Abstract
The current standard of care for treatment of organophosphate (OP) poisoning includes pretreatment with the weak reversible acetylcholinesterase (AChE) inhibitor pyridostigmine bromide. Because this drug is an AChE inhibitor, similar side effects exist as with OP poisoning. In an attempt to provide a therapeutic capable of mitigating AChE inhibition without such side effects, high-throughput screening was performed to identify a compound capable of increasing the catalytic activity of AChE. Herein, two such novel positive allosteric modulators (PAMs) of AChE are presented. These PAMs increase AChE activity threefold, but they fail to upshift the apparent IC50 of a variety of OPs. Further development and optimization of these compounds may lead to pre- and/or postexposure therapeutics with broad-spectrum efficacy against pesticide and nerve agent poisoning. In addition, they could be used to complement the current therapeutic standard of care to increase the activity of uninhibited AChE, potentially increasing the efficacy of current therapeutics in addition to altering the therapeutic window.
Keywords
Introduction
Organophosphate (OP) pesticides and chemical warfare nerve agents irreversibly inhibit the enzyme acetylcholinesterase (AChE, EC 3.1.1.7), which is responsible for hydrolysis of the neurotransmitter acetylcholine. 1 This leads to excessive cholinergic neurotransmission, resulting in paralysis of neuromuscular function and a cholinergic crisis. Widespread use of OPs in agriculture, and as weapons during military conflicts and by terrorists, illustrates this threat to the global population and the need for an effective, broad-spectrum medical countermeasure. 2
Currently, the standard procedure used by the U.S. Armed Forces for reducing the damage inflicted by OP poisoning is pretreatment with the reversible AChE inhibitor pyridostigmine bromide (PB). 3 Following suspected exposure to OP agents, treatment is initiated through the use of the Mark I autoinjector, providing doses of atropine and pralidoxime to compete against the signal caused by excess acetylcholine and to reactivate inhibited AChE. 4 There is currently much interest in developing new therapies to counter OP poisoning, including catalytic bioscavengers using recombinant AChE, 5 butyrylcholinesterase, 5 and noncholinesterases, 6 as well as in modifying oxime-based reactivators. 7
A positive allosteric modulator (PAM), which binds AChE distant from the active site, may offer a substantial improvement to currently available therapeutic options for the treatment of OP toxicity. We have recently developed a rules-based computational model that hypothesizes that developing an allosteric (nonactive site) modulator of AChE can provide sufficient protection to act as an OP countermeasure. 8 In this model, we suggested that by reducing the binding affinities or phosphorylation rates of inhibitors, we could generate broad-spectrum therapeutics. Positive allosteric modulation may also occur through a mechanism that increases the rate of the native substrate reaction, through either a decrease in the Michaelis constant (Km) or an increase in the enzyme maximum velocity (Vmax). Experimental evidence supporting such a hypothesis has shown that mutations or binding at allosteric sites can have dramatic effects on enzyme actions, including altering binding affinities.9,10 Indeed, AChE has at least one allosteric site, the peripheral anionic site, that can exert activating and inhibiting effects. 11
Materials and Methods
Compound Library
The University of Cincinnati Drug Discovery Center is housed at the Metabolic Disease Institute (MDI), formerly known as the Genome Research Institute (GRI). The chemical library contains 207,000 bottles of pure compounds and 340,000 compounds in DMSO solution for rapid dispensing. Compounds in DMSO are stored frozen at 4 °C in an inert, low-humidity, argon atmosphere. The library was purchased from Proctor & Gamble in 2006 with support from Cincinnati Children’s Hospital Medical Center and was constructed to ensure diversity in druggable chemical space, as defined by comparisons to Derwent’s World Drug Index. 12 Chemicals with reactive and unstable features associated with toxicities were removed from the library. As compounds are diminished, they are replaced through purchase from commercial vendors found within the ZINC database. 13 The quality control process measures weight, solubility, and liquid chromatography–mass spectrometry (LC-MS) purity and identity to ensure that all compounds are of high quality.
High-Throughput Screening and Orthogonal Confirmation
High-throughput screening (HTS) for initial modulators of AChE activity was performed using the Amplex Red AChE activity assay (Life Technologies, Carlsbad, CA) as per the manufacturer’s instructions. Briefly, purified electric eel (Electrophorus electricus) AChE was treated with acetylcholine, which it converts to choline, which is then oxidized by choline oxidase, forming betaine and hydrogen peroxide. The hydrogen peroxide is then used by horseradish peroxidase to oxidize resazurin to the fluorescent reporter resorufin. Fluorescence of resorufin was monitored using a kinetic read at 544 nm and 590 nm with the PerkinElmer Plate::Explorer Automated HTS system (PerkinElmer, Waltham, MA).
