Abstract
RNA-protein interactions are vital to the replication of the flaviviral genome. Discovery focused on small molecules that disrupt these interactions represent a viable path for identification of new inhibitors. The viral RNA (vRNA) cap methyltransferase (MTase) of the flaviviruses has been validated as a suitable drug target. Here we report the development of a high-throughput screen for the discovery of compounds that target the RNA binding site of flaviviral protein NS5A. The assay described here is based on displacement of an MT-bound polynucleotide aptamer, decathymidylate derivatized at its 5′ end with fluorescein (FL-dT10). Based on the measurement of fluorescence polarization, FL-dT10 bound to yellow fever virus (YFV) MTase in a saturable manner with a Kd = 231 nM. The binding was reversed by a 250-nucleotide YFV messenger RNA (mRNA) transcript and by the triphenylmethane dye aurintricarboxylic acid (ATA). The EC50 for ATA displacement was 1.54 µM. The MTase cofactors guanosine-5′-triphosphate and S-adenosyl-methionine failed to displace FL-dT10. Analysis by electrophoretic mobility shift assay (EMSA) suggests that ATA binds YFV MTase so as to displace the vRNA. The assay was determined to have a Z′ of 0.83 and was successfully used to screen a library of known bioactives.
Introduction
The Flaviviridae, including yellow fever virus (YFV), dengue virus (DENV), and West Nile viruses (WNV), are a serious public health threat worldwide, with at least 50 million infections annually from the four dengue serotypes alone. Currently, vaccines exist only for YFV, tick-borne encephalitis virus (TBE), and Japanese encephalitis virus (JEV). In the absence of a fully protective dengue vaccine 1 and with millions at risk of infection, there remains a critical need for the development of effective chemotherapeutics for both dengue and other flaviviral infections.
The flaviviral genome is organized as a (+)-strand RNA encoding three structural proteins and seven nonstructural proteins. Current strategies and targets for the development of flaviviral inhibitors are summarized.2–7 Notable targets include the viral protease/helicase activities of (NS2B/NS3) and the methyltransferase/polymerase activities of NS5. The viral genome contains a virally methylated 5′–cap 1 structure, necessary for messenger RNA (mRNA) stability and efficient translation. 8 Using S-adenosyl-L-methionine (SAM) as the methyl donor, the MTase domain catalyzes the methylation of cap (GpppA) genomic RNA to form cap 1 (m7GpppAm) at the 5′ end of the flaviviral genome. The NS5 methyltransferase is able to methylate at both the guanine N-7 9 and adenosine 2′-O 10 positions sequentially, first at N-7 followed by 2′-O. 11 Inhibition of N-7 or 2′-O methylation in WNV results in virus that is nonreplicative or attenuated. 11 DENV deficient in cap methylation also results in attenuated or nonreplicative virus.12,13 The MTase also contains guanylyltransferase (capping) activity. 14 The requirement for methylation of the cap of the viral genome therefore makes NS5A an attractive target for discovery of antiviral drugs.
Inhibition of methylation can occur when blocking guanosine-5′-triphosphate (GTP) binding, which includes inhibition of capping activity, SAM binding, or binding of the viral RNA (vRNA) substrate. Recent assays for the discovery of inhibitors of the flaviviral MTases include those that target the GTP binding and capping activities15,16 and those based on inhibition of enzymatic activity using both short capped PolyC ribo-octanucleotides, 17 as well as longer 74–base pair (bp) vRNA transcripts. 18 The use of fluorescein 3′ end–labeled five-base RNAs to examine RNA binding showed negative effects on binding by GTP, with weak positive effects by SAM. 19 For reasons not yet understood, the use of short, capped RNA substrates limits analysis of cap methylation to 2′-O-methylation only. To assay N-7 methylation, longer vRNA transcripts are required. 18
The inhibitor of nucleic acid binding, aurintricarboxylic acid (ATA), was first identified as a potent inhibitor of DENV and WNV MTases by means of in silico screening. 20 Here we describe a simple high-throughput assay for discovery of inhibitors of flavivirus MTases in which fluorescein-labeled dT10 (FL-dT10) bound to the YFV MTase is used as a surrogate for the vRNA for the discovery of a new class of flaviviral inhibitors, compounds with vRNA displacement activity. This high-throughput assay works without the immediate need for radioactivity-based enzymatic reactions and is suitable for miniaturization. The assay can be deployed as both a primary assay for the discovery of inhibitors that displace the vRNA from the MTase or as a secondary assay for mechanistic, descriptive, and confirmatory studies of inhibitors discovered by other means.
