Abstract
There is a growing interest in alopecia prevention strategies, as the number of alopecia patients is increasing. We examine the efficacy of herbal medicine for hair growth promotion/hair loss inhibition in two cell lines via Western blot and high-content screening (HCS). Nine herbal extracts were obtained from three different herbal medicine mixtures using 3 different extraction methods. Five target proteins—IGF-1 (insulin-like growth factor–1), TGF-β2 (transforming growth factor–β2), VEGF (vascular endothelial growth factor), DKK-1 (Dickkopf-1), and Wnt5α—were observed for the assessment of hair growth promotion/hair loss inhibition efficacy. The efficacies of nine extracts were compared with minoxidil as control. Efficacy was defined as a rise in the expression levels of IGF-1, VEGF, and Wnt5α but a decrease in DKK-1 and TGF-β2. Intracellular concurrent imaging of these proteins was successfully achieved using HCS, employing visible-to-near infrared probing based on quantum-antibody conjugates and hypermulticolor imaging.
Keywords
Introduction
Recently, high-content screening (HCS) has received great attention in the process of drug development due to its capability to provide real-time monitoring of intracellular events using fluorescent probes. 1 The complex intracellular responses involved in drug-induced efficacy or cytotoxicity can be observed in organ-specific cells by HCS.2,3 This capability of HCS allows the acquisition of cellular information in more detail and provides a useful basis for making better decisions on progressing compounds. However, HCS has a practical limitation in simultaneously monitoring complex intracellular responses in intact cells. This is because HCS measurement is dependent on fluorescent probes. Most probing markers are generally fluorescent dyes with a broad wavelength range in both absorption and emission spectrum.4,5 This spectral property of probing dyes makes it difficult to simultaneously observe a lot of intracellular events using HCS due to serious overlapping among the emission spectra of probing dyes. Until now, simultaneous monitoring of more than four intracellular events by HCS has been rare. To generalize the use of HCS as a cell-based drug screening method, it is important to address this issue.
In recent years, the incidence of alopecia has increased. Alopecia occurs due to a nutritional imbalance induced by a combination of environmental pollution, stress, frequent dyeing and perm, drinking, smoking, and diet. The age at the onset of hair loss is decreasing, indicating an increased prevalence in the younger generation. Forty percent of patients with alopecia require treatment. Alopecia can be cured by various hair loss treatments, including hair transplant; cosmetics such as shampoo, rinse, essence, and soap; and medication. Among these methods, medical treatment has been shown to be efficacious in improving hair growth. Some drugs prevent alopecia by inhibiting the male hormone. Finasteride and minoxidil are two well-known drugs for hair loss.6,7 Finasteride is a synthetic 5-α-reductase inhibitor, and minoxidil is a vasodilator, both of which suppress male hormones and are used to treat alopecia. However, the use of two drugs should be limited due to strong side effects. Finasteride causes sexual dysfunction and minoxidil induces itching, redness, and inflammation. Because hair loss treatment requires a long-term remedy, minimizing adverse side effects is crucial in drug development. Accordingly, new ingredients were discovered from natural products with less adverse effects. Many companies have improved product development by using herbal medicines: RiUP containing minoxidil (Taisho Pharmaceutical Co., Ltd., Tokyo, Japan), Yakuyo Shidenkai Z containing eugenol and eugenyl glucoside (Kanebo Ltd., Tokyo, Japan) extracted from Syringa patula var. kamibayshii, and Yakuyo Furorin XG (Shiseido Cosmetic Co., Yokohama, Japan) extracted from Sophora japonica L.
In this study, we investigated the effects on the expression level of proteins involved in hair loss and hair growth using Western blot and hypermulticolor single-cell imaging cytometry. Herbal medicine was extracted with viscozyme or pectinex, which was expected to improve physiological activities by changing glycosides to the active aglycone form. Target proteins used to identify drug efficacy were IGF-1 (insulin-like growth factor–1), TGF-β2 (transforming growth factor–β2), VEGF (vascular endothelial growth factor), DKK-1 (Dickkopf-1), and Wnt5α. These five target proteins were related to the hair growth cycle in human follicle dermal papilla cells (HFDPC) and the human keratinocyte cell line, HaCaT. TGF-β2 is expressed in the catagen phase and leads to the apoptotic death of hair cells. 8 DKK-1 is a Wnt pathway regulator and induces apoptosis and inhibits proliferation in human keratinocytes. 9 IGF-1 is a substance that activates cells in the hair root and suppresses the catagen and telogen phases of the hair growth cycle. 10 VEGF is a growth factor that stimulates vasculogenesis and angiogenesis and triggers hair growth. 11 Wnt5α is a key factor to regenerate hair follicles, control the hair growth cycle, induce the progression from the catagen phase to the anagen phase, prolong the anagen phase, and inhibit apoptosis in dermal papilla cells. 12 The efficacy of several herbal medicines was first screened by Western blot. Among these, only the candidates that showed capability as a hair growth promoter and/or hair loss inhibitor were analyzed by HCS with single-cell imaging cytometry. After treatment of HFDFC and HaCaT cells with herbal medicine, altered expressions of the five proteins were observed to assess their efficacies of hair growth promotion and hair loss inhibition. The intracellular activations/deactivations of five target proteins by herbal medicines were monitored concurrently by HCS. As mentioned above, simultaneous monitoring of the five target proteins in intact cells remains a challenge for HCS. This was achieved by the use of quantum dot–antibody conjugates as fluorescent probes in addition to near-infrared (IR) probing based on hypermulticolor cellular imaging. First of all, compared with conventional fluorescent dyes, the quantum dot has a very narrow emission wavelength range, which is advantageous for using a larger number of quantum dots at the same time. 13 Despite this advantage, their emission spectra still provide spectral overlap among them. Acquisition of cellular imaging at a particular wavelength other than spectral overlap is an important factor to consider in order to accomplish concurrent monitoring of a larger number of cellular events than HCS at the current stage. Securing room to acquire cellular imaging at a particular wavelength makes it possible to use a larger number of probes. Hypermulticolor cellular imaging based on an acousto-optic tunable filter (AOTF) is an attractive solution to solve this problem. The quantum dot is also well organized for application to a variety of surface chemistries, which minimizes nonspecific binding to intracellular proteins. Visible-to-near IR probing is another important strategy to establish HCS, allowing a larger number of probes than the current stage. This is because it is difficult to exceed four or five probes in the visible range even though quantum dots and hypermulticolor imaging are employed for HCS. In this work, the intracellular concurrent monitoring of five target proteins responsible for hair growth promotion and hair loss inhibition was attempted at the single-cell level with visible-to-near IR probing of quantum dot–antibody conjugates in addition to hypermulticolor imaging. The employed AOTF can cover near-IR cellular imaging up to 1000 nm. Compared with Western blot, which requires cell lysis, target proteins can be monitored intracellularly by HCS. It is expected that the accuracy to assess herbal medicine efficacy with respect to hair growth promotion/hair loss inhibition can be enhanced by the HCS approach, eliminating protein loss due to cell lysis common during Western blot.
