Abstract
Modulating the bone morphogenetic protein 2 (BMP-2) and transforming growth factor–β1 (TGF-β1) signaling pathways is essential during tendon/ligament (T/L) healing. Unfortunately, growth factor delivery in situ is far from trivial and, in many cases, the necessary growth factors are not approved for clinical use. Here we used a BMP-2 and a TGF-β1 reporter cell line to screen a library of 1280 Food and Drug Administration–approved small molecules and identify modulators of both signaling pathways. We identified four compounds capable of modulating BMP and TGF signaling on primary human tendon–derived cells (huTCs) and describe their effects on proliferation and differentiation of these cells.
Introduction
Despite significant advances in unraveling the mechanisms governing tendon/ligament (T/L) healing and improving the clinical management of these injuries, the outcome is far from ideal. Animal and cellular models developed to identify the molecular drivers of T/L healing and repair lend evidence to the importance of specific growth factors in the process. Secreted growth factors (GFs) such as bone morphogenetic protein (BMP) and transforming growth factor–β (TGF-β) have been identified as key players in the process of T/L specification during embryonic development as well as on the healing and repair upon injury.1,2 Several reports demonstrated that GFs promoted cell differentiation, upregulated cartilage/bone markers known to be involved in enthesis formation and induce cytoskeletal organization, thus contributing to the restoration of tissue strength and functionality.3–5 Nonetheless, the concentration and spatial distribution of these endogenous GFs during T/L healing are not sufficient to shorten the repair period that may take from several months to a year. 6 In an attempt to overcome these limitations, several groups suggested that a combination of scaffolds and recombinant human GFs, such as rhTGF-β1 and rhBMP-2, could accelerate T/L healing.7,8 However, even though recombinant human material has shown great potential in tissue regeneration, their biological nature poses several challenges for their production regarding batch variability, stability, biological activity, and production costs, 9 emphasizing the need for an alternative such as the identification and/or development of small molecules with the desired biological properties.
Consequently, we screened a library of 1280 Food and Drug Administration (FDA)–approved small molecules using a BMP-2 and a TGF-β1 reporter cell line and identified small molecules capable of modulating both pathways. Subsequent assays used human tendon–derived cells (huTCs) to investigate the effect of the selected molecules in huTC metabolism as well as differentiation potential. A schematic overview of the experiment design is represented in Figure 1 .
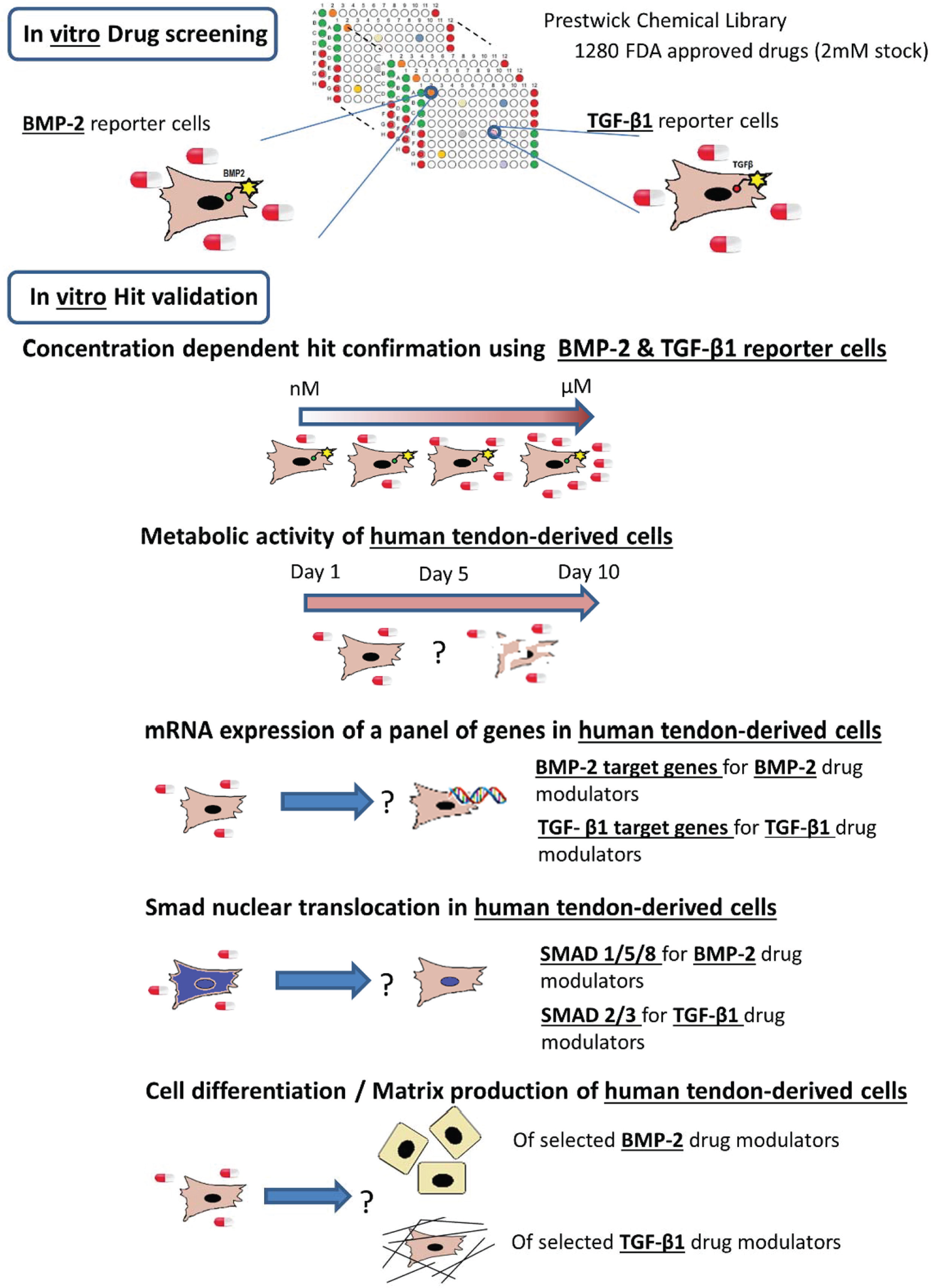
Schematic overview of the experimental design.
