Abstract
Spheroid cultures of cancer cells reproduce the spatial dimension–induced in vivo tumor traits more effectively than the conventional two-dimensional cell cultures. With growing interest in spheroids for high-throughput screening (HTS) assays, there is an increasing demand for cost-effective miniaturization of reproducible spheroids in microtiter plates (MPs). However, well-to-well variability in spheroid size, shape, and growth is a frequently encountered problem with almost every culture method that has prevented the transfer of spheroids to the HTS platform. This variability partly arises due to increased susceptibility of MPs to edge effects and evaporation-induced changes in the growth of spheroids. In this study, we examined the effect of evaporation on the reproducibility of spheroids of tumor and nontumor cell lines in 384-well plates, and show that culture conditions that prevent evaporation-induced medium loss result in the formation of uniform spheroids across the plate. Additionally, we also present a few technical improvements to increase the scalability of the liquid-overlay spheroid culturing technique in MPs, together with a simple software routine for the quantification of spheroid size. We believe that these cost-effective improvements will aid in further improvement of spheroid cultures for HTS drug discovery.
Introduction
Three-dimensional (3D) cultures of tumor cell lines have gained a pivotal role in cancer research and drug discovery due to their relative closeness to physiological conditions. 1 Cancer cell spheroids reproduce the complexities of solid tumors, such as cell–cell interactions, zones of proliferating and quiescent cells, diffusion barriers and multicellular resistance, and regions of hypoxia, more effectively than two-dimensional (2D) cultures. 1 Due to these tumor-like properties, spheroids are considered biochemically and pathophysiologically more relevant in vitro tumor models for rational testing of drug efficacy and toxicity alongside 2D cultures.2,3
A number of standardized protocols have been developed for 3D cultures of cells that have undergone significant modifications for high-throughput (HT)/high-content adaptability.1,3,4 However, well-to-well variability in spheroid size, shape, and growth is a daunting challenge that has prevented the reliable adoption of spheroids for HT screening (HTS) assays.3 –5 Nonetheless, continuous endeavor to tackle these challenges over the last few years has resulted in increased use of spheroid cultures in mainstream drug discovery.
Among various 3D culture methods, liquid overlay on agarose-coated plates is a frequently used technique of spheroid formation. 3 Agarose-based cultures are relatively inexpensive compared to commercial 3D culture plates, and easier to adapt from standard multiwell plates to microtiter plates (MPs) than using scaffolds.2,6,7 Recent reports that show the formation of uniform spheroids in 96- 7 and 384-well plates (WPs) with low standard deviation of spheroid area 2 make liquid overlay on agarose amenable to HTS assays. The potential use of this culturing technique for HTS was further validated by pilot screens of small-molecule libraries in 96- and 384WP formats.2,7 Interestingly, the agarose method has been miniaturized to 1536WPs with an additional step that includes coating of cell aggregates with growth factor–reduced Matrigel, and has resulted in uniform spheroid formation. 7
Although miniaturization of assays is highly desirable for HTS, edge effect is one of the common challenges often encountered in MPs.8,9 A longer incubation of plates over 24 h is susceptible to faster evaporation of the medium from peripheral wells, which results in inconsistent readouts.10,11 Furthermore, small culture volumes in MPs are more prone to evaporation-induced osmolality shifts in the medium that critically affect the growth and metabolic activities of cells.9,10,12,13 Sustainable improvements in culture conditions that prevent uneven loss of the medium decrease well-to-well variability in assay readouts.9,10 Additionally, use of embryo-grade mineral oil is another alternative cost-effective approach of reducing medium evaporation from MPs.
Various studies have reported the formation of reproducible spheroids by agarose-overlay and other culture methods under standard cell culture conditions in MPs;1,2,7 however, there have been no in-depth studies to investigate the variability in spheroid formation across wells. In this study, we examined the reproducibility and uniformity of spheroid size of different tumor and nontumor cell lines across a 384WP cultured under different combinations of plate lids and incubators. Furthermore, we investigated the effect of spheroid size variability on drug response to show that culture condition that prevents excessive loss of medium reduces well-to-well variability in drug response.
