Abstract
Small-molecule inhibitors have been previously investigated to identify possible therapeutics for the treatment of chronic pain. In the present study, known nerve growth factor (NGF) inhibitors identified by 125I-NGF binding were characterized using affinity and binding evaluations by surface plasmon resonance (SPR) spectroscopy. A novel strategy for characterizing NGF inhibitors was used to determine the binding affinity (KD) and saturation ability of each compound with immobilized NGF. Seventy-four percent of compounds screened demonstrated a positive binding event to NGF. A KD less than 10 μM and a percent saturation greater than 50% were used as thresholds to identify inhibitors that would warrant further investigation. This study details for the first time a methodology that can be used to directly characterize the binding event between small-molecule inhibitors and NGF.
Introduction
Neurotrophins play a crucial developmental role in regulating the survival and differentiation of neurons in both the peripheral and central nervous systems. 1 Nerve growth factor (NGF) is a member of the neurotrophin family and has been well established with a role in development in sympathetic and sensory neurons. 2 NGF has also been associated with pain-signaling systems in adult animals and in humans by binding and activating high-affinity tyrosine kinase receptor TrkA. 3
Inhibiting the activity of NGF may have significant therapeutic potential for pathologies related to neuropathic pain, including congenital insensitivity to pain with anhidrosis (CIPA) or hyperalgesia, where a dysfunction of the NGF–TrkA signaling pathway is observed. 4 One method of inhibiting the effect of NGF is with the use of the anti-NGF monoclonal antibody tanezumab. Clinically, tanezumab has demonstrated efficiency in the treatment of pain in patients with osteoarthritis, chronic lower back pain, and diabetic peripheral neuropathy. 5 However, there remains limitations with the use of tanezumab with respect to autoimmune responses, variability in pharmacokinetics, as well as drawbacks in safe administration of the antibody and production cost. 6 Tanezumab was also under several clinical holds during its phase studies due to evidence of peripheral nerve effects. Using an approach to inhibiting NGF with the use of small molecules may have significant pharmacological, practical, and economic advantages over monoclonal antibodies. Small molecules such as ALE 0540, 7 PD 90780, 8 PQC 083, 9 Ro 08-2750, 10 and Y1036 11 have previously demonstrated an ability to inhibit NGF activity in vitro, and several of these have been suggested to bind the ligand NGF rather than the receptor.
The mature form of NGF is a symmetrical dimer consisting of two identical monomers 12 that associate by hydrophobic interactions. 13 The structure of NGF, proposed by McDonald and colleagues, describes a 118-amino-acid sequence that forms a monomer with four distinct loop regions and two β-pleated sheet strands. 13 Molecular modeling of PD 90780 and PQC 083 binding to NGF suggest a binding site at the loop I–loop IV cleft of NGF,8,9 which would suggest a 2:1 stoichiometry (two small molecules to one NGF dimer). Originally, ALE 0540 was thought to bind to the binding sites on TrkA and p75 receptors 7 (rather than to NGF), and Ro 08-2750 was suggested to inhibit NGF at the hydrophobic dimer interface. 10 Others have suggested, by using molecular modeling evidence, that this is not likely to be the mechanism of inhibition and that ALE 0540 and Ro 08-2750 bind similarly to NGF as PD 90780. 12 Y1036 has been suggested to bind near the hydrophobic dimer interface, 11 more specifically at Lysine-57, 14 suggesting a 2:1 stoichiometry.
Previously used screening strategies for NGF inhibitors required the use of radioisotopes, such as 125I.7–9,11 Inhibition constants were calculated based on the amount of radioactive decay detected as labeled NGF bound to receptors, a process that would be inhibited when NGF was previously exposed to active small molecules. However, using radioisotopes has the potential to alter the molecular function of NGF, and it has the limitation that the small molecules may be interacting with the receptor rather than NGF, which would give similar inhibitory results.