Materials and Methods
Cell Types
Mink lung epithelial cells (MLECs) previously transfected with a reporter plasmid consisting of TGF-β–responsive elements from the PAI-1 promoter fused to a luciferase reporter gene 10 and C2C12 transfected with a reporter plasmid consisting of BMP-responsive elements from the Id1 promoter fused to a luciferase reporter gene 11 were a generous gift from D. B. Rifkin.
huTCs were isolated from human hamstring tendon tissue using 0.15% (w/v) collagenase type II solution (Worthington, Biochemical Corp., Lakewood, NJ) digestion in basic media (see below) as previously described. 12 The collection and anonymous use of the tissue was performed according to the medical ethical regulations and the “good use of redundant tissue for research of the Dutch Federation of Medical Research Societies” guideline.
All cells were cultured in basic media (BM) consisting of Dulbecco’s modified Eagle’s medium (DMEM) (Gibco) supplemented with 10% fetal bovine serum (FBS) (Gibco), 100 U/mL penicillin and 100 µg/mL streptomycin (Gibco), and 0.2 mM ascorbic acid (Sigma).
Small-Molecule High-Throughput Screening
The Prestwick Chemical Library, consisting of 1280 FDA-approved small molecules (Prestwick Chemical, Inc., Washington, DC), was dissolved in DMSO to a final concentration of 2 mM.
BMP-2 reporter cells were seeded onto 96-well cell culture plates at 10,000 cells/well in BM and allowed to attach for 24 h at 37 °C, 5% CO2, and 95% humidity. Small molecules were then added to the wells at a final concentration of 5 µM, and DMSO-treated cells were used as a negative control, while cells treated with 2.5 nM rhBMP-2 were used as a positive control. Cells were further cultured for 24 h, upon which media containing the small molecules were removed, cells were washed one time with phosphate-buffered saline (PBS), and Luciferase assay (Promega, Madison, WI) was used according to the manufacturer’s protocol.
Similarly, TGF-β1 reporter cells were seeded onto 96-well cell culture plates at 20,000 cells/well and allowed to attach for 24 h at 37 °C, 5% CO2, and 95% humidity. Small molecules were then added to a final concentration of 5 µM, and DMSO-treated cells were used as the negative control, while cells treated with 0.8 nM rhTGF-β1 were used as a positive control. Cells were further cultured for 24 h, upon which media containing small molecules was removed, cells were washed one time with PBS and Luciferase assay (Promega) was used according to the manufacturer’s protocol.
Luminescence was read at 590 nm on Victor plate reads (PerkinElmer, Wellesley, MA) and reported as relative light units (RLU). A hit was identified if, upon exposure to a small molecule, the resulting luciferase activity was at least two times higher than the negative control. Small molecules that fulfilled this threshold were purchased from Prestwick Chemical, Inc. and retested in a dose-dependent manner (from nM to µM) under the same seeding and culture conditions. The compounds showing an increased luciferase activity compared to the negative control were selected for further investigation using huTCs.
Metabolic Activity
huTCs were seeded on to 96-well cell culture plates at 4,000 cells/well in BM and allowed to attach for 24 h. The next day, cells were treated with different concentrations of the selected compounds in a dose-dependent manner (from nM to µM). After 1, 5 and 10 days, metabolic activity was measured after incubation for 1 h with 10% (v/v) Presto Blue Cell Viability Reagent (Invitrogen, Carlsbad, CA) in BM and absorbance was measured at 590 nm (n = 3).
Gene Expression Analysis
huTCs were seeded on to 6-well cell culture plates at 50,000 cells/well in BM and cultured until confluent. BM was then replaced with starvation media (SM) consisting of BM without FBS and further cultured for 24 h, upon which the selected compounds were added in BM for 24 h.
Total RNA was isolated using the NucleoSpin RNA II isolation kit (Macherey-Nagel, Düren, Germany), according to the manufacturer’s instructions. RNA was collected in RNase-free water and a quantitative analysis was performed using spectrophotometry (Nanodrop, Thermo Fisher Scientific, Waltham, MA). First-strand complementary DNA (cDNA) was synthesized from 0.6 µg total RNA/sample, using iScript (Bio-Rad, Hercules, CA) according to the manufacturer’s instructions. PCR was performed on a real-time PCR detection system (Bio-Rad), using iQ Syber green supermix (Bio-Rad) for different genes, as presented in
SMAD2/3 and SMAD 1/5/8 Nuclear Translocation
huTCs were seeded on to 96-well cell culture plates at 5,000 cells/well in BM and allowed to attach for 24 h. The next day, BM was replaced with SM for 5 h, followed by 30 min or 2 h of incubation in BM in the presence of quinacrine (5 µM), niclosamide (100, 500 nM or 1 µM), vorinostat (5 or 10 µM), or hycanthone (5 or 10 µM). Cells exposed to 2.5 nM rhBMP-2 or 0.2 nM rhTGF-β1 were used as positive control. Following, the cells were washed with PBS and fixed with 10% neutral buffered formalin solution (Sigma) for 15 min, after which they were stained for SMAD2/3 (for TGF
Fluorescence images were taken using BD Pathway 435 (BD Bioscience) after adjusting the staining intensity to the isotype (secondary antibody alone) and quantification of SMAD nuclear translocation was assessed using CellProfiler open-source software. 13 Following, the integrated intensity measured for the cytoplasmic and the nuclear SMAD was used to calculate the nucleus/cytoplasm ratio of SMAD.
