Abstract
Chemotherapeutics tumor resistance is a principal reason for treatment failure, and clinical and experimental data indicate that multidrug transporters such as ATP-binding cassette (ABC) B1 and ABCG2 play a leading role by preventing cytotoxic intracellular drug concentrations. Functional efflux inhibition of existing chemotherapeutics by these pumps continues to present a promising approach for treatment. A contributing factor to the failure of existing inhibitors in clinical applications is limited understanding of specific substrate/inhibitor/pump interactions. We have identified selective efflux inhibitors by profiling multiple ABC transporters against a library of small molecules to find molecular probes to further explore such interactions. In our primary screening protocol using JC-1 as a dual-pump fluorescent reporter substrate, we identified a piperazine-substituted pyrazolo[1,5-a]pyrimidine substructure with promise for selective efflux inhibition. As a result of a focused structure-activity relationship (SAR)–driven chemistry effort, we describe compound
Introduction
More than 50 members of the ATP-binding cassette (ABC) transporter superfamily have been identified, and three major subfamilies (ABCB, ABCC, and ABCG) have been linked to human multidrug resistance (MDR). These efflux pumps are expressed in many human tumors, where they likely contribute to chemotherapy resistance. The transporters ABCB1, ABCC1, and ABCG2 are highly expressed in the gut, liver, and kidneys and may restrict the oral bioavailability of administered drugs. ABCB1 and ABCG2 are also expressed in the epithelia of the brain and placenta, as well as in stem cells, where they perform a barrier function. 1 They influence oral absorption and disposition of a wide variety of drugs, and as a result, their expression levels have important consequences for susceptibility to drug-induced side effects, interactions, and treatment efficacy. The specific subclass members ABCB1 (Pgp, MDR1), ABCC1 (MRP1), and ABCG2 (BCRP, MXR) are known to significantly influence the efficacy of drugs and have unambiguously been shown to contribute to cancer multidrug resistance. 2 Dual treatment with ABC transporter inhibitors in conjunction with chemotherapeutics is a common treatment strategy to circumvent MDR in cancers. 3 Although a large number of compounds have been identified as possessing ABC transporter inhibitory properties, only a few of these agents are appropriate candidates for clinical use as MDR reversing agents. 4 The clinical failures observed with the current class of drugs provide ample justification for identifying new classes of modulators and exploring the biology around them.
The development of ABC efflux transporter inhibitors is now in its third generation, with the major focus still on ABCB1. Progress over the past decade has renewed interest in the efflux inhibition field, and a variety of modulators have been identified. A large number of structurally and functionally diverse compounds act as substrates or modulators of these pumps.
5
A representative subset of these compounds will be briefly discussed here. The first generation of chemosensitizers was discovered from marketed drugs and included the calcium channel blockers verapamil and nicardipine, cyclosporin A, and progesterone; however, dose-related toxicity and solubility challenges prevented progress into the clinic. Second- and third-generation inhibitors were drawn predominantly from the derivatization of first-generation molecules, as well as from combinatorial chemistry, targeted primarily at ABCB1. Some of the higher profile examples include (i) the cyclosporin A derivative valspodar (PSC-833)
6
; (ii) Vertex Pharmaceutical’s biricodar (VX-710; Vertex Pharmaceuticals, Cambridge, MA)
7
; (iii) the anthranilamide-based modulators XR9051 (
As a specialty screening center in the National Institutes of Health Molecular Libraries Probe Production Centers Network (NIH MLPCN), the University of New Mexico Center for Molecular Discovery (UNMCMD) and its Chemistry Center partners are tasked with finding small-molecule probe compounds for academic investigators seeking improved tools for interrogating biological systems. In collaboration with the University of Kansas Specialized Chemistry Center (KU SCC), we set out to develop new small-molecule scaffolds with distinct efflux inhibition selectivity profiles based on multiplex transporter target assays. Early in the postscreen follow-up, it was evident that ABCG2 was the desirable focus for a probe campaign based on promising preliminary selectivity. Although there has been significant progress with ABCB1 inhibitors, similar progress has not been achieved with ABCG2 inhibitors. An example was noted with the Aspergillus fumigatus mycotoxin fumitremorgin C (FTC,