Materials and Methods
Reagents
ATA, SAM, S-adenosyl-L-homocysteine (SAH), and GTP were purchased from Sigma-Aldrich (St. Louis, MO). FL-dT10 (5′ FAM-TTT-TTT-TTT-T-OH 3′) was synthesized by IDT, Inc. (Coralville, IA).
MTase Purification
The DNA sequence that specifies the first 296 amino acids of YFV NS5 (17D) was cloned into pet28B with an N-terminal poly His tag, and protein was expressed in the BL21 Rosetta strain from Invitrogen (Madison WI). Cultures were induced with 0.5 mM isopropyl β-D-1-thiogalactopyranoside for 6 h at room temperature (RT), pelleted, and frozen. Frozen pellets were resuspended in lysis buffer and sonicated. Following centrifugation to remove insoluble material, the enzyme was purified by affinity chromatography on Ni++ resin from Thermo Scientific (Madison, WI). Protein was dialyzed against methyltransferase buffer (50 mM HEPES [pH 7.2], 1 mM MgCl2, and 6 mM KCl) and stored at −80 °C until use. WNV and DENV type 2 MTases were expressed in the M15 expression system from Qiagen (Valencia, CA) and purified as described above.
Fluorescence Polarization Assay
Fluorescence polarization (FP) assays were performed in 20 mM HEPES (pH 7.2) and 5 mM MgCl2, using purified YFV MTase protein, and 10 nM FL-dT10 in a total volume of 30 µL dispensed into 384-well black microtiter plates. Binding saturation experiments were performed by dilution of purified YFV MTase serially into MTase buffer containing FL-dT10. Plates were incubated at 37 °C for 15 min and read on a Tecan Safire2 fluorescence microtiter plate reader (Tecan, Männedorf, Switzerland). For experiments containing small molecules, 1 µL of a 30× stock of compound was added to the wells prior to addition of the reaction mixture. Determination of EC50 for ATA was performed as described above, using 500 nM YFV MTase. For FP experiments containing vRNA, 80 ng of YFV RNA transcript representing the first 250 nucleotides of the 5′-terminal YFV genome was added to each test well. WNV and DENV MTase were used at 500 nM for binding experiments.
Capped RNA Substrates
Capped RNA in vitro transcripts for methyltransferase assays were generated by a procedure similar to that described by Ray et al. 9 Briefly, capped RNA substrates representing the first 250 nucleotides of the 5′-terminal YFV genome were transcribed in vitro from polymerase chain reaction (PCR) products containing a bacteriophage T7 class II φ2.5 promoter that allows an adenosine triphosphate (ATP)–initiated transcript using T7 RNA polymerase. 21 In vitro YFV RNA transcripts were capped in the presence or absence of SAM with α-[32P]-GTP and Vaccinia virus capping enzyme from CellScript (Madison, WI) to generate GpppA-RNA and m7GpppA-RNA. The resulting capped RNA was purified through a G-50 column from GE Healthcare (Madison, WI), followed by an RNeasy column obtained from Qiagen prior to use in methylation assays.
Cap Methylation Assays
The methylation reaction was performed at 37 °C for 1 h in MTase buffer, containing 5000 cpm GpppA-YFV RNA, 1.0 µM YFV MTase, and 100 µM SAM. For testing of inhibitors, MTase was preincubated with ATA for 10 min at 37 °C, and reactions were initiated by the addition of RNA and SAM. Methylation reactions were digested for 1 h at 68 °C with Nuclease P1 (US Biological, Swampscott, MA), and the resultant cap modification products were analyzed by polyethyleneimine cellulose thin-layer chromatography in 0.2 M ammonium sulfate.