Alkaline Phosphatase Activity
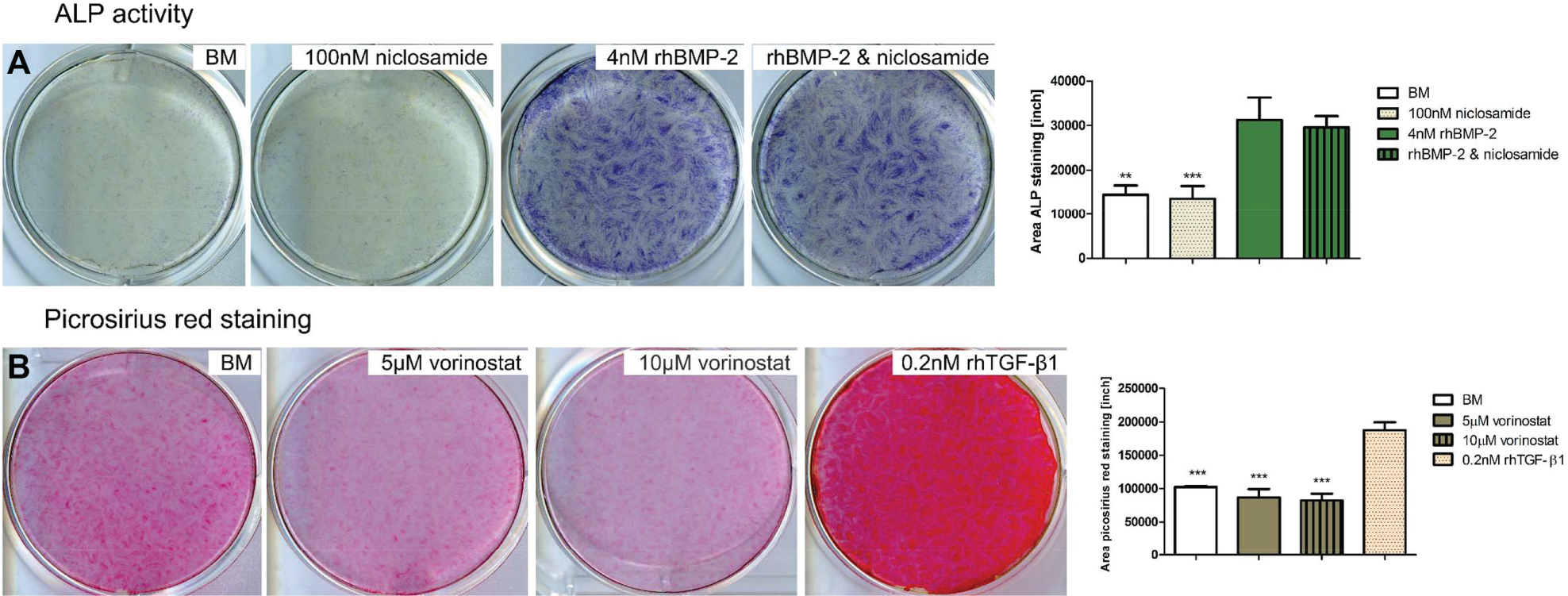
huTCs were seeded on to 12-well cell culture plates at 50,000 cells/well in BM. The next day, BM was replaced with fresh media and 100 nM niclosamide or 4 nM rhBMP-2 was added to the wells (n = 3). Cells were cultured for 7 days with one media change at day 3. At day 7, cells were fixed in 10% neutral buffered formalin solution (Sigma) for 15 min and stained using the Leukocyte Alkaline Phosphatase Kit-LAP (85L2-1KT; Sigma) following the manufacturer’s instructions. Images of the stained wells were acquired using an Epson Perfection V750 PRO scanner (Epson, Long Beach, CA), and quantification was performed using Fiji open-source software.
14
Equal-sized squares were cropped from the wells, converted to 8-bit, and background subtracted using the rolling ball radius function. The image was then converted into binary, the area of alkaline phosphatase (ALP) staining was selected and the total number of pixels was measured (
Collagen Content Analysis
huTCs were seeded on to 6-well cell culture plates at 50,000 cells/well in BM and cultured until confluent, upon which media was refreshed and vorinostat (5 and 10 µM) or 0.2 nM rhTGF-β1 was added to the wells (n = 3). Vorinostat or rhTGF-β1 was freshly added every 3 days upon media refreshment and at day 10 cells were fixed in 10% formalin for 15 min at room temperature and stained for collagen deposition using the picrosirius red stain kit (Polyscience, Inc., Warrington, PA) following the manufacturer’s instructions. Images of the stained wells were acquired and quantified using Fiji open-source software as described above.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 6 software (GraphPad Software, La Jolla, CA). Two-way analysis of variance (ANOVA) and a Tukey’s post hoc test were used to compare the different conditions. A p value of ≤0.05 indicates a statistically significant difference. Results are shown as mean ± standard deviation.
