Abstract
High-throughput screening (HTS) of 42 865 compounds was performed to identify compounds that inhibit formation of or kill Staphylococcus epidermidis RP62a biofilms. Three biological processes were assayed, including (1) growth of planktonic/biofilm bacteria, (2) assessment of metabolically active biofilm bacteria using a resazurin assay, and (3) assessment of biofilm biomass by crystal violet staining. After completing the three tiers (primary screening, hit confirmation, and dose-response curves), 352 compounds (representing ~0.8%) were selected as confirmed hit compounds from the HTS assay. The compounds were divided into groups based on their effectiveness on S. epidermidis biofilm properties. The majority of these affected both inhibition and killing of bacterial biofilm cultures. Only 16 of the confirmed hit compounds that have either an AC50 lower than 10 µM and/or Sconst ≥70 from those processed were selected for further study by confocal laser scanning microscopy (CLSM). The CLSM was used to evaluate the confirmed hit compounds on (1) inhibition of biofilm formation and (2) killing of preexisting S. epidermidis biofilms. Taken together, with further testing (e.g., disease-related conditions), such compounds may have applications as broad antimicrobial/antibiofilm use for prophylactic or therapeutic intervention to combat infections in surgical and intensive care clinics and battlefield settings.
Virtually all microorganisms and especially bacteria use multiple strategies and gene products for survival in myriad potentially hazardous environmental settings and also during human infection. One such bacterial tactic is the formation of complex, surface-attached communities known as biofilms. 1 Bacteria can form highly refractory biofilms on numerous environmental surfaces including piping, drains, sinks, toilet bowls, and other commonly used items. They also can cause microbial-induced corrosion that can affect the operation and structural properties of bridges, vehicles, ships, cannons, and so forth for either civilian or military use. Such biofilms are also notorious for causing a variety of highly problematic human disease states, contributing to an estimated 80% of all human infections. 2 During human disease, surface-attached biofilms (known as mode I biofilms 3 ) are commonly found on tubing, contact lenses, catheters, stents, prosthetic devices, bone, teeth, and a variety of human tissues and cells. Some common disease states associated with mode I biofilm infections include chronic otitis media, endocarditis, urinary tract infections, and oropharyngeal infections (e.g., plaque and gingivitis 4 ).
Bacterial biofilms consist of both viable and dead organisms enmeshed within a complex combination of protein, DNA, lipid, and carbohydrate that can serve as a protective shield in which the encased organisms can thrive while being sheltered from potentially detrimental and/or lethal conditions. In clinical settings, bacterial biofilms can be highly refractory to even the most potent antibiotic regimens, in part because of dramatic changes in bacterial metabolism, inadequate penetration of the antibiotic(s), and/or expression of either innate or acquired resistance genes. In addition, the polymeric matrix enhances the ability of biofilm bacteria to survive stresses such as temperature/pH fluxes, antibiotics, heavy metals, and many biocides typically used to kill bacteria. 1 Further, the enmeshed biofilm bacteria possess an innate ability to sense potentially toxic agents and invoke an active process known as biofilm dispersion of what are then termed planktonic (unattached or free-living) organisms. 1
Because of the myriad special challenges presented by bacterial biofilms in clinical, environmental, and industrial settings, eradication of such biofilm has been the research focus of thousands of academic scientists around the world. Biofilm-associated problems can be alleviated via three specific strategies: (1) preventing the attachment and initial growth of biofilm-forming microorganisms, (2) preventing the spread of biofilm bacteria by killing organisms enmeshed within the biofilm, and (3) disrupting preexisting highly refractory biofilms by triggering the process of dispersion (dispersed/exposed bacteria are more susceptible to most antimicrobial agents 5 ).
Staphylococcus epidermidis is the most common of more than 30 members of the genus Staphylococcus that typically constitute the majority of the human skin commensal microbial flora. However, depending on the organism and the immune status of the patient, certain strains can cause problematic hospital-acquired infections. 6 The organism synthesizes several biofilm-related factors, including polysaccharide intercellular adhesin (PIA), 7 Aap, 8 and SasG, 9 the latter two of which have G5 domains that possess zinc-dependent biofilm-forming properties. 10 S. epidermidis biofilms, similar to those of the related methicillin-resistant Staphylococcus aureus (MRSA; e.g., strain COL 11 ), are often highly refractory to the β-lactam class of antibiotics (e.g., methicillin, amoxicillin, etc.). 12
Given the aforementioned problems associated with S. epidermidis biofilms, we undertook a high-throughput screen (HTS) approach of a diverse library of about 40 000 compounds to identify the compounds that can be inhibiting and/or killing of the S. epidermidis biofilm culture. The experiment encompassed a multifactorial approach, examining multiple stages of attachment, growth, viability, and metabolism, a unique and unparalleled examination of nearly every aspect of biofilm development. The confirmed hit compounds were then also studied further by confocal laser scanning microscopy (CLSM). Collectively, this study was used to confirm the ability of compounds resulting from HTS and also to study further the potential efficacy in killing and/or dispersion of the preexisting biofilm cultures, studies that were not conducted in the HTS assay.
We are fully cognizant that the substrata and media used in this study do not closely mimic those of a human host, yet they do represent the first critical steps in the elucidation of compounds with the potential to one day offer hope for treatment of complications due to infections by this highly problematic opportunist bacteria.
Materials and Methods
Bacteria
Staphylococcus epidermidis RP62A (herein S. epidermidis) and a mutant defective for biofilm formation (herein termed M7) were routinely cultured in tryptic soy broth (TSB) from MP Biomedicals LLC (Salon, OH) prior to the HTS analyses described in detail below. All organisms were propagated from −80 °C frozen stocks maintained in 20% glycerol.
Chemical Library Used in These Studies
A total of 42 865 compounds were used in this study. The compounds were selected as a highly diverse representation of different classes of the University of Cincinnati Drug Discovery Center’s compound collection, containing more than 360 000 compounds. About 2000 compounds were specially selected from the UC compound collection because of their chemical similarity to known biofilm inhibitors and/or biocides.