The present work deals with the application of biosensor technology to observe the direct interaction between small molecules and NGF without the use of radiolabels. Having an alternative label-free method for measuring the small-molecule–NGF binding event rather than the downstream binding, signaling, or functional effect of the event would be of great benefit to understanding the mechanisms of these inhibitors.
Materials and Methods
Binding of 125I-NGF to PC12 Cells
Full-length (1–118) mouse NGF purified by high-performance liquid chromatography from 2.5S NGF (purity greater than 95%) was obtained from Cedarlane Laboratories (Toronto, ON). The iodination of NGF was performed as previously described. 15 The 125I-NGF obtained (typically 80–120 c.p.m./pg) was purified by size exculsion chromatography on a PD10 column (Pharmacia) preequilibrated with HEPES Krebs–Ringer (HKR) buffer [10 mM HEPES (pH 7.35) containing 125 mM NaCl, 4.8 mM KCl. 1.3 mM CaCl2, 1.2 mM MgSO4, 1.2 mM KH2PO4, 1 g/l glucose, and 1 g/l BSA]. PC12 cells were grown in RPMI with 10% fetal calf serum. Each data point was set up in a single tube containing 125I-NGF (at the required concentration), 400,000 cells (for a final concentration of 106/ml) and NGF (at 10 nM for nonspecific binding) as required. The tubes were incubated for 2 h at 4 °C, and 100 µl aliquots (providing triplicate data points for each sample) were transferred to 400 µl microcentrifuge tubes containing 200 µl of 10% glycerol in HKR. Tubes were centrifuged for 2 min at 5000 rpm, the tip containing the cell pellet was cut off, and radioactivity bound to the cells was determined.
Surface Plasmon Resonance Spectroscopy
A Biacore T200 spectrometer was obtained from GE Healthcare Life Sciences (Piscataway, NJ, USA). Synthetic organic compounds were synthesized by Sussex Research (Ottawa, ON, Canada). DMSO was obtained from Sigma-Aldrich (Oakville, ON, Canada). A Biacore Amine Coupling Kit was obtained from GE Healthcare Life Sciences. Other analytical-grade compounds were obtained from Fisher Scientific (Ottawa, ON, Canada). Deionized water from a Milli-Q Water Purification System was used for making all solutions. A series S Sensor Chip CM5 was used. HBS-EP buffer containing 0.5% DMSO was used as the running buffer with pH 7.5 (0.01 M HEPES, 0.15 M NaCl, 3 mM EDTA, and 0.05% v/v Tween 20). The running buffer was degassed using a vacuum chamber. Sensor Chip CM5 was docked and normalized with a 70% w/w glycerol solution for stabilization. The instrument was primed with running buffer twice before use.
Surface Preparation with Immobilization of NGF
The NGF protein was coupled using the amine-coupling method. Sensor Chip CM5 was activated using a 7 min contact time of 0.4 M EDC and 0.1 M NHS from the Biacore Amine Coupling Kit, at a flow rate of 10 μL/min. A solution of NGF in acetic acid was first made by dissolving 1 mg NGF into 50 μL of 0.02% acetic acid solution. The NGF–acetic acid solution was then mixed with HBS-EP buffer to yield 10 μg/mL solution. Following activation, the described NGF solution was injected over the activated sensor chip surface for 9 min at a flow rate of 10 μL/min. Immobilization was measured at 6645 response units (RUs) (1 RU = 1 pg/mm2). After the injection, the surface was washed with 1 M NaCl to remove any uncoupled or non-covalently bound material from the surface. The excess hydroxysuccinimidyl groups on the surface were deactivated with 1 M ethanolamine hydrochloride, pH 8.5, for 7 min at a flow rate of 10 μL/min. The surface of a reference flow cell was activated with 0.4 M EDC/0.1 M NHS for 7 min with a flow rate of 10 μL/min, and then deactivated with a 7 min exposure of 1 M ethanolamine at a flow rate of 10 μL/min. With no ligand bound to the flow path, the control flow cell was used to detect nonspecific binding of the small molecules to the sensor chip surface during screening affinity assays.