Materials and Methods
Evaporation Check
Plate wells were categorized into six groups based on their distance from the plate boundary ( Fig. 1A ). To check for the immediate effect of evaporation on medium volume across the six groups, 384WPs (CellCarrier, clear-bottom; PerkinElmer, Waltham, MA) with/without sterile H2O in the liquid reservoir in the periphery of plates ( Fig. 1A ) were filled with an equal volume of medium by Multidrop Combi Reagent Dispenser (Thermo Fisher Scientific, Inc., Waltham, MA). The plates were then incubated at 37 °C for 4 days under four different combinations of plate lids and incubators: regular plate lid and standard incubator (Combination 1); regular plate lid and SteriStore Rotary incubator with 95% humidity, 5% CO2, and 20% O2 (HighRes Biosolutions, Woburn, MA; Combination 2); MicroClime Environmental lid (Labcyte, Inc., Sunnyvale, CA) and standard incubator (Combination 3); and MicroClime Environmental lid and SteriStore incubator (Combination 4). Prior to use, MicroClime lids were filled with sterile H2O according to the manufacturer’s protocol. The residual volume of medium per well was measured manually on day 4 by a multichannel pipette, and the percent medium loss relative to day 0 was considered a measure of evaporation. 8
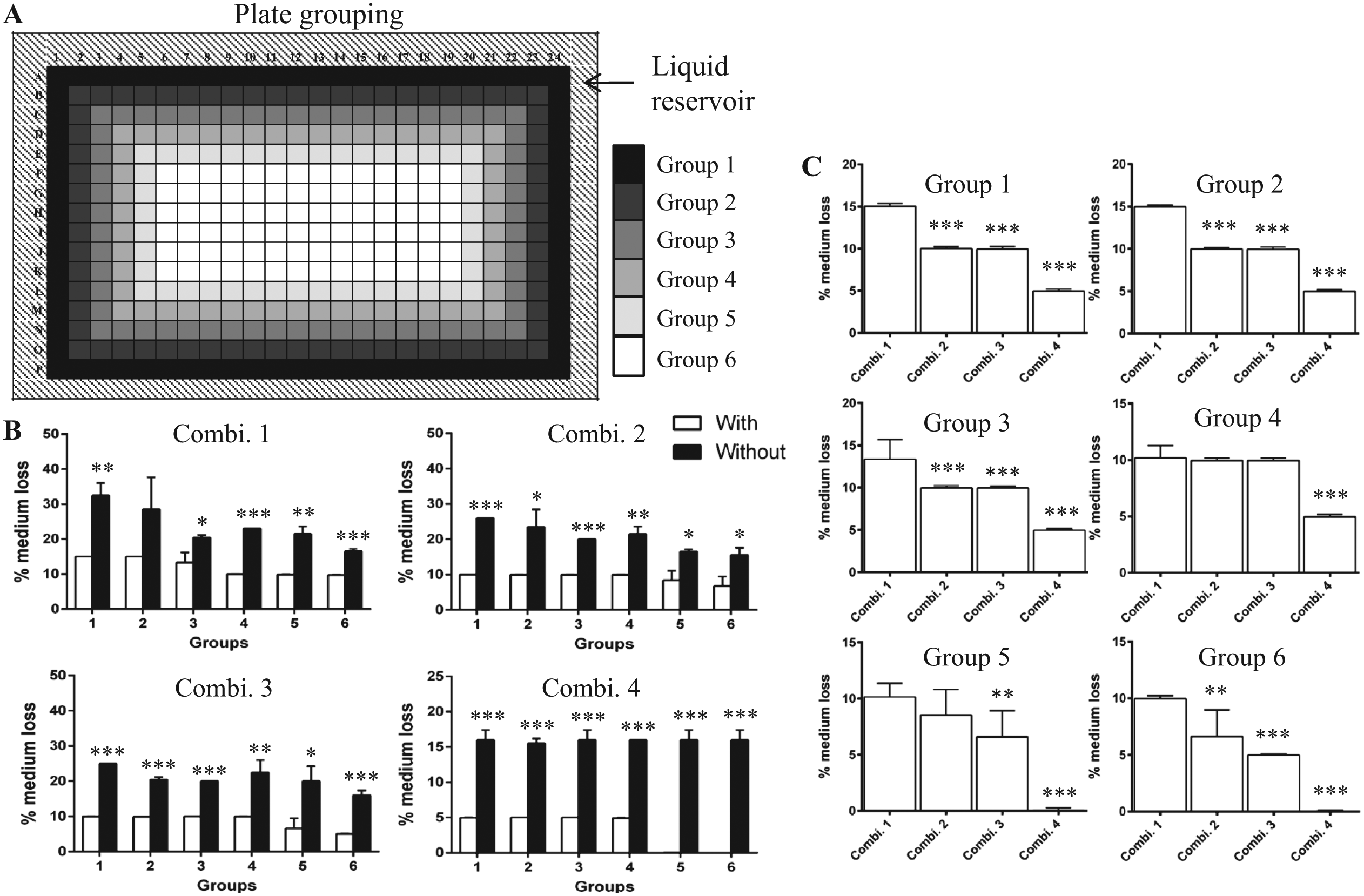
Uneven loss of cell culture medium. (
Cell Culture
Human colorectal HCT116 and HT29 tumor cells and osteosarcoma U-2OS cells were cultured in McCoy’s 5A medium (Sigma-Aldrich, Prague, Czech Republic) supplemented with 10% fetal bovine serum (FBS; Gibco, Thermo Fisher Scientific, Inc.) and 2 mM
Generation of Spheroids Using a Modified Liquid-Overlay Technique
Spheroids were formed in CellCarrier clear-bottom 384WPs using a modified version of liquid-overlay technique. 14 For plate coating, a 0.75% (w/v) low-melting agarose (Sigma-Aldrich) stock solution was prepared in phenol red–free McCoy’s medium without FBS. The agarose solution was then autoclaved to sterilize, and filtered through a 0.22 µm “rapid” Filtermax 500 mL filter top (TPP Techno Plastic Products AG, Trasadingen, Switzerland). Plates were then coated with 15 µL of filtered 0.75% agarose by Multidrop Combi Reagent Dispenser using a Small Tube Plastic Tip Dispensing Cassette (Thermo Fisher Scientific Oy, Vantaa, Finland) under sterile conditions. The coated plates were either used the same day or aseptically stored at 4 °C for up to 2 weeks.