Results
Identification and Validation of Small Molecules Capable of Modulating TGF-β1 and BMP-2 Reporter Cell Lines
A library of 1280 FDA-approved small molecules was screened to identify modulators of BMP-2 and TGF-β1 signaling. BMP-2 and TGF-β reporter cell lines were exposed for 24 h to the small molecules (5 µM) and hits were selected if a compound’s luciferase activity was at least two times higher than the negative controls. Seven BMP-2 and three TGF-β1 small molecules were identified as primary hits. Pirlindole mesylate, pramipexole, nylidrin, butylscopolammonium (n-) bromide, quinacrine dihydrochloride dehydrate (quinacrine), chlorhexidine and niclosamide were identified as modulators of the BMP-2 reporter cell line, whereas hycanthone, etoposide and vorinostat were identified as modulators of the TGF-β1 reporter cell line (
Fig. 2A
,
C
and
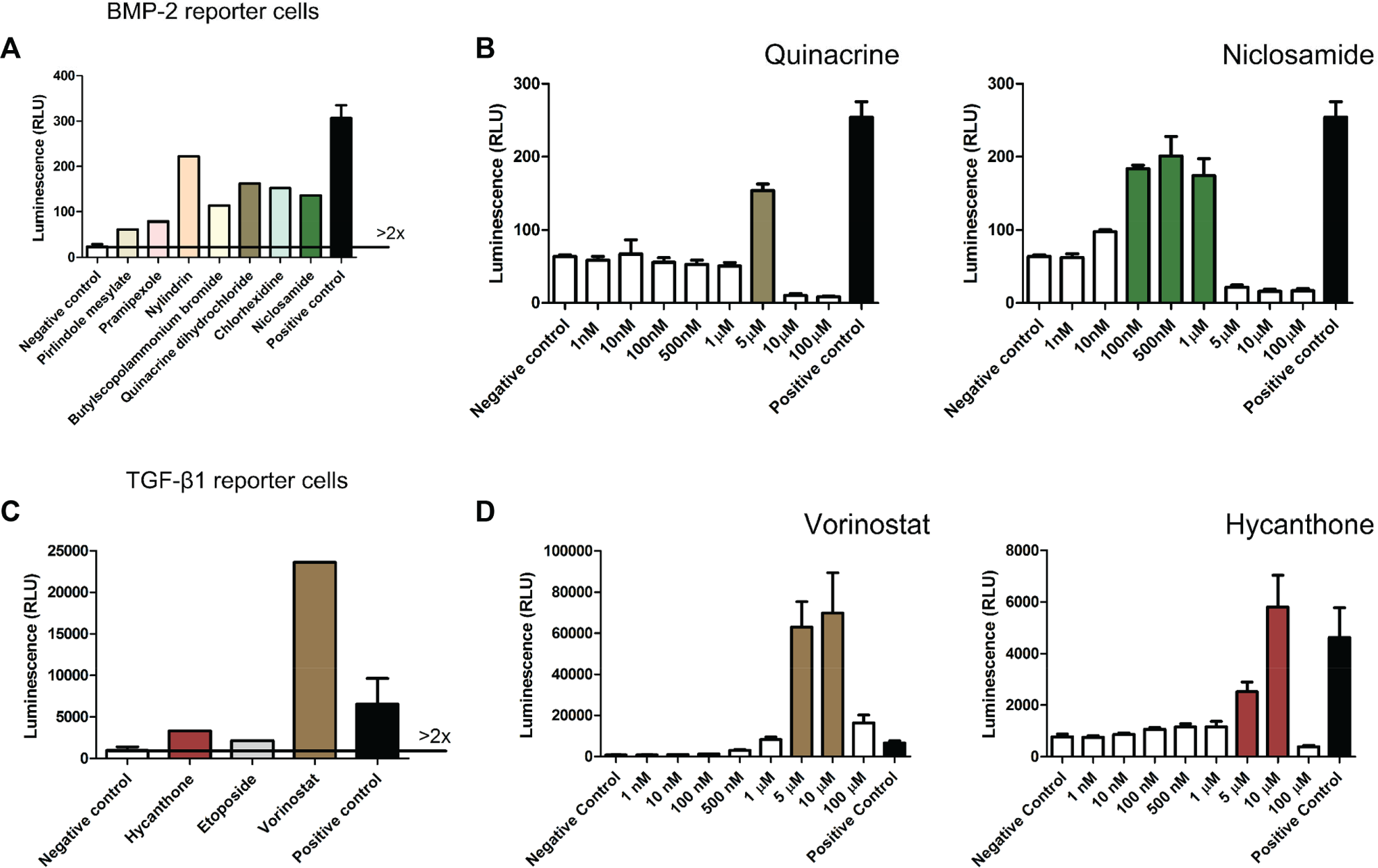
Small-molecule library screening for bone morphogenetic protein 2 (BMP-2) and transforming growth factor–β1 (TGF-β1) modulators. BMP-2 and TGF-β1 reporter cell lines were independently plated on to 96-well cell culture plates and compounds were added for 24 h at a concentration of 5 µM, after which cells where lysed and luciferase activity measured. Positive control are cells cultured in the presence of 2.5 nM recombinant human BMP-2 (rhBMP-2) or 0.8 nM recombinant human TGF-β1 (rhTGF-β1), while negative control are cells cultured in basic media. A hit was identified if, upon exposure to a small molecule, the resulting luciferase activity was at least two times higher than the negative control. (
Subsequently, dose-response curves (1 nM to 100 µM, n = 3) were generated for the selected primary hits. Quinacrine and niclosamide were validated as potential BMP-2 modulators while hycanthone and vorinostat were validated as potential TGF-β1 modulators ( Fig. 2B , D ).
Effect of the Validated Hits on Metabolic Activity of huTCs
Given that our final goal is to use the identified hits for T/L regeneration, we tested them on huTCs previously isolated and described. 12 To that end, a time-dependent dose-response curve was performed and metabolic activity was measured at days 1, 5 and 10. For BMP-2 modulators our results showed a statistically significant increase in metabolic activity for quinacrine (250 nM) and niclosamide (100 nM) compared to rhBMP-2 ( Fig. 3A ). Moreover, for the time and concentrations tested herein, an increase in metabolic activity during time was observed for all concentrations with the exception of quinacrine (5 µM) and niclosamide (1 µM), which inhibited metabolic activity both at days 5 and 10. Noteworthy, all the groups treated with rhBMP-2 or the BMP-2 modulators showed lower metabolic activity relative to BM suggesting that both BMP-2 and BMP-2 modulators reduce the metabolic activity of huTC cultures, possibly by reducing cell proliferation in favor of cell differentiation.
