Abstract
Because neurons are difficult to obtain from humans, generating functional neurons from human induced pluripotent stem cells (hiPSCs) is important for establishing physiological or disease-relevant screening systems for drug discovery. To examine the culture conditions leading to efficient differentiation of functional neural cells, we investigated the effects of oxygen stress (2% or 20% O2) and differentiation medium (DMEM/F12:Neurobasal-based [DN] or commercial [PhoenixSongs Biologicals; PS]) on the expression of genes related to neural differentiation, glutamate receptor function, and the formation of networks of neurons differentiated from hiPSCs (201B7) via long-term self-renewing neuroepithelial-like stem (lt-NES) cells. Expression of genes related to neural differentiation occurred more quickly in PS and/or 2% O2 than in DN and/or 20% O2, resulting in high responsiveness of neural cells to glutamate, N-methyl-
Keywords
Introduction
The advent of induced pluripotent stem cell (iPSC) technology has changed the study of human diseases. Drug development using cells differentiated from human iPSCs (hiPSCs), including patient-derived and genome-edited hiPSCs,1,2 has recently become feasible. Neurons are among the most sought-after cell types for target differentiated cells because they are not easily accessible from healthy donors or patients with neurological disorders. Neurons are electrically excitable cells that process and transmit electrical and chemical signals, forming networks connected via synapses. These characteristics of neurons are mainly mediated by voltage-gated ion channels and synaptic receptors, such as glutamate receptors. When creating neurons from hiPSCs for functional assays, it is crucial to ensure that the differentiated cells are both morphologically and functionally representative of neurons. This requires both optimizing the differentiation conditions to produce functional neurons and examining to what degree and how early these channels and receptors are functionally expressed.
Oxygen (O2) is an important physiological factor for cell function. An O2 level of 20% has generally been used in conventional in vitro cell culture systems. In mammalian brains, however, interstitial tissue O2 levels range from ~1% to 5%. 3 Recent studies have revealed that O2 plays crucial roles in the proliferation and differentiation of stem cells. For example, hypoxic conditions (2% O2) enhance the synthesis of cartilage proteins (collagen I and II and glycosaminoglycans) and mechanical functionality (tensile and compressive properties) in chondrogenic differentiation from human embryonic stem cells (ESCs) 4 ; 3% O2 inhibits maturation of differentiating cardiomyocytes derived from mouse iPSCs 5 ; and 1% O2 hampers adipogenic, osteogenic, and chondrogenic differentiation from human bone marrow mesenchymal stem cells. 6
Hypoxic conditions also affect the differentiation of neuron subtypes from neural stem cells (NSCs) and neural progenitor cells (NPCs). Several papers have shown that hypoxic conditions promote differentiation of dopaminergic neurons from NSCs/NPCs—either collected from rat embryonic mesencephalon 3 or human fetal mesencephalon and striatum, 7 or derived from mouse ESCs. 8 Horie et al. 9 reported that anoxia/hypoxia (0%–3% O2) in mouse NSCs collected from the ganglionic eminence shifted the neuron subtype of differentiation from γ-aminobutyric acid (GABA)–positive neurons to glutamate-positive neurons. Xie et al. 10 published findings that hypoxia (2% O2) shifted differentiation to glial fibrillary acidic protein (GFAP)–positive glia from βIII-tubulin-positive neurons during neural differentiation from human ESC- and hiPSC-derived NPCs.
In addition to O2, compounds and factors used to supplement culture media can affect neural differentiation and maturation. For example, dibutyl cyclic adenosine monophosphate (db-cAMP) increased the βIII-tubulin-positive cell population in NSCs/NPCs collected from the subventricular zone of adult rats. 11 Lepski et al. 12 showed that forskolin or 3-isobutyl-1-methylxanthine treatment increased the number of microtubule-associated protein 2 (MAP2)–positive cells and facilitated electrophysiological neuronal maturation of NSCs obtained from the subventricular zone of rat embryos. Ascorbic acid increases the percentage of βIII-tubulin-, tyrosine hydroxylase–, and serotonin-positive cells during neuronal differentiation from mouse ESCs. 13 Indeed, cAMP derivatives and ascorbic acid are used as supplements in some neural differentiation media to improve differentiation. 14
These results of the effects of O2 and other factors on neuronal differentiation were mainly determined with immunocytochemical methods. However, there have been few studies on how hypoxia affects the function of the differentiated neuronal cells. To establish physiological or disease-relevant assay systems using human neuronal cells in vitro, it is important that they not only exhibit neuronal morphology and express immunological markers, but also function normally. Therefore, in this study we compared the expression of genes involved in neural differentiation, the functions of ionotropic and metabotropic glutamate receptors, and the formation of networks of neurons differentiated from hiPSC-derived NSCs. We generated long-term self-renewing neuroepithelial-like stem (lt-NES) cells as an hiPSC-derived source of NSCs, because lt-NES cells have unique characteristics, including continuous expandability, ease of culturing (because they are adherent cells), and stable neuronal and glial differentiation potency. 15 These characteristics are suitable for the construction of systems for screening compounds during drug discovery. In this study, we differentiated lt-NES cells in different media (one prepared in house and one commercial) and O2 conditions (20% and 2% O2) to investigate the effects of these conditions on functional differentiation and maturation of hiPSC-derived neural cells in order to establish more efficient and robust screening systems.