HTS Assays
S. epidermidis was grown overnight in TSB media and then diluted 1:400 in TSB containing 0.25% glucose. Bacteria were inoculated into 384-well plates with or without 10 µM of each compound. One column of each plate contained only the S. epidermidis M7 biofilm-deficient mutant control, 13 and another served as the medium control, which has no test compound or bacteria.
Total Bacterial Growth
After incubation for 20 to 22 h at 37 °C without shaking, the optical density of the bacterial suspensions was recorded using the Plate::Vision plate reader (Evotech, Irvine, CA) equipped with a 560 BP filter (560 nm, 2.5% light intensity, 1 mm focus to bottom of plate, exposure time 100 ms). The “focus to bottom” is a parameter used to adjust the focus height of the Plate::Vision detector. This parameter was optimized during the assay development stages and validated to obtain the best results with the sample volume used in our studies.
Resazurin Metabolic Assay
After measurement of total bacterial growth, the plates were washed six times with 50 µL phosphate-buffered saline (PBS) from Sigma-Aldrich (St. Louis, MO) using a BioTeck Plate washer. After the addition of 50 µL of 2.5 mg/mL of resazurin solution from Sigma-Aldrich (St. Louis, MO) in 0.1X TSB, the plates were incubated for 50 min at room temperature. The fluorescence signal was measured using an Envision plate reader (PerkinElmer, Santa Clara, CA) with excitation and emission wavelengths of 535 nm and 590 nm, respectively.
Crystal Violet Staining Assay for Biofilm Formation
The resazurin solution from the above metabolic assays was removed by washing the plate wells three times with 50 µL PBS. Finally, 0.005% of crystal violet (CV) from Sigma-Aldrich was added to each well and incubated for 20 min at room temperature. The unbound CV to the bacteria was removed by washing three times with PBS. The bound CV was then dissolved in 50 µL of 95% ethanol, and the optical density (560 nm) was recorded using a Plate::Vision plate reader using the same setting for total bacterial growth measurements.
Primary Screening, Hit Confirmation, and Dose-Response Experiments
First, a total of 42 865 compounds were performed HTS as a single-point compound concentration of 10 µM for the primary screening., Next, all of the hit compounds were retested in triplicate for hit confirmation using the same concentration. The compounds from the hit confirmation studies were then used at 10 different concentrations (or points) for dose-response assessment (0.1–50 µM) measurements in triplicate. All experiments of primary screening, hit confirmation, and dose response were performed in three assay steps: (1) total bacteria growth, (2) resazurin metabolic assay, and (3) CV staining for biofilm formation, as mentioned previously. Finally, the confirmed hit compounds that were effective at inhibiting and/or killing the bacterial biofilm cultures were identified.
Data Analysis
Data calculations and normalization were performed using Genedata Screener Assay Analyzer Software (version 9.0.0 standard). For each of the three aforementioned assays, the measured results were normalized to the controls of each microtiter plate for data analysis. To determine dose responses, the Smart Fit Model (Genedata, Lexington, MA) was used for fitting the data derived from each compound and obtaining the dose-response curves. Sinf (infinite activity, i.e., the fitted activity level at infinite concentration) and S0 (zero activity, i.e., the fitted activity level with no compound) were set to −100 and 0, respectively. The AC50 value (µM) was defined as the concentration of compound that led to a 50% reduction in each assay (as compared with untreated controls) and was determined by Genedata Screener Condoseo Software.
Determination of Signal-to-Background Ratios and Z′ Values
The signal-to-background (S/B) ratios and Z′ values were calculated based on Zhang et al. 14
DMSO Tolerance
Because each compound was dissolved in DMSO, the effect of DMSO on the S. epidermidis RP62a biofilms was also investigated. The bacteria were grown in the presence of different concentrations of DMSO (0%–6%). Then, total bacteria growth, resazurin metabolic, and CV assays were performed as mentioned above.
Confirmation of “Hit” Compounds by CLSM
CLSM was used to investigate two different yet fundamental features of the underlying biofilm maturation process: (1) the effect of each compound on inhibition of biofilm formation and (2) its ability to kill preexisting biofilms. The biofilm inhibition experiments entailed growing S. epidermidis biofilms in the presence of 10 µM of each compound for 18 h at 37 °C without shaking in PBS.15,16 The killing experiments involving preformed biofilms involved culturing biofilms without test compounds. Next, the biofilms were washed once with PBS and incubated for an additional 24 h at 37 °C in PBS containing 300 µM of each compound, due to the inherent refractory nature of biofilm relative to free-living planktonic bacteria. Biofilm cultures were then washed twice with 100 µL of PBS and stained using the LIVE/DEAD BacLight Bacterial Viability Kit (Invitrogen, Carlsbad, CA). Finally, biofilm images were obtained using a Zeiss LSM510 META Confocal Microscope (Carl Zeiss, Inc., Germany). A 488 nm laser was used in conjunction with a Plan-Apochromat 100×/1.4 Oil DIC objective. Z-stacks were eight bits and measured 127.3 µm × 127.3 µm × 16.0 µm in size. The excitation/emission settings for the dyes were 480/500 nm for SYTO9 (green fluorescence/live cells) and 490/635 nm for propidium iodide (red fluorescence/dead cells). Three-dimensional images were generated using the “Projection” ability of the LSM Image Browser software (version 1.4.0.121; Carl Zeiss, Inc.), and Live/Dead calculations were conducted based on these images using MacBiophotonics ImageJ following the guidelines in the cell-counting guide by C. Labno at the University of Chicago Integrated Light Microscopy Core (http://digital.bsd.uchicago.edu).
