Abstract
The development of a macrophage-based, antitubercular high-throughput screening system could expedite discovery programs for identifying novel inhibitors. In this study, the kinetics of nitrate reduction (NR) by Mycobacterium tuberculosis during growth in Thp1 macrophages was found to be almost parallel to viable bacilli count. NR in the culture medium containing 50 mM of nitrate was found to be optimum on the fifth day after infection with M. tuberculosis. The signal-to-noise (S/N) ratio and Z-factor obtained from this macrophage-based assay were 5.4 and 0.965, respectively, which confirms the robustness of the assay protocol. The protocol was further validated by using standard antitubercular inhibitors such as rifampicin, isoniazid, streptomycin, ethambutol, and pyrazinamide, added at their IC90 value, on the day of infection. These inhibitors were not able to kill the bacilli when added to the culture on the fifth day after infection. Interestingly, pentachlorophenol and rifampicin killed the bacilli immediately after addition on the fifth day of infection. Altogether, this assay protocol using M. tuberculosis–infected Thp-1 macrophages provides a novel, cost-efficient, robust, and easy-to-perform screening platform for the identification of both active and hypoxic stage-specific inhibitors against tuberculosis.
Introduction
Mycobacterium tuberculosis (MTB) is the most important life-threatening bacterial pathogen we face today, responsible for three million deaths annually. 1 An increase in the number of people having double infections with MTB and human immunodeficiency virus warns us about the consequences, and therefore emphasizes, the importance of controlling this infection. 2 Furthermore, the emergence of multidrug-resistant strains of MTB has led to the expansion of this disease. The World Health Organization has declared as a priority the need to immediately control tuberculosis infection to prevent the spread of drug-resistant strains. 3 Latency in tubercle bacilli is the principal cause for many of the problems associated with the disease. 4 Drug-induced latency of the bacilli may be considered one of the most important hurdles in reducing the duration of therapy. 5 Therefore, the need to develop new inhibitors or antibiotics for tuberculosis is inevitable.
Nonreplicating M. tuberculosis bacilli under in vitro culture conditions are characteristically resistant to most of the antitubercular agents and usually known as dormant bacilli. 4 It is well recognized that nonpulmonary tissue oxygen concentrations within the human body are well below the oxygen concentration in ambient room air. 6 Furthermore, the oxygen concentration in the phagosome of activated macrophages is lower than the extracellular oxygen concentration. 7 It has also been reported that MTB cells within lipid-loaded macrophages lose acid-fast staining, becoming phenotypically resistant to the two frontline antimycobacterial drugs rifampicin and isoniazid, and induce gene transcripts involved in dormancy and lipid metabolism within the pathogen.8,9 The pathogen acquires the phenotypically drug-resistant nonreplicating state during latent infection and creates major hindrance to curing the disease. 10 Wayne’s hypoxia and nutrient starvation–induced dormancy models were developed to explain certain features in persistent tubercular bacilli obtained from hosts. 10 Pioneering work by Wayne showed that nitrate reductase (NarGHJI) played an important role during transition from the aerobic to anaerobic dormant stage and that this transition occurs during initial exposure to the asymptomatic pathogenesis as well as during exposure to antitubercular medicines.10,11 Recent reports suggest that nitric oxide (NO) and superoxide (O2–) are generated inside host macrophages and kill the intracellular bacilli after infection by combining to form highly unstable peroxynitrite (ONOO–), which subsequently rearranges to produce NO3– . 9 Currently, there are no reports of nitrate reduction that takes place inside infected macrophages due to hypoxia-induced dormancy.
