Abstract
Plastic labware is used in all processes of modern pharmaceutical research, including compound storage and biological assays. The use of these plastics has created vast increases in productivity and cost savings as experiments moved from glass test tubes and capillary pipettes to plastic microplates and multichannel liquid handlers. One consequence of the use of plastic labware, however, is the potential release of contaminants and their resultant effects on biological assays. We report herein the identification of biologically active substances released from a commonly used plastic microplate. The active contaminants were identified by gas chromatography–mass spectroscopy as dodecan-1-ol, dodecyl 3-(3-dodecoxy-3-oxopropyl)sulfanylpropanoate, and dodecanoic acid, and they were found to be selective monoamine oxidase-B inhibitors.
Keywords
Introduction
Plastic materials are extensively used as disposable labware in pharmaceutical research. Although generally considered chemically inert, trace amounts of releasing agents or stabilizing antioxidants from the production process are known to be present in plastics and may leach into solutions that come into contact with the plastic. 1 These trace chemical additives that can be released from plastics, also described as extractables and leachables, may have a significant effect on the utility of plasticware as evidenced by bioassay-specific interferences identified while performing routine pharmaceutical drug discovery assays.2–5
In this communication, we report on the identification of extractables and leachables from disposable plastic labware that possesses selective inhibitory activity against monoamine oxidase-B (MAO-B) as compared to monoamine oxidase-A (MAO-A).
MAO enzymes exist in humans in two isoforms: MAO-A and MAO-B. MAO enzymes are present in the outer mitochondrial membrane of both neuronal and nonneuronal cells, and are responsible for the oxidative deamination of amines.6,7 Monoamine oxidase inhibitors (MAOIs) are used in the treatment of depressive disorders, anxiety disorders, Parkinson’s disease, and Alzheimer’s disease. Although there are many therapeutic benefits associated with the use of MAOIs, they can also have serious side effects, such as orthostatic hypotension, hepatotoxicity, and hypertensive crisis. 8
Due to the potential seriousness of the side effects associated with inhibition of MAO enzymes as well as the potential costs and complications associated with development of a compound with MAO-A and/or MAO-B inhibition as an off-target activity, high-throughput assays for both MAO-A and MAO-B inhibition have been developed at Bristol-Myers Squibb.
During routine operation of the MAO-A and MAO-B inhibition assays, a drastic and abrupt change in assay performance was noticed. The changes observed were an attenuation of the MAO-B assay high (maximal) signal (enzyme and substrate wells) and a significant negative shift in all of the values of the reference inhibitor concentration–response curves that are tested with every assay.
An analytical investigation using gas chromatography–mass spectroscopy (GC/MS) provided data on three closely related substances being present in the newly introduced assay plates that were not detected in the plate type previously used in the assay process. The three compounds were identified as dodecan-1-ol, dodecanoic acid, and dodecyl 3-(3-dodecoxy-3-oxopropyl)sulfanylpropanoate, the latter of which is used as an antioxidant in the manufacture of plastic products. Furthermore, we show that commercially obtained reference samples of these three agents possess selective inhibitory activity against MAO-B as compared to the MAO-A enzyme.
These results contribute to the increasing number of reports demonstrating that interfering substances are introduced into drug discovery assays by leaching from disposable plastic labware under routine conditions. In addition, we demonstrate that leachables and extractables from disposable plastic labware can possess highly specific biological activity against just one of a pair of closely related biological targets, such as the MAO-A and MAO-B enzymes, between which there is nearly 70% sequence homology. 9 Therefore, routine validation of labware for nonspecific activities may no longer guarantee the compatibility of that labware among assays or target classes. This emphasizes the importance of rigorous validation of both specific reagents and consumables for individual assays to ensure the successful deployment of pharmaceutical drug discovery assays.