Small-Molecule Sample Preparation
Measureable amounts of each small molecule were obtained with an analytical balance and dissolved in an appropriate amount of DMSO to give a 10 mM solution. The samples were diluted with the running buffer to yield small-molecule solutions for the assay of concentrations that varied from 0.7 μM to 200 μM.
Analyte Injection
Prior to analyte injection, the series S CM5 chip was conditioned with three 30 s cycles of running buffer followed by three startup cycles, allowing the response to stabilize before analyte injection. Data were collected at a temperature of 25 °C. Individual compound samples were tested from lowest to highest concentrations, separated by a 15 s stabilization period after each sample in each compound series. During each sample cycle, analyte was injected for 60 s at a flow rate of 30 μL/min. A dissociation period was monitored for 30 s after analyte injection before regeneration with 1.0 M NaCl for 120 s at a flow rate of 30 μL/min to wash any remaining analyte from the sensor chip before running the next sample.
Two-Site Steady-State Binding Affinity Analysis
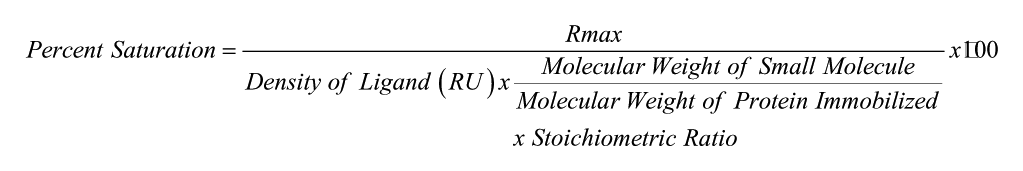
The Biacore T200 was programmed to run an automated assay with the various small-molecule samples. Once the responses were measured, they were processed using Biacore T200 Evaluation Software Version 1.0. The responses measured in the blank flow cell (control) were subtracted from the response measured in the flow cell with protein immobilized. The binding affinities (KD) of each small molecule were obtained by plotting the subtracted responses against the concentration and fitting the curve with a two-site steady-state affinity fit. Each maximal response (Rmax) was compared to the theoretical Rmax calculated for each compound to determine percent saturation of available NGF on the chip. Percent saturation was calculated using the following equation, in which density of ligand refers to the immobilization response of NGF:
Results and Discussion
Fifty novel compounds of an analogue series and four previously reported compounds (PQC 083, PD 90780, ALE 0540, and Ro 08-2750) were screened using the described protocol. 125I analysis identified 21 novel NGF inhibitors. However, with 125I-NGF being an estimation of the binding event, SPR analysis was required to identify NGF-binding inhibitors as opposed to receptor inhibitors.
Figure 1 represents the blank-subtracted sensogram for the concentration-dependent binding of PQC 083 to the NGF immobilized on the sensor chips. Figure 2 represents the two-site steady-state affinity plot for the binding of varying concentrations of PQC 083 to NGF. The sensograms and affinity plots of the remaining 53 compounds binding to the immobilized NGF were of similar quality to that of PQC 083. Binding affinities were calculated from the affinity plots using the Biacore T200 Evaluation Software.
