Abstract
More therapeutic monoclonal antibodies and antibody-based modalities are in development today than ever before, and a faster and more accurate drug discovery process will ensure that the number of candidates coming to the biopharmaceutical pipeline will increase in the future. The process of drug product development and, specifically, formulation development is a critical bottleneck on the way from candidate selection to fully commercialized medicines. This article reviews the latest advances in methods of formulation screening, which allow not only the high-throughput selection of the most suitable formulation but also the prediction of stability properties under manufacturing and long-term storage conditions. We describe how the combination of automation technologies and high-throughput assays creates the opportunity to streamline the formulation development process starting from early preformulation screening through to commercial formulation development. The application of quality by design (QbD) concepts and modern statistical tools are also shown here to be very effective in accelerated formulation development of both typical antibodies and complex modalities derived from them.
Keywords
Introduction
Biotherapeutic products based on monoclonal antibodies (mAbs) are the fastest growing category of therapeutics entering clinical study.1,2 Since the first introduction of technology to produce monoclonal antibodies in the 1970s and early 1980s, the number of antibody-based modalities on the market and in development has reached several hundred. Because of their high affinity to target molecules and low immunogenicity, they have dramatically changed the practice of medicine, improving and saving the lives of countless patients. At the same time that applications of antibodies in medical therapy and biotechnology increased, it became clear that mAbs have some disadvantages associated with the relatively large size and complexity of the protein. These disadvantages may include differences in tissue penetration due to their large size, complicated cloning procedures for two chains of an IgG, and complex glycosylation patterns often resulting in low titer, requiring significant resources for optimization of production. The constant Fc domain is necessary for immunological effector functions and prolongs the half-life, which in some cases can be undesired for specific applications. The antigen binding site is constructed of six hypervariable loops, and this can be difficult to engineer. In part, due to these features, there is growing demand for full-length antibody alternatives with improved structural and functional features. In Figure 1 , several different mAb-based modalities are shown. The combination of existing mAb domains and the addition of other protein constructs create many possibilities for engineering to improve stability, to alter half-life time, and to direct to a specific target. Multispecificity can be added to functionality and numerous varieties of constructs that could be made based on antibody domains.3,4 To successfully use antibody-based biologic therapeutics in medicinal practice, it is necessary to maintain conformational, chemical, and colloidal integrity during all steps of the development, manufacturing, and commercialization processes. 5 It is also critical for monoclonal antibodies to have antigen binding and biological activity through the formulation development. Biotherapeutic formulations provide the storage solution and delivery carrier for the drug through the development process. Drug candidates should be formulated for preclinical and clinical studies. Early screening for the most stable formulation can save a lot of resources during the late stages when the cost of instability could be extremely high. In biopharmaceutical companies, formulation development is often focused on long-term stability studies during preclinical and clinical phases. 6 In those studies, the drug product formulated in vials or syringes is exposed to different stress conditions and stored at several temperatures for long periods of time, often up to 2 years. The activities of early preformulation development, if any, usually are performed on a small scale and not on a systematic basis. In many cases, the analytical and stress methods used in early and late formulation studies are not comparable. Often the information collected on formulations and molecules is not shared and is hard to access, even within a single company, since it is obtained by different departments. The lack of comparability and accessibility makes it difficult to make science-based decisions on formulations most suitable for specific drug candidates. A streamlined process with characterization and stress methods appropriate for each development step and with easily accessible information flow between early and late-stage teams will significantly accelerate the formulation development process. Systematically collected data during the entire formulation screening/selection process from early preformulation to commercial development will establish the basis for correlations between structure and stability properties. It is possible not only to simply correlate structural data with formulation stability but also to predict such stability during manufacturing, storage, and delivery. In this review, we describe the means to make formulation development faster and more efficient by combining modern advancements in automation, miniaturization, and computerization technologies into one seamless process of accelerated formulation development.
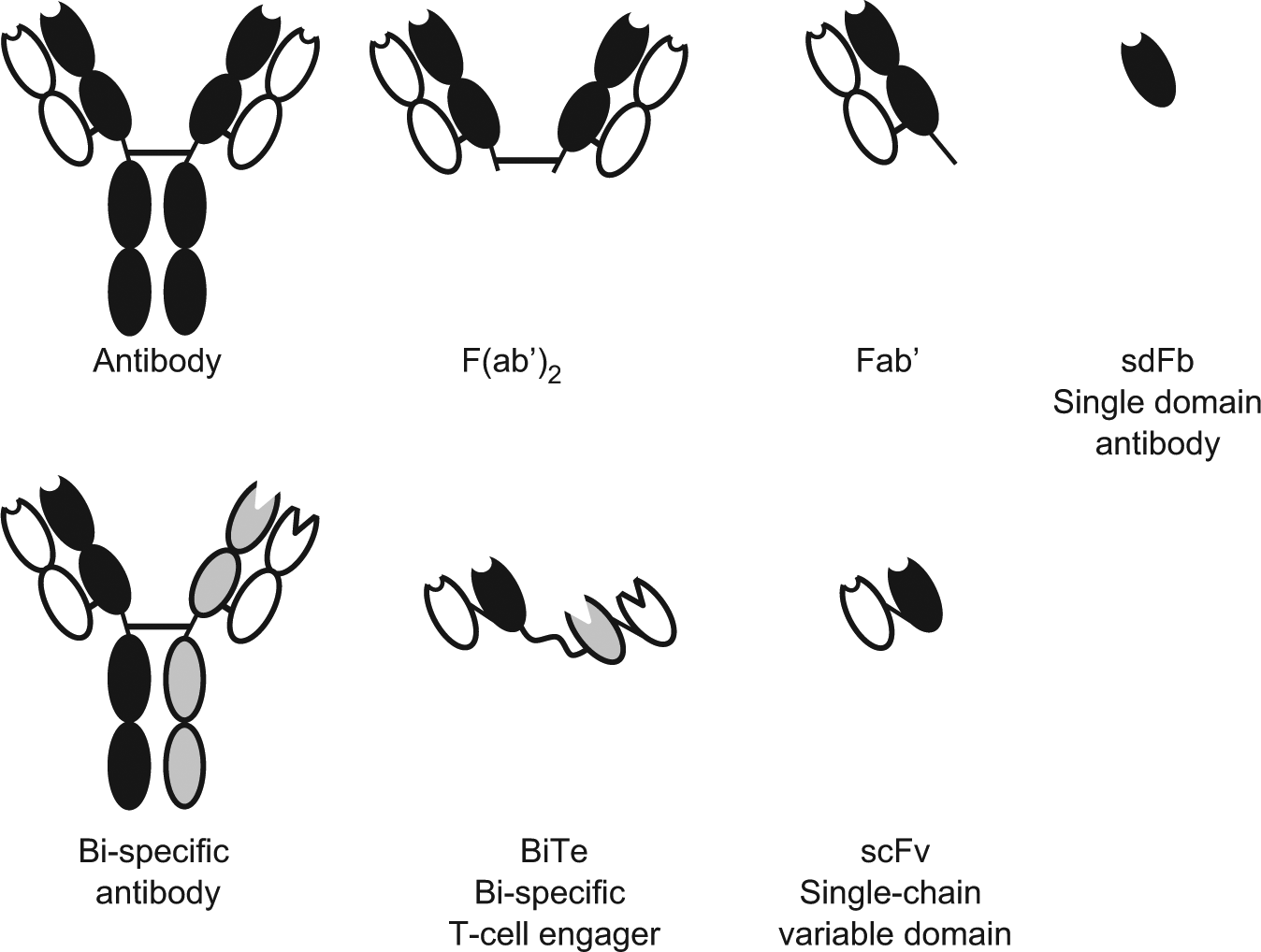
mAb-Based modalities.
