Abstract
Acute lymphoblastic leukemia (ALL) is the most common pediatric malignancy. Glucocorticoids (e.g., dexamethasone) form a critical component of chemotherapy regimens for pediatric ALL, and the initial response to glucocorticoid therapy is a major prognostic factor, where resistance is predictive of poor outcome. We have previously established a clinically relevant ALL xenograft model, consisting of primary pediatric ALL biopsies engrafted into immune-deficient mice, in which in vitro and in vivo dexamethasone sensitivity significantly correlated with patient outcome. In this study, we used high-throughput screening (HTS) to identify novel compounds that reverse dexamethasone resistance in a xenograft (ALL-19) derived from a chemoresistant pediatric ALL patient that is representative of the most common pediatric ALL subtype (B-cell precursor [BCP-ALL]). The compound 2-(4-chlorophenoxy)-2-methyl-N-(2-(piperidin-1-yl)phenyl)propanamide showed little cytotoxic activity alone (IC50 = 31 µM), but when combined with dexamethasone, it caused a marked decrease in cell viability. Fixed-ratio combination assays were performed against a broad panel of dexamethasone-resistant and -sensitive xenografts representative of BCP-ALL, T-cell ALL, and Mixed Lineage Leukemia–rearranged ALL, and synergy was observed in six of seven xenografts. We describe here the development of a novel 384-well cell-based high-throughput screening assay for identifying potential dexamethasone sensitizers using a clinically relevant ALL xenograft model.
Introduction
Improvements in treatment of the most common pediatric cancer, 1 acute lymphoblastic leukemia (ALL), have led to 5-year event-free survival rates of more than 85% and 5-year overall survival rates approaching 90%. 2 Despite the high survival rates, ALL claims more children’s lives than any other cancer, with approximately 600 deaths yearly in the United States. 1 Treatment of ALL consists of 2 to 2.5 years of chemotherapy, which is composed of three phases: remission induction, intensification (consolidation), and continuation (maintenance) therapy. 3 Glucocorticoids such as prednisone and dexamethasone are critical components of remission induction therapy protocols and are frequently used in both intensification and continuation therapies. 3 Although prednisone has been traditionally used, dexamethasone is now increasingly used as it provides better control of central nervous system leukemia. 4
Response to initial glucocorticoid therapy is also used as a powerful prognostic factor in pediatric ALL, where resistance is predictive of poor outcome.5,6 Approximately 5% to 10% of pediatric ALL patients display intrinsic resistance to glucocorticoids and, although placed on high-risk treatment protocols, have an inferior 5-year event-free survival of approximately 55%. 7 Approximately 15% of pediatric ALL patients eventually relapse, and survival after relapse is dismal, with a 5-year event-free survival of approximately 15% for some high-risk subgroups. 8 The high incidence of pediatric ALL means that although fewer patients are relapsing due to improved treatments, relapsed ALL is still ranked as the fifth most common pediatric cancer. 9 Acquired resistance to glucocorticoids is common at relapse and is disproportionate compared with other chemotherapeutic drugs. 10 Therefore, although glucocorticoids can be extremely effective, the emergence of resistance remains a significant barrier to cure.
Glucocorticoids are steroid hormones that induce apoptosis in normal and malignant lymphoid cells. 11 Glucocorticoids enter the cell through passive diffusion, where they bind to the glucocorticoid receptor (GR), a member of the nuclear receptor family of ligand-dependent transcription factors. 11 Studies on the mechanisms of glucocorticoid resistance in leukemia cell lines have shown that resistance is generally associated with GR defects, such as down-regulation of the GR or defective ligand binding to the GR.11–13 This mechanism of glucocorticoid resistance has been proven less relevant in the clinical disease setting, however, because GR defects are rarely observed in primary patient material.11,14,15
We have previously established a clinically relevant ALL xenograft model, consisting of primary pediatric ALL biopsies engrafted into immune-deficient mice.16,17 Glucocorticoid resistance in patient-derived xenografts was not due to GR defects and occurred downstream of ligand-induced nuclear translocation of the GR.18,19 In this model, the in vitro and in vivo sensitivity of xenografts to dexamethasone reflected the outcome of the patients from whom they were derived, which supports the use of this model in a high-throughput screening (HTS) assay to identify dexamethasone sensitizers. 16 We have previously shown that dexamethasone resistance can be partially reversed both in vitro and in vivo using the histone deacetylase inhibitor vorinostat (suberoylanilide hydroxamic acid) or the receptor tyrosine kinase inhibitor SU11657, which indicates that dexamethasone resistance can be reversed by pharmacologic intervention.19,20 A number of strategies have been adopted to identify drugs that overcome glucocorticoid resistance, including bioinformatics and testing candidates based on mechanism of action.21–24 However, as none of these candidates were specifically developed as glucocorticoid sensitizers, we developed a novel HTS assay to identify these compounds.
Cell-based HTS assays have risen in popularity and now represent approximately half of all HTS campaigns. 25 We chose to perform a cell-based assay as there is no prior bias of known molecular targets, therefore allowing for the selection of compounds that can be used as probes to further elucidate the mechanisms of glucocorticoid resistance or sensitivity. 25 By using a human pediatric ALL xenograft, we intended to identify compounds that sensitize leukemia cells to dexamethasone in a clinically relevant model. The xenograft (ALL-19) selected for use in the screen exhibits robust and high-level dexamethasone resistance both in vitro and in vivo and is representative of the most common pediatric ALL subtype, B-cell precursor ALL (BCP-ALL).16,17 HTS compounds were initially tested for their ability to reduce cell viability at one concentration in the presence of a fixed concentration of dexamethasone. Hit compounds were then tested for their ability to reduce cell viability preferentially in the presence of dexamethasone. Confirmed hit compounds were then tested in a dose-response format, with and without a set concentration of dexamethasone to identify the most effective sensitizers. Of the 40,000 compounds initially tested, 4 hit compounds were identified. These compounds may exhibit some cytotoxic effects when administered alone but at higher concentrations than was required to potentiate the effects of dexamethasone. This report details the development and validation of the novel HTS assay, selection and confirmation of dexamethasone sensitizers, and analysis of the applicability of the lead compound. Ultimately, the pharmacologic reversal of dexamethasone resistance may lead to improved outcomes for patients with intrinsic/acquired lymphoid malignancies.