Electrophoretic Mobility Shift Assay
Electrophoretic mobility shift assay (EMSA) analysis was performed by incubating 1 µM YFV MTase and 5000 cpm cap RNA representing the first 250 bp of the YFV genome with ATA and SAH for 30 min at 25 °C in 10 mM Tris (pH 9.0) and 1 mM MgCl2. After incubation, samples were fractionated on a 7% polyacrylamide gel in 100 mM Tris (pH 9.5) and 10 mM NaCl as buffer. After electrophoresis was complete, the gel was analyzed by autoradiography for RNA binding to MTase.
Bioactives Screen
Screening of the National Institutes of Health (NIH) clinical collection was performed as described for FP assays above, using 500 nM YFV MTase, 10 nM FL-dT10, and library test compound, 33 µM. After dispensing 30-µL reaction mixtures to microtiter plate wells, library compounds were added to the wells by pin transfer of 100 nL. Plates were incubated at 37 °C for 30 min and read on a Tecan Safire2 for fluorescence measurements. ATA was used at a concentration of 10 µM as positive control for displacement of FL-dT10.
Z′ Analysis
The Z′ of the assay was calculated using the equation 1 – [3(SD1 + SD2)]/|µ1 – µ2| to compare untreated and ATA-treated wells. 22 In this formula, |µ1 – µ2| represents the absolute value of the difference between the average measurement of FP in the test and control reactions, and (SD1 + SD2) represents the sum of the standard deviations of the same two measurements. Under the conditions of the assay, FP was measured with and without test compound in a reaction containing 500 nM YFV MTase and 10 nM FL-dT10, incubated alone or with 4 µM ATA for 30 min at 37 °C; 30 wells were used for each condition tested.
Results and Discussion
The diverse critical roles played by MTases make them attractive targets for the discovery of compounds that differentially modulate their activity. To date, no MTase inhibitors have been successfully developed into effective therapeutics. Here we describe an assay for the discovery of inhibitors of the nucleic acid binding site of the MTase, representing an alternate avenue for the discovery of effective nucleic acid MTase inhibitors.
Decathymidylate (dT10), a YFV RNA Surrogate Probe to Test the RNA Binding Site of YFV MTase
In undertaking the present studies, we have validated displacement of a surrogate ligand in the form of a short DNA sequence that occupies the vRNA binding site on the MTase. The selection of dT10 as a probe was based on the observed action of phosphocellulose as a pseudo-affinity medium for the absorption of DNA- or RNA-modifying enzymes.23–26 We asked whether dT10, based on its phosphate content, might specifically bind to the YFV capping MTase via the RNA binding site. Oligo dT10 adopts a random coil configuration, with minimal ability to assume a secondary structure; it represents approximately one helical turn. Decathymidylate was derivatized at its 5′ end with fluorescein to form the probe, FL-dT10, that was then tested for its ability to bind to YFV MTase. Complex formation between FL-dT10 and YFV MTase as a function of added protein was monitored by FP, with results shown in Figure 1A . The Kd for FL-dT10 was 231 ± 12 nM, calculated directly as a function of FL-dT10 binding using nonlinear regression analysis. The Kd of binding of the FL-dT10 is similar to that described for the binding of five-base RNA by DENV, WNV, and YFV MTases. 19 Comparison of DENV, WNV, and YFV MTases for FL-dT10 binding in Figure 1B shows that DENV and WNV MTases are also capable of binding FL-dT10. The assay described above establishes that a negatively charged DNA oligonucleotide can act as a surrogate for the vRNA substrate.