Materials and Methods
Reagents
N-Methyl-
Cell Line
We used an hiPSC line, 201B7, 16 which was obtained at passage 25 (P25) from Kyoto University, Kyoto, Japan. In addition, we utilized an hiPSC line, P11025 at P13 introduced from Cellectis AB (now Takara Bio Europe, Gotebörg, Sweden). P11025 iPSCs were generated from primary fibroblasts of a healthy Caucasian male, reprogrammed by using episomal vector combined with SOX2, KLF4, OCT4, LIN28, L-MYC, and GLIS-1. These hiPSCs were maintained in Essential 8 (Thermo Fisher Scientific) at 37 °C in a humidified atmosphere of 20% O2. This study was approved by the Eisai Research Ethics Committee (approval numbers 2013-0013, 2013-193, 2014-193, 2014-0211, 2015-193, 2015-0211, 2016-193, and 2016-0211).
Generation of lt-NES Cells
We generated human lt-NES cells from 201B7 and P11025 cells according to the methods of Falk et al.
15
The generation procedures were as follows: 1.5 × 106 cells were plated in Aggrewell 800 plates (STEMCELL Technologies, Vancouver, Canada) in STEMdiff Neural Induction medium (STEMCELL Technologies). Neural aggregates were harvested on day 5 and cultured in wells coated with poly-
Neural Differentiation of lt-NES Cells
The following two neural differentiation media were investigated: DMEM/F12:Neurobasal-based medium17-20 (hereafter referred to as DN medium or simply DN) and a commercial neural differentiation medium (Neural Differentiation Medium for pan-neural differentiation, PhoenixSongs Biologicals, Branford, CT; hereafter referred to as PS medium or simply PS). The DN medium contained a 1:1 mixture of DMEM/F12 and Neurobasal (Thermo Fisher Scientific) supplemented with 0.5× N2 supplement, 0.5× B27 supplement, brain-derived neurotropic factor (BDNF, 20 ng/mL; Wako), glial cell–derived neurotropic factor (GDNF, 20 ng/mL; Wako), neurotrophin-3 (10 ng/mL; Wako), and laminin (1 μg/mL). PS was supplemented with supplement cocktail (base supplement, 20 ng/mL BDNF, 20 ng/mL GDNF, and 1 μg/mL laminin). Lt-NES cells were differentiated in DN or PS under 20% O2 or 2% O2. After neural differentiation, cells were used for immunocytochemical examination, microarray, RT-qPCR, calcium flux assays, and multielectrode array (MEA; see Supplementary Materials and Methods).
Results
Immunocytochemistry
Lt-NES cells were positive for the neural progenitor markers Nestin and SOX2 (
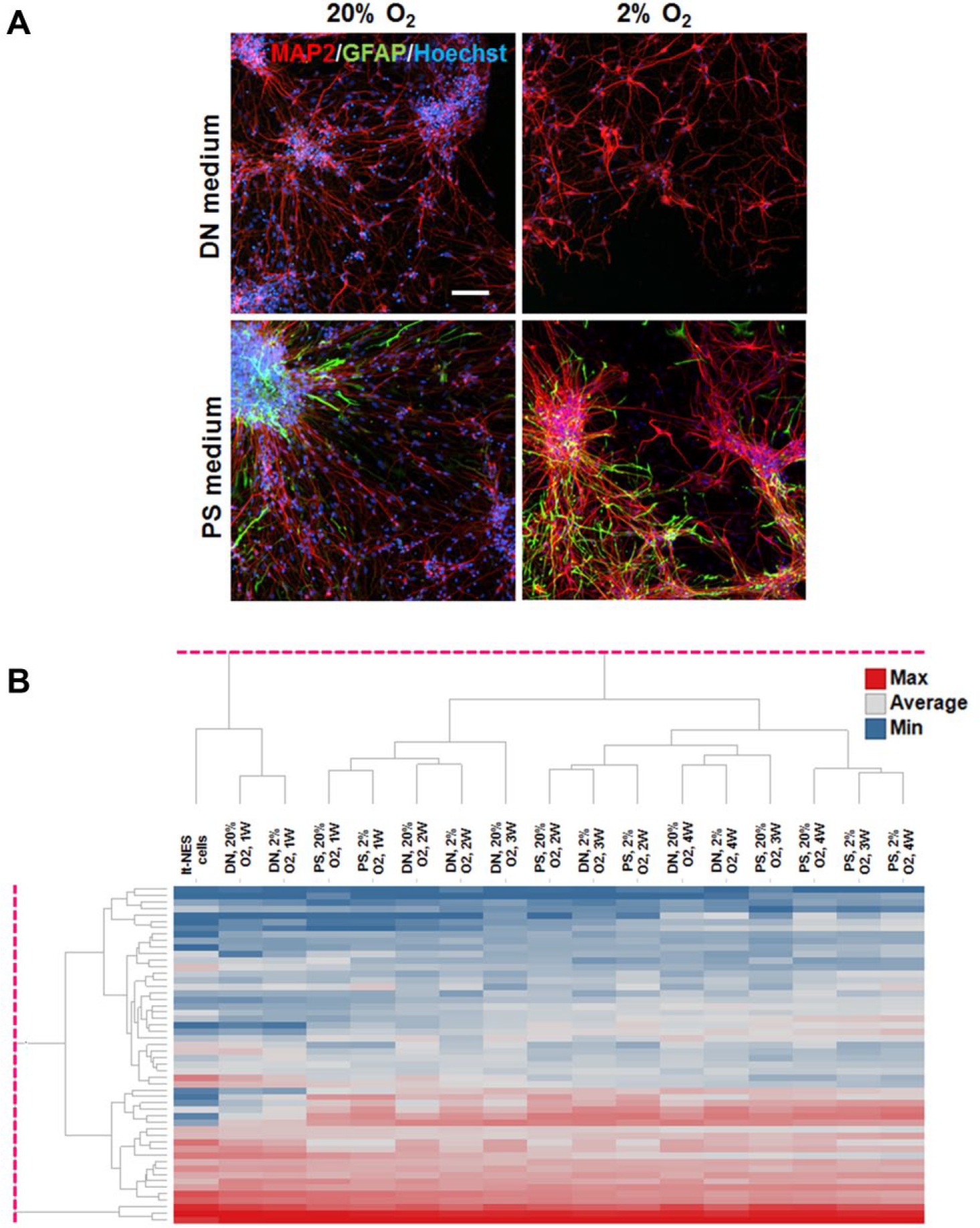
(
Microarray Results
The expression of genes related to neural differentiation differed by medium, O2 tension, and time in culture (1, 2, 3, or 4 weeks) ( Fig. 1B ). The dendrogram shows five main clusters, suggesting that neural differentiation progressed more rapidly in 2% O2 than in 20% O2, and in PS than in DN. But there were no increases of gene expression related to oligodendrocytes and oligodendrocyte progenitor cells.