Consistent with other HTS efforts,14,15 two rounds of screening were performed with an initial selection for activators and inhibitors using a 13 µM single dose on a 5000-compound diversity set from the University of Cincinnati Drug Discovery Center. Following the single high-dose screen and t-test selection (compound AChE activity different from controls; p ≤ 0.05), a second round of screening was performed with those hits using a 10-point dose–response titration from 2 nM to 40 µM. A 200% increase in activity was chosen as a threshold for secondary screened compounds to reduce the number of hits by approximately 40%. A greater activity increase was hypothesized to afford more protection at higher OP exposure concentrations. Orthogonal confirmation, using Ellman’s method, 16 in the presence and absence of paraoxon (PXN) inhibition was used to identify those compounds that were able to rescue AChE from toxicity. Because each compound was dissolved in DMSO, the effect of DMSO on AChE was also investigated. The source of enzyme for orthogonal screening was human erythrocyte (C0663; Sigma-Aldrich, St. Louis, MO), and recombinant human and mouse AChE obtained from Dr. Jonah Cheung (New York Structural Biology Center, New York, NY). 17
Data Analysis
Data calculations and normalizations were performed using Genedata Screener Assay Analyzer software (Genedata, Basel, Switzerland). Raw data were normalized to controls and corrected for systematic errors. Assay performance and robustness were evaluated, and the quality of the data was checked by calculating the coefficient of variation between sample wells, the signal-to-background ratio, the Z’ factor, and the reproducibility of controls.
Liquid Chromatography, Mass Spectrometry, and Nuclear Magnetic Resonance
High-performance liquid chromatography (HPLC) separation of the compounds was performed on a Waters Alliance HT 2795 Separations Module (Waters, Milford, MA) using an Aquasil C18 column (250 mm × 2.1 mm; Thermo Scientific, Waltham, MA). The injection volume of the sample was 50 µl. The sample was a 10 mM solution of N-(1,1,1,3,3,3-hexafluoro-2-((4-hydroxyphenyl)amino)propan-2-yl)benzamide (compound I; Fig. 1 ) in DMSO aged for 17 months at 4 °C. Chromatographic separation of the compounds was achieved with a binary mobile phase under gradient conditions. The mobile phases, A and B, were 0.1% formic acid (Sigma-Aldrich) in water and 0.1% formic acid in acetonitrile (Sigma-Aldrich), respectively. The gradient elution program was as follows: 0–3 min, 2% B; 3–20 min, linear gradient from 2% B to 65% B; 20–25 min, 98% B; and 25–30 min, 2% B. The flow rate was maintained at 1.5 ml/min throughout, and the column temperature was maintained at 35 °C throughout. Flow was split post column using a needle valve, with a small portion directed to a Waters QTOF micro mass spectrometer with a LockSpray dual-electrospray source, and the rest of the flow was directed to a Waters 996 photodiode array (PDA) detector. Mass calibration was maintained throughout the experiment using sodium iodide as the calibrant. Fractions were collected manually by simultaneously monitoring both absorbance and mass.