Results
The goal of this study was to identify small-molecule compounds that inhibit biofilm formation and/or kill preexisting biofilms of the important human opportunistic pathogen S. epidermidis. We used an HTS to identify such hit compounds. The assays involved (1) initial primary HTS hit identification (single point), (2) hit confirmation in triplicate, and finally (3) determination of AC50 values via 10-point dose-response curves, which were also performed in triplicate. Each tier consisted of three specific assay steps: (1) measurement of total bacterial growth, (2) assessment of bacterial viability in biofilms using a resazurin solution, and (3) the measurement of biofilm formation by a classical CV staining assay, respectively (see schematic in
Fig. 1
or
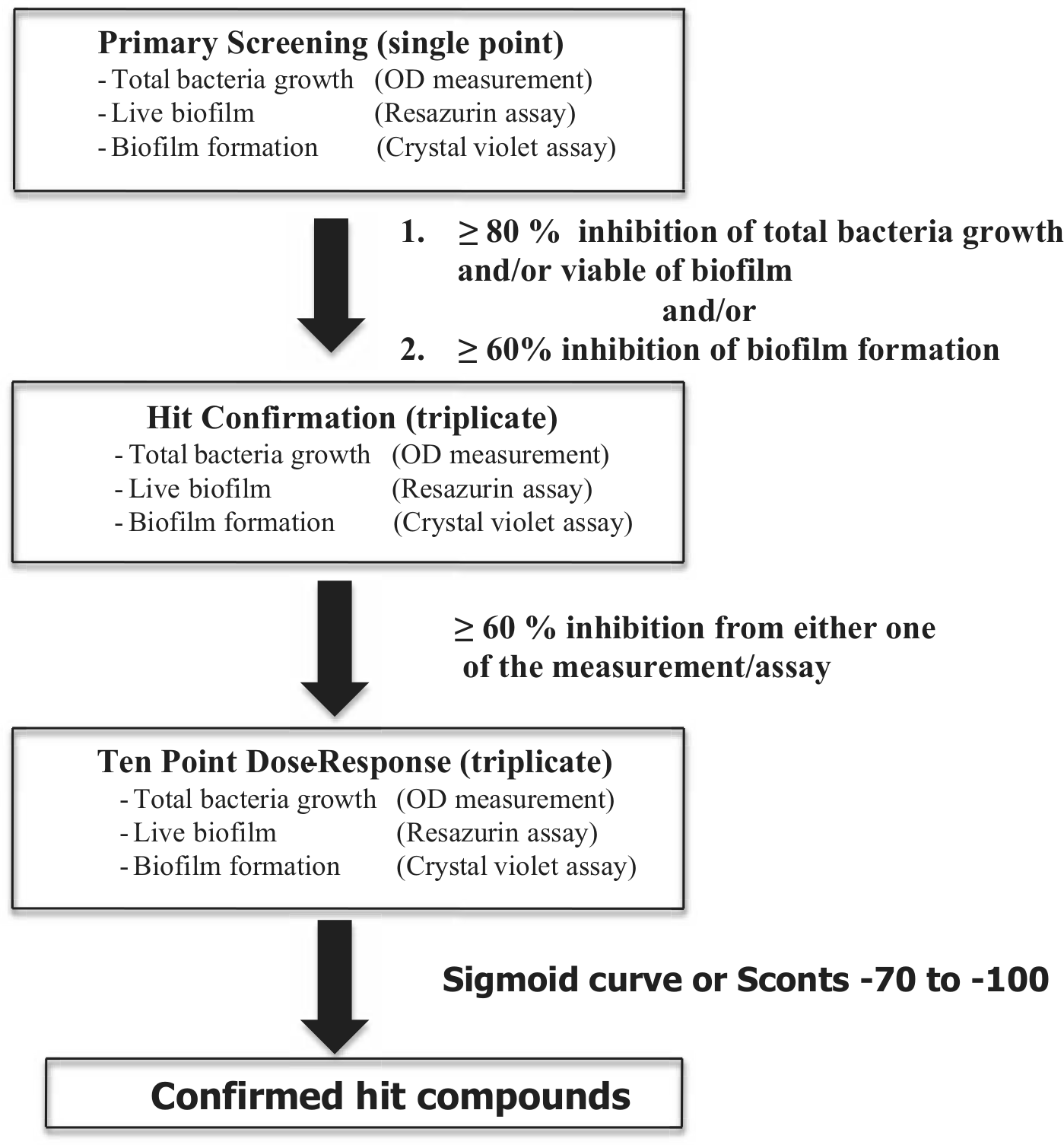
Flow chart of high-throughput screening assay to identify the effective compounds inhibiting formation and/or killing of S. epidermidis biofilm. The 42 865 compounds in the primary screening were used to identify inhibitors of S. epidermidis biofilm formation. Each test compound was evaluated in a single concentration at 10 µM. The effects of compounds on total bacteria growth (OD measurements), live biofilm metabolism (resazurin assay), and biofilm formation (crystal violet assay) were determined. Hit confirmation was performed in triplicate at the same concentration of compound to confirm those compounds that demonstrated ≥80% inhibition of either total bacteria growth and/or live biofilm and/or ≥60% biofilm formation in the primary screening. Finally, dose responses were determined from inhibition measurements across 10 concentrations in triplicate (0.1–50 µM) and were performed for all compounds passing hit confirmation that showed ≥60% inhibition of total bacteria growth and/or live biofilm and/or inhibition of biofilm formation.
Bacteria Biofilm Assay Development
The media used, bacteria inoculum size, as well as resazurin and CV concentrations were optimized to achieve an S/B ratio and Z′ factor that exceeded the generally accepted minimums of S/B >3 and a Z′ >0.5 for a successful HTS.
