Abstract
Gamma amino butyric acid receptors (GABA) are major therapeutic targets for the development of drugs in neurological and psychiatric disorders. The new generation of GABAA modulators is targeting subtype selectivity and low/partial efficacy on the receptor to potentially overcome the adverse effects described for drugs with full agonist profile. We evaluated a screening approach to measure the relative efficacy of GABAA positive allosteric modulators (PAM) using automated patch clamp and fluorescence membrane potential assays. We determined that the use of an internal comparator (zolpidem), tested on each cell in parallel to the test compound, provides a reliable approach to measure and compare the relative efficacy of PAM ligands. Patch clamp recordings on recombinant GABAA receptors, using a multiple drug addition protocol, allows us to rank PAM ligands with different levels of efficacies. We observed that fluorescence membrane potential assays are not predictive of the relative efficacies of GABAA PAM ligands.
Introduction
Gamma amino butyric acid (Gaba) is a major neurotransmitter released in the central nervous system (CNS) and contributes to the inhibition of neuronal networks in the CNS. The GABA A receptors are complex pentameric proteins, composed of a variety of subunits (α1-6, β1-3, γ1-3, δ, ϵ, π, θ), which are differentially expressed in the CNS.1,2 Activation of GABA A receptors by the endogenous agonist Gaba triggers an inward chloride current that hyperpolarizes the cell and contributes at the synaptic level to the generation of inhibitory postsynaptic currents. GABA A receptors have been the key focus of drug discovery efforts over the past decades, and major pharmacological tools have been developed for therapeutic use in epilepsy, depression, anxiety, and sleep disorders.2–4 Benzodiazepines are a major drug class that act as positive allosteric modulators (PAMs) of the Gaba site by binding at the interface between the α and γ subunit of GABA A receptors.5,6
The chronic activation of GABAA receptors by use of agonists or PAMs leads to major tolerability issues and abuse liabilities, which often limit the clinical use of such drugs. To overcome these issues, recent strategies have focused on the development of drugs with low relative intrinsic efficacy or that selectively potentiate a specific GABA A receptor subtype to a partial level. 4 Such compounds may either act through the agonist site or bind to the allosteric site and should potentially alleviate the adverse effects mediated by the use of classical benzodiazepines. The functional characterization of such compounds can be achieved by recording GABAA currents in recombinant systems using standard patch clamp recordings. 7 However, the identification of novel GABA A modulators involves the screening of large chemical libraries and consequently the development of appropriate high-throughput screening assays. Different methods have been described that allow for the functional screening of GABA A receptors including 36Cl− flux, 8 I− influx,9,10 fluorescence resonance energy transfer assay, 11 FLIPR assay, 12 and automated patch clamp. 13 Although these screening methods have been successfully used to identify and to determine GABAA modulator potencies, only a few have so far studied the accurate measurement of the relative efficacy of the GABAA modulators. Analyzing the relative efficacy of GABAA PAMs requires appropriate experimental protocols in which agonist concentration, reference compound, and GABAA receptor expression are monitored. In this study, we describe the design of an experimental approach to screen for the relative efficacy of GABAA modulators using an automated patch clamp system. In addition, we investigate whether the relative efficacy could be determined by using a high-throughput fluorescence membrane potential (FMP) assay.
Materials and Methods
GABAA Cell Line
The human GABAA receptor subunits α1 (NM_000806), β2 (accession number NM_021911), and γ2S (accession number BC_059389) have been stably expressed in a CHO-K1 cell line using the Fugene 6 (Roche, Basel, Switzerland) transfection protocol.
CHO-K1 cells, stably expressing the α1/β2/γ2S subunits of the human GABAA receptor, were cultured using standard culture media Ham’s F-12 with glutamine (GIBCO, Carlsbad, CA) containing 10% fetal bovine serum (FBS; Lonza, Basel, Switzerland), 1% penicillin + streptomycin (Lonza), and 400 μg/mL G418 (GIBCO) + 250 μg/mL Zeocin (Invitrogen, Carlsbad, CA). Cells were passaged using trypsin EDTA 0.05% solutions and plated at 10,000 cells/cm2 or 5000 cells/cm2 density in a T-75 flask (Sarstedt, Nümbrecht, Germany) 2 to 3 days before use on the automated patch clamp platform (PatchXpress 7000A; Axon Instrument, Union City, CA). The day of the experiment, cells were harvested using Accumax treatment (Sigma, St. Louis, MO) and allowed to recover for 90 min at room temperature before being used on the PatchXpress system.