Several methods are used to evaluate the in vitro and in vivo antimycobacterial activity of drugs.12–15 In vitro methods require colony forming unit (CFU) determinations; however, such experiments generally require an incubation period of 3 to 4 wk before colonies can be accurately counted. Because these studies are extremely laborious, require multiple serial dilutions, and use a large number of agar plates or culture tubes, it is difficult to test more than a few compounds in any one experiment. More recently, radiometric methods based on the measurement of 14CO2 release from a radiolabeled metabolic substrate, such as 14C-palmitic acid (BACTEC TB 460 system), or the measurement of enzymatic activity of a reporter gene, are used as a measure of the killing activity inside the macrophage cell line, enabling the rapid evaluation of drug activity; however, these methods are still of limited use.16–20 Problems associated with these methods are the unavailability of facilities for isotopic measurements and the high cost and inconsistencies of the assays. Methods based on reporter genes, such as β-galactosidase and luciferase, are rapid and sensitive, and the results correlate well with those of culture-based methods.13,16,19,21–24 However, these methods require recombinant bacilli, and various factors affect the measurement of the enzymatic activity. Recently, a procedure that avoids the necessity of measuring enzymatic activity has been reported using a green fluorescent protein gene20,25; however, measurement of both the antimycobacterial effect on the bacilli within host cells and the cytotoxic effect of drugs on host cells cannot be performed simultaneously. Although a simple fibroblast-based assay has been established for antitubercular screening, the bacilli follow a very different infection characteristic in these host (MRC-5) cells. 15 This assay is not popular, mainly because it employs an unusual host, requires a very high multiplicity of infection (MOI), and reports the death of the host cells, which is the last event that happens during infection. Consequently, there is an urgent need for a high-throughput antitubercular screening protocol using a human macrophage infection model, which would be advantageous for the identification of lead inhibitors.
Interestingly, an antitubercular screening assay in microplate format has been recently established by monitoring the nitrate reduction (NR) of Mycobacterium bovis BCG adopted in Wayne’s hypoxic model. 12 Extending this work, the authors have shown how nitrate reduction by intracellular MTB in Thp-1 macrophages can be used to develop an antitubercular screening protocol. Advantageously, this assay enables the identification of inhibitors at both stages of the bacilli within the host intracellular environment.
Materials and Methods
Reagents
Rifampicin, isoniazid, streptomycin, ethambutol, and pyrazinamid were purchased from Sigma (USA).
These compounds were solubilized according to the manufacturer’s recommendations, and stock solutions were filter sterilized and stored in aliquots at −80°C. Sulphanilic acid, N-(1-napthyl)ethylenediamine dihydrochloride (NEDD), was purchased from Merck (India). Dubos broth base and Dubos albumin supplements were purchased from Difco (USA). Standard sterile flat-bottom 96-well plates were purchased from Tarsons (India). Fetal bovine serum (FBS) and minimum essential medium (MEM; without phenol red) were purchased from GIBCO Biosciences.
Cell Cultures and Their Maintenance
Human acute monocyte leukemia cell line Thp1, obtained from the National Center for Cell Science (NCCS; Pune, India), was maintained in a 25 cm2 tissue culture flask containing MEM cell culture medium with 10% heat-inactivated FBS. M. tuberculosis H37Ra (ATCC 25177), obtained from the Microbial Type Culture Collection (MTCC; Chandigarh, India), was routinely maintained on Dubos albumin agar medium at 37 °C. The inoculum was prepared by inoculating three to four colonies from a 21-day-old Dubos agar slant into a 100 mL glass conical flask containing 30 mL Dubos albumin broth followed by incubation at 37 °C and shaken at 150 rpm using an incubator shaker (Model 431; Thermo Electron Corporation, USA).
Infection of M. tuberculosis Macrophage
Human acute monocytic leukemia cell line Thp1 was used to infect and cultivate MTB following a reported method. 26 Briefly, Thp1 cells (1 × 104 cells/mL) were grown in a 96-well microplate containing 200 µL MEM medium at pH 7.4 and for tissue culture flask 3 mL of the same medium at 37 °C for 4 days in the presence of 5% CO2/95% air and 95% relative humidity to reach the required cell density (5 × 104 cells/mL) followed by treatment with 100 nM of phorbol myristate acetate (PMA). The culture was then incubated for a further 24 h to allow macrophage conversion in the plate/flask, then incubated for another 12 h with MTB at 1:100 MOI (i.e., 100 bacilli per macrophage). Extracellular bacilli were removed by washing three to four times with phosphate-buffered saline (PBS) at pH 7.2 followed by the addition of 200 µL or 3 mL fresh MEM medium containing 50 mM sodium nitrate to the microplate wells or tissue culture flask, respectively. To check the effect of inhibitors on the growth of intracellular bacilli, compounds were added at their respective IC90 value (defined as the lowest concentration of the agent required to inhibit 90% growth of the bacilli) at the start of incubation (after the extracellular bacilli were removed by PBS). Unless mentioned otherwise, the macrophages were lysed after 8 d of incubation, using 200 µL or 3 mL of hypotonic buffer pH 7.4 (10 mM HEPES buffer containing 1.5 mM MgCl2 and 10 mM KCl) in the microplate wells or tissue culture flask, respectively. The 100 µL lysate was then spread on Dubos albumin agar plates to get the CFU after 3 to 4 wk of incubation at 37 °C.