Materials and Methods
Reagent
VEGFR antibody, Wnt5α antibody, DKK-1 antibody, IGF-1 antibody, and TGF-β2 antibody were from Abcam (Cambridge, UK). Qdot 525/565/625/655/800 antibody conjugation kits were purchased from Invitrogen (Eugene, OR).
Cell Culture
HFDPC and HaCaT cells were used to assess the effects of drug as hair restorers. HFDPC, purchased from Promo Cell (Heidelberg, Germany), was cultivated in HFDPC medium (Promo Cell) containing fetal calf serum, basal medium, basic fibroblast growth factor, and insulin. When the cells reached 80% confluence, they were subcultured using HBSS (HEPES buffered saline solution), trypsin/EDTA solution, and neutralizing solution (Promo Cell). HFDPC was used within 5 to 6 passages for Western blot and HCS because more than 10 passage numbers of HFDPC affect the cell activation. HaCaT cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM; Gibco BRL, Gaithersburg, MD) supplemented with 10% fetal bovine serum (FBS), 100 units/mL penicillin, and 0.1 mg/mL streptomycin (Invitrogen). Cells were maintained at 37 °C with humidified air under an atmosphere of 5% CO2.
Quantum Dot (Qdot)–Antibody Conjugation
Five Qdots of different colors were conjugated to five different antibodies using the Qdot antibody conjugation kit according to the manufacturer’s instruction: Qdot 525 to VEGFR, Qdot 565 to Wnt5α, Qdot 625 to DKK-1, Qdot 655 to IGF-1, and Qdot 800 to TGF-β2. Briefly, Qdots were activated with 4-(maleimidomethyl)-1-cyclohexanecarboxylic acid N-hydroxysuccinimide ester (SMCC) as an amine-to-thiol crosslinker to the maleimide-nanocrystal surface. Antibodies were treated with dithiothreitol (DTT) to break the disulfide bond in antibodies and to expose free sulfhydryls. Excess SMCC and DTT were removed by size exclusion chromatography. Maleimide-Qdots were incubated with reduced antibodies for 1 h; therefore, antibodies were covalently bound to Qdots. The excess maleimide groups were removed by β-mercaptoethanol. Unbounded Qdots and antibodies were removed by a separation media column, supplied in the kit. Finally, five Qdot-antibody complexes were manufactured (VEGFR–Qdot 525, Wnt5α–Qdot 565, DKK-1–Qdot 625, IGF-1–Qdot 655, and TGF-β2–Qdot 800). The maximum emission wavelengths of VEGFR, Wnt5α, DKK-1, IGF-1, and TGF-β2 were 525, 565, 625, 655, and 800 nm for each.
Extraction of Candidate Agents as a Hair Restorer and/or Hair Loss Inhibitor from Medicinal Herbs
Twenty-three dried herbal medicines purchased from the local markets were mixed and ground with a mixer mill. The ratios are listed in
Table 1
. A schematic diagram of the extraction process is represented in
Herbal Medicines and Composition of the Extracts.
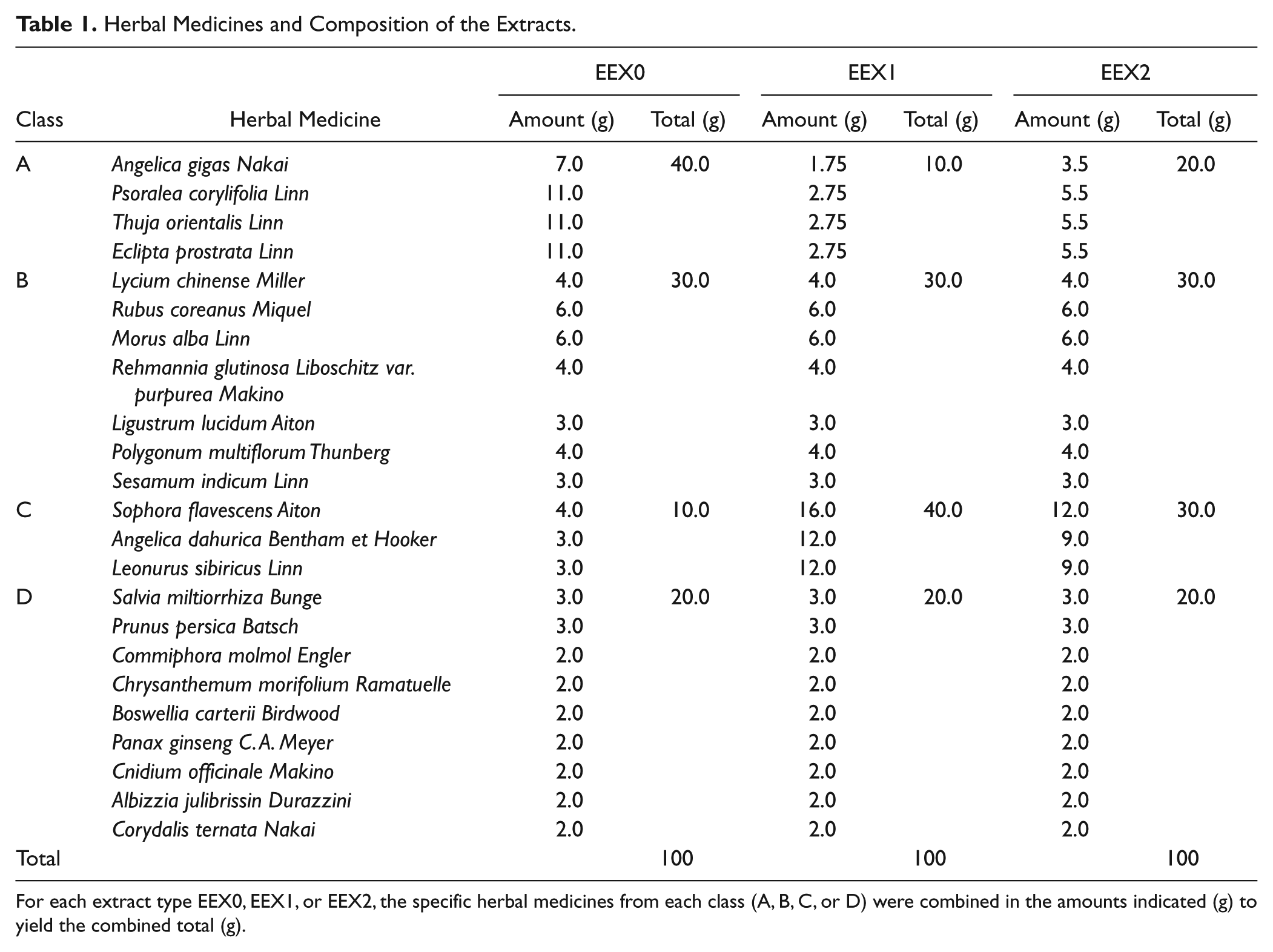
For each extract type EEX0, EEX1, or EEX2, the specific herbal medicines from each class (A, B, C, or D) were combined in the amounts indicated (g) to yield the combined total (g).