Materials and Methods
Extraction and GC/MS Analysis
Eighty microliters (80 µL) of neat DMSO (Sigma-Aldrich, St. Louis, MO) were added to empty wells of either a 384-well Matrix deep-well storage plate (Matrix Technologies, Hudson, NH) or a REMP 384-well microplate (VWR, Visalia, CA). Plates were sealed and incubated at room temperature in the dark for 12 days. Like-samples from each respective plate were collected and combined to yield approximately 1 mL of each sample in glass dram vials (VWR). The samples were then evaporated to dryness under a nitrogen stream at room temperature, and the resulting residues were dissolved in 100 µL of a high-performance liquid chromatography solvent grade chloroform–methanol 1:1 mixture.
In preparation for GC/MS analysis, the chloroform–methanol extracts were again evaporated to dryness under a nitrogen stream to remove any aqueous residue. Using a standard derivatization procedure for GC/MS analysis, 100 µL of MSTFA (N-methyl-N-trimethylsilyl-trifluoro acetamide; Fluka, St. Louis, MO) was added to the sample residues and gently shaken for 1 h at 37 °C. The samples were then analyzed on a Thermo Scientific GC/MS system (San Jose, CA) composed of a TriPlus autosampler, a Trace gas chromatographer, and a DSQII single quadrupole mass spectrometer. The GC column was a Phenomenex ZB-5ms phase, 0.25 mm ID, 0.25 µm film thickness (FT), 30 m length (Torrance, CA), and the carrier gas was helium. The GC inlet was set at 250 °C and a split ratio of 50. The GC column was initially set at 60 °C for 1 min and then ramped at 10 °C/min to 325 °C. After a 3 min solvent delay, positive electron impact (EI) ionization with 70 eV ion energy and a scan range from m/z 50 to 700 was used. The GC/MS operating software Xcalibur 1.4 and the NIST Mass Spectral Library version 2.0 were used for identification of the extracted materials.
Test Compound Preparation
Test compounds [deprenyl (Sigma-Aldrich), clorgyline (Invitrogen, Grand Island, NY), dodecan-1-ol (Sigma-Aldrich), dodecyl 3-(3-dodecoxy-3-oxopropyl)sulfanylpropanoate (Sigma-Aldrich), and dodecanoic acid (Fluka)] were solubilized to 3 mM in neat DMSO. One hundred and twenty microliters (120 µL) were then transferred to their own respective wells located in column 1 (wells A1–E1) of a Matrix deep-well storage plate. Eighty microliters (80 µL) of neat DMSO were then added to wells A2–E12 of the same Matrix storage plate. Serial dilutions were carried out by transferring 40 µL of each of the test compounds from column 1 to column 2, mixing thoroughly, then transferring 40 µL of the mixed samples from column 2 to column 3, and so on until an 11-point serially diluted concentration–response curve was created for each of the test compounds. Five hundred nanoliters (500 nL) of each of the samples were immediately transferred to two Corning standard surface black–black flat-bottom 384-well assay plates (Corning, NY) for testing in the MAO-A and MAO-B inhibition assays. Final concentrations of each test compound were 30 µM, 10 µM, 3.33 µM. 1.11 µM, 370 nM, 120 nM, 40 nM, 13.7 nM, 4.5 nM, 1.5 nM, and 0.5 nM.
MAO-A and MAO-B Inhibition Assays
MAO assay kits were obtained from Invitrogen, and MAO-A and MAO-B supersomes were from Corning. The assay detects hydrogen peroxide, one of the products of MAO activity. Hydrogen peroxide reacts with Amplex Red, generating the fluorescent product resorufin. The assay was run according to the manufacturer’s instructions with the modifications described in the next paragraph, which were determined from linearity experiments conducted in-house.
Briefly, enzyme was preincubated with test compound for 15 min at room temperature prior to initiation of the assays (final enzyme concentrations were 0.0625 µg/well for MAO-A or 0.25 µg/well for MAO-B, 1% DMSO). Following the preincubation, either MAO-A or MAO-B substrate solutions were added to initiate the assays [final concentrations were 20 or 40 µM Kynuramine (for MAO-B and MAO-A, respectively), 1 U/mL horseradish peroxidase, and 200 µM Amplex Red], and the assays were incubated at room temperature for an additional 60 min, protected from light. At the end of the 60 min incubation, the fluorescence intensity of each sample was measured with a BMG Pherastar plate reader (Cary, NC) using a 540Ex/590Em filter module.