Estimation of Nitrite in Culture
The nitrite liberated following the reduction of sodium nitrate in the culture medium at pH 6.8 and 37 °C was determined using a reported method. 27 An 80 µL aliquot of the culture was taken out from the microplate wells or flask and placed in a separate microplate, to which was added 80 µL of 1% sulphanilic acid solution (prepared in 20% v/v HCl) and 80 µL of 0.1% w/v N-(1-naphthyl)ethylenediamine dihydrochloride (NEDD) solution (prepared in distilled water) at different time intervals of incubation. The plate was then incubated for 15 min to develop a pink color, and the absorbance was read at 540 nm (A540nm) using a plate reader (SpectraMax Plus384; Molecular Devices, USA). The nitrite present in the culture reacts with sulphanilic acid to form a colorless nitrite-sulphanilic acid adduct that further reacts with NEED to form colored prontosil, which exhibits an absorbance at 540 nm. 27 The remaining cell culture in the microplate (120 µL) was used to determine the bacterial load within the macrophages by lysing with hypotonic buffer (10 mM HEPES, 1.5 mM MgCl2, and 10 mM KCl) and spreading 100 µL samples on Dubos agar plates at the same time intervals of incubation to enumerate colonies after 21 days.
Assay Protocol
Two hundred microliters of Thp1 monocyte culture (5 × 104 cells/mL) was treated with 100 nM PMA solution in each well of 96-well microplates and kept for 12 h. The macrophage cultures were infected with 20 µL of diluted M. tuberculosis at 1:100 MOI and kept for another 12 h. The infected macrophage cultures were washed with 1X PBS solution two to three times, then 200 µL of fresh medium (MEM) containing 50 mM of sodium nitrate was added. Inhibitors of different dosages were transferred to each well of the infected macrophage cultures either at 0 h or after 5 d of infection, and the plate was incubated for 8 d. Eighty microliter aliquots of each of the cultures were transferred to a separate microplate, and 80 µL of sulphanilic acid (1% in 20% HCl) and 80 µL of NEDD solution (0.1% in distilled water) were added and incubated for a further 15 min. The microplate wells where macrophages were not infected with MTB were used as blanks. The absorbance of the plate at 540 nm was recorded in a plate reader.
Calculation of % Inhibition
where and blank = A540 of the macrophage culture not infected with MTB
negative control = A540 of M. tuberculosis infected macrophage culture without inhibitor.
Results
Correlation between Nitrate Reduction and Viable Count of M. tuberculosis during Its Growth inside the Thp-1 Macrophage in the Presence of Different Nitrate Concentrations in a Tissue Culture Flask and Microplate
An earlier report suggested that the NR of M. bovis BCG could successfully reflect the viability of dormant bacilli in the microplate format. 12 The active-stage inhibitors of tubercle bacilli could also be identified from the same assay. 12 The concept of developing high-throughput screening in a macrophage-based assay was first tested using a tissue culture flask, which was then tested with a microplate, where intracellular MTB showed similar growth kinetics. During the growth of intracellular bacilli, nitrate reductase activity was also found to follow a similar pattern in both the culture flask and microplate experiments ( Fig. 1 ). Although nitrate is known to be produced within infected macrophage, the maximum concentration of 0.09 optical density (OD) after 8 d is not sufficient to provide the expected S/N ratio from the nitrite estimation. Both CFU and nitrate production increased with increasing nitrate concentration from 10 to 40 mM. Nitrate of 50 mM gave a similar nitrite release to 40 mM, but at 100 mM nitrate, both the nitrite production and bacterial number (CFU) decreased to below the 20 mM nitrate level, suggesting this concentration was toxic ( Fig. 1 ). In a hypoxic environment, intracellular bacilli convert the nitrate with the help of nitrate reductase enzyme to nitrite, which is gradually accumulated within the cell. At a certain concentration, nitrite appears to be toxic to the bacilli. A concentration of 50 mM of nitrate was selected as the optimum substrate concentration required for the assay. From the kinetics of nitrite production, it was shown that both the nitrite production and CFU increased up to 120 h, and after 192 h, a saturation level was achieved, which was similar up to 200 h. Therefore, 192 h (8 d) could be chosen as the optimum incubation time for the assay.