14
Then, we first investigated the tolerance of S. epidermidis biofilms to DMSO in each of the three assays (total bacterial growth, resazurin metabolic, and CV staining assays for biofilm formation) because all test compounds were initially dissolved in DMSO. Our results show that there were no adverse effects at DMSO concentrations up to 2% for all three assay steps (
Primary Screening, Hit Confirmation, and Dose Response of HTS
A summary of the number of test and hit compounds and the percentage of those that were ultimately designated true hit compounds in the three assays of the S. epidermidis biofilm screening from a single-point primary screening (10 µM) is listed in
Table 1
. Those compounds that showed ≥80% inhibition of bacterial growth and/or resazurin metabolism and/or ≥60% CV assay compared with controls (715 hits compounds, but only 693 compounds were available for further study, or ~1.6% of the primary screen compounds) were advanced to the next tier, the hit confirmation studies. In this tier, each compound was assayed in triplicate at a concentration of 10 µM. There were 352 compounds, approximately 50% of the compounds tested in the hit conformation (693 compounds), which showed ≥60% inhibition of bacterial growth and/or resazurin metabolic assay and/or inhibition in the CV assay when compared with control treatments. These compounds were confirmed with sufficient potency to be considered as solid candidates for advancement to the dose-response experiments. After the dose-response tier of experimentation, all compounds with dose-response curves indicating a decrease in growth with an increase in compound concentration are considered confirmed hits together with the compounds that yielded Sconst values ≥–70. These criteria were used for all three assays in this tier. Sconst is the percentage activity of each compound when it does not demonstrate a sigmoidal curve within the range of the compound concentrations tested. The media-only group was assigned a value of −100, whereas the bacterial control was assigned a value of zero. The criteria for selection of the hit compounds from each tier (either primary screening to hit conformation or hit conformation to dose response) are summarized in
Summary of the Results of the High-Throughput Screening Assay.
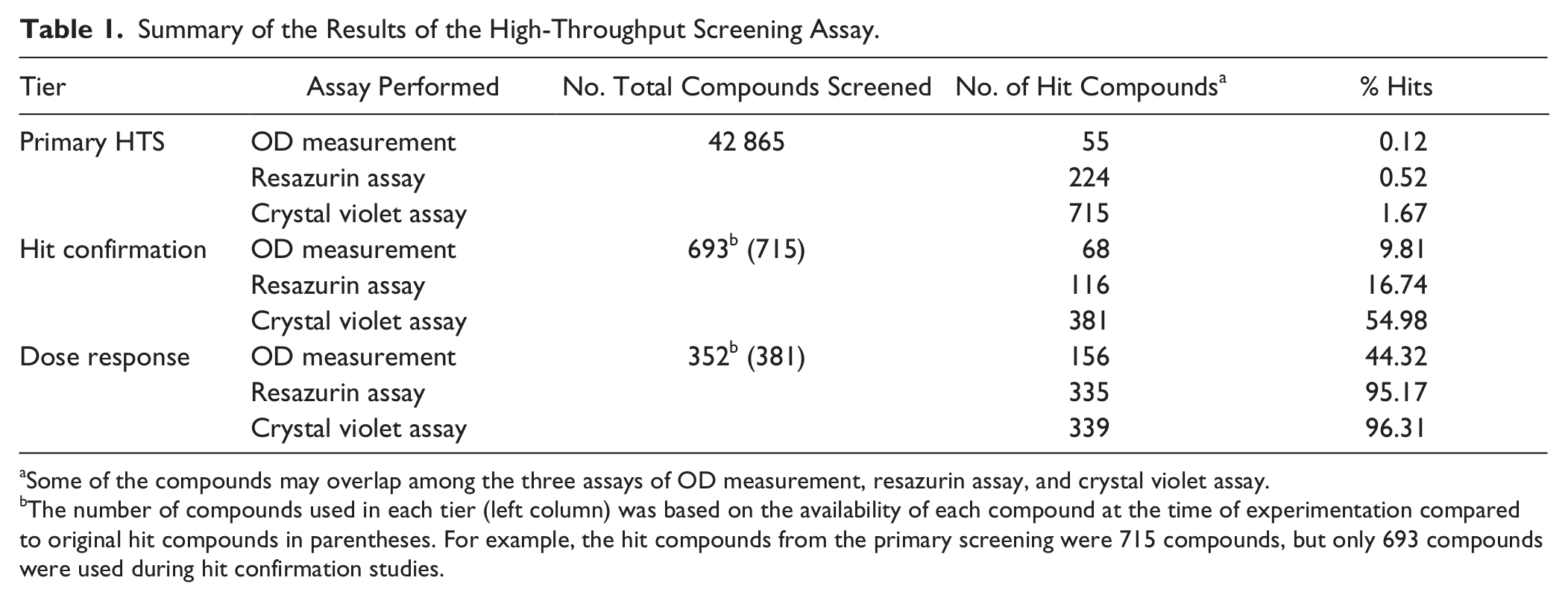
Some of the compounds may overlap among the three assays of OD measurement, resazurin assay, and crystal violet assay.
The number of compounds used in each tier (left column) was based on the availability of each compound at the time of experimentation compared to original hit compounds in parentheses. For example, the hit compounds from the primary screening were 715 compounds, but only 693 compounds were used during hit confirmation studies.
After the last tier, the dose response, the compounds were grouped based on their effectiveness on total bacteria growth and/or live biofilms and/or biofilm formation
Selected Confirmed Hit Compounds for CLSM
To move forward from our HTS results to further characterize and confirm the effects of the confirmed hit compounds in the inhibition of biofilm formation and killing of preexisting S. epidermidis biofilms, 16 random, lead, confirmed-hit compounds ( Fig. 2 , Se-1 through Se-16) were selected for further experimentation using CLSM. Our priority was to validate these confirmed hit compounds based on their effectiveness at either inhibiting and/or killing S. epidermidis biofilms at low concentrations because of cost-effectiveness and medical/material application concerns. Therefore, the compounds evaluated by CLSM were selected based on an AC50 lower than 10 µM using the criteria of either OD measurements and/or resazurin assays and/or CV staining assays and/or the Sconst (≥–70) in which the compounds showed no dose effect in dose-response experiments. The AC50 is the concentration of compound that inhibits 50% of bacterial growth, kills live bacteria in biofilms, or inhibits biofilm formation. The confirmed hit compounds that were effective in only one of the assay systems used in this study (e.g., killing of the biofilm [live biofilm assay] or inhibition of biofilm formation [CV assay]) were not performed in the CLSM studies yet may be conducted in future studies.
