooling, 0.3 mL water and 2.7 mL of the sulfuric acid-sodium molybdate solution were added and mixed thoroughly and then heated in a boiling water bath for 5 minutes. After centrifugation (1500 rpm, 5 minutes), the absorbance of the supernatant was measured at 815 nm against a blank. The concentrations of inorganic phosphorus were calculated according to a standard phosphorus curve. The amount of each phospholipid was calculated using the amount of recovered phosphate in each spot, and the results were expressed as nanomoles or micromoles of phosphorus per gram of wet weight of the nervous tissues.
Statistical Analysis
Data were compared by 1-way analysis of variance using SPSS 13.0, followed by a Student-Newman-Keuls post hoc test. A difference between means was considered significant at P < .05.
Results
TOCP, PMSF, and PXN+MPX Inhibit NTE Activity of Mouse Nervous Tissues In Vitro
To examine the effect of TOCP, PMSF, PXN, and PXN+MPX on NTE activity in vitro, homogenates of mouse brain, spinal cord, and sciatic nerve were treated with these toxicants, and then the enzyme activity was determined. As shown in Figure 2, the NTE activity in all 3 tissues was clearly inhibited by PMSF and PXN+MPX; the activity decreased by 93.4% (P < .01), 93.5% (P < .01), and 87.6% (P < .05), respectively, in brain, spinal cord, and sciatic nerve after PMSF treatment and by 86.0% (P < .01), 96.1% (P < .01), and 83.8% (P < .05), respectively, after treatment with PXN+MPX. There was no significant difference between the inhibition of NTE activity in the same nervous tissue between the 2 treatment groups of PMSF and PXN+MPX. TOCP