High-Throughput Methods for Formulation Screening
Formulation development of monoclonal antibodies and their modalities incorporates many standard analytical biochemical and biophysical methods traditionally applied for protein characterization. Originally, many of these methods were developed in a relatively low-throughput format to be used on samples containing high amounts of protein, but effective screening of multiple formulation conditions requires methods that can generate information fast using low amounts of protein available without significantly sacrificing accuracy, precision, and specificity. Monoclonal antibodies have very complex structures, both chemically and physically, to maintain their functionality and stability during their lifetime. 7 The native structures of mAbs and their domains have to adapt to changes in factors such as ionic strength, pH, and others. While mAb flexibility is necessary for biological function, it presents many challenges in formulation development. Under various conditions of a development process, the conformation, protein-solution interactions, and even chemical structure of a mAb can change, creating new variants with possible adverse effects on efficacy and stability. A full understanding of all possible degradation pathways under different stresses is still not complete, but many years of research by both academic and biopharmaceutical scientists have established the general scientific foundation for understanding protein properties in different environmental conditions.8,9 In formulation development, it is critical to have information on multidimensional properties of protein and their dependence on both formulation and stress factors. Different conditions can affect different aspects of protein stability. 10 Generally, three types of stability can be distinguished. Conformational stability describes the ability of protein to maintain its native structure, to be properly folded. Colloidal stability is the stability of protein with native structure in solution to avoid aggregation, precipitation, or phase separations. Chemical stability is the stability of amino acids and the covalent bonds between them and between different protein domains. These are arbitrary definitions, but they have some practical sense since there are distinct high-throughput methods to characterize each type of stability. Also conformational, colloidal, and chemical stabilities are not necessarily independent of each other. For example, protein unfolding can expose hydrophobic regions of a protein, leading to self-association often followed by total precipitation of the protein. Chemical modifications can create hydrophobic areas with the described above consequences. But a thorough knowledge of each stability contribution to overall stability properties can help significantly in the selection of the best formulation.
Screening for Conformational Stability
Conformational stability of monoclonal antibodies is high relative to other proteins. Normally, it is hard to achieve complete unfolding of an antibody molecule even at temperatures up to 100 °C. The technique of choice for measuring unfolding temperatures and thermodynamic parameters during protein unfolding is differential scanning calorimetry (DSC). A typical DSC profile for an IgG antibody molecule contains three peaks identified as Fab, CH2, and CH3 domains. 11 The melting temperature (Tm) of each domain is not a constant but can be shifted depending on the formulation factors. Under typical formulation conditions, the most stable domain is usually the CH3 domain, and the least stable is the CH2 domain. The pH of the solution and the presence of salt significantly decrease the Tm. 12 Some excipients can also change the Tm. The DSC method requires approximately 0.5 to 1.0 mL of sample at a protein concentration of 0.5 mg/mL for sufficient sensitivity, and the entire scan requires greater than 1 h to complete. Even though there are plate-based and automated versions of DSC instruments, the assay is still very demanding in terms of protein amount and time. Several high-throughput methods have been developed to screen for conformational stability of proteins in general and monoclonal antibodies specifically. 13 One method is based on the measurement of extrinsic probe fluorescence during a gradual temperature increase. During the process of protein unfolding, hydrophobic regions, normally buried in the interior of the protein, become exposed to the solution, and fluorescence of a probe sensitive to hydrophobicity increases significantly. Originally, the method was developed for screening of binding of small molecules to proteins. The melting temperature shifts after compound binding, and the magnitude of the shift depends on the affinity of the small molecule for the protein, 14 and this was called a thermal shift assay or differential scanning fluorimetry (DSF). The first application of this technique in thermal stability testing was the screening of formulations of monoclonal antibodies to identify a formulation that provided optimal conformational stability. 15 The dye, SYPRO Orange (Molecular Probes, Eugene, OR), is commonly used for this application, and mAb formulations have a low fluorescent background when the antibodies are properly folded. However, unfolding of the antibody leads to a fluorescent signal that can be hundreds of times greater than the background. DSF data from such an experiment correlate well with DSC results for monoclonal antibodies. Both the onset of fluorescence and the temperature of hydrophobic exposure (Th) can serve as characteristics of thermal stability. The Th value is determined by the minimal value of the first derivative of fluorescence dependence on temperature. The domain (usually the CH2 domain for mAbs) with the lowest Th is well resolved on the scan. Other domains, however, are less visible because of a quenching effect at higher temperatures in the presence of unfolded protein. DSF can be run on a standard reverse transcriptase (RT)–PCR instrument with a standard thermostat and fluorescence measurement capability. The protein concentration range used in the method varies from 0.05 to 200 mg/mL. The whole scan of a 96-well plate takes 1 to 2 h depending on the scan rates used. The optimal temperature increase is around 0.1 to 0.5 °C per minute. One drawback to the method is that surfactants, widely used components in mAb formulations to prevent surface or interface interactions, result in a high background signal when probes such as SYPRO Orange are used. These probes are thought to interact with micelles formed by the surfactants, interfering with the signal due to protein unfolding. To avoid interference with surfactants, new probes have been introduced. 16 4-(Dicyanovinyl) julolidine (DCVJ) has been successfully used in the screening of antibody formulations containing several surfactants. The DCVJ-based DSF method was able to determine the Th values in formulations where SYPRO Orange failed. Control formulations without protein should be used in accurate determinations of unfolding temperature and fluorescence onset, in cases when intensity increases above the background. For some antibody-based modalities, it is still difficult to apply DSF because of the high background of fluorescence at low temperatures. Highly hydrophobic regions can be exposed in the constructs, which are different from the typical IgG-like molecules. The increase of fluorescence in the presence of unfolded protein has been used for the direct measurement of mAb aggregation in the study by He et al. 17 In this study, the aggregation caused by high temperature has been detected at a level as low as 3% and was confirmed by size exclusion chromatography. Turbidity measurement is an older, established method for the measurement of protein aggregation. Traditionally, the method has involved using a cuvette and a standard spectrophotometer taking measurements at a visible wavelength (350–400 nm), but the method is easy to apply in a multiwell plate format. 