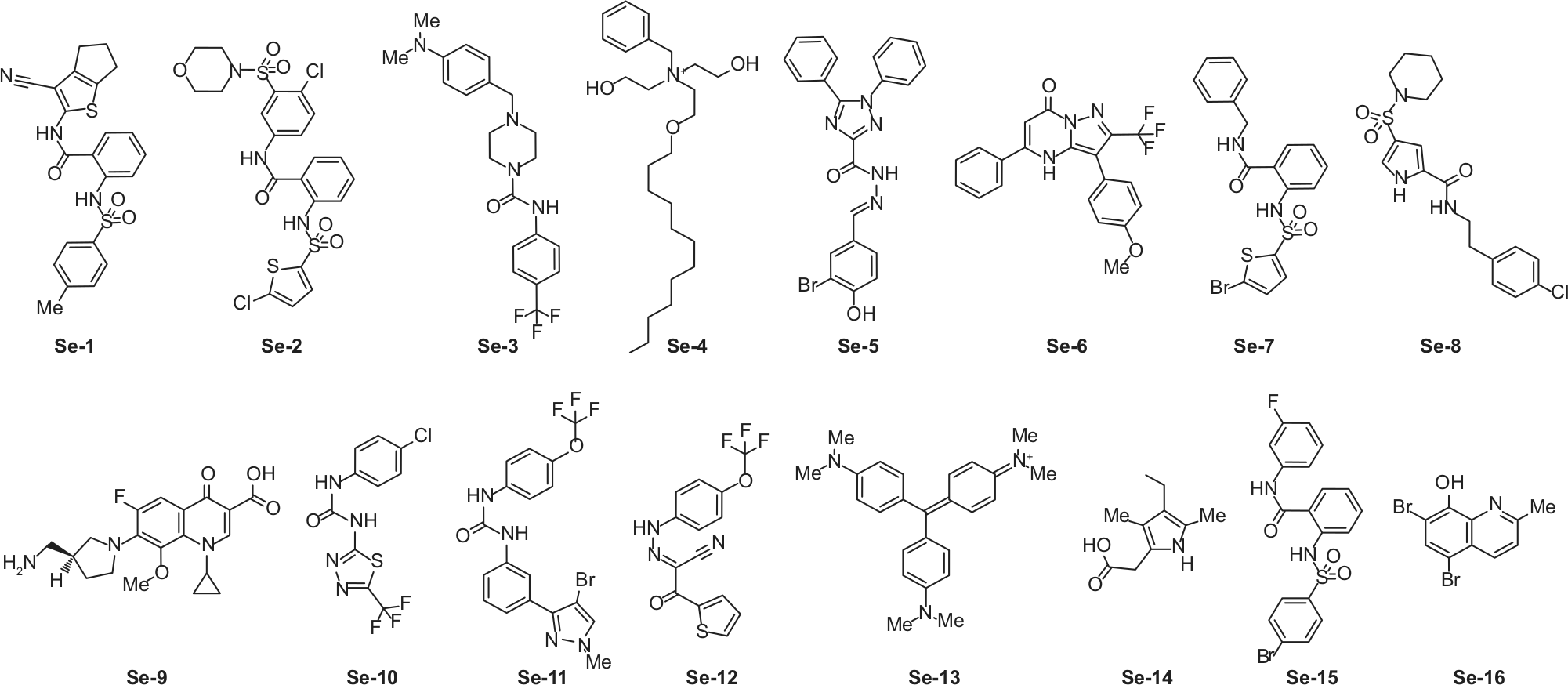
Molecular structure of the 16 selected hit compounds from the high-throughput screening (HTS) assay and used in confocal laser scanning microscopy (CLSM) analysis (see Fig. 3 ). After completing the last tier of the HTS assay (the dose response), these 16 confirmed hit compounds (designated Se-1 to Se-16), which showed effectiveness at either inhibiting and/or killing of S. epidermidis biofilms, were chosen for the subsequent biofilm studies using CLSM.
The AC50, Sconst values, and chemical classification of each compound of the 16 confirmed hit compounds from dose-response experiments are shown in
Table 2
. The AC50 curves and the chemical names are listed in
Summary of Compound Results from Dose-Response and CLSM Experiments.
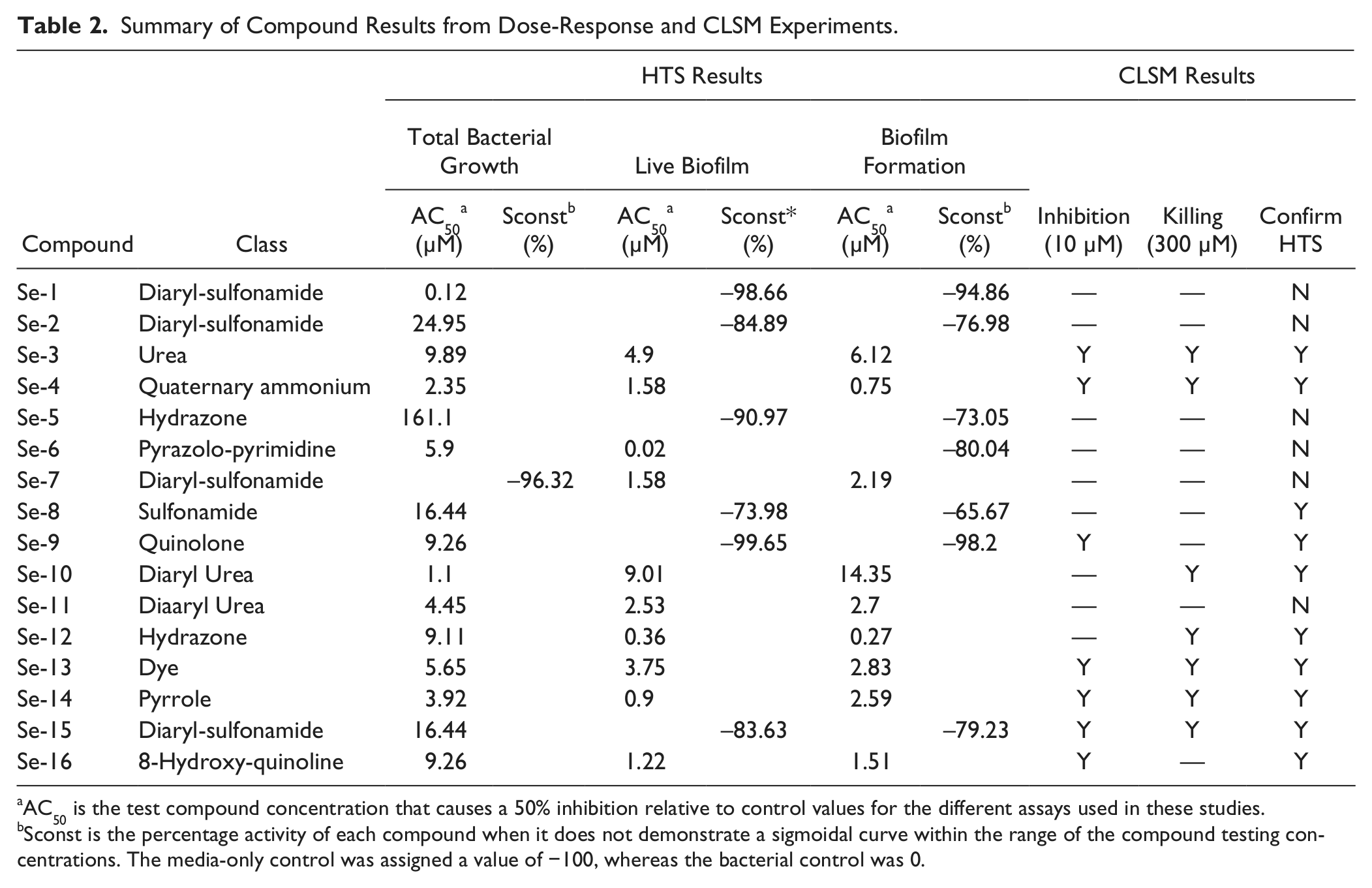
AC50 is the test compound concentration that causes a 50% inhibition relative to control values for the different assays used in these studies.
Sconst is the percentage activity of each compound when it does not demonstrate a sigmoidal curve within the range of the compound testing concentrations. The media-only control was assigned a value of −100, whereas the bacterial control was 0.
Examination of Confirmed Hit Compounds by CLSM