Treatment of Cells with Minoxidil and Herbal Medicine Extracts
HFDPC and HaCaT cells were cultured for 24 h in serum-free DMEM and then treated with minoxidil and the extracts of herbal medicines with various concentrations for 24 h. The cells were treated with 2, 6, and 12 µM minoxidil. Then, 10, 25, and 50 µg/mL of herbal medicine extracts (EEX0_Vis_EA, EEX1_Vis_EA, EEX2_Vis_EA, EEX0_Pec_EA, EEX1_Pec_EA, and EEX2_Pec_EA) were used to treat the cells while 10 and 50 µg/mL EEX0_Vis_EA_7.5, EEX1_Vis_EA_7.5, and EEX2_Vis_EA_7.5 were used to treat the cells. The control was the cells treated with 0.1% DMSO.
Western Blot
The extract-treated cells from the 12-well plate were collected and resuspended in lysis buffer (complete lysis-M; Roche, Manheim, Germany) at 4 °C for 15 min. The proteins were separated, and the supernatant protein content was measured by the Bradford protein assay. Then, 30 µg protein was separated on a 12.5% sodium dodecyl sulfate–polyacrylamide gel (SDS-PAGE). Then, protein detection was performed with five purified primary antibodies: anti-VEGFR (1:2000), anti-Wnt5α (1:400), anti–DKK-1 (1:400), anti–IGF-1 (1:400), and anti–TGF-β2 (1:1000) at room temperature for 2 h. The membranes were then washed with TBS-T (Tris-buffered saline and Tween-20) buffer and incubated with horseradish peroxidase–linked anti-rabbit secondary antibody (1:1000) at room temperature for 1 h. The bound antibodies were quantified with an enhanced chemiluminescence (ECL) detection kit (Roche, Basel, Switzerland) and a Luminescent Image Analyzer (LAS-1000; Fujifilm, Tokyo, Japan). All the Western blot assays were repeated three times in this study.
HCS
The extract-treated cells were detached from the 12-well plate using accutase (Thermo Electron Corporation, Waltham, MA) for 10 min at 37 °C. Then, quantitative analysis was carried out using hyperspectral single-cell imaging cytometry described in our earlier publication.2,14,15 Briefly, the mercury lamp beam purified by an excitation filter (330WB80; Omega Filters, Brattleboro, VT) was focused onto the cells using a microscope objective lens. The fluorescence emissions of Qdots were collected by the identical microscope objective lens and detected by a charge-coupled device (CCD) camera (CoolSNAP cf mono, A05F871008; Photometrics, Tucson, AZ) through an AOTF (TEAF10-0.4-1.00-SD; Brimrose, Sparks, MD). The spectral range of NIR-AOTF covers from 400 to 1000 nm. The beam of a particular wavelength was transmitted through the AOTF, and the transmitted beam was scanned by the applied acoustic filed at a rate of 1 wavelength/s and detected by a CCD camera (CoolSNAP cf mono, A05F871008; Photometrics). Fluorescence cellular images were obtained as a function of wavelength and analyzed automatically using commercially available software (MetaMorph, Version 7.1.3.0; Molecular Devices, Downingtown, PA).
Results
Minoxidil-Induced Activation/Deactivation of Target Proteins
The activation/deactivation of target proteins (TGF-β2, DKK-1, VEGF, IGF-1, and Wnt5α) by minoxidil was detected in HFDPC and HaCaT cells with Western blot (
Figure 1A
,
B
shows quantification of band intensities on Western blots in
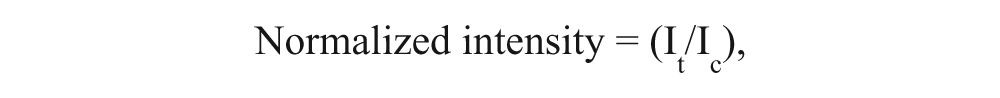