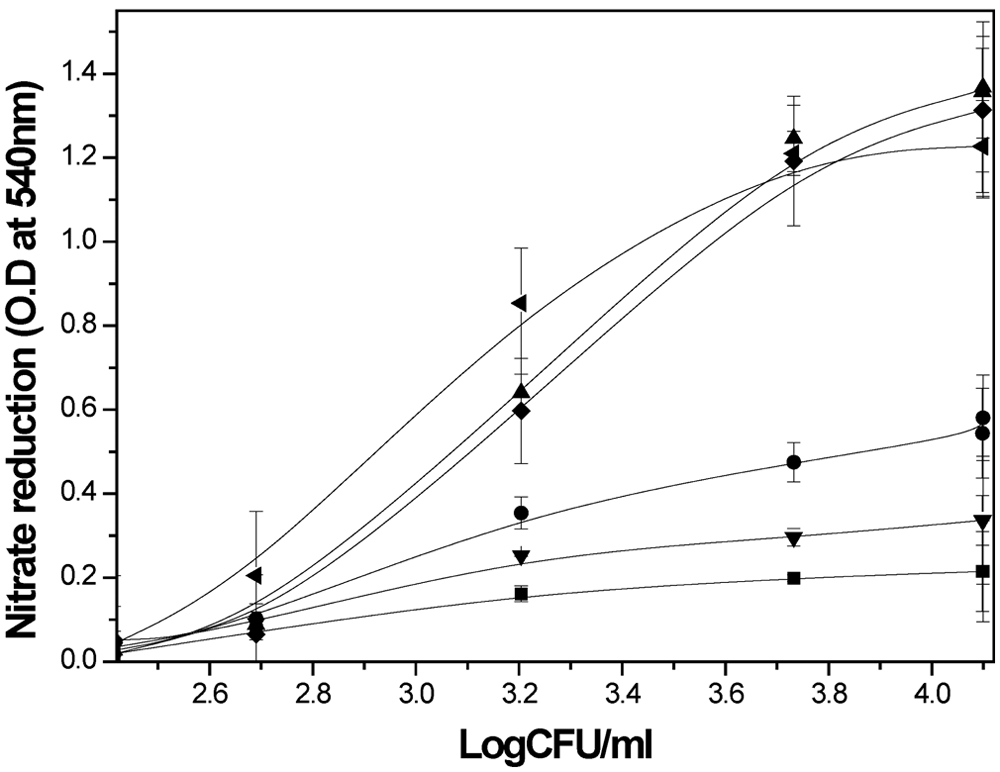
Plot showing the correlation between nitrate reduction and viable count of M. tuberculosis during its growth inside THP1 macrophages. Thp1 macrophage cells were infected with MTB bacilli and incubated for 8 d in the presence of 10 mM (▼), 30 mM (•), 40 mM (♦), 50 mM (▲), and 100 mM (■) nitrate concentrations. The growth of the bacilli was monitored by determining the CFU values as well as measuring the optical density (OD) at 540 nm after addition of Greiss reagent to the microplate cultures at different time points. The plot was made by using colony-forming unit values with the corresponding OD values obtained at 540 nm. A parallel set of experiments was carried out in a tissue culture flask (◄) with 50 mM nitrate in the medium while keeping other parameters the same, which are also included in the plot. Other experimental details are described in the Materials and Method section. Experiments were carried out more than three times, and the results shown are the mean ± SD.
Robustness of Assay
The robustness of this screening protocol in microplate format was assessed by determining the S/N ratio and Z-factor. 12 When the S/N ratio for NR was determined at different concentrations of nitrate in the medium, a maximum value of 5.4 was obtained for 50 mM of nitrate in the medium ( Table 1 ). The Z-factor also attained a peak level of 0.965 at the same nitrate concentration.