RT-qPCR Analysis
To confirm the microarray results, we quantified gene expression with RT-qRCR. Changes in representative genes are illustrated in
Figure 2
. Detailed values and statistical results from two-way factorial analysis of variance (ANOVA) and Tukey’s test are shown in
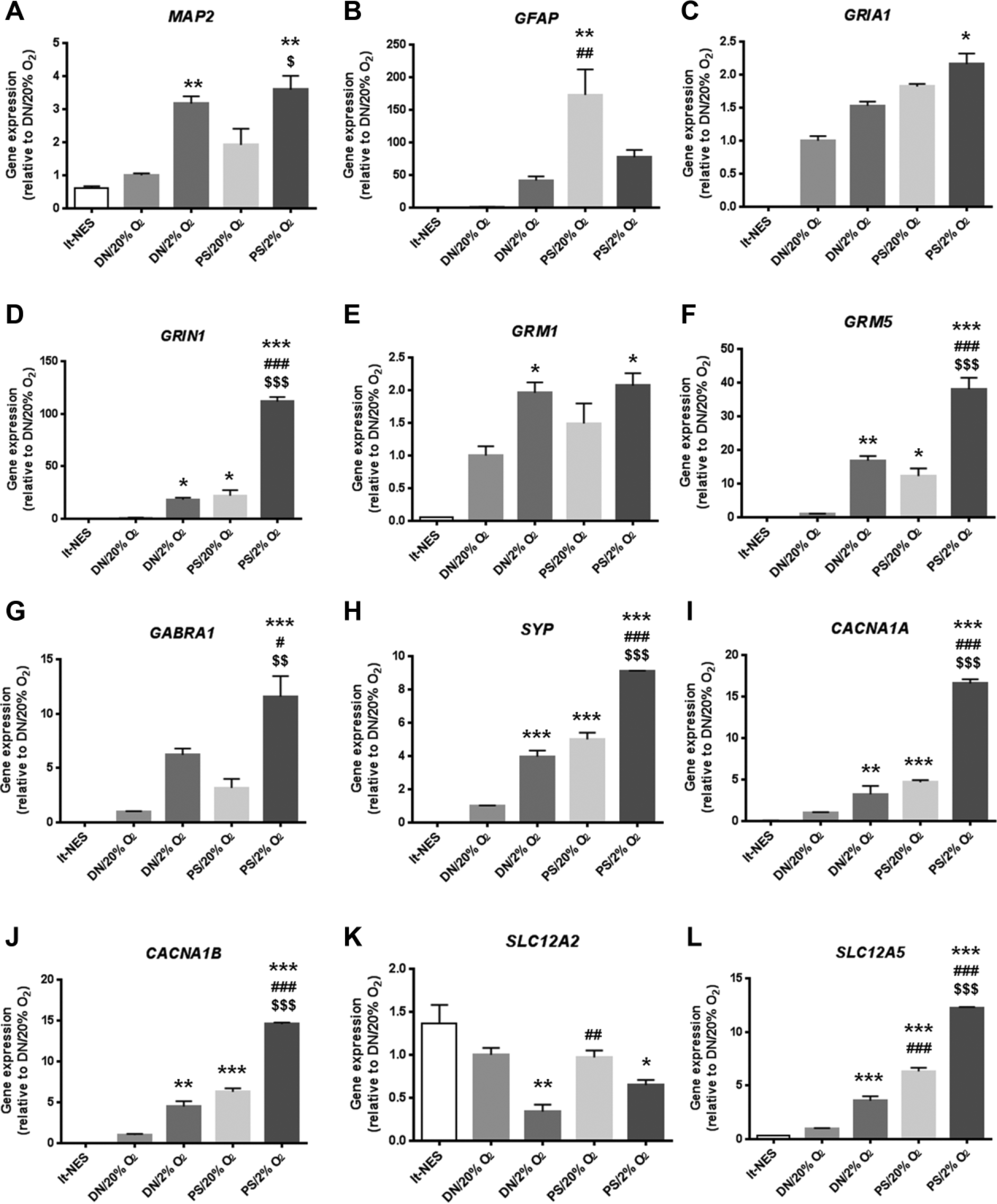
Expression levels of representative genes involved in neural differentiation and maturation in hiPSC-derived lt-NES cells and lt-NES cell–derived neural cells differentiated in PS or DN under 2% or 20% O2 for 4 weeks. (
The expression of the neuronal marker gene, MAP2, increased in 2% O2 ( Fig. 2A ). The medium had a clear effect on the astrocyte marker, GFAP, although O2 effects were also observed; the highest gene expression was found in PS/20% O2 ( Fig. 2B ). Several genes related to AMPA receptors (GRIA1) ( Fig. 2C ) and kainate receptors (GRIK3 and GRIK5) showed increased expression in 2% O2, some of which were also affected positively by PS, resulting in additive increases of gene expression. The combination of 2% O2 and PS produced synergistic increases for NMDA receptor-related genes (GRIN1 [ Fig. 2D ], GRIN2A, and GRIN3A). For metabotropic glutamate receptors, GRM1 expression was elevated by hypoxia ( Fig. 2E ), and GRM5 expression was amplified by the combination of 2% O2 and PS ( Fig. 2F ).