where It represents the intensity of the band obtained from the minoxidil-treated cell, and Ic is the intensity of the band obtained from the control cell without the minoxidil treatment. The normalized intensities were plotted as a function of minoxidil concentration. (It/Ic) > 1 indicates a higher expression level of the target proteins in minoxidil-treated cells than in the control cell. On the other hand, (It/Ic) < 1 indicates a lower expression level of the target proteins in minoxidil-treated cells than in the control.
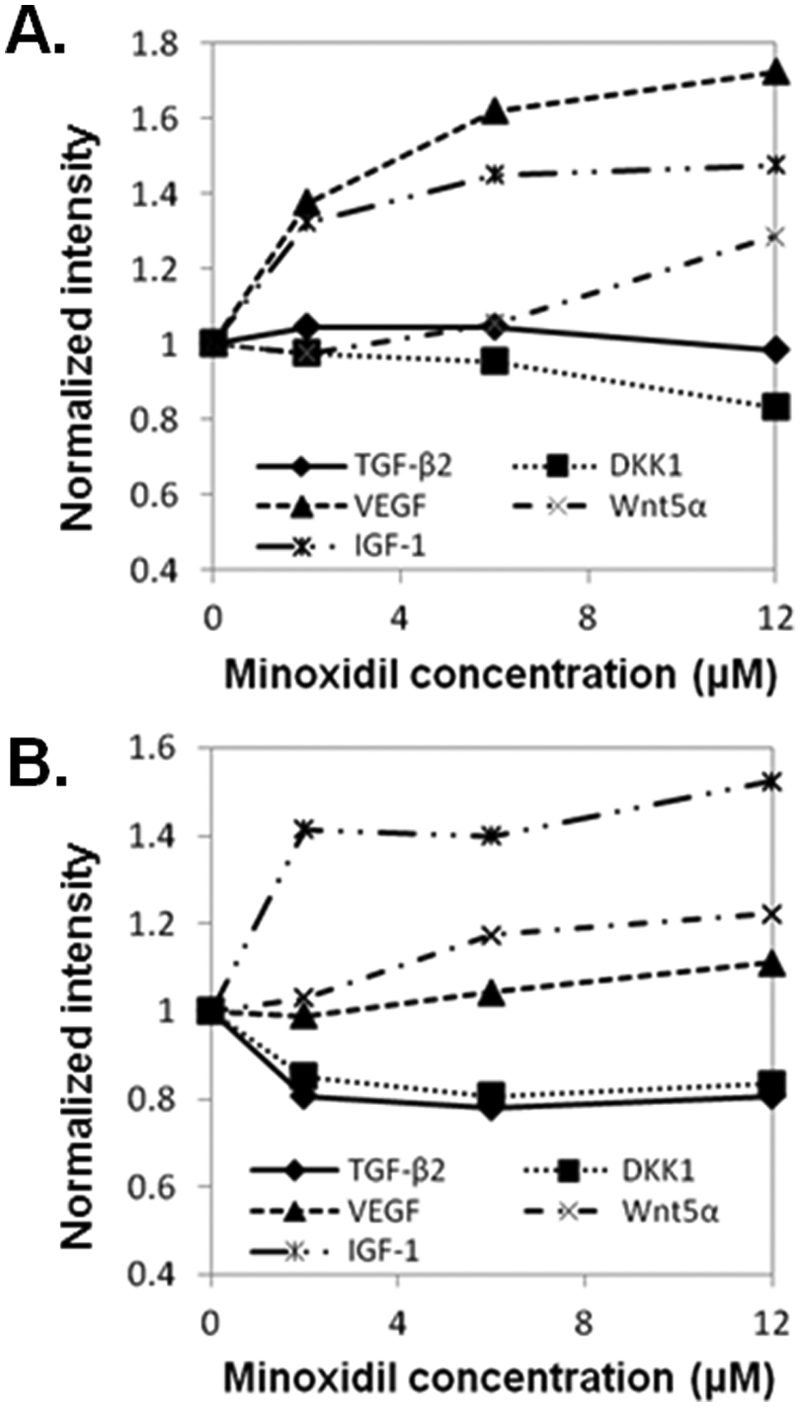
Quantification of Western blots for five target proteins in minoxidil-treated human follicle dermal papilla cells (HFDPC) (
In HFDPC, four target proteins showed a significant increase or decrease compared with the control. The expression level of DKK-1 was reduced, but those of VEGF, IGF-1, and Wnt5α increased after minoxidil treatment. In HaCaT cells, activation of all target proteins appeared to be altered by minoxidil. The expression levels of DKK-1 and TGF-β2 in minoxidil-treated HaCaT cells were lower than the control, whereas those of VEGF, IGF-1, and Wnt5α in minoxidil-treated HaCaT cells were higher than the control.
Effects of Herbal Medicines on Hair Growth
To investigate the effects of the extracts (EEX0_Vis_EA, EEX1_Vis_EA, EEX2_Vis_EA, EEX0_Pec_EA, EEX1_Pec_EA, EEX2_Pec_EA, EEX0_Vis_EA_7.5, EEX1_Vis_ EA_7.5, and EEX2_Vis_EA_7.5) as a hair restorer or hair loss inhibitor, HFDPC and HaCaT cells were treated with different concentrations of each extract for 24 h. The concentrations of Vis_EA samples and Pec_EA samples were 10, 25, and 50 µg/mL. Concentrations of Vis_EA_7.5 samples were 10 and 50 µg/mL, and 0.1% DMSO was used as a control.
To compare the efficacy of an extract of herbal medicines with minoxidil directly, cells were treated with minoxidil or extracts and analyzed using Western blot. All extracts and minoxidil were treated with the same concentration (50 µg/mL). Control cells were treated with DMSO.
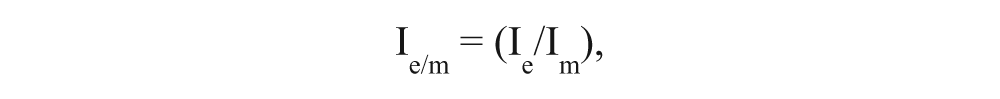
where Ie is the band intensity of the target protein in the extract-treated cells, and Im represents the band intensity of the target protein in minoxidil-treated cells. Ie/m > 1 indicates a higher expression level of the target proteins with herbal extract treatment than with minoxidil treatment, whereas Ie/m < 1 indicates a lower expression level of the target proteins with herbal extract treatment than with minoxidil treatment. In all the cases, TGF-β2 and DKK-1 showed >1, and VEGF, IGF-1, and Wnt5α showed <1. Consequently, the efficacy of these three extracts was lower than that of minoxidil. The efficacy comparison of herbal extracts in HFDPC and HaCaT cells is shown in Table 2 . Nine kinds of herbal extracts affected HFDPC more than HaCaT cells. Based on the efficacy of herbal extracts in both cells, EEX0_Vis_EA, EEX2_Vis_EA, and EEX1_Vis_EA_7.5 showed therapeutic effects and potential to be used as a hair growth–promoting and/or hair loss–inhibiting agent.
Quantitative Changes in Expression of Five Proteins in Promoting Hair Growth and Preventing Hair Loss in HFDPC and HaCaT Cells.
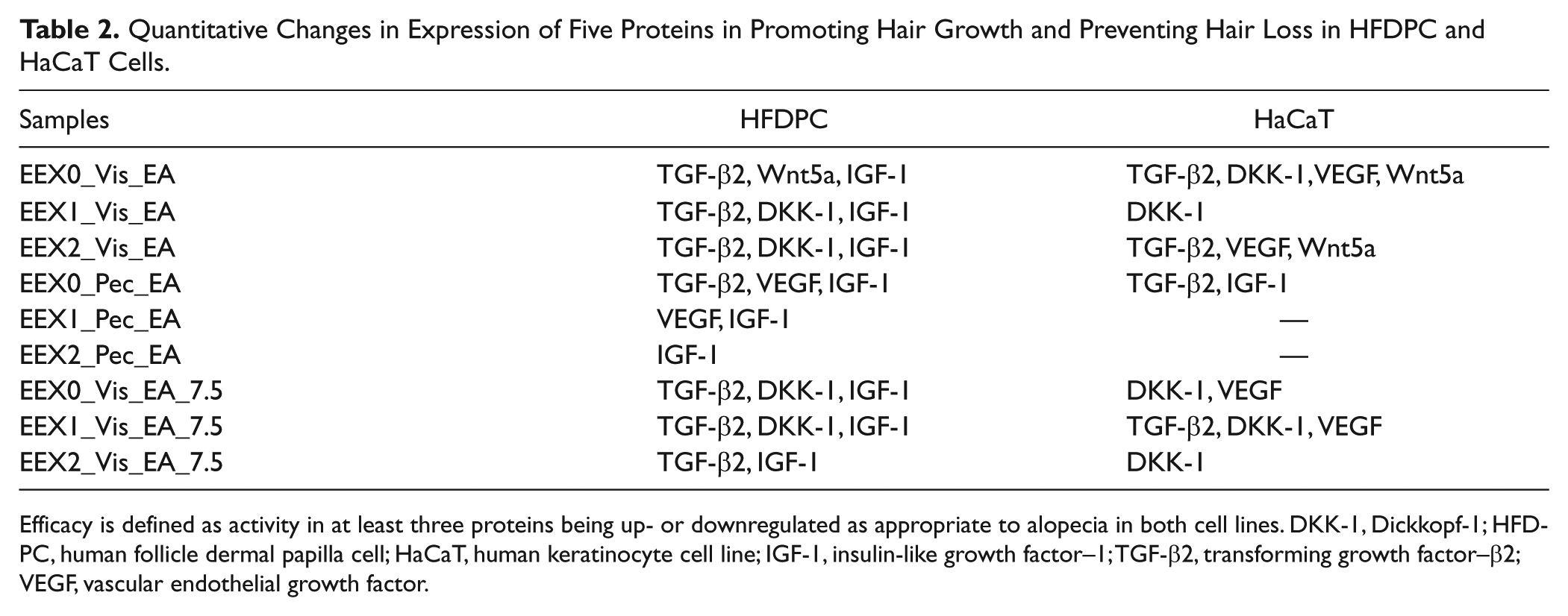
Efficacy is defined as activity in at least three proteins being up- or downregulated as appropriate to alopecia in both cell lines. DKK-1, Dickkopf-1; HFDPC, human follicle dermal papilla cell; HaCaT, human keratinocyte cell line; IGF-1, insulin-like growth factor–1; TGF-β2, transforming growth factor–β2; VEGF, vascular endothelial growth factor.