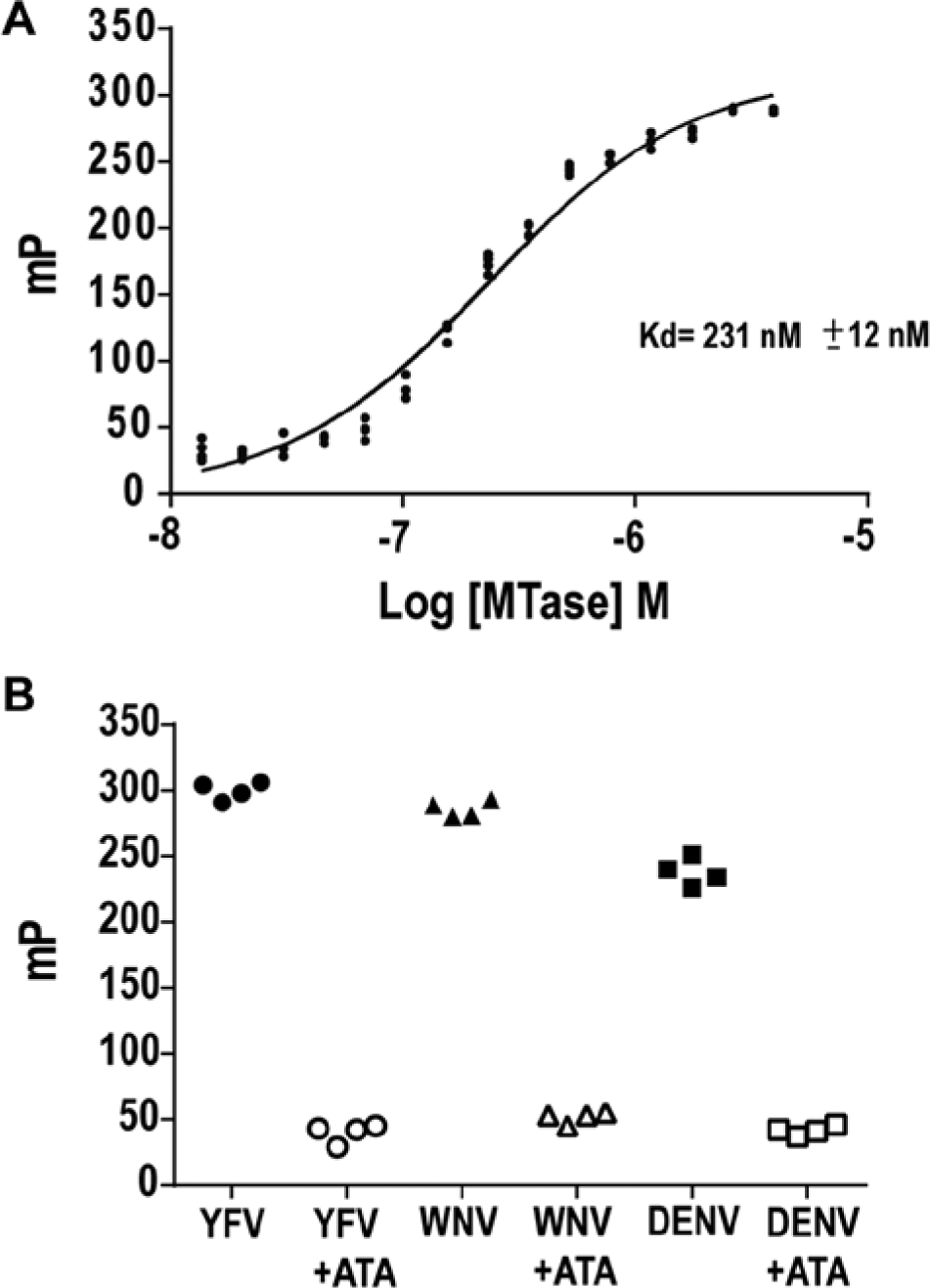
Binding of FL-dT10 to yellow fever virus (YFV) MTase measured by fluorescence polarization (FP). (
YFV MTase uses both the YFV RNA and GTP together, with the guanylyltransferase activity inherent in NS5A, as substrates to form a cap (GpppA) at the 5′-end of the vRNA. This reaction is followed by methylation of the cap, with SAM as the methyl donor, first on guanine to form cap 0 (N-7MeGpppA), followed by methylation on the 2′-O of adenosine to form cap 1 (N-7GpppA2′-O). To learn whether the MTase ligands GTP and SAM displace FL-dT10, we incubated YFV MTase with FL-dT10 to form complexes in the presence of known ligands as competitors. Results, shown in
Figure 2A
, indicate that the complex is disrupted by ATA (a known nucleic acid binding protein inhibitor)27–31 but not by GTP or SAH, suggesting that FL-dT10 selectively interacts with the binding site for vRNA and does not affect the binding sites for GTP and SAM/SAH. The ability of ATA to disrupt the complex is mimicked by YFV RNA, as shown in
Figure 2B
. Interference from GTP on fluorescein 3′ end–labeled five-base RNA binding and a weak positive effect on binding in the presence of SAM were noted previously.