Additive increases of gene expression by the combination of 2% O2 and PS were seen for GABA receptor genes (GABRA1 [ Fig. 2G ], GABRA2, GABRB2, GABRB3, and GABRG2) and synapse-related proteins (SYP [ Fig. 2H ], SYN1, and DLG4). Gene expression of the P/Q- and N-type calcium channels (CACNA1A [ Fig. 2I ] and CACNA1B [ Fig. 2J ], respectively) was increased synergistically by the combination of 2% O2 and PS. Expression of SLC12A2 and SLC12A5, which encode a sodium-potassium-chloride transporter and a potassium-chloride transporter, respectively, showed contrasting changes: SLC12A2 expression decreased in 2% O2 ( Fig. 2K ), whereas SLC12A5 expression synergistically increased under the combined influence of 2% O2 and PS ( Fig. 2L ).
Calcium Flux Imaging for Receptor Function Assays
Glutamate application increased calcium signals in a concentration-dependent manner (
Fig. 3A
). Large responses were observed in PS/2% O2 and PS/20% O2. In DN/2% O2, glutamate-induced calcium flux was also observed, but the maximum response was only ~30% of that in PS/2% O2. The calcium rise was smallest in DN/20% O2. The maximum responses and EC50 values are summarized in
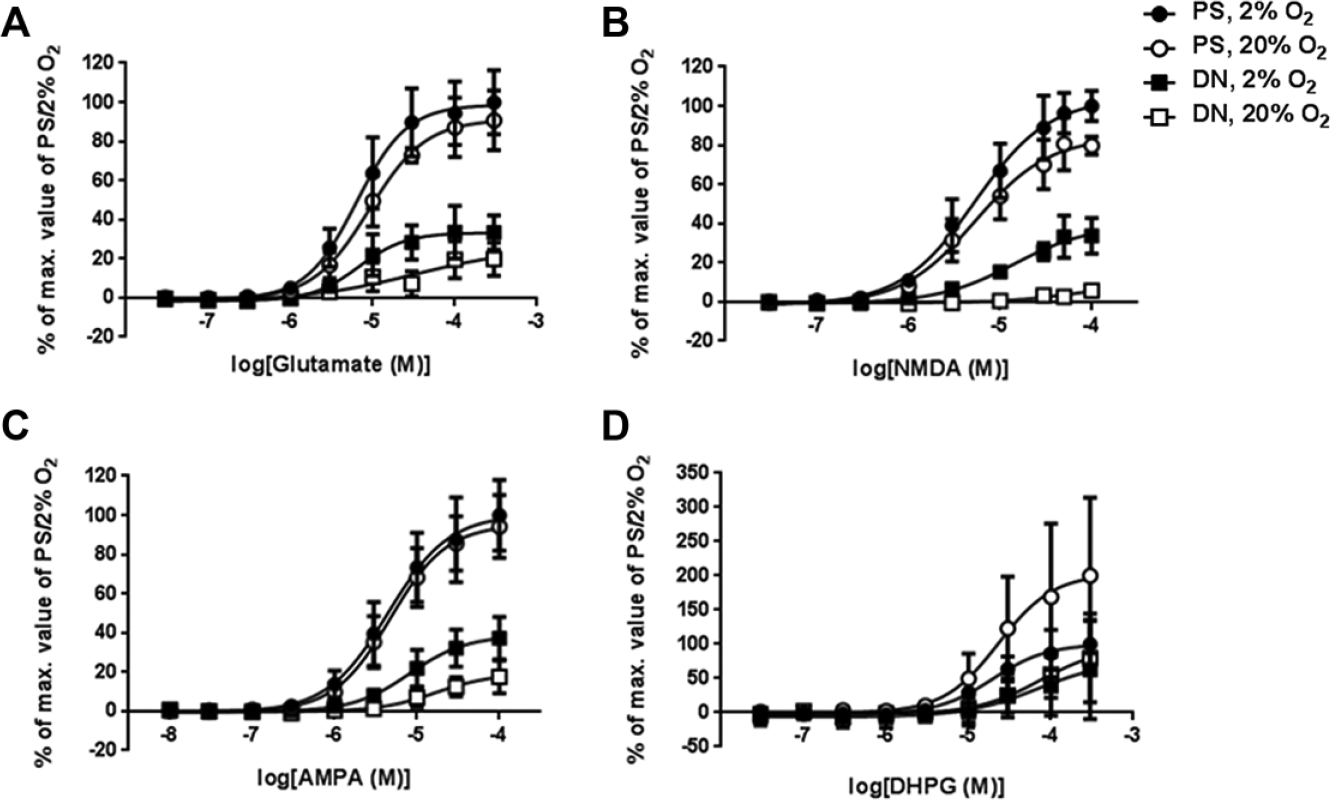
Calcium rise assays of ionotropic and metabotropic glutamate receptor function in human neural cells differentiated from lt-NES cells. Lt-NES cells were differentiated in DN or commercial neural differentiation medium (PS) under 2% or 20% O2 for 4 weeks. Concentration–response curves were drawn based on the means of three independent experiments (n = 3 in each experiment). Values are expressed as means ± SEM. Cells were stimulated with (
To further characterize the responses of lt-NES cell–derived neuronal cells to glutamate, we treated the cells with specific agonists. Response patterns to NMDA ( Fig. 3B ) and AMPA ( Fig. 3C ) in these culture conditions were similar to those with glutamate treatment. In general, the induced calcium flux was greatest in PS/2% O2 and PS/20% O2, followed by DN/2% O2, and then DN/20% O2. The NMDA-induced signal was particularly small in DN/20% O2, corresponding to only ~10% of that in PS/2% O2. The calcium flux induced by NMDA and AMPA (30 μM in both cases) was completely inhibited by specific antagonists for NMDA receptors (MK-801, Fig. 4A ) and AMPA receptors (NBQX, Fig. 4B ). IC50 values for MK-801 and NBQX were 0.53 μM and 1.20 μM, respectively, when using neural cells differentiated in PS/2% O2. In the case of PS/20% O2, IC50 values for MK-801 and NBQX were 0.50 μM and 0.80 μM, respectively.