18 Unfortunately, sensitivity of the turbidity method is low for smaller aggregates such as dimers, tetramers, and so on, and it is impossible to observe a size distribution. Unfolding and aggregation of antibodies are closely linked, and this phenomenon can be used in the evaluation of conformational stability by aggregation monitoring with the help of a light-scattering technique. The gradual heating of the sample and simultaneous measurement of the light-scattering signal has been employed in the OPTIM 2 instrument manufactured by Avacta Analytical (Wetherby, UK) to screen small-volume samples simultaneously for both unfolding temperature and aggregation temperature. In the work by Goldberg et al., 19 a static light-scattering method was applied using a static light plate reader, the StarGazer-384 (Harbinger Biotechnology and Engineering Corporation, Markham, Ontario, Canada). A small volume (25 µL) was added to a 384-well plate and heated. Protein aggregation was monitored by measuring the intensity of the scattered light. The results were used for evaluation of both conformational and colloidal stabilities. Nuclear magnetic resonance (NMR) is the technique often considered to be sophisticated, laborious, and time-consuming. But recent advances and improvements in automation, sensitivity, resolution, and spectral processing make NMR a tool for fast and especially very accurate characterization of biopharmaceutical products. 20 Fourier transform infrared spectroscopy (FT-IR) and microscopy has been applied for aggregate characterization by imaging the area containing protein samples incubated at high temperature. FT-IR spectra of multiwell plate with protein samples was obtained during stability assay. A field of view 128 by 128 pixels and 7.0 by 9.8 mm was used to probe 12 sample wells simultaneously in this study. The image can be taken in time intervals, and aggregation kinetics is easy to follow and quantify. The pixels contained in the well area of color-coded polygons were averaged to obtain the one spectrum per well region for every image collected at a time interval. 21 Raman microscopy spectroscopy has also been used in a high-throughput format to determine size and chemical characteristics of large particles. 22 Instability under stress conditions can lead to structural changes, which otherwise require significant energy. The stress can be caused by elevated temperature, chemical agents, and surface or interphase interactions. On the other hand, monoclonal antibodies are very stable structurally under typical storage conditions at 4 to 8 °C for long periods of time. The major degradation pathway for these molecules under these conditions is aggregation or formation of large particles. 23 Although aggregation of monoclonal antibodies after long-term storage at low temperature can sometimes be caused by unfolding, 24 the major cause is colloidal instability. Generally, the structure of engineered mAb-based modalities is less stable than the original antibody molecule. For example, an antibody-drug conjugate (ADC) includes a native mAbs with a small-molecule drug (sometimes referred to as the payload) attached through a linker. The position of the linker connection with the antibody is important for stability.25,26 Covalent linkage to solvent-accessible cysteine residues may significantly disrupt the integrity of molecule conformation and change overall thermodynamics of unfolding. 27 The hydrophobicity of the payload and the inherent heterogeneity of the number of payloads per antibody molecule are additional factors for instability. 28 Another type of modality, the bispecific antibody, 29 is a construct consisting of domains from different molecules. This arrangement and any other combinations of various antibody domains usually have lower conformational stability than the original mAbs.
Screening for Colloidal Stability
Monoclonal antibodies are very complex proteins with multidomain structures. The most common IgG type contains 12 domains that can be divided into heavy and light chains, variable and constant domains. There are a total of four isotypes and two light chain classes expressed in humans. But those differences in isotype structures cannot explain the high variability in IgG stabilities. 30 The mechanism of colloidal stability in biotherapeutic formulations is still poorly understood,31,32 despite an abundance of fundamental knowledge of physical processes in protein solutions. Only recently, systematic approaches have been applied to study the colloidal stability of mAbs in formulations. The approaches combine computational and experimental methods to understand protein-protein and protein-solution interactions, including mAb interactions with different formulation components. The most common computational method is calculation of the theoretical pI, which can serve as a reference value to estimate the stable pH range of potential formulations. At a pH close to the pI, the protein tends to precipitate from solution, 33 and to avoid this, it is necessary to adjust the ionic strength by addition of salt or other excipients.34,35 For most mAb molecules, the pI range is between 6.0 and 9.0, but it is possible that various new mAb modalities may fall outside of this range.
Colloidal stability can be measured in several ways. Most of the approaches are based on the evaluation of a protein’s propensity for self-association, leading to subsequent aggregation or phase separation. The second virial coefficient B22 (B2 and A22 can also be used) is the parameter used in the estimation of interaction between protein molecules. 36 Basically, the sign of B22 is an indication of the direction of intermolecular forces. A negative second virial coefficient is related to dominant repulsion between proteins, and a positive B22 value shows protein-protein attraction. An ideal protein solution results in no interaction with B22 = 0. The second virial coefficient can be measured by multiple methods, including self-interaction chromatography, static light scattering, and analytical ultracentrifugation.37–39 In a study by Saito et al., 40 colloidal stabilities for four different monoclonal antibodies were evaluated based on the second virial coefficient and the net charge in formulations containing different concentrations of NaCl and sugars. Colloidal stability was partly correlated with net charge, and the presence of salt changed aggregation propensity according to the second virial coefficient. The problem for formulation development is that the B22 methods take significant time and expertise to obtain compatible and reproducible results. Throughput is relatively low, and sample preparation can be tedious. An alternative to B22 is the interaction parameter kD that can be measured by a dynamic light-scattering technique. 41 kD is derived from the dependence of the protein diffusion coefficient on protein concentration, which can be easily determined using a dynamic light-scattering (DLS) plate reader in a high-throughput format. 42 Usually, four to five concentrations in the range of 2 to 15 mg/mL are required to reliably determine the interaction parameter for one sample. For a single 96-well plate, the analysis of 24 formulations is possible, taking about 1 to 2 h depending on the acquisition time and formulation conditions. Stable antibody formulations typically have positive kD values. The propensity for aggregation is increased after addition of salt, and the kD can become negative, indicating repulsion of the protein molecules. In one study, agitation-induced particle formation of an IgG1 monoclonal antibody was greatly increased at kD values less than 7 mL/g in the presence of 60 mM NaCl. 43 Methods for the measurements of second virial coefficients and interaction parameters normally use low concentrations of proteins. Antibody formulations require relatively high protein concentrations. At high mAb concentrations, the crowding effect, including short-range intermolecular forces, can give a greater contribution than at low concentrations, complicating the prediction of colloidal stability by this method. Another parameter, G22, is dependent on protein concentration and can serve as a better predictor of protein-protein interactions. 44 G22 like B22 can also be determined by static or dynamic light scattering, and it is possible to apply a high-throughput format with the help of a plate reader.