19
Owing to the need for longer RNAs for N-7 methylation, and in the absence of defined RNA binding sites on the MTase, we propose that FL-dT10 may only partially overlap with the binding site of these short RNAs. Fixation of the FL-dT10 to YFV MTase confirmed that the site of FL-dT10 binding was critical for MTase activity (
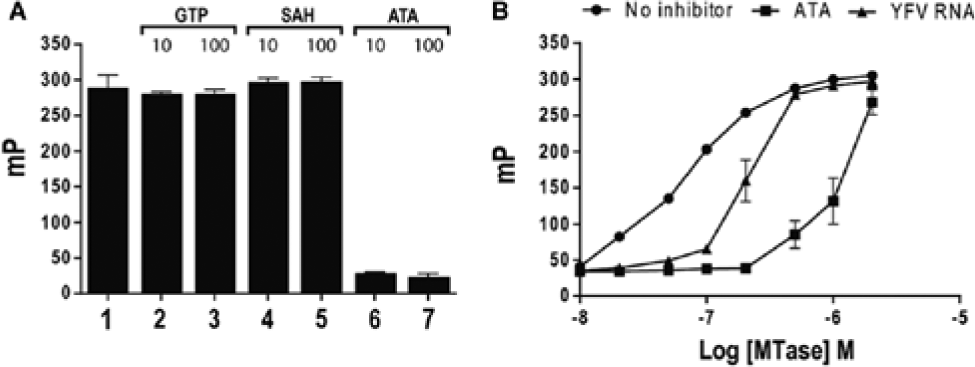
Specificity of FL-dT10 displacement by MTase ligands. (
ATA, a Small-Molecule Inhibitor of Protein Nucleic Acid Interactions
ATA has been described as generally inhibiting nucleic acid–protein interactions. ATA was also shown to inhibit the DENV and WNV MTases, with a proposed binding site linked to the KDKE RNA-binding motif. 20 We therefore tested the ability of ATA to disrupt FL-dT10–YFV MTase complexes. Results, shown in Figure 3 , indicate that ATA disrupted the binding of FL-dT10 to YFV MTase with an EC50 = 1.54 ± 0.16 µM, which is close to the IC50 for ATA inhibition of human DNA MTases DNMT1, 0.68 µM, and DNMT3a, 1.4 µM. 32 In addition, the IC50 for ATA inhibition of short capped RNA 2′-O methylation was 2.3 ± 0.3 µM for DENV MTase and 4.2 ± 0.2 µM for WNV MTase. 20
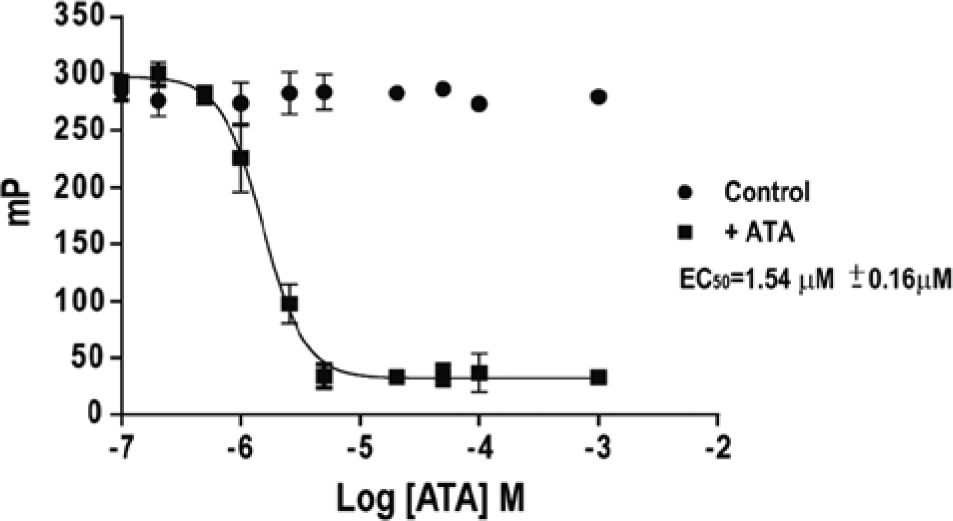
Displacement of FL-dT10 from yellow fever virus (YFV) MTase by aurintricarboxylic acid (ATA). The EC50 for destabilization of FL-dT10-YFVMTase complexes by ATA was determined by fluorescence polarization and calculated using nonlinear regression analysis.