Materials and Methods
Reagents and Equipment
Cell culture–tested, water-insoluble dexamethasone was used in all experiments (Sigma-Aldrich, Castle Hill, NSW, Australia). Cell viability was determined using ViaCount (Merck Millipore, Billerica, MA) or resazurin reagent. Flow cytometry was performed using a Guava easyCyte flow cytometer (Merck Millipore). Screened compounds were a random selection of 40,000 compounds from the Australian Cancer Research Foundation Drug Discovery Centre for Childhood Cancer diversity library. Screened compounds were predissolved in DMSO (5 mM) and tested at a final concentration of 10 µM (unless otherwise stated).
In Vitro Cell Culture
The development and characterization of a series of pediatric ALL xenografts derived from patient biopsies have been previously described.16,17 For all experiments described in this study, xenograft cells were retrieved from cryostorage and resuspended in RPMI-1640 medium (Invitrogen Life Technologies, Gaithersburg, MD) supplemented with 10% fetal bovine serum (Invitrogen Life Technologies), penicillin (100 U/mL), streptomycin (100 µg/mL), and L-glutamine (2 mM; complete RPMI). Cells were centrifuged at 490 × g for 5 min, aspirated, and washed with complete RPMI. The cells were resuspended in QBSF-60 medium (Quality Biological, Gaithersburg, MD) supplemented with Flt-3 ligand (20 ng/mL; Amgen, Thousand Oaks, CA), penicillin (100 U/mL), streptomycin (100 µg/mL), and L-glutamine (2 mM; complete QBSF) at a cell concentration previously optimized for each xenograft (1–5 × 106 cells/mL). Viability was determined by the exclusion of 0.2% trypan blue (Sigma-Aldrich). For all experiments, cells were seeded and equilibrated at 37 °C, 5% CO2, for 12 to 16 h prior to drug treatment.
Primary HTS Assay
All HTS assays were performed using mycoplasma-free and validated stocks of xenograft cells, using a Hamilton Microlab STAR liquid-handling robot. All HTS assays were performed in 384-well black, clear-bottom tissue-culture treated plates (Sigma-Aldrich) using fluorescence as the readout for cellular viability as measured with the resazurin cell viability assay. ALL-19 xenograft cells, passage 5, were seeded at 50,000 cells/well in 50 µL complete QBSF medium and were allowed to equilibrate overnight (12–16 h).
Test compounds (5 mM stock concentration in DMSO) were added to each assay plate using a pintool (V&P Scientific Inc., San Diego, CA) at 100 nL/well for a final concentration of 10 µM. Immediately after test compound addition, dexamethasone (0.5 mM stock concentration in DMSO) was added to each assay plate using a pintool (V&P Scientific, Inc.) at 100 nL/well for a final concentration of 1 µM and a final DMSO concentration of 0.4% (v/v). Plates were incubated at 37°C, 5% CO2, for 24 h, and then resazurin reagent was added using an automated Multidrop Combi Reagent Dispenser (Thermo Fisher Scientific, Scoresby, VIC, Australia) at 5 µL/well. Initial fluorescence (0 h read) was measured using a plate fluorimeter (Molecular Devices, Sunnyvale, CA) with excitation at 530 nm and emission at 590 nm. Plates were incubated for 24 h before the final fluorescence was measured (24 h read). At both time points, plates were incubated at room temperature for 5 minutes in the absence of light before reading fluorescence. See Figure 1A for a schematic of the experimental timeline.
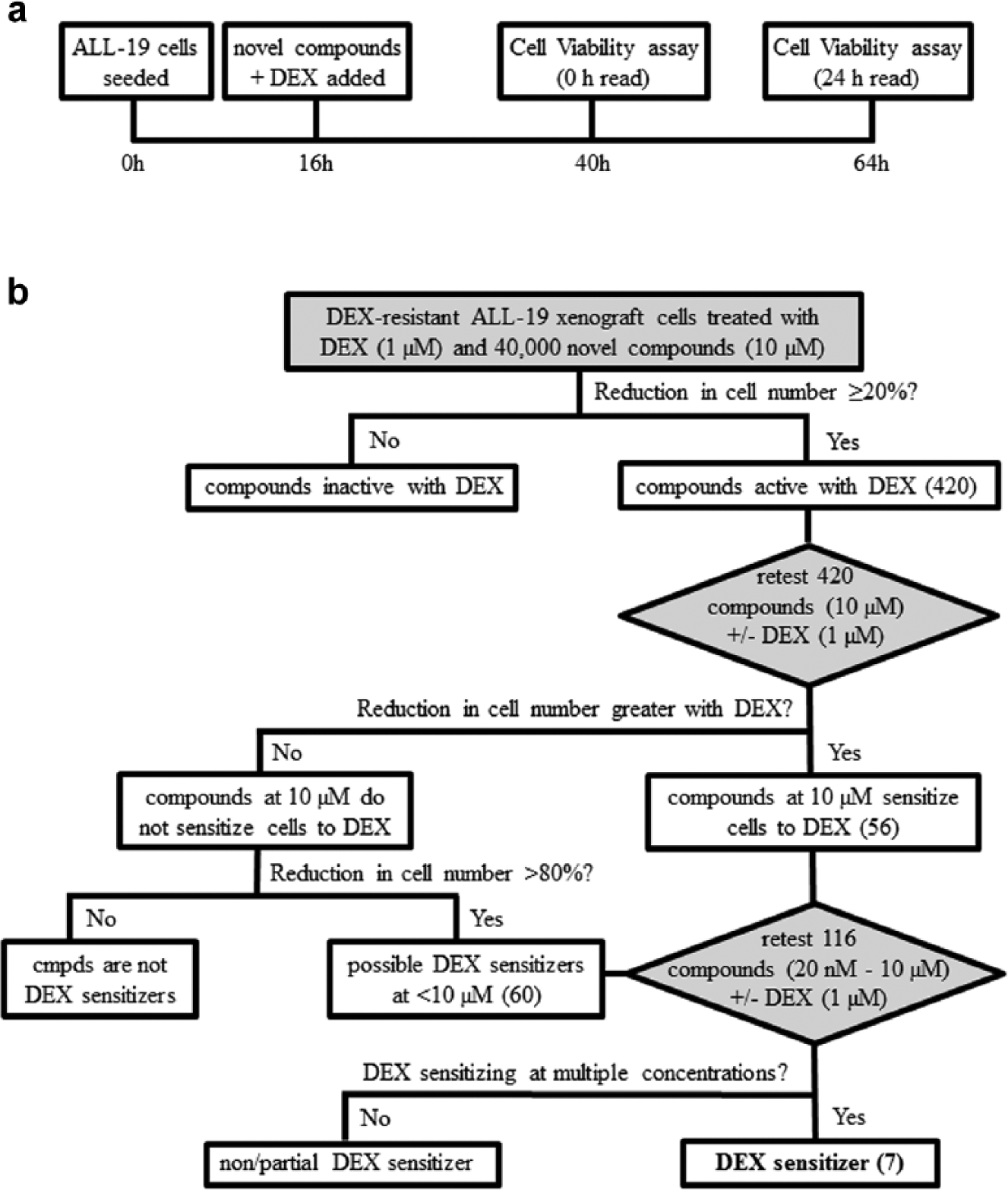
Graphical representation of high-throughput screening (HTS) campaign. (
The following controls were included per assay plate:
HTS assays were conducted in batches of up to 20 assay plates with a dexamethasone control plate included at the beginning and end of each batch. The dexamethasone control plates consisted of dexamethasone-sensitive, ALL-3 xenograft cells, 16 passage 4, seeded at 100,000 cells/well in 50 µL complete QBSF medium. Half of the wells on each plate were treated with dexamethasone (1 µM final concentration, positive control), whereas the other half were treated with a comparable concentration of DMSO carrier (negative control). This control ensured that the dexamethasone was active and was added to wells evenly.