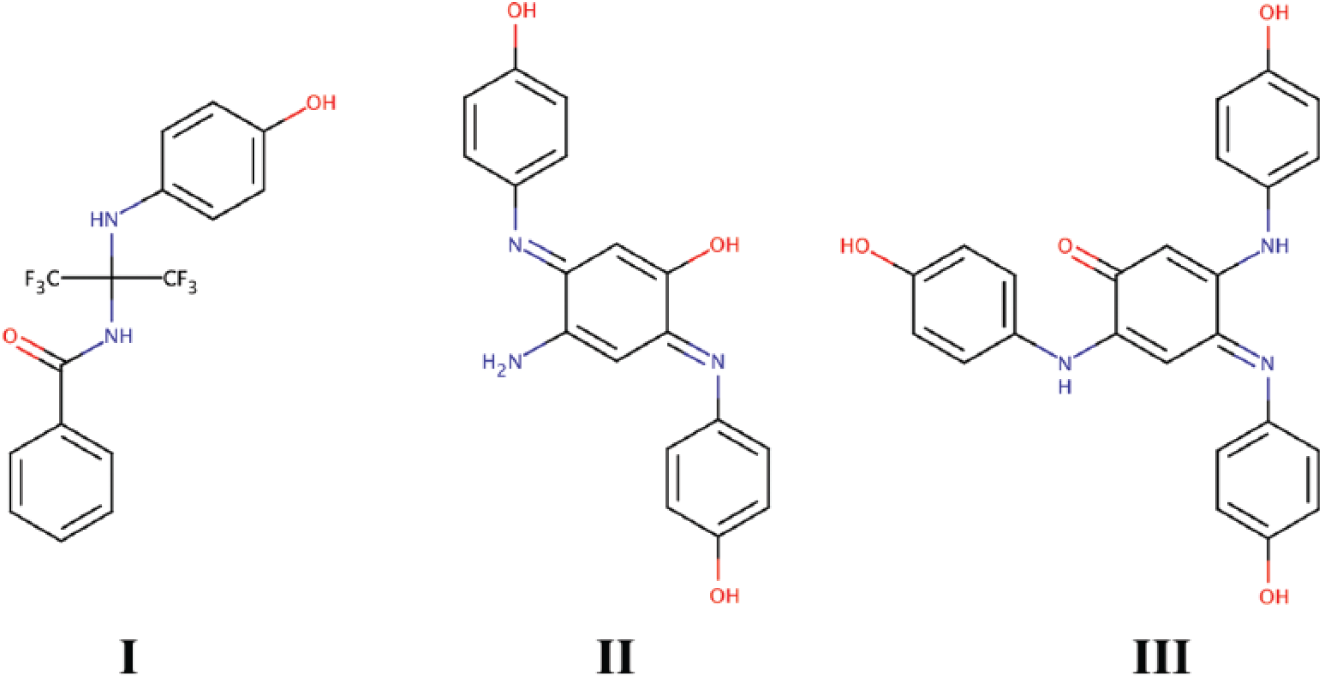
Structures of allosteric acetylcholinesterase (AChE) activators I, II, and III.
The molecular formula for compound II ( Fig. 1 ) was determined using high-resolution MS on a Waters QTOF micro mass spectrometer. The structure was inferred from the molecular formula, the mass fragmentation data, and the structure of compound III ( Fig. 1 ). The structure was confirmed after synthesis from HPLC retention time, MS, and mass fragmentation data. The structure of compound III was determined by nuclear magnetic resonance (NMR) spectroscopy from the proton, carbon, DEPT 90, DEPT 135, COSY, HSQC, and HMBC spectra. All NMR spectra were collected on a Bruker Avance III HD 400 MHz NMR spectrometer (Bruker, Billerica, MA) with two radiofrequency channels, a programmable lock channel, a single axis gradient controller, a waveform memory, a variable temperature controller, and a 5 mm autotune broadband (BBFO) Smart probe with a z-axis gradient. Insufficient quantities of compound II were isolated to allow for NMR structure determination. Synthesis methods for II and III are provided as supplemental information.
AChE Activity Enhancement and Inhibition
Post-HTS AChE activity and inhibition assays were performed according to the Ellman method. 16 Final assay conditions included 40 ng/mL recombinant human or mouse AChE (or AChE from human erythrocytes), 1 mM acetylthiocholine (ATC; Sigma-Aldrich), 0.5 mM 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB; Sigma-Aldrich), and 0.5% DMSO (Sigma-Aldrich) in phosphate buffered saline (PBS; Gibco, Carlsbad, CA). Activity assays were performed in 384-well plates with PAMs and OPs delivered using an Echo 550 liquid handler with the Omics 2 upgrade (Labcyte, Sunnyvale, CA). PAM concentrations ranged from 50 µM to 4 nM. Activation in the presence of inhibitor was performed at each PAM’s EC90, and the inhibitor was titrated from 10 µM to 0.2 nM. AChE and AChE-free control experiments (with and without vehicle control) were performed to confirm that the compounds do not interact with the assay materials. AChE was incubated with PAMs and OPs for 10 min, in the presence of DTNB, prior to reaction initiation by the addition of ATC using the Echo 550. The initiated plate was immediately transferred to a Flexstation III (Molecular Devices, Sunnyvale, CA), and a kinetic read of the absorbance at 412 nm was performed for 10 min, at which point the velocity curve had begun to plateau. Reaction rates were calculated from the linear portion of the curve and imported into Prism 5 (GraphPad Software, San Diego, CA) for three-parameter Hill curve fitting and analysis.