In addition to formation of soluble aggregates and large particles, there is another manifestation of colloidal instability—phase separation. Massive precipitation and formation of insoluble large particles is one form of phase separation from the soluble protein fraction. Recently, a predictive method for aggregation propensity under long-term storage at low temperature has been developed and applied to the study of monoclonal antibody formulations. 45 In this method, various antibodies were precipitated by ammonium sulfate in the presence of different excipients. Ammonium sulfate was added from a 4-M stock solution to a final concentration ranging from 1 to 1.72 M. The final protein concentration of each formulation was 5 mg/mL. A good linear correlation was observed between mAb solubility and aggregation level, as measured by size exclusion chromatography (SEC), after long-term storage. An even better correlation was obtained between the apparent transfer-free energy (ΔG) of the native state from water into each of the excipients in the mAb formulations. Based on a 3D model structure of an antibody molecule, ΔG values for those amino acids exposed to the solution were used in the calculations of the apparent transfer-free energies. This good correlation suggests that the estimation of thermodynamic parameters of protein-solvent interactions may be a good analytical way to predict colloidal stability. Polyethylene glycol (PEG) is another precipitating agent to evaluate protein solubility. Buffer and pH formulation conditions have been ranked by a PEG-induced high-throughput precipitation method in the screening of various IgG1 monoclonal antibodies. 46 PEG can also be used not only to precipitate antibodies but also to induce liquid-liquid phase separation. Similar to ammonium sulfate precipitation, the thermodynamic parameters of the condensed phase obtained after PEG-induced separation can be used to evaluate colloidal stability. 47 Unfortunately, even with all the assays currently available or in development to predict colloidal stability of proteins in formulations, there is no single method that is accurate enough to rely on. A combination of computational and experimental techniques will likely be used address this vexing problem, leading to the development of optimal protein formulations.
There are many methods for direct detection and measurement of protein aggregation. Antibody formulations stored at low temperatures for a long period of time can form soluble dimers, subvisible particles, and large visible particles. The process of native aggregation can be very fast and caused by significant and sudden changes in protein concentration, pH, or ionic strength. It can be slow, taking many months, and worst, it can also be unpredictable, induced by local fluctuations in surface properties. Formulation screening almost always includes sample analysis by SEC to measure the amount of soluble aggregates. The SEC method can detect mAb aggregates of sizes from dimers up to heptamers, with larger aggregates being filtered by the column. Modern SEC methods are based on ultra-performance liquid chromatography (UPLC) instruments, and the runtime for each sample can be as short as 2 to 6 min. With an autosampler capable of handling multiwell plates, the analysis of one plate containing 96 formulations can take about 4 to 5 h, which is 10 times faster than traditional low-throughput assays. A typical sample volume per well in a 96-well plate is about 50 to 200 µL. Protein interaction with the SEC column can result in aggregation or aggregate dissociation. 48 An orthogonal method such as analytical ultracentrifugation is necessary to control reversibility of the aggregates. 49 The detection of subvisible particles with the sizes in the range of 100 to 1000 nm requires methods other than SEC. Methods based on static and dynamic light-scattering techniques are very well suited for subvisible particle measurements in antibody formulations. 50 A DLS instrument, the DynaPro Plate Reader (Wyatt Technology, Santa Barbara, CA), can perform measurements on 96-, 384-, or even 1536-well plates. Unfortunately, the DLS assay is extremely sensitive to the presence of large aggregates, and the signal from even a very small number of subvisible particles can easily overwhelm the scattering signal from the monomer species. Furthermore, the resolution of DLS is quite low, and it is difficult to resolve the mAb monomer peak from its dimer peak Also, the quantitative measurements are very dependent on the model of instrument used for the measurements, and an accurate determination of the amount of particles is impossible in the case of multiple peaks and wide peak distributions. However, the throughput of the method is high, and it is convenient for fast selection of formulation samples with both high and low amounts of large aggregates.
Liquid-liquid phase separation (LLPS), 51 as one form of colloidal instability induced by PEG, has been used to evaluate antibody solutions. 52 LLPS in monoclonal antibody solutions occurs only under certain combinations of temperature, concentration, and specific properties of protein molecules. Attractive forces between antibody molecules are too weak to result in LLPS at temperatures above the freezing point of the solution. But the phase separation can be induced by the presence of PEG. The screening can be done in a high-throughput setting on a multiwell plate. Particles larger than 2 to 5 µm can be detected by methods based on microscopy such as micro-flow imaging (MFI) 53 or light obscuration methods.54,55 Both methods require a relatively large volume of sample and take significant time to acquire and analyze the data. Recently, automated versions of both methods were introduced for commercial use, but careful calibration is necessary to achieve the desired accuracy and consistency. For example, particle counts made by MFI are usually higher than counts obtained on the same sample by the light obscuration technique. 56 Another fast method to characterize large protein particles is based on flow cytometry instruments, which can count unlabeled particles. 57 Various formulations containing subvisible antibody particles with sizes ranging from 500 nm and upward were analyzed by this method, and the results were compared with MFI and light obscuration assays and demonstrated a good correlation between cytometry-based methods and more standard techniques. Although not truly a high-throughput technique, this method can be automated, saving analysis time.