Given the absence of clinically relevant ABCB1 or ABCG2 specific inhibitors and as there remain gaps in understanding how inhibition of these efflux pumps can be best exploited for therapeutic gain, our team focused on vetting and optimizing novel hit scaffolds with promising preliminary ABCG2 or ABCB1 selectivity and potency. As part of that effort, several benchmark compounds were chosen for comparison during development of the pyrazolopyrimidinylpiperazine scaffold,
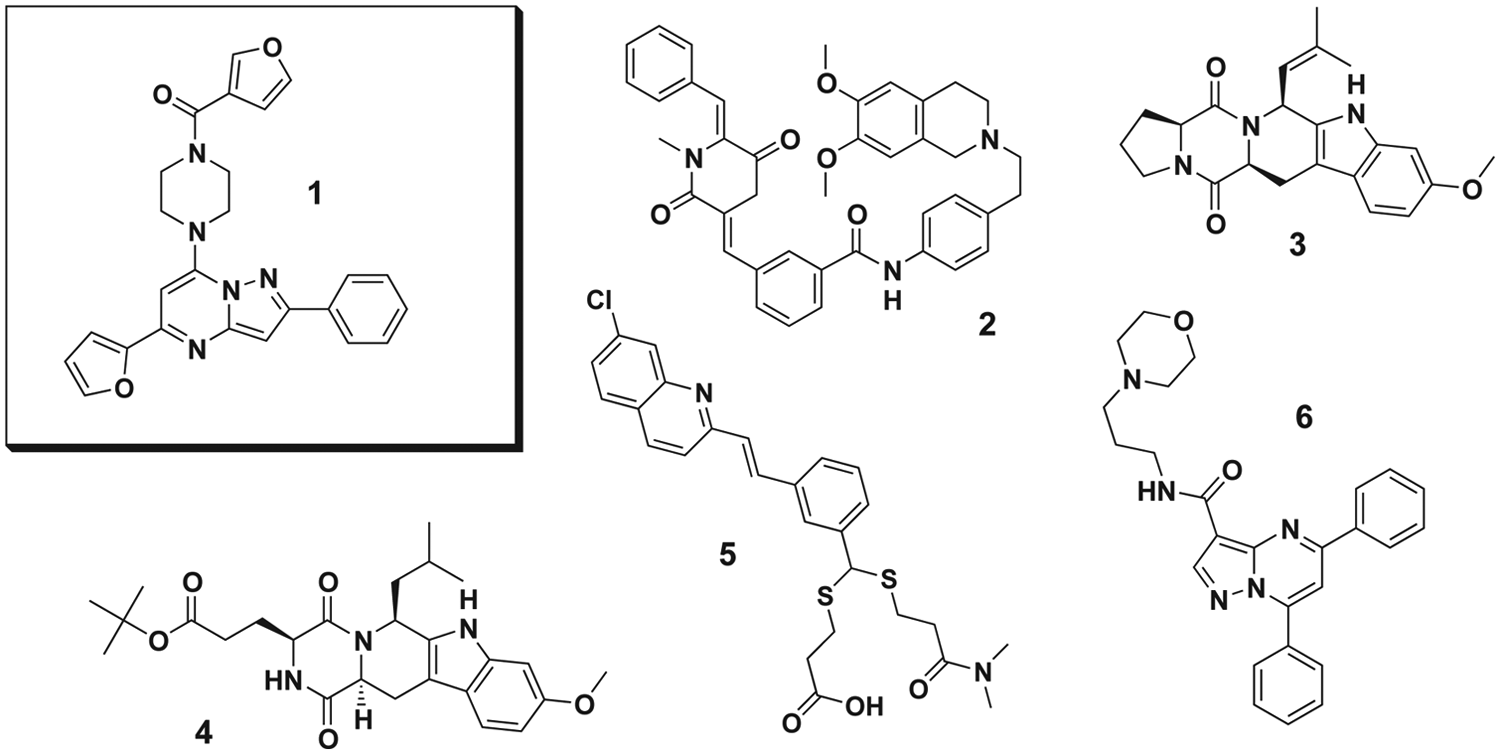
Structures of small molecules chosen for direct experimental comparison. Probe compound CID44640177 (
Materials and Methods
General Information
The ABCB1-overexpressing drug-resistant cell line, CCRF-Adr 5000, and its parental CCRF-CEM cells were kindly provided by Dr. T. Efferth (Pharmaceutical Biology, German Cancer Research Center, Heidelberg, Germany). We have previously described the generation of the Jurkat-DNR ABCB1-overexpressing cell line. 27 Ovarian ABCG2-overexpressing Ig-MXP3 and Igrov1/T8 cells as well as the parental Igrov1-sensitive cells were kindly provided by Dr. D. Ross (Department of Medicine, University of Maryland Greenebaum Cancer Center, Baltimore, MD). Cells were grown in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS; Hyclone, Logan, UT), 2 mM L-glutamine, 10 mM HEPES, 10 U mL−1 penicillin, 10 µg mL−1 streptomycin, and 4 µg mL−1 ciprofloxacin. Selective pressure for the ABCB1-overexpressing CCRF-ADR 5000 and Jurkat-DNR cells was maintained by growth in 20 nM daunorubicin hydrochloride (DNR). Selective pressure for the ABCG2-overexpressing Ig-MXP3 cells was maintained by treatment with 340 nM mitoxantrone dihydrochloride (MTX) for 1 h prior to harvest.
The fluorescent reporter dye JC-1 and cell-type differentiation dye CellTrace Far Red DDAO-SE were obtained from Invitrogen (Carlsbad, CA). Nicardipine hydrochloride, DNR, MTX, topotecan hydrochloride hydrate (TPT), and FTC (
The HyperCyt high-throughput flow cytometry platform (IntelliCyt, Albuquerque, NM) was used to sequentially sample cells from 384-well microplates (2 µL per sample) for flow cytometer presentation at a rate of ~40 samples per minute.28,29 Flow cytometric analysis was performed on a CyAn flow cytometer (Beckman-Coulter). The resulting time-gated data files were analyzed with HyperView (IntelliCyt, Albuquerque, NM) software to determine compound activity in each well. Inhibition response curves were fitted by Prism software (GraphPad Software, Inc., La Jolla, CA) using nonlinear least squares regression in a sigmoidal dose-response model with variable slope, also known as the four-parameter logistic equation. This type of time-gated flow cytometric data analysis was described in detail for a previous ABC transporter screen from our group. 30
Primary Assay Conditions
To facilitate a shortened screening timeline, the single-point assay was performed as a duplex allowing for data from both cell lines to be collected in one screening campaign. The assay was conducted in 384-well format microplates in a total volume of 15.1 µL dispensed sequentially as follows: (1) JC-1 substrate (10 µL per well), (2) test compound (100 nL per well), and (3) drug-resistant cells (5 µL per well). CCRF-Adr cells (ABCB1) were color-coded with 0.5 ng mL−1 CellTrace Far Red DDAO-SE for 15 min at room temperature, washed twice by centrifugation, and then combined with unlabeled Ig-MXP3 cells (ABCG2) in the assay buffer. Final in-well concentration of test compound was 6.6 µM; JC-1 concentration was ~ 1 µM. JC-1 previously proved to be an ideal fluorescent reporter substrate for both ABCB1 and ABCG2. 30 The cell concentration was 3 × 106 cells mL−1 (1:1 ratio of the two cell types). Nicardipine was used as an on-plate control for both pumps at 50 µM. The plate contents were mixed and rotated end-over-end at 4 rpm at 25 °C for 10 min, and then cell samples were immediately analyzed. This resulted in analysis of approximately 1000 cells of each cell type from each well. Flow cytometric data of light scatter and fluorescence emission at 530 ± 20 nm (488 nm excitation, FL1) and 665 ± 10 nm (633 nm excitation, FL8) were collected.