Data Analysis
The signal-to-background ratio was defined as the ratio between the assay maximal signal (totals) and minimal signal (blanks).
The assay maximal signal (totals) was defined as the fluorescence intensity generated when combining enzyme and substrate solutions in the absence of test compound; it is measured in relative fluorescent units (RFUs).
The assay minimal signal (blanks) was defined as the fluorescence intensity generated in the absence of test compound and enzyme but in the presence of substrate solution; it is also measured in RFUs.
Concentration–response curves for test compounds were analyzed, and IC50 values were calculated using four-parameter linear regression analysis.
Results and Discussion
During the routine performance of MAO-A and MAO-B inhibition assays, a drastic and abrupt decrease in the assay signal-to-background ratio was observed. This loss in signal was not due to increased signal in the minimal signal wells, nor was it due to reduced enzyme activity because the wells that contained the lowest concentrations of the reference inhibitor had expected levels of activity (eightfold greater than background; see Fig. 1 ).
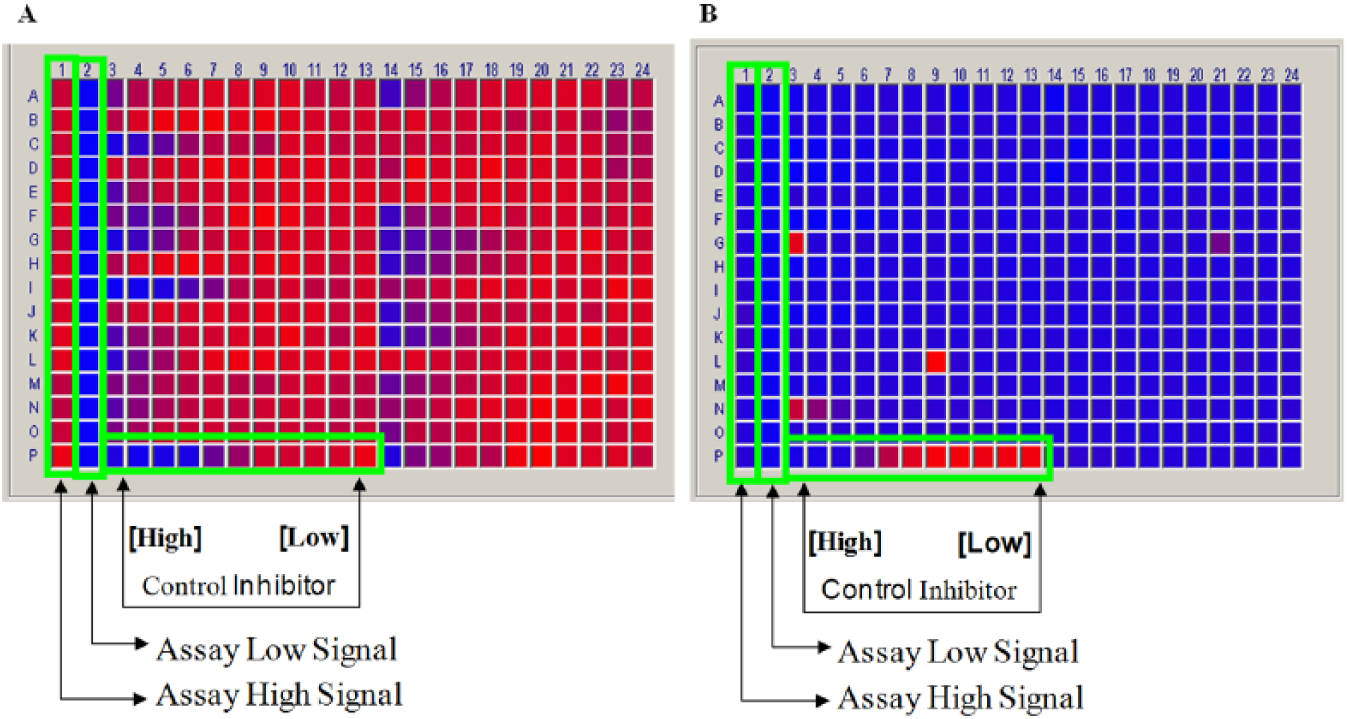
Heatmaps from a monoamine oxidase-B (MAO-B) inhibition assay before (
A detailed investigation revealed and confirmed that a recent substitution of microplates used in the compound preparation process (from Matrix microplates to REMP microplates) was the root cause of the change in assay performance. It should be noted that both of these plate types (REMP and Matrix) are routinely used for a variety of assays and were considered to be interchangeable labware in our laboratory.