Calculations Employed in HTS Analysis
The change in relative fluorescence units (RFU) was calculated for each well by deducting the initial fluorescence (0 h read) from the final fluorescence (24 h read). Percentage cell viability was calculated for the test samples based on the negative control (100% signal), which permitted correction for any minimal effect of 1 µM dexamethasone. Percentage cell viability was calculated as follows:
where Avg RFU = average relative fluorescence units.
The Z-factor was calculated for each assay plate, according to the method by Zhang et al. 26
Secondary HTS Assay
Test compounds (420) from the primary screen that resulted in ≥20% reduction in cell viability were selected for the secondary HTS assay. Compounds were tested on ALL-19 cells (50,000 cells/well) in triplicate, with 1 µM dexamethasone (final concentration) and without dexamethasone (DMSO treated), following the same protocol and time course employed in the primary HTS assay. Percentage cell viability for test compounds + dexamethasone were calculated based on control wells treated with 1 µM dexamethasone (final concentration), where percentage cell viability for test compounds – dexamethasone was calculated based on control wells treated with DMSO carrier.
Hits were defined as follows:
Compounds that caused an additional reduction in cell viability (≥15%) when combined with 1 µM dexamethasone, when compared with cell viability in the absence of dexamethasone.
“Potent” compounds with ≥80% reduction in cell viability ± 1 µM dexamethasone.
Compounds with a fold change increase in activity with dexamethasone >1 standard deviation from the mean ratio. Fold change increase in activity (decrease in cell viability) was calculated by dividing the percentage cell viability – dexamethasone by the percentage cell viability + dexamethasone.
Tertiary HTS Assay
Test compounds (116) that were defined as hits in the secondary screen were selected for the tertiary HTS assay. Compounds were serially diluted in DMSO using a pintool (V&P Scientific, Inc.) to a range of final concentrations (10 µM–20 nM). ALL-19 cells were tested with a dose response of each compound in triplicate, with 1 µM dexamethasone (final concentration) and without (DMSO treated), following the same protocol and time course used in the primary HTS assay. Percentage cell viability for test compounds + dexamethasone was calculated based on control wells treated with 1 µM dexamethasone (final concentration) alone, where percentage cell viability for test compounds – dexamethasone was calculated based on control wells treated with DMSO carrier. Compounds (7) that exhibited greater cytotoxicity with 1 µM dexamethasone at a range of concentrations were defined as hits.
In Vitro Cytotoxicity Assays
Xenograft cells were retrieved from cryostorage, washed with complete RPMI, and resuspended in complete QBSF at a cell concentration previously optimized for each xenograft (1–5 × 106 cells/mL), and 100 µL was seeded in 96-well clear, U-bottom tissue-culture–treated plates (In Vitro Technologies, Noble Park North, VIC, Australia). Plates were equilibrated at 37 °C, 5% CO2, for 12 to 16 h prior to compound treatment ( Fig. 1A ). Compounds were serially diluted in complete QBSF medium and added in triplicate wells. Following 48 h incubation at 37°C, 5% CO2, cell viability was assessed by mitochondrial activity assay (resazurin cell viability assay, 6 h incubation) or by flow cytometry (ViaCount reagent). Cell viability was calculated as a percentage of untreated controls (unless otherwise stated). Results presented are the mean ± standard error of the mean of a minimum of two independent experiments. IC50 values were calculated from cumulative survival curves.
In Vitro Fixed-Ratio Combination Cytotoxicity Assays and Assessment of Combination Index
Xenograft cells were retrieved from cryostorage, washed with complete RPMI, and resuspended in complete QBSF at a cell concentration previously optimized for each xenograft (1–5 × 106 cells/mL), and 100 µL was seeded in 96-well clear, U-bottom tissue-culture–treated plates (In Vitro Technologies). After overnight equilibration, cells were treated with compound and dexamethasone simultaneously at a fixed-ratio of concentrations corresponding to ¼, ½, 1, 2, and 4 times the IC50 values for each drug (unless otherwise stated) independently and in combination. Cytotoxicity was assessed using resazurin cell viability assay or flow cytometry (as previously described). The fraction affected was calculated for each drug concentration, and data were subsequently analyzed by the median-effect method using CalcuSyn software (Biosoft, Cambridge, UK). 27 Combination Index (CI) values were calculated for each drug combination, with the assumption that the drugs act on distinct targets, where synergy is defined as a CI <0.9, additivity as CI 0.9 to 1.1, and antagonism as CI >1.1. 27
Results
HTS Assay Development and Validation
A number of factors were considered when designing the HTS assays. A final concentration of 1 µM dexamethasone was used in the assays, as this concentration resulted in a clear difference in activity between dexamethasone-resistant and -sensitive ALL xenografts. Although a higher dexamethasone concentration may have resulted in more hits, these would be less clinically applicable. Generally, HTS compounds are screened at 1 to 50 µM; however, solubility issues can frequently occur at higher compound concentrations. Both HTS compounds and dexamethasone were dissolved in DMSO. Although this unfortunately increased the final concentration of DMSO in the well to 0.4% (v/v), the pintool used to transfer 100 nL of each compound is most accurate when transferring nonaqueous liquids. The maximum DMSO concentration tolerated by xenograft cells in culture was determined to be 0.8% (v/v), and the DMSO concentration was maintained at ≤0.4% (v/v) in all experiments.