Before cell seeding, agarose-coated plates from 4 °C were allowed to equilibrate to room temperature (RT). Cells were seeded at a density of 2.5 × 104 cells/mL per well in growth medium by Multidrop Reagent Dispenser using a Standard Tube Dispensing Cassette (Thermo Fisher Scientific Oy) at medium speed to prevent disruption of agarose layer. The plates were then centrifuged at 4
Prewarmed medium was added to each well on day 4 using an EL406 Microplate Washer Dispenser (BioTek, Winooski, VT). Medium was replaced regularly every 3 days by the EL406 washer manifold, and an equal volume of media was added by the peripump dispenser of the EL406. The
High-Content Imaging and Image Analyses
Spheroids in agarose-coated plates were imaged on days 4 and 14 in a fully automated CellVoyager High-Content Imaging System (Model CV7000; Yokogawa Electric Corporation, Tokyo, Japan) using a 4× air objective (N.A. 16). The 4× objective with higher numerical aperture and sCMOS camera enable imaging of a complete well, thereby capturing spheroids that are occasionally not in the center of the well. Bright-field
Images were stored in the TIFF format, and spheroid characteristics were analyzed using an in-house algorithm developed in MatLab R2013b (MathWorks, Inc., Natick, MA). Briefly, the sharpest image with the maximum L1-norm of the image gradient was selected from the
Drug Treatment and Cytotoxicity Assay
HCT116 spheroids on day 6 cultured under different plate lid and incubator combinations were treated with 10 µg/mL oxaliplatin (Ebewe Pharma Ges.m.b.H., Unterach, Austria) for 48 h. Following drug treatment, 10 µL of medium was transferred from each drug-treated and untreated/control wells to a new flat-bottom 384WP (CellCarrier) using a robotic liquid handling system (Tecan Freedom EVO 150; Tecan Group Ltd., Männedorf, Switzerland). Cytotoxicity was measured using a Thermo Scientific Pierce LDH Cytotoxicity Assay Kit (Thermo Fisher Scientific, Inc., Rockford, IL) following manufacturer’s instructions, and the absorbance of assay plates was read in an EnVision Multimode Plate Reader (PerkinElmer). Percent cytotoxicity was calculated according to the instructions of the LDH Cytotoxicity Assay Kit.