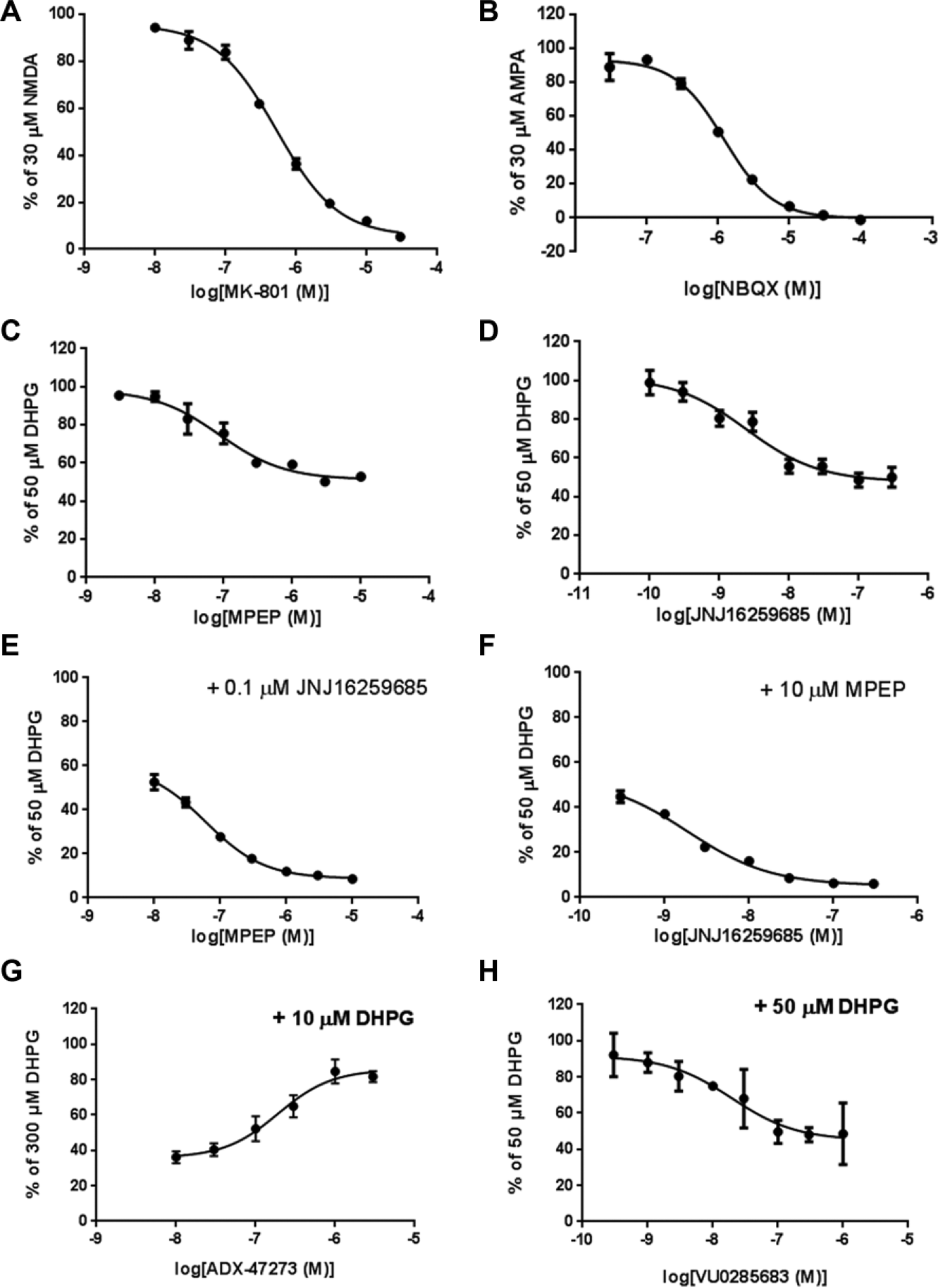
Validation of ionotropic and metabotropic glutamate receptor function in neural cells differentiated from lt-NES cells by using antagonists against these receptors. (
We also examined the responsiveness of metabotropic glutamate receptors by using DHPG, an agonist for mGluR1/5. Neural cells cultured in the four conditions responded to DHPG in a concentration-dependent fashion (
Fig. 3D
), but EC50 values of DHPG in DN/2% O2 and DN/20% O2 were ~3-fold larger than those in PS/2% O2 and PS/20% O2 (
MEA Recordings
We recorded the neural activity of lt-NES cell–derived neurons differentiated in PS under 20% or 2% O2 for 7 weeks. More spikes and bursts were visible in the raster plot at week 6 than at week 4, and they occurred more frequently under 2% O2 than under 20% O2 conditions (
Fig. 5A
). Synchrony of spikes was observed in lt-NES cell–derived neurons differentiated under 2% O2 at week 6, indicating formation of a neuronal network. Spontaneous spikes occurred in the third week of differentiation under both conditions (
Fig. 5B