The CCRF-Adr cell line proved optimal for the duplex-Far Red DDAO-SE staining protocol, but in the single-plex follow-up, we preferentially used our ABCB1-overexpressing Jurkat-DNR cell line for confirmatory dose response. Each cell line (Jurkat-DNR and Ig-MXP3) was run separately against all compounds (no differential cell staining) in dose response. The protocol differed from the single-point screen as described here. Cells and reagents were added sequentially as follows: (1) phosphate-buffered saline (PBS) buffer (5 µL per well), (2) test compound (100 nL per well), and (3) drug-resistant cells (10 µL per well) preexposed to the JC-1 substrate at 1 µM just prior to the well addition. Final in-well concentrations of test compound ranged from 50 µM to 69 nM over an 18-point dose response, and the cell concentration was 1 × 106 cells mL−1. Dose-response IC50 values were averaged for multiple runs (average n = 2–4).
Chemoreversal Secondary Assay
Cells (ABCB1, Jurkat-DNR, or ABCG2 Ig-MXP3) were incubated with the test compound in a 3–order of magnitude concentration range over 3- and 7-day periods in the presence of the inhibitor and chemotherapeutic (ABCB1, 100 nM DNR or ABCG2, 30 nM MTX), such that a cell concentration of at least 1 × 105 cells mL−1 was maintained. Cell viability was determined by trypan blue staining and enumeration under light microscopy. At day 3, wells with greater than 2 × 105 cells were refreshed with medium, to include readjustment of chemotherapeutic and inhibitor concentration. A chemoreversal index (Chemoreversal 50, CR50) was determined from the viability assessment. Using a similar approach, a direct cytotoxicity index (Toxic Dose 50, TD50) was determined by assessment