Microscale Thermophoresis
Dissociation constants of the leads were determined using microscale thermophoresis (MST). Recombinant human or mouse AChE was fluorescently labeled using a Pierce DyLight 650 (Life Technologies) according to the manufacturer’s directions. Briefly, 1 mg/mL recombinant human or mouse AChE was incubated at 4 °C with the dye for 1 h in the dark before two rounds of purification by centrifuge. A 16-point serial concentration screen was performed using a Monolith NT.115 (NanoTemper Technologies, Munich, Germany) to measure thermophoresis. PAMs were diluted in MST buffer (NanoTemper Technologies) to a highest concentration of 10 mM with a final DMSO concentration of 1%. This concentration was serially diluted 1:1 in MST buffer with 1% DMSO. Finally, an 8 nM solution of fluorescently labeled AChE was combined 1:1 with the PAM solutions to produce a 16-point dilution series with a final DMSO concentration of 0.5%. Serial dilutions were added to standard glass capillaries via capillary action, thermophoretic time traces were acquired with infrared laser power at 60%, and fluorescence was monitored for 15 s. Data analysis was conducted using the “Thermophoresis with Jump” instrument setting, and the baseline was subtracted and data were normalized before averaging. All binding data were imported into Prism 5 for curve-fitting analysis using the four-parameter Hill curve.
Propidium Displacement
AChE was incubated with II or III and propidium iodide (PI; Sigma-Aldrich) at a concentration 10-fold higher than the dissociation constant ([PI]final = 10 µM) and in excess of the available AChE binding sites. 11 2,2′,2″-[benzene-1,2,3-triyltris(oxy)]tris(N,N,N-triethylethanaminium) triiodide (Gallamine triethiodide) served as a positive control because it has been shown to interact with the PI binding site. 18 The PAM concentrations were titrated from 50 µM to 7.62 nM in threefold serial dilutions and performed at room temperature in PBS with a final DMSO concentration of 0.5%. PAMs and PI were added simultaneously in alternate wells of a 384-well plate (to ease pipetting with a multichannel pipette) in quadruplicate and allowed to incubate with AChE for 10 min before measuring fluorescence intensity (emission monitored at 602 nm and excitation at both 287 and 488 nm, individually) using a FlexStation III. One-way ANOVA comparisons were performed using Prism 5. Two sets of 0.5% DMSO-only controls (eight total samples) were included to ensure reproducibility of the control values.
Results
High-Throughput Screening
This screen produced 142 hits (activators and inhibitors altering AChE activity) that were passed into a second round of HTS with a 10-point dose response. Of the 142 possible hits, 139 were confirmed as having a dose response, of which 39 were positive modulators (