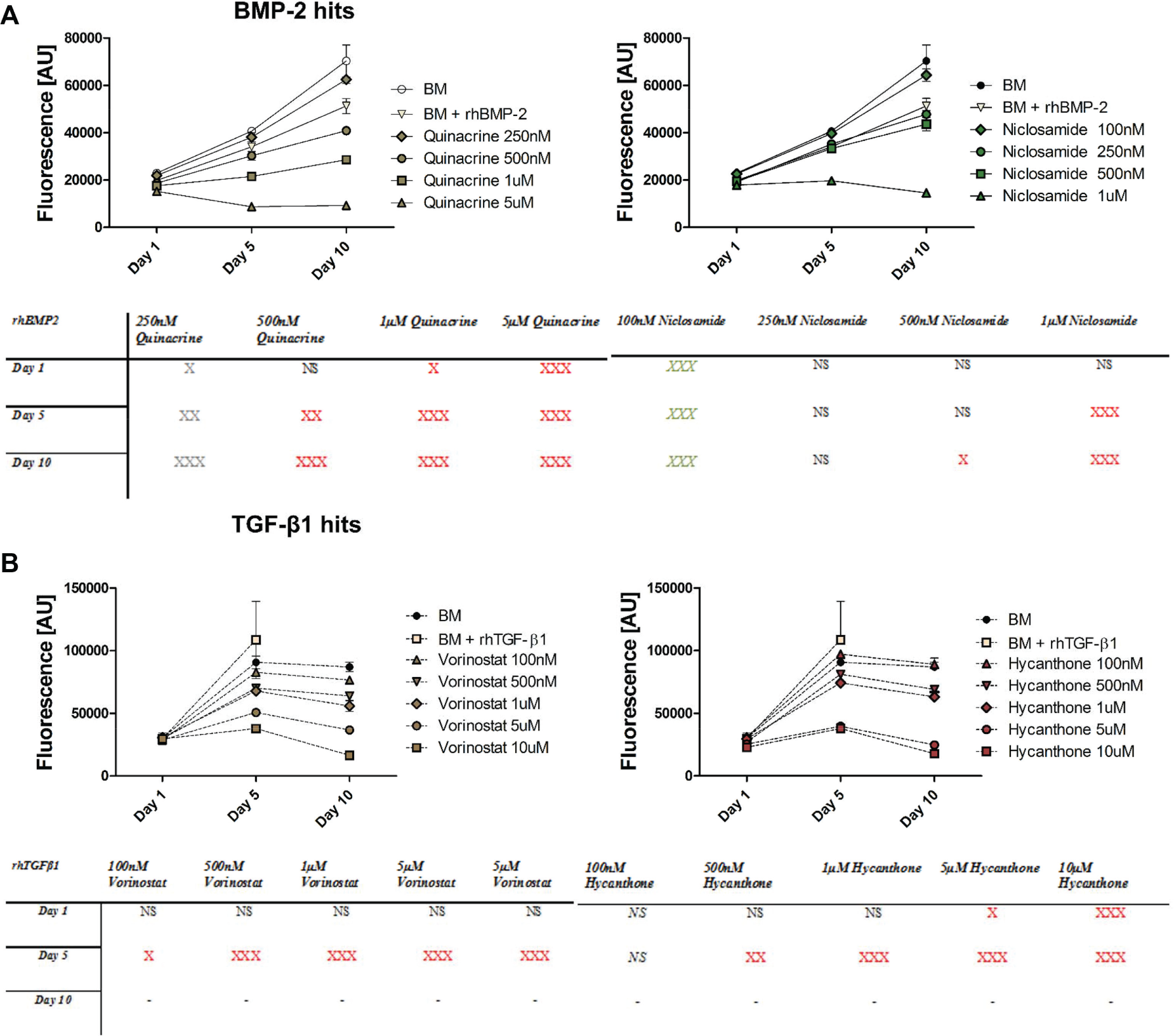
Metabolic activity of human tendon–derived cells (huTCs) exposed to (
For TGF-β1 modulators all concentrations showed an increase in metabolic activity during the first 5 days. However, compared to rhTGF-β1 and BM control, the metabolic activity for all the concentrations tested was statistically significantly reduced with the exception of 100 nM hycanthone. At day 10, metabolic activity either reached a plateau or became partially inhibited possible due to high number of cells that, in the case of the TGF-β1 control, led to cell detachment and pellet formation by day 10 ( Fig. 3B ).
Following, as the concentrations found to induce BMP-2 and TGF-β1 activation in the reporter cells inhibited the metabolic activity of huTCs, we decided to test whether acute exposure (24 h) rather than chronic exposure (10 days) could modulate both signaling pathways in huTCs.
Identified Hits Modulate BMP-2 and TGF-β1 Target Gene Expression
To examine whether the identified hits modulate the expression of genes involved in BMP-2 or TGF-β1 pathway signaling, we analyzed the expression of a panel of genes after acute exposure (24 h) of huTCs to the selected hits.
Our results showed that 100 nM niclosamide significantly increased the expression of collagen IA1 (COL1A1) by 1.3 ± 0.2-fold, followed by a trend in increased expression in runt-related transcription factor 2 (RUNX2) by 0.7 ± 0.3-fold; alkaline phosphatase, liver/bone/kidney (ALPL) by 0.6 ± 0.3-fold; BMP-2 by 0.4 ± 0.3-fold; and BMP-4 by 0.4 ± 0.2-fold compared to both BM and rhBMP-2 control. Next, we found that 5 µM quinacrine significantly increased the expression of BMP-2 and BMP-4 by 2.8 ± 0.4-fold but not the expression of other osteogenic target genes, which were significantly downregulated compared to both BM and rhBMP-2 control ( Fig. 4A ).