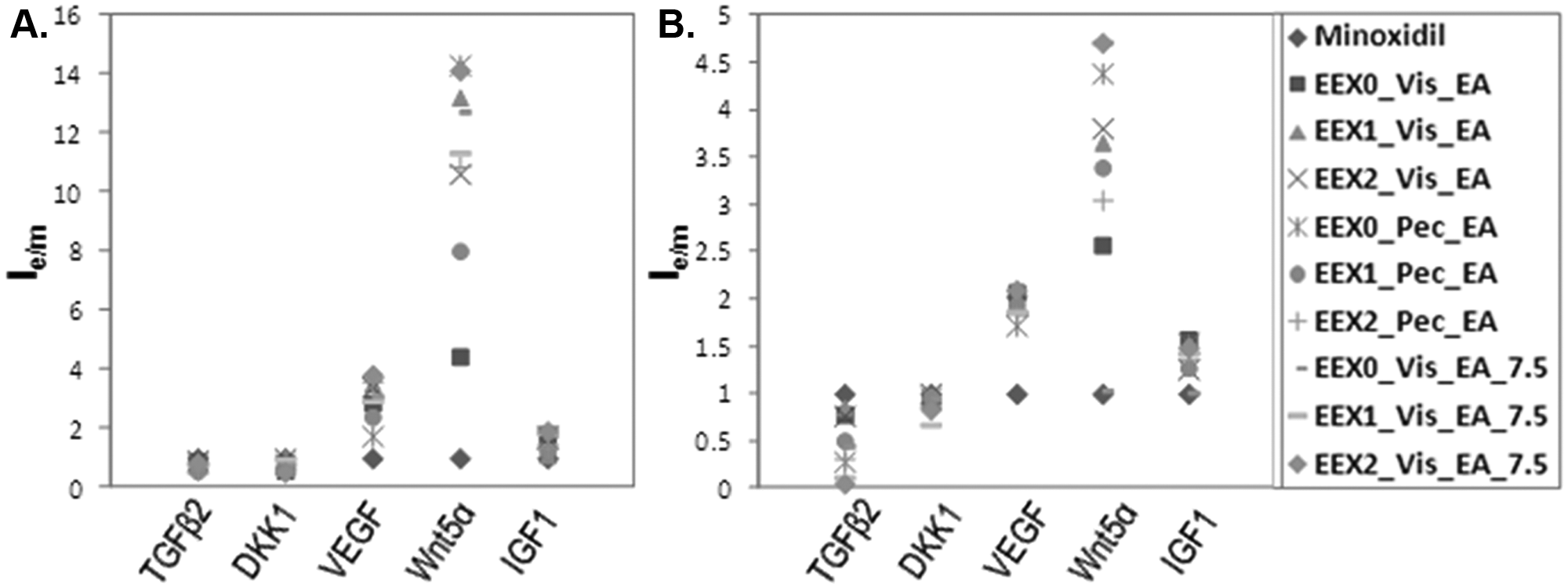
Comparison of herbal medicine extract- and minoxidil-induced upregulation/downregulation of five target proteins in treated human follicle dermal papilla cells (HFDPC) and the human keratinocyte cell line (HaCaT). HFDPC (
HCS of Herbal Medicines
The Western blot results suggested that EEX0_Vis_EA, EEX2_Vis_EA, and EEX1_Vis_EA_7.5 extracts had drug efficacy as a hair restorer and/or hair loss inhibitor in both cell lines. HCS was performed to monitor intracellular activations of target proteins in these three samples at the single-cell level. VEGF, Wnt5α, DKK-1, IGF-1, and TGF-β2 antibodies were conjugated to Qdot 525, 565, 625, 655, and 800, individually. The herbal extract–treated two cells were treated with Qdot-tagged antibodies. Intracellular imaging acquired from target proteins labeled with fluorescent Qdot-antibody conjugates was automatically analyzed using hyperspectral single-cell imaging cytometry. 16 Figures 3 to 5 show the HCS results in the extract-treated cells. The y-axes in Figures 3 to 5 (B , D ) indicate the normalized intensities of Qdot-linked target proteins in the cells. The y-axis was calculated according to the following equation:
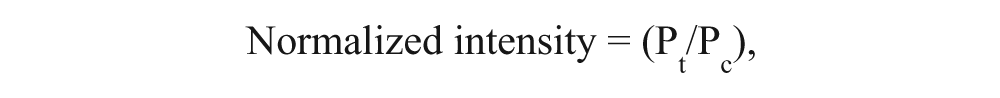
where Pt represents the percentage of Qdot-bound single cells in the extract-treated cells with various concentrations, and Pc is the percentage of Qdot-linked single cells in the control (no extract treatment).