Determination of Signal/Noise (S/N) Ratio and Z-Factor from a 96-Well Plate Format for the Nitrate Reductase Macrophage-Based Assay (NRMQA) Done at Different Nitrate Concentrations
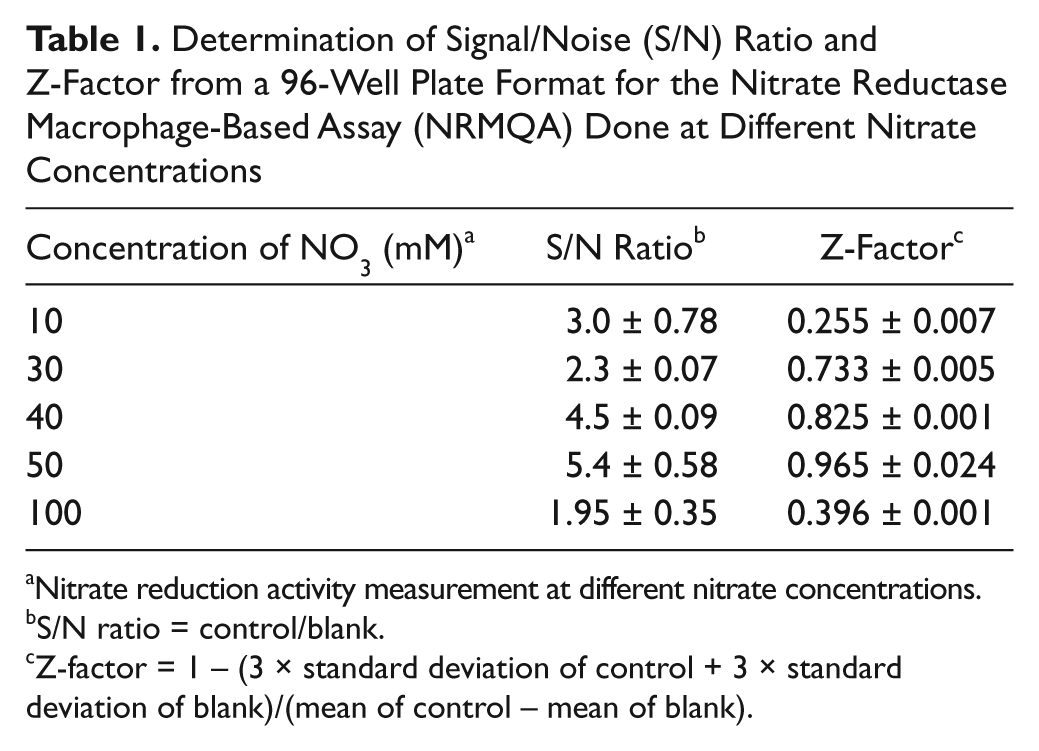
Nitrate reduction activity measurement at different nitrate concentrations.
S/N ratio = control/blank.
Z-factor = 1 − (3 × standard deviation of control + 3 × standard deviation of blank)/(mean of control – mean of blank).
The compounds to be screened for the assay were generally prepared in DMSO solution. Hence, the maximum tolerable dose of DMSO for NR and growth in the assay should be calculated before the screening of compounds. The DMSO dose-response curve obtained clearly indicated that the growth and nitrite production was not significantly affected at up to 1% of DMSO in the assay mix (data not shown). Consequently, the maximum volume of compound solution that could be used in the assay was 1% of the total assay mixture.
Validation of the Assay Protocol by Using Stage-Specific Inhibitors
It was earlier noticed that actively growing bacilli are killed by most of the standard antitubercular drugs (rifampicin, isoniazid, streptomycin, ethambutol, and pyrazinamid),8,9 whereas our data suggest that nitrate reductase (pentachlorophenol)–specific inhibitors kill the pathogen more efficiently after hypoxia is reached within the macrophage (data not published). To estimate their effect on nitrite production by MTB bacilli within macrophage, the IC90 values of the inhibitors were determined from the nitrate reductase macrophage-based assay (NRMQA) by applying them at day 0 of infection, and nitrite production was monitored for 8 d after incubation ( Table 2 ). The CFU values ( Table 2 ) obtained from the same set of cultures were compared with the values reported for a simple fibroblast-based assay ( Table 2 ). 15 The results of nitrite production obtained from this experiment clearly indicated that the IC90 values of rifampicin, isoniazid, streptomycin, and pentachlorophenol were lower and that the values for ethambutol and pyrazinamide were higher than the CFU values obtained from the same assay. The difference between NR and CFU values were not significant, nor did they show any uniform pattern, because earlier reports indicated a range of minimum inhibitory concentration (MIC) values for most of these drugs.28–31 Therefore, it was concluded that NR detection nicely parallels the CFU-based method.