Data Analysis
Data were analyzed using GraphPad Prism (version 6; San Diego, CA) and MatLab R2013b (MathWorks Inc.). Statistical difference was considered significant at
Results and Discussion
Despite the use of automated liquid handling systems to maximize spheroid reproducibility, uneven culture conditions potentially due to medium evaporation result in well-to-well variability in spheroids in microplates. 14 To address these concerns, we examined the effect of four different combinations of plate lids and incubators on medium evaporation and subsequently on the reproducibility of uniform spheroid formation of tumor and noncancerous cell lines.
First, we quantified the loss of medium across the six groups of wells under the different conditions as an immediate effect of evaporation. Incubation of 384WPs without H2O in the outer liquid reservoir resulted in significant loss of medium across the six groups of wells compared to plates with H2O in the reservoir under all four combinations of plate lids and incubators ( Fig. 1B ). We then performed a comparison of plate lid and incubator combinations within individual groups for 384WPs incubated with H2O in the outer reservoir. There was significant loss of medium from Groups 1–3 and 6 (vs Combinations 2–4), Group 4 (vs Combination 4), and Group 5 (vs Combinations 3 and 4) of 384WPs cultured in standard incubators with regular plate lids ( Fig. 1C ). Altogether, our data suggest a reduced loss of medium across plates with MicroClime lids cultured in the SteriStore incubator with a well-controlled humidified atmosphere. These findings are in agreement with those of Walzl et al., 10 who showed that culturing 96WPs in a humidity chamber placed inside standard incubators abolished the uneven loss of medium from the outer wells of plates.