As mentioned before, other mAb-based modalities might have stability properties completely different form the properties of the original monoclonal antibodies. Any changes in hydrophobicity or charge introduced by conjugates in ADCs change the protein-solvent interaction and therefore the colloidal stability. Some constructs of peptides and an Fc domain can carry the hydrophobic properties of the peptides 58 and hardly be soluble in formulations designed for antibodies. 59 The colloidal properties of Fc-fusion proteins 60 can also be determined by the fused protein rather than the Fc domain. 61 Peptide linkers are often hydrophobic and can induce the aggregation of ADCs. Doxorubicin conjugate with Phe-Lys or Val-Cit linkers was found to form noncovalent dimers. 62 At the same time, the addition of a hydrophilic methoxytriethylene glycol chain onto doxorubicin inhibits its aggregation. 63
Screening for Chemical Stability
Chemical modifications of monoclonal antibodies can be predicted in many cases by analysis of the primary sequence. 64 So-called hot spots of chemically unstable regions can be identified during early stages of development and, if possible, they can be engineered out. Higher order structural information is also critical and very helpful to determine the proximity of amino acid residues to each other and their exposure to the solvent, providing clues to the likelihood of chemical reactions occurring at given residues.65,66 Stress formulation conditions can create additional degradation pathways not readily predictable by computational methods. C-terminal processing of lysine residues,67,68 N-terminal pyroglutamate formation, 69 deamidation, 70 glycation, 71 and oxidation72,73 are common chemical modifications of mAbs and their modalities. Many traditional analytical methods are available to characterize chemical modifications of monoclonal antibodies during development steps.74,75 There are well-established chromatographic methods for the analysis of chemical modifications in antibodies, including reversed phase, ion exchange, SEC, hydrophobic, and hydrophilic interaction chromatography, all widely used in formulation development. Although these chromatographic separations can take a relatively long time, a new generation of resins and high-pressure instrumentation has been introduced in recent years, allowing a much shorter runtime with comparable peak resolution to the older methods. Reversed-phase chromatography with a phenyl column has been used in the UPLC format with 1.7-µm particles. The cycle time was reduced to 5 min compared to 40 to 60 min for typical reversed-phase separations. 76 These short cycle times allowed the effective quantitative analysis of IgG1 and IgG2 clipping, and the same method has shown promise for the analysis of other chemical modifications such as oxidation. The advantages of high pressure and small resin particles have also been applied in ion exchange chromatography where the runtime for one antibody sample was reduced to 5 to 10 min instead of 30 to 40 min with traditional methods. The columns Bio WCX NP3 4.6 × 50 mm (Agilent Technologies, Santa Clara, CA) and Bio Pro SP-F 4.6 × 50 mm (YMC America, Allentown, PA) are good for these high-throughput applications. Ion exchange chromatography with a pH gradient has been successfully used to analyze monoclonal antibody charge variants. 77 The method has much better resolution and peak capacity than ion exchange chromatography based on the typical ionic strength gradient.
The gold standard for the analysis of chemical modifications of monoclonal antibodies is mass spectrometry (MS), where the mAb molecule is digested by an appropriate protease prior to analysis. This method is called peptide mapping and typically uses liquid chromatography (LC) coupled with a mass spectrometer to analyze the peptide mixture. LC-MS peptide mapping takes a long time and expertise to prepare samples, run the LC separation, and analyze the samples by MS. Recently, each step of peptide mapping has been improved to increase its overall throughput. In addition to above-mentioned advances for chromatography assays, automated liquid handling systems have been used for tedious sample preparation.78,79 The method used different channels in a 96-capillary array, and the full analysis was completed within 45 min. Modern mass spectrometers have very high resolution and, in some cases, allow the elimination of the chromatography step. Direct infusion has been used for glycan analysis 80 and peptide mapping of mAbs. 81 Separation by mass should be enough to distinguish the majority of chemical modifications. The new, advanced methods of MS data analysis combined with fast sample preparation and data acquisition can create a high-throughput platform for the screening of chemical modifications caused by stress and formulation conditions. In fact, formulation analysis can be greatly accelerated since the same protein sequence can be used in simple comparison of modified and control, nonmodified samples in different formulations. The pattern of modifications collected by high-throughput methods should be a powerful tool for formulation development. The types of chemical modifications can depend on the type of mAb-based modality. Possible degradations of “payloads” are the major concerns for ADC formulation development. 82 The minimized construct of nanobodies, another mAb-related modality, creates little room for covalent modifications since any changes in chemical structure will affect the critical area responsible for functionality. 83
Screening for Viscosity
Many biotherapeutic drugs based on monoclonal antibodies must be formulated and delivered at high concentrations, typically higher than 50 to 100 mg/mL. At such high concentrations, the distance between protein molecules become comparable to the size of individual protein molecules. Short intermolecular forces play a significant role in the determination of the overall properties of mAb solutions. One of the results of the increased influence of short-ranged interactions may be high viscosity of formulations that can create serious problems during drug purification and delivery. The exact mechanisms involved in the formation of high viscosities are still poorly understood. Charge distribution changes on the surface of an antibody are thought to play some role in dramatic changes of viscosity, 84 and dipole-dipole interactions have been suggested to be responsible for increased viscosity of antibody formulations. 85 Currently, there are no computational methods to accurately predict high viscosity, and traditional methods to measure viscosity take a long time, requiring substantial amounts of protein. Experimental methods to measure the viscosity of protein solutions are borrowed from polymer chemistry and include the falling ball method, 86 cone and plate assay, 87 and others. 88,89 A good review on high-viscosity problems associated with highly concentrated protein solutions and on the methods to measure viscosity has been published by Jezek et al. 90 Although not a major issue in the late stages of formulation development, the amount of available protein can be critical during the early stages. It is desirable to know as early as possible during development if a candidate molecule has a high viscosity at the concentrations that will be used in the formulation. At the early stage, the amount of available protein is simply not enough to reach the high-viscosity concentrations in the volumes required for traditional measurements. While the addition of certain ions or excipients can significantly decrease viscosity, 91 it is usually necessary to screen many compositions to find the right formulation, specific to the molecule of interest, that results in a lowered viscosity. This is why new methods have been developed to minimize the sample volume necessary for reliable viscosity measurement and to maximize the number of samples that can be tested. The Viscosizer 200 (Malvern Instruments Ltd., Worcestershire, UK) employs a UV imaging sensor to capture real-time data, improving the signal-to-noise ratio and eliminating a lamp effect. A protein-specific wavelength option enables the analysis of mAb formulations using a sample volume of approximately 10 µL. The Viscometer-Rheometer-On-a-Chip (VROC) introduced by RheoSense (San Ramon, CA) is a microfluidic chip-based instrument that can handle 100 µL of sample. DLS measurements can also be used to determine the viscosity of concentrated proteins. The addition of large polystyrene beads to a solution being studied results in a light-scattering signal that is easy to measure and quantify, and the diffusion coefficient can be determined by measurements of fluctuations of scattering light. 92 Usually, DLS is used to determine the size of aggregates or particles in a solution of known viscosity at a known temperature. But the measured diffusion coefficient can be used in the Stokes-Einstein equation (D = kT/6πηR) to calculate viscosity. The method has been specifically applied to antibody formulations.93,94 The DynaPro Platereader instrument (Wyatt Technology) was used in these studies. Only 20 to 50 µL of solution is needed to perform the measurements, and a 384-well plate can accommodate enough samples to screen many formulations. The major disadvantage of the DLS-based method is the possible interaction between the polystyrene beads and the proteins being studied that may lead to aggregation of a bead-protein complex. 95 Surface modifications of the beads can mitigate this problem.