). The number of electrodes detecting spikes and bursts depended on the O2 condition (main effects: F1,4 = 7.912, p = 0.0482 for number of electrodes detecting spikes; F1,4 = 8.696, p = 0.0420 for those detecting bursts) and differentiation time (main effects: F3,12 = 10.00, p = 0.0014 for numbers of electrodes detecting spikes; F3,12 = 5.962, p = 0.0099 for those detecting bursts;
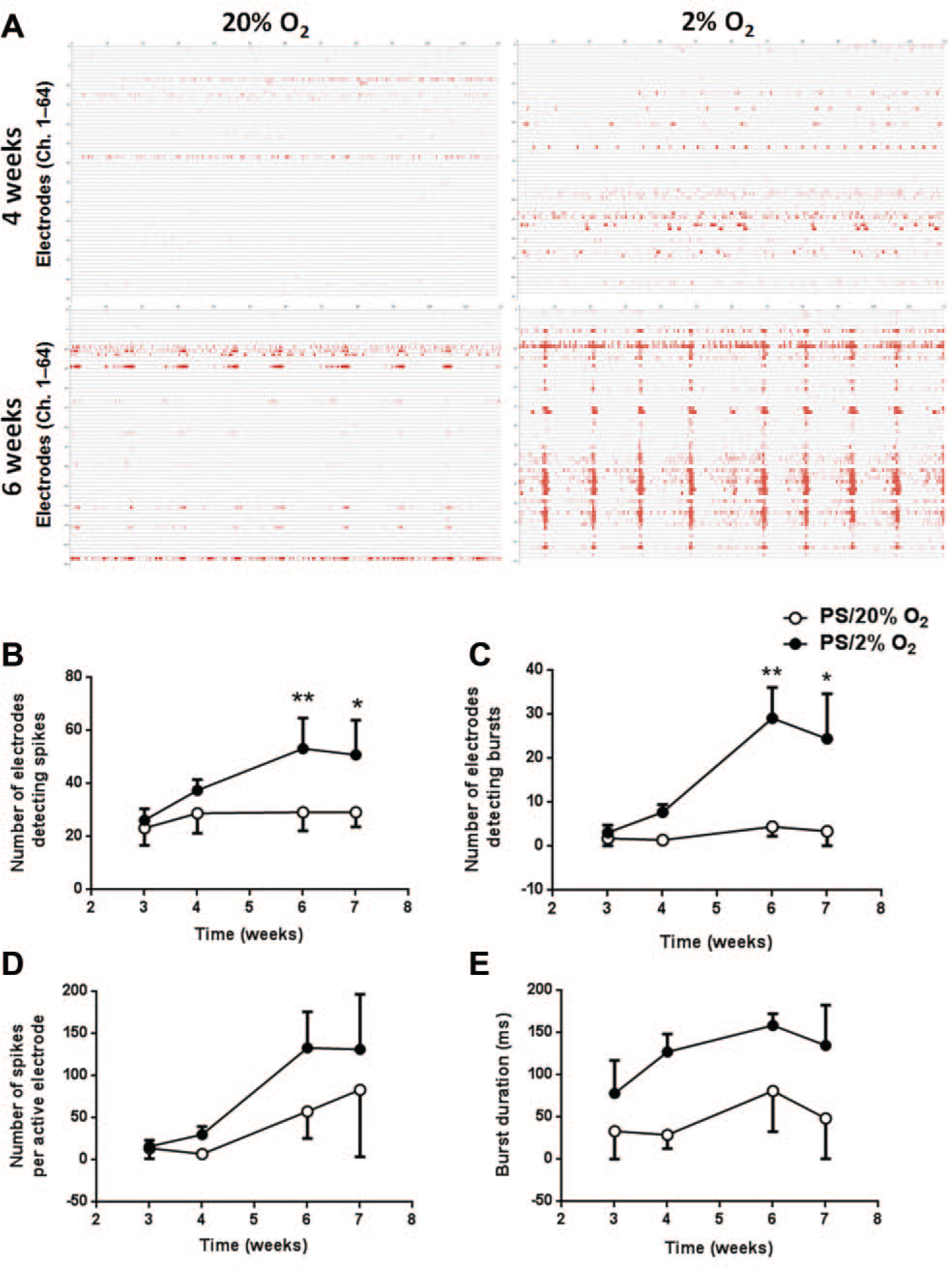
MEA analysis of spontaneous firing in neurons differentiated from lt-NES cells. (
To investigate whether inhibitory effects of GABA were reproduced in lt-NES neurons differentiated in PS/2% O2, we treated neurons with GABA and measured firing numbers with the MEA. After treatment with vehicle, we observed bursts in addition to many spikes ( Fig. 6A ). GABA treatment (0.2, 1, 5, and 20 μM) reduced spike counts. Although bursts were still seen with 0.2 and 1 μM GABA, burst duration and spike counts per burst decreased with increasing GABA concentration, with bursts disappearing at 5 μM GABA. The remaining spikes were almost completely abolished by the addition of 100 nM TTX. GABA treatment significantly reduced spike counts in a concentration-dependent manner from 0.2 μM (p < 0.001 vs the vehicle treatment [0.1% distilled water]; Fig. 6B ).