of cell death of cells grown in media alone. Results were compared with the survival of parental cells in the presence of the selective agent (chemotherapeutic; 100% cell death), as well as survival of drug-resistant cells in the presence of the chemotherapeutic drug (control yields 100% viability). As previously described by our group for an ABCB1-reversal agent, the difference between the CR50 and the TD50 affords an approximation of the in vitro therapeutic index for the test compound. 31 The threshold for a “good” therapeutic window when comparing CR50 and TD50 somewhat depends on the end-point use. For cancer treatment, a low threshold, in the 10-fold (or greater) range, can still be considered acceptable due to the severity and life-threatening nature of the disease.
Preliminary In Vivo Study
Igrov1/T8 cells were injected into the hind limbs of CB-17 SCID mice at a concentration of 1 × 107 cells in 200 µL, n = 3 per condition. The tumor was grown until the volume was in the range of 75 to 250 mm3. The volume of the tumor was verified with a Scienceware Digi-Max slide caliper obtained from Sigma-Aldrich, and the tumor volumes were calculated by the following equation: (W2/2)*L.
21
Tumor-bearing mice were injected intratumorally with 150 nM topotecan alone, as well as with either 100 nM of
Representative Synthesis
Probe compound
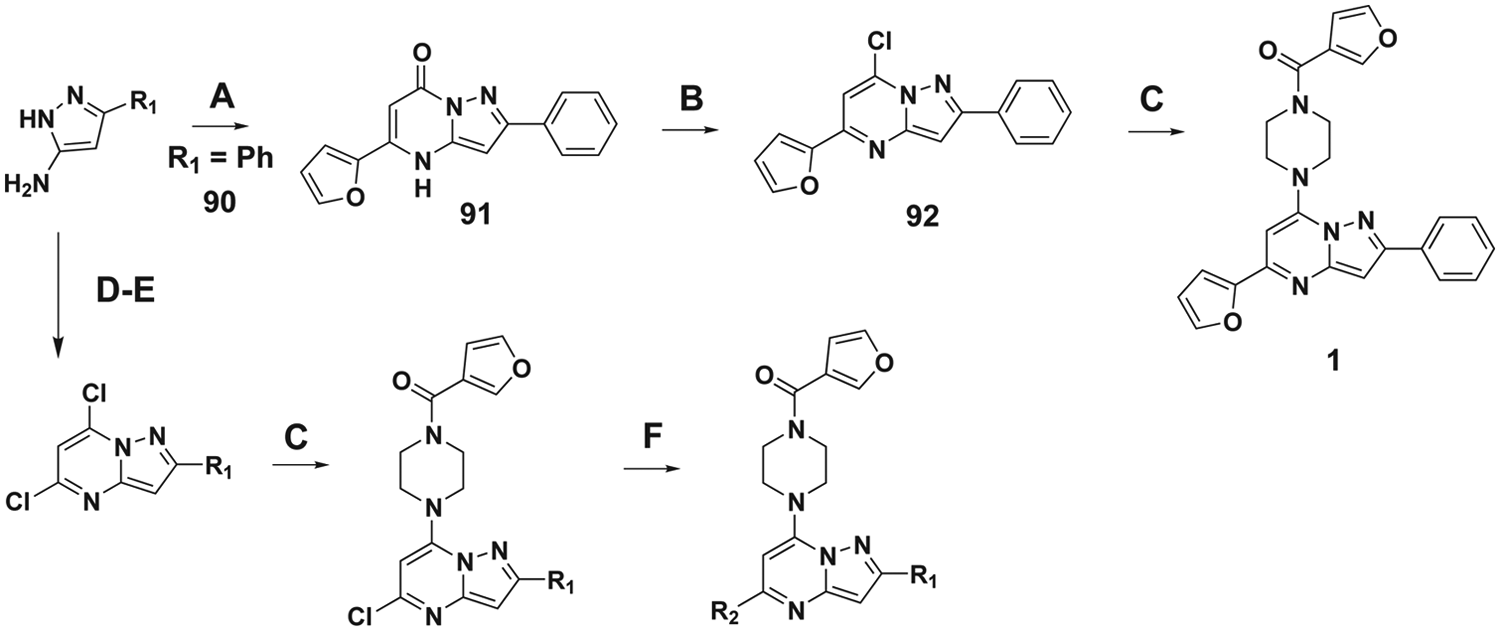
Representative synthetic route for compound
Results and Discussion
Primary Screening