Electrophysiology Recordings
GABAA receptor chloride currents were recorded using the automated patch clamp platform PatchXpress 7000A (Axon Instrument). During all procedures, the holding potential was set to −60 mV. Whole-cell compensation was automatically set before each experiment. Current traces were recorded by patch clamp amplifier (Multiclamp 700A Computer-Controlled Patch-Clamp Dual Headstage Amplifier, Axon Instruments) at a sampling rate of 2 KHz. Recordings were performed at room temperature (~25 °C). Data analysis was performed using DataXpress 2 software (version 2.0.4.2; Molecular Devices, Sunnyvale, CA).
Extracellular solution contained (in mM) 135 NaCl, 4.7 KCl, 2.5 CaCl2, 1.2 MgCl2, 11 glucose, and 10 HEPES (adjusted at pH 7.4 with NaOH 5N, ~290 milliosmolar). Intracellular solution contained (in mM) 100 KCl, 40 KF, 2 MgCl2, 11 EGTA, 10 glucose, and 10 HEPES (pH 7.2 with KOH 1N, ~295 milliosmolar).
The potentiation of GABAA-evoked chloride currents in the presence of drug was determined and compared with the maximum potentiation in the presence of 1 µM zolpidem. The procedure for sequential application of 5 µM Gaba alone or co-applied with drugs is shown in Figure 1A . First, the vehicle DMSO 0.5% is tested followed by 2 rapid applications (45 µL/s) of 5 µM Gaba (G1, G2) for 5 s at an 80 s interval. After each application, Gaba is washed out for 30 s with extracellular solution. The cells are subsequently incubated in the presence of 1 µM zolipem for 1 min followed by one coadministration of Gaba 5 µM + zolpidem 1 µM (Z+G3) to evoke maximal chloride currents. Cells are subsequently washed with extracellular solution to remove zolpidem followed by a new application of 5 µM Gaba (G4). The cells for which G4 responds to 80% to 120% of G2 are included into the analysis. The effects of test compounds alone are evaluated during the 5 min incubation to record baseline chloride currents followed by a coapplication of test compound and 5 µM Gaba (C+G5) to record maximal GABAA current potentiation.