Automation Tools
Automation can dramatically decrease the time of formulation development by replacing tedious and laborious manual processes with computer-controlled steps. Modern advancements in robotics and computerization make it possible to perform manipulations and measurements that were unthinkable 10 years ago. Robots and other elements of automation have been leveraged for a wide range of drug development processes, including drug screening, manufacturing, and purification. Formulation development is the ideal field for full implementation of automation and high-throughput technologies. Aliquotting numerous samples, preparing controls and standards in large volumes and multiple replicates, and repetitive tasks of sample transfer can be automated and controlled by software with a high level of precision and accuracy, completely without or with minimum human involvement and, of course, without the introduction of human error. The first steps of formulation studies include preparation of various solutions containing different components and their mixtures. The combination of pH, types of buffers, presence of ions, excipients, and surfactants creates a formulation space with multiple coordinates. To cover this space completely, many samples are required, and a multiwell plate platform is well suited for this purpose. The typical multiwell platform is 96-well based and requires only a small volume of protein solution. A standard footprint for this approach, adaptable for automation applications, has been developed by the Society for Biomolecular Screening (SBS). 96 The plates used in formulation studies often have a surface material, geometry, and other properties different from the final drug containers. Some caution should be taken, and the results of plate-based and container-based assays should be carefully compared before full implementation of formulation plate screening. There are many basic 96-well pipetting workstations to handle liquid transfer and mixing. Also, more sophisticated platforms manufactured by companies such as Tecan Group Ltd. (Männedorf, Switzerland), Eppendorf (Hamburg, Germany), and Hamilton (Bonaduz GR, Switzerland) are available for complex tasks of distribution of very small and large volumes. For those platforms, the addition of many deckware components can expand the capabilities of the system to include features such as plate agitation, centrifugation, and incubation at different temperatures. Many automation-friendly sample holders are available for fast characterization and formulation screening. 97 Analytical instruments can be coupled with the liquid handling platforms to automate the entire screening process. The integration of both liquid handling systems and analytical instrumentation can create a continuous process, and such a process has been used for high-throughput formulation screening of monoclonal antibodies. 98 Aggregation behavior of several mAbs in different formulations was characterized by multiple assays. In another study, IgG1 and IgG2 formulations were optimized with the help of high-throughput methods, and a Freedom EVO liquid handling station (Tecan Group Ltd.) was used to prepare 96 formulations for stability studies. 99 After incubation at 4, 25, and 40 °C, the 96-well plates were analyzed by high-throughput size exclusion chromatography using a temperature-controlled sampler storage unit and an autosampler. A good example of an integrated system specifically designed for formulation development of monoclonal antibodies is the system described by Zhao et al. 100 In the publication, the authors presented the custom-made high-throughput laboratory (HTL) based on the liquid handling robotic instrument, Star, produced by Hamilton and several separate analytical instruments, including DLS, lab-on-chip platform (5100 ALP from Agilent Technologies), SEC–high-performance liquid chromatography (HPLC), and cation exchange chromatography (CEX)–HPLC. The samples were prepared using a robotic system and were then transferred to storage before analysis. It is not clear whether the transfer was performed manually or automatically. All instruments had temperature-controlled plate holders and autosamplers for sample storage and injections. According to the authors, about 500 liquid formulations can be prepared in deep-well plates, distributed, and stored under different temperatures for degradation studies. A more automated high-throughput system is commercially available from Freeslate (Sunnyvale, CA). This system combines liquid handling instrumentation with integrated capabilities for pH measurements, viscosity measurements, and visual inspection/particle counting. Also, additional third-party instruments can easily be added to the platform to enhance the process of formulation screening. The company also developed its own Lab Execution and Analysis (LEA) software to handle all data obtained from the integrated analytical platform. It is important to keep such an automation platform open and flexible so that new advanced modules can be added to replace old ones, making it unnecessary to replace an entire obsolete system when new techniques and instruments become available. Automated systems are ideal for the development of formulation platforms to be applied to well-characterized candidates. Considering flexibility and openness of automation platforms, they can also be applied to the new molecules with different properties. Comparability studies could be easily performed using a platform approach and harmonization across various analytical techniques integrated into automated system.101–103 Quality assurance techniques can be used along with automation methods, and human errors are significantly decreased during sample preparation. Z′ trend 104 and plate pattern recognition 105 algorithms help to detect outliers and instrumental errors. High level of automation can be applied to ultra-low-volume systems based on the lab-on-chip (LOC) concept. Development of microelectromechanical systems (MEMS) has contributed significantly to LOC implementation in pharmaceutical and biopharmaceutical industries. 106 The major advantages of LOC include small sample volume, improved control, and lower energy consumption.
Analysis of High-Throughput Formulation Data
The analysis of data starts with the planning of an experiment and the experimental design of that experiment. It is critical to collect all available information about the stability of a molecule before starting any formulation studies. Usually, initial results on protein purification and solubility are available even during early stages of development. Amino acid sequence, theoretical pI, and total charge can be used for “hot spot” analysis for potential chemical modifications and for estimates of physical stability. Furthermore, it is highly desirable to use basic quality by design (QbD) principles in the experimental planning.107,108 This means that it would be possible to predict the formulation properties from the results of a well-designed experiment. In formulation development, the combination of formulation components creates the formulation space, and the data obtained from stability studies create the formulation stability space. In cases where the number of formulations is too large to screen, a design of experiment (DOE) approach can significantly save limited resources. The data obtained from experiments designed by DOE methods can produce a model of output parameters (e.g., physical or chemical stability) that will cover formulation space not included in experimental conditions. With the tools of statistical analysis, one can estimate the significance of formulation factors and confidence intervals for the predictive models. The more data points obtained by high-throughput methods, the more accurate the prediction model and the higher the efficiency of the formulation development experiment. Fractional factorial design was used in a study of a lyophilized IgG3 antibody. 109 DOE and specifically Box-Behnken experimental design were applied to screen stability parameters such as the glass transition temperature, level of aggregation and the unfolding temperature of the lyophilized product, and particle size of the reconstituted product. Simultaneous screening of colloidal and thermal stability resulted in effective formulation optimization in the study of IgG2 formulations. 110 When formulation factors have opposite effects on characterization parameters, a thorough statistical analysis is necessary to minimize the negative and maximize the positive influence of all factors. The presence of salt can decrease thermostability, 111 but at the same time, the salt can reduce the viscosity of some highly concentrated antibody solutions. 112 In another example, the prediction model was built based on experimental measurements of multiple formulations of a mAb at three concentrations. 113 The models showed a dependence of thermostability and viscosity on coordinates of pH values and protein concentrations in the presence of ions and excipients. The optimal combination of all formulation factors could be found, and any adjustments in values were easy to make according to the model. As shown in the studies cited above, for most formulation stability screenings, formulation factors and interactions between them are included as linear terms in multivariable regression models, and the range of factor values is narrow. Normally, the effect of pH is strong enough and sufficiently statistically significant to also be included as a quadratic term, which creates some curvature in the response analysis. For example, 96 formulations of IgG1 and IgG2 were screened before and after incubation at different temperatures. 99 Size exclusion chromatography was used to measure the percentage of monomer in each formulation, and a model was created with monomer percentage as the response value. In plots of monomer percentage versus pH, the maximum values were dependent on formulation compositions and were in the range of pH 5.2 to 6.0, consistent with results previously reported on mAb stability. In many cases, the right presentation and visualization of multidimensional data can help in the understanding of experimental results, leading to the selection of an optimal formulation. Multiple analytical techniques were used to characterize IgG1 particle formation, and radar charts array analysis was applied to visualize the effect of formulation components. 114 More complex analysis of characterization data obtained by different methods is based on statistical evaluation of spectroscopic and other biophysical results. Presentation of the data can be done using empirical phase diagrams, which depict the changes in biophysical properties by color. This approach was used for the analysis of highly concentrated antibody solutions115,116 and in other studies of antibody structure and stability.117,118