A high-throughput, no-wash, duplex assay was constructed in which both the ABCB1 and ABCG2 transporters were evaluated in parallel using fluorescent JC-1 as the efflux reporting substrate. ABCB1-overexpressing CCRF-Adr cells were color-coded to allow their distinction from Ig-MXP3 ABCG2-overexpressing cells as previously described. 30 Figure 3 briefly summarizes the primary, duplex screening protocol. The primary screening results were uploaded as PubChem BioAssay Database Identifiers (AID) 1325 and 1326 for ABCG2 and ABCB1, respectively (Summary AID 1818). 32 A total of 194 393 Molecular Libraries Small Molecule Repository (MLSMR, http://mlsmr.glpg.com) compounds were tested with Z′ values of 0.74 ± 0.10 and 0.64 ± 0.13 for ABCB1 and ABCG2, respectively. A total of 200 and 130 actives were noted in ABCG2 and ABCB1, respectively. Compounds were deemed active if the percent inhibition was greater than 80%. A subsequent cherry pick resulted in single-point confirmatory testing of 273 compounds (AIDs 1453 and 1451), resulting in 16 and 18 actives in ABCG2 and ABCB1, respectively. As a fluorescence counterscreen, a set of related 488/530-nm fluorescence compound profiling data was also associated with the SMR cherry pick set in which compound fluorescence was assessed in the absence of JC-1 to rule out false positives versus actual efflux inhibitors. These data were uploaded as two AIDs (1480 and 1483) where the 273 compounds were tested with 89 and 83 compounds noted as active (i.e., fluorescent) in ABCG2 and ABCB1, respectively. Based on these single-point screening data, confirmatory dose-response analysis was subsequently performed on 40 compounds (AIDs 1690 and 1689), resulting in 16 actives for ABCB1 and 9 actives for ABCG2.
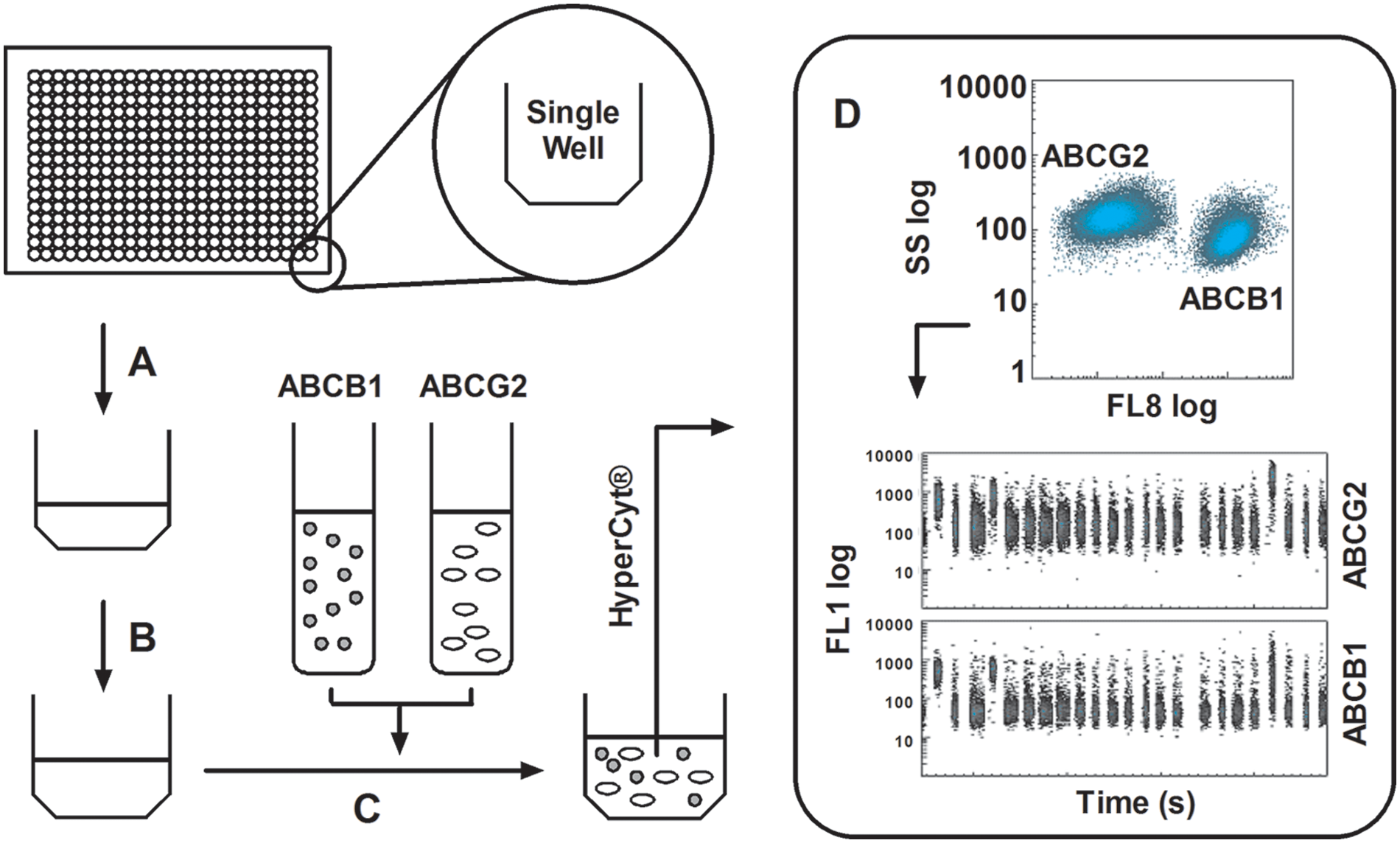
General scheme for the duplex high-throughput screening flow cytometric screening campaign. (
Efflux Inhibition-Driven SAR