Although spheroids effectively reproduce pathophysiological traits of solid tumors, differences in spheroid size and shape alter their response to treatments.4,15 Therefore, generation of a uniform spheroid is imperative for reliable intra- and interplate data interpretation of HTS assays. To check for the effect of medium loss across the six groups on the uniformity of spheroid formation, we performed a groupwise analysis of area of spheroids of HCT116 cells from the sharpest
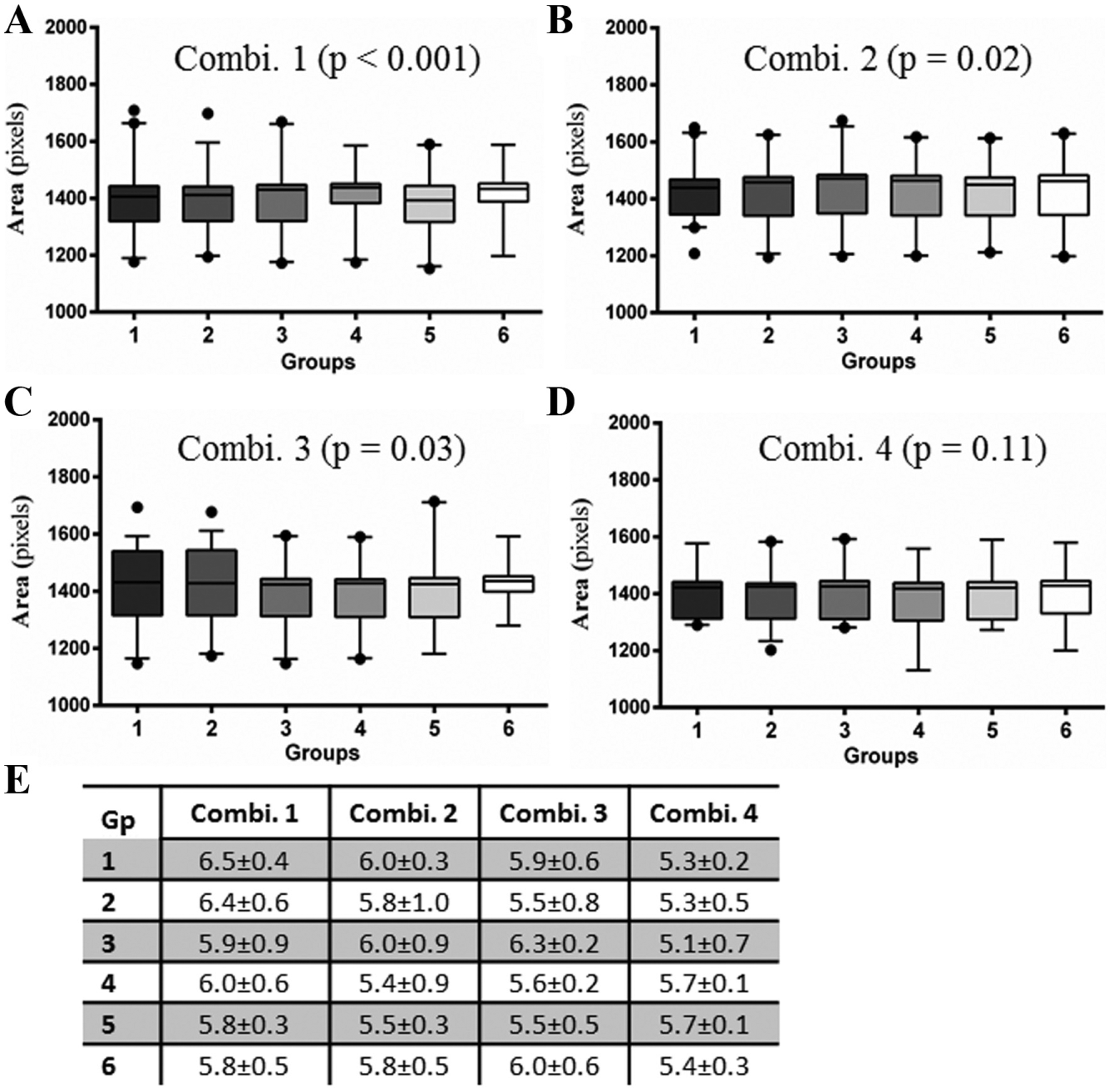
Comparison of spheroid size uniformity and reproducibility across 384WPs. On day 4, spheroids of HCT116 cells cultured under the four different combinations of plate lids and incubators were compared for uniformity of size across the six groups of wells. (
Since long-term cultures of cells are more prone to evaporation-induced changes in the culture medium that alter cell growth,12,13 we allowed 4-day-old spheroids to grow for an additional 10 days with regular change of medium to determine the variability in spheroids within the six groups. The CSA of day 14 spheroids significantly differed among six groups in plates under Combinations 1, 2, and 4 (Fig. 3Ai,ii,iv). Surprisingly, there was no statistically significant difference in spheroids in culture Combination 3 (
Fig. 3Aiii
). Unlike day 4, the intragroup CVs were smaller in plates cultured under Combination 1 than in Combination 4 (
Fig. 3B
). Additionally, spheroids in plate–incubator Combination 1 were of larger CSA and consequently of larger size (Fig. 3C,D), whereas those in Combination 3 were significantly smaller than those in other combinations (
Fig. 3D