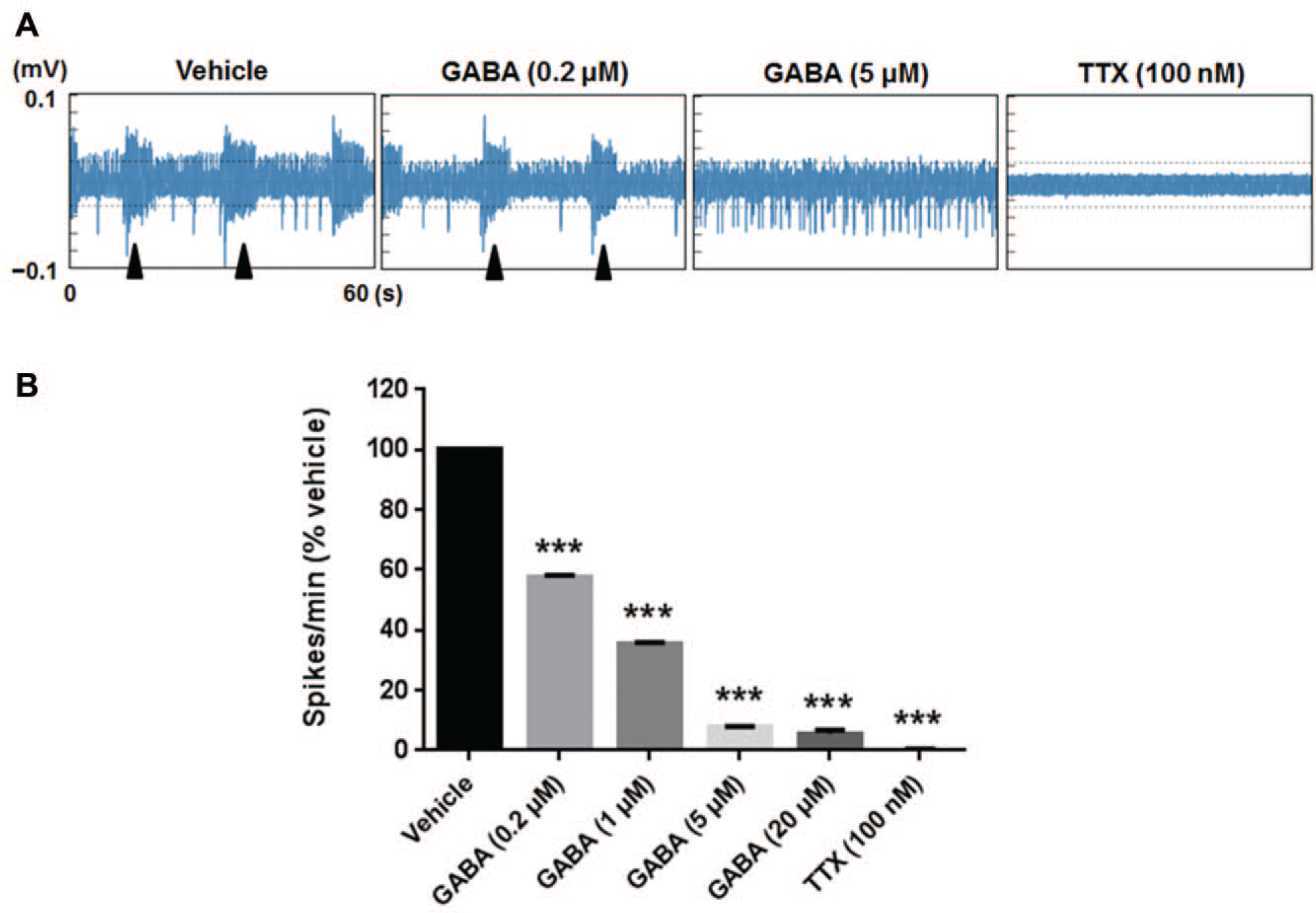
Effects of GABA on spontaneous firing in neurons differentiated from lt-NES cells. Lt-NES cells were differentiated in commercial neural differentiation medium (PS) under 2% O2 for 7 weeks. After baseline levels of spontaneous firing were measured, the neurons were treated with GABA at increasing concentrations (0.2, 1, 5, and 20 μM), and then with TTX at 100 nM. (
Discussion
Immunocytochemical and gene expression analyses revealed that both the medium and the O2 condition profoundly affected the direction of differentiation of neural cells from hiPSC-derived lt-NES cells and the expression levels of genes involved in neural differentiation and maturation. Lt-NES cells differentiated into neurons in both DN and PS media, but more GFAP+ cells were generated in PS medium than in DN medium. PS medium also supported higher expression levels of many genes related to receptors (e.g., AMPA, NMDA, kainate, and GABA receptors) and synapse formation (e.g., synapsin and PSD-95) than did DN medium, and expression of these genes under 2% O2 was higher than under 20% O2. Clustering analysis of gene expression in lt-NES cells and lt-NES cell–derived neural cells differentiated under these different conditions showed that PS medium and hypoxia each promoted neural differentiation separately and in an additive or synergistic manner.
SLC12A5 and SLC12A2 encode a potassium-chloride transporter (KCC2) and a sodium-potassium-chloride transporter (NKCC1), respectively. NKCC1 is expressed in neurons and nonneuronal cells, whereas KCC2 is neuron specific and is particularly expressed in mature neurons, where it contributes to mature dendritic spine formation and functional excitatory synapses. 21 A developmental switch from NKCC1 to KCC2 occurs during neuronal maturation, 22 and we observed that 2% O2 reduced SLC12A2 expression and increased SLC12A5 expression, further supporting the contribution of hypoxia to neuronal maturation and network formation (e.g., more frequent spontaneous synchrony of firings). Moreover, the neuronal maturation process was evidenced by the inhibition of spontaneous firing in lt-NES neurons by GABA, indicating that the developmental shift from depolarization to hyperpolarization in response to GABA had occurred. 22
Of the four conditions tested, PS/2% O2 generated the most mature neurons. Recently, Sun et al. 23 demonstrated that culturing mouse NPCs in 5% O2 enhanced their neuronal differentiation and survival relative to when mouse NPCs were cultured in 20% O2. In 5% O2, mitochondrial oxidative metabolism was high, decreasing glycolysis and reducing the concentration of reactive oxygen species (ROS). It was reported that ROS levels were increased in neural differentiation in 20% O2.24,25 Therefore, inhibition of differentiation-induced ROS by hypoxia may be beneficial for neural differentiation of NSCs. The enhanced neuronal differentiation and function under 2% O2 observed in our study are consistent with these findings.