Comparison of IC90 Values Determined by a Nitrate Reductase Macrophage-Based Assay (NRMQA) with MIC Values Reported Earlier in a Simple Fibroblast-Based Assay (SFA) of Standard Inhibitors against M. tuberculosis
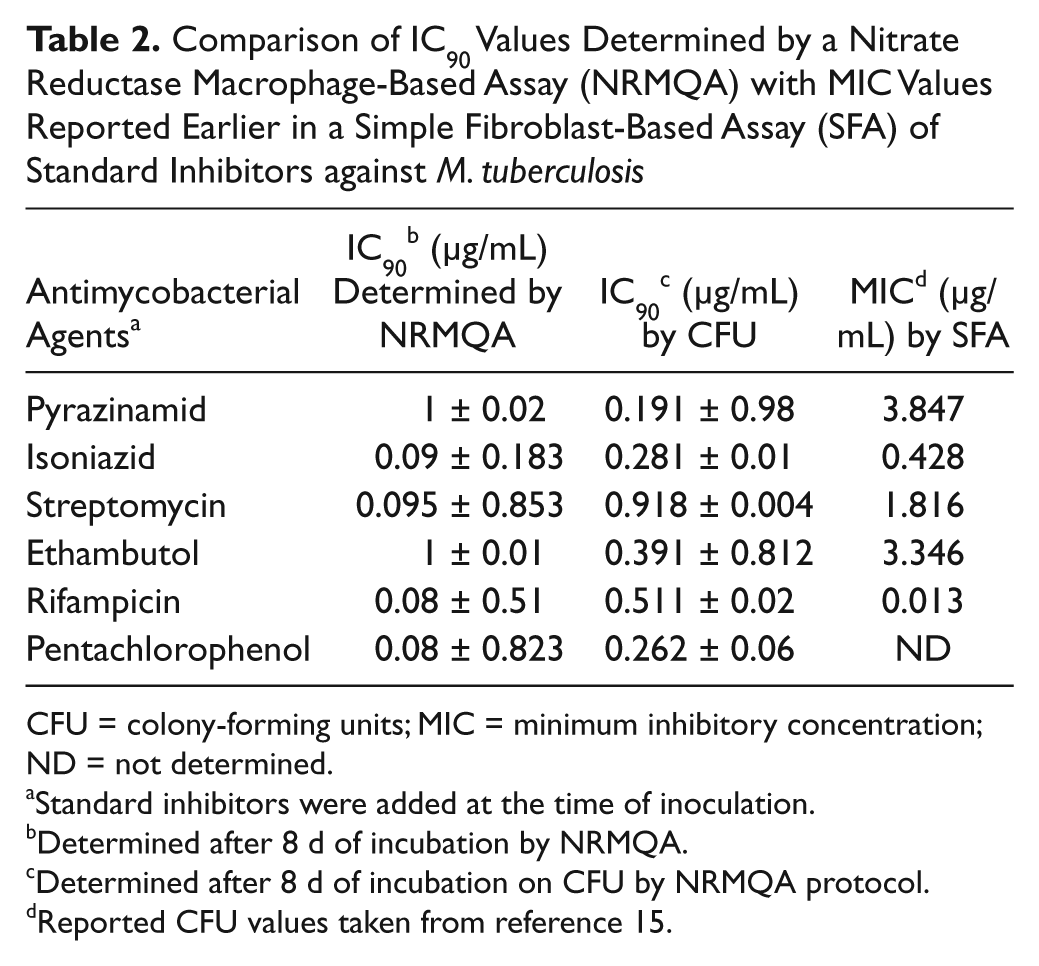
CFU = colony-forming units; MIC = minimum inhibitory concentration; ND = not determined.
Standard inhibitors were added at the time of inoculation.
Determined after 8 d of incubation by NRMQA.
Determined after 8 d of incubation on CFU by NRMQA protocol.
Reported CFU values taken from reference 15.
Intracellular bacilli after the fifth day of infection was exposed to all these inhibitors at the concentration that inhibits 90% growth of bacilli at the aerobic stage, and the viability pattern and NR were followed for a further 3 d ( Fig. 2 ). The kinetics of NR clearly indicated the ineffectiveness of aerobic stage-specific inhibitors such as streptomycin, isoniazid, and ethambutol on the NR, which was validated from the CFU values. Earlier reports suggested that nitrate reductase enzyme is constitutively expressed in MTB, and rapid induction in its activity was observed during hypoxic shift down to the nonreplicative dormant stage. 32 In the presence of nitrate in the medium, the bacilli become completely dependent on nitrate respiration under hypoxic conditions. Under such conditions, exposure to inhibitors of nitrate reductase such as pentachlorophenol is able to kill the bacilli (unpublished data). The specificity in the mode of action of pentachlorophenol was also envisaged from its inability to affect the growth of intracellular bacilli for up to 4 d when added at day 0 after infection. Rifampicin, known for its action against both the actively growing as well as the nonreplicating state of the bacilli, 19 was also found to inhibit nitrate reduction, albeit at a milder level compared with pentachlorophenol, when added after 5 d of infection, confirming that the bacilli were mainly in the dormant stage.