By using a coupled enzymatic reaction system, we were able to rapidly screen through compounds and identify such activators; however, the screening method used provided the appearance of activators making AChE “unsaturable.” This is not a true behavior but, rather, is a consequence of the coupled enzymatic reaction assay, in which activated AChE is consistently turning over substrate and therefore increasing the signal of the second reaction. Because the AChE active site is narrow and contains an even narrower neck in the active-site cleft, we suggest that if a small molecule were to have an activating effect as its concentration was increased to saturation, it must be exerting its effect through an allosteric mechanism. 19
Lead Identification and Confirmation
From the HTS and an orthogonal screen using Ellman’s AChE activity assay in the presence of inhibitor to confirm specific interaction, we identified a single lead compound (compound I;
Fig. 1
) as effective at reducing PXN inhibition. Activator compound I was then evaluated for stability by MS and NMR. Identity-confirming mass spectra revealed a nonnative structure of I, and detailed investigations of the structure resulted in identifying II and III as the active products (
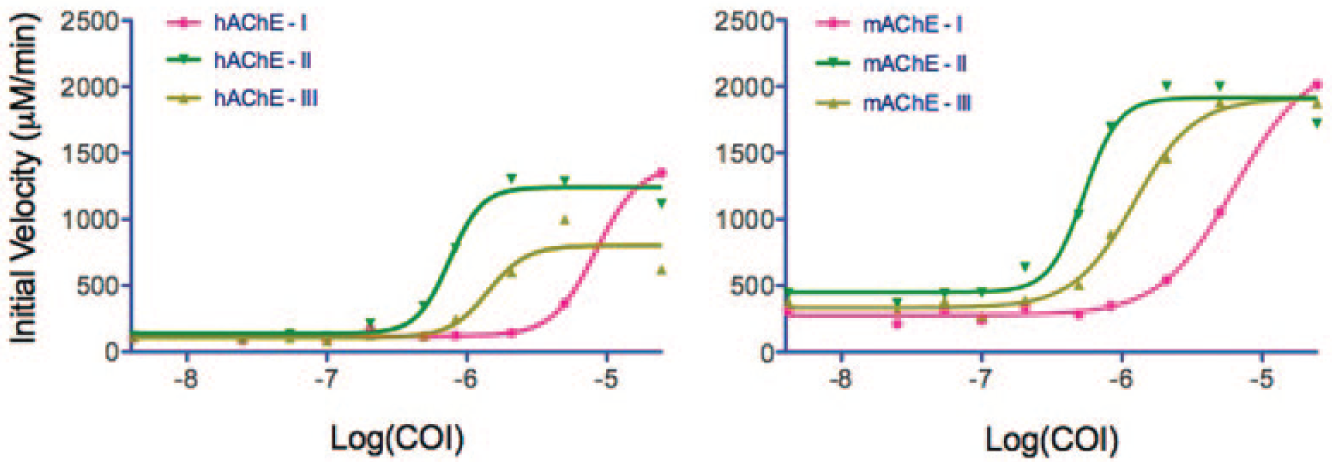
Modulation of human (left) and mouse (right) allosteric acetylcholinesterase (AChE) activity by compounds I, II, and III, as measured using the Ellman assay. Compounds II and III, used here, were the synthesized oxidation products of compound I, which was used after fresh dilution from DMSO into aqueous solution (prior to oxidation). Curves are fit to the data using three-parameter Hill equations. COI, compound of interest; concentration is log molar.
AChE Allosteric Binding
Enzyme–modulator binding interactions were characterized using MST for the pure compounds. As was expected from the initial activation assays, the dissociation constants for all compound–enzyme pairs were in the low micromolar range. As an example, the Kd values for the pairs II-hAChE and II-mAChE (human and mouse AChE, respectively) were shown to be 5 µM and 4 µM, respectively ( Fig. 3 ). The other compound–enzyme pairs exhibited similarly moderate binding affinities.
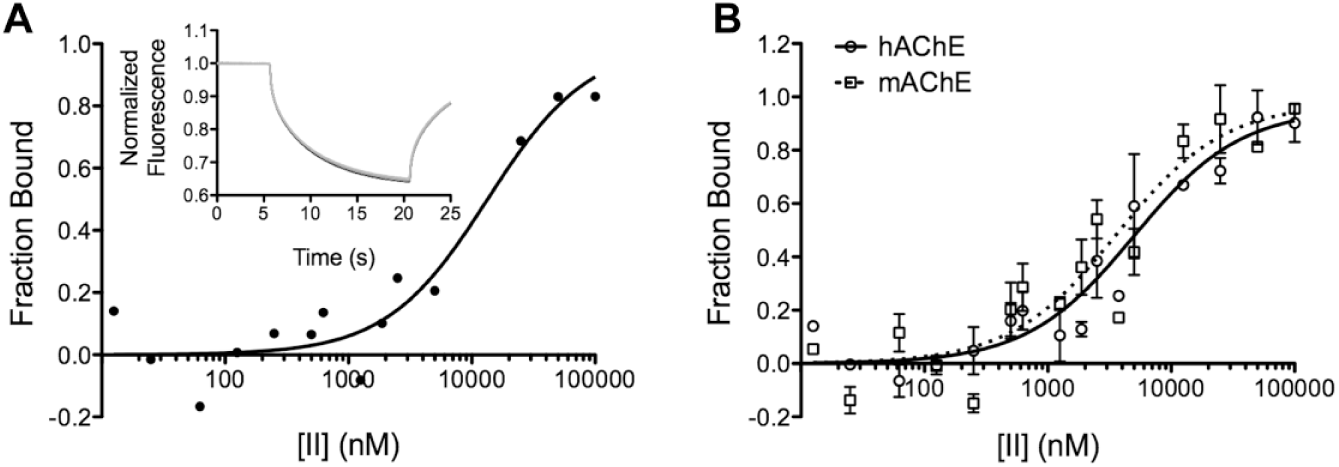
Microscale thermophoretic analysis of compound II binding to allosteric acetylcholinesterases (AChEs). (
The AChE peripheral anionic site (PAS) is a site in close proximity (>20 Ǻ) to the active site that has been shown previously to bind both activators and inhibitors. 20 Using a propidium displacement assay 11 to ascertain whether the PAMs were binding at the PAS, it was found that these compounds act through a potentially novel allosteric site. For the PAS to be the binding site for the PAM, the PI inflection points of the displacement curve ( Fig. 4 ) and the activity curve ( Fig. 2 ) should be the same. In our results, however, we do not find evidence of an inflection point in the PI displacement data ( Fig. 4 ), and in fact we observe significant differences from the control experiments only for the 50 µM samples of both II and III (P ≤ 0.05).