In calcium imaging assays, glutamate, NMDA, AMPA, and DHPG all produced concentration-dependent calcium rises in neural cells differentiated from human lt-NES cells in all conditions tested. The activities of NMDA, AMPA, and DHPG were inhibited by treatment with the specific antagonists MK-801, NBQX, and JNJ16259685 and MPEP in a concentration-dependent fashion. Thus, NMDA and AMPA receptors, as well as mGluR1 and mGluR5, were pharmacologically functional in these cells. The maximum calcium signal evoked by glutamate, NMDA, or AMPA stimulation was highest in the order of PS/2% O2 ≥ PS/20% O2 > DN/2% O2 > DN/20% O2. For DHPG treatment, the maximum activity in PS/20% O2 was about twofold that in PS/2% O2, but this difference was not statistically significant because of the large variation in the PS/20% O2 samples. The order of EC50 values was similar to that of the maximum activities. The maximum activities are presumably affected by the functional maturation of neural cells and the number of living cells on which assays are performed. Fewer cells were present in DN than in PS medium, which may have resulted in lower maximum activities. However, the degree of responsiveness to glutamate receptor agonists corresponded to the expression level of genes related to the receptors, suggesting that the responsiveness was not merely an artifact of cell number.
mGluR5 was functional in human neural cells derived from 201B7 hiPSC-derived lt-NES cells. This receptor has been a therapeutic target molecule for several disorders of the central nervous system, such as schizophrenia and cognitive deficits associated with drug abuse. PAMs of mGluR5 have been developed because of increased receptor subtype selectivity and reduced potential for receptor sensitization.26,27 The NMDA receptor and mGluR5 are coupled physiologically and biochemically. 28 Activation of mGluR5 leads to functional potentiation of the NMDA receptor, which is expected to alleviate dysfunction of glutamatergic receptors.28-30 To examine whether candidate PAMs of mGluR5 actually enhance neuronal activity by potentiating NMDA receptors in human neurons, assays that use cell lines that overexpress human mGluR5 or human NMDA receptor, or both, and human astrocytes are inappropriate, because these cells cannot provide readouts reflecting neuronal function, such as electric signals. Human neurons expressing both functional endogenous NMDA receptors and mGluR5 will be indispensable as a robust and relevant evaluation model, but there have been few reports regarding such neurons derived from human pluripotent stem cells. In the present study, we observed that both mGluR5 and NMDA receptors were functional in 201B7-derived human neural cells, particularly those cultured in PS/2% O2 or PS/20% O2. These cells may therefore be useful for basic studies and drug development that target mGluR5. Moreover, we detected mGluR5 NAM activity using differentiated lt-NES neural cells. mGluR5 NAMs may be useful for treating a variety of conditions, including pain, anxiety, fragile X syndrome, autism, and depression. 31 Therefore, lt-NES neural cells will also be useful for screening compounds that possess mGluR5 NAM activity.
We found that the medium affected astrocyte differentiation: PS medium induced more GFAP+ cells and higher GFAP gene expression than did DN medium. Together with the neuronal differentiation and maturation described above, these results suggest that PS medium contains factors that induce the differentiation of both neurons and astrocytes. Although the detailed ingredients and compositions of PS medium have not been published, PhoenixSongs Biologicals has provided information that it contains db-cAMP and ascorbic acid. Because DN medium does not contain either compound, both db-cAMP and ascorbic acid could play important roles in the acceleration of neural differentiation of lt-NES cells. Novel neural differentiation media have been developed to improve the quality of neurons derived from human pluripotent stem cells. Under these circumstances, Bardy et al. 14 recently reported that classic basal media (DMEM/F12 and Neurobasal) and serum impair electrophysiological activity and synaptic function of neurons derived from hiPSCs. This prompted them to design a novel medium (BrainPhys containing a serum-free supplement), in which concentrations of inorganic salts, neuroactive amino acids, and energy substances were adjusted to overcome these impairments.
Our results show that PS medium has several advantages over DN for screening compounds: PS medium requires fewer lt-NES cells and reduces the differentiation period. The effects of medium (or its components) and O2 condition on gene expression, functional maturation of glutamate receptors, and neural activity can directly affect signal-to-noise ratios and Z factors in compound screening. Therefore, the conditions under which we cultivate cells may be as important as the type of cells we use to optimize screening procedures. Optimizing the combination of medium and O2 condition will enable us to establish a novel compound screening system. Such a system could provide functional hiPSC-derived neurons or neurons derived from patient iPSCs or genome-edited iPSCs, which are physiologically relevant and potentially related to specific diseases.
Footnotes
Acknowledgements
We thank Marsha Roach of PhoenixSongs Biologicals for providing us information on the contents of the PS medium.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: H.O. is a scientific consultant for San Bio Co., Ltd. All other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by grants from the Program for Intractable Disease Research Utilizing Disease-Specific iPS Cells funded by the Japan Science and Technology Agency (JST)/Japan Agency for Medical Research and Development (A-MED) to H.O., and the Ministry of Health, Labor and Welfare (MHLW) of Japan to H.O. J.K. and H.O. received research funding from Eisai Co., Ltd.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