From Early Preformulation to Commercial Formulation Development
In this work, we describe various tools and methods that can significantly reduce the time and resources necessary for formulation development of mAbs and other modalities based on antibody domains. We cannot overestimate the importance of early formulation screening for candidates entering the development cycle. It is important to have the molecules that are being evaluated for further development in a stable solution for the various studies that are used for candidate selection. Solution stability of the candidates can affect the results of early bioactivity tests, giving erroneous results that do not reflect the true bioactivity of the molecule being tested, instead giving results for a degradation product of that molecule. Chemical modifications in the complementarity determining region (CDR) of an antibody directly change the affinity of antibody-antigen binding, often resulting in a loss of activity. Another potential problem caused by instability is an increase in immunogenicity due to the degradation product that may be absent in the original molecule.119–121 Conformational changes can lead to unfolding and aggregation or precipitation, which can decrease the apparent protein concentration in solution or lead to higher binding constants 109 and also can trigger immunogenic reactions. 122 Colloidal instability causes proteins to precipitate from solution or form separate liquid phases. Animal toxicology and early pharmacokinetics studies require stable formulations. The protease activity can be a serious problem in antibody formulations, with a residual amount of cell proteases left after the purification process. 123 The fragmentation of monoclonal antibodies seriously affects the quality of the final drug product, and preliminary screening for possible protease cleavage sites should be important part of early development.
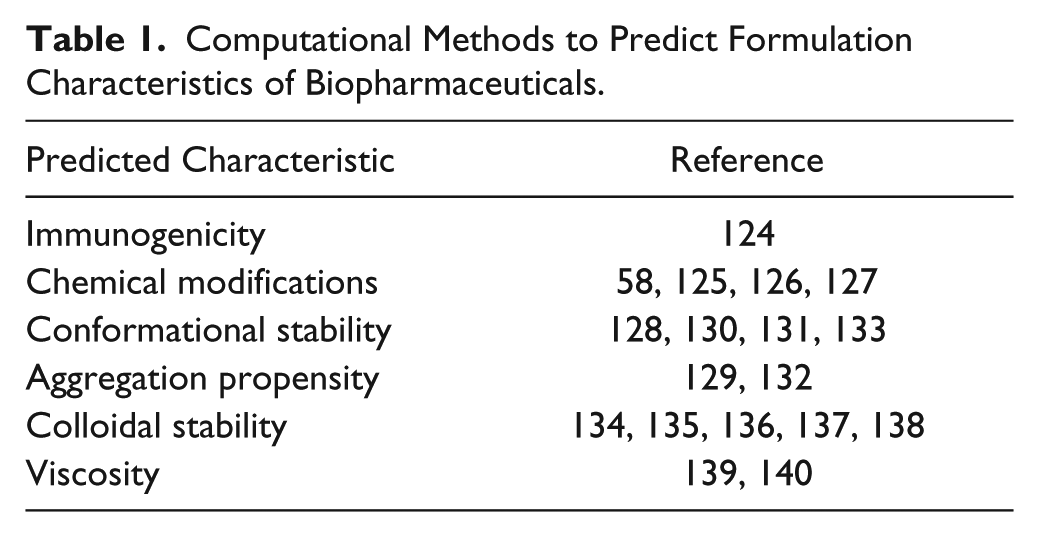
Modern methods of computer-based analysis can be employed as soon as the sequence and preliminary structure of a candidate molecule is known. Current sequence analysis and computational methods allow an accurate prediction of immunogenicity 124 and many types of chemical degradations.58,125–127 Structural stability and aggregation propensity can be also estimated by computer models.128–133 Prediction of colloidal stability, viscosity, and aggregation of native proteins is more difficult to do. Recently, several works have been published on the calculation and modeling of propensities for native aggregation,134,135 phase separation,136–138 and high viscosity.139,140 Some of the references to recent works on sequence-based computerized predictions of various characteristics that could affect formulation properties of proteins are presented in Table 1 .
Computational Methods to Predict Formulation Characteristics of Biopharmaceuticals.
Despite all of these recent developments, there is still no highly accurate method to predict the overall stability of a molecule under all possible stresses and in any formulation. Generally, even after applying predictive assays, it is usually prudent to carry out actual experiments to either prove the theoretical predictions or to create data to make additional predictions. With many candidates and limiting amounts of protein available during early development, formulation screening should be focused on both the selection of a stable formulation and the selection of suitable candidates for further development. Limiting amounts of protein dictates the use of small volumes and low concentrations. A multiwell plate platform is ideal for preformulation studies when the priority is the comparison and the ranking of multiple formulations and candidates. Various combinations of formulations and candidates can be screened during preformulation development. Typically, conformational stability is screened by DSF assay. 141 A heat map can be constructed based on Tm numbers obtained by DSF, and visual inspection immediately shows which formulations result in the highest and lowest thermal stability. Temperature ramping can be combined with the measurements of turbidity or light scattering by available plate readers. The temperatures for unfolding, aggregation, and precipitation can serve as characterization parameters for antibody formulations. 142
The plate with the formulations and candidates tested by DSF can be placed on a short-term stability study at increased temperature. After incubation, the aggregation level is measured by SEC and DLS, producing a table of monomer and aggregate peak percentage. The numbers are available for both visual and a more thorough statistical analysis. Static light-scattering (SLS) methods 143 coupled to chromatographic techniques can be very useful when accurate size determination is necessary. SLS analysis has been applied not only to protein formulations but also to virus and virus particle–containing vaccines.144,145
Precipitation by ammonium sulfate or PEG is a solubility test and is also a way to evaluate colloidal stability.46,146 On that stage, it is reasonable to limit the number of formulations to a minimum unless it is desired to stress candidates under conditions such as pH or ionic strength. The selected formulations can be used for preclinical studies. A wider formulation screening should be performed when the number of candidates is reduced to only a few or, in some cases, a single one and the candidates are ready to go to clinical development. For most therapeutic IgG1 and IgG2 antibodies, it is possible to use a platform formulation approach. 147 This means that one formulation is selected as the platform and screening is carried out on conditions closely related to the platform formulation. Platform analytical methods are typically also available. 148 Usually, other modalities are less stable than IgG, and a wider screening is required to find the stable solution for each molecule since the modifications and mixture of different domains produce a construct with hard to predict stability properties.148–150 After wide screening and identification of a stable formulation, a more narrow range of components can be tested with higher resolution, that is, with more data points between formulation factor variables. Commercial formulation development normally comes with the studies of several formulations and stress conditions to explore. 151 High-throughput methods help to accelerate those studies and to obtain statistically reliable results on stability. Instead of 1 to 2 years, the commercial development based on high-throughput assays could take just few months. It is possible to use multiwell plate-based and real container-based instrumentation together. Control studies should be performed to confirm that results obtained by plates and containers are comparable and not affected by the difference in surface or geometry properties. Plate-based methods can expand the formulation and stability space and can be complementary to containers studies. A highly automated sample handling system with integrated instrumentation for stress applications and analytical characterization will reduce analyst time and increase the accuracy of sampling and measurements. Also, automation reduces human error and provides many opportunities for detection of outliers and their sources. At any stage of formulation development, it is imperative to have as much information as possible prior to experimental design. Such information determines both output parameters and variable factors in the studies. It is also important to have a modular structure for stress and characterization techniques. Depending on the development stage, the modules can be selected to fit the requirements and needs of formulation studies. For example, for preformulation development, DSF and the precipitation assay should be enough to screen for conformational and colloidal stability. A 96-well plate is a good choice for the placement of formulations and candidates. Sixteen candidates can be screened in six formulations. If any specific problem appears, the selection of high-throughput methods and formulation factors are to be adjusted accordingly. In Table 2 , various automated and high-throughput methods that can be used to accelerate formulation development are presented.