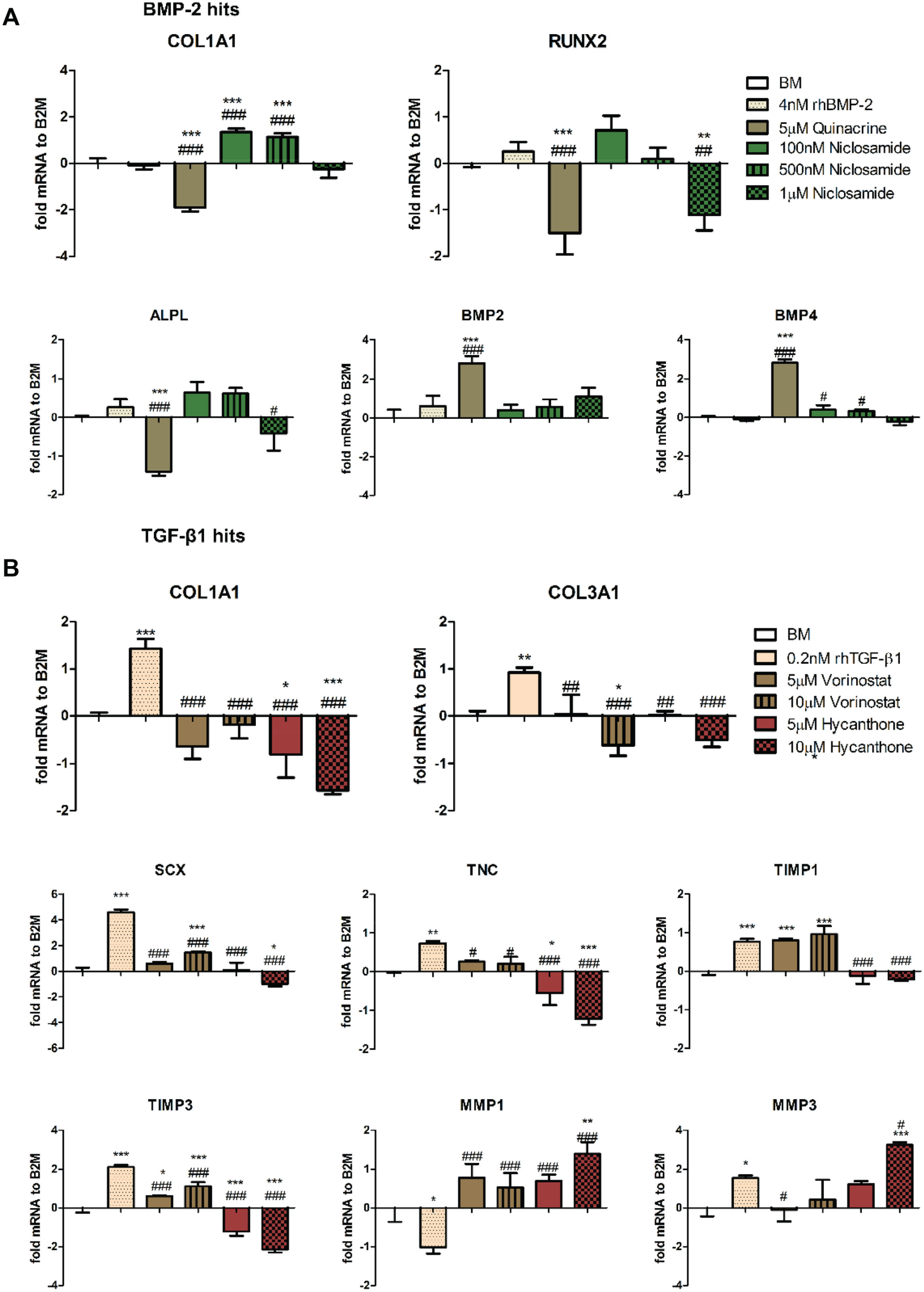
Gene expression analysis of human tendon–derived cells (huTCs) exposed for 24 h to (
For the TGF-β1 hits, our results showed that exposing huTCs to vorinostat partly induced expression of tendon and extracellular matrix (ECM) genes. Compared to BM, both 5 and 10 µM vorinostat significantly increased the expression of SCX by 0.6 ± 0.1-fold and 1.5 ± 0.1-fold, TIMP metallopeptidase inhibitor 1 (TIMP1) by 0.8 ± 0.04-fold and 1 ± 0.2-fold, and TIMP3 by 0.6 ± 0.03-fold and 1.1 ± 0.2-fold. In addition, further increase, although not statistically significant, was observed for tenascin c (TNC) by 0.3 ± 0.03-fold and 0.2 ± 0.2-fold and matrix metallopeptidase 1 (MMP1) by 0.7 ± 0.4-fold and 0.5 ± 0.4-fold. COL1A1, COL3A1, and MMP3 remained at comparable levels to BM. In contrast to vorinostat, 5 and 10 µM hycanthone showed an inhibition of all genes with the exception of MMP1 and MMP3, in which a statistically significant increase was observed for the highest concentration tested ( Fig. 4B ).
Similar results were obtained with a second donor (
BMP-2 and TGF-β1 Hits Modulate SMAD Nuclear Translocation
To disclose the mechanism of action of the identified BMP-2 and TGF-β1 hits we investigated how they modulate SMAD nuclear translocation in huTCs. To do so, cells were exposed to the selected hits for 30 min or 2 h and SMAD1/5/8 (BMP-2 signaling pathway) or SMAD2/3 (TGF-β1 signaling pathway) nuclear translocation was analyzed using immunocytochemistry.