No discernable SAR was revealed through the first round of cherry pick analysis or the powder resupply, and many of the compounds were observed to be fluorescent artifacts. However, preliminary chemoreversal secondary screening efforts confirmed activity of several compounds, including
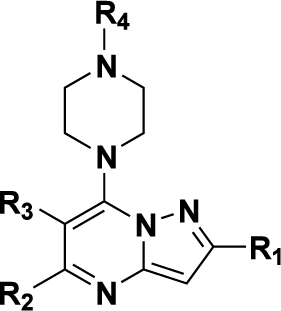
The pyrazolo[1,5-a]pyrimidine core was preserved, and exchange of the peripheral substituents was surveyed, depicted as shaded regions (
Fig. 4A
). Exploratory commercial SAR expansion resulted in compounds with selectivity profiles significantly biased toward ABCG2. The initial set of hit-related compounds screened from the MLSMR and purchased from vendors predominately possessed structural differences in R1 to R3 and the furan ring of R4. On the basis of these structural variations around the core, we chose functional groups that would fill in the SAR gaps and further reveal pharmacological preferences based on steric interactions, lipophilicity, hydrogen bond donating or accepting character, and modulating the electronic nature of aryl substituents. Of the hits identified through this endeavor,
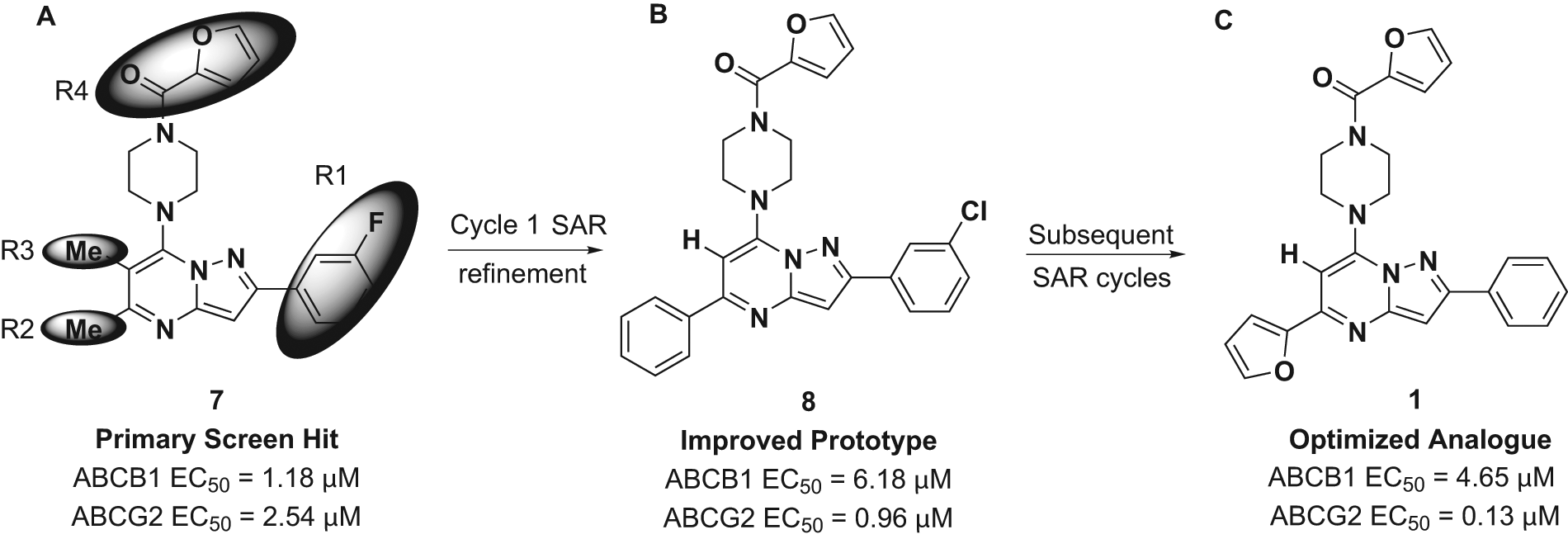
Scaffold modification summary from primary hit to probe compound. (
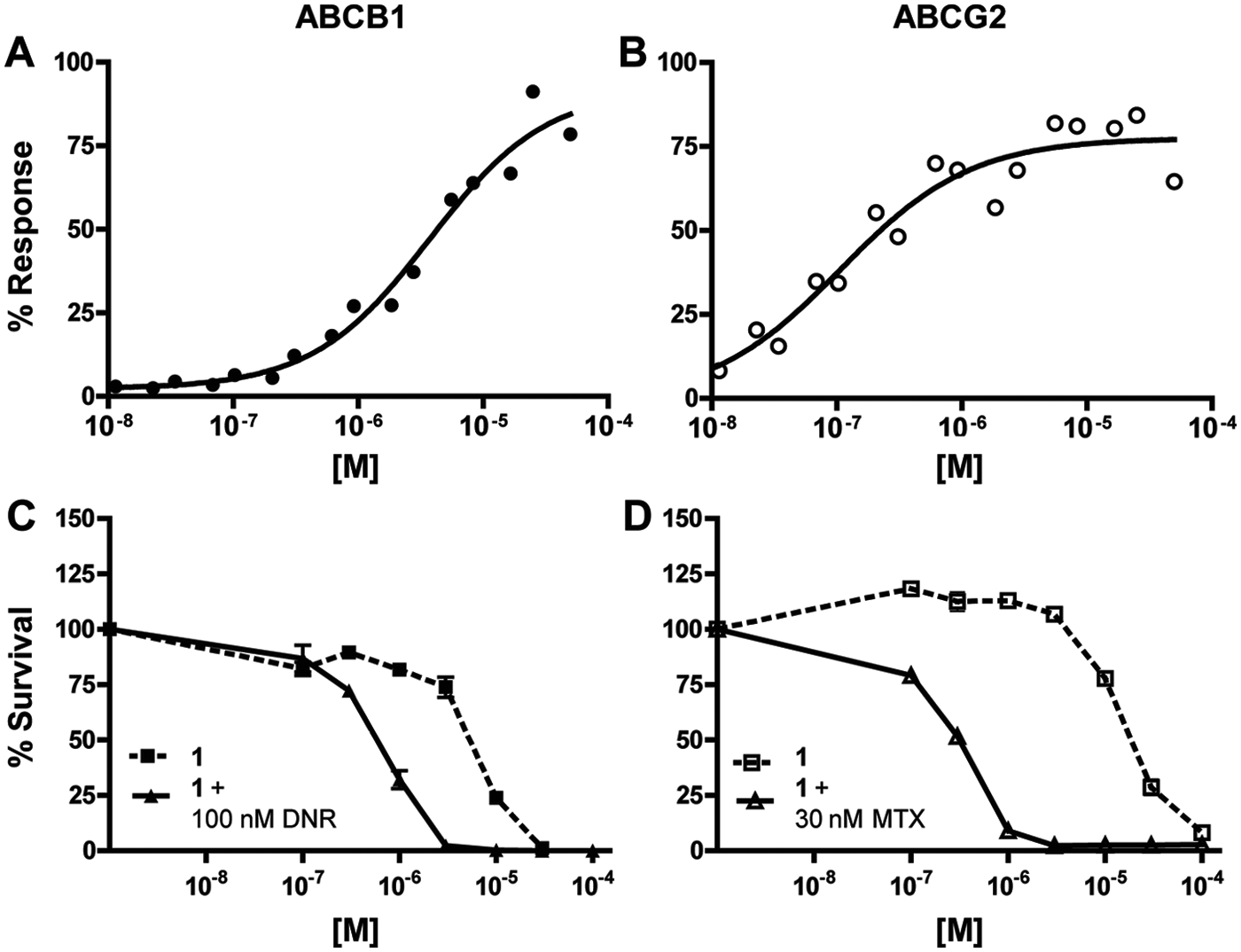
Efflux inhibition and chemotherapeutic potentiation of
Of the approximately 160 compounds assessed in the primary efflux dose-response assay (AIDs 489002, 489003, 504566, and 504569), 126 were synthesized by the KU SCC. Several compounds in the purchased collection contained a 3-chlorophenyl substituent at R1, analogous to the R1 moiety present in hit
Structure-Activity Relationship Expansion on Initial Hit Compound
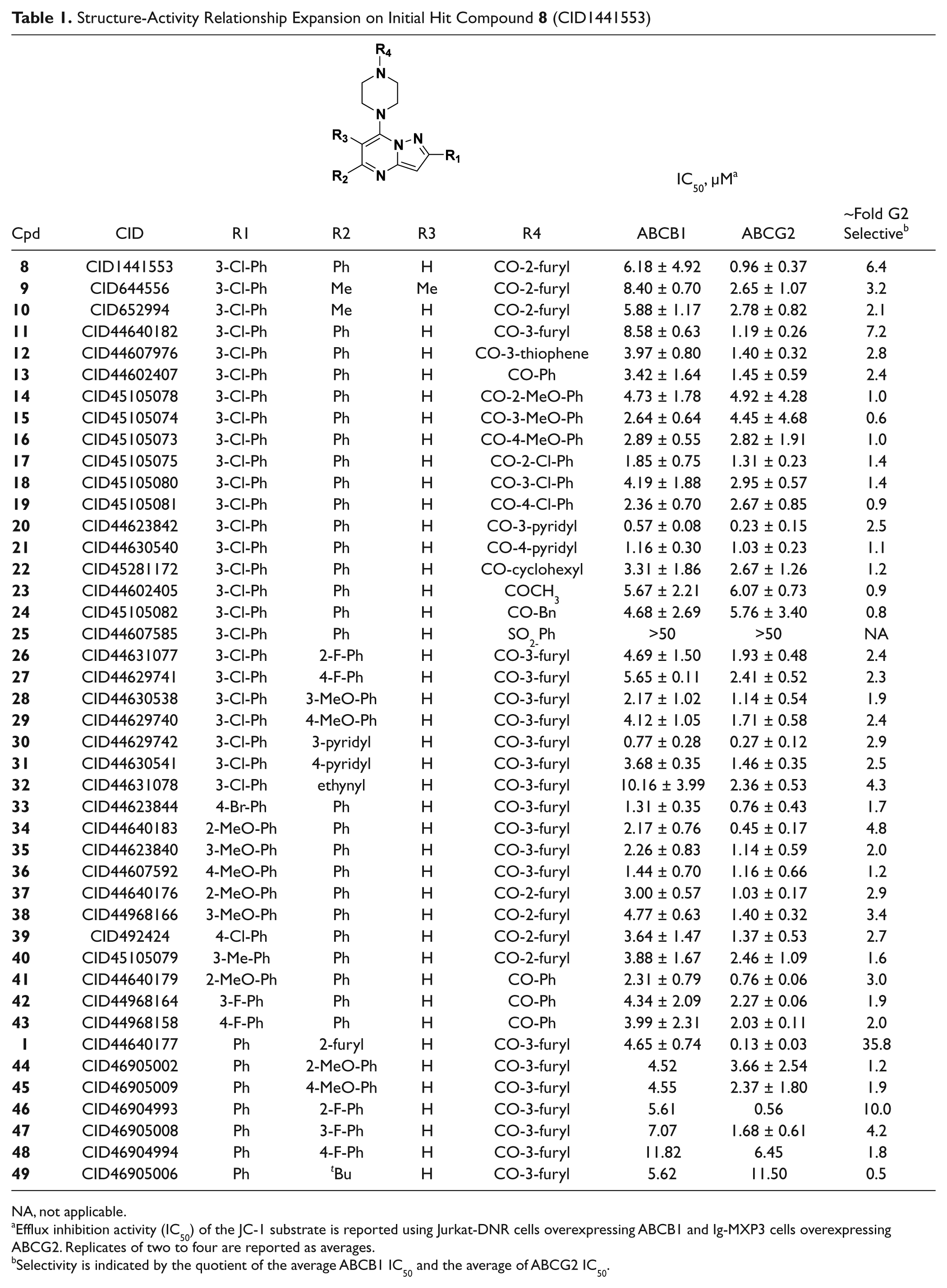
NA, not applicable.