). These data show that the growth of 14-day spheroids is greatly affected in standard incubators with the MicroClime plate lid combination. As an equal volume of media was replaced every 3 days for each plate, the difference in spheroid size between combination types could have presumably resulted due to differences in incubator conditions. It is to be noted that the CSA of spheroids did not differ significantly between plates with regular (Combination 2) and MicroClime (Combination 4) plate lids in the SteriStore incubator (
Fig. 3D
). The pooled CVs for the four combinations (
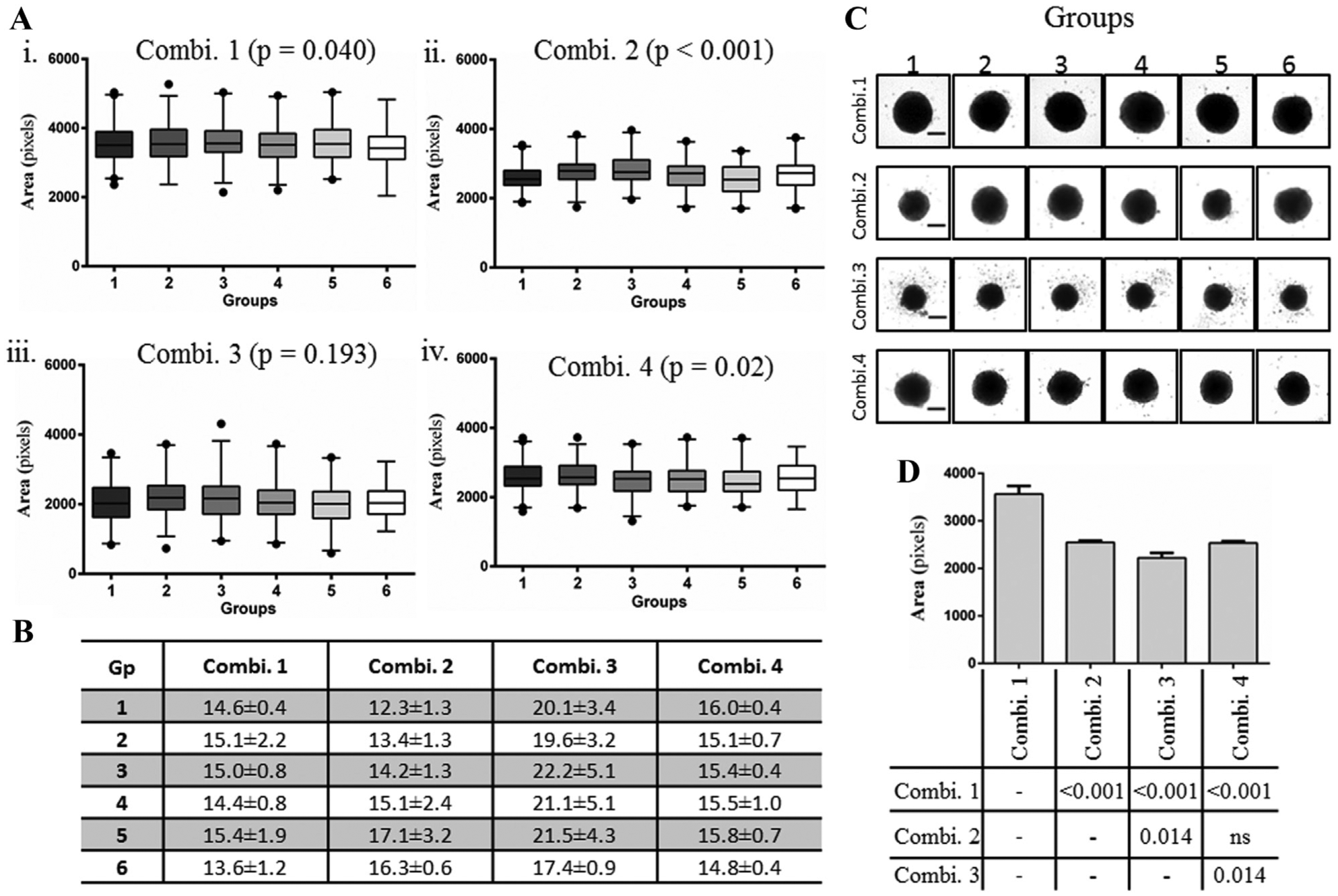
Increase in variability in long-term cultures of HCT116 spheroids. Day 4 spheroids were allowed to grow for an additional 10 days with medium change every 3 days, and the spheroid size was measured on day 14. (
It has been well documented that not all cell types are suitable for a particular 3D culture method,
1
and different tumor cell lines form spheroids that show differences in size, shape, histology, and growth kinetics. Therefore, we next determined the suitability of the MicroClime plate lid and SteriStore incubator (Combination 4) for uniform spheroid formation for additional tumor and noncancerous cell lines that we use frequently for drug toxicity studies. HT29, HeLa, and HepG2 cells formed uniform-sized spheroids across the six groups on day 4 (
Fig. 4A
). Likewise, the nontumor HEK293 and BJ cells also formed uniform spheroids under the culture Combination 4 (
Fig. 4A
). However, both Caco-2 and U-2OS did not aggregate into spheroids on day 4 or even after 10 days in culture (
Fig. 4B
). The CVs of CSA of spheroids of HT29, HeLa, HepG2, HEK293, and BJ cells were less than 7% in the six groups on day 4, indicating greater reproducibility of spheroid formation for these cell types under Combination 4 culture condition (
Fig. 4C