led to a slight but significant reduction of NTE activity in brain. This activity decreased by 17.7% (P < .05); however, there was no significant alteration of NTE activity in spinal cord and sciatic nerve after TOCP treatment. No significant inhibition of NTE activity was observed in any of tissues after PXN treatment.
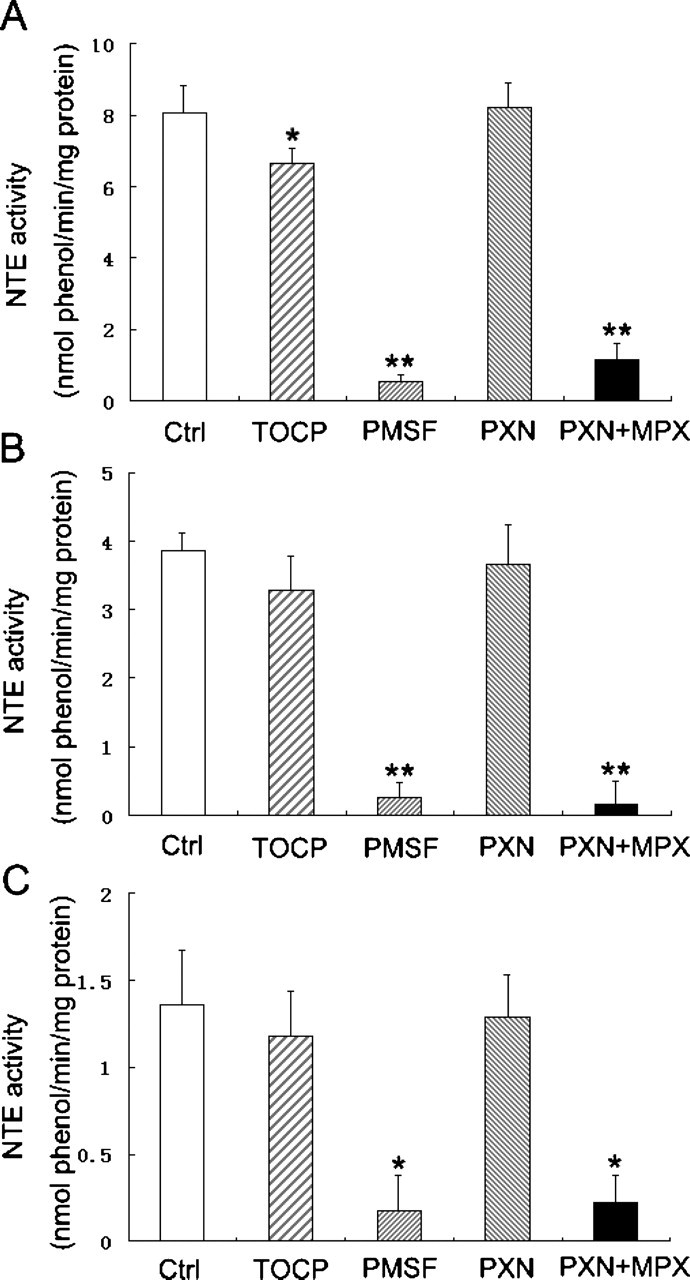
The alteration of neuropathy target esterase (NTE) activity in mouse nervous tissues after treatment with tri-ortho-cresyl phosphate (TOCP), phenylmethylsulfonyl fluoride (PMSF), paraoxon (PXN), and PXN + mipafox (MPX). The homogenates of (A) mouse brain, (B) spinal cord, and (C) sciatic nerve were treated with TOCP (1 mM), PMSF (1 mM), PXN (40 µM), and PXN (40 µM) + MPX (50 µM) for 30 minutes at 37°C, and then the NTE activity was determined as described under Method. Data were expressed as mean ± SD of 3 independent experiments. *Significantly different at P < .05 and **significantly different at P < .01 compared with control.
PMSF Treatment Enhances the PC and LPC Levels of Mouse Brain and Spinal Cord In Vitro
To investigate the effect of NTE inhibition in vitro on the homeostasis of PC and LPC in nervous tissues, the levels of these 2 phospholipids in mouse brain, spinal cord, and sciatic nerve were measured after TOCP, PMSF, PXN, and PXN+MPX treatment. As shown in Figure 3, the levels of PC and LPC in all of the tissues did not significantly change after treatment of TOCP, PXN, and PXN+MPX. However, significant alterations were observed in PC and LPC levels in brain and spinal cord after PMSF treatment, and the levels of these 2 phospholipids were