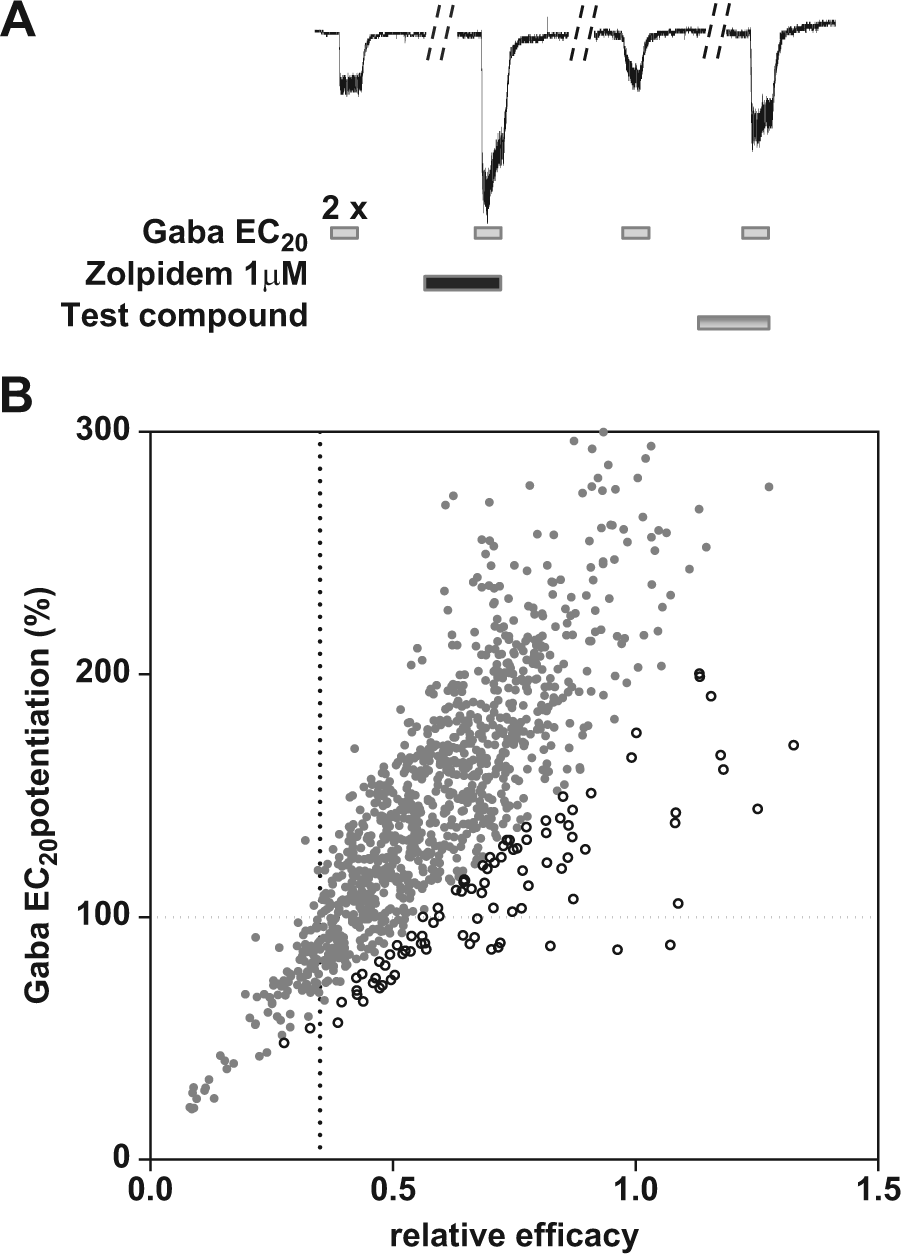
(
The relative efficacy is calculated as the ratio of the maximal current amplitudes evoked in the presence of test compound and zolpidem [(C + G5)/(Z + G3)].
Validation criteria for each tested cell were as follows: (1) current amplitude higher than 150 pA, (2) stable leak current ≤200 pA, and (3) potentiation of Gaba response by zolpidem >180% [(Z + G3)/G2].
Half-maximal effective concentration (EC50) values were determined by nonlinear regression analysis using a sigmoidal dose-response (variable slope) equation (GraphPad software version 5 for windows, San Diego, CA).
Fluorescence Measurements
GABAA receptor activation was measured using an FMP dye (Molecular Devices) as previously described. 14 Cells were seeded into 96-well clear-bottom plates the day before the experiment at a density of 35,000 cells/well. The cell culture media were removed and replaced with 180 µL of Hanks balanced salt solution (HBSS) containing Hepes 20 mM pH 7.4 and FMP dye. Test compounds were prepared in HBSS Hepes 20 mM pH 7.4 (1% DMSO), and 20 µL were added to the cell plate (in triplicate) and incubated for 30 min at room temperature. Each test plate contained the positive controls zolpidem (1 µM) and bretazenil (0.1 µM). Basal fluorescence (F0) was recorded in a Flexstation reader (Molecular Devices) using an excitation and emission wavelength of 545 nm and 572 nm, respectively. Gaba (20 µL) was added with a Multidrop dispenser (Thermo Scientific, Waltham, MA) system and incubated for 6 min at room temperature before final fluorescence (Ffinal) measurement. The addition of Gaba to the GABAA receptor cell line induces an increase of the FMP dye fluorescence response that reaches maximal levels after 6 min. The Gaba EC20 value was checked before each experiment using a dose range of 10 nM to 100 µM. Fluorescence data were normalized using the equation F = (Ffinal − F0)/F0.
All data are represented as mean ± standard deviation (SD). Power analyses have been performed with SAS 9.2 Software.