Following up on the finding that the REMP plate was the source of the agents interfering with MAO-B activity, GC/MS was used to identify three closely related agents extracted from the REMP plates that were not observed in comparable extracts from the Matrix deep-well plates. These extracted agents were identified as dodecan-1-ol (CAS 112-53-8), dodecyl 3-(3-dodecoxy-3-oxopropyl)sulfanylpropanoate [CAS 53571-83-8, common name: dilauryl thiodipropionate (DLTDP)], and dodecanoic acid (CAS 143-07-7) (
Figs. 2
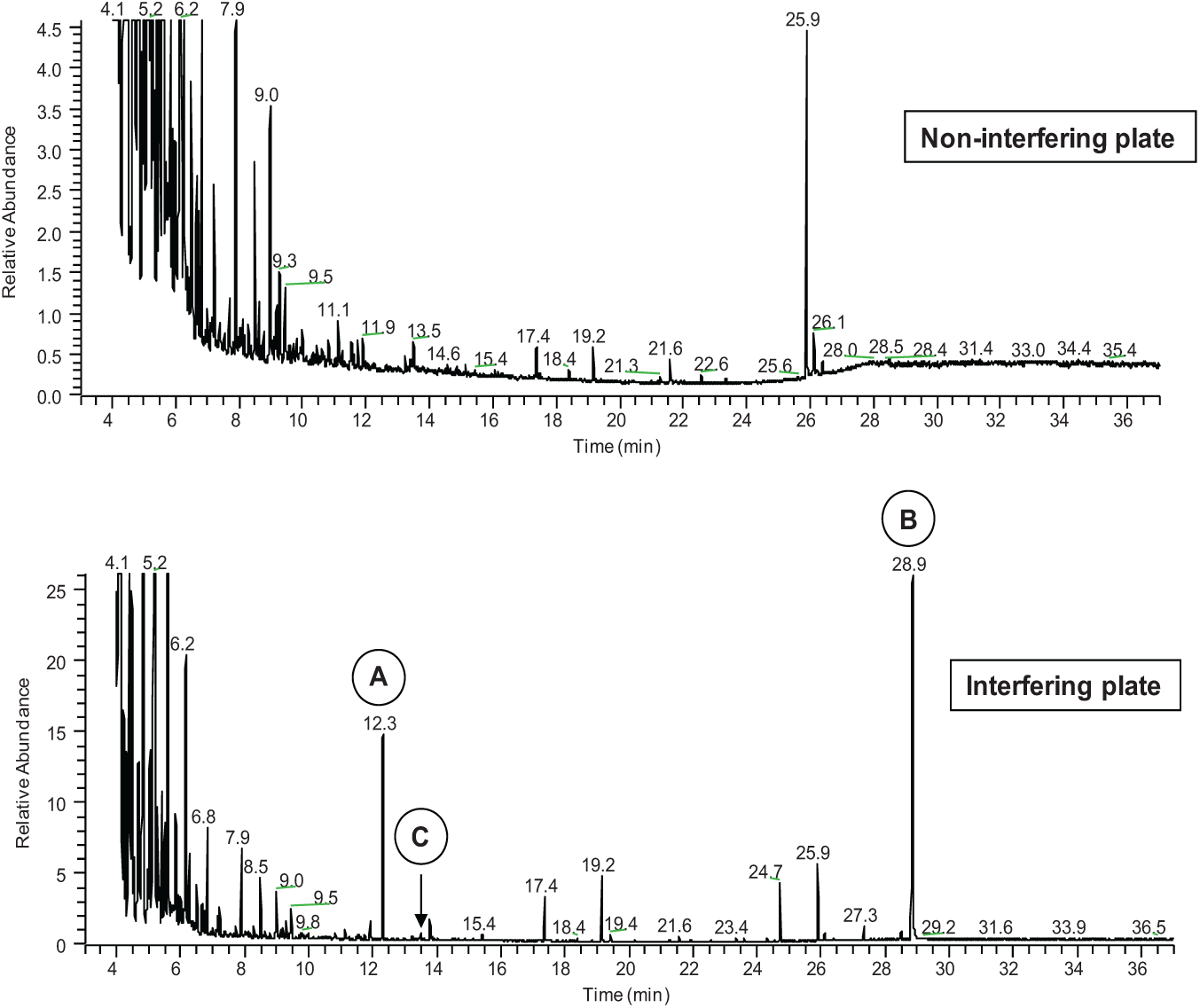
Microplate extractions confirm the presence of unknown species. A comparison of Matrix microplate (non-interfering) and REMP microplate (interfering) sample extractions showing unknown species in the REMP microplate extract at 12.3 min, 28.9 min, and 13.3 min.