The two parameters, inhibition of S. epidermidis biofilm formation and killing of preexisting S. epidermidis biofilms, were further investigated using CLSM. First, to evaluate the effectiveness of compounds at inhibiting S. epidermidis biofilm formation, bacteria were again grown in the presence of 10 µM of each compound, the same concentration used in first two tiers of the HTS assay. Second, the ability to kill preexisting S. epidermidis biofilms was evaluated at a concentration of 300 µM of each compound (significantly higher concentrations of antimicrobial agents are required to kill biofilm relative to planktonic bacteria 17 ).
The media used for the study of S. epidermidis biofilms in the CLSM experiments were optimized for maximal titers of live biofilm bacteria before treatment with designated compound. The bacteria were grown overnight in TSB and diluted in 0.2% to 10% TSB or PBS for both inhibition of biofilm formation and killing of preexisting biofilm experiments. Our results showed that S. epidermidis biofilms formed a thicker biofilm in PBS than diluted TSB for both inhibition of biofilm formation and killing of preexisting biofilms (
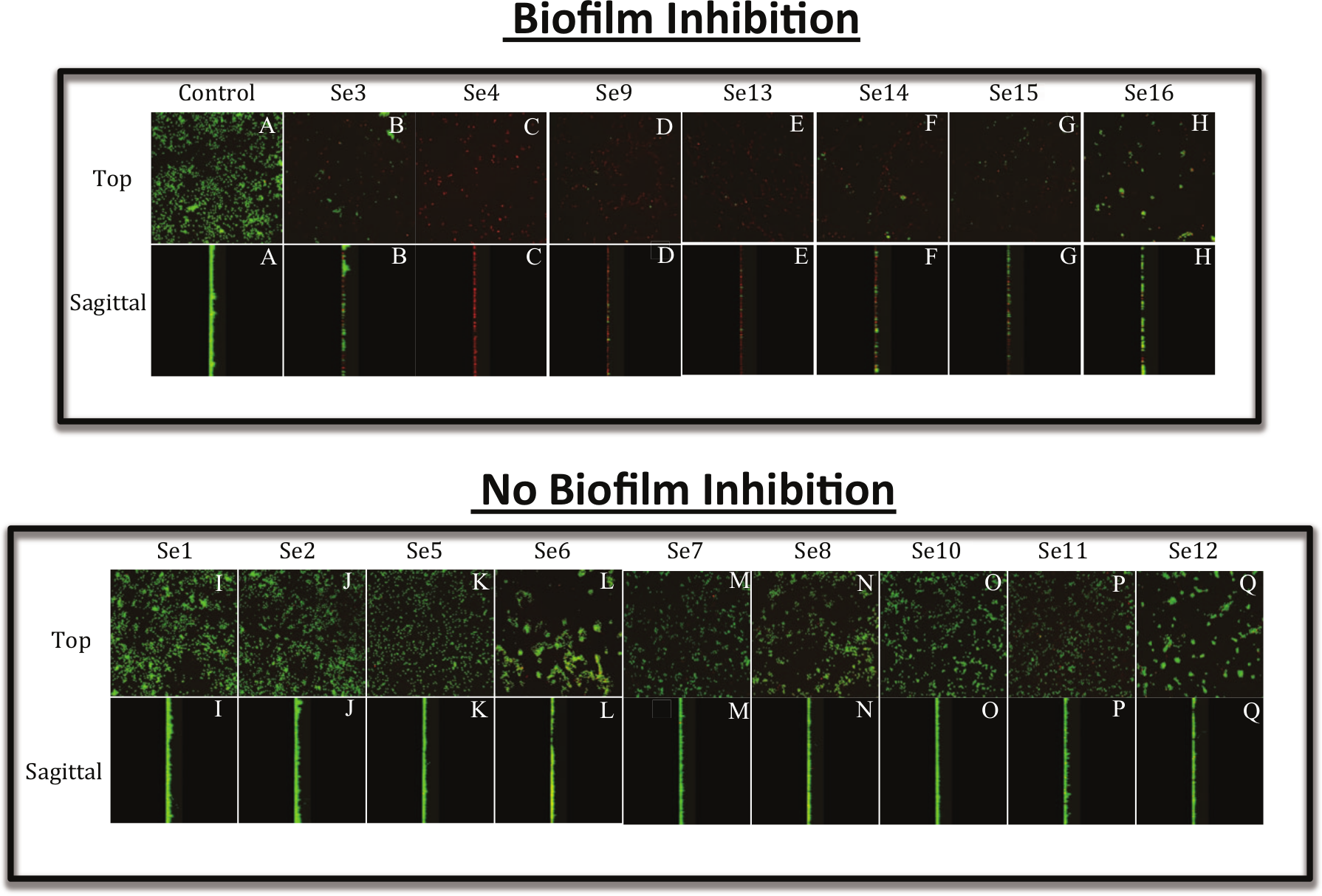
Confocal laser scanning microscopic (CLSM) images of S. epidermidis biofilms when co-cultured with the compound of interest (for inhibition of biofilm formation). S. epidermidis biofilms were grown on glass substrata in the presence of 10 µM of each compound for 18 h. The biofilms were then stained with a viability stain according to the Materials and Methods section. Then, biofilm images of both live and dead bacteria were taken by CLSM. The top panels (both top and sagittal views) show the compounds that inhibited S. epidermidis biofilm formation compared with control bacteria with DMSO (