Efflux inhibition activity (IC50) of the JC-1 substrate is reported using Jurkat-DNR cells overexpressing ABCB1 and Ig-MXP3 cells overexpressing ABCG2. Replicates of two to four are reported as averages.
Selectivity is indicated by the quotient of the average ABCB1 IC50 and the average of ABCG2 IC50.
Alterations in the 3-chlorophenyl R1 substituent were then made while assessing three R4 head groups, specifically alternating between acyl-2-furan, acyl-3-furan, or benzoyl functionalities (
With this information in hand, the team followed up with an SAR effort aimed at demonstrating supportive SAR for compounds bearing an R2 = 2-furyl group while also attempting to improve upon the profile of the most promising analogue,
An examination of 2-furan replacements at R2 was also undertaken (
The commercial set of compounds contained a few scaffolds bearing a methyl group at R3. SAR data generated in the early experimental phases demonstrated some benefit to the presence of small alkyl groups at R3; however, this was highly dependent on the identity of groups at R1, R2, and R4. To understand the functionality changes around
Attention was then turned to investigating the effect of different R4 functionality appended to the piperazine (
In a more aggressive effort, the entire “top piece” of the scaffold, consisting of the piperazine and the R4 group, was modified (
In the process of evaluating these structural modifications, several compounds were prepared singly to target possible oversights in SAR, as every possible R1 to R4 combination cannot be prepared and assessed in a timely way. Others were targeted as a means of inserting the best combinations as gleaned from the preceding generations of SAR. These compounds were more recently pursued to probe specific structural combinations and are summarized (
Prior Art Comparison and Potentiation
In parallel with the above efforts, selected compounds were also assessed in secondary assays; however, this chemoreversal assay based on potentiation of a given chemotherapeutic is a very low-throughput assay, and compound data from this assay could not be used to drive the SAR program. Key data have been collected (AIDs 504476 and 504477) for some of the most promising compounds (
Table 2
). Evaluation of probe compound
Potentiation Data and Prior Art Comparison
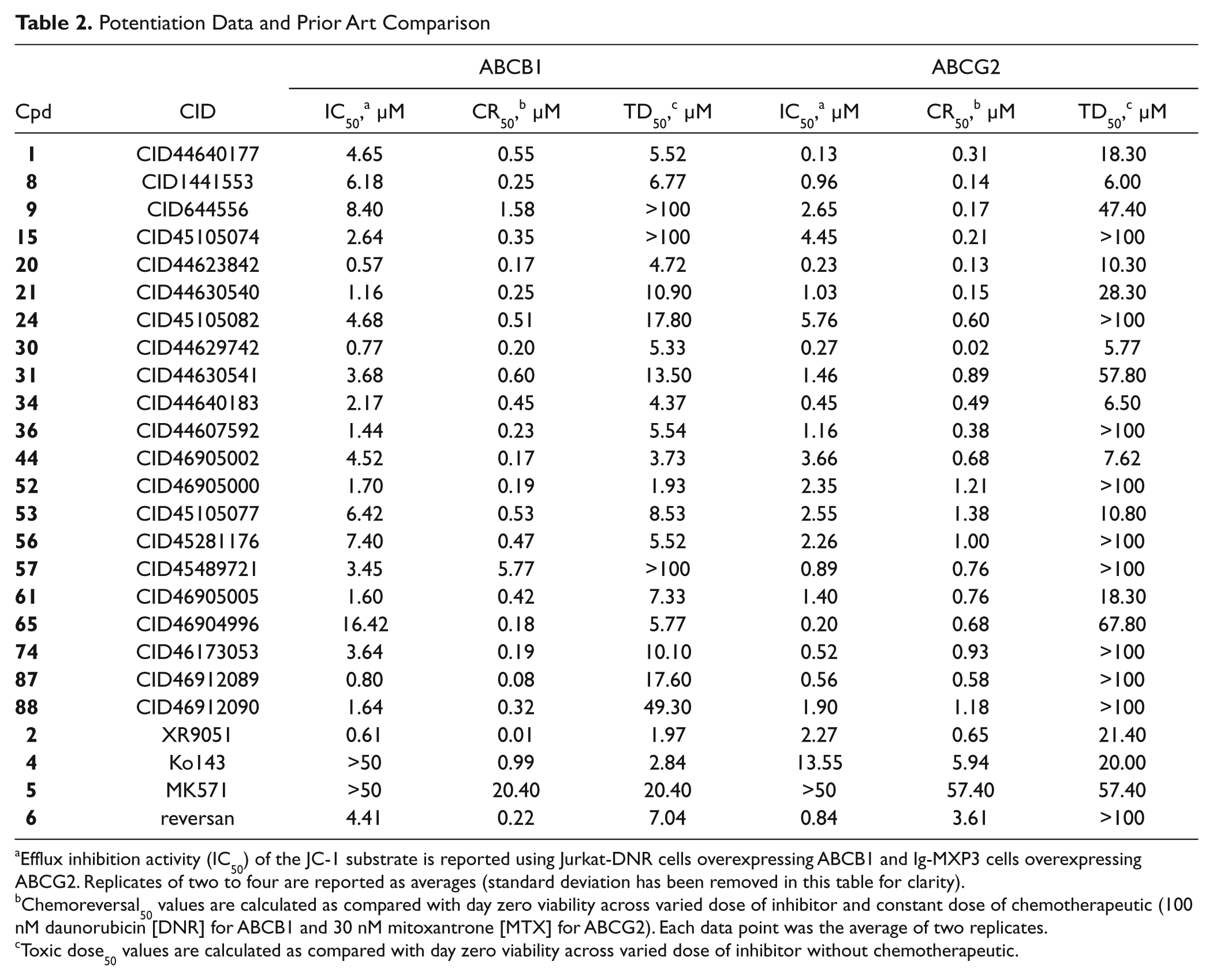