Results and Discussion
Automated Patch Clamp Studies
In this study, we describe an automated patch clamp protocol to determine the relative efficacy of PAMs of the GABAA receptor in order to rank ligands with various relative efficacies and especially compounds with low potentiation levels such as bretazenil. Such compounds are often referred as “partial” PAMs (or wrongly termed partial agonists), and efficacy has been determined in the absence 15 or in the presence of a comparator, 7 which makes direct comparison quite difficult. For the screening of GABAA agonists, a maximal dose of Gaba is generally used to calculate functional efficacy, whereas for the screening of positive allosteric modulators, compounds acting on the benzodiazepine site, Gaba concentrations at EC5 to EC20 should be considered. Indeed, the cellular response to Gaba (e.g., Gaba at EC20) is dependent on GABAA receptor expression levels and receptor combinations (different subtypes or αβ vs. αβγ containing receptors), and such heterogeneity may be expected during expression of recombinant GABAA receptors. 13
In this study, we selected zolpidem as an internal comparator because of its medium GABAA affinity and its convenient washout properties. We evaluated the activity of zolpidem on the human α1β2γ2S GABAA receptor stably expressed in a CHO cell line by using the automated patch clamp system. Figure 2A shows the potentiation of GABAA currents by 1 µM zolpidem, inducing a leftward shift of the Gaba dose-response curve. Zolpidem produced GABAA potentiation with a pEC50 value of 7.7 ± 0.15 (n = 5). The potentiation by zolpidem (1 µM) is highest at Gaba EC20 and decreases at higher Gaba concentrations ( Fig. 2A ), and the analysis of 1776 patched cells indicates that zolpidem potentiates Gaba EC20 by 254% ± 68% ( Fig. 2B ). The response of zolpidem was also very stable over a large number of cell passages ( Fig. 2B ; passage 8–28), reflecting the stability of the GABA A receptor expression levels. Less than 5% of the cells showed a response inferior to 150%, most probably corresponding to cells with altered GABAA receptor pharmacology, and therefore we generally used a cutoff of 180% response (average − 1 SD) of zolpidem (1 µM) to validate the cells in our screening assay.
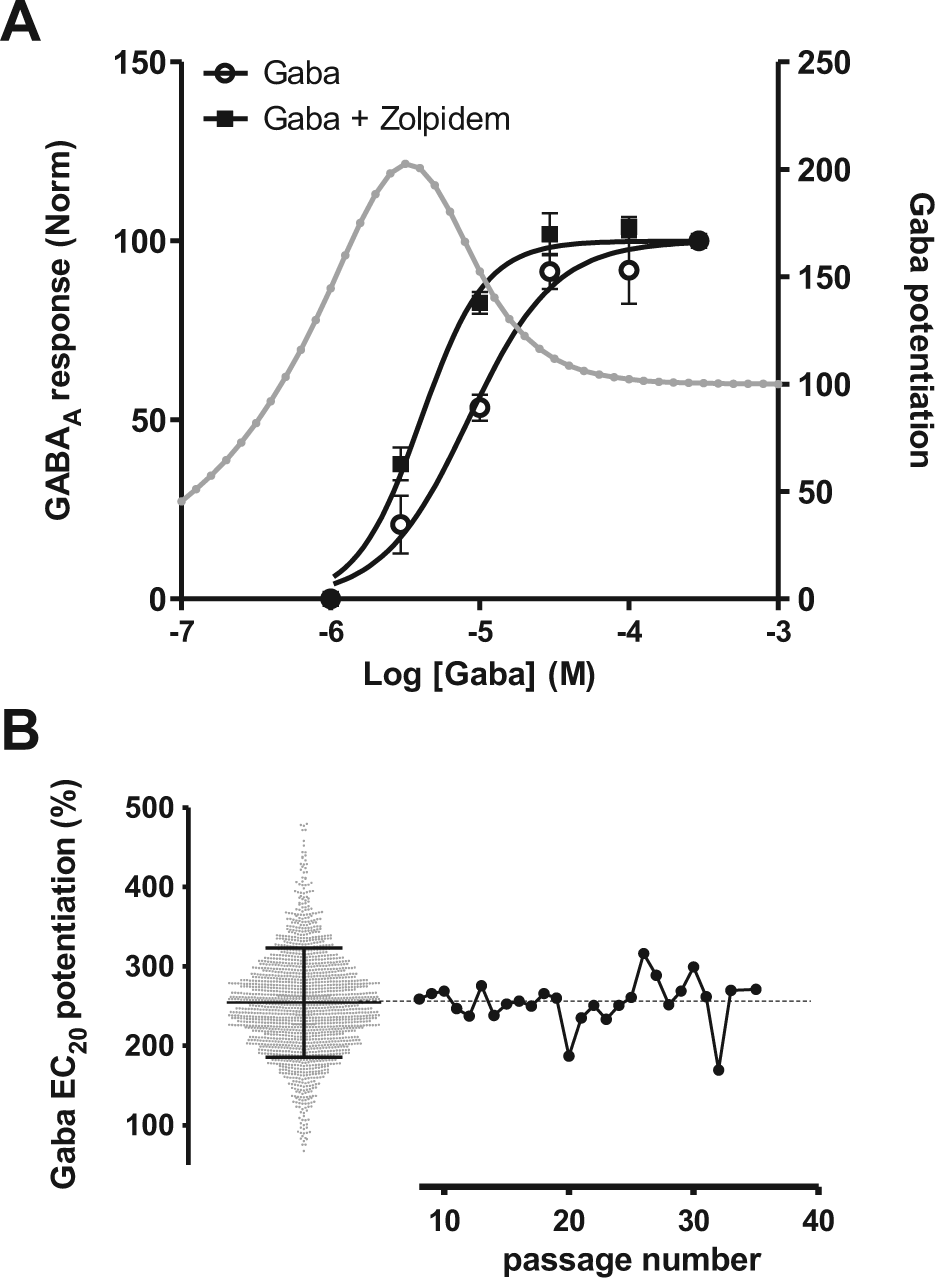
(
The relative efficacy of a PAM may vary from cell to cell based on receptor combination and expression levels, and consequently, the use of an internal comparator is key to normalize the measured potentiation values. We decided to develop a novel protocol in which the efficacy of a PAM will be calculated based on the maximal response of zolpidem (1 µM) for each cell.