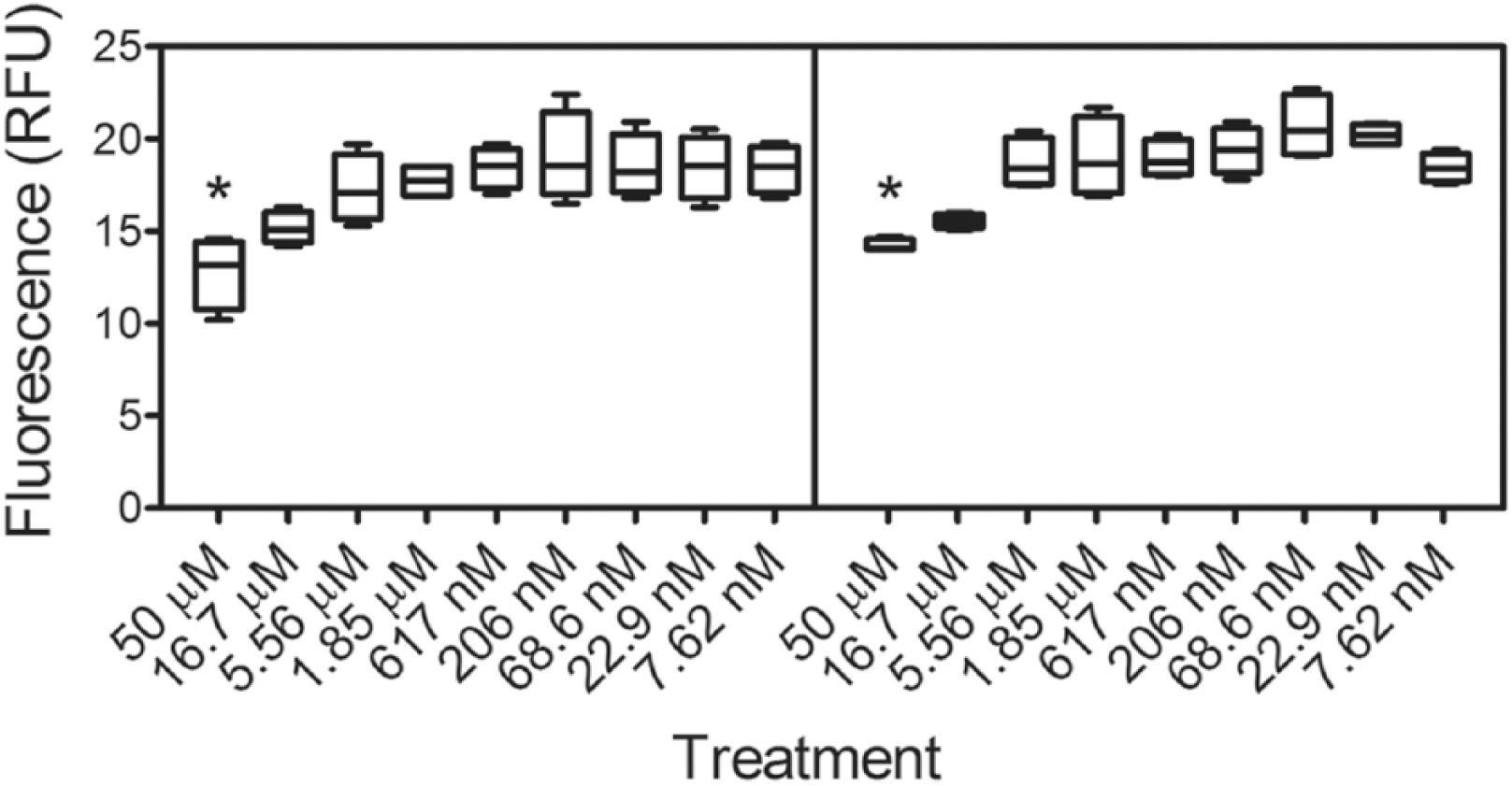
Box-and-whisker plot showing the propidium displacement as a function of compound II (left) and III (right) concentrations. Four replicates are represented (* = P < 0.01). A shift in the excitation wavelength follows the complexation of propidium and allosteric acetylcholinesterase (AChE). Displacement of propidium results in a reduction of fluorescence response units (RFUs) measured at an emission wavelength of 595 nm on excitation at 535 nm. Gallamine triethiodide was used as a positive control.