Automated and High-Throughput Methods for Formulation Characterization Used in Various Stages of Biopharmaceutical Formulation Development.
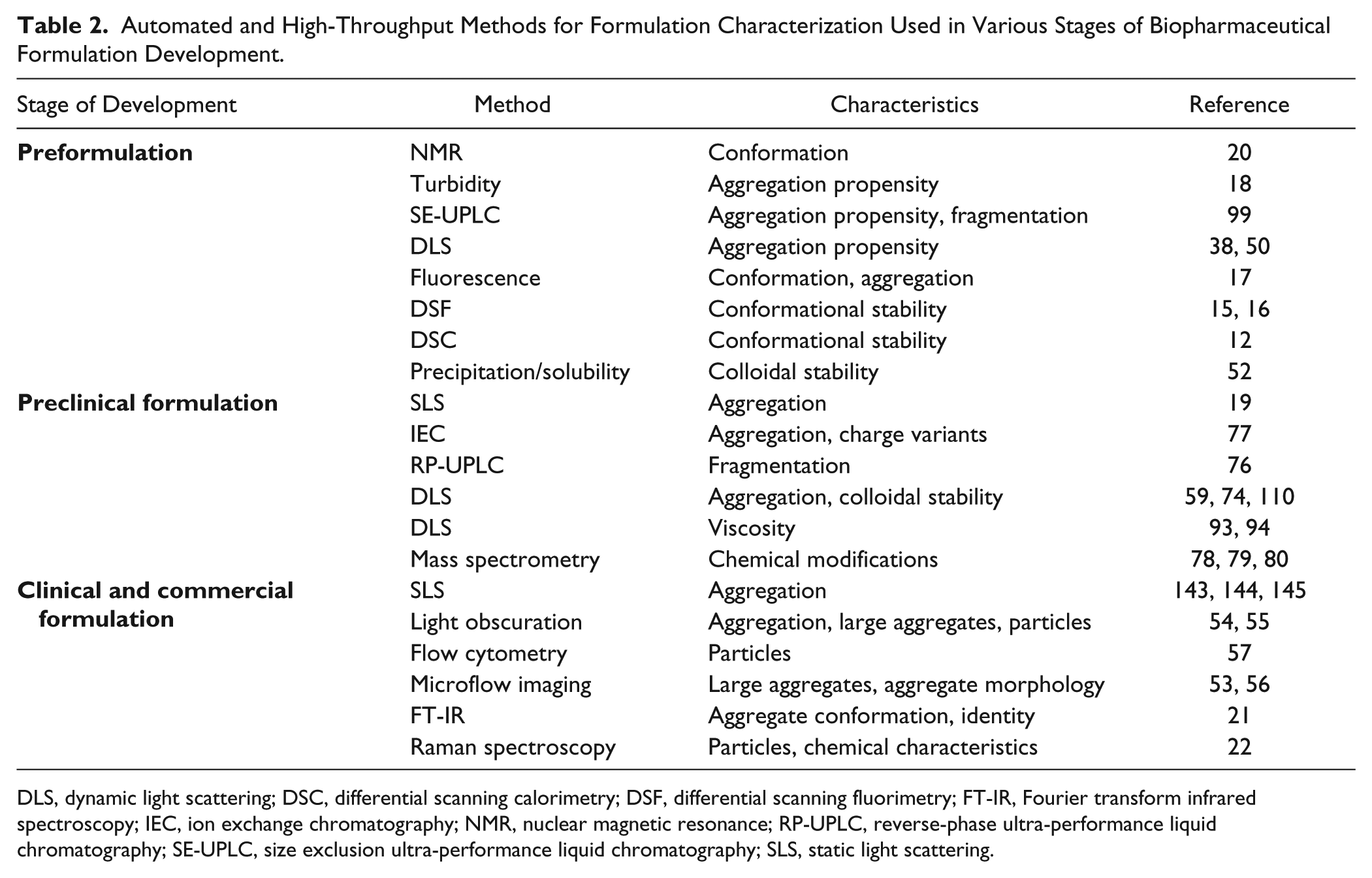
DLS, dynamic light scattering; DSC, differential scanning calorimetry; DSF, differential scanning fluorimetry; FT-IR, Fourier transform infrared spectroscopy; IEC, ion exchange chromatography; NMR, nuclear magnetic resonance; RP-UPLC, reverse-phase ultra-performance liquid chromatography; SE-UPLC, size exclusion ultra-performance liquid chromatography; SLS, static light scattering.
Implementation of high-throughput and automation technologies in formulation development of monoclonal antibodies creates a unique opportunity to move new drug candidates much faster through the development process with fewer resources being expended. It also generates an easily accessible and consistent pool of data that can be used for future projects and for the prediction of long-term stability.
Footnotes
Acknowledgements
We thank Dr. R. Matthew Fesinmeyer, Dr. Bruce Kerwin, and Dr. Ramil Latypov for fruitful discussion regarding methods of protein characterization.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