Figure 1A shows the screening protocol that has been established to measure the relative efficacy of test compounds. First, two additions of Gaba at EC20 are added to the cells in order to establish a stable GABA A response. This step is followed by an incubation of the positive control zolpidem (1 µM, 1 min) and the potentiation with Gaba, which provides the maximal GABAA potentiation value of the cell. After washout of the positive control, a new Gaba EC20 is added to ensure return to control values followed by incubation of the test compound and a final potentiation with Gaba EC20. A preincubation step of 5 min with the test compounds should reduce the risk of underestimating the efficacy of ligands with slow binding kinetics. Coapplication is convenient when testing at rather high ligand concentrations, but it has been reported that, for example, abecarnil has slow kinetics 16 and therefore requires several minutes to produce its maximal effect.
We selected a set of GABAA reference compounds, all described to bind to the benzodiazepine site, and evaluated their relative efficacy to zolpidem (1 µM) in our PatchXpress protocol. All compounds were tested at a single dose, which produces maximal potentiation in our experiments, and their relative efficacy was calculated as described in the “Material and Methods” section. The control values in our experiments showed a ratio of 0.35 ± 0.07 (n = 12), which should not be interpreted as a potentiation effect of the vehicle but results from the contribution of the Gaba EC20 response. Bretazenil (0.1 µM) produces the lowest potentiation with a relative efficacy of 0.46 ± 0.09 (n = 52), in agreement with its described profile as a PAM with partial efficacy. 17 All other compounds showed relative efficacies between 0.61 and 0.82 (see Table 1 ). The effects of zolpidem in these experiments were quite reproducible, as shown by the relative efficacy of 0.96 ± 0.19 (n = 14) when tested against itself. The results provide a direct comparison between GABAA PAMs and show that different levels of efficacies can be measured using this protocol. Power analyses indicated that a minimum sample size of 14 is needed to reach the level of significance (Student t test, α = 0.05; two-tailed; power 80%, SD = 0.089) when the difference of relative efficacy is equal to 0.1 between two compounds. The relative efficacies of the ligands determined in our assay are in agreement with the ranking of potentiation levels previously determined in a chloride flux assay on GABAA and in which chlordiazepoxide was used an internal comparator. 7 Chlordiazepoxide, which produces similar potentiation levels at different alpha-containing receptors, has also been described as a good internal standard when it comes to the comparison of compound efficacies between different GABAA receptor subtypes. 7
Relative efficacy to zolpidem of GABAA PAM ligands in patch clamp experiments.