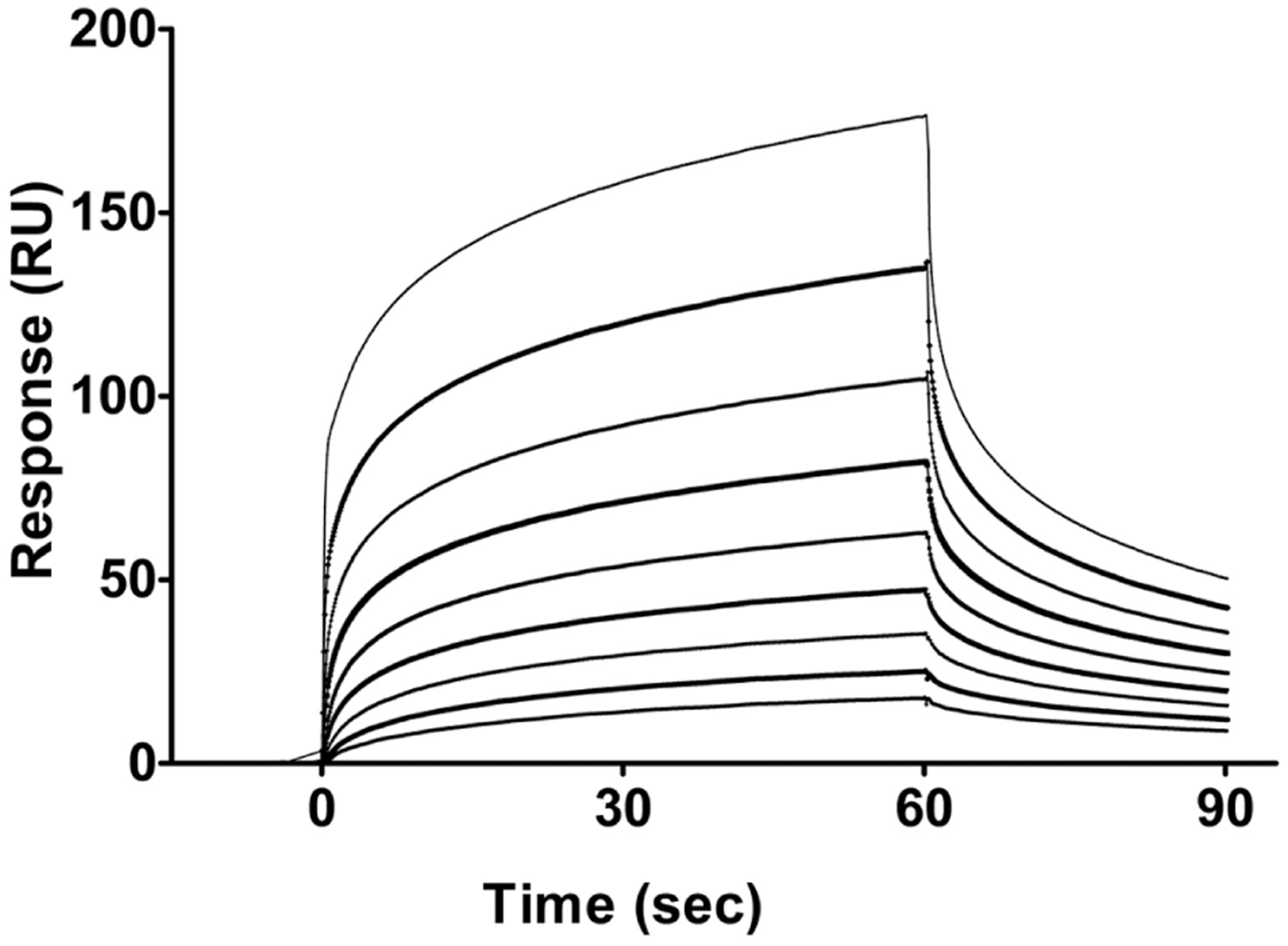
Blank subtracted sensogram that describes the association and dissociation of each concentration of PQC 083 tested in the assay. Time (0–60 s) represents the association of PQC 083 to nerve growth factor (NGF); at 60 s, the analyte flow stops. A dissociation period of 60–90 s is monitored before regeneration of the chip occurs before the next sample is run. Top line: 200 μM. Second line from top: 100 μM. Third line from top: 50 μM. Fourth line from top: 25 μM. Fifth line from top: 12.5 μM. Fourth line from bottom: 6.25 μM. Third line from bottom: 3.13 μM. Second line from bottom: 1.56 μM. Bottom line: 0.78 μM.