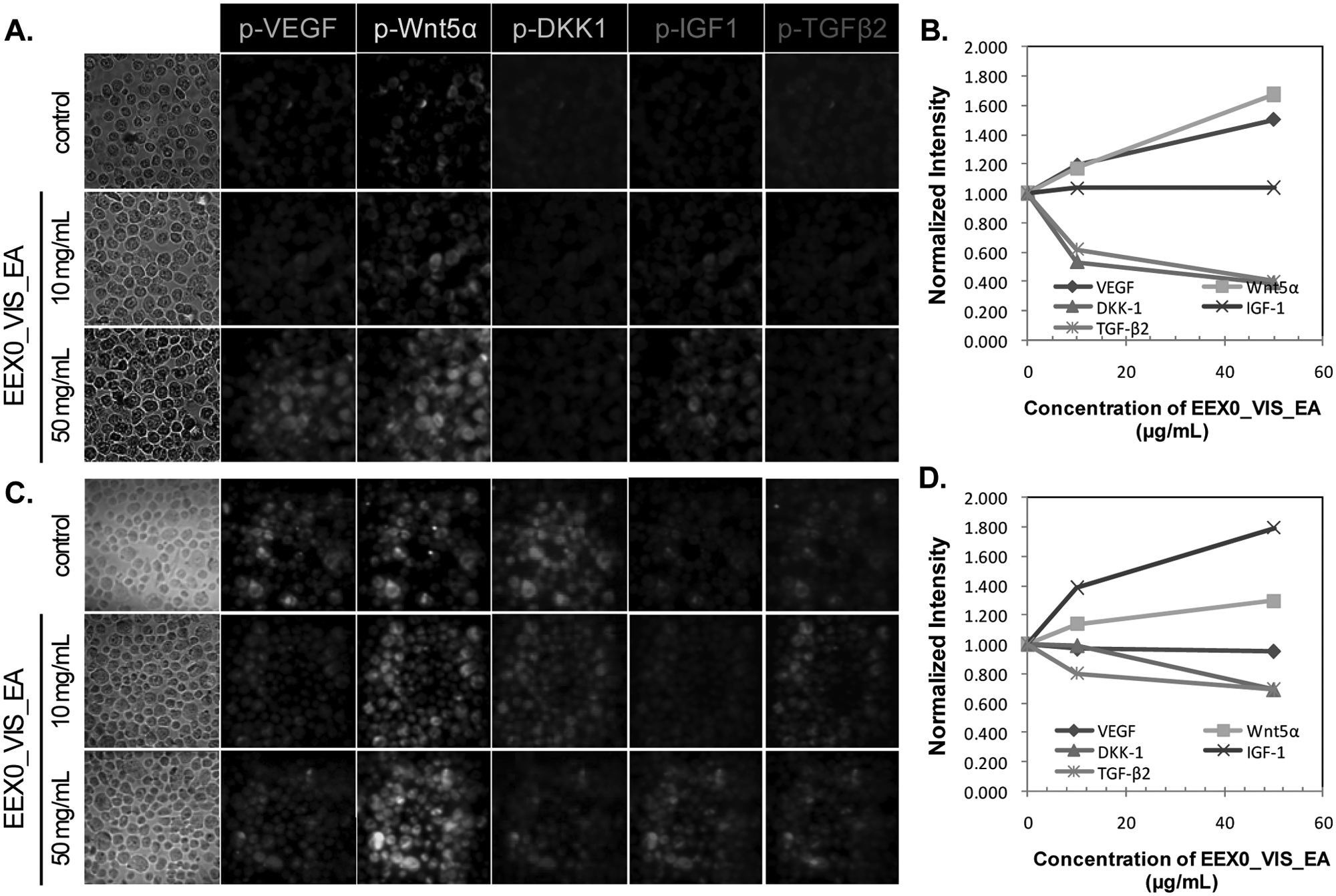
High-content screening of upregulation/downregulation of five target proteins by EEX0_Vis_EA using hyperspectral single-cell imaging cytometry. The human keratinocyte cell line (HaCaT) (
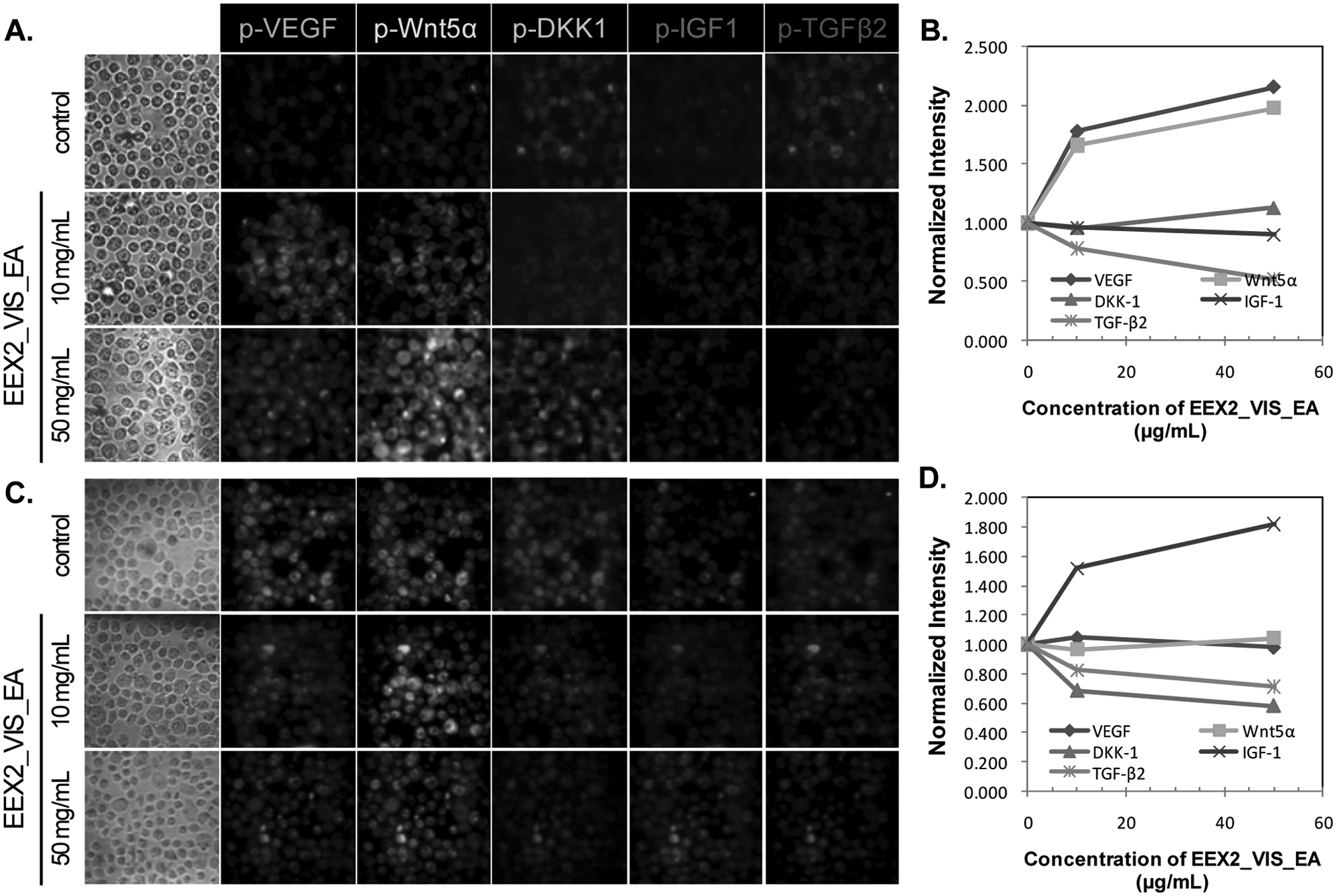
High-content screening of upregulation/downregulation of five target proteins by EEX2_Vis_EA using hyperspectral single-cell imaging cytometry. The human keratinocyte cell line (HaCaT) (
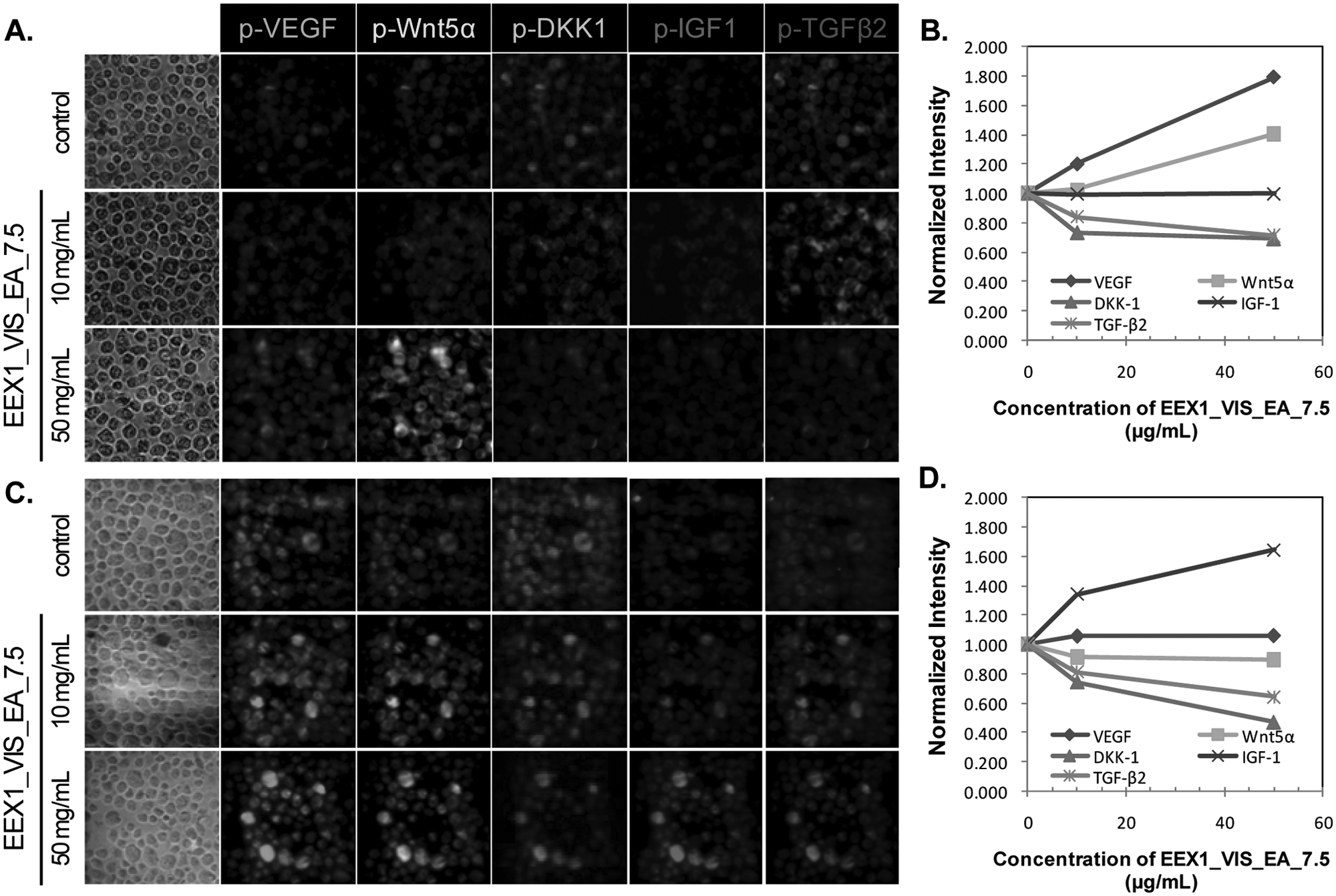
High-content screening of upregulation/downregulation of five target proteins by EEX1_VIS_EA_7.5 using hyperspectral single-cell imaging cytometry. The human keratinocyte cell line (HaCaT) (
In HFDPC, EEX0_Vis_EA induced changes in TGF-β2, Wnt5α, and IGF-1 expression. EEX2_Vis_EA and EEX1_Vis_EA_7.5 caused changes in TGF-β2, DKK-1, and IGF-1 expression. In HaCaT cells, EEX0_Vis_EA caused changes in TGF-β2, DKK-1, VEGF, and Wnt5α expression. EEX2_Vis_EA led to changes in TGF-β2, VEGF, and Wnt5α expression. EEX1_Vis_EA_7.5 caused the changes in TGF-β2, DKK-1, and VEGF expression. The activation changes of target proteins in both cells induced by herbal extracts were the same as the results obtained by the Western blot.
Discussion
In this study, efficacies of the extracts of herbal medicines on hair growth promotion/hair loss inhibition were monitored by Western blot, and quantitative hyperspectral single-cell imaging cytometry was used to confirm such efficacies. Although hair growth and hair loss have been studied during the past decade, it has not been clear which molecular mechanism is responsible for hair