We next determined whether YFV MTase–vRNA bound complexes are also sensitive to the disruptive effects of ATA. [32P]-GTP-labeled cap YFV RNA was incubated with YFV MTase, resulting in the formation of MTase-RNA complexes. Reactions containing the formed complexes were supplemented with ATA, and the resultant products of the reaction were fractionated by polyacrylamide gel electrophoresis (
Fig. 4
). As shown in
Figure 4
, lane 1 contains [32P]-GTP–labeled cap YFV RNA alone, while lane 2 displays the retardation of the RNA by added YFV MTase in the absence of inhibitor. Lanes 3 to 5 show the breakdown of the complex by addition of 10, 50, and 100 µM ATA. In contrast, 10, 50, and 100 µM SAH (lanes 6–8) failed to destabilize the complex. Experiments performed with 0.1-µM and 1-µM concentrations of ATA did not show displacement of the YFV RNA from the MTase, with displacement of RNA occurring at 10 µM ATA (
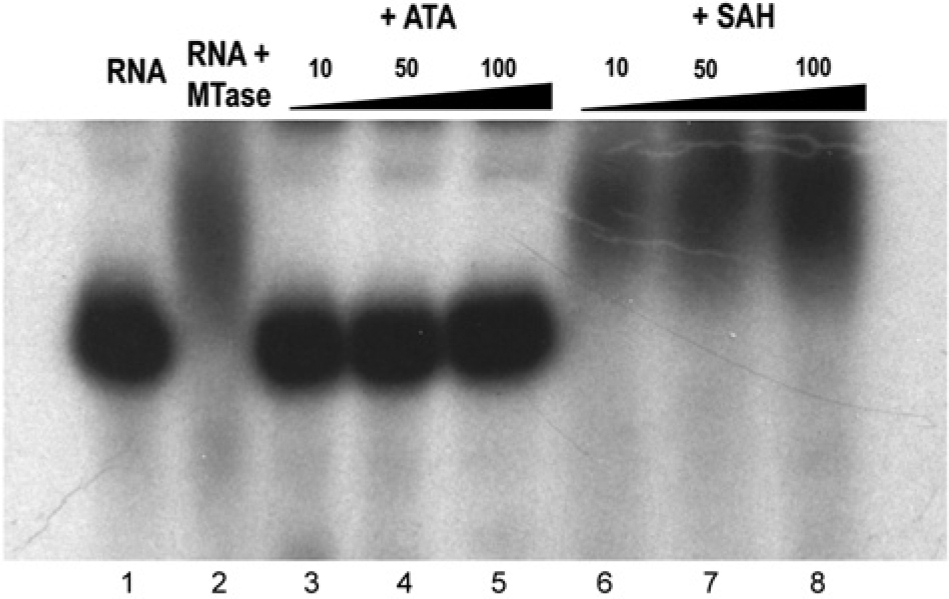
Displacement of yellow fever virus (YFV) RNA from YFV MTase by aurintricarboxylic acid (ATA) or S-adenosyl-L-homocysteine (SAH). Destabilization of YFV RNA-YFV MTase complexes by ATA. The YFV RNA was incubated with YFV MTase and supplemented with increasing concentrations of ATA or SAH as shown. Reaction mixtures were analyzed by polyacrylamide gel electrophoresis gel electrophoresis followed by autoradiography. Lane 1, YFV cap RNA; Lane 2, cap RNA + MTase (complete); lanes 3–5, complete plus 10, 50, and 100 µM ATA, respectively; lanes 6–8, complete plus 10, 50, and 100 µM SAH, respectively.