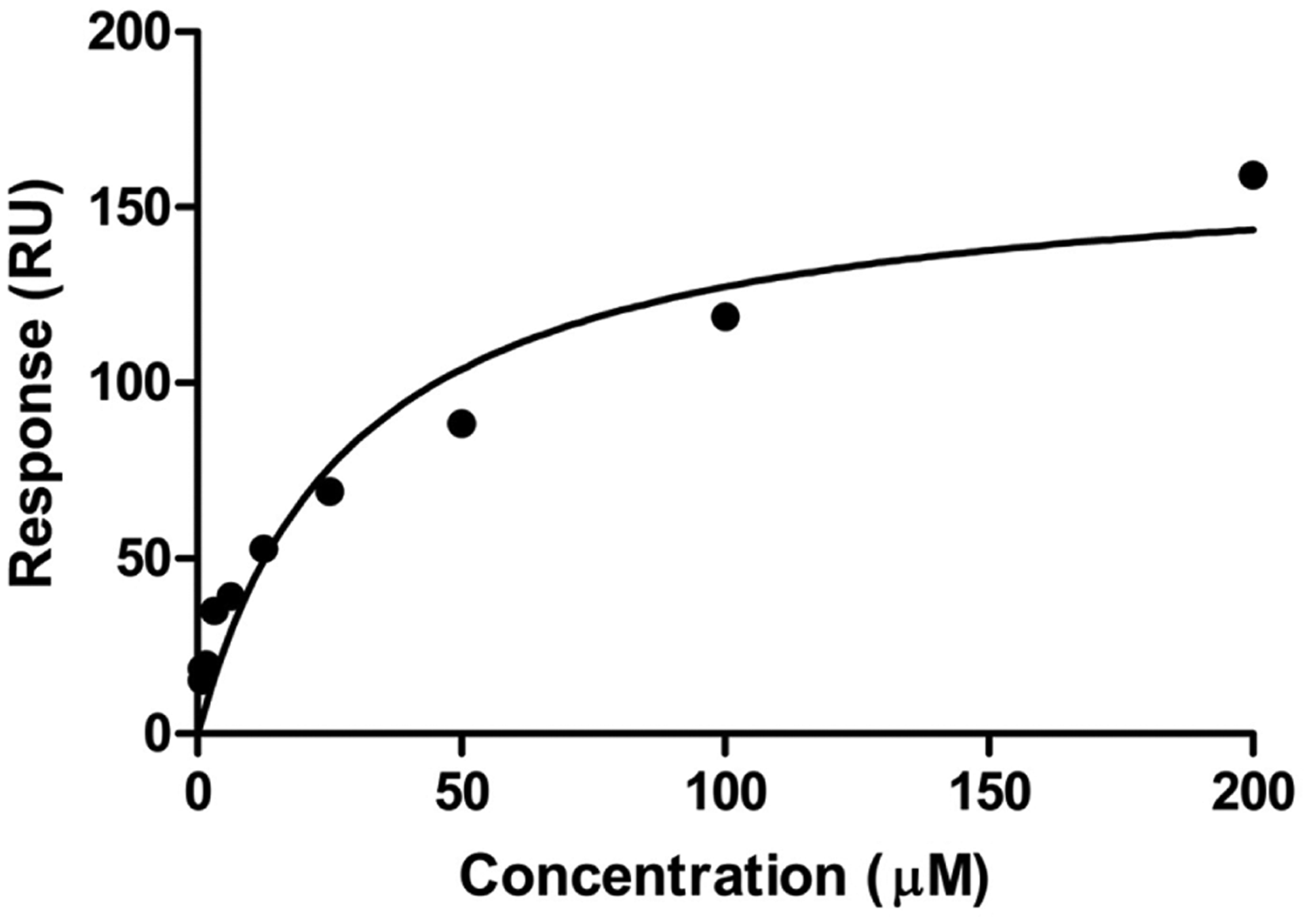
Two-site steady-state binding model describing PQC083 binding to nerve growth factor (NGF); x-axis: concentration (μM); y-axis: subtracted response (response from active flow cell minus response from reference flow cell). Each data point represents the subtracted response from the saturation (last 10 s of the association phase) of PQC 083 at varying concentrations (Figure 1).