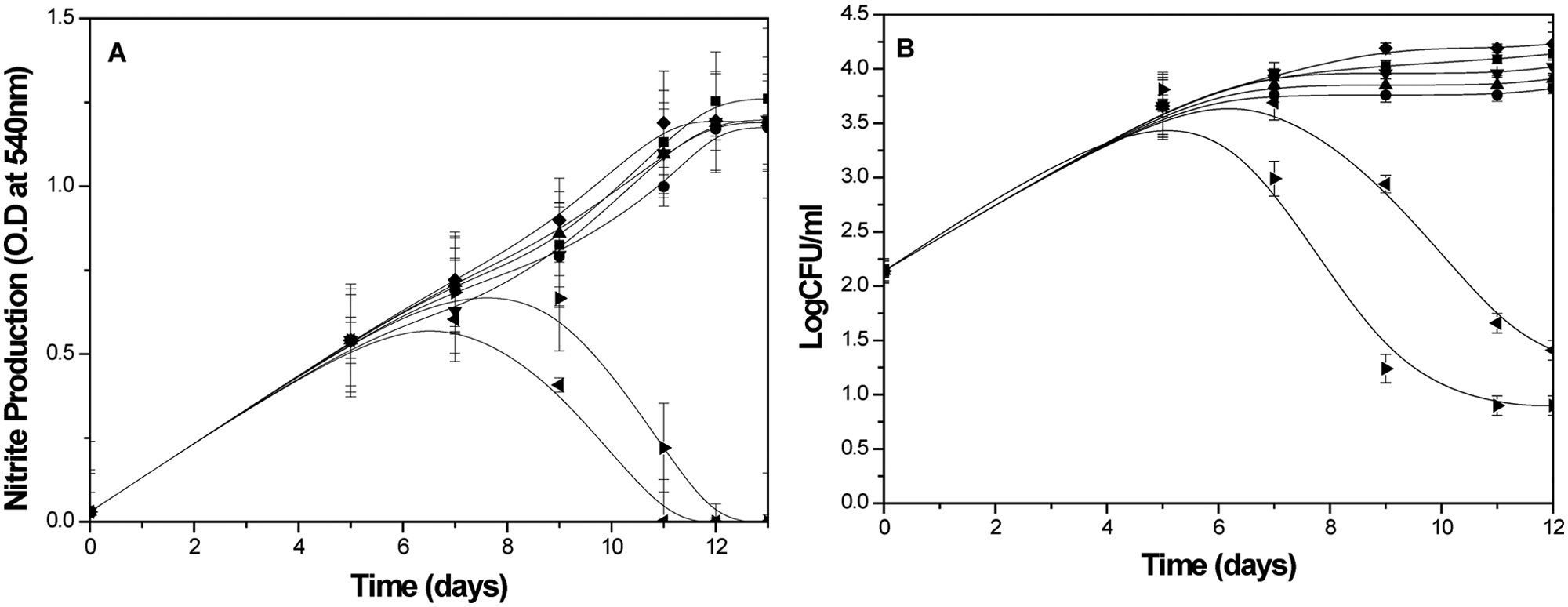
Stage specificity of standard inhibitors on the viability of M. tuberculosis within THP1 macrophages monitored using the nitrate reductase macrophage-based assay protocol. Rifampicin (0.08 µg/mL, ►), pentachlorophenol (0.08 µg/mL, ◄), streptomycin (0.095 µg/mL, ♦), ionized (0.09 µg/mL, ▲), ethambutol (1 µg/mL, ●), and pyrazinamid (1 µg/mL, ▼) were added after 5 d of infection, and nitrite production (A) as well as CFU values (B) of intracellular MTB were monitored with the ninth day after infection. The nitrite production and intracellular bacilli were measured in the control (■) from 0 day after infection of macrophages. The rest of the details of the experiment are described in the Materials and Methods section. The values are shown as mean ± SD of three identical experiments.