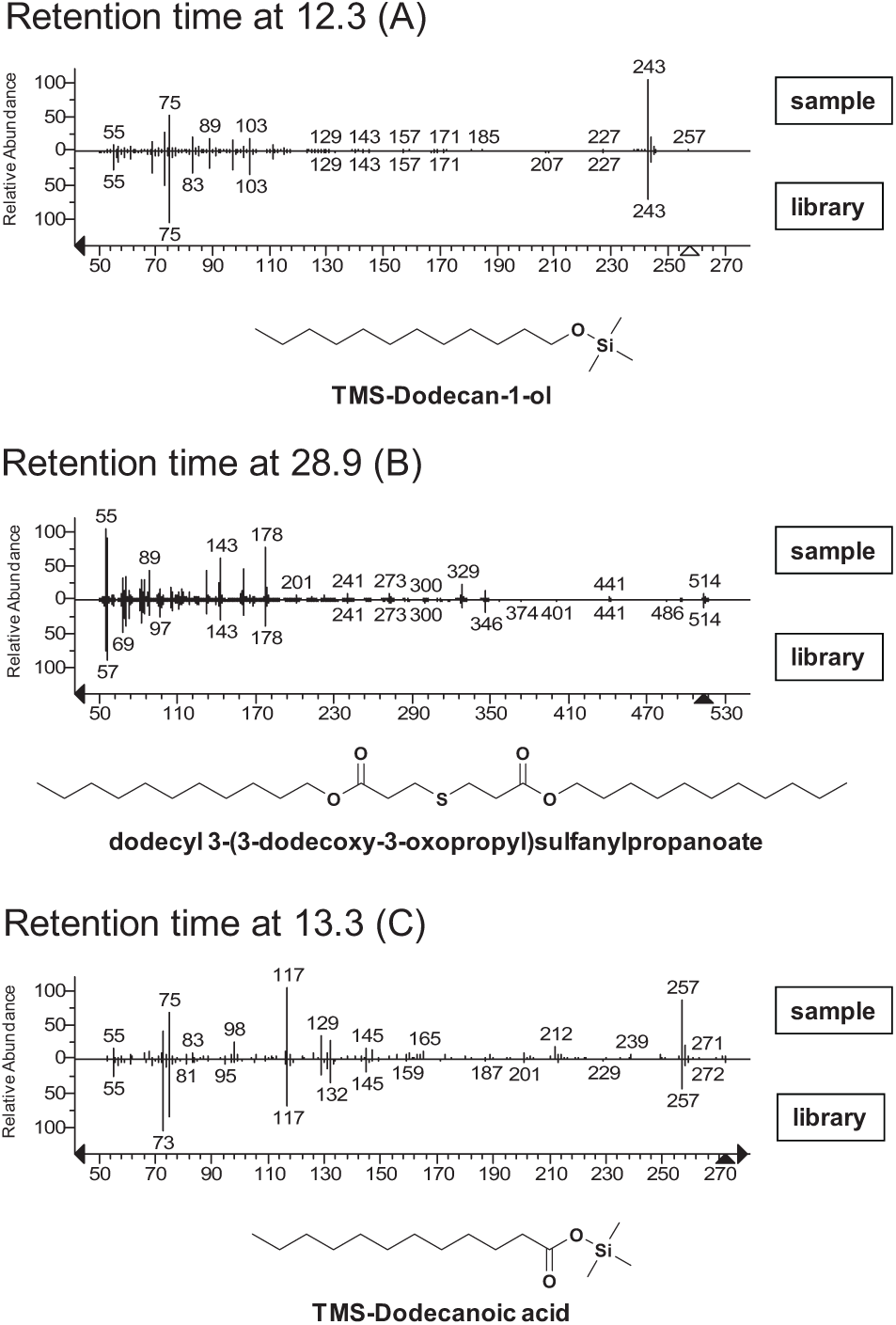
Identification of unknown analytes. The three unknown analytes from the REMP microplate extract were identified as TMS-dodecan-1-ol, dodecyl 3-(3-dodecoxy-3-oxopropyl)sulfanylpropanoate, and TMS-dodecanoic acid, respectively.