Of the 54 compounds analyzed, 74% resulted in a response characterized as direct binding to NGF while using SPR analysis. It was determined that the novel analogues that bound to NGF and the previously reported compounds fit best to a 2:1 stoichiometry (two small molecules to one NGF dimer) and were analyzed using a two-site binding model with the Biacore T200 Evaluation Software. Eighteen percent of the investigated compounds displayed a KD lower than the previously reported compounds (<10 μM). However, the degree to which the compounds were able to saturate the available NGF immobilized varied (0.3–150% saturation; Figure 3). When calculating the percent saturation, a 2:1 stoichiometry was considered, and of the 54 compounds screened, 16% of them were able to saturate the available NGF immobilized to a level of more than 50%. Of these compounds, two had a KD lower than 10 μM (circled in Figure 3) and, therefore, are good candidates for further investigation.
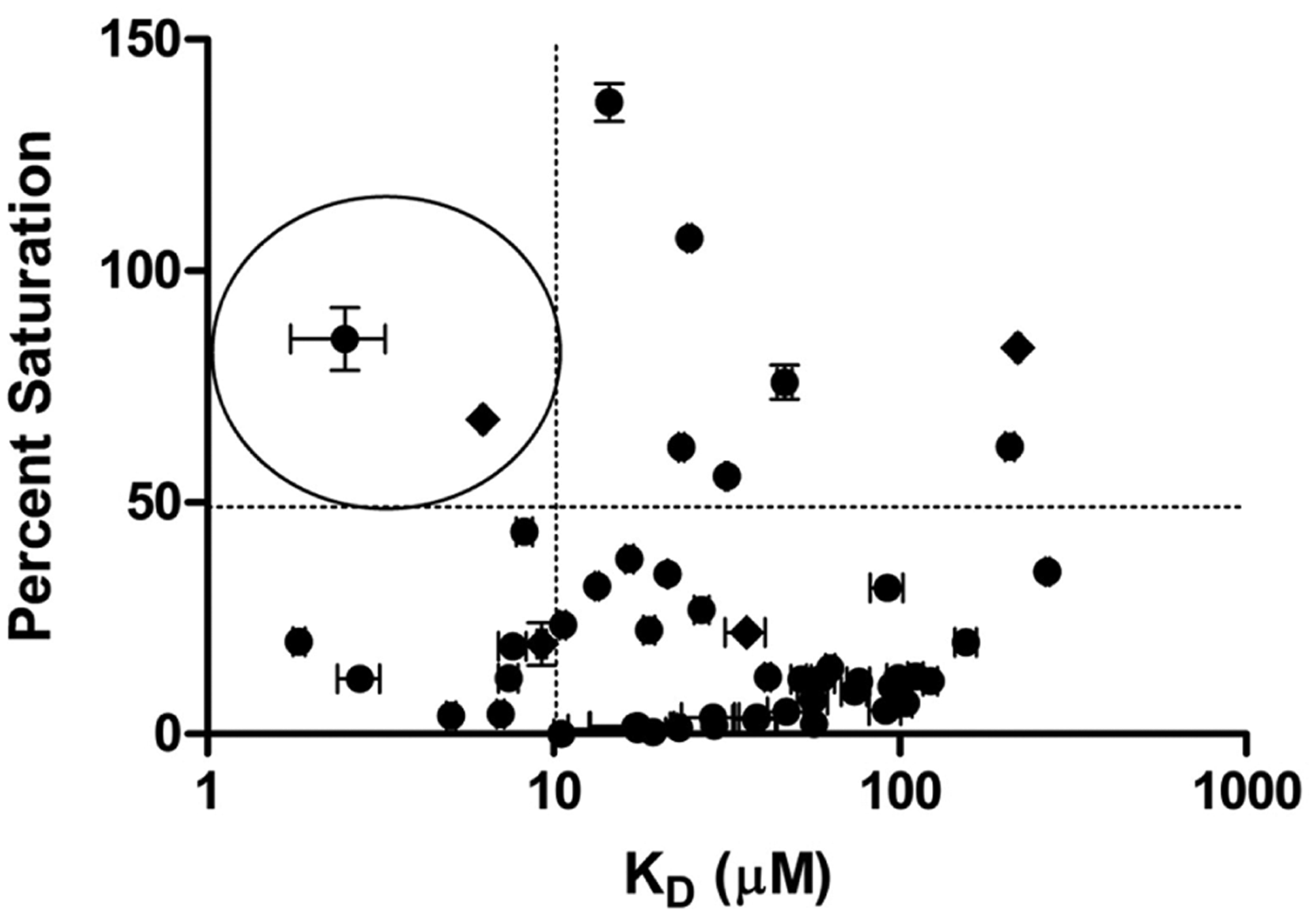
Ranking of small molecules screened using percent saturation and binding affinity [log(M)] to nerve growth factor (NGF). Diamond: previously reported small-molecule inhibitors. Circles: small synthetic analogue series. Circled: two compounds that were able to saturate the available NGF >50% and bind with an affinity <10 μM. Each compound was measured n = 4.