AChE Activation and Inhibition Protection
To test our hypothesis that PAMs could provide protection against OP intoxication, we evaluated the inhibition characteristics of three OP pesticides with the following activators: PXN, diisopropyl fluorophosphate (DFP), and dicrotophos (DCO), as listed in Table 1 . The activator concentration was held constant at 5 µM, approximately equal to the 95% effective concentration (EC95) for all modulator–enzyme pairs. The ability of therapeutics to “shift” apparent IC50 values has been used to evaluate the efficacy of oxime reactivators. 21 As expected from the higher activation efficiency, II was observed to have the strongest apparent IC50 shift potential of the inhibitors tested ( Table 1 ), albeit only a small shift. It is important to note that these apparent IC50 values are reported as absolute concentrations. The smaller size of II in comparison to III may allow for greater flexibility in binding to the enzyme variants, which could explain its effectiveness in both enzymes. mAChE was seen to be the more amenable to allosteric protection by III, demonstrating the divergent behavior of allostery and emphasizing how critical it is to test for in vitro efficacy using the enzyme variant from the intended animal model in addition to the human enzyme.
Apparent IC50 Values of OP Pesticides by Allosteric AChE Activators.
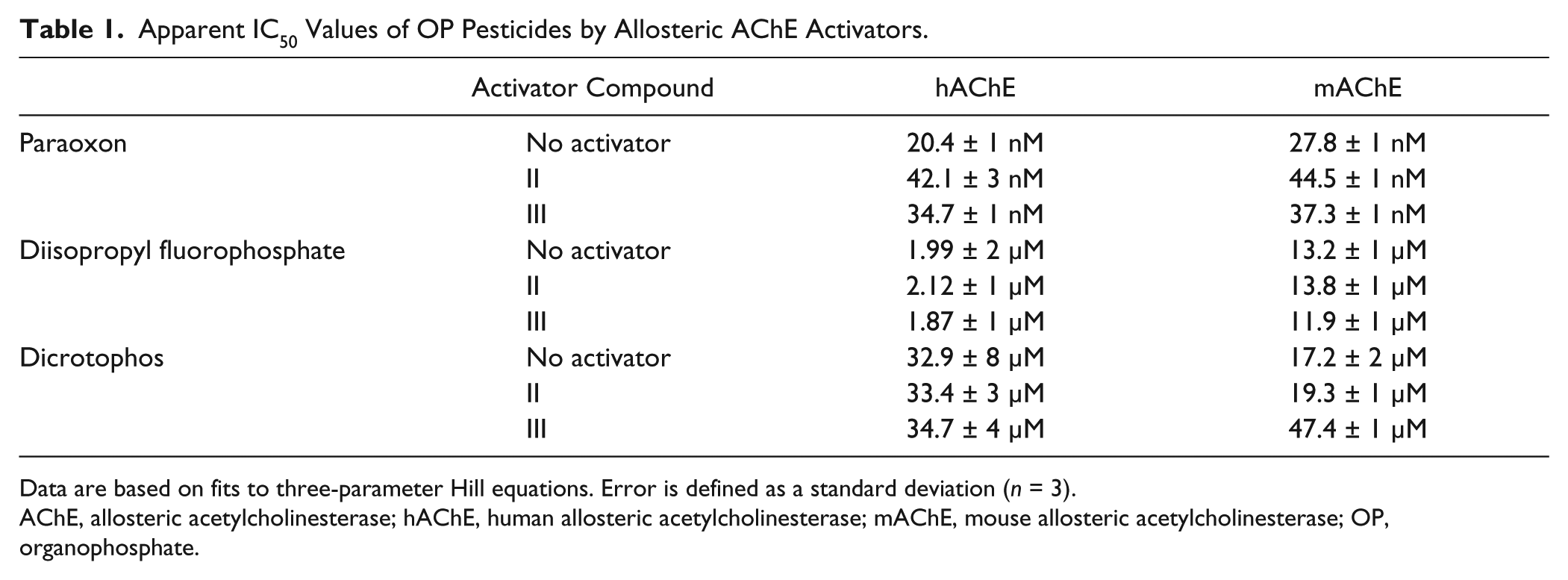
Data are based on fits to three-parameter Hill equations. Error is defined as a standard deviation (n = 3).