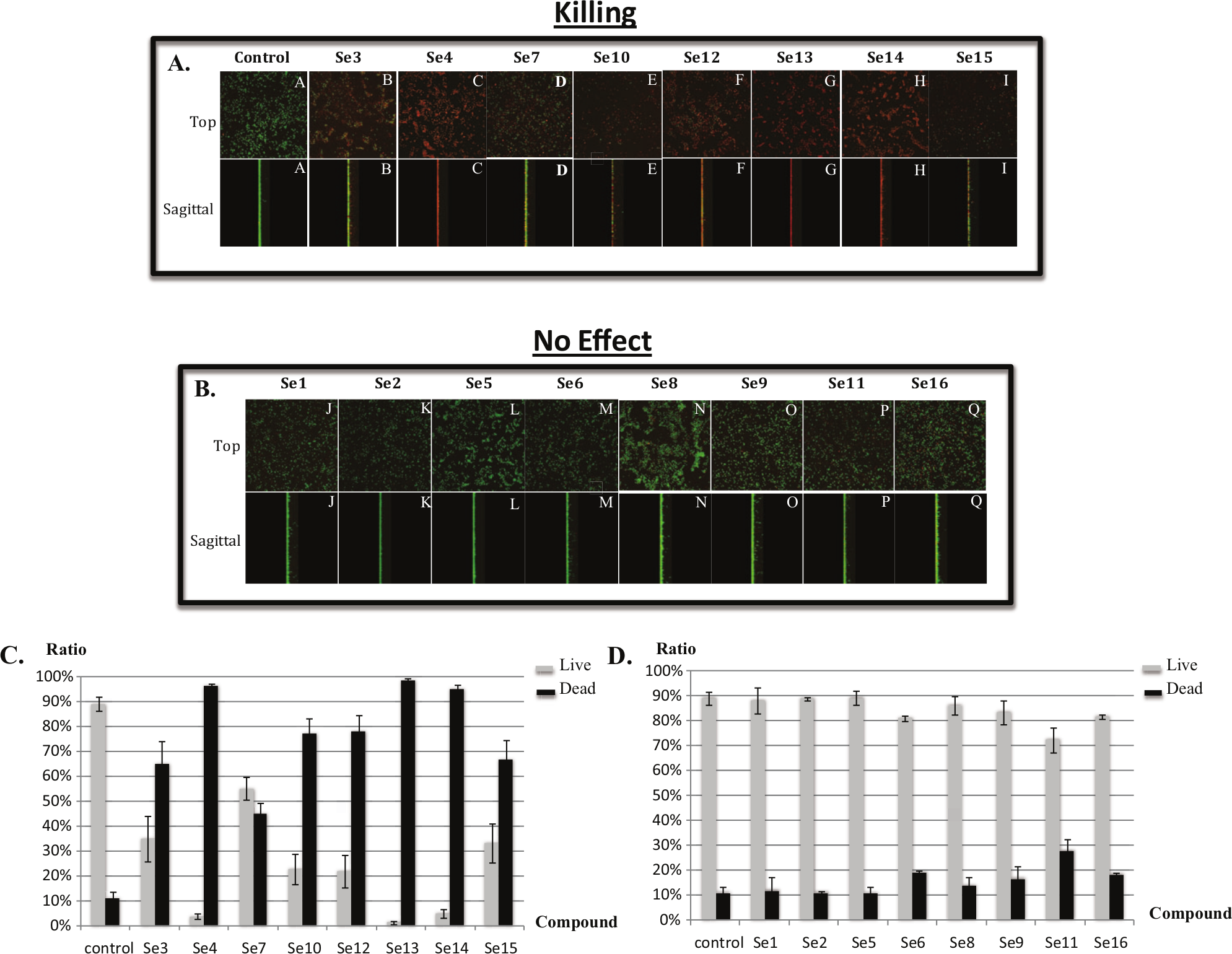
Efficacy of compounds at killing of preexisting S. epidermidis biofilms using CLSM. S. epidermidis biofilms were grown in PBS on glass substrata for 18 h. After washing with PBS, 300 µM of compound was added to the biofilm culture. After 24 h, the biofilms were then stained with a viability stain according to the Materials and Methods section. The biofilm images of both live and dead bacteria were taken using CLSM. The top panel (
Discussion
Inhibition and/or killing of biofilm bacteria is a topic of intensive research on a global level because of their role in a vast number of health, environmental, and industrial problems and also because of their innate, highly refractory antibiotic- and biocide-resistance properties. Numerous laboratories have studied biofilms through their various stages of development (for reviews, refer to1,3), the types of organisms and/or microbial consortia used in their complex assembly, and, most recently, the various genes that are (1) required for biofilm formation, (2) expressed or repressed within biofilms, (3) required for dispersal, (4) involved in matrix formation, and (5) involved in antibiotic or biocide resistance/susceptibility.