By integrating the SPR analysis into the results determined from 125I-NGF binding analysis, 14 novel compounds and three of the previously reported small molecules were identified as NGF-binding inhibitors (Figure 4). It was also determined that there is a relationship between the high-affinity binding of the two-site binding analysis from the novel compounds to the IC50 of the same compounds determined by 125I-NGF (Figure 4) (F(1,16) = 8.381; p = 0.0111; R2 = 0.3585).
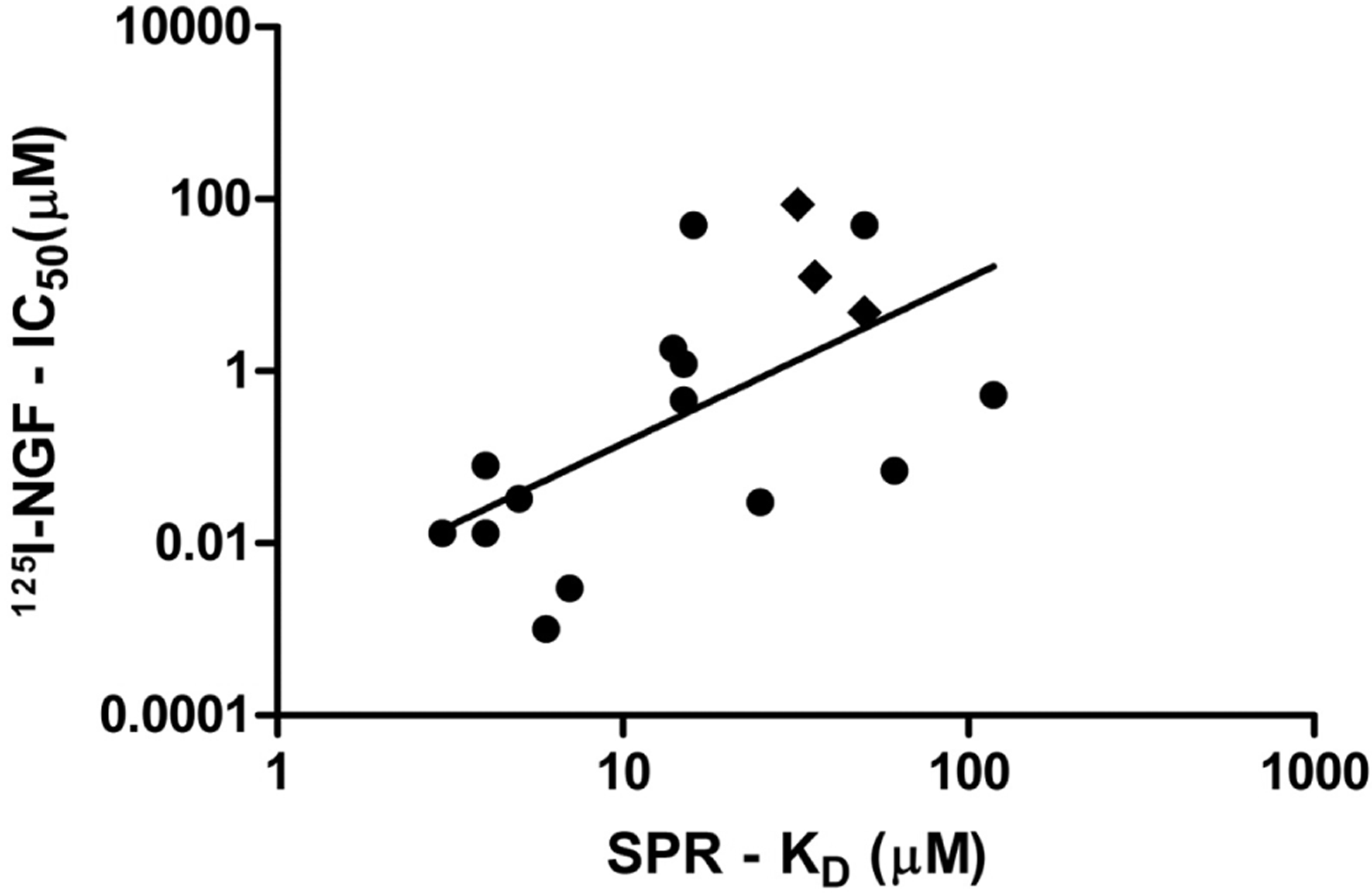
Demonstration of the relationship of 14 novel compounds and three previously reported compounds between their IC50 measured using 125I-NGF and a high-affinity binding affinity (KD) measured using surface plasmon resonance (SPR) two-site analysis. Diamond: previously reported small-molecule inhibitors. Circles: Small synthetic analogue series (F(1,16) = 8.381; p = 0.0111; R2 = 0.3585).
Previous methods for screening small-molecule inhibitors used radioisotopes and have the limitation of altering the molecular function of NGF in an assay. By using biosensor technology sensitive enough to analyze the biomolecular interactions between small molecules (~300 D) and NGF (~26 kD), we can identify further characteristics about these small-molecule inhibitors including identifying whether the molecules are NGF-binding inhibitors or bind elsewhere in the complex.
This article describes the ability to characterize novel small-molecule inhibitors by determining their ability to bind and saturate NGF. By being able to identify the stoichiometry of this relationship, the ability for these small molecules to saturate NGF can be measured. A 2:1 stoichiometry (two small molecules to one NGF dimer) is hypothesized, which confirms previous work with molecular modeling techniques.8,9,11 Small molecules can bind to proteins at a relatively low affinity to nonspecific sites as well as to a high affinity at specific sites, which can be measured using a two-site affinity model. This model groups together the low affinity as one weak binding site that provides a much more accurate KD value for the high-affinity site, which is usually the site of interest. By using the two-site affinity model to describe the binding of these small molecules to NGF, we provide evidence that when binding occurs to one of the available sites on a NGF monomer, a conformational change alters binding affinity for the secondary binding site on the adjacent monomer. However, it is also a possibility that immobilization of NGF to the sensor chip for screening occupies one of the potential binding sites, altering the measured affinity.
SPR affinity assays are demonstrated to be useful in the characterization of compounds that bind and inhibit NGF. Without the use of radiolabels, we have provided further evidence of a theoretical 2:1 stoichiometry and a model by which these small molecules are able to saturate the available NGF in assay. Identifying small molecules with the ability to highly saturate the available NGF, in addition to having a high binding affinity, would be ideal for the use of a therapeutic. These properties allow for smaller dosing yet still yield therapeutic effects in patients displaying symptoms associated with a dysregulation in NGF.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.