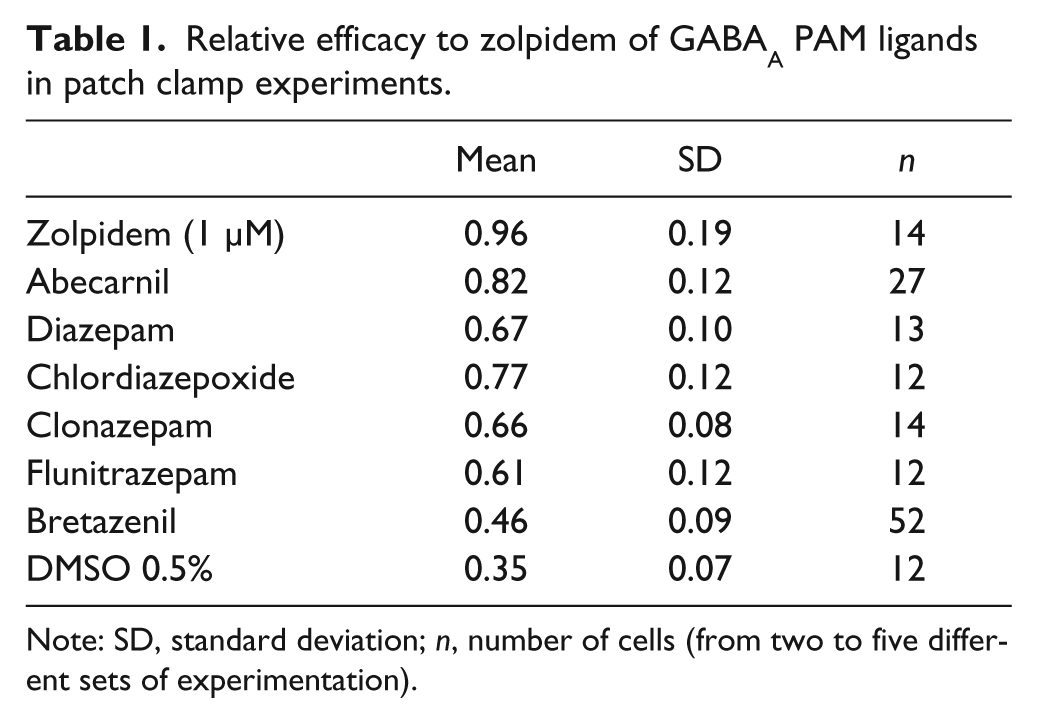
Note: SD, standard deviation; n, number of cells (from two to five different sets of experimentation).
To what extent do Gaba EC20 potentiation levels predict the relative efficacy of a PAM? To address that question, we compared the calculated relative efficacies (vs. zolpidem) with the Gaba EC20 potentiation levels obtained from a data set of ~1100 cells that have been measured during screening activities. Figure 1B shows the relationship between the calculated efficacy levels and Gaba EC20 potentiation values. We conclude that the two efficacy values correlate but only if the cells that show a low response to zolpidem (open circles) have been discarded. Indeed, the cells that show a response to zolpidem inferior to 180% have a general tendency to underestimate the efficacy levels when using Gaba EC20 potentiation measurements.
FMP Approach for the Screening of GABAA PAMs
We further evaluated the use of a different technology platform that should enable the screening of GABAA PAMs with a higher throughput. The screening of voltage- and ligand-gated ion channels has been facilitated by the introduction of FMP dyes. Such dyes respond to membrane potential changes induced by the channel activation, and the resulting fluorescence changes can be easily measured with standard fluorescence readers. Activation of the GABAA receptor cell line by Gaba leads to a strong and dose-dependent increase of the FMP dye fluorescence (EC50 ~1 µM), reaching a maximal level of 68% over baseline (data not shown). The GABAA response, when stimulated with a low dose of Gaba at EC20, is potentiated by a set of PAM references known to act at the benzodiazepine site ( Fig. 3A ). The pEC50 values of the reference drugs calculated from these experiments correlate with the pIC50 values measured from binding of the drugs to the [3H]flunitrazepam site in the rat brain membranes ( Fig. 3B ), confirming that the pharmacologic activity of the GABAA PAMs is reproduced in our stable cell line. Furthermore, the testing of a larger set of GABAA compounds, known to bind to the central benzodiazepine binding site, resulted in a good correlation (r2 = 0.75) between function and binding activity. However, we were interested to determine if the FMP assay may also predict the functional efficacy of a compound when compared with patch clamp data. We tested the effects of bretazenil, described as a PAM with low efficacy on GABAA, and observed that this drug potentiated the Gaba EC20 response to about half of the response of zolpidem (25% ± 7% vs. 52% ± 14%, n = 61).