Discussion
Our study revealed that the kinetics of nitrate reduction and viable count of bacilli inside the infected macrophages was almost parallel ( Fig. 1 ). Earlier studies from our laboratory had clearly shown that nitrate reduction in Wayne’s model is directly related to the viability of dormant Mycobacterium smegmatis in the presence of nitrate in the medium and could be used as a reporter system to monitor the dormant bacilli. 33 The Km value of partially purified nitrate reductase was found to be 384 µM (unpublished work), which suggested that a 10-fold increase in Km value (i.e., 3.83 mM) inside the cell could achieve optimum nitrate reduction (V max). Our present data show that the maximum reduction of nitrate occurs at the 50 mM extracellular level, suggesting the existence of a barrier to nitrate penetration into the cell. The difference in nitrate reduction ability among MTB, M. bovis BCG, and M. smegmatis has long been used as a diagnostic tool to identify different mycobacterial species. 34 MTB is the strongest reducer of nitrate in the medium, possibly because of the point mutation in the promoter region of narGHJI (respiratory type nitrate reductase). 32
Recent observations on the absence of nitrate reductase activity in MTB are probably due to the use of 5 mM nitrate in the medium, 32 which is also supported from our results ( Fig. 1 ). Measurement of NR did not require cell lysis and could be monitored colorimetrically without any specialized instrumental requirement. The color also remained stable at least for 24 h. Hence, NR activity determinations are easy, cost-efficient, and simple assays for monitoring the viability of dormant stage-specific bacilli. However, in the case of MTB, nitrate reduction occurs at a lower rate in the actively growing stage and induced many fold as the availability of oxygen in the environment was depleted. Interestingly, NR did not increase to the extent generally observed with respect to the number of MTB bacilli under in vitro conditions, as reported earlier. 35 Instead, NR followed a very similar pattern of increase along with the number of bacilli inside the macrophages. As the bacilli ceased to grow after the fourth day after infection, probably because of hypoxia, the entry of nitrate into host cells also reached the plateau. So the primary role of NR could be redox balancing, or it may serve a temporary function to provide energy during the shift down to the NPR stage. 35 The present macrophage-based assay demonstrates a similar pattern of growth and induction of NR activity in microplate assays, representing the transition between aerobic to microaerophilic stage (NRP-1) of the bacilli. The major advantage of this assay is that we could pick up both the aerobic as well as NRP-1 stage inhibitors in the first assay, where the inhibitors will be used at day 0 of infection. Once the hits of the inhibitors will be identified, they could be used on day 5 after infection of macrophages using the same protocol to identify the dormant inhibitors ( Fig. 2 ; Table 2 ). No other assays currently available are able to identify inhibitors of both stages of MTB. Thus, along with its application in the screening of a chemical library, the macrophage-based assay could also be used in biochemical experiments at the microplate level to the advantage of having a large number of data points compared with the low throughput tissue culture flask model, making it cost-effective. The S/N ratio and Z-factor clearly indicate the robustness of the assay protocol ( Table 1 ).
The most valuable information regarding validation comes from the sensitivity of the assay against standard inhibitors. IC90 values for aerobic-stage inhibitors determined by this microplate assay were very close, and sometimes better, than the values determined by other drug susceptibility assays, 13 further validating the protocol in favor of its use in screening ( Fig. 2 ; Table 2 ) against both the actively replicating as well as the dormant stage of MTB. The microplate format of the assay allows for the screening of a large number of compounds using an automated robotic high-throughput screening platform, which may enormously expedite drug discovery programs for the discovery of novel antitubercular molecules. Altogether, the exclusion of CFU counting, a minimum number of steps within 8 days, the use of cheaper reagents, and no specialized safety requirements could make the present assay the most acceptable among all available screening protocols and can enable identification of both aerobic replicating phase inhibitors as well as the NRP-1 stage of MTB.
Footnotes
Acknowledgements
The authors are thankful to Microbial Type Culture Collection (MTCC), India, for providing M. tuberculosis H37Ra (ATCC 25177) and the National Center for Cell Science (NCCS) for providing the THP1 monocyte cell line. SS is highly thankful to the Indian Council of Medical Research, (ICMR), India, for providing the senior research fellowship. Both the authors are also thankful to the director of the National Chemical Laboratory, Pune, India, for providing financial assistance through an in-house project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