Efflux inhibition activity (IC50) of the JC-1 substrate is reported using Jurkat-DNR cells overexpressing ABCB1 and Ig-MXP3 cells overexpressing ABCG2. Replicates of two to four are reported as averages (standard deviation has been removed in this table for clarity).
Chemoreversal50 values are calculated as compared with day zero viability across varied dose of inhibitor and constant dose of chemotherapeutic (100 nM daunorubicin [DNR] for ABCB1 and 30 nM mitoxantrone [MTX] for ABCG2). Each data point was the average of two replicates.
Toxic dose50 values are calculated as compared with day zero viability across varied dose of inhibitor without chemotherapeutic.
Prior art for
Preliminary In Vivo Mouse Data
To specifically demonstrate the direct effect of a chemotherapeutic agent relevant to these studies, we administered an intratumoral dose of TPT (150 nM). This concentration of TPT was slow to kill nonresistant (ABCG2-nonexpressing) parental tumor cells but not the drug-resistant (ABCG2-expressing) tumor cells (EC50 for parental is 7 nM vs. 311 nM for resistant cells). To demonstrate efficacy of the inhibitors, we grew ABCG2-resistant tumor cells to a volume between 75 and 250 mm3 in CB-17 SCID mice. Tumor size was determined immediately prior to the first injection. Tumor-bearing mice were injected with 150 nM TPT and either 100 nM of
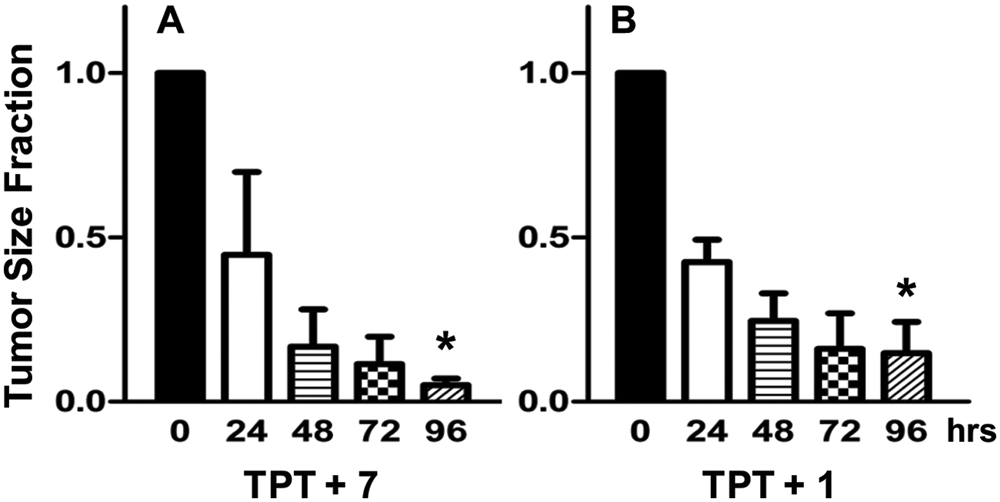
Response of ABCG2-resistant Igrov1/T8-derived tumors in mice to combination therapy of 150 nM topotecan (TPT). The tumor size at 0, 24, 48, 72, and 96 h is indicated (n = 3) along with the standard error of the mean (SEM). The significant difference between the mean values from 0 to 96 h is indicated by an asterisk (p < 0.001). Inhibitor concentration was selected based on potentiation efficacy balanced with apparent cellular toxicity. Significant tumor reduction was noted in both cases. (
In summary, as a result of a duplex, high-throughput flow cytometric screening campaign and subsequent medicinal chemistry optimization, we report herein the discovery of an ABCG2 efflux inhibitor
Footnotes
Acknowledgements
CID 44640177 (ML230) is available from the University of Kansas Specialized Chemistry Center.
Declaration of Conflicting Interests
Bruce Edwards and Larry Sklar are founders of IntelliCyt.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nation Institutes of Health grants U54MH084690 (LAS, UNMCMD), R03 MH081228-01A1 (RSL), and U54HG0050311 (JA, KU SCC).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.