AChE, allosteric acetylcholinesterase; hAChE, human allosteric acetylcholinesterase; mAChE, mouse allosteric acetylcholinesterase; OP, organophosphate.
Discussion
Allosterically acting therapeutic candidates are well established in recent literature, including targets for pain regulation, 22 inflammation, 23 and Alzheimer’s disease. 24 Examples of both positive and negative allosteric modulators are currently in clinical trials.25,26 One advantage of using allostery as a mode of action lies in the ability of the therapeutic to not interfere competitively with the native enzyme function, in contrast with the current modes of action for treatments that serve as reversible AChE inhibitors (for pretreatment) or active-site oxime reactivators. In addition, using a PAM as a chemical countermeasure against OP intoxication could potentially reduce the onset of poisoning symptoms, increase the chances of patient survival, and assist in the efficacy of oxime and nonoxime AChE reactivators. Finally, because PAMs do not interact directly with the active site but instead alter the shape or dynamics of that site, using allosteric therapeutics may provide broad-spectrum efficacy against OP inhibitors without regard to inhibitor structure.
Oxidation of compound I, forming compound II, is believed to have occurred because the chemical library is exposed to numerous freeze–thaw cycles and humidified air. Compound III is believed to have formed from a similar three-step synthesis scheme, outlined in the Supplemental Information, during either the synthesis or degradation of compound I. It is well known that freeze–thaw cycles and room-temperature storage alter the stability of compounds in DMSO, leading to degradation of products.27,28
Calculations of the specificity constant (kcat/KM) show that hAChE and mAChE are highly efficient (1.32×108 and 1.20×108 M−1 s−1, respectively); however, a kinetically perfect enzyme specificity constant is on the order of 108 to 1010 M−1 s−1. 29 These calculations suggest that it may be possible to increase AChE’s rate from 10- to 100-fold higher than its native rate. 29 One possible explanation for its high activity, for which evidence exists, is that ACh is electrostatically steered into the active site. 30 This suggests that the apparent rate of diffusion may be increased by an electrostatic attraction force, resulting in the enzyme no longer being limited by simple diffusion: the AChE reaction rate for its charged substrate can also depend on ionic strength and substrate concentration. 31 As with all other proteins, AChE is metastable and flexible, opening the possibility that AChE can displace substrate via “breathing” motions, 32 which may lead to further enhanced enzyme activity if the PAMs discovered here alter the breathing mechanism. It may also be possible for the PAMs to interact with the putative “back door,” 33 altering the rate of substrate entrance or product clearance in the active-site gorge. Finally, an eightfold selective enhancement of the activity (likely associated with an interaction of the enhancer with the PAS) was found in a C-terminally truncated version of AChE, 34 which is consistent with our (and others’) 29 hypothesis that a 10–100-fold enhancement of activity is possible for AChE.
In conclusion, by using an HTS campaign targeted to find positive allosteric modulators of acetylcholinesterase, we have identified two potential therapeutic leads with the ability to reduce the impact of OP-mediated AChE inhibition and potentially prevent activation of the parasympathetic nervous system. These compounds share a common core feature generated from N-polymerization of the p-aminophenol, which we hypothesize is a feature that either contains or forms the pharmacophore, and investigations are underway to confirm or rebut this hypothesis. Structure–activity relationship (SAR) studies are currently being performed to optimize binding and increase the efficacy of protection. The ability of these compounds to protect against nerve agents in vivo is also being actively pursued, and preliminary cell culture experiments suggest limited toxicity. We expect that through the SAR and in vivo studies, a valuable new treatment option will be developed, potentially saving the lives of warfighters, agricultural pesticide workers, and the civilian population from mass-casualty releases or self-administered exposures.
Footnotes
Acknowledgements
The authors would like to thank Dr. David Riddle for critical comment, and Drs. William Seibel and Matthew Wortman at the University of Cincinnati for assistance with HTS.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this work was provided by the Defense Threat Reduction Agency (CBM.Neuro.THR3) to JMG and through a Cooperative Research Agreement between the Henry M. Jackson Foundation for the Advancement of Military Medicine and the US Air Force. The opinions expressed in this article are those of the authors and do not necessarily reflect the opinions of the US Air Force, the US Department of Defense, or the US government.
Supplemental Materials
HTS results; oxidation product characterization mass and NMR spectra; synthesis procedures.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