). Interestingly, irrespective of seeding an equal cell number per well on day 0, each cell type formed spheroids of different size by day 4 (
Fig. 4D
). This indicates the inherent property of different cell lines to form spheroids of different size and compactness. Similarly to HCT116 spheroids, day 4 spheroids of HT29, HeLa, HepG2, HEK293, and BJ were allowed to grow for an additional 10 days. Except BJ cells, there was no significant difference in the size of day 14 spheroids of all other cell lines (
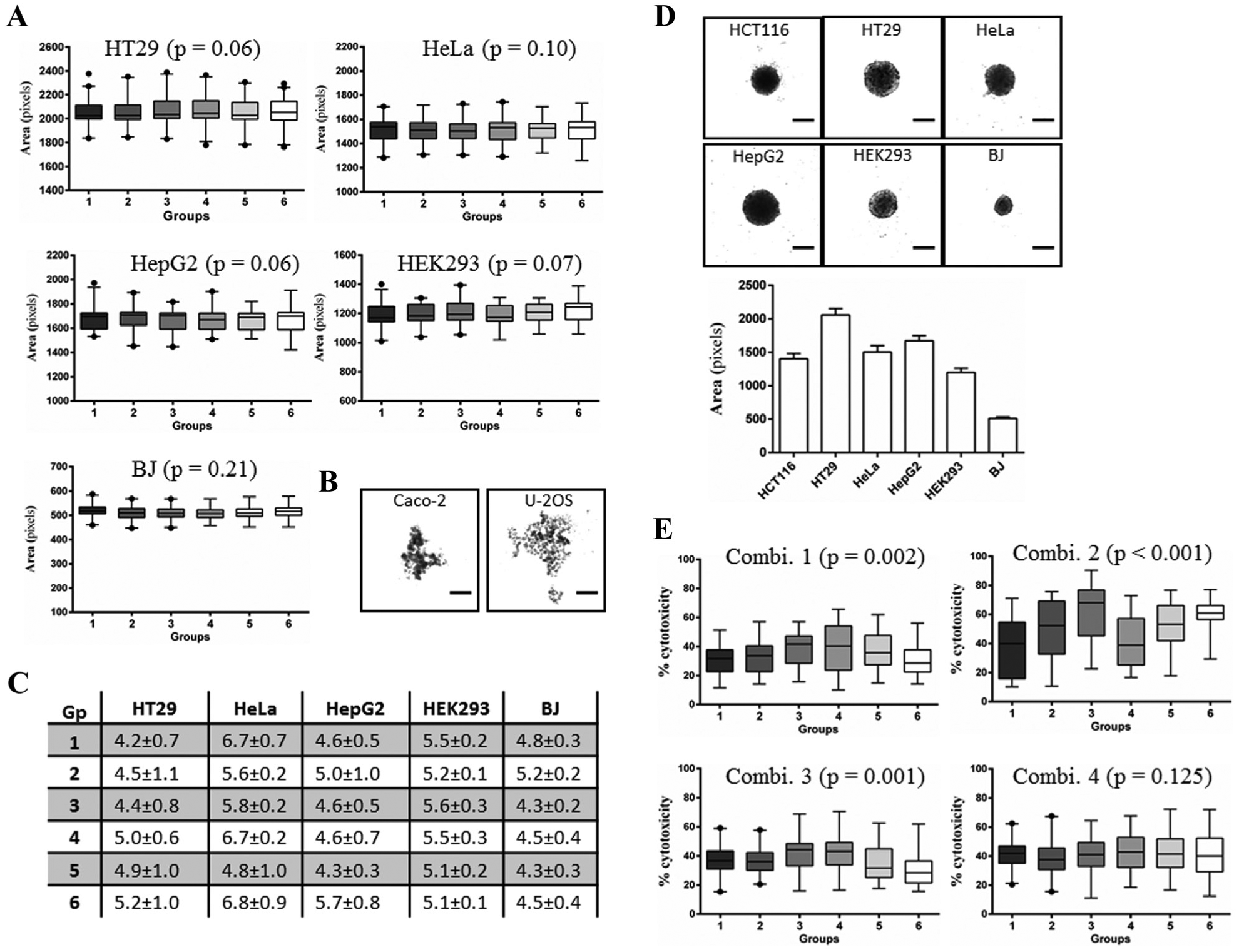
Reproducibility of spheroids of other cell lines under Combination 4. To examine the general suitability of the MicroClime lid and SteriStore incubator combination, spheroids of five tumor and two noncancerous cell lines were compared for uniformity and reproducibility of size. (
As a proof of concept of the effect of variable spheroid size on drug respsone, 15 we examined the effect of oxaliplatin on spheroids across 384WPs under the four different combinations of plate lids and incubators. HCT116 spheroids were treated with 10 µg/mL oxaliplatin for 48 h on day 6, and the cytotoxicity determined by LDH assay. Not surprisingly, cytotoxicity of oxaliplatin significantly differed across the six groups in 384WPs cultures under Combinations 1–3, which resulted in the formation of variable-sized spheroids across wells ( Fig. 4E ). In contrast, there was no significant difference in the response of spheroids across wells to oxaliplatin in the Combination 4 culture condition, which did not affect HCT116 spheroid size homogeneity ( Fig. 4E ).