enhanced by 28.9% (P < .05) and 22.5% (P < .05), respectively, in the brain and 30.3% (P < .05) and 20.3% (P < .05), respectively, in the spinal cord. Nevertheless, the levels of PC and LPC in the sciatic nerve were not significantly altered after PMSF treatment.
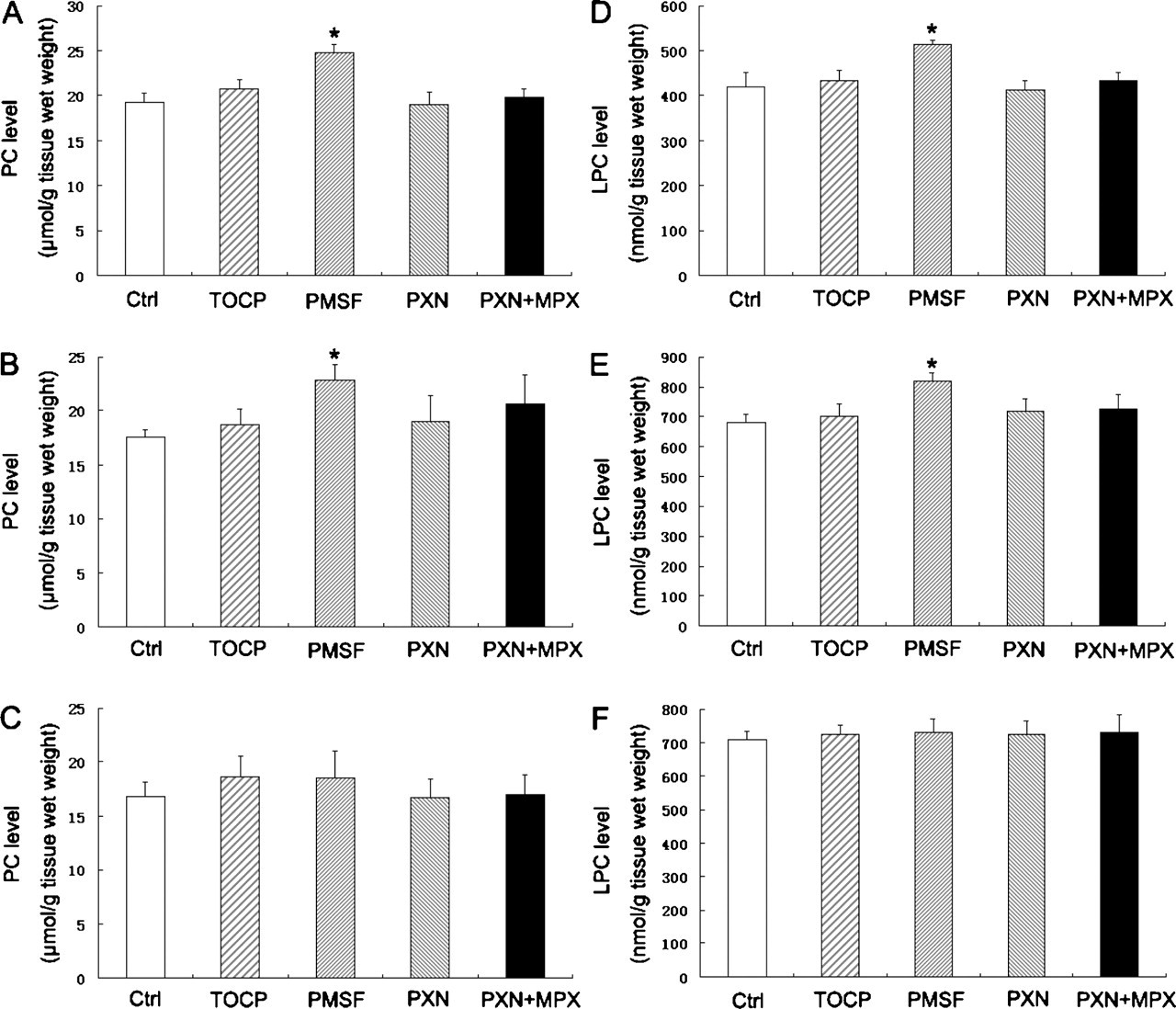
The alteration of phosphatidylcholine (PC) and lysophosphatidylcholine (LPC) levels in mouse nervous tissues after treatment with tri-ortho-cresyl phosphate (TOCP), phenylmethylsulfonyl fluoride (PMSF), paraoxon (PXN), and PXN + mipafox (MPX). The homogenates of mouse brain (A, D), spinal cord (B, E), and sciatic nerve (C, F) were incubated at 37°C for 30 minutes with TOCP (1 mM), PMSF (1 mM), PXN (40 µM), and PXN (40 µM) + MPX (50 µM), and then the contents of PC (A, B, C) and LPC (D, E, F) were measured as described under Method. Data were expressed as mean ± SD of 3 independent experiments. *Significantly different at P < .05 compared with control.
Discussion
NTE is a neural protein with esterasic activity and is anchored to the cytoplasmic face of the endoplasmic reticulum. 6,7 NTE is mainly present in nervous tissues, although significant amounts of NTE are also present in some nonneural tissues, particularly intestine, kidney, and lymphocytes. The highest specific activities of NTE are found in brain tissue, with substantially less in spinal cord and sciatic nerve, in which relative specific activities are about 20% to 30% and 3% to 5% of brain in the hen. 5,34 Here, we confirmed these early observations in the mouse. The NTE activity in the brain is much higher than in the spinal cord and sciatic nerve.