Xenograft cells were plated in serum-free medium to avoid false hits, which frequently occur in HTS assays because of compound-serum interactions. 28 Multiple resazurin cell viability assay incubation times were tested in the 384-well assay format, and an incubation time of 24 h was required because of the limited proliferative ability of xenograft cells in culture and low seeding density employed (ALL-19 cells seeded at 1 × 106 cells/mL, usually 2 × 106 cells/mL). The initial fluorescence (0 h read) of each well was deducted from the final fluorescence (24 h read), as this was deemed more accurate then subtracting an averaged blank from all wells. Outer wells of the assay plate were not included in the HTS because of potential evaporation effects.
To evaluate the reproducibility of the HTS assay, ALL-19 cells were plated and half of the wells were treated with 1 µM dexamethasone (negative control, 100% signal), whereas the other half were treated with 1 µM dexamethasone and 1 µM daunorubicin (positive control, 0% signal). Well-to-well within-plate, plate-to-plate within-day, day-to-day, and cell stock-to-cell stock variability were all assessed; the minimum Z-factor achieved was 0.78, which represents a highly robust assay. To further validate the HTS assay, a pilot screen was performed twice on different days and with different cell stocks. ALL-19 cells were screened with 1000 compounds (10 µM) in the presence of 1 µM dexamethasone, under the same conditions as the primary HTS, and the same hits were observed in both assays.
HTS for the Identification of Dexamethasone Sensitizers
The xenograft ALL-19 was derived from an aggressive and chemoresistant pediatric BCP-ALL that induced early fatality in the patient and was used to develop an HTS assay for identifying novel compounds that reverse dexamethasone resistance. ALL-19 cells are resistant to dexamethasone at concentrations up to 850 µM (data not shown), whereas ALL-3 cells are derived from a chemosensitive BCP-ALL and are sensitive to subnanomolar concentrations of dexamethasone (IC50 = 12 nM). 16 In the primary HTS assay, ALL-19 cells were exposed in vitro to dexamethasone at a concentration ineffective in cell killing against this xenograft (1 µM) but that is about 100-fold greater than the IC50 for sensitive xenografts, simultaneously with 1 of 40,000 HTS compounds (10 µM; Fig. 1B ). A fluorescent readout for cell viability (resazurin, also known as Alamar Blue) was chosen as a greater spectral range is achieved using fluorescence than with absorbance-based viability assays.
As xenograft cells display limited proliferative capacity in culture, cells were seeded at a relatively high density of 50,000 cells/well. Xenograft cells are expanded by passage through immune-deficient mice, and cells are harvested from the spleen. 17 Because of the large volume of cells required to carry out the HTS campaign, cells were pooled from multiple spleens to create a number of ALL-19 cell stocks. These cell stocks were mycoplasma free and validated using single nucleotide polymorphism arrays. In addition, each pooled ALL-19 cell stock and nonpooled ALL-19 cells (control) were tested in vitro against a panel of commonly used chemotherapeutics (dexamethasone, daunorubicin, vincristine, and staurosporine), and all cell stocks showed similar responses to all chemotherapeutics.
A number of controls were included per assay plate. The negative control (100% signal) consists of ALL-19 cells treated with 1 µM dexamethasone, which defines the higher limit of the assay. The positive control (0% signal) consists of ALL-19 cells treated with 1 µM dexamethasone and 1 µM daunorubicin, which defines the lower limit of the assay, as 1 µM daunorubicin kills 100% of ALL-19 cells. These 2 controls provided the dynamic range of the assay and allowed the Z-factor to be calculated per assay plate. Cellular assays with a Z-factor ≥0.5 are sufficiently robust for single measurements.25,26 Each assay plate displayed a Z-factor of ≥0.75; thus, the HTS assay was sufficiently robust.
As ALL-19 cells are resistant to dexamethasone, a control was required to ensure that dexamethasone was added to each well uniformly. A dexamethasone control plate consisting of dexamethasone-sensitive ALL-3 xenograft cells, half of the wells treated with 1 µM dexamethasone (kills 100% of cells) and half treated with an equivalent amount of DMSO (control), was included at the beginning and end of each batch run. Primary HTS assays were conducted in batches of 20 assay plates, including a dexamethasone control plate at the end of each batch run that ensured the dexamethasone stock remained active during the entire batch.