Thiodipropionates are used as antioxidants in the manufacture of plastics as a preservative that acts by decomposing hydroperoxides.10–12 Dodecan-1-ol is a known precursor of DLTDP. The trace dodecanoic acid detected is presumed to be derived from its corresponding alcohol.
All three compounds identified from the REMP microplate extraction demonstrated significant and selective inhibition of the MAO-B enzyme as compared to the MAO-A isoform, which shares ~70% sequence homology with MAO-B. 9 Dodecan-1-ol is a highly selective and potent inhibitor of MAO-B with a demonstrated IC50 value of 0.2 µM ( Fig. 4 ). DLTDP is a 40-fold weaker selective inhibitor of MAO-B than dodecan-1-ol, with an IC50 value of 7.8 µM. Dodecanoic acid is a very weak selective inhibitor of MAO-B demonstrating an IC50 value greater than 30 µM. Clorgyline and deprenyl were used as reference inhibitors to monitor assay performance, including enzyme activity and selectivity in all test occasions. Clorgyline demonstrated IC50 values of 1.1±0.3 nM and >250 nM against MAO-A and MAO-B, respectively, and deprenyl demonstrated IC50 values of >1 µM and 9.3±0.9 nM against MAO-A and MAO-B, respectively.
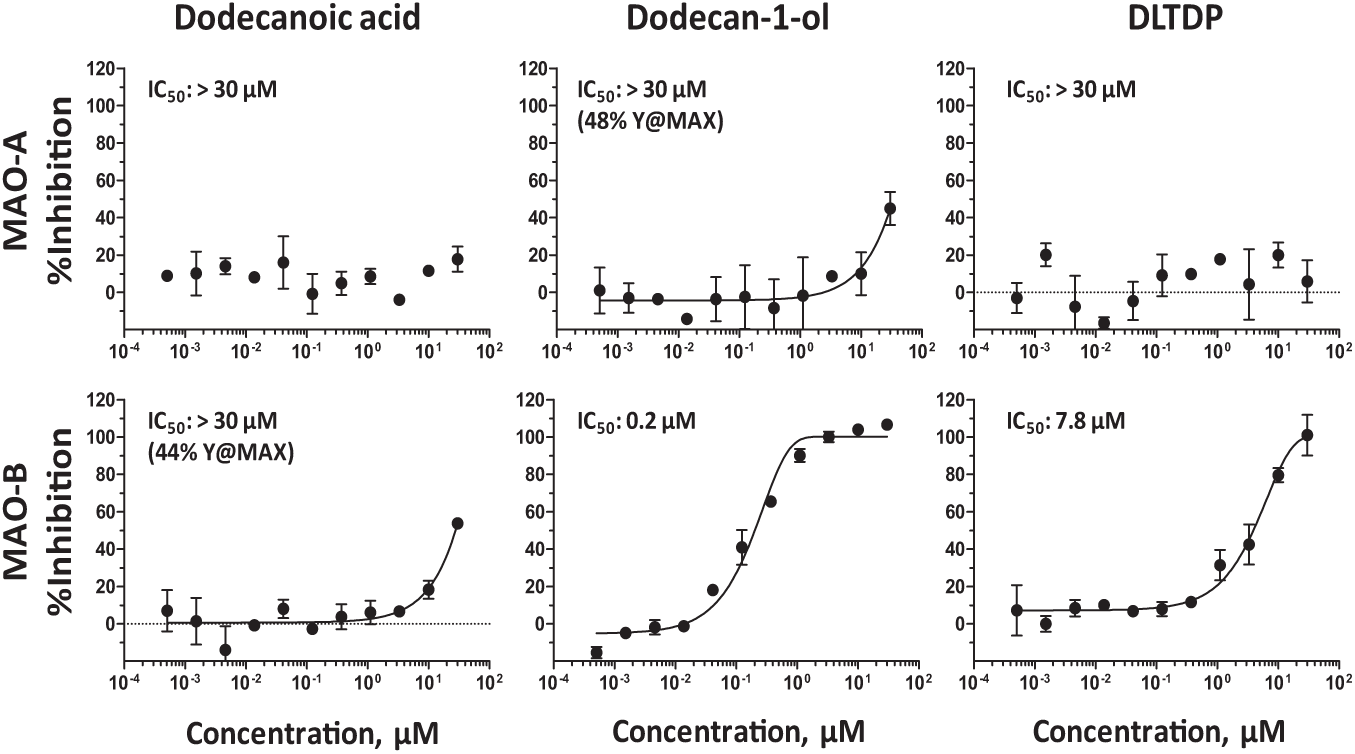
Activity of extracted compounds against monoamine oxidase-A (MAO-A) and monoamine oxidase-B (MAO-B) enzymes. The analytes identified from the REMP microplate extraction were commercially sourced and assayed for activity in the MAO-A and MAO-B enzyme assays. Dodecan-1-ol demonstrated >150-fold potency against MAO-B compared to MAO-A. DLTDP, dodecyl 3-(3-dodecoxy-3-oxopropyl)sulfanylpropanoate; Y@Max, the percentage of inhibition observed at the highest concentration tested.
Based on these findings, the use of Matrix deep-well microplates for test compound storage and delivery was resumed, and MAO-B assay performance immediately returned to normal.