Our initial goal was to evaluate the effect of NTE on the homeostasis of PC and LPC in vitro by comparing the effect of several different toxicants on PC and LPC levels when NTE activity was mostly inhibited. No significant NTE activity inhibition was observed when the mouse brain homogenate was treated with TOCP at less than 1 mM, whereas the activity was significantly inhibited after exposure to 1 mM TOCP. However, NTE activity was not further inhibited even with exposure to more than 1 mM TOCP. Thus, 1 mM TOCP was used in this experiment. NTE activity was partially inhibited when the tissue was treated with PMSF at a concentration of less than 1 mM, whereas more than 90% of NTE activity was inhibited after treatment with 1 mM PMSF, the concentration of PMSF chosen for use in the experiments. NTE activity was operationally defined as PXN-resistant, MPX-sensitive phenyl valerate–hydrolyzing activity. In the assay, 40 µM PXN almost inhibited all of phenyl valerate–hydrolyzing activity except NTE activity, and 40 µM PXN plus 50 µM MPX almost inhibited all phenyl valerate–hydrolyzing activity. 26,27 Therefore, 40 µM PXN and 50 µM MPX were chosen. The NTE activity in mouse brain, spinal cord, and sciatic nerve was clearly inhibited in vitro by TOCP, PMSF, and PXN+MPX; however, TOCP was a relatively less potent NTE inhibitor than PMSF and MPX. Only 17.7% of NTE activity in the brain was inhibited, whereas no significant change was observed in the spinal cord and sciatic nerve after TOCP treatment. This may be because TOCP requires metabolic activation and cannot be fully transformed to active neurotoxic metabolite in a short assay time of 30 minutes. 22 Mouse brain may contain higher enzymatic activity for TOCP metabolism than the spinal cord and sciatic nerve. However, PMSF was a very potent inhibitor of NTE and inhibited approximately 90% of the NTE activity in the nervous tissues. PMSF led to an increase in the contents of PC and LPC in the brain and spinal cord, whereas TOCP did not, although it inhibited NTE activity in the brain. However, we could not conclude whether NTE plays a key role in PC and LPC homeostasis, because PMSF, a broad-spectrum protease inhibitor, may inhibit the activities of other phospholipases besides NTE and because TOCP only slightly inhibited NTE activity. To investigate whether the inhibition of NTE was the reason for the increased content of PC and LPC in mouse brain after PMSF treatment, the mouse nervous tissue homogenates were treated with PXN (40 µM) and PXN (40 µM) + MPX (50 µM). Both PXN and MPX can inhibit many esterases, and a difference between these 2 toxicants is that MPX can inhibit NTE whereas PXN cannot. No significant NTE inhibition was observed, and the contents of PC and LPC were not significantly altered in mouse nervous tissues after PXN treatment. Similar to PMSF, PXN+MPX potently inhibited the NTE activity of mouse brain, and no significant difference of NTE inhibition was observed between treatment with PMSF and treatment with PXN+MPX. However, PXN+MPX did not enhance levels of PC and LPC in mouse brain compared with the control group or the PXN-only treatment group. These results indicate that inhibition of NTE in mouse nervous tissues in vitro is not enough to disrupt the homeostasis of PC and LPC. The increase of PC and LPC contents caused by PMSF in mouse brain and spinal cord may be the consequence of combined inhibition of NTE and other phospholipases. Previous studies showed that NTE, as a lysophospholipase, accounts for only 17% of total lysophospholipase in mice brain. 35 This suggests that NTE may not be a central phospholipase involved in PC and LPC metabolism. PMSF did not alter PC and LPC levels in the sciatic nerve, although it potently inhibited the NTE activity, which may be attributable to the relatively low level of phospholipases in sciatic nerve. Although some phospholipids may be hydrolyzed enzymatically in axons, many phospholipids presumably return to the nerve cell bodies to be metabolized. 36
We investigated the effect of NTE inhibition on the homeostasis of PC and LPC in mouse nervous tissues in vitro based on recent data showing that NTE may act as a LysoPLA or PLB. PMSF led to an increase in the contents of PC and LPC in the brain and spinal cord, whereas MPX did not. We conclude that inhibition of NTE did not disrupt the homeostasis of PC and LPC in mouse nervous tissues under the present experimental conditions.
Footnotes
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (30470228, 30870537) and a grant from National High Technologies R&D Program (2006AA06Z423).