Screened compounds were assessed for their ability to reduce cell viability at 10 µM in the presence of 1 µM dexamethasone. The percentage cell viability was calculated for each compound relative to the dexamethasone-treated control (negative control); thus, the minimal effect of 1 µM dexamethasone on ALL-19 cell viability was corrected for. The primary HTS assay resulted in 420 compounds that reduced cell viability by ≥20% (
Figs. 1B
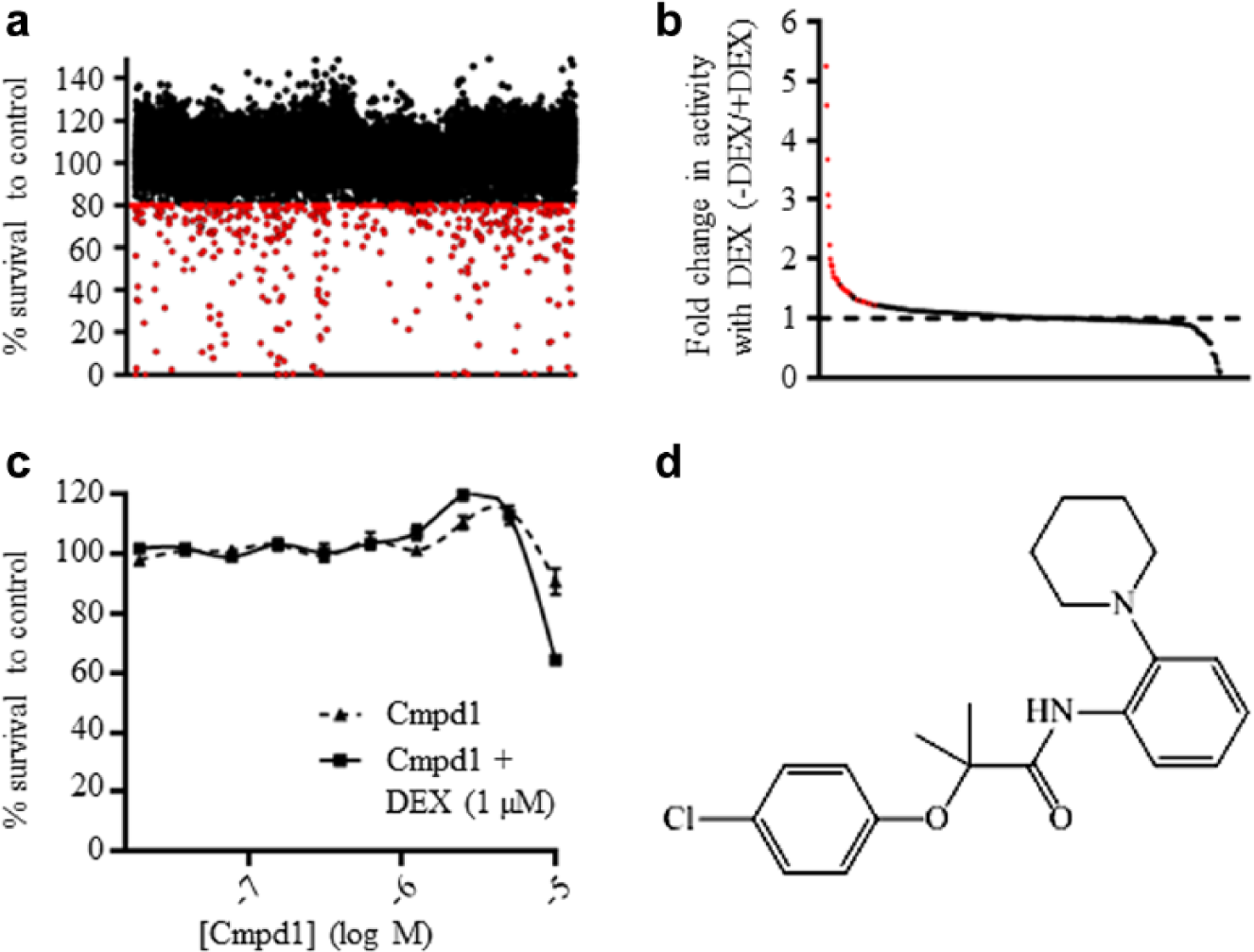
Identification of novel dexamethasone sensitizers by high-throughput screening (HTS). (
As expected, most compounds displayed the same activity ± dexamethasone ( Fig. 2B ). Potential dexamethasone sensitizers were defined as compounds that showed an additional reduction in cell viability in the presence of 1 µM dexamethasone of at least 15% or compounds with a ratio of percentage cell viability – dexamethasone/percentage cell viability + dexamethasone >1 standard deviation from the mean ( Fig. 2B ). This second cutoff allowed the inclusion of compounds that may not have shown a large reduction in cell viability + dexamethasone at 10 µM but display a large fold change in activity. For example, a compound with 16% cell viability – dexamethasone and 4% cell viability + dexamethasone displays a fourfold increase in activity with dexamethasone but only a 12% reduction in cell viability. Potent compounds (≥80% reduction in cell number ± 1 µM dexamethasone) were also included in the tertiary HTS assay, as dexamethasone sensitization could be masked due to high toxicity at 10 µM. The secondary screen resulted in 56 potential dexamethasone sensitizers and 60 potent compounds ( Fig. 1B ).
The 116 compounds were then retested on ALL-19 cells in triplicate in a dose response (20 nM – 10 µM) ± 1 µM dexamethasone. None of the 60 potent compounds were dexamethasone sensitizers at low concentrations. This tertiary HTS assay served two purposes: to confirm the secondary HTS results and to assess the extent of dexamethasone sensitization. The tertiary HTS assay identified seven dexamethasone sensitizers; Cmpd1 (2-(4-chlorophenoxy)-2-methyl-N-(2-(piperidin-1-yl)phenyl)propanamide) showed a modest dexamethasone-sensitizing effect at higher concentrations ( Fig. 2C ). The structure of Cmpd1 is shown in Figure 2D .
Confirmation of Dexamethasone Sensitizer Cmpd1
The seven hit compounds identified from the HTS assay were reordered from their suppliers and retested in standard 96-well cytotoxicity assay conditions. We could not reacquire one compound because of resupply issues. Of the six retested compounds, only four were found to have a dexamethasone-sensitizing effect similar to that observed in the tertiary HTS. Further experiments focused on Cmpd1 (2-(4-chlorophenoxy)-2-methyl-N-(2-(piperidin-1-yl)phenyl)propanamide) ( Fig. 2D ). The structure and purity of Cmpd1 were verified using nuclear magnetic resonance (NMR) spectroscopy and liquid chromatography–mass spectrometry (LC-MS).