The growing frequency and number of bioactive contaminants identified and reported in the literature to be leaching from plastic have attracted the attention of the pharmaceutical industry. There are international industry conferences dedicated to the subject of extractables and leachables, and in 2007 a group of pharmaceutical, biotech, and medical device companies formed an exchange [the Extractables and Leachables Safety Information Exchange (ELSIE), www.elsiedata.org] for the purpose of capturing a list of bioactive extractables and leachables.
Previously, simple and long-chain fatty amide leachables such as oleamide and erucamide (commonly used as slip agents in plastics manufacture) had been reported to effect enzyme or G protein-coupled receptor signaling, but biological assay interference from antioxidant leachables such as DLTDP (used for preservation of the end product) has not been widely discussed.5,13 Here, we report the identification of three leachables that are selective inhibitors of MAO-B. These findings further buttress the literature suggesting that the magnitude of the extractables and leachables problem is growing in both size and scope and affecting many diverse industries.14,15
In conclusion, we report the identification of three leachables from disposable plastic labware that were found to be MAO-B-specific inhibitors, among which was the antioxidant DLTDP. Bioactive contaminants in drug discovery assays pose many challenges, including costly loss of time, productivity, and efficiency due to investigating the source of interfering agents and resolving conflicting data sets. It is incumbent on all of us in the research community to be aware of the issues described herein and perform proper due diligence to ensure that valid biological results are obtained from our research efforts. We recommend continuing and strengthening collaborations between research organizations and labware manufacturers to address the problem and mitigate the significant impact that extractables and leachables are having on research.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Leads Discovery & Optimization, Pharmaceutical Candidate Optimization, and Synthesis & Analytical Technology Team organizations at Bristol-Myers Squibb Company, Wallingford, CT.