To determine the functional significance of the FL-dT10 binding studies described, we tested inhibition of the enzymatic activity of YF MTase by ATA. Test samples containing MTase, [32P]-GTP–labeled cap RNA, and SAM were assembled. The capped vRNA comprised the 250-nt 5′UTR region of the YFV genome. Results are shown in Figure 5 . The same inhibitory reagents that displace FL-dT10 from YFV MTase were shown to inhibit its enzymatic activity, further validating the use of FL-dT10 as a surrogate ligand for the vRNA. As shown in Figure 5 , ATA inhibited both N-7 and 2′-O methylation at approximately the same concentration of 12.5 µM. The IC50s for ATA inhibition were previously reported for WNV to be 4.2 µM for 2′-O and 38 µM for N-7, as well as 2.3 µM and 127 µM for DENV. 20 However, these values were collected with two different lengths of RNA substrates. It would be interesting to know whether WNV and DENV show a similar IC50 profile for both N-7 and 2′-O when using only long RNA substrates, as shown here with YFV MTase. The need for higher concentrations of ATA for inhibition of longer RNA substrates is consistent with the polymeric binding characteristics of ATA. Taken together, these data support a mechanism of ATA fully displacing or competing for binding with the vRNA on the MTase. The studies reported above support the use of FL-dT10 as a convenient surrogate probe to replace vRNA in a high-throughput screen to discover YFV MTase inhibitors.
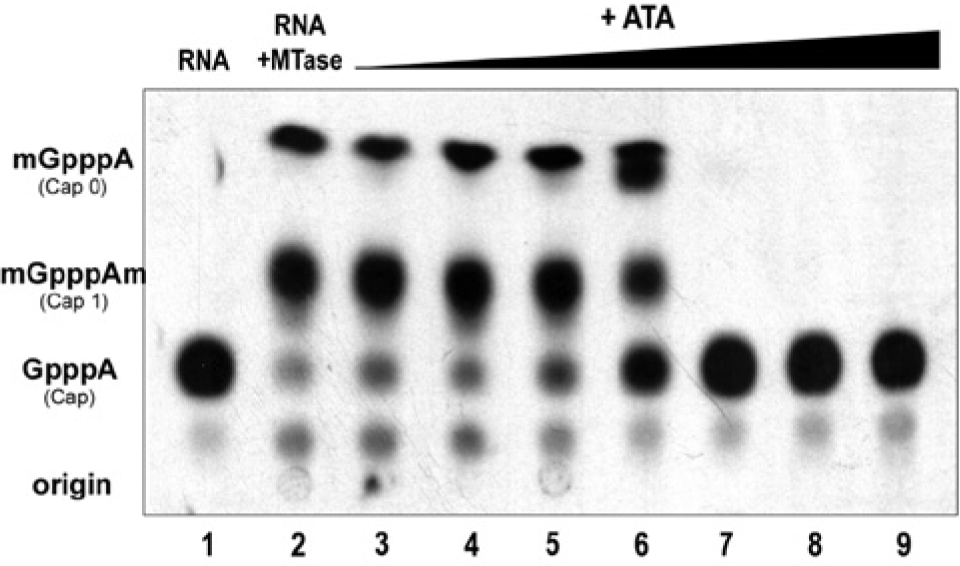
Inhibition of yellow fever virus (YFV) MTase activity by aurintricarboxylic acid (ATA). N-7 and 2′-O activity of YFV MTase was measured in the presence of ATA. The methylation reaction was allowed to proceed for a sufficient time to obtain significant cap 1 (N-7 and 2′-O methylation). ATA was added to the reaction at the concentrations indicated. The IC50 was obtained by densitometry and nonlinear regression analysis. The IC50 for ATA was determined to be 12.5 ± 2 µM. Data shown represent three independent experiments. Lane 1, [32P]-GTP labeled cap; lane 2 complete, cap 0 and cap 1 formed by the YFV MTase from labeled cap; lanes 3–9 complete, supplemented with 1, 2, 5, 10, 20, 50, and 100 µM ATA, respectively.