The order of addition experiments confirmed that sensitization to dexamethasone was maximal when cells were treated simultaneously with Cmpd1 and dexamethasone (
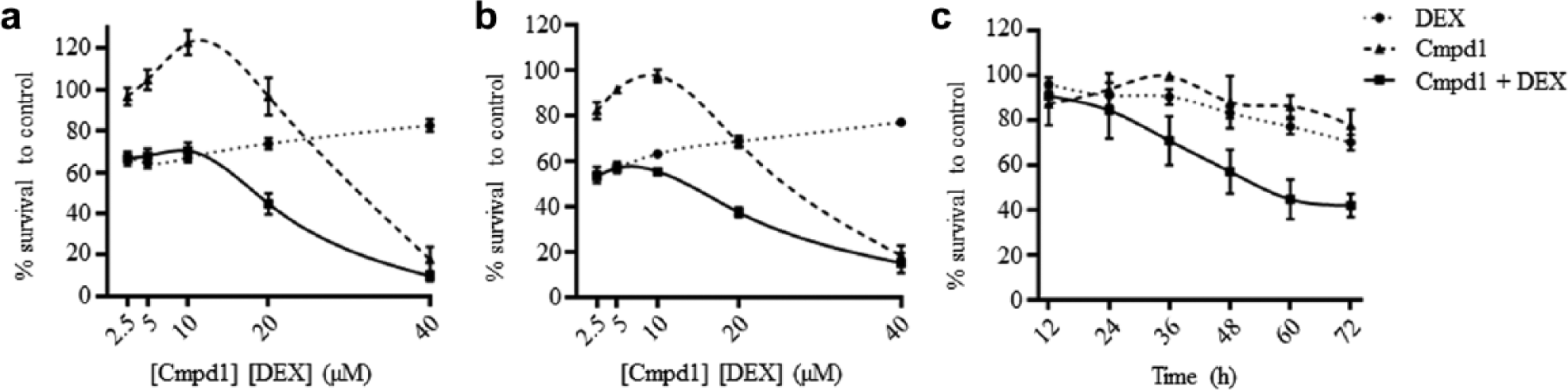
Synergistic antileukemia effects of Cmpd1 and dexamethasone in vitro against acute lymphoblastic leukemia xenograft cells (ALL-19). (
Characteristics of the xenografts used in this study and their responses to Cmpd1 and dexamethasone.
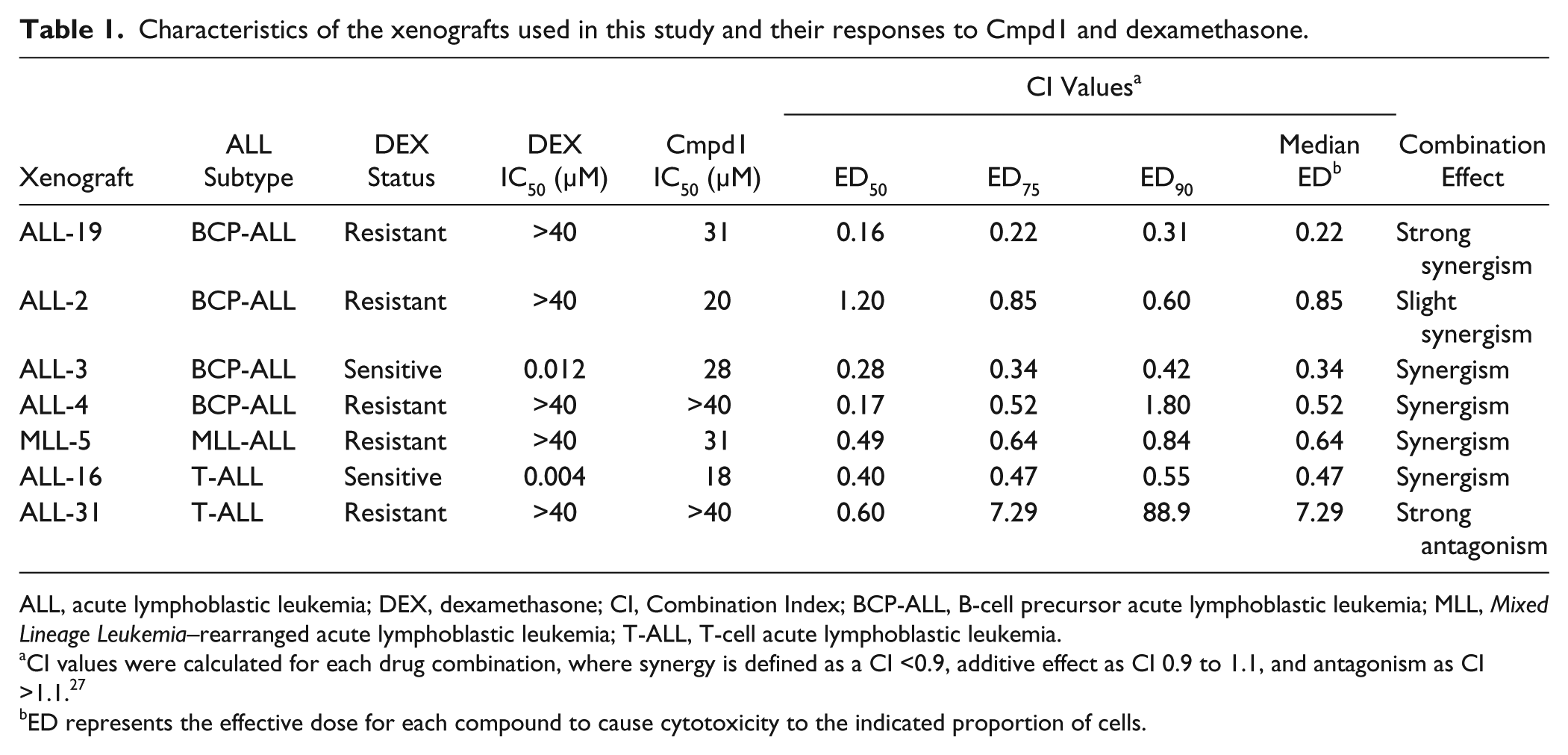
ALL, acute lymphoblastic leukemia; DEX, dexamethasone; CI, Combination Index; BCP-ALL, B-cell precursor acute lymphoblastic leukemia; MLL, Mixed Lineage Leukemia–rearranged acute lymphoblastic leukemia; T-ALL, T-cell acute lymphoblastic leukemia.
CI values were calculated for each drug combination, where synergy is defined as a CI <0.9, additive effect as CI 0.9 to 1.1, and antagonism as CI >1.1. 27
ED represents the effective dose for each compound to cause cytotoxicity to the indicated proportion of cells.
To assess the temporal relationship of interactions between Cmpd1 and dexamethasone, we next performed a time course experiment in which cell viability was assessed at various times following exposure of ALL-19 cells to fixed concentrations (20 µM) of Cmpd1 and dexamethasone, alone and in combination. The combination caused a marked decrease in cell viability compared with the single agents alone, with <50% viable cells remaining after combination treatment for 60 h ( Fig. 3C ).