Exposure of huTCs to BMP-2 hits for 30 min or 2 h in BM or SM did not affect SMAD1/5/8 nuclear translocation. Although not statistically significant, a trend towards increased SMAD1/5/8 nuclear translocation was observed for 5 µM quinacrine and 1 µM niclosamide after 30 min and for 100 and 500 nM niclosamide after 2 h.
For the identified TGF-β1 hits, exposure of huTCs to the selected hits for 30 min did not increase SMAD2/3 nuclear translocation compared to BM. However, after 2 h of exposure to 5 µM vorinostat and 10 µM hycanthone, a statistically significant increase in SMAD2/3 nuclear translocation was observed compared to BM. Interestingly, this increase was similar to the one observed with rhTGF-β1 control ( Fig. 5 ).
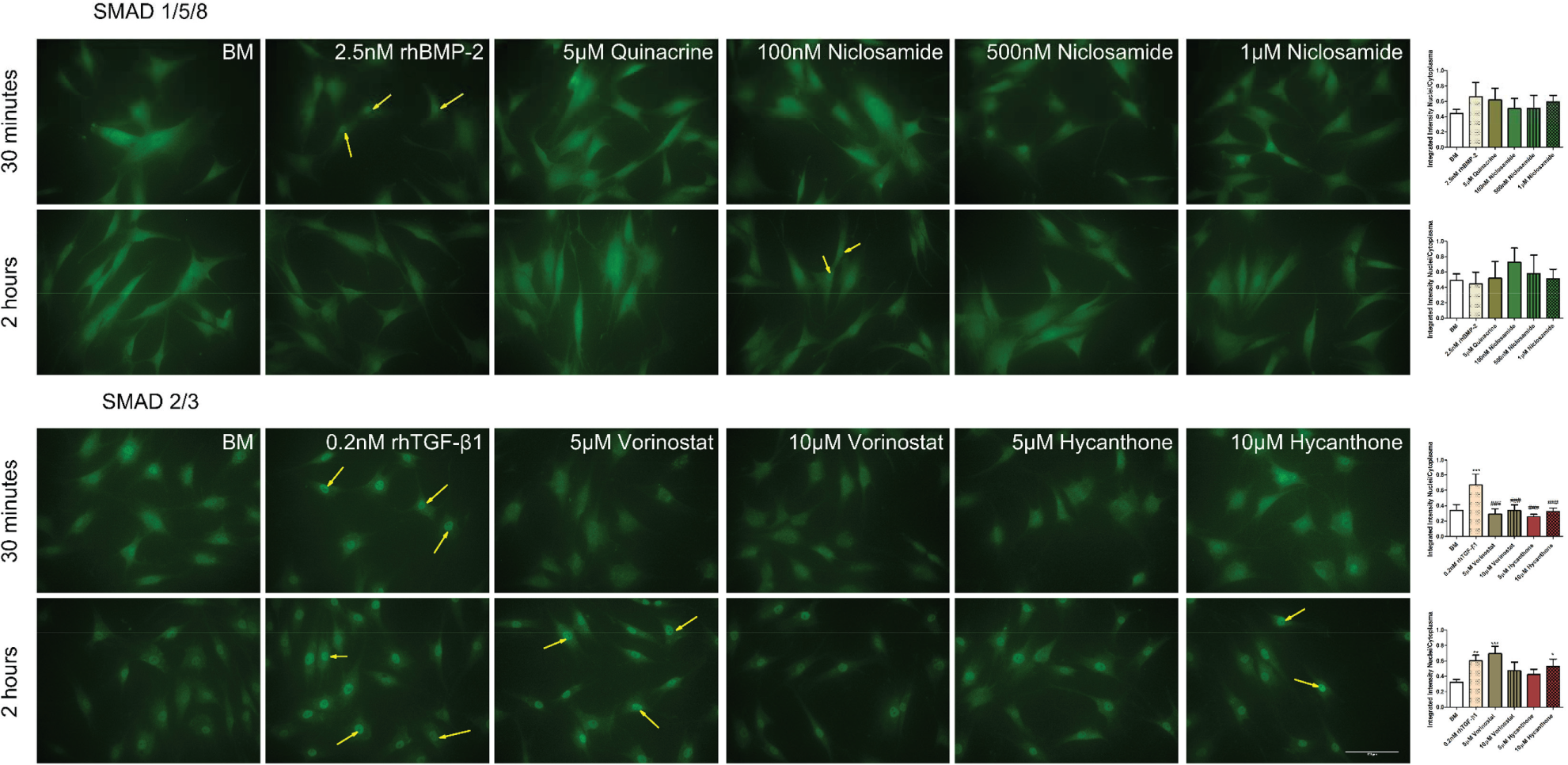
Immunofluorescence staining of SMAD1/5/8 and SMAD2/3 on human tendon–derived cells (huTCs) exposed to bone morphogenetic protein 2 (BMP-2) or transforming growth factor–β1 (TGF-β1) modulators for 30 min or 2 h. Yellow arrows indicate SMAD nuclear translocation. Quantification of SMAD1/5/8 or SMAD2/3 nuclear translocation is presented as a ratio between integrated staining intensity between nuclei and cytoplasm. Values are represented as mean ± standard deviation of at least three independent pictures from different wells (n ≥ 3). Statistically significant differences were found only when comparing BM with TGF-β1 modulators (*) or recombinant human TGF-β1 (rhTGF-β1) with TGF-β1 modulators (#). Scale bar corresponds to 100 µm.
BMP-2 Hits Modulate ALP Activity
Following the increase in metabolic activity and in the expression of osteogenic genes upon exposure of huTCs to 100 nM niclosamide, we decided to analyze its effect on ALP activity, an early marker for in vitro osteogenesis. A 7-day exposure of huTCs to 100 nM niclosamide did not increase nor interfered with the BMP-2–mediated increase in ALP activity ( Fig. 6A ). In addition, exposure of huTCs to 5 µM quinacrine during 7 days led to cell death and consequently no ALP assay was performed.