S. epidermidis can acquire resistance to β-lactams as well as vancomycin, potentially leading to problems analogous to those of other antibiotic-resistant staphylococcal strains, the most notable being methicillin-resistant (MRSA), vancomycin-intermediate (VISA), and vancomycin-resistant (VRSA) S. aureus strains. Despite excellent progress in the discovery of effective new antibiotics, resistance unfortunately typically develops within 10 to 20 y or even sooner (as little as 1 y), thereby creating huge challenges for basic and clinical scientists alike. 18 Therefore, it has become a major priority in the past 10 y to use the most modern technologies and strategies to identify novel compounds that may interfere with fundamental and/or essential processes involved in biofilm formation in S. epidermidis and other pathogenic bacteria.
To identify compounds with the potential to inhibit and/or kill S. epidermidis in biofilms, an HTS assay was developed and performed in this study. All of the three steps of the HTS assay demonstrated a Z′ factor greater than 0.5 and an S/B ratio more than three (
The compounds were divided into four groups based on the activities of inhibition and/or killing of S. epidermidis biofilms based on the CLSM results. First, we identified those compounds effective at both killing and inhibition of S. epidermidis biofilms. These represented compounds Se-3, Se-4, Se-13, Se-14, and Se-15. The second group consisted of two compounds (Se-9 and Se-16) that inhibited S. epidermidis biofilm formation but did not kill the bacteria biofilm. Third, three compounds (Se-7, Se-10, and Se-12) were able to kill only the preexisting S. epidermidis biofilms. Compounds Se-3, Se-4, Se-10, and Se-12 to -15 were able to kill biofilms during the early stages of development (e.g., bacterial attachment, HTS results) as well as preexisting S. epidermidis biofilms (CLSM results), whereas the remaining compounds were able to kill S. epidermidis biofilms during only the early stages of biofilm development (HTS results, Table 2 ).
Because of such a diverse group of compounds from a structural perspective, the set of compounds showed considerable variation. This would be expected in screening such a diverse library (42 865 of more than 300 000 compounds). Nevertheless, two compound classes clearly emerged. Se-1, Se-2, Se-7, and Se-15, termed diaryl sulfonamides, are all aryl amides and aryl sulfonamides of anthranilic acid. We were also puzzled that some compounds such as Se-6, Se-10, Se-11, and Se-12 ( Table 2 ), which demonstrated inhibitory and/or killing activity in the HTS screen, were not observed in the CLSM study. Thus, further analysis and repetition using a structure-specific theme is certainly worthy of future examination and perhaps under conditions on different substrata. In contrast, both Se-10 and Se-11 are halogenated diaryl ureas, a class known to have numerous examples of biocidal activity. Se-3 and Se-12 bear a more speculative general similarity to the diaryl ureas as well. Se-4 is clearly a lipophilic quaternary ammonium salt, a common motif in surfactants, and it is not surprising for a surfactant to display both biofilm and/or antibacterial efficacy (even against highly refractory Pseudomonas aeruginosa biofilm infections). 19 Se-13 is a colored lipophilic dye, so its perceived activity may be an assay artifact. However, it is highly appropriate that Se-9 was detected in the assay, serving almost as a positive control. Se-9 is a known quinolone antibiotic with potent activity against both gram-positive and gram-negative bacteria.
Still, most of the compounds (Se-3, Se-4, Se-8, Se-9, Se-10, Se-12, Se-13, Se-14, Se-15, Se-16) displayed the same qualitative results at inhibiting and/or killing S. epidermidis biofilms as measured by both HTS and CLSM methods ( Table 2 ). This group of compounds may hold promise as candidates for the treatment of S. epidermidis biofilms based on their respective differences in efficacy using two different surfaces (polystyrene and glass) and two media used in the study (TSB and PBS). The other group of compounds, Se-1, Se-2, Se-5, Se-6, Se-7, and Se-11, were not effective at either inhibiting or killing S. epidermidis biofilms in PBS on glass surfaces in our CLSM results, but this group of compounds was effective when cells were grown on a polystyrene surface and in a TSB plus glucose medium in the HTS results ( Table 2 ).
In summary, from an HTS of 42 865 compounds, a number of heretofore unknown compounds were found to effectively inhibit and/or kill S. epidermidis biofilms, those belonging to a range of chemical classes. Other compounds that were identified as effective in the HTS but were not examined in the CLSM analyses are shown in
Logically, our future studies will involve the identification of the mechanistic basis using some of the top confirmed hit compounds against S. epidermidis RP62a and determine the effectiveness of these compounds in inhibiting and/or killing other medically important biofilm-forming bacteria. Obviously, the effectiveness of each of these compounds could be determined under different growth conditions, such as flow-through biofilms (representing blood or urinary tract infections) or static, anaerobic biofilms (representing abscess infections), because S. epidermidis and many other organisms are able to grow under such conditions. We also plan to determine the effectiveness of these compounds at inhibiting and/or killing other bacterial biofilms such as those generated by P. aeruginosa and subsequently biofilm co-culture of these two important pathogens affecting a plethora of human diseases. For example, cystic fibrosis and chronic obstructive pulmonary disease patients can be infected with both P. aeruginosa and S. aureus in their thick airway mucus. 20 Thus, we strongly believe that many different species of biofilm-forming bacteria should be tested with these compounds to determine their ability to inhibit and/or kill these organisms in biofilms. With such additional information in hand, it is possible that such developmental research will ultimately lead to benefit a variety of patients, from preventive, prophylactic, and treatment strategies.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the United States Air Force Research Laboratory (AFRL) to T.J.L. and R.K. The authors report no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