Applicability of Dexamethasone Sensitizer Cmpd1
To determine whether Cmpd1 was broadly active or if it selectively sensitized only ALL-19 cells to dexamethasone, fixed-ratio combination cytotoxicity assays were performed against an additional six xenografts. The panel consisted of dexamethasone-sensitive and -resistant xenografts, representative of BCP-ALL, T-cell ALL (T-ALL), and Mixed Lineage Leukemia–rearranged ALL (MLL-ALL;
Fig. 4
;
Table 1
;
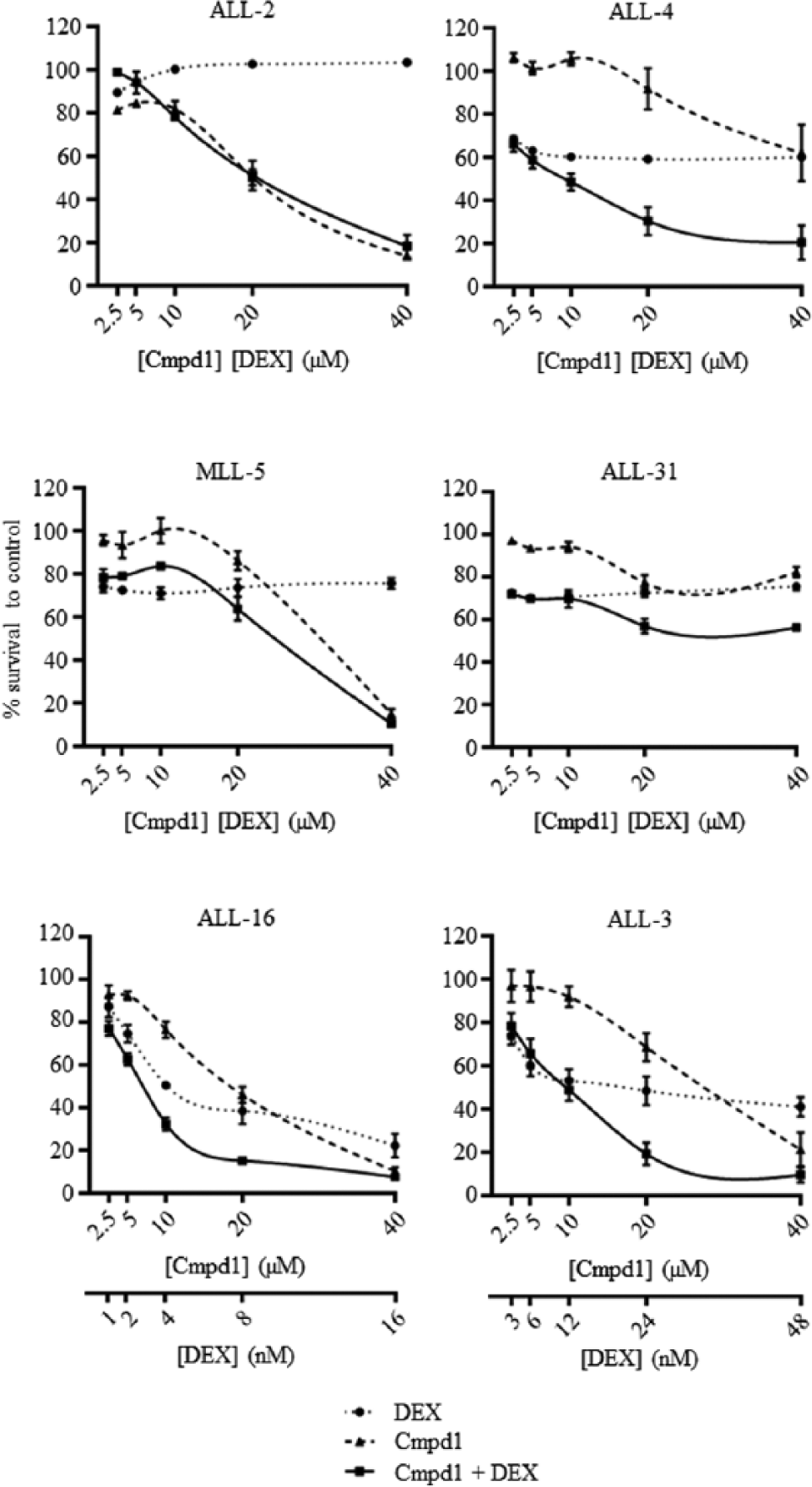
Cmpd1 exhibits broad dexamethasone-sensitizing effects against acute lymphoblastic leukemia (ALL) xenograft cells. Xenograft cells were exposed to Cmpd1, dexamethasone, or both in combination at a fixed ratio of concentrations for 48 h, and cell viability was then assessed by resazurin assay. IC50 values were calculated from cumulative survival curves. Each data point represents the mean ± SEM of three independent experiments.
Synergy between Cmpd1 and dexamethasone was observed in all four BCP-ALL xenografts tested, which included a dexamethasone-sensitive xenograft (ALL-3) and three dexamethasone-resistant xenografts derived from patients who succumbed to their disease (
Fig. 4
;
Table 1
;
Discussion
We have previously reported that the in vitro and in vivo dexamethasone responses of pediatric ALL xenografts significantly correlated with the clinical outcome of the patients from whom they are derived.16,17 Therefore, it is likely that the mechanism of compounds found to reverse dexamethasone resistance in vitro in dexamethasone-resistant xenografts will be relevant to patients who present with dexamethasone-resistant leukemia. The focus of this study was to develop an HTS assay that could identify compounds that sensitize dexamethasone-resistant ALL xenografts to dexamethasone, to confirm these findings, and to determine if the hit compound showed wider applicability.
Although it is known that glucocorticoids (e.g., dexamethasone) induce apoptosis in lymphoid cells, the signaling pathway remains incompletely defined, and the lesions underlying resistance remain poorly understood. For this reason, a cell-based assay was chosen for the screen, as this allows for selection of compounds with no prior bias of molecular targets and that are cell permeable. By performing a cell-based HTS assay, entire pathways of interest were probed, which provides the opportunity for identifying novel molecular targets.