Lastly, preparation of agarose-coated MPs using robotic systems is another practical challenge of the agarose-overlay technique of spheroid culture for HTS assays. 4 Clogging of dispensing tubes with agarose is of potential concern and requires agarose solution or the whole dispensing unit to be kept heated.2,14 To address this issue, we used a 0.75% (w/v) filtered agarose solution that does not gel as rapidly as unfiltered 1%–1.5% (w/v) agarose, and needs no heating during dispensing. Using filtered 0.75% agarose solution and a Multidrop dispenser, we were able to coat a package of 384WPs containing 16 plates in less than 5 min without the issue of clogging of dispensing cassette tubing. Any remaining agarose present in the dispensing cassette can be easily washed out from cassettes by priming with 70 °C–80 °C distilled H2O.
The goal of the present study was to examine the well-to-well variability in spheroid size using the agarose-overlay technique of spheroid culture in 384WPs. We show that a cell culture condition that prevents significant loss of medium due to evaporation improves the reproducibility of uniform spheroid formation across plates. Additionally, culturing spheroids of certain cell types under this condition does not affect their uniform growth in long-term cultures. Furthermore, we also show that a significant variability in spheroid size results in altered drug response across assay plates. Minor technical modifications, such as the use of a 0.75% filtered agarose solution, increase the throughput of the plate preparation step of the agarose-overlay culturing method. The modifications presented in our study require no additional equipment and consumables, and can be set up with the existing facilities available in HTS laboratories.
Footnotes
Acknowledgements
This work was supported by grants from the Czech Ministry of Education, Youth and Sports (LO1304) and the Technological Agency of the Czech Republic (TE02000028).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