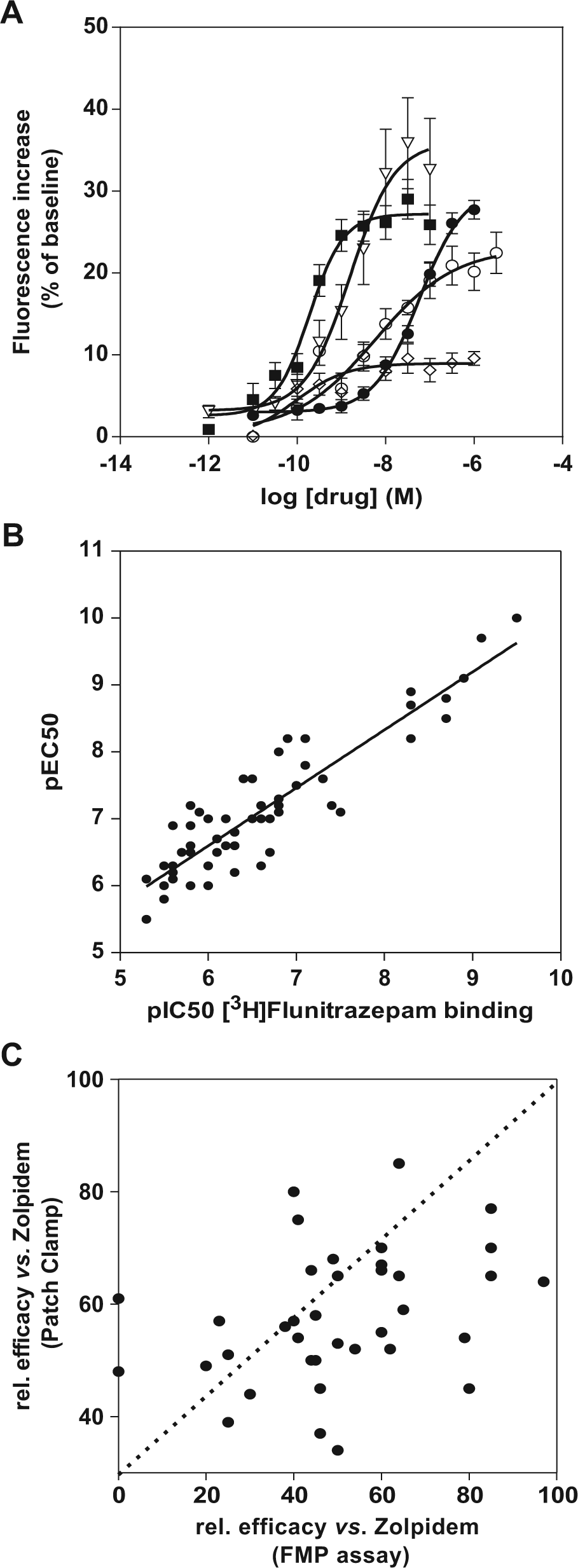
Fluorescence membrane potential assay on α1β2γ2S GABAA receptors stably expressed in a CHO cell line. (
We further evaluated the fluorescence approach for screening GABAA PAMs with the aim of establishing whether the measured efficacies are comparable to the data observed in patch clamp experiments. We selected a set of ~40 compounds that have been identified by [3H]flunitrazepam binding to rat brain membranes and determined their functional efficacy on GABAA receptors using the FMP assay. The relative efficacy (vs. zolpidem) was calculated based on the maximal response, and we then retested the compound at the same concentration using the PatchXpress platform. Figure 3C shows the relationship between relative efficacies, all normalized to the maximal response of zolpidem, measured by two different screening platforms. No correlation was observed between the two approaches, and some compounds were even completely inactive in the FMP assay, although they produced significant potentiation in patch clamp experiments ( Fig. 3C , data points on y axis).
The results indicate that the FMP assay may be suitable for screening the functional effects of GABAA PAMs acting at the benzodiazepine site but may not be a good platform to characterize and compare the efficacy of test compounds.
The protocol described in this study provides a first approach for the determination of PAM efficacy on GABAA receptors using automated patch clamp platforms. Although the study was limited to the testing of ligands acting at the benzodiazepine site, it would be interesting to further validate the approach with a larger and more diverse set of PAMs and to explore different GABAA receptor subtypes.
GABAA ligands still offer a large panel of therapeutic opportunities, and hence, the profile of agonists and PAMs in terms of binding affinity, receptor selectivity, and intrinsic efficacy needs to be carefully established. The automated patch clamp protocol that we characterized in this study should permit us to identify compounds with low, medium, or high efficacy and thus provide a rational way to select compounds with the desired profile.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