The novel HTS strategy was designed to maintain the appropriate biological context of dexamethasone resistance while balancing feasibility in terms of number of cells used and adaptation to automation. A dexamethasone-resistant ALL xenograft (ALL-19) was employed in the HTS assay as, unlike most glucocorticoid-resistant leukemia cell lines, the mechanism of resistance in ALL xenografts appears more relevant to the clinical disease setting.11,14,15 The primary HTS assay identified compounds that reduced cell viability in the presence of dexamethasone. It was not until the secondary screen, in which the active compounds were rescreened ± dexamethasone, that we were able to determine if any compounds were dexamethasone sensitizers. It would have been ideal to test the compounds ± dexamethasone in the primary screen; however, this would have required double the number of cells and reagents. Unlike cell lines, xenografts have limited proliferative capacity in culture and are expanded by engraftment in immune-deficient mice, and human leukemia cells are harvested at a high level of purity (usually >90% 17 ) from the spleens of highly engrafted mice. Also, because of their limited proliferative capacity, xenograft cells must be seeded at considerably higher densities than cell lines, typically 1–5 × 106 cells/mL. Thus, the HTS strategy was designed to conserve the use of xenograft cells while being able to screen at least 40,000 compounds.
A number of controls were included to ensure confidence in the HTS assay and robustness. The Z-factor was calculated per assay plate, and with a minimum Z-factor of 0.75 achieved, the HTS assay was sufficiently robust for single measurement analysis.25,26 Additional control plates seeded with dexamethasone-sensitive xenografts cells and tested ± dexamethasone were included at the beginning and end of each batch run to ensure that dexamethasone was added evenly to the test plates (as ALL-19 is dexamethasone resistant) and remained active throughout the entire batch run. To further validate the primary HTS design, two pilot screens of 1000 compounds were conducted on separate days and with different cell stocks and yielded the same hits. The retesting HTS assays were also designed to confirm the results of each previous assay. In the primary screen, 40,000 compounds were tested at 10 µM + 1 µM dexamethasone. In the secondary screen, the 420 hits were tested again at 10 µM + 1 µM dexamethasone but also at 10 µM without dexamethasone. In the third and final screen, the 116 hits were tested at 10 µM ± 1 µM dexamethasone as in the secondary screen, however they were also tested at a range of other concentrations. The hit rates were 1%, 13%, and 13% for the primary, secondary, and tertiary screens, respectively.
The seven hit compounds identified in the tertiary screen to be more effective in the presence of dexamethasone at a range of concentrations were reordered from their suppliers and retested. Of the six we were able to reacquire, only four were found to have dexamethasone-sensitizing effects when retested. This is a common occurrence in HTS assays and can be due to compound degradation or compound labeling error. 28 Although we were able to confirm the structure and purity of the reordered compounds using NMR and LC-MS, we did not perform any such confirmations on the library compounds used in the HTS assay as these compounds are received predissolved in DMSO.
We further characterized the dexamethasone-sensitizing ability of the hit compound, Cmpd1, 2-(4-chlorophenoxy)-2-methyl-N-(2-(piperidin-1-yl)phenyl)propanamide. Fixed-ratio combination assays with Cmpd1 and dexamethasone on ALL-19 showed strong synergism (median CI = 0.22). ALL-19 is derived from a relapse BCP-ALL, which induced early fatality in the patient. 16 The observation of strong synergy between Cmpd1 and dexamethasone in ALL-19 is highly encouraging, as previous compounds found to reverse dexamethasone resistance in other ALL xenografts have shown dexamethasone additivity at best against ALL-19. 20 The combination also showed slight synergism (median CI = 0.85) against ALL-2, also a dexamethasone-resistant relapse BCP-ALL xenograft that induced early fatality in the patient.16,17
The combination of Cmpd1 and dexamethasone was also tested against 2 BCP-ALL xenografts derived from diagnosis samples, ALL-3 and ALL-4. Although ALL-3 is a dexamethasone-sensitive xenograft derived from a patient who remains in complete remission, the addition of Cmpd1 further potentiated the effects of dexamethasone, with a synergistic effect observed (median CI = 0.34). ALL-4 is derived from a very-high-risk subtype of BCP-ALL that is Philadelphia chromosome–positive (Ph+), which has a poor outcome, even though patients are placed on high-risk treatment protocols. 29 ALL-4 is dexamethasone resistant, and the combination was synergistic in this xenograft (median CI = 0.52), causing >80% reduction in cell number in combination, but <50% cell reduction as single agents at the highest concentration tested. Therefore, Cmpd1 shows broad applicability, inducing synergy when in combination with dexamethasone against all four BCP-ALL xenografts tested.
The combination was then tested against two T-ALL xenografts, ALL-16 and ALL-31. T-ALL patients are assigned as a high-risk subgroup, regardless of dexamethasone sensitivity status. 3 However, the survival of T-ALL patients is now comparable with BCP-ALL patients, but intensive treatment is required. 29 The combination of Cmpd1 and dexamethasone showed synergism (median CI = 0.47) against the dexamethasone-sensitive T-ALL xenograft, ALL-16. Unfortunately, the combination displayed antagonism (median CI = 7.29) in the dexamethasone-resistant T-ALL xenograft, ALL-31. However, the combination did show some dexamethasone sensitization in ALL-31 at a lower ED value, and the high median CI value could be attributed to the combination curve plateauing at higher concentrations. The combination was also synergistic (median CI = 0.64) against a dexamethasone-resistant, infant MLL xenograft, MLL-5. This was a positive finding as infants with MLL-ALL are assigned as high risk and have an inferior outcome, with 5-year event-free survival rates of approximately 35%.3,29,30
A somewhat surprising finding was that Cmpd1 exerted its best synergistic effects with dexamethasone when administered simultaneously. Although investigation into the mechanism of action of Cmpd1 is required, it is plausible that Cmpd1 blocks the efflux of dexamethasone from the cell, therefore displaying best synergistic effects when administered simultaneously with dexamethasone. It is unlikely that Cmpd1 is an epigenetic modifier, as pretreatment would enhance the synergistic antileukemic efficacy with dexamethasone. 27 In conclusion, using a clinically relevant xenograft model in a cell-based screen, we have identified a novel compound that potentiates the cytotoxicity of dexamethasone against aggressive and glucocorticoid-resistant xenografts. Future development of this novel class of compound may lead to its use as a molecular tool to further define mechanisms of glucocorticoid action, as well as to enhance its potential to reverse glucocorticoid resistance in lymphoid malignancies.
Footnotes
Acknowledgements
Children’s Cancer Institute Australia for Medical Research is affiliated with the University of New South Wales and the Sydney Children’s Hospitals Network.
Declaration of Conflicting Interests
The authors have declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a grant from the National Health and Medical Research Council. R.B.L. is supported by a Fellowship from the National Health and Medical Research Council. C.E.T. is supported by a UNSW Research Excellence Award from the University of New South Wales.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