growth and hair loss at the cellular level. Several proteins in the cell are known to affect hair growth and hair loss. One of these proteins, TGF-β2, which is a cytokine, is related to many cellular functions, especially in fetal development.8,17 In hair follicle physiology, dihydrotestosterone (DHT) stimulates the synthesis of TGF-β2 in the dermal papilla, in the catagen phase. TGF-β2 suppresses proliferation of the hair follicle, which induces the mitochondria-mediated caspase activation (intrinsic apoptosis pathway) and subsequently elicits apoptotic cell death of hair follicle physiology.
Wnt5α is one of the typical noncanonical Wnt proteins that suppresses the canonical Wnt signaling pathway and plays a critical role in the homeostasis and proliferation of various tissues.18–20 Among proteins associated with the Wnt signaling pathway, Wnt5α promotes hair cycle and hair follicle growth. Wnt protein binds to the Wnt receptor, Frizzled (FZ) proteins, and the low-density lipoprotein receptor-related protein (LRP) family and activates the canonical Wnt signaling pathway. These complexes elicit the stabilization of β-catenin. Subsequently, β-catenin is translocated to the nucleus and combines with the members of the T-cell factor (TCF)/lymphoid enhancer factor (LEF) family to form active transcription complexes. Wnt5α mRNA is tremendously expressed in dermal papilla cells and also exists in inner root sheath (IRS) and outer root sheath (ORS) of mouse hair follicles. The expression level of Wnt5α mRNA is especially the highest in the anagen phase but decreases in the catagen and telogen phases.