High-Throughput Screen of the NIH Clinical Collection
To validate the FP assay for use as a high-throughput discovery screen, we calculated a Z′ using 4 µM ATA as our control inhibitor. This analysis resulted in a Z′ of 0.83 (
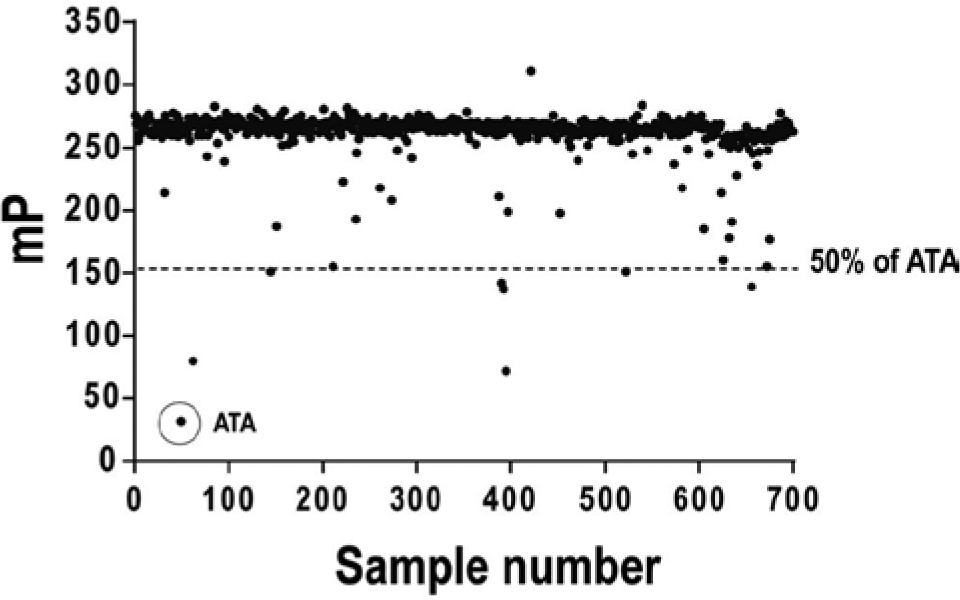
High-throughput screen of the National Institutes of Health Clinical Collection. The test reaction contained 500 nM MTase and 10 nM FL-dT10. In total, 30 µL of test reaction per well was dispensed into 384-well plates and supplemented with 100 nL library test compound added by pin transfer. Fluorescence polarization was determined and plotted as a function of sample number. ATA, aurintricarboxylic acid.
Multiple screening hits involving cephalosporins and benzofurans were obtained, suggesting that upon suitable modification, they might be repurposed for the treatment of flaviviral infections. Taken together, these data establish the use of a fluorescein-labeled dT10 as a suitable surrogate ligand for the YFV vRNA. Although this assay does not directly measure MTase activity, it is well suited to HTS and also represents a new way to screen for inhibitors of nucleic acid binding proteins. DNA methyltransferases have been considered targets for cancer chemotherapy.33–35 It will be interesting to learn the extent to which the primary assay described here will be adaptable to the discovery of other nucleic acid–modifying enzymes.
Footnotes
Acknowledgements
We thank the Small Molecule Screening Facility (SMSF) at the University of Wisconsin Madison for technical assistance with HTS of the NIH Clinical Collection. We thank Rob Striker in the Department of Medicine at the University of Wisconsin Madison for WNV and DENV MTase expression clones.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded, in part, by grant 5R03AI079638 from the National Institutes of Health.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