(
TGF-β1 Hits Modulate Collagen Production
To investigate whether vorinostat influences the production of ECM we exposed huTCs to 5 and 10 µM vorinostat, intermittently for 2 h a day for 10 days and analyzed collagen production using a picrosirius red staining. Our results demonstrate that, as expected, exposure of huTCs to rhTGF-β1 led to an increase in collagen production whereas exposure to vorinostat, regardless of the concentration used, did not increase collagen production ( Fig. 6B ).
Discussion
In this study, we aimed to identify FDA-approved compounds capable of modulating BMP and TGF-β signaling. The identification of such molecules could pave the way to new therapies capable of inducing bone and matrix formation, key actors in tendon/ligament enthesis formation 15 and middle core repair.16,17 Currently, the only approved drug that stimulates bone formation is parathyroid hormone (PTH), which was lately reported to stimulate bone resorption while increasing bone mineral density. 18 Furthermore, rhBMP-2 was approved as a bone graft substitute to achieve fracture repair in clinical use. 19 However, the need to use supra-physiological doses and the high production costs create a barrier for routine clinical application and emphasize the need to discover new bone anabolic agents that either replace or reduce the basal concentration of rhBMP-2 used in patients. rhTGF-β has not been approved for clinical application. Other than the previously described compounds, no other FDA-approved compounds modulate the TGF-β pathway.
We screened a library of commercially available FDA-approved compounds using genetically modified reporter cell lines for BMP-2 and TGF-β1 and identified several small molecules that modulate BMP and TGF-β signaling pathways. Dose-response curve analysis led to the identification of two BMP-2 (quinacrine dihydrochloride dehydrate and niclosamide) and two TGF-β1 (hycanthone and vorinostat) candidates. All four compounds have been previously described in literature to have antineoplastic potential, showing different mechanisms of action. Quinacrine has been described to intercalate with DNA, inhibit some flavoprotein enzymes and uncouple oxidation and phosphorylation. 20 Niclosamide has been described to inhibit glucose uptake, oxidative phosphorylation, and anaerobic metabolism.21,22 Hycanthone has been described to intercalate into DNA and inhibit RNA synthesis in vitro 23 and vorinostat is a powerful histone deacetylase inhibitor. 24
According to their antineoplastic properties, we were concerned that the antiproliferative properties of the identified compounds may dominate our screen which we identified by a decrease in metabolic activity for quinacrine, vorinostat and hycanthone but not for niclosamide. Following, we decided to test whether acute exposure (24 h) rather than chronic exposure (10 days) could modulate both signaling pathways in tendon-derived cells and investigated transcriptional activation of BMP and TGF-β responsive genes. Niclosamide (100 nM) was found to induce expression of BMP signaling genes, in line with a recent study that screened for osteogenic compounds using a SP7:luciferase transgenic zebrafish model and where niclosamide was identified as a potential lead osteogenic compound. 25 In an attempt to better understand its function, we screened the literature and found that niclosamide treatment does not inhibit the Wnt/β-catenin signaling pathway, 26 which can act downstream of BMP-2 signaling to promote osteoblast cell fate decision in multipotent progenitors. 27 We also showed that quinacrine induced expression of BMP signaling genes (BMP-2 and BMP-4) in both donors and reached up to four times higher levels than the addition of rhBMP-2 alone. This finding lends support to a previous study reporting that quinacrine stimulates endogenous BMP-2 upregulation in vitro. 28 According to the literature and based on our findings, we believe that quinacrine specificity to intercalate with the DNA 29 is not random but might be selective, as we and others have shown that it promotes BMP transcription. Further work is necessary to confirm this hypothesis.
From the TGF-β1 modulators, we have shown that only vorinostat, but not hycanthone, induces the expression of TGF-β responsive genes, like SCX, TNC, TIMPs and MMPs. As a HDAC inhibitor, vorinostat upregulates the cellular acetylation level and therefore accelerates the natural epigenetic process 30 that we believe to potentially be of great help during tendon remodeling.
So far, we have shown that our identified compounds modulate the expression of BMP-2 and TGF-β1 target genes. However, we could not observe an increase in ALP activity for niclosamide and quinacrine, nor did we observe an increase in ECM production in huTCs exposed to vorinostat. Our concern that the antiproliferative or cytotoxic effects of the compounds might hinder downstream assays to study the functionality of the cells turned out to be true for quinacrine but not for niclosamide or vorinostat. Even though we did not observe an increase in ALP activity in the presence of niclosamide or collagen production in the presence of vorinostat, the protein expression levels remained at comparable levels with BM with no observable changes in cell morphology. By investigating whether the identified hits modulate BMP or TGF-β signaling by modulating SMAD nuclear translocation, we found that even if vorinostat—but not niclosamide—affected SMAD nucleus translocation, their mechanisms of action differed from the native TGF-β1 and BMP-2, suggesting that other mechanisms may be involved.
In addition, the reason why niclosamide and vorinostat, in contrast with rhBMP-2 and rhTGF-β1, did not show an effect on the selected functional assays—ALP and ECM production, respectively—suggests that an increase in messenger RNA production does not necessarily translate to an increase in protein levels. These results warrant further studies to unravel the relation between transcription and translation of the target proteins in huTCs with special emphasis on the molecular regulators of ALP activity and ECM production and turnover.
Moreover, with the advent of gene editing technologies such as the CRISPR/Cas9, one could accelerate the discovery of compounds capable of modulating the endogenous promoters in the cellular context of interest rather than using minimal promoter(s) cloned on reporter plasmids. This is highly relevant for future work since it is well recognized that the genetic and epigenetic landscape of a given promoter is a key modulator of its activity. 31
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Twente.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