Compared with Wnt5α, DKK-1 also affects Wnt signaling, with the opposite effects on hair growth. 21 DKK-1 as a Wnt antagonist binds to Wnt coreceptors of the LRP family and suppresses the canonical Wnt signaling pathway. Therefore, the growth of ORS cells is inhibited by DKK-1, and balding dermal papilla cells (DPC) are activated. Kwack et al. 22 demonstrated that 50–100 nM DHT increased the expression level of DKK-1 mRNA in 3 to 6 h in HFDPC. The upregulation of DKK-1 induced by DHT in DPC induced apoptotic cell death.
VEGF protein is a key regulator of angiogenesis. 23 The expression of VEGF in DPC is strong in the anagen phase. 24 Yano et al 11 reported that the expression of VEGF protein in the outer root sheath of the murine hair follicles affected cell proliferation in the perifollicular capillary network. Transgenic overexpression of VEGF upregulated perifollicular vascularization and accelerated hair growth in mice after depilation.
IGF-1, a hormone expressed in the hair follicle, is involved in differentiation, cell growth, and survival. In the human hair follicle, proteins in the IGF signaling pathway, including IGF-1, IGF–1 receptor (IGF-1R), and IGF–binding protein 3, are expressed in the dermal papilla. 25 Weger and Schlake 25 demonstrated the effect of IGF-1 on hair growth using IGF-1–expressed transgenic mice in the IRS. In transgenic mice, fine hairs are significantly elongated, and hair shaft bending is suppressed. Furthermore, IGF-1 prevents apoptotic cell death in hair follicles.
Minoxidil is the most popular drug to promote hair growth and suppress hair loss. Our results showed that minoxidil upregulated VEGF, IGF-1, and Wnt5α and downregulated DKK-1 in HFDPC. In addition, DKK-1 and TGF-β2 were downregulated, and VEGF, IGF-1, and Wnt5α were upregulated by minoxidil in HaCaT cells. Some studies demonstrated the effects of minoxidil on hair growth/hair loss–relevant proteins in vitro and in vivo. Han et al. 26 reported minoxidil-induced changes in proliferation-associated factors, including Akt, extracellular signal–regulated kinase (ERK), Bcl-2, and Bax on HFDPC. Minoxidil caused an increase in phosphorylated ERK and Akt and a decrease in the Bcl-2/Bax ratio. Consequently, minoxidil stimulated cell proliferation and prevented cell death. Kwack et al. 27 found that the phosphorylation of GSK3β, PKA, and PKB and expression of Axin2, Lef-1, and EP2 were accelerated by minoxidil in HFDPC. Lachgar et al. 28 demonstrated that minoxidil in the range of 0.2 to 12 µM raised VEGF mRNA expression and protein in HFDPC. Previously, no concrete molecular evidence existed that minoxidil directly affects the upregulation/downregulation of DKK-1, IGF-1, TGF-β2, and Wnt5α. In this study, we found for the first time that minoxidil in the range of 0 to 12 µM affected the expression of DKK-1, IGF-1, TGF-β2, and Wnt5α in HFDPC and HaCaT cells.
We also tested the effect of herbal extracts in HFDPC and HaCaT cells to develop a hair restorer or hair loss inhibitor. Herbal medicine is known to exhibit fewer side effects. 29 Therefore, the efforts to develop herbal-based hair loss medication have been quite active in the industry. We focused on drug development and discovery using diverse herbal medicines based on observation of upregulation/downregulation of five proteins related to hair growth/hair loss. As shown in Table 1 , 23 dried herbal medicines were used and divided into four groups. Herbal medicines in group A activated hair follicles, prevented hair loss, improved hair growth, and made shiny and healthy hair. Herbal medicines in group B play a role in gray hair prevention. Herbal medicines in group C have an effect on skin diseases. Herbal medicines in group D promote blood circulation. These groups were mixed in different ratios and subsequently treated with viscozyme or pectinex. The animal study was carried out using each group. The mixing ratios of groups A to D in three herbal mixtures were determined based on the safety and efficacy of extracts obtained through the animal study. The enzymes were used to overcome the natural tendencies of phytochemicals in plants, such as low physiological activity and less absorption to the basal layer of the skin. 30 After the enzyme treatment, phytochemicals were converted from the inactive glycosides to the active aglycone form, which increased the active ingredients and physiological activity. The enzyme catalyzed the hydrolysis of phytochemicals and decomposed them into a smaller size suitable for easier penetration into the skin. The hair growth promotion/hair loss inhibition effects of these herbal medicines on HFDPC and HaCaT cells were observed using Western blot and confirmed with HCS using hyperspectral single-imaging cytometry. In this study, six different extracts of herbal medicines (EEX0_Vis_EA, EEX1_Vis_EA, EEX2_Vis_EA, EEX0_Pec_EA, EEX0_Vis_EA_7.5, and EEX1_Vis_EA_7.5) showed drug efficacy as a hair restorer/hair loss inhibitor in HFDPC, and three different extracts of herbal medicines (EEX0_Vis_EA, EEX2_Vis_EA, and EEX1_Vis_EA_7.5) had drug efficacy in HaCaT cells. Three extracts of herbal medicines (EEX0_Vis_EA, EEX2_Vis_EA, and EEX1_Vis_EA_7.5) prevented hair loss and promoted hair growth in both cell lines. HCS was carried out using these three extracts. The HCS results were consistent with the Western blot results. The visualized changes in five target proteins indicated that apoptotic cell death was suppressed, but cell proliferation was stimulated. Therefore, EEX0_Vis_EA, EEX2_Vis_EA, and EEX1_Vis_EA_7.5 can be considered as hair restorers or hair loss inhibitors.
The drug efficacy of the three extracts of herbal medicine was compared with minoxidil efficacy at the same concentration (50 µg/mL). The expression levels of target proteins showed that minoxidil, which is the only chemical product, had better efficacy than the herbal medicines. On the other hand, herbal medicine has various chemical compounds, and 23 dried herbal medicines have a great deal of chemicals. Although diverse chemicals exist in the 23 herbal medicines, the total amount of chemicals in herbal medicines is far less than 50 µg in 1 mL of the extracts. Furthermore, these herbal medicine extracts do not have side effects demonstrated by the use of minoxidil. Further study is warranted to analyze the active ingredients in herbal medicines and to test their efficacy.
Footnotes
Acknowledgements
We are grateful to the Research Institute of Pharmaceutical Sciences at Seoul National University for providing some experimental equipment.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Ministry of Education, Science and Technology (MEST) (2012-0005653 and 2008-0061858) and by the Small and Medium Business Administration, South Korea (